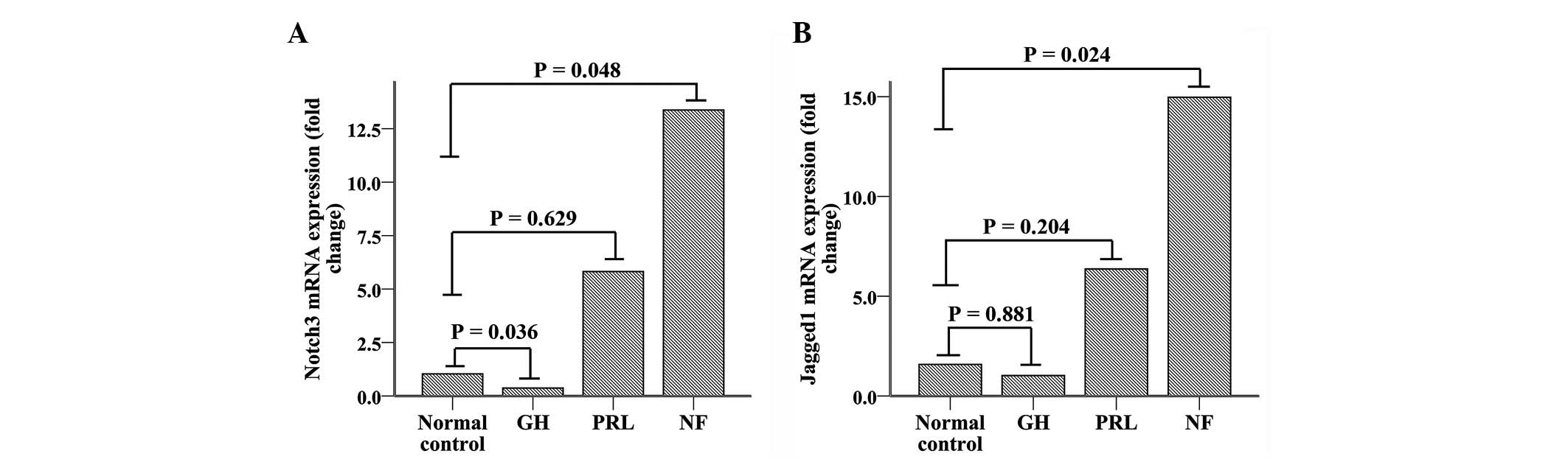
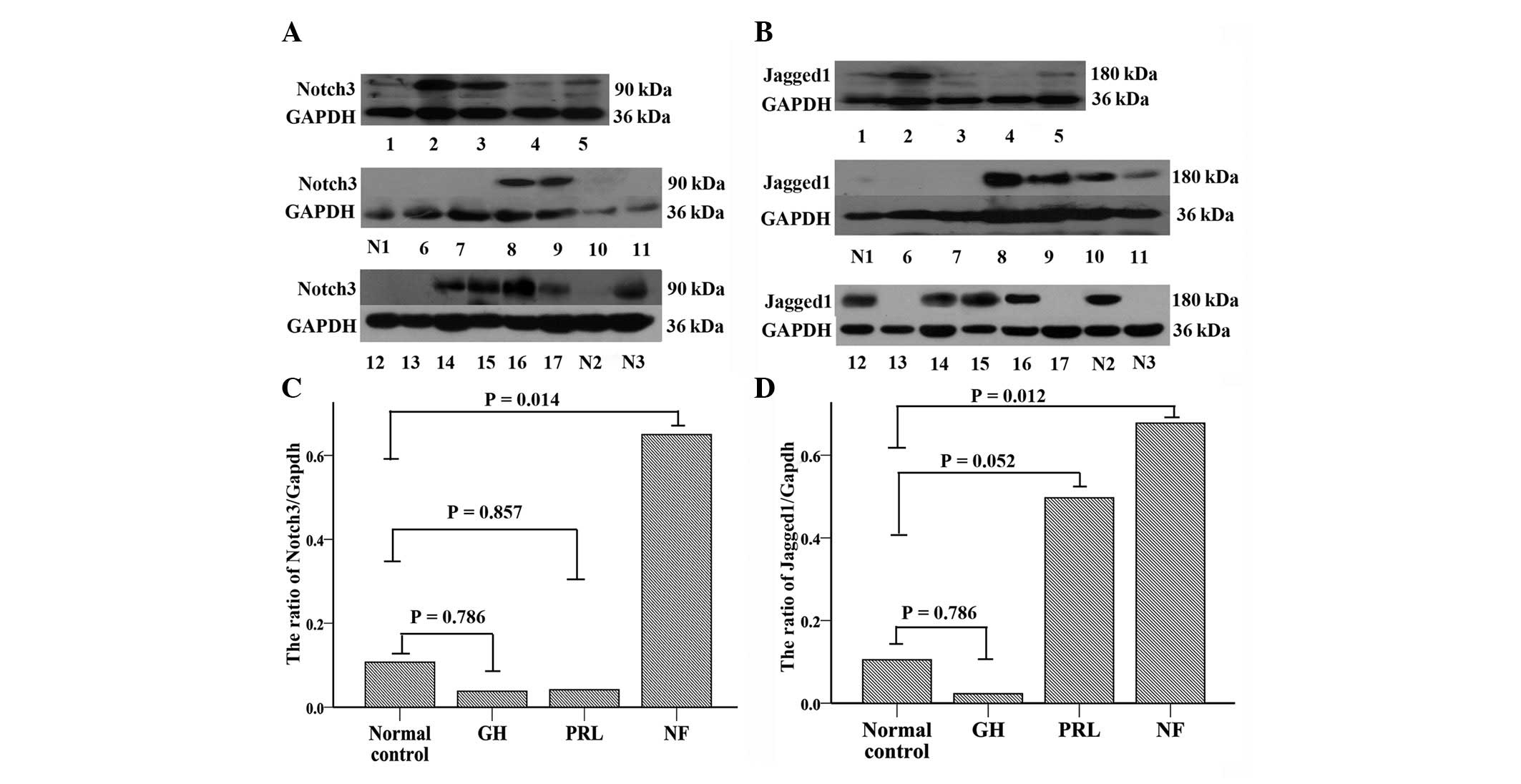
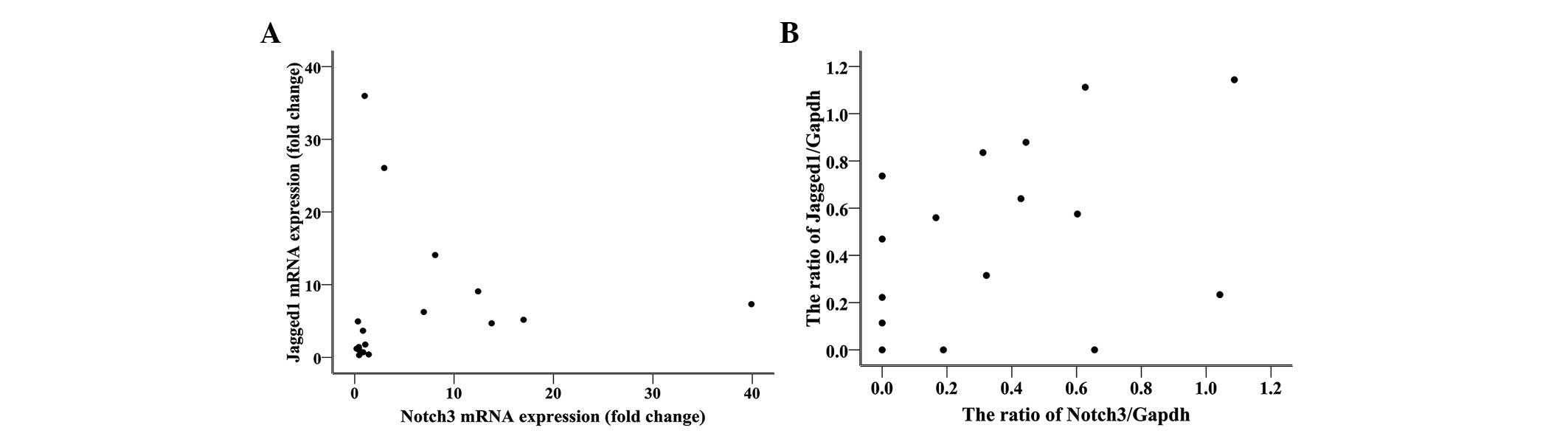
|
1
|
Buurman H and Saeger W: Subclinical
adenomas in postmortem pituitaries: classification and correlations
to clinical data. Eur J Endocrinol. 154:753–758. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Saeger W, Lüdecke DK, Buchfelder M,
Fahlbusch R, Quabbe HJ and Petersenn S: Pathohistological
classification of pituitary tumors: 10 years of experience with the
German Pituitary Tumor Registry. Eur J Endocrinol. 156:203–216.
2007.PubMed/NCBI
|
|
3
|
Greenman Y and Stern N: Non-functioning
pituitary adenomas. Best Pract Res Clin Endocrinol Metab.
23:625–638. 2009. View Article : Google Scholar
|
|
4
|
Jaffe CA: Clinically non-functioning
pituitary adenoma. Pituitary. 9:317–321. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Black PM, Hsu DW, Klibanski A, et al:
Hormone production in clinically nonfunctioning pituitary adenomas.
J Neurosurg. 66:244–250. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gittoes NJ: Current perspectives on the
pathogenesis of clinically non-functioning pituitary tumours. J
Endocrinol. 157:177–186. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Katznelson L, Alexander JM and Klibanski
A: Clinical review 45: Clinically nonfunctioning pituitary
adenomas. J Clin Endocrinol Metab. 76:1089–1094. 1993.PubMed/NCBI
|
|
8
|
Korbonits M and Carlsen E: Recent clinical
and pathophysiological advances in non-functioning pituitary
adenomas. Horm Res. 71(Suppl 2): 123–130. 2009. View Article : Google Scholar
|
|
9
|
Colao A, Pivonello R, Di Somma C,
Savastano S, Grasso LF and Lombardi G: Medical therapy of pituitary
adenomas: effects on tumor shrinkage. Rev Endocr Metab Disord.
10:111–123. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Colao A, Di Somma C, Pivonello R, Faggiano
A, Lombardi G and Savastano S: Medical therapy for clinically
non-functioning pituitary adenomas. Endocr Relat Cancer.
15:905–915. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bianchi S, Dotti MT and Federico A:
Physiology and pathology of notch signalling system. J Cell
Physiol. 207:300–308. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lai EC: Notch signaling: control of cell
communication and cell fate. Development. 131:965–973. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Artavanis-Tsakonas S, Rand MD and Lake RJ:
Notch signaling: cell fate control and signal integration in
development. Science. 284:7701999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Greenwald I: LIN-12/Notch signaling:
lessons from worms and flies. Genes Dev. 12:1751–1762. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Miele L, Miao H and Nickoloff BJ: NOTCH
signaling as a novel cancer therapeutic target. Curr Cancer Drug
Targets. 6:313–323. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Indraccolo S, Minuzzo S, Masiero M and
Amadori A: Ligand-driven activation of the notch pathway in T-ALL
and solid tumors: why Not(ch)? Cell Cycle. 9:80–85. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Park JT, Li M, Nakayama K, et al: Notch3
gene amplification in ovarian cancer. Cancer Res. 66:6312–6318.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamaguchi N, Oyama T, Ito E, et al: NOTCH3
signaling pathway plays crucial roles in the proliferation of
ErbB2-negative human breast cancer cells. Cancer Res. 68:1881–1888.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Serafin V, Persano L, Moserle L, et al:
Notch3 signalling promotes tumour growth in colorectal cancer. J
Pathol. 224:448–460. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen X, Stoeck A, Lee SJ, Shih Ie M, Wang
MM and Wang TL: Jagged1 expression regulated by Notch3 and
Wnt/beta-catenin signaling pathways in ovarian cancer. Oncotarget.
1:210–218. 2010.PubMed/NCBI
|
|
21
|
Rodilla V, Villanueva A, Obrador-Hevia A,
et al: Jagged1 is the pathological link between Wnt and Notch
pathways in colorectal cancer. Proc Natl Acad Sci USA.
106:6315–6320. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jundt F, Probsting KS, Anagnostopoulos I,
et al: Jagged1-induced Notch signaling drives proliferation of
multiple myeloma cells. Blood. 103:3511–3515. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Choi JH, Park JT, Davidson B, Morin PJ,
Shih Ie M and Wang TL: Jagged-1 and Notch3 juxtacrine loop
regulates ovarian tumor growth and adhesion. Cancer Res.
68:5716–5723. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Moreno CS, Evans CO, Zhan X, Okor M,
Desiderio DM and Oyesiku NM: Novel molecular signaling and
classification of human clinically nonfunctional pituitary adenomas
identified by gene expression profiling and proteomic analyses.
Cancer Res. 65:10214–10222. 2005. View Article : Google Scholar
|
|
25
|
Evans CO, Moreno CS, Zhan X, et al:
Molecular pathogenesis of human prolactinomas identified by gene
expression profiling, RT-qPCR, and proteomic analyses. Pituitary.
11:231–245. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Miao Z, Miao Y, Lin Y and Lu X:
Overexpression of the Notch3 receptor in non-functioning pituitary
tumours. J Clin Neurosci. 19:107–110. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
|
|
28
|
Konishi J, Yi F, Chen X, Vo H, Carbone DP
and Dang TP: Notch3 cooperates with the EGFR pathway to modulate
apoptosis through the induction of Bim. Oncogene. 29:589–596. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Haruki N, Kawaguchi KS, Eichenberger S, et
al: Dominant-negative Notch3 receptor inhibits mitogen-activated
protein kinase pathway and the growth of human lung cancers. Cancer
Res. 65:3555–3561. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ohashi S, Natsuizaka M, Yashiro-Ohtani Y,
et al: NOTCH1 and NOTCH3 coordinate esophageal squamous
differentiation through a CSL-dependent transcriptional network.
Gastroenterology. 139:2113–2123. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Masieron M, Minuzzo S, Pusceddu I, et al:
Notch3-mediated regulation of MKP-1 levels promotes survival of T
acute lymphoblastic leukemia cells. Leukemia. 25:588–598. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Desiderio DM and Zhan X: The human
pituitary proteome: the characterization of differentially
expressed proteins in an adenoma compared to a control. Cell Mol
Biol (Noisy-le-grand). 49:689–712. 2003.PubMed/NCBI
|
|
33
|
Goldberg LB, Aujla PK and Raetzman LT:
Persistent expression of activated Notch inhibits corticotrope and
melanotrope differentiation and results in dysfunction of the HPA
axis. Dev Biol. 358:23–32. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhu X, Zhang J, Tollkuhn J, et al:
Sustained Notch signaling in progenitors is required for sequential
emergence of distinct cell lineages during organogenesis. Genes
Dev. 20:2739–2753. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dutta S, Dietrich JE, Westerfield M and
Varga ZM: Notch signaling regulates endocrine cell specification in
the zebrafish anterior pituitary. Dev Biol. 319:248–257. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zeng Q, Li S, Chepeha DB, et al: Crosstalk
between tumor and endothelial cells promotes tumor angiogenesis by
MAPK activation of Notch signaling. Cancer Cell. 8:13–23. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Steg AD, Katre AA, Goodman B, et al:
Targeting the notch ligand JAGGED1 in both tumor cells and stroma
in ovarian cancer. Clin Cancer Res. 17:5674–5685. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bellavia D, Campese AF, Alesse E, et al:
Constitutive activation of NF-kappaB and T-cell leukemia/lymphoma
in Notch3 transgenic mice. EMBO J. 19:3337–3348. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Vacca A, Felli MP, Palermo R, et al:
Notch3 and pre-TCR interaction unveils distinct NF-kappaB pathways
in T-cell development and leukemia. EMBO J. 25:1000–1008. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Monahan P, Rybak S and Raetzman LT: The
notch target gene HES1 regulates cell cycle inhibitor expression in
the developing pituitary. Endocrinology. 150:4386–4394. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kita A, Imayoshi I, Hojo M, et al: Hes1
and Hes5 control the progenitor pool, intermediate lobe
specification, and posterior lobe formation in the pituitary
development. Mol Endocrinol. 21:1458–1466. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Jordan S, Lidhar K, Korbonits M, Lowe DG
and Grossman AB: Cyclin D and cyclin E expression in normal and
adenomatous pituitary. Eur J Endocrinol. 143:R1–R6. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Shih IeM and Wang TL: Notch signaling,
gamma-secretase inhibitors, and cancer therapy. Cancer Res.
67:1879–1882. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tammam J, Ware C, Efferson C, et al:
Down-regulation of the Notch pathway mediated by a gamma-secretase
inhibitor induces anti-tumour effects in mouse models of T-cell
leukaemia. Br J Pharmacol. 158:1183–1195. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Konishi J, Kawaguchi KS, Vo H, et al:
Gamma-secretase inhibitor prevents Notch3 activation and reduces
proliferation in human lung cancers. Cancer Res. 67:8051–8057.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kondratyev M, Kreso A, Hallett RM, et al:
Gamma-secretase inhibitors target tumor-initiating cells in a mouse
model of ERBB2 breast cancer. Oncogene. 31:93–103. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ramakrishnan V, Ansell S, Haug J, et al:
MRK003, a γ-secretase inhibitor exhibits promising in vitro
pre-clinical activity in multiple myeloma and non-Hodgkin’s
lymphoma. Leukemia. 26:340–348. 2012.
|