Introduction
Radiation therapy (RT) is currently the cornerstone
of the treatment of malignant thoracic diseases, including lung
cancer, breast cancer, malignant lymphoma, esophageal cancer and
thymoma. To ensure coverage of the tumor, the irradiation of normal
tissues surrounding the tumor is unavoidable and may result in
symptomatic injury (1).
Radiation-induced pulmonary injuries (RIPIs), including
radiation-induced pneumonitis and lung fibrosis, limit the
therapeutic ratios of tumor treatment and reduce the quality of
life in long-term survivors (1).
Thus, effective prevention and control of RIPIs is extremely
important in these patients.
The pathological process of RIPIs is complex,
beginning with an acute inflammatory response that includes
alveolar cell depletion and interstitial inflammation in the lung.
Irreversible fibrosis, including fibroblast proliferation with
collagen accumulation, occurs in the late stages of this process
and eventually leads to the loss of normal lung structure (2). The biological effects of ionizing
radiation begin with the direct generation of various reactive
oxygen species (ROS), which cause oxidative damage to DNA, proteins
and lipids, as well as the activation of transcription factors and
signal transduction pathways (3).
Oxidative damage to cellular components in the lung leads to cell
damage and even cell death, and triggers inflammation that induces
reparative processes and results in radiation-induced lung fibrosis
(4). Thus, molecules with
radical-scavenging properties show particular promise as
radio-protectors (5). Animal
studies have shown that antioxidant therapy reduces the extent of
radiation-induced lung damage: hydrogen therapy has been shown to
reduce cell damage, improve the viability of ionizing A549 cells
and attenuate irradiation-induced damage by reducing oxidative
stress (6); a superoxide dismutase
(SOD) mimetic has been demonstrated to increase the tolerance of
ionizing radiation in the lungs of rats (7); and amifostine was shown to reduce
radiation-induced damage by scavenging oxygen and oxygen free
radicals (8).
Quercetin, or 3,3’,4’,5,7-pentahydroxyflavone, is
categorized as a flavonol, one of the six subclasses of flavonoid
compounds (9). The protective
effects of flavonoids in biological systems are ascribed to their
capacity for transferring electrons to free radicals, activating
antioxidant enzymes and inhibiting oxidative stress (10). Quercetin has a superior antioxidant
activity due to the presence of the catechol group in the B ring
and the OH group at position 3 on the AC ring. These structural
features allow quercetin to donate hydrogen to scavenge free
radicals and increase the stability of flavonoid radicals (11). Quercetin is known to possess marked
antioxidative, anti-inflammatory and antifibrotic capacities.
Animal experiments have demonstrated its ability to scavenge oxygen
free radicals, inhibit lipid oxidation and affect the glutathione
redox status (12,13). Quercetin has been shown to improve
the inflammatory status by reducing tumor necrosis factor (TNF)-α
and inducible nitric oxide synthase production in obese Zucker rats
(14). In vitro, quercetin
inhibits keloid fibroblast proliferation, collagen production and
keloid contraction by suppressing transforming growth factor
(TGF)-β/Smad signaling (15). In
vivo, quercetin has been shown to improve liver histology and
reduce collagen content in rats with carbon tetrachloride-induced
cirrhosis (16). We thus
hypothesized that quercetin would be an ideal candidate for the
amelioration of RIPIs.
At present, the routine treatment for acute
radiation pneumonitis includes a combined regime of adrenal cortex
hormones and antibiotics, but this treatment does not effectively
prevent or cure radiation pneumonitis or fibrosis. The present
study aimed to investigate the effect and potential mechanism of
the action of quercetin liposomes on RIPIs in a murine model.
Materials and methods
Quercetin liposome preparation
Since quercetin naturally has poor water solubility,
laboratory-prepared quercetin liposomes characterized by improved
solubility and increased in vivo absorbability (State Key
Laboratory of Biotherapy, West China Hospital, Sichuan University,
Chengdu, Sichuan, China) were used.
The quercetin liposomes were prepared as described
previously (17). Briefly, mixtures
of lecithin/cholesterol/PEG 4000/quercetin in 13:4:1:6 weight
ratios were dissolved in chloroform/methanol (3:1, v/v) and
evaporated until dry under reduced pressure in a rotary evaporator.
The dried lipid films were sonicated in 5% glucose solution in a
homothermal container. The final products were concentrated,
lyophilized under vacuum for 5 h and stored at −20°C. This
end-product has good solubility and may be used directly or
dissolved in saline intraperitoneally.
Animal model and experimental
protocol
All animal procedures were approved by the
Laboratory Animal Care Committee of Sichuan Province. Female C57BL
mice (Experimental Animal Center of Sichuan University, Chengdu,
Sichuan, China) aged 6–8 weeks, with approximate body weights of
18–20 g, were used in this study.
A total of 69 mice were randomly divided into three
groups: a control group; an RT plus saline (RT+NS) group that
received intraperitoneal injections of 200 μl saline 2 h
prior to irradiation and on days 1–28 subsequent to RT; and an RT
plus quercetin liposome (RT+QU) group that received intraperitoneal
injections of 5 mg/kg quercetin liposome, based on a previous study
(17), 2 h prior to irradiation and
on days 1–28 subsequent to RT.
For the thoracic irradiation, the mice were
anesthetized by the intraperitoneal administration of 10 ml/kg 3.5%
chloral hydrate. A single dose of cobalt-60 γ radiation (GWXJ80;
Nuclear Power Institute of China, Chengdu, China) was administered
to the entire thorax (0.8953 Gy/min; source-skin distance, 80 cm)
of each mouse. Organs above and below the thorax were shielded.
At 1, 4, 8 and 24 weeks post-RT, four or five mice
in each group were sacrificed. Peripheral blood samples and
bronchoalveolar lavage fluid (BALF) were obtained, the left lung
was fixed in 4% paraformaldehyde and the right lung was
cryopreserved at −80°C.
Malondialdehyde (MDA) content and SOD and
glutathione peroxidase (GSH-PX) activities
Tissue from one lobe of each right lung was
homogenized in 5% phosphate-buffered saline. The homogenate was
then centrifuged at 800 × g for 10 min and the clear upper
supernatant fluid was used. The MDA content and the SOD and GSH-PX
activities in the lung were measured using respective kits (Nanjing
Jiancheng Bioengineering Institute, Nanjing, China) according to
the manufacturer’s instructions.
BALF analysis
At 1, 4, 8 and 24 weeks post-irradiation, the mice
were sacrificed, an open tracheotomy was performed and a small
plastic tube was inserted into the trachea. BALF was extracted
three times with 2 ml physiological saline. The BALF was
centrifuged (400 × g, 15 min) and the cell pellet was suspended in
1 ml modified Hank’s balanced salt solution. Total nucleated and
differential cell counts were performed on cellular monolayers
prepared by cytocentrifugation at 800 rpm for 10 min, followed by
hematoxylin and eosin (HE) staining. The percentages of
inflammatory cell types (including neutrophils and lymphocytes)
that were present were assessed by differential counts of 400
cells.
TNF-α and TGF-β1 concentrations in
plasma
The TGF-β1 and TNF-α contents of the plasma were
measured by sandwich enzyme-linked immunosorbent assays (ABC-ELISA,
R&D Systems, Minneapolis, MN, USA), according to the
manufacturer’s instructions.
Hydroxyproline (HP) assay
Collagen deposition was estimated by determining the
total HP content of one lobe of each right lung using alkaline
hydrolysis (Nanjing Jiancheng Bioengineering Institute). Briefly,
the lung tissue in the test tube was weighed, 1 ml hydrolyzate was
added and hydrolysis was performed in a boiling water bath for 20
min to regulate the pH (pH 6.0–6.8). The hydrolyzate was diluted by
adding activated carbon, then the contents of the tube were mixed
thoroughly and centrifuged at 3,500 rpm for 10 min and 1 ml
supernatant was tested. Once the reagents had been added to the
reaction mixture, the supernatant absorbance was measured at 550
nm.
Histology and immunocytochemistry
The left lungs were fixed by an intratracheal
instillation of 4% paraformaldehyde, allowed to cure overnight,
embedded in paraffin and cut into 5-μm thick sections.
Certain sections were stained with HE and Masson’s trichrome for
the determination of collagen content. Pulmonary fibrosis was
scored using the scale developed by Ashcroft et al (18). Briefly, entire fields of 15 sections
were scanned and each field was graded visually on a scale ranging
from 0 (normal) to 8 (total fibrotic obliteration of the field).
The mean of the scores obtained for all fields was used as the
visual fibrosis score. The remaining sections were
immunocytochemically stained with anti-TGF-β1 antibody (Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA) to detect active TGF-β1
expression. Five fields were randomly selected for each mouse and
three mice from each group were examined; thus, a total of 15
sections were analyzed for each group. The number of cells showing
active TGF-β1 expression within each field was counted under a
light microscope at ×400 magnification (CX41RF; Olympus; Tokyo,
Japan).
Statistical analysis
Data are presented as the mean ± standard deviation.
The statistical analysis was performed by a one-way analysis of
variance, followed by Dunnet’s t-test. P<0.05 was considered to
indicate a statistically significant difference.
Results
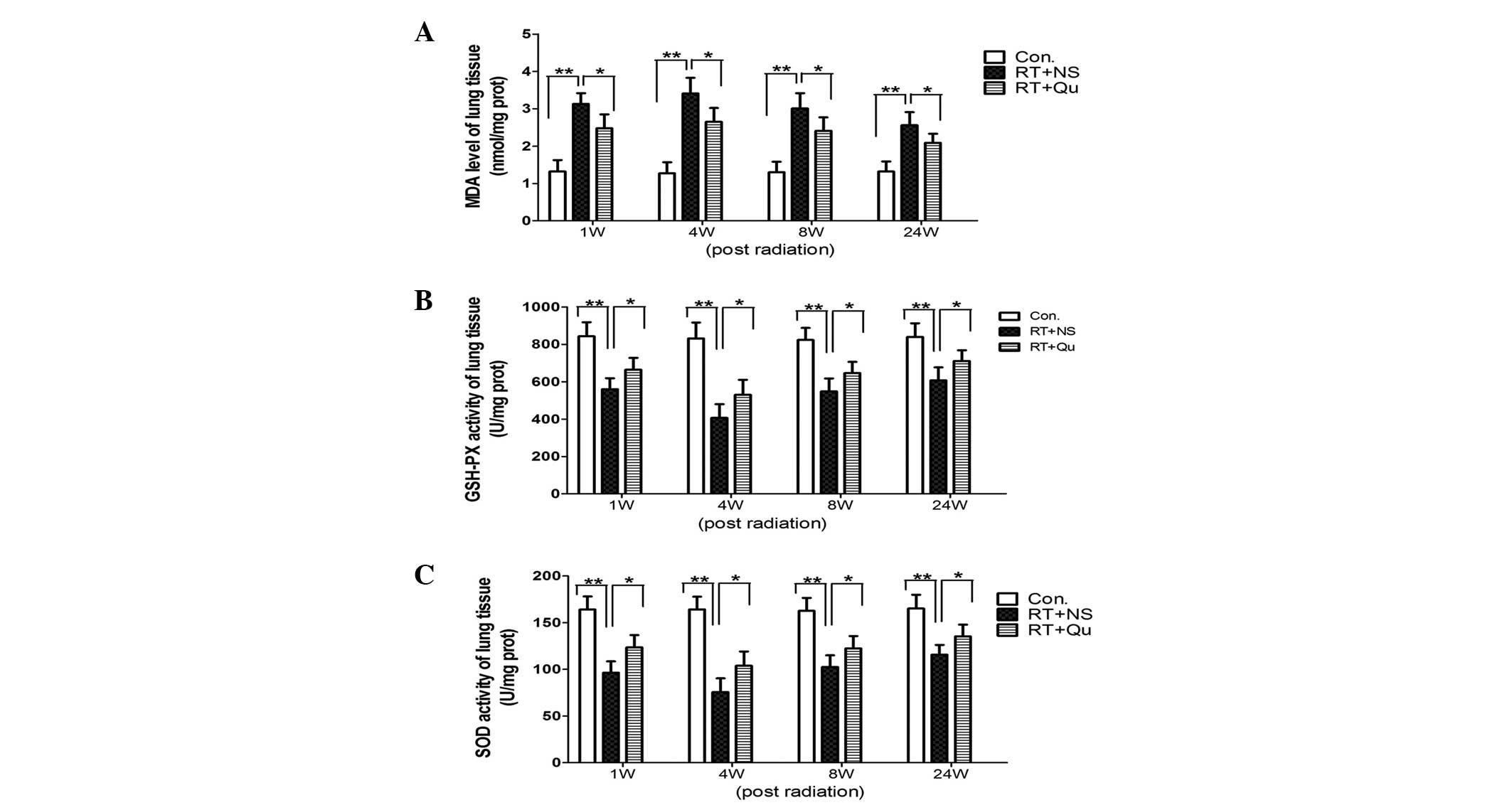
MDA content and SOD and GSH-PX activity
in lung tissue
From 1 to 24 weeks post-RT, the MDA content of the
lung tissues increased significantly (all P<0.01 vs. control
group; Fig. 1). Quercetin liposome
administration significantly reduced the MDA content (all P<0.05
vs. RT+NS group).
From 1 to 24 weeks post-RT, the SOD and GSH-PX
activities in the lung tissue significantly decreased (all
P<0.01 vs. control group; Fig.
1). Quercetin liposome administration significantly increased
the SOD and GSH-PX activities (all P<0.05 vs. RT+NS group).
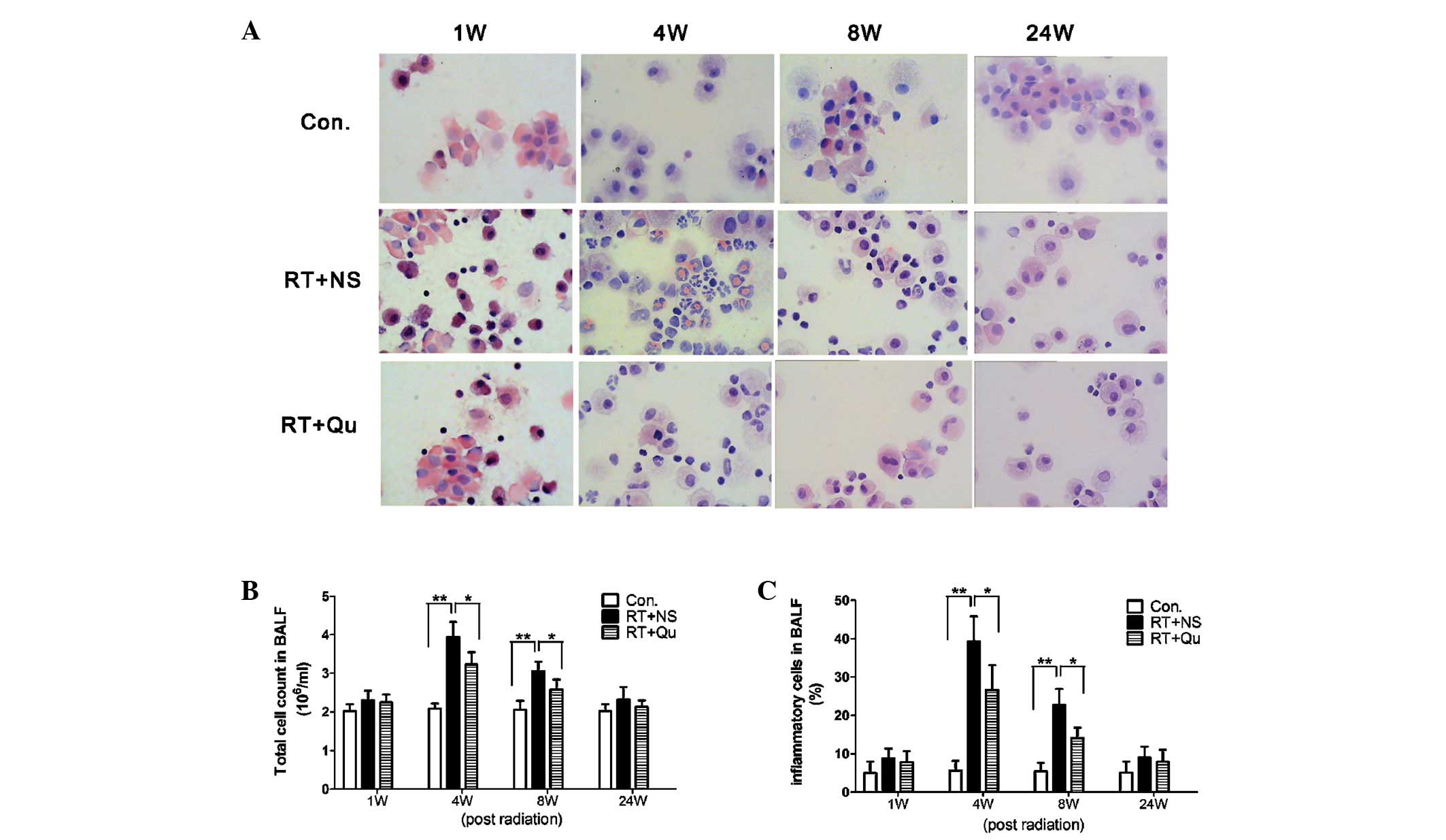
Total cell counts and proportions of
inflammatory cells in BALF
Epithelial cells and macrophages were the main cell
types identified in the BALF from rats in the control group and the
presence of lymphocytes were rare (Fig.
2). At 4 and 8 weeks post-RT, the total cell counts of the BALF
and the percentages of inflammatory cells were increased
significantly (all P<0.01 vs. control group). In the RT+QU
group, the total cell counts of the BALF and the percentages of
inflammatory cells were significantly reduced (all P<0.05 vs.
RT+NS group) at 4 and 8 weeks post-RT.
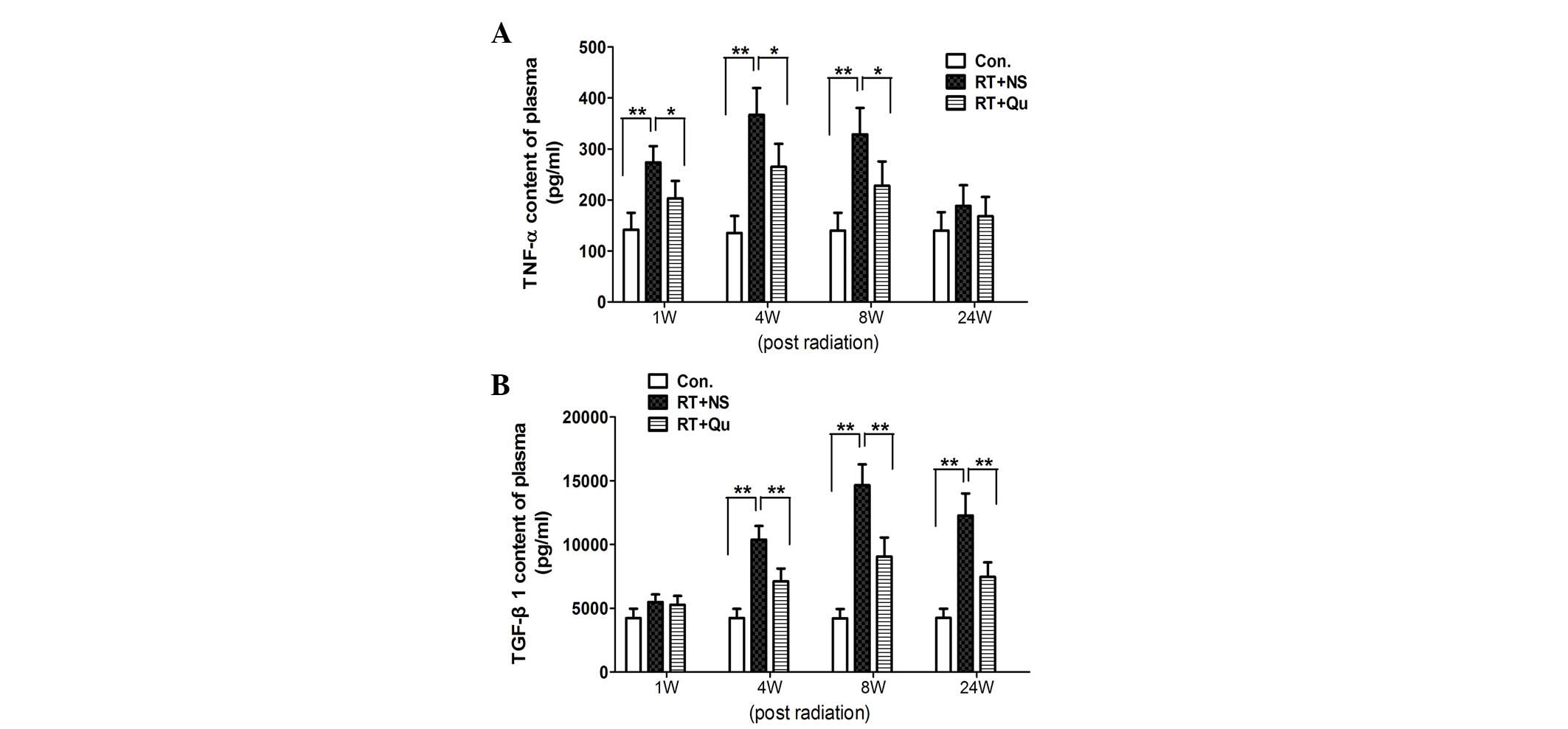
TNF-α and TGF-β1 concentrations in
plasma
In the control group, the TNF-α concentration in
plasma was 135.1±33.6 pg/ml (Fig.
3). At 1, 4 and 8 weeks post-RT, the TNF-α concentrations
increased significantly, resulting in concentrations of 273.4±32.2,
367.0±52.5 and 328.8±51.7 pg/ml, respectively (all P<0.01 vs.
control group). In the RT+QU group, the TNF-α concentrations
declined significantly, with results of 203.1±34.2, 264.7±45.4 and
228.0±47.3 pg/ml, respectively (all P<0.05 vs. RT+NS group).
In the control group, the TGF-β1 concentration in
plasma was 4,207.2±732.1 pg/ml (Fig.
3). At 4, 8 and 24 weeks post-RT, the TGF-β1 concentrations
increased significantly to 10,373.2±1,084.8, 14,650.6±1,632.6 and
12,262.5±1,740.7 pg/ml, respectively (all P<0.01 vs. control
group). In the RT+QU group, the TGF-β1 concentrations declined
significantly to 7,100.8±1,009.4, 9,056.6±1,484.5 and
7,466.8±1,138.5 pg/ml, respectively (all P<0.01 vs. RT+NS
group).
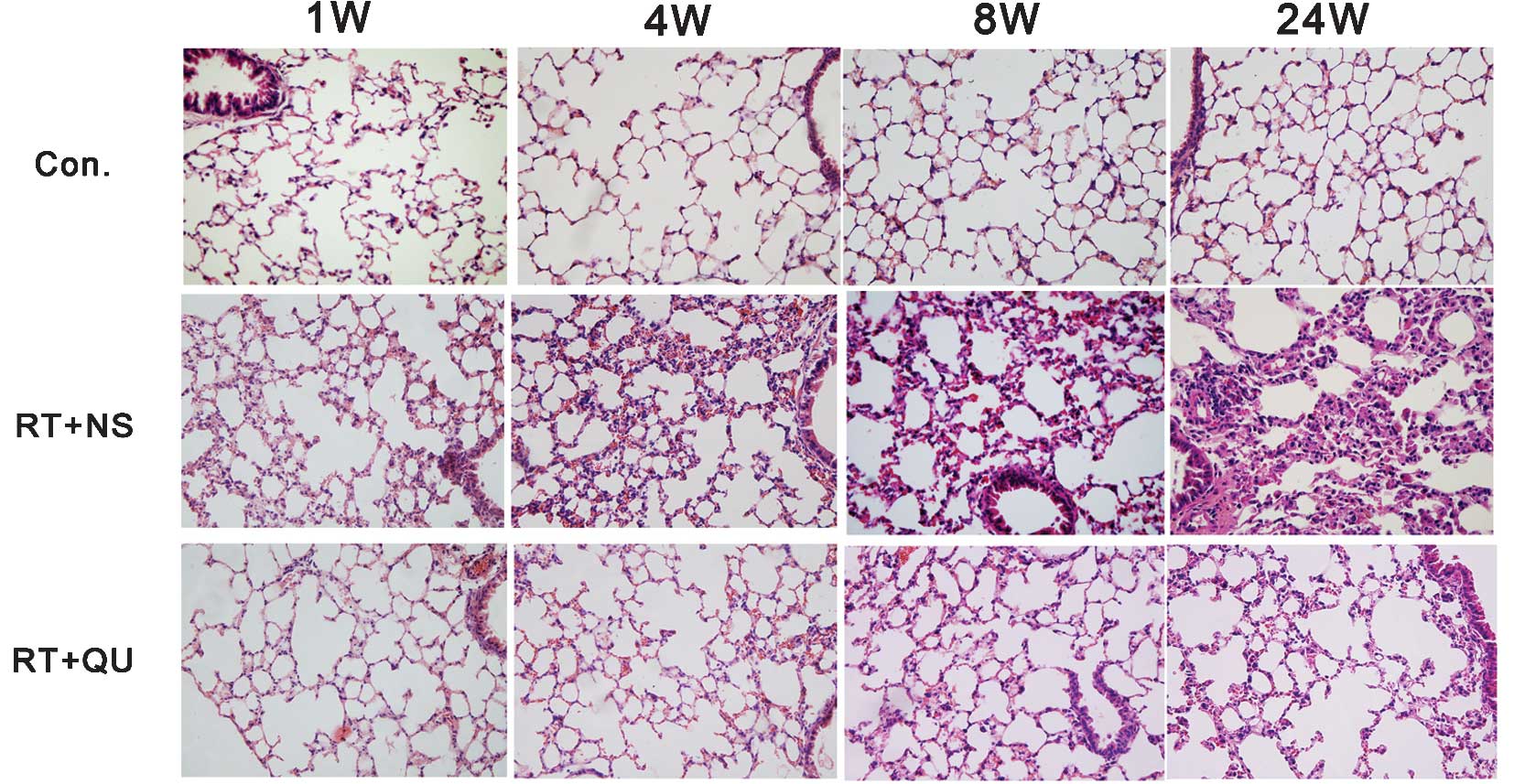
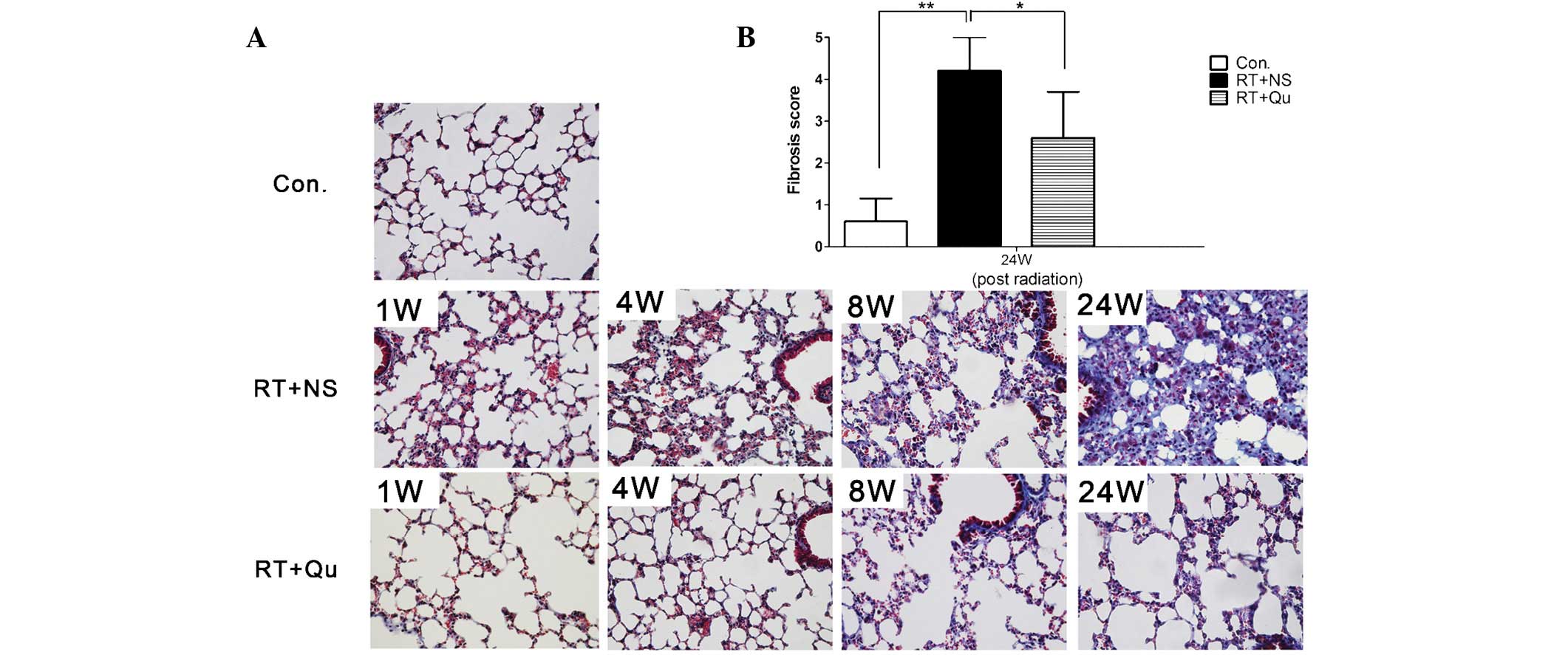
Histological changes in lung tissue
Subsequent to irradiation, two pathological phases
of RIPIs were observed; an initial phase of acute and subacute
pneumonitis (1–8 weeks), followed by late fibrosis (24 weeks;
Figs. 4 and 5). In the pneumonitis phase, numerous
inflammatory cells infiltrated the alveoli and alveolar walls and
the lung interstitium was thickened, although minimal fibrosis was
present on the alveolar walls, which were stained with Masson’s
trichrome. In the late fibrosis stage, lung interstitium thickening
and inflammatory cell infiltration were observed and the alveolar
structure became disordered and collapsed. Notably, Masson’s
trichrome staining revealed diffuse fibrous changes in the alveolar
walls. The lung fibrosis score at 24 weeks post-irradiation was
significantly higher (4.2±0.8) compared with the control group
(0.6±0.55; P<0.01). However, the damage was clearly minor in the
RT+QU group, with a lung fibrosis score of 2.6±1.1 at 24 weeks
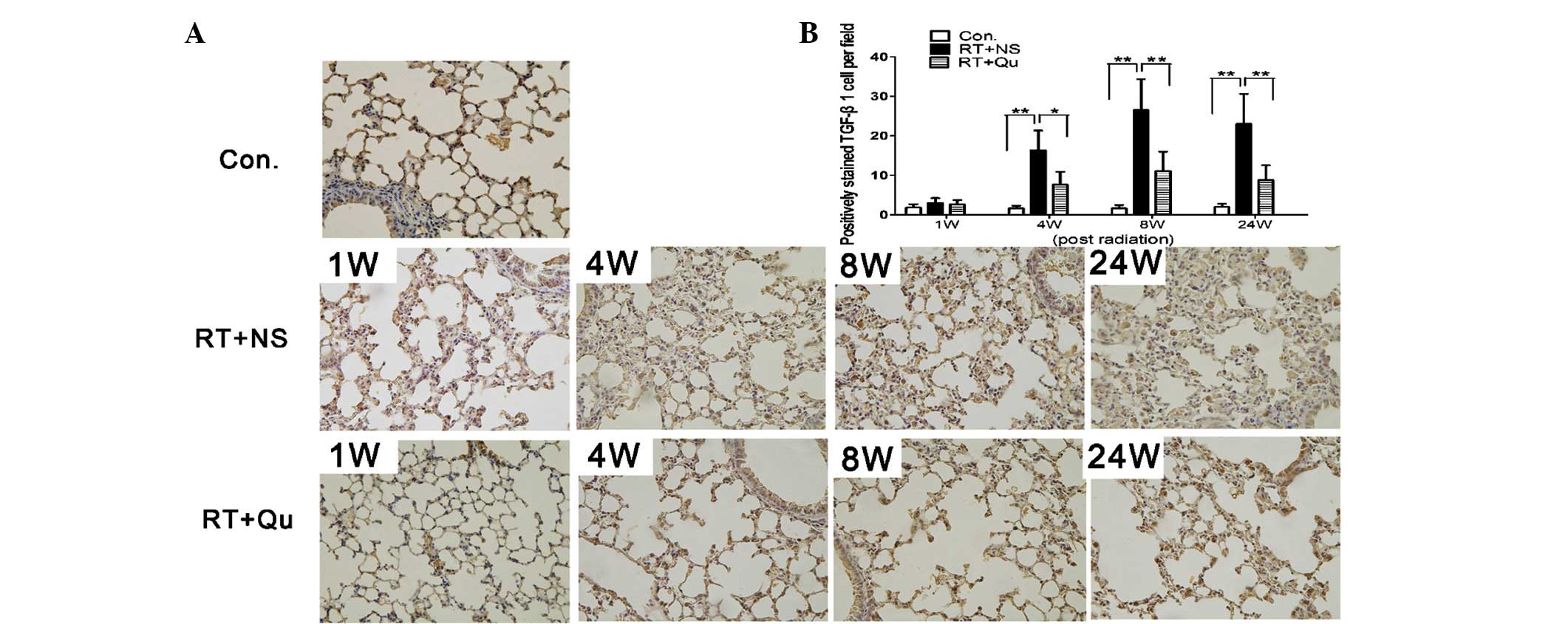
post-irradiation (P<0.05). The cells with active TGF-β1
expression infiltrated the lung tissue between 4 and 24 weeks
post-irradiation (Fig. 6), although
the degree of infiltration was significantly lower in the RT+QU
group compared with the RT+NS group (all P<0.05 vs. RT+NS
group).
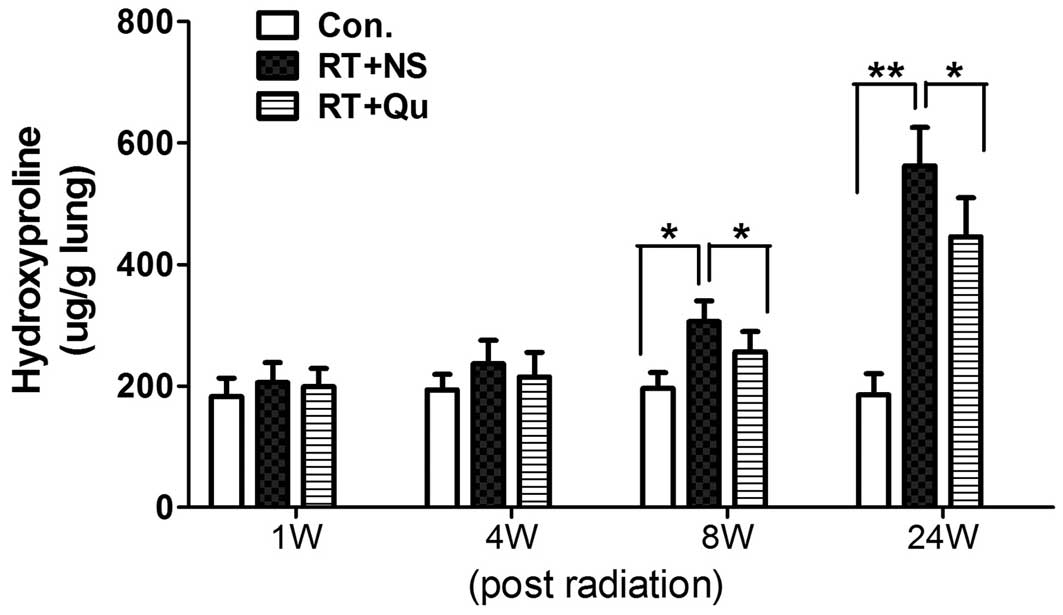
HP content in the lung tissue
Fibrosis is characterized by collagen deposition,
and the HP content in the lung tissues reflects the proportion of
tissue with collagen fibers. The lung tissues in the control group
contained 185.5±34.4 μg HP/g lung (Fig. 7). The HP content began to increase
significantly in the first 8 weeks post-irradiation and peaked at
24 weeks (562.7±63.2 μg/g wet tissue; P<0.01). Quercetin
liposome administration noticeably reduced the HP content of the
lung tissue (446.0±64.1 μg/g lung at 24 weeks;
P<0.05).
Discussion
The present findings of marked increases in MDA
content and reductions in SOD and GSH-PX activities between 1 and
24 weeks after whole-lung irradiation demonstrated oxidative stress
sustained from radiation-induced pneumonitis and lung fibrosis.
Ionizing radiation causes DNA damage through direct and indirect
mechanisms (19); sensitive
molecules in cells are directly damaged and interactions between
radiation and water molecules in cells lead to the production of
ROS, including superoxide anion radicals, hydrogen peroxide and
hydroxyl radicals. Hydroxyl radicals are responsible for an
estimated 60–70% of all ionizing radiation-induced cell damage
(3,19). The radiation-induced burst of ROS
generation is transient, but radiation also damages critical
biomolecules governing the metabolic production of ROS, including
mitochondria and oxidoreductase enzymes. Oxidative stress also
contributes to the biological effects of ionizing radiation long
after exposure (20). Leach et
al (21) reported that the
transient generation of ROS occurs within minutes of cell exposure
to ionizing radiation, damaging mitochondrial permeability and
resulting in the constant enhancement of ROS generation. Previous
studies have shown that oxidative damage is increased and
antioxidative capacities are decreased in radiation-induced lung
injury (6,22). The effective protection of
antioxidants have also been shown to indirectly reflect the
potential causative role of oxidative stress in the development of
RIPIs (7).
The antioxidant activities of quercetin are
attributed to numerous factors, including free radical scavenging,
protection against lipid oxidation (23), up-regulation of anti-oxidant enzymes
and ROS trapping by direct hydrogen ion donation (12). However, quercetin administration has
been hampered by its extreme water insolubility. The encapsulation
of quercetin in liposomes improves its water solubility, prolonging
circulation times in the blood and accumulation times in the lung
(17). Significantly, the use of
liposomal quercetin was shown to reduce the injection dose compared
with free quercetin (17). Hence,
in the present study, intraperitoneal injections of quercetin
liposome were administered prior to and following RT. A lower MDA
content and higher SOD and GSH-PX activities were observed in the
lung tissue in the RT+QU group compared with the RT+NS group,
demonstrating that quercetin inhibited pulmonary oxidative
damage.
Inflammation may be central in the initiation and
establishment of RIPIs (2). Changes
in cell populations in the BALF have often been considered to
reflect inflammatory changes in the lung (24). As a pro-inflammatory cytokine, TNF-α
is likely to be involved in the early phase of RIPIs. Hence, the
proportions of inflammatory cells in the BALF and TNF-α
concentrations in the plasma were measured to estimate the extent
of the inflammatory response. It was observed that the quercetin
liposome significantly decreased the total cell counts and the
proportion of inflammatory cells in the BALF, and also reduced
plasma TNF-α concentrations. A histological examination revealed
the suppression of the inflammatory response in the RT+QU group.
The anti-inflammatory effects of quercetin may be attributed to the
interplay between oxidative stress and inflammation (23). ROS are involved not only in the
occurrence of oxidative stress, but also in the promotion of
inflammatory processes. ROS are key mediators of inflammatory
reactions in atherosclerosis (25).
They are able to activate transcription factors such as nuclear
factor (NF)-κB and activator protein-1, which induce the production
of cytokines such as TNF-α (26).
Consequently, ROS scavenging not only prevents oxidative stress,
but also mitigates inflammation. Quercetin has been reported to
inhibit TNF-α production and gene expression via NF-κB modulation
(27). In animal models of allergic
airway inflammation and asthma, quercetin has been demonstrated to
reduce inflammatory cell infiltration and inflammatory cytokine
production (28).
Tissue fibrosis is the excessive accumulation of
collagen. TGF-β1 is a key cytokine in the fibrotic process that
activates myofibroblast progenitors and upregulates collagen
protein synthesis (2). Hence, in
the present study, the plasma TGF-β1 concentrations and HP content
in the lung tissue were measured, and Masson’s trichrome staining
was used to estimate the extent of lung fibrosis. It was observed
that quercetin liposomes significantly reduced the plasma TGF-β1
concentrations and HP content in the lung tissue. A histological
examination revealed the suppression of TGF-β1 expression and
collagen deposition in the lung. The lung fibrosis scores were
significantly lower in the RT+QU group compared with the RT+NS
group. The mechanism of quercetin’s antifibrotic effects may also
be associated in part with the reduction of oxidative stress
(29). Oxidative stress is
postulated to play an important role in a wide range of fibrotic
diseases, including atherosclerosis, cardiac fibrosis and
idiopathic lung fibrosis (30). ROS
and lipid peroxidation products stimulate fibrogenic cytokines that
act as chemoattractants, mitogens and differentiating agents for
smooth muscle cells (25,31). TGF-β isoforms are secreted in a
latent complex, and the release of TGF-β from this complex is
called activation. The ROS-mediated oxidation of a methionine
residue in the latent complex releases TGF-β from extracellular
reservoirs (32). In vitro,
quercetin has been shown to suppress TGF-β-induced collagen
production in normal human lung fibroblasts (33). In biliary-obstructed rats, quercetin
has been shown to maintain an antioxidant defense and reduce
oxidative damage, ameliorating liver fibrosis (29).
In conclusion, oxidative stress in the lung leads to
radiation-induced pneumonitis and lung fibrosis. The present study
demonstrated that quercetin liposomes inhibit pulmonary oxidative
stress, alleviating radiation-induced acute pneumonitis and late
fibrosis. Thus, quercetin effectively protected lung tissue against
RIPIs.
Acknowledgements
The present study was supported by the
National Major Project of China (No.2011ZX09302-001-01), and the
Natural Science Fund of China (No.81172131).
References
|
1.
|
Stone HB, Coleman CN, Anscher MS and
McBride WH: Effects of radiation on normal tissue: consequences and
mechanisms. Lancet Oncol. 4:529–536. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Tsoutsou PG and Koukourakis MI: Radiation
pneumonitis and fibrosis: mechanisms underlying its pathogenesis
and implications for future research. Int J Radiat Oncol Biol Phys.
66:1281–1293. 2006. View Article : Google Scholar
|
|
3.
|
Zhao W and Robbins ME: Inflammation and
chronic oxidative stress in radiation-induced late normal tissue
injury: therapeutic implications. Curr Med Chem. 16:130–143. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Graves PR, Siddiqui F, Anscher MS and
Movsas B: Radiation pulmonary toxicity: from mechanisms to
management. Semin Radiat Oncol. 20:201–207. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Karbownik M and Reiter RJ: Antioxidative
effects of melatonin in protection against cellular damage caused
by ionizing radiation. Proc Soc Exp Biol Med. 225:9–22. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Terasaki Y, Ohsawa I, Terasaki M, et al:
Hydrogen therapy attenuates irradiation-induced lung damage by
reducing oxidative stress. Am J Physiol Lung Cell Mol Physiol.
301:L415–L426. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Vujaskovic Z, Batinic-Haberle I, Rabbani
ZN, et al: A small molecular weight catalytic metalloporphyrin
antioxidant with superoxide dismutase (SOD) mimetic properties
protects lungs from radiation-induced injury. Free Radic Biol Med.
33:857–863. 2002. View Article : Google Scholar
|
|
8.
|
Koukourakis MI: Amifostine in clinical
oncology: current use and future applications. Anticancer Drugs.
13:181–209. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Kelly GS: Quercetin. Monograph Altern Med
Rev. 16:172–194. 2011.
|
|
10.
|
Heim KE, Tagliaferro AR and Bobilya DJ:
Flavonoid antioxidants: chemistry, metabolism and
structure-activity relationships. J Nutr Biochem. 13:572–584. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Heijnen CG, Haenen GR, Oostveen RM,
Stalpers EM and Bast A: Protection of flavonoids against lipid
peroxidation: the structure activity relationship revisited. Free
Radic Res. 36:575–581. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Cai Q, Rahn RO and Zhang R: Dietary
flavonoids, quercetin, luteolin and genistein, reduce oxidative DNA
damage and lipid peroxidation and quench free radicals. Cancer
Lett. 119:99–107. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Meyers KJ, Rudolf JL and Mitchell AE:
Influence of dietary quercetin on glutathione redox status in mice.
J Agric Food Chem. 56:830–836. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Rivera L, Morón R, Sánchez M, Zarzuelo A
and Galisteo M: Quercetin ameliorates metabolic syndrome and
improves the inflammatory status in obese Zucker rats. Obesity
(Silver Spring). 16:2081–2087. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Phan TT, Lim IJ, Chan SY, Tan EK, Lee ST
and Longaker MT: Suppression of transforming growth factor
beta/smad signaling in keloid-derived fibroblasts by quercetin:
implications for the treatment of excessive scars. J Trauma.
57:1032–1037. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Pavanato A, Tuñón MJ, Sánchez-Campos S, et
al: Effects of quercetin on liver damage in rats with carbon
tetrachloride-induced cirrhosis. Dig Dis Sci. 48:824–829. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Yuan ZP, Chen LJ, Fan LY, et al: Liposomal
quercetin efficiently suppresses growth of solid tumors in murine
models. Clin Cancer Res. 12:3193–3199. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Ashcroft T, Simpson JM and Timbrell V:
Simple method of estimating severity of pulmonary fibrosis on a
numerical scale. J Clin Pathol. 41:467–470. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Ward JF: DNA damage produced by ionizing
radiation in mammalian cells: identities, mechanisms of formation,
and reparability. Prog Nucleic Acid Res Mol Biol. 35:95–125. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Spitz DR, Azzam EI, Li JJ and Gius D:
Metabolic oxidation/reduction reactions and cellular responses to
ionizing radiation: a unifying concept in stress response biology.
Cancer Metastasis Rev. 23:311–322. 2004. View Article : Google Scholar
|
|
21.
|
Leach JK, Van Tuyle G, Lin PS,
Schmidt-Ullrich R and Mikkelsen RB: Ionizing radiation-induced,
mitochondria-dependent generation of reactive oxygen/nitrogen.
Cancer Res. 61:3894–3901. 2001.PubMed/NCBI
|
|
22.
|
De AK, Rajan RR, Krishnamoorthy L, Bhatt
MB and Singh BB: Oxidative stress in radiation-induced interstitial
pneumonitis in the rat. Int J Radiat Biol. 68:405–409. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Boots AW, Haenen GR and Bast A: Health
effects of quercetin: from antioxidant to nutraceutical. Eur J
Pharmacol. 585:325–337. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Hong JH, Jung SM, Tsao TC, et al:
Bronchoalveolar lavage and interstitial cells have different roles
in radiation-induced lung injury. Int J Radiat Biol. 79:159–167.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Madamanchi NR, Hakim ZS and Runge MS:
Oxidative stress in atherogenesis and arterial thrombosis: the
disconnect between cellular studies and clinical outcomes. J Thromb
Haemost. 3:254–267. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Rahman I: Oxidative stress, transcription
factors and chromatin remodelling in lung inflammation. Biochem
Pharmacol. 64:935–942. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Nair MP, Mahajan S, Reynolds JL, et al:
The flavonoid quercetin inhibits proinflammatory cytokine (tumor
necrosis factor alpha) gene expression in normal peripheral blood
mononuclear cells via modulation of the NF-kappa beta system. Clin
Vaccine Immunol. 13:319–328. 2006. View Article : Google Scholar
|
|
28.
|
Rogerio AP, Kanashiro A, Fontanari C, et
al: Anti-inflammatory activity of quercetin and isoquercitrin in
experimental murine allergic asthma. Inflamm Res. 56:402–408. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Peres W, Tuñón MJ, Collado PS, Herrmann S,
Marroni N and González-Gallego J: The flavonoid quercetin
ameliorates liver damage in rats with biliary obstruction. J
Hepatol. 33:742–750. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Yarnold J and Brotons MC: Pathogenetic
mechanisms in radiation fibrosis. Radiother Oncol. 97:149–161.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Poli G and Parola M: Oxidative damage and
fibrogenesis. Free Radic Biol Med. 22:287–305. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Jobling MF, Mott JD, Finnegan MT, et al:
Isoform-specific activation of latent transforming growth factor
beta (LTGF-beta) by reactive oxygen species. Radiat Res.
166:839–848. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Nakamura T, Matsushima M, Hayashi Y, et
al: Attenuation of transforming growth factor-β-]stimulated
collagen production in fibroblasts by quercetin-induced heme
oxygenase-1. Am J Respir Cell Mol Biol. 44:614–620. 2011.
|