Introduction
Hepatoblastoma (HB) is the most common malignancy of
the liver in infants and children, comprising ~1% of all pediatric
cancers (1–3). It is well known that a complete
surgical resection is vital to cure patients with HB. However, 50%
of cases do not have this chance due to advanced and unresectable
HB at the initial presentation (4–6). In
this situation, it is considered that a poor patient prognosis is
associated with a large tumor size, porta hepatis invasion and
metastatic spread (5–7). HB has been recognized to be a highly
chemosensitive tumor since the 1970s (8). Therefore, pre-operative systemic
chemotherapy acts to improve the prognosis of children with HB by
reducing the tumor size and increasing the feasibility of complete
resection. However, the toxicity of systemic chemotherapy,
including cardiac and bone marrow damage, is occasionally
significant (9). In order to reduce
these disadvantages, a new chemotherapeutic approach that is able
to improve the survival rates of these patients is urgently
required. Transcatheter arterial chemoembolization (TACE) has been
attempted for the treatment of unresectable HB and may be an
alternative to systemic chemotherapy (10–12).
The present study analyses the use of pre-operative TACE in 24
patients with HB who were treated over the last four years at the
Guangzhou Women and Children’s Medical Center (Guangzhou Children’s
Hospital, Guangzhou, Guangdong, China). According to the results of
these patients, the feasibility and effectiveness of pre-operative
TACE for infants and children with HB was investigated.
Materials and methods
Patients
The present study was approved by the Ethical
Committee of Guangzhou Women and Children’s Medical Center and
written informed consent was obtained from all patients’ legal
guardians. Between March 2007 and March 2011, 24 cases with HB were
considered suitable for the present study. These cases consisted of
14 males and 10 females, with ages ranging between 26 days and 41
months, with a mean age of 14.2 months. The chief clinical
manifestations of all patients were an abdominal mass, bloating and
significant elevation of the serum α-fetoprotein (AFP) level. All
cases were confirmed by computed tomography (CT) and live tumor
biopsy prior to TACE. Tumors were considered unresectable if they
were bilobar or multicenteric. According to the Children’s Cancer
Study Group (CCSG) staging system (13), there were three cases of stage IIA,
11 cases of stage IIB, five cases of stage IIIA and five cases of
stage IIIB.
Transcatheter arterial
chemoembolization
Under general anesthesia and continued monitoring,
including observations of ECG, blood pressure and oxygen saturation
measurements, the femoral artery was cannulated using the Seldinger
technique. A 4-Fr catheter was inserted through the femoral artery
under fluoroscopy via digital subtraction angiography (DSA). A
2.7-Fr microcatheter was selectively inserted through the catheter
into the tumor-feeding artery. Pirarubicin and cisplatin were used
as anticancer agents. According to the volume of the tumor,
pirarubicin (20–30 mg/m2) and cisplatin (50–60
mg/m2) emulsified in lipiodol were injected into the
artery supplying the tumor, followed by superselective embolization
using polyvinyl alcohol (PVA) until the blood flow was completely
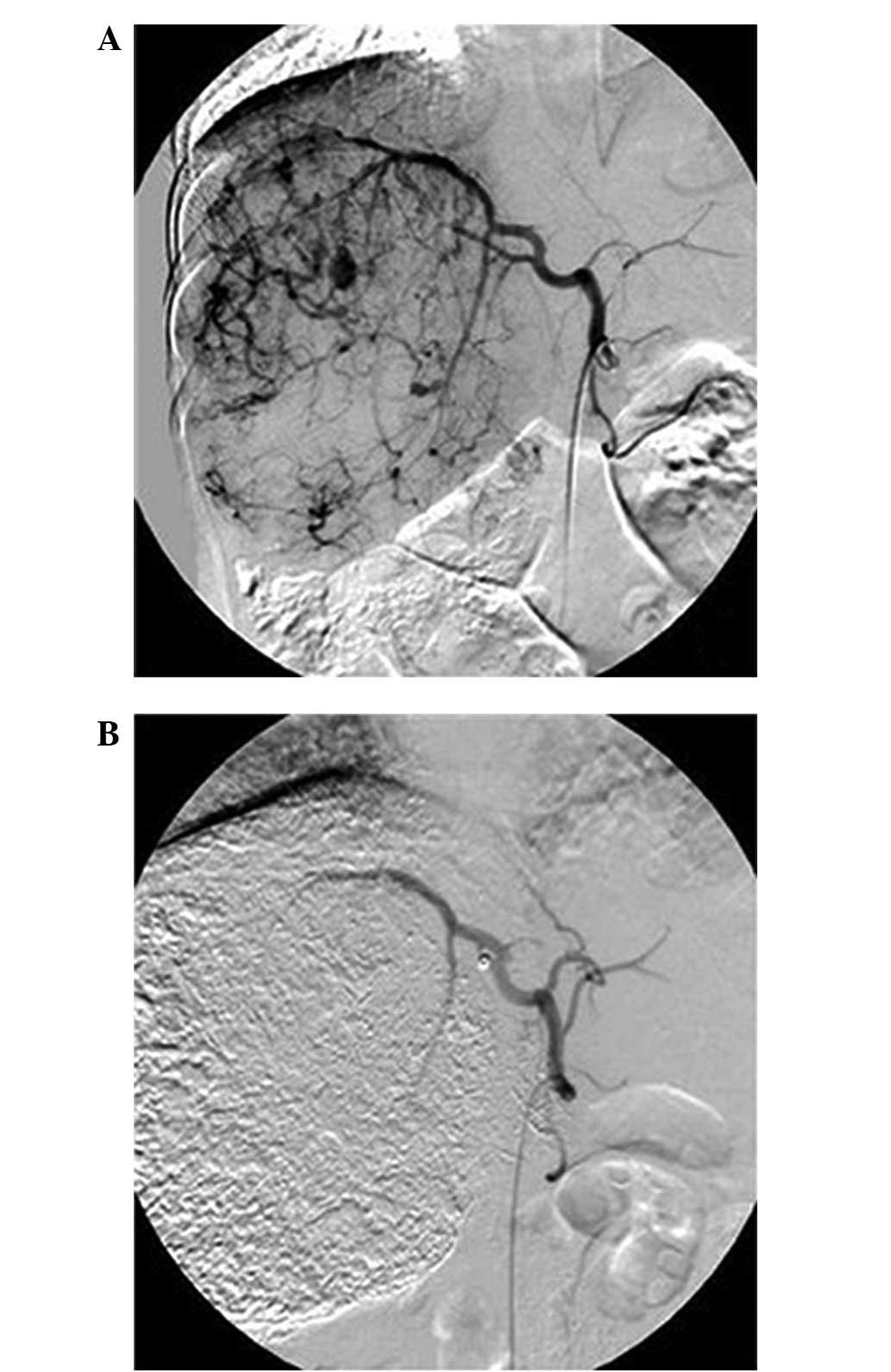
stagnated (Fig. 1). In order to
achieve a tumor status that was acceptable for surgery, the TACE
procedure was performed one to four times depending on the
patient’s response. The interval between the last TACE and surgery
was approximately six weeks.
Post-treatment assessment
Four to six weeks after TACE, the tumor volume, as
measured by abdominal CT, and the serum AFP level were regarded as
indicators for judging the treatment outcome. The tumor volume was
calculated by the following formula: Volume = 1/2 × (transverse
diameter)2 × length. The tumor response was classified
into four categories using the following criteria: Complete
response (CR), complete disappearance of all tumors and normal
level of AFP lasting more than four weeks; partial response (PR), a
decrease of ≥50% in tumor volume and a significant reduction in AFP
level, with no evidence of new lesions or progression in any
lesion; non-response (NR), a decrease of <50% in tumor volume,
with no evidence of new lesions; and progressive disease (PD), an
increase of the tumor size by >25% or appearance of a new
lesion.
To evaluate the toxicity of TACE, the alanine
aminotransferase (ALT), creatinine (Cr) and creatine kinase (CK)
levels of the 24 patients were tested prior to the first TACE and
subsequent to the last TACE to assess liver, renal and cardiac
function, respectively.
Statistical analysis
All data were analyzed using SPSS software (version
11, SPSS Inc., Chicago, USA). The differences between pre- and
post-TACE groups were evaluated by Student’s t-test. P<0.05 was
considered to indicate a statistically significant difference.
Results
Tumor response to TACE
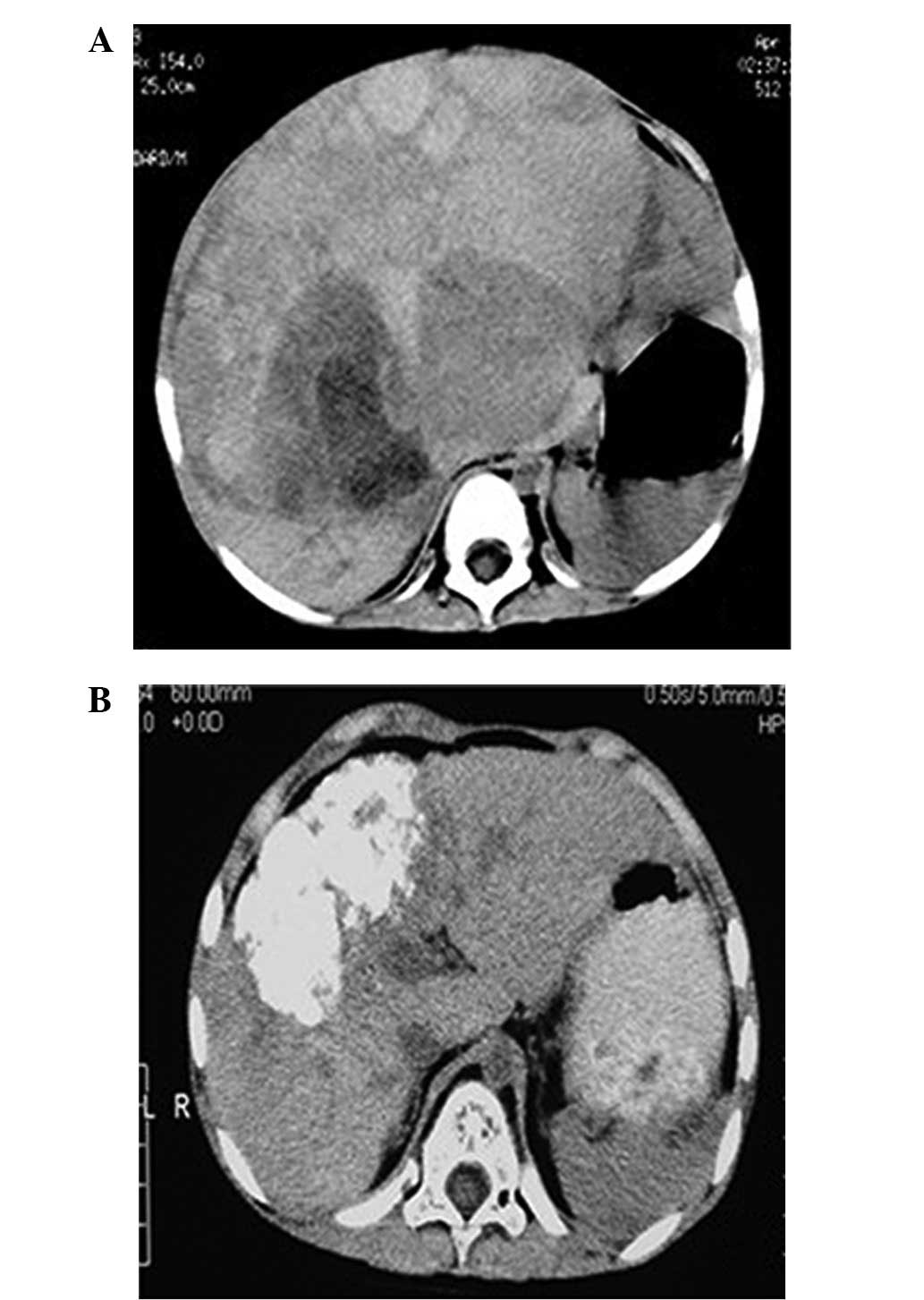
Following TACE, there was a visible reduction in
tumor size associated with a decrease in AFP levels (Table I). The responses of the tumors were
favorable, as shown by the non-contrast abdominal CT (Fig. 2). The tumor volumes decreased by
between 46.1 and 90.2%, with a mean value of 72%. The AFP levels
fell by between 63.8 and 99.9%, with a mean value of 95.7%.
According to the aforementioned criteria, there was a PR in 91.7%
cases (22/24), although there were NRs in 8.3% cases (2/24). A
total of 22 cases (PRs) underwent a subsequent safe complete
surgical resection and the remaining two patients (NRs) accepted a
partial resection. No delay to surgery was experienced.
 | Table ITherapeutic effects of TACE in 24
patients. |
Table I
Therapeutic effects of TACE in 24
patients.
| | | | | Tumor volume
(cm3) | AFP level
(ng/ml) | | |
|---|
| | | | |
|
| | |
|---|
| Case No. | Gender | Age | No. of TACEs | CCSG stage | Pre-TACE (first
time) | Post-TACE (last
time) | Pre-TACE (first
time) | Post-TACE (last
time) | Tumor shrinkage rate
(%) | AFP decrease (%) |
|---|
| 1 | M | 16 m | 4 | IIB | 264.96 | 66.83 | 132002.00 | 374 | 74.78 | 99.72 |
| 2 | M | 13 m | 3 | IIB | 685.45 | 160.62 | 484000.00 | 266.00 | 76.57 | 99.95 |
| 3 | F | 13 m | 2 | IIB | 486.41 | 109.65 | 385462.00 | 4768.00 | 77.46 | 98.76 |
| 4 | F | 14 m | 2 | IIB | 510.30 | 167.24 | 12568.00 | 9.55 | 67.23 | 99.92 |
| 5 | F | 13 m | 4 | IIIB | 331.61 | 32.42 | 363000.00 | 45.80 | 90.22 | 99.99 |
| 6 | F | 19 m | 1 | IIIA | 399.42 | 215.13 | 484000.00 | 175307.00 | 46.14 | 63.78 |
| 7 | M | 9 m | 3 | IIIA | 329.16 | 106.02 | 34892.00 | 1023.00 | 67.79 | 97.07 |
| 8 | M | 22 m | 2 | IIIB | 467.54 | 138.65 | 25066.00 | 262.00 | 70.34 | 98.95 |
| 9 | M | 13 m | 2 | IIA | 740.70 | 90.32 | 127278.00 | 61.40 | 87.81 | 99.95 |
| 10 | M | 3 m | 2 | IIA | 268.80 | 135.05 | 8920.00 | 453.00 | 49.76 | 94.92 |
| 11 | M | 1 m | 4 | IIB | 375.56 | 87.72 | 52320.00 | 52.50 | 76.64 | 99.90 |
| 12 | M | 9 m | 3 | IIIB | 305.92 | 78.46 | 24200.00 | 32.00 | 74.35 | 99.87 |
| 13 | M | 27 m | 2 | IIIB | 330.75 | 124.93 | 12934.00 | 243.00 | 62.23 | 98.17 |
| 14 | M | 16 m | 2 | IIIA | 700.05 | 206.54 | 4309.00 | 103.00 | 70.50 | 97.61 |
| 15 | F | 16 m | 2 | IIA | 304.70 | 55.27 | 7074.00 | 4.78 | 81.86 | 99.93 |
| 16 | M | 26 d | 3 | IIB | 205.22 | 72.86 | 10043.00 | 63.00 | 64.50 | 99.37 |
| 17 | F | 5 m | 2 | IIB | 301.83 | 88.51 | 8652.00 | 31.10 | 70.68 | 99.64 |
| 18 | F | 9 m | 3 | IIB | 575.09 | 89.21 | 20700.00 | 58.70 | 84.49 | 99.72 |
| 19 | M | 6 m | 2 | IIB | 451.64 | 130.70 | 425226.00 | 1100.00 | 71.06 | 99.74 |
| 20 | F | 41 m | 2 | IIIA | 518.64 | 175.73 | 294588.00 | 1203.00 | 66.12 | 99.59 |
| 21 | F | 37 m | 2 | IIIB | 1466.45 | 268.35 | 310000.00 | 91800.00 | 81.70 | 70.39 |
| 22 | F | 10 m | 1 | IIIA | 445.56 | 199.82 | 2390.00 | 340.00 | 55.15 | 85.77 |
| 23 | M | 5 m | 2 | IIB | 336.73 | 58.14 | 1004.00 | 46.00 | 82.73 | 95.42 |
| 24 | M | 16 m | 2 | IIB | 371.70 | 85.72 | 3658.00 | 15.00 | 76.94 | 99.59 |
| Mean | - | 13.91 m | - | - | 465.48 | 122.62 | 134761.92 | 11569.24 | 71.96 | 95.73 |
Adverse effects
Through statistical analysis, it was observed that
no significant differences existed between the pre- and post-TACE
groups with regard to Cr and CK. A significant difference was
observed in the ALT levels (P<0.05), but this was not clinically
significant as it was within the normal range (Table II). All patients presented with a
fever following TACE and their temperatures ranged from 37.5 to
40ºC for two to 16 days. Other clinical symptoms included nausea
and vomiting in 22 patients who recovered within three days
following suitable treatment. No marked chemotherapeutic
agent-induced toxicity was noted during TACE.
 | Table IIToxicity of TACE. |
Table II
Toxicity of TACE.
| Variable | Prior to first
TACE | Following last
TACE | P-value |
|---|
| ALT (U/l) | 26.33±17.24 | 33.04±13.71 | 0.029 |
| Cr (umol/l) | 22.71±5.03 | 21.75±4.42 | 0.694 |
| CK (U/l) | 116.70±31.57 | 120.39±26.08 | 0.472 |
Discussion
As previously mentioned, HB is one of the most
common malignant tumors in children worldwide. Complete tumor
resection is considered to be the most important treatment for the
long-term survival of children with HB. However, approximately half
of all children with HB have unresectable tumors at presentation
due to a huge tumor size or extensive infiltration. Pre-operative
systemic chemotherapy is vital for reducing tumor size and is able
to convert an unresectable tumor to a resectable one (14,15).
However, the systemic adverse effects may lead to a delayed surgery
and even chemotherapy-related mortality (6,8,9,16).
To reduce these disadvantages of systemic chemotherapy, an
alternative targeting therapy is required.
TACE has been demonstrated to be a valuable approach
and been used extensively in hepatocellular carcinoma (HCC) in
adults (17,18). However, the application of TACE in
children is limited as the setting and equipment must be adapted to
infants and children. The small vessel diameter of pediatric
vessels demands more experience and skill to avoid dissection or
perforation. Pediatric TACE has become feasible with the
development of the micro-catheter technique. In the present series,
all of the cases showed a significant response to TACE, with a
reduction in tumor size and a decrease in the AFP levels. The
majority of cases underwent subsequent safe complete surgical
resection and no delay to surgery was experienced. Angiography
provides a comprehensive understanding of the nutrient vessels of
the tumor, enabling subsequent intra-arterial injection with
chemotherapy drugs and embolic agents. TACE exposes the tumor cells
to high concentrations of drugs, which cannot be achieved by
systemic administration using the same dose. For the purpose of
chemoembolization, the ethiodized oil was conjugated with
pirarubicin and cisplatin. The selectively accumulated emulsion
releases the anticancer drugs slowly and decreases the transit time
of the drugs through the tumor vasculature, thus resulting in an
increased local concentration and tumor contact time for the drugs
(16,19–21).
As an effective embolic agent, PVA lead to the stenosis and
occlusion of the artery supply to the tumor and its subsequent
necrosis.
Although TACE is an outstanding method, there are
certain complications that require attention. In the present study,
a ‘post-embolization syndrome’ consisting of fever, abdominal pain,
nausea, vomiting and elevated AST, ALT and CRP levels occurred in
almost all patients, which may have been due to the massive
necrosis of the tumor, although these symptoms were minimal and
transient (6,22). More seriously, major complications
associated with the use of chemoembolic agents, including acute
liver failure, tumor rupture or pulmonary embolism, may also
occasionally occur (22,23). In the present series, no marked
chemotherapeutic agent-induced toxicity was observed during TACE.
Serious adverse effects, including liver dysfunction, renal
function failure, cardiac damage and myelosuppression, did not
occur. Thus TACE may be considered a safe pre-operative treatment
instead of systemic chemotherapy, particularly for patients without
distant metastasis.
In conclusion, pre-operative TACE may be considered
as an effective, feasible and safe treatment, instead of systemic
chemotherapy, for inducing the surgical resectability of
unresectable HB in pediatric patients during the surgical resection
waiting period. However, further experience and research into its
use is necessary prior to this approach becoming accepted as a
first-line therapy.
References
|
1
|
Schnater JM, Kohler SE, Lamers WH, et al:
Where do we stand with hepatoblastoma? A review. Cancer.
98:668–678. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Derek J and Perilongo G: Hepatoblastoma:
an oncological review. Pediatr Radiol. 36:183–186. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Czauderna P, Otte JB, Roebuck DJ, et al:
Surgical treatment of hepatoblastoma in children. Pediatr Radiol.
36:187–191. 2006. View Article : Google Scholar
|
|
4
|
Nakagawa N, Cornelius AS, Kao SCS, et al:
Transcatheter oily chemoembolization for unresectable malignant
liver tumors in children. J Vasc Intervent Radiol. 4:353–358. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Arcement CM, Towbin RB, Meza MP, et al:
Intrahepatic chemoembolization in unresectable pediatric liver
malignancies. Pediatr Radiol. 30:779–785. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Oue T, Fukuzawa M, Kusafuka T, et al:
Transcatheter arterial chemoembolization in the treatment of
hepatoblastoma. J Pediatr Surg. 33:1771–1775. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tashjian DB, Moriarty KP, Courtney RA, et
al: Preoperative chemoembolization for unresectable hepatoblastoma.
Pediatr Surg Int. 18:187–189. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Exelby PR, Filler RM and Grosfeld JL:
Liver tumors in children in the particular reference to
hepatoblastoma and hepatocellular carcinoma: American Academy of
Pediatrics surgical section survey - 1974. J Pediatr Surg.
10:329–337. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Evans AE, Land VJ, Newton WA, et al:
Combination chemotherapy (vincristine, adriamycin,
cyclophosphamide, and 5-fluorouracil) in the treatment of children
with malignant hepatoma. Cancer. 50:821–826. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Vogl TJ, Scheller A, Jakob U, et al:
Transarterial chemoembolization in the treatment of hepatoblastoma
in children. Eur Radiol. 16:1393–1396. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ohtsuka Y, Matsunaga T, Yoshida H, et al:
Optimal strategy of preoperative transcatheter arterial
chemoembolization for hepatoblastoma. Surg Today. 34:127–133. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li JP, Chu JP, Yang JY, et al:
Preoperative transcatheter selective arterial chemoembolization in
treatment of unresectable hepatoblastoma in infants and children.
Cardiovasc Intervent Radiol. 31:1117–1123. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ortega JA, Krailo MD, Haas JE, et al:
Effective treatment of unresectable or metastatic hepatoblastoma
with cisplatin and continuous infusion doxorubicin chemotherapy: a
report from the Children’s Cancer Study Group. J Clin Oncol.
9:2167–2176. 1991.
|
|
14
|
Seo T, Ando H, Watanabe Y, et al:
Treatment of hepatoblastoma: less extensive hepatectomy after
effective preoperative chemotherapy with cisplatin and adriamycin.
Surgery. 123:407–414. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ehrlich PF, Greenberg ML and Filler RM:
Improved long-term survival with preoperative chemotherapy for
hepatoblastoma. J Pediatr Surg. 32:999–1003. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ogita S, Tokiwa K, Taniguchi H and
Takahashi T: Intraarterial chemotherapy with lipid contrast medium
for hepatic malignancies in infants. Cancer. 60:2886–2890. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Groupe d’Etude et de Traitement du
Carcinome Hépatocellulaire. A comparison of lipiodol
chemoembolization and conservative treatment for unresectable
hepatocellular carcinoma. N Engl J Med. 332:1256–1261. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Llovet JM, Real MI, Montaña X, et al:
Arterial embolization or chemoembolisation versus symptomatic
treatment in patients with unresectable hepatocellular carcinoma: a
randomized controlled trial. Lancet. 359:1734–1739. 2002.
View Article : Google Scholar
|
|
19
|
Iwai K, Maeda H and Konno T: Use of oily
contrast medium for selective drug targeting to tumor: enhanced
therapeutic effect and X-ray image. Cancer Res. 44:2115–2121.
1984.PubMed/NCBI
|
|
20
|
Nakamura H, Hashimoto T, Oi H and Sawada
S: Transcatheter oily chemoembolization of hepatocellular
carcinoma. Radiology. 170:783–786. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sasaki Y, Imaoka S, Kasugai H, et al: A
new approach to chemoembolization therapy for hepatoma using
ethiodized oil, cisplatin, and gelatin sponge. Cancer.
60:1194–1203. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sakamoto I, Aso N, Nagaoki K, et al:
Complications associated with transcatheter arterial embolization
for hepatic tumors. Radiographics. 18:605–619. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chung JW, Park JH, Im JG, et al: Pulmonary
oil embolism after transcatheter oily chemoembolization of
hepatocellular carcinoma. Radiology. 187:689–693. 1993. View Article : Google Scholar : PubMed/NCBI
|