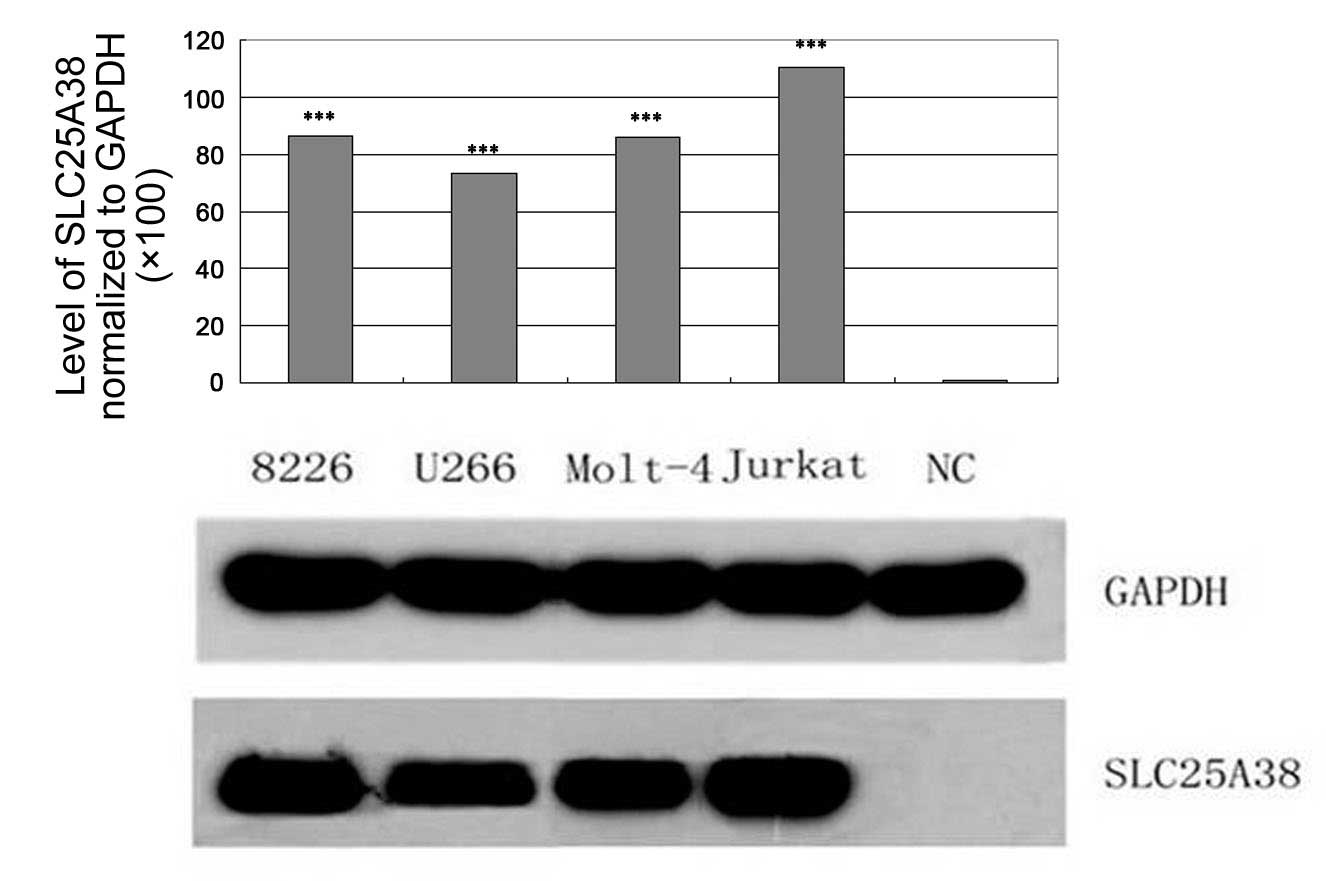
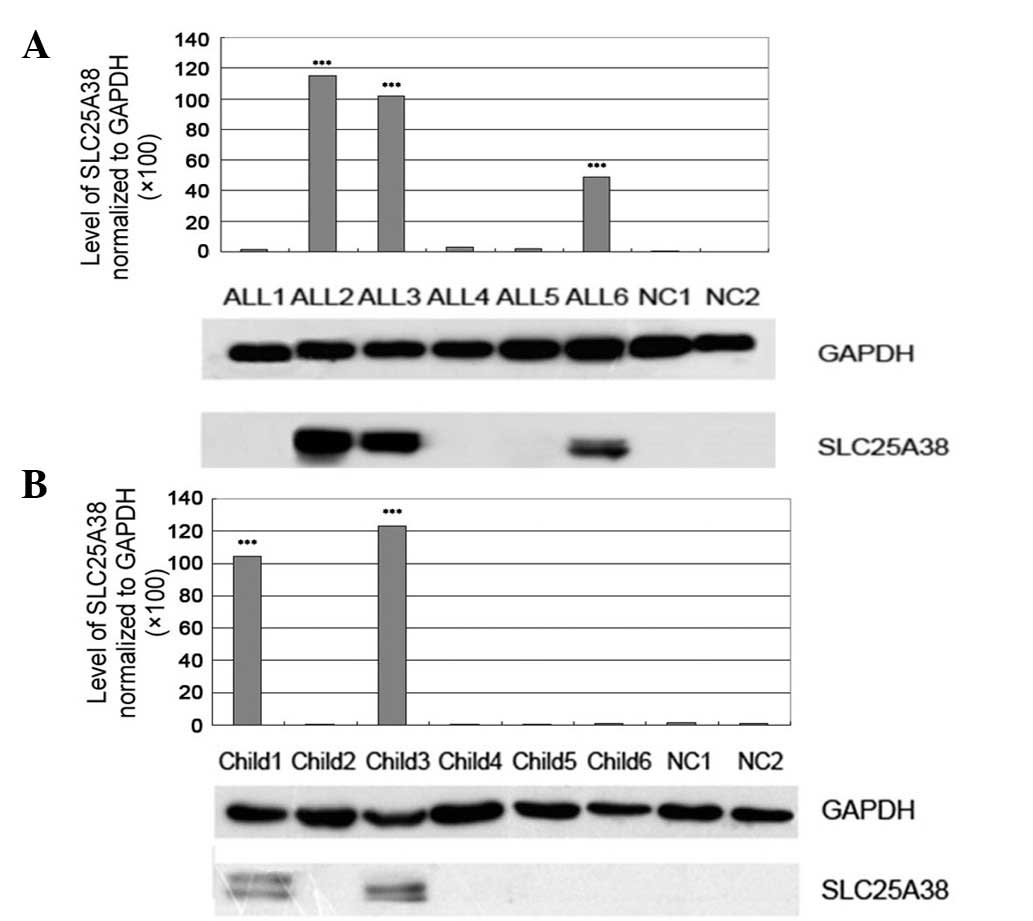
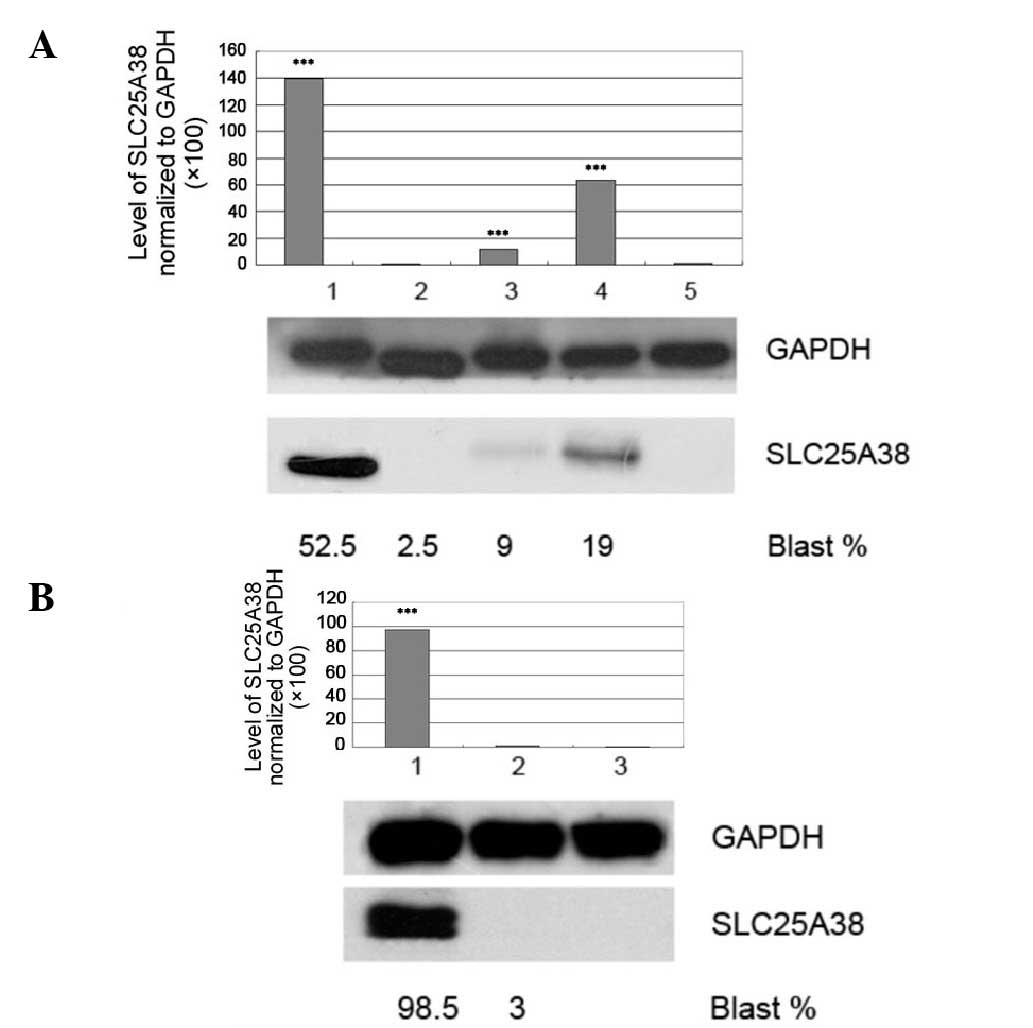
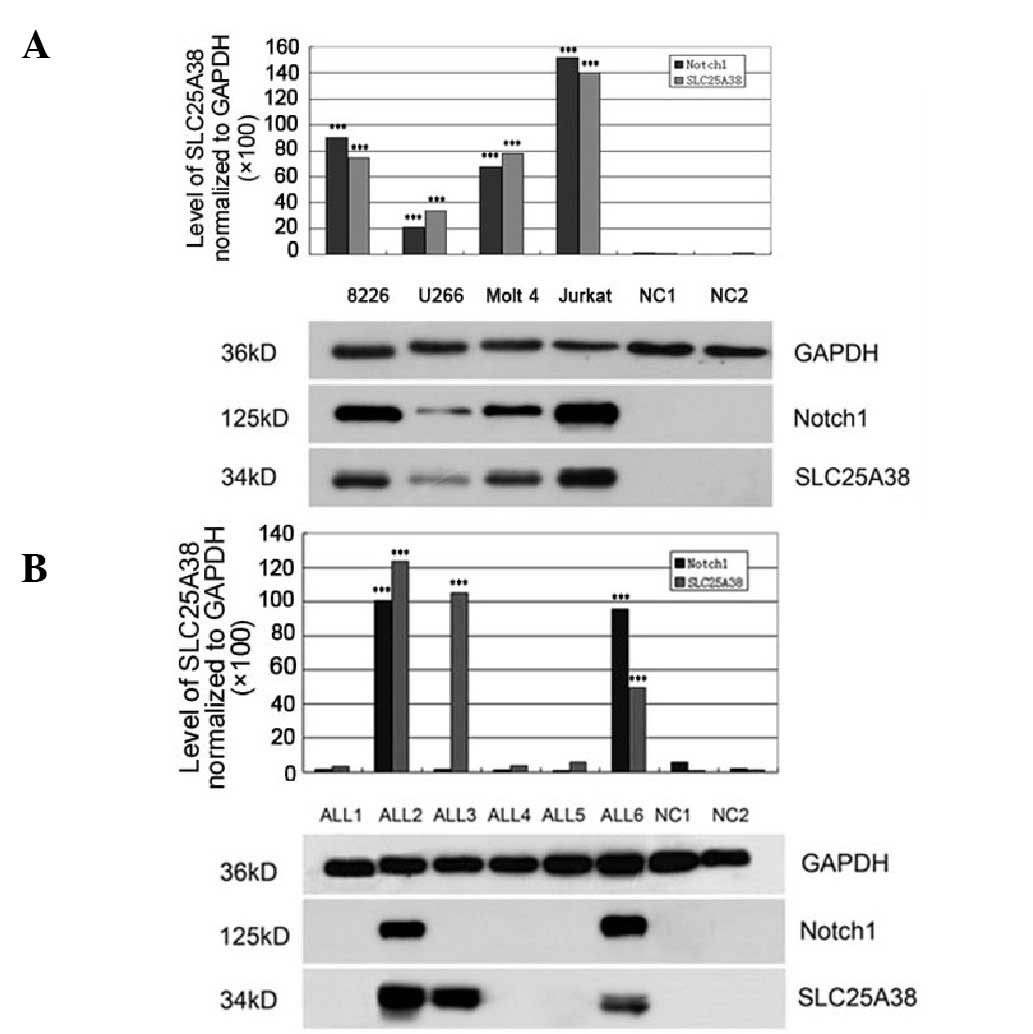
|
1
|
Barrett AJ, Horowitz MM, Pollock BH, et
al: Bone marrow transplants from HLA-identical siblings as compared
with chemotherapy for children with acute lymphoblastic leukemia in
a second remission. N Engl J Med. 331:1253–1258. 1994. View Article : Google Scholar
|
|
2
|
Schroeder H, Gustafsson G,
Saarinen-Pihkala UM, et al: Allogeneic bone marrow transplantation
in second remission of childhood acute lymphoblastic leukemia: a
population-based case control study from the Nordic countries. Bone
Marrow Transpl. 23:555–560. 1999. View Article : Google Scholar
|
|
3
|
McNeil DE, Coté TR, Clegg L and Mauer A:
SEER update of incidence and trends in pediatric malignancies:
acute lymphoblastic leukemia. Med Pediatr Oncol. 39:554–557. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bassan R: Evolving strategies for the
management of high-risk adult acute lymphoblastic leukemia.
Haematologica. 90:12992005.PubMed/NCBI
|
|
5
|
Pui CH and Evans WE: Treatment of actue
lymphoblastic leukemia. N Engl J Med. 354:166–178. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pui CH, Robison LL and Look AT: Acute
lymphoblastic leukemia. Lancet. 371:1030–1043. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pullarkat V, Slovak ML, Kopecky KJ, et al:
Impact of cytogenetics on the outcome of adult actue lymphoblastic
leukemia: results of Southwest Oncology Group 9400 study. Blood.
111:2563–2572. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Marks DI, Paietta EM, Moorman AV, et al:
T-cell acute lymphoblastic leukemia in adults: clinical features,
immunophenotype, cytpgenetics, and outcome from the large
randomized prospective trial (UKALLXII/ECOG 2993). Blood.
114:5136–5145. 2009. View Article : Google Scholar
|
|
9
|
Pui CH, Pei D, Sandlund JT, et al:
Long-term results of St Jude Total Therapy Studies 11, 12, 13A,
13B, and 14 for childhood acute lymphoblastic leukemia. Leukemia.
24:371–382. 2009.
|
|
10
|
Hediger MA, Romero MF, Peng JB, et al: The
ABCs of solute carriers: physiological, pathological and
therapeutic implications of human membrane transport proteins.
Pflugers Arch. 447:465–468. 2004. View Article : Google Scholar
|
|
11
|
Palmieri F: The mitochondrial transporter
family (SLC25): physiological and pathological implications.
Pflugers Arch. 447:689–709. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pebay-Peyroula E, Dahout-Gonzalez C, Kahn
R, et al: Structure of mitochondrial ADP/ATP carrier in complex
with carboxyatractyloside. Nature. 426:39–44. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Haitina T, Lindblom J, Renström T and
Fredriksson R: Fourteen novel human members of mitochondrial solute
carrier family 25 (SLC25) widely expressed in the central nervous
system. Genomics. 88:779–790. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guernsey DL, Jiang H, Campagna DR, et al:
Mutations in mitochondrial carrier family gene SLC25A38 cause
nonsyndromic autosomal recessive congenital sideroblastic anemia.
Nat Genet. 41:651–653. 2009. View
Article : Google Scholar
|
|
15
|
Bergmann AK, Campagna DR, Mcloughlin EM,
et al: Systematic molecular genetic analysis of congenital
sideroblastic anemia: evidence for genetic heterogeneity and
identification of novel mutations. Pediatr Blood Cancer.
54:273–278. 2010.
|
|
16
|
Kannengiesser C, Sanchez M, Sweeney M, et
al: Missense SLC25A38 variations play an important role in
autosomal recessive inherited sideroblastic anemia. Haematoligica.
96:33242011.
|
|
17
|
Atamna H: Heme, iron, and the
mitochondrial decay of ageing. Ageing Res Rev. 3:303–318. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ryter SW and Tyrrell RM: The heme
synthesis and degradation pathways: role in oxidant sensitivity.
Heme oxygenase has both pro- and antioxidant properties. Free
Radical Biol Med. 28:289–309. 2000. View Article : Google Scholar
|
|
19
|
Leong KG and Karsan A: Recent insights
into the role of Notch signaling in tumorigenesis. Blood.
107:2223–2233. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Aster JC, Blacklow SC and Pear WS: Notch
signalling in T-cell lymphoblastic leukaemia/lymphoma and other
haematological malignancies. J Pathol. 223:262–273. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kanamori E, Itoh M, Tojo N, et al: Flow
cytometric analysis of Notch1 and Jagged1 expression in normal
blood cells and leukemia cells. Exp Ther Med. 4:397–400.
2012.PubMed/NCBI
|
|
22
|
Mansour MR, Sulis ML, Duke V, et al:
Prognostic implications of NOTCH1 and FBXW7 mutations in adults
with T-cell acute lymphoblastic leukemia treated on the MRC
UKALLXII/ECOG E2993 protocol. J Clin Oncol. 27:4352–4356. 2009.
View Article : Google Scholar
|
|
23
|
Zhang H, Zhang YW, Chen Y, et al:
Appoptosin is a novel pro-apoptotic protein and mediates cell death
in neurodegeneration. J Neurosci. 32:15565–15576. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bennett JM, Catovsky D, Daniel MT, et al:
Proposals for the classification of the acute leukaemias.
French-American-British (FAB) co-operative group. Br J Haematol.
33:451–458. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Vardiman JW, Thiele J, Arber DA, et al:
The 2008 revision of the World Health Organization (WHO)
classification of myeloid neoplasms and acute leukemia: rationale
and important changes. Blood. 114:937–951. 2009. View Article : Google Scholar
|
|
26
|
Ferrando AA and Look AT: Clinical
implications of recurring chromosomal and associated molecular
abnormalities in acute lymphoblastic leukemia. Semin Hematol.
37:381–395. 2000. View Article : Google Scholar
|
|
27
|
Ferrando AA, Neuberg DS, Staunton J, et
al: Gene expression signatures define novel oncogenic pathways in T
cell acute lymphoblastic leukemia. Cancer Cell. 1:75–87. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Soulier J, Clappier E, Cayuela JM, et al:
HOXA genes are included in genetic and biologic networks defining
human acute T-cell leukemia (T-ALL). Blood. 106:274–286. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ferrando AA, Armstrong SA, Neuberg DS, et
al: Gene expression signatures in MLL-rearranged T-lineage and
B-precursor acute leukemias: dominance of HOX dysregulation. Blood.
102:262–268. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Aifantis I, Raetz E and Buonamici S:
Molecular pathogenesis of T-cell leukaemia and lymphoma. Nat Rev
Immunol. 8:380–390. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Budhu A, Ji J and Wang XW: The clinical
potential of microRNAs. J Hematol Oncol. 3:372010. View Article : Google Scholar
|
|
32
|
Fernando TR, Rodriguez-Malave NI and Rao
DS: MicroRNAs in B cell development and malignancy. J Hematol
Oncol. 5:72012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Whitehead KA, Langer R and Anderson DG:
Knocking down barriers: advances in siRNA delivery. Nat Rev Drug
Discov. 8:129–138. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yin L, Velazquez OC and Liu ZJ: Notch
signaling: emerging molecular targets for cancer therapy. Biochem
Pharmacol. 80:690–701. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Breit S, Stanulla M, Flohr T, et al:
Activating NOTCH1 mutations predict favorable early treatment
response and long-term outcome in childhood precursor T-cell
lymphoblastic leukemia. Blood. 108:1151–1157. 2006. View Article : Google Scholar
|
|
36
|
Weng AP, Ferrando AA, Lee W, et al:
Activating mutations of NOTCH1 in human T cell acute lymphoblastic
leukemia. Science. 306:269–271. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Aster JC, Pear WS and Blacklow SC: Notch
signaling in leukemia. Annu Rev Pathol. 3:587–613. 2008. View Article : Google Scholar
|
|
38
|
Zou J, Li P, Lu F, et al: Notch1 is
required for hypoxia-induced proliferation, invasion and
chemoresistance of T-cell acute lymphoblastic leukemia cells. J
Hematol Oncol. 6:32013. View Article : Google Scholar : PubMed/NCBI
|