Introduction
Synovial sarcoma is a malignant tumor that accounts
for 10% of all soft-tissue tumors (1). In total, <3 cases per million
individuals are diagnosed each year. The disease can occur at any
age, but is more common in teenagers and young adults. Despite its
name, synovial sarcoma does not develop in the synovial tissues of
the joints. The disease mostly presents in the extremities,
particularly the periarticular regions. However, synovial sarcoma
is also found in the head and neck, mediastinum, heart, esophagus,
lungs, abdominal wall, mesentery, forearms, thighs, and muscle and
fascia of the waist and back (1–5). In 1996,
Gaertner et al reported the first case of synovial sarcoma
in the literature (3). The diagnosis
of synovial sarcoma is difficult due to its rarity and the fact
that its clinical and histological characteristics are similar to
other spindle cell pleural tumors. Histopathology and/or
immunohistochemistry investigations are essential to diagnose
synovial sarcoma; the diagnosis in the present study was based on
immunohistochemistry, but cytogenetics can be used to detect
chromosomal translocations specific to synovial sarcoma, which aids
in the confirmation of the diagnosis (2). Cytogenetic studies are useful for
differential diagnosis. Chromosomal translocation (chromosome X or
18) is observed in >90% of synovial sarcomas and may be detected
by the FISH method (fluorescence in-situ hybridization) w5x
(2). In spite of its high
sensitivity, the FISH method is not obligatory for the diagnosis,
and it is indicated in cases of histological and
inmunohistochemical overlapping.
The main poor prognostic factors are complete
resection, a tumor size >5 cm, the male gender, a high-grade
tumor and an age of >20 years. The patient in the present study
did not undergo a complete resection due to the presence of a giant
tumor. These factors are likely reasons for the disease progressing
in a relatively short period of time.
Case report
A 49-year-old male was admitted on October 5 2010,
to the Harbin Thoracic Hospital due to left-sided chest pain that
had persisted for 1 month. Computed tomography (CT) chest scans
showed bilateral pleural effusion. Between October 2010 and August
2011, the patient was treated with anti-inflammatory therapy 7
times for a duration of 2 weeks (penicillin, 4.5 mg, twice a day
intravenous drops; quinolines, 100 ml, once per day intravenous
drops) and underwent pleural drainage surgery, which caused a
subsequent partial remission. The patient underwent an evacuation
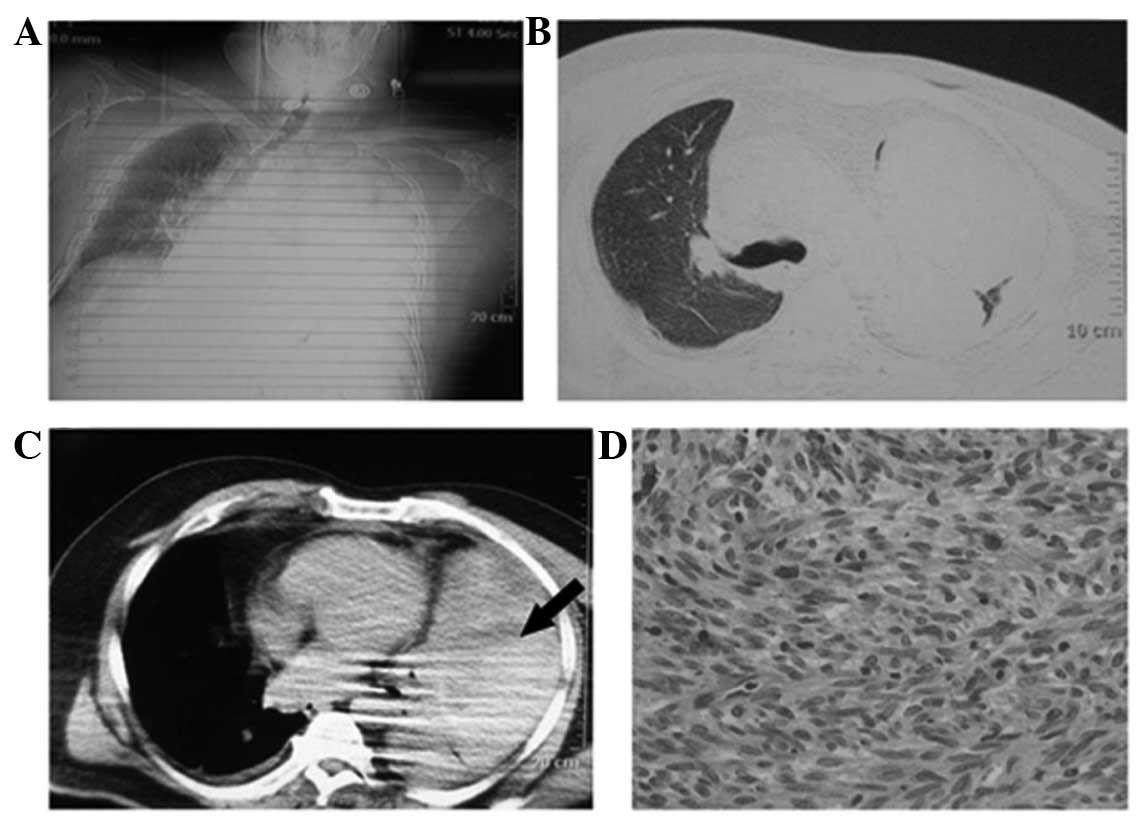
of a hematoma on April 28, 2011, as CT showed a huge low-density
mass in the left pleural space (Fig.
1A–C). The histopathological specimen showed (left pleural)
fibrous wall tissue, with hemorrhage, necrosis, collagen
degeneration and a diffuse spindle cell proliferative zone.
Following morphological and immunohistochemical analysis, the
patient was diagnosed with synovial sarcoma. The tumor cells
exhibited immunohistochemical positivity for vimentin, epithelial
membrane antigen and calponin, but negativity for S-100 and cluster
of differentiation 34 (Fig. 1D). The
patient's pain was relieved after the surgery and the follow-up
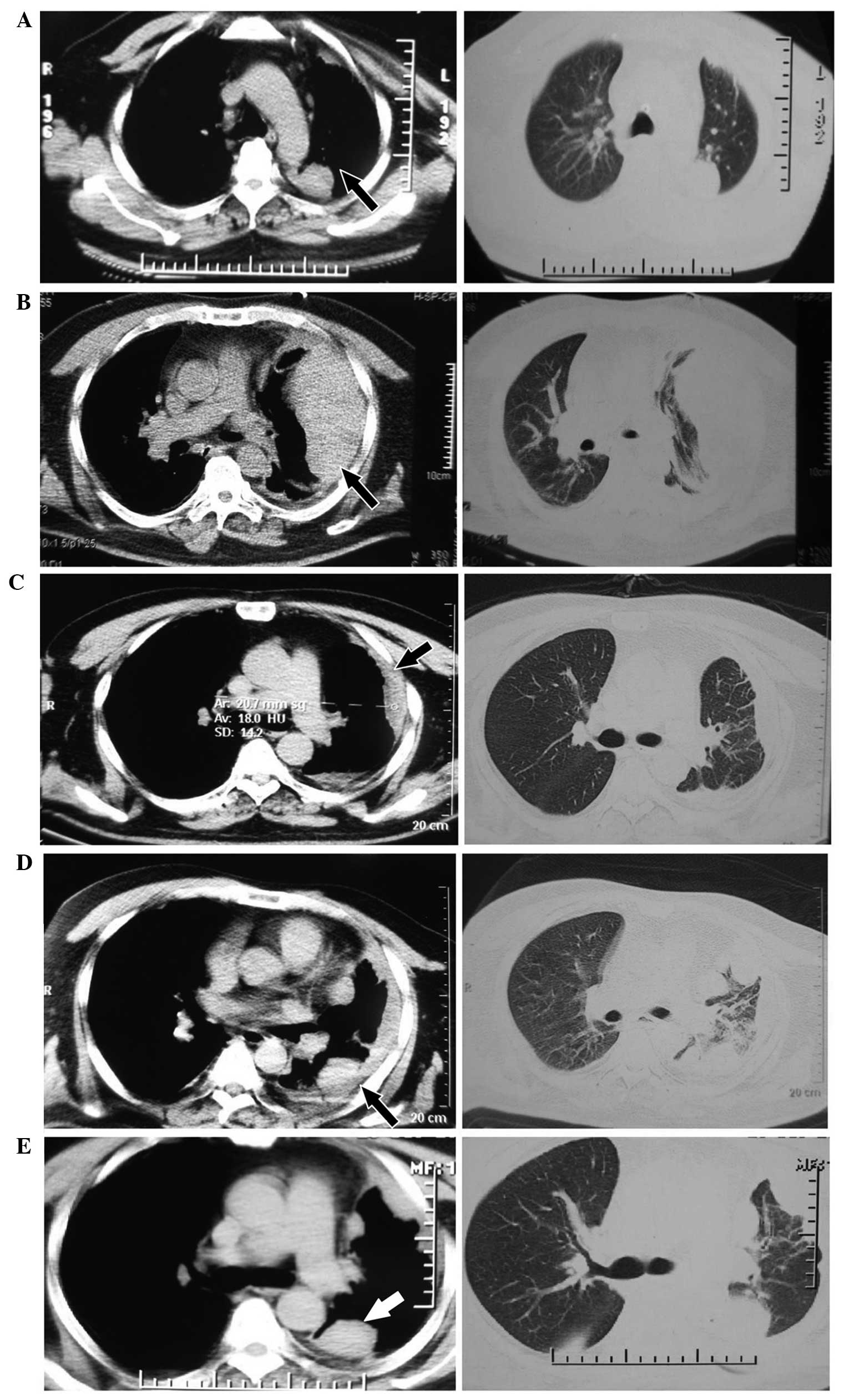
chest CT showed that the mass was significantly reduced in size
(Fig. 2A). The patient went home
after the surgery on May 17, 2011, without further treatment.
After 2 months, a follow-up chest CT scan showed
local recurrence (Fig. 2B). Between
July 15, 2011, and September 23, 2011, the patient received four
cycles of chemotherapy consisting of 7.5 mg/m2 Endostar
intravenous guttae (ivgtt) on days 1–14 and 1.0 g/m2
ifosfamide ivgtt on days 1–6. During the treatment, the imaging
evaluation was of a partial response (Fig. 2C). The main adverse effects of the
treatment were grade IV nausea, vomiting and dizziness.
Between November 14, 2011, and February 2, 2012, due
to chest pain and disease progression on chest CT (Fig. 2D), the patient was administered 3
cycles of chemotherapy (250 mg/m2 pemetrexed ivgtt on
days 1–8 and 7.5 mg/m2 Endostar ivgtt on days 1–14). The
subsequent therapeutic evaluation was of stable disease (Fig. 2E). On February 2, 2012, the patient
began to receive sunitinib (375 mg/m2) as maintenance
therapy. On July 23, 2013, the patient succumbed to respiratory and
circulatory failure due to tumor progression.
Discussion
In the present study, sunitinib was administered to
the patient following combined treatment with chemotherapy and
Endostar. To the best of our knowledge, this is the first reported
use of sunitinib for maintenance therapy in pleural synovial
sarcoma and it achieved a good curative effect. An imaging
examination showed that the tumor size was significantly reduced
and the patient's survival time was 25 months when the tumor
recurred after surgery. The main adverse effects during treatment
were hypertension and neutropenia.
Although the treatment was effective, the patient's
quality of life was poor. From March 2012 onwards, the patient was
unable to function independently due to chest pain. The sarcoma had
a brittle texture, and as a result any careless action may have
lead to tumor rupture and severe pain. The experience of bleeding
greatly affected the psychology of the patient, who became afraid
and depressed due to the pain and had to use a Fentanyl transdermal
system for a prolonged period of time, with a maximum dose of 134.4
mg. In April 2013, the patient accepted a nerve block and nerve
ablation, but the procedures did not produce the desired
therapeutic effect.
The incidence of pleural synovial sarcoma has been
shown to be increasing and the most common therapy is a surgical
resection (6,7), followed by chemotherapy. In general,
synovial sarcomas are chemosensitive to ifosfamide and doxorubicin,
but there is no established gold standard for the treatment of
pleural synovial sarcoma. The rarity of synovial sarcoma means that
there is no consensus on the role of chemotherapy in preventing
metastases and improving survival.
However, antiangiogenic therapy in general shows
promise for the treatment of malignancy, tumor invasion and
metastasis of a number of different types of cancer. O'Reilly et
al first identified endostatin in hemangioendothelioma cells as
an antiangiogenic molecule (8). The
targets of endostatin are the newly formed capillary endothelial
cells, which are intimately associated with tumors (9). Endostar (YH-16) is a novel recombinant
human endostatin, which has a broad spectrum of antiangiogenic
activity; its mechanism of action is to stimulate the migration of
endothelial cells by inhibiting the formation of blood vessels and
tumor neovascularization, thus blocking the nutrient supply of the
tumor, inhibiting tumor proliferation and metastasis. Endostar also
exhibits anti-xenograft activity against human tumor cells (SPC2A4
lung cancer, SGC7901 gastric cancer, HeLa cervical cancer, and
SMMC27721 and Bel7402 hepatoma cells). Endostar has also shown
antitumor activity in vitro in a murine tumor model of
broad-spectrum cancers (such as S180 sarcoma and H22 hepatocellular
carcinoma cells).
Sunitinib is an orally active tyrosine kinase
inhibitor (TKI) that affects various signaling cascades involved in
antiangiogenesis and tumor proliferation, including vascular
endothelial growth factors and platelet-derived growth factors. The
drug is used in the first-line therapy of clear cell renal
carcinoma and imatinib-resistant gastrointestinal stromal tumors,
and is also used to treat numerous other types of solid tumors,
including breast, lung, prostatic and colorectal cancer (10–13). For
example, the efficacy of the TKIs imatinib (Gleevec) and sunitinib
(Sutent) in patients with advanced gastrointestinal stromal tumors
(GISTs) has been demonstrated by observations in the clinic, a
disease for which there were previously no highly effective
treatment options for metastatic disease. There is a paucity of
data in the literature regarding the role of sunitinib in synovial
sarcoma, therefore the present study investigated its efficacy in
synovial sarcoma. One previous report was located in the literature
about a patient with synovial sarcoma, who also had a non-GIST and
stable disease following 16 weeks of therapy with sunitinib
(14).
In conclusion, it is worth considering using
Endostar and targeted therapy following chemotherapy to optimize
the treatment of pleural synovial sarcoma. Sunitinib can be used in
pleural synovial sarcomas as maintenance therapy. The optimal dose
and treatment time with Endostar and sunitinib remains to be
established in clinical trials with more patients. Sunitinib is a
useful practical adjuvant for the management of pleural synovial
sarcomas.
Acknowledgements
This study is supported by the fund of the Natural
Science Foundation of Heilongjiang (grant no. D201223).
References
|
1
|
Mankin HJ and Hornicek FJ: Diagnosis,
classification, and management of soft tissue sarcomas. Cancer
Control. 12:5–21. 2005.PubMed/NCBI
|
|
2
|
Aubry MC, Bridge JA, Wickert R and
Tazelaar HD: Primary monophasic synovial sarcoma of the pleura:
Five cases confirmed by the presence of SYT-SSX fusion transcript.
Am J Surg Pathol. 25:776–781. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gaertner E, Zeren EH, Fleming MV, Colby TV
and Travis WD: Biphasic synovial sarcomas arising in the pleural
cavity. A clinicopathologic study of five cases. Am J Surg Pathol.
20:36–45. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jawahar DA, Vuletin JC, Gorecki P,
Persechino F, Macera M and Magazeh P: Primary biphasic synovial
sarcoma of the pleura. Respir Med. 91:568–570. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nicholson AG, Goldstraw P and Fisher C:
Synovial sarcoma of the pleura and its differentiation from other
primary pleural tumours: A clinicopathological and
immunohistochemical review of three cases. Histopathology.
33:508–513. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Loscertales J, Triviño A, Gallardo G and
Congregado M: Primary monophasic synovial sarcoma of the pleura:
Diagnosis and treatment. Interact Cardiovasc Thorac Surg.
12:885–887. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nishiyama N, Iwata T, Izumi N, Tsukioka T,
Nagano K and Suehiro S: Aggressive repeat surgery for a recurrent
synovial sarcoma in the pleura. Ann Thorac Cardiovasc Surg.
15:46–49. 2009.PubMed/NCBI
|
|
8
|
O'Reilly MS, Boehm T, Shing Y, et al:
Endostatin: an endogenous inhibitor of angiogenesis and tumor
growth. Cell. 88:277–285. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhuo W, Chen Y, Song X and Luo Y:
Endostatin specifically targets both tumor blood vessels and
lymphatic vessels. Front Med. 5:336–340. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cardoso F, Canon JL, Amadori D, et al: An
exploratory study of sunitinib in combination with docetaxel and
trastuzumab as first-line therapy for HER2-positive metastatic
breast cancer. Breast. 21:716–723. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Corn PG, Song DY, Heath E, et al:
Sunitinib plus androgen deprivation and radiation therapy for
patients with localized high-risk prostate cancer: results from a
multi-institutional phase 1 study. Int J Radiat Oncol Biol Phys.
86:540–545. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Modest DP, Camaj P, Heinemann V, et al:
KRAS allel-specific activity of sunitinib in an isogenic disease
model of colorectal cancer. J Cancer Res Clin Oncol. 139:953–961.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Waqar SN, Gopalan PK, Williams K,
Devarakonda S and Govindan R: A phase I trial of sunitinib and
rapamycin in patients with advanced non-small cell lung cancer.
Chemotherapy. 59:8–13. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
George S, Merriam P, Maki RG, et al:
Multicenter phase II trial of sunitinib in the treatment of
nongastrointestinal stromal tumor sarcomas. J Clin Oncol.
27:3154–3160. 2009. View Article : Google Scholar : PubMed/NCBI
|