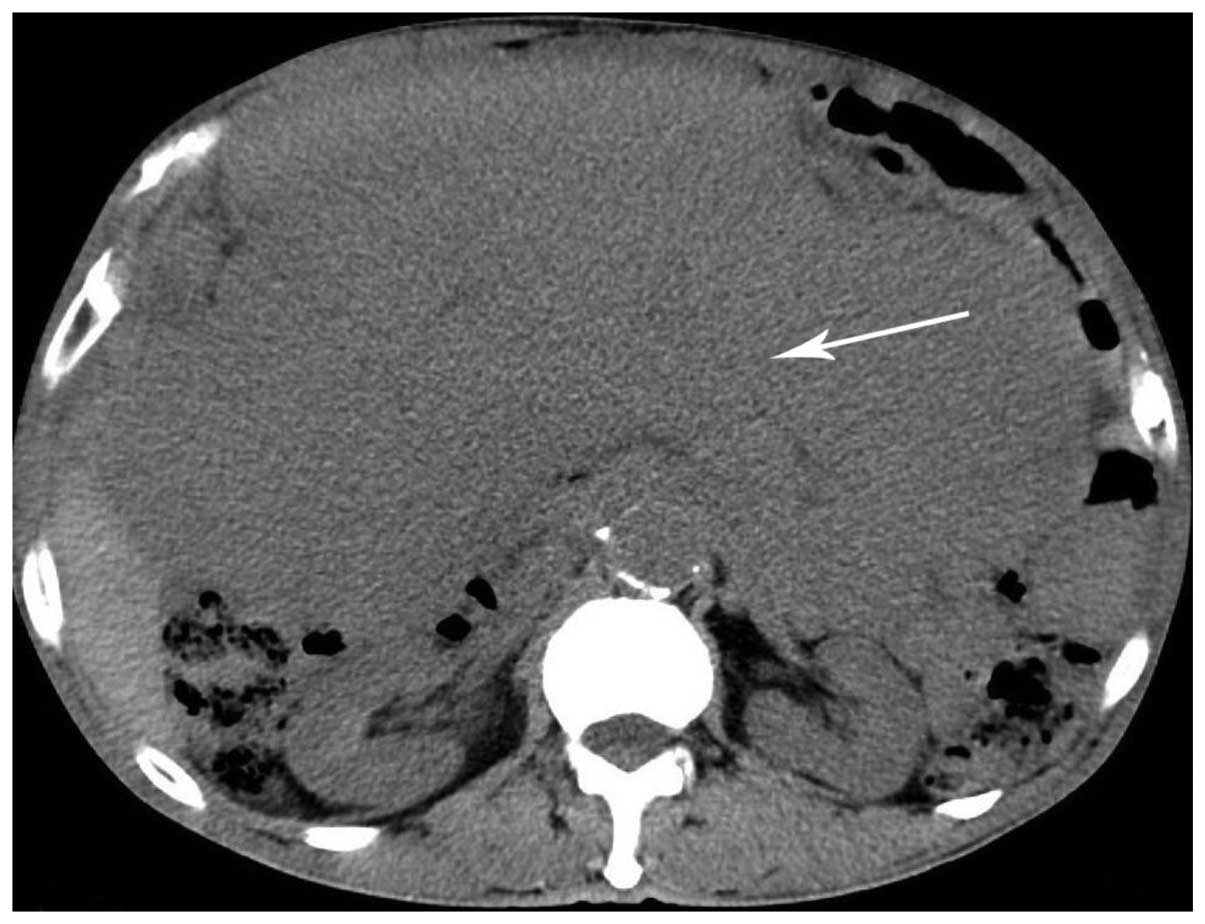
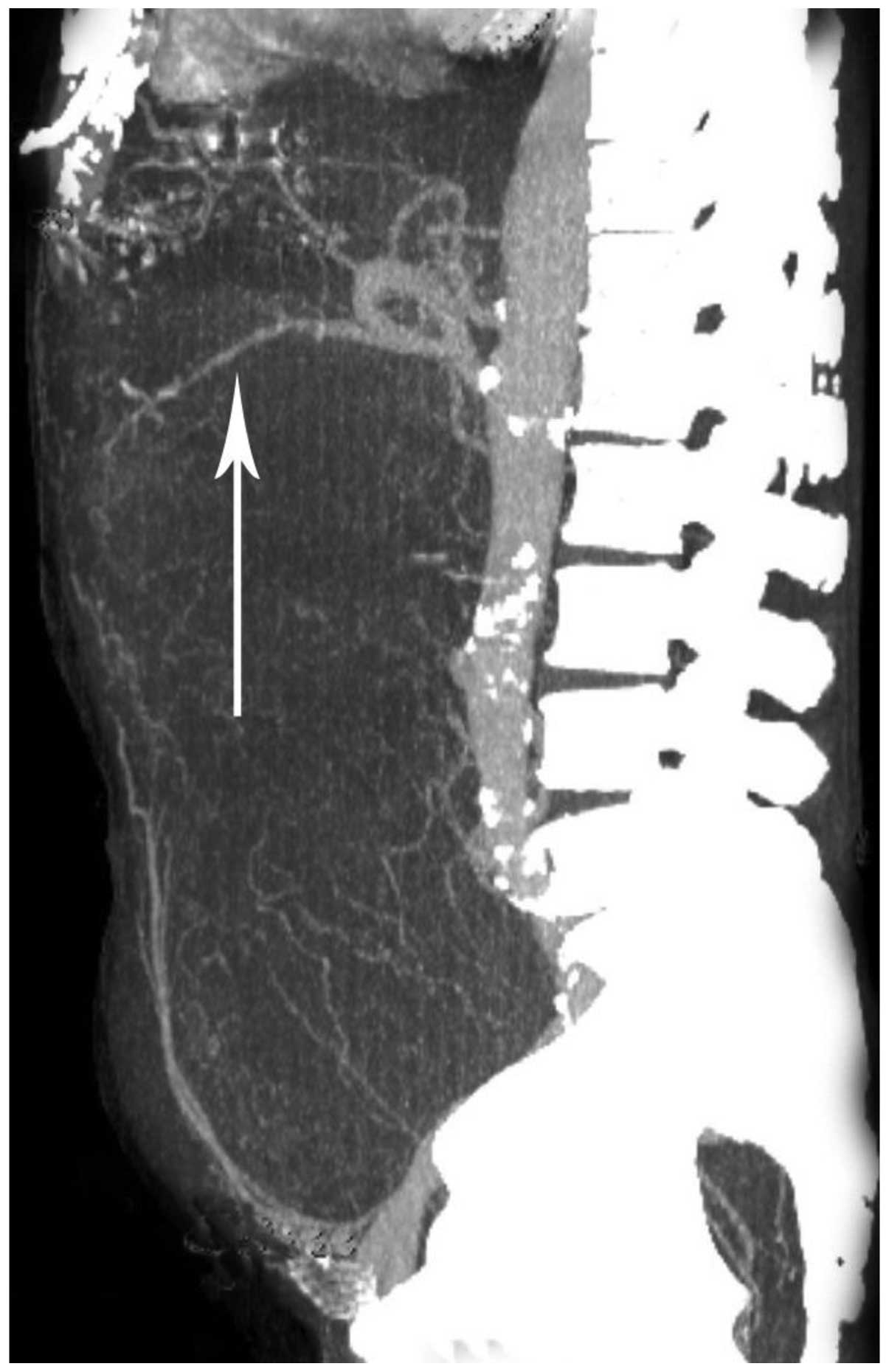
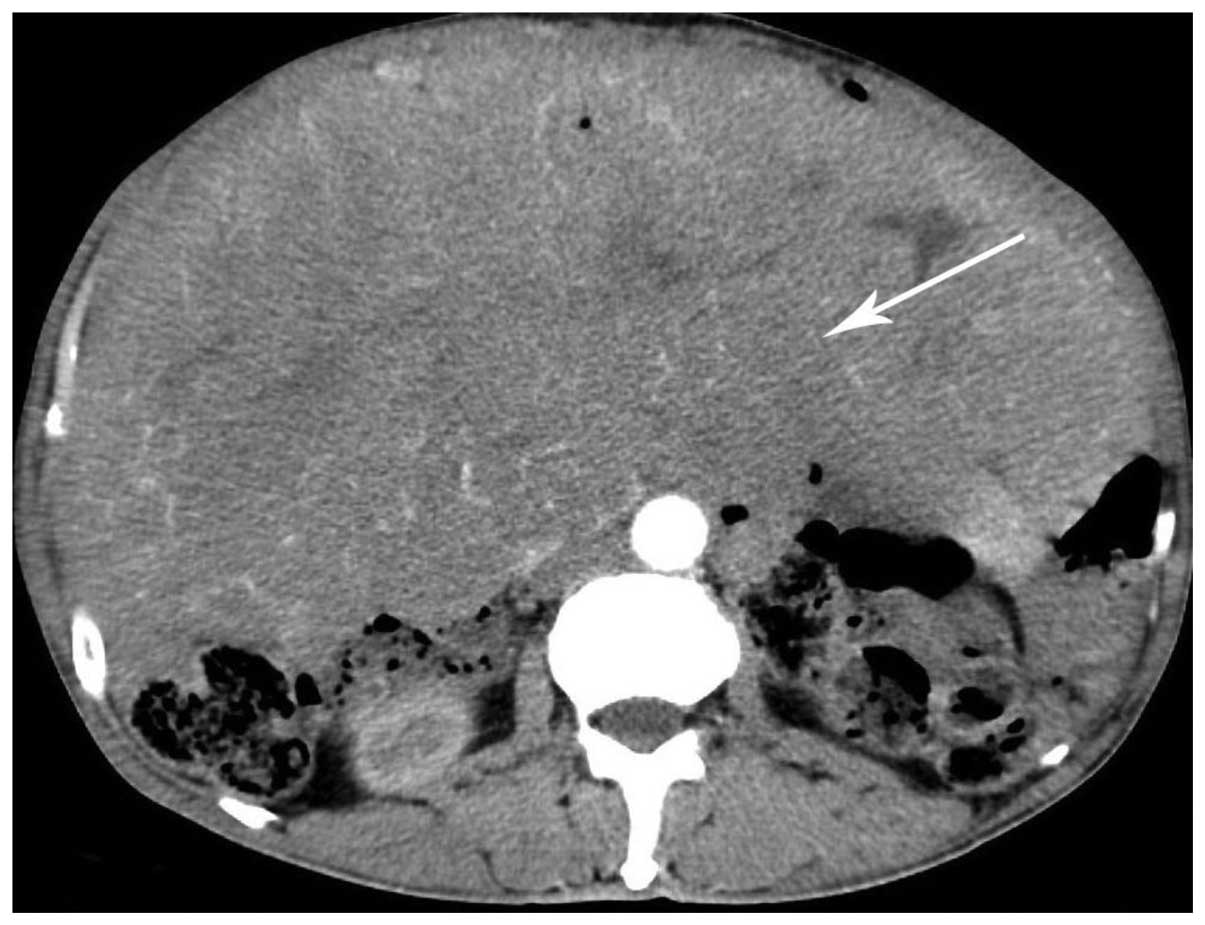
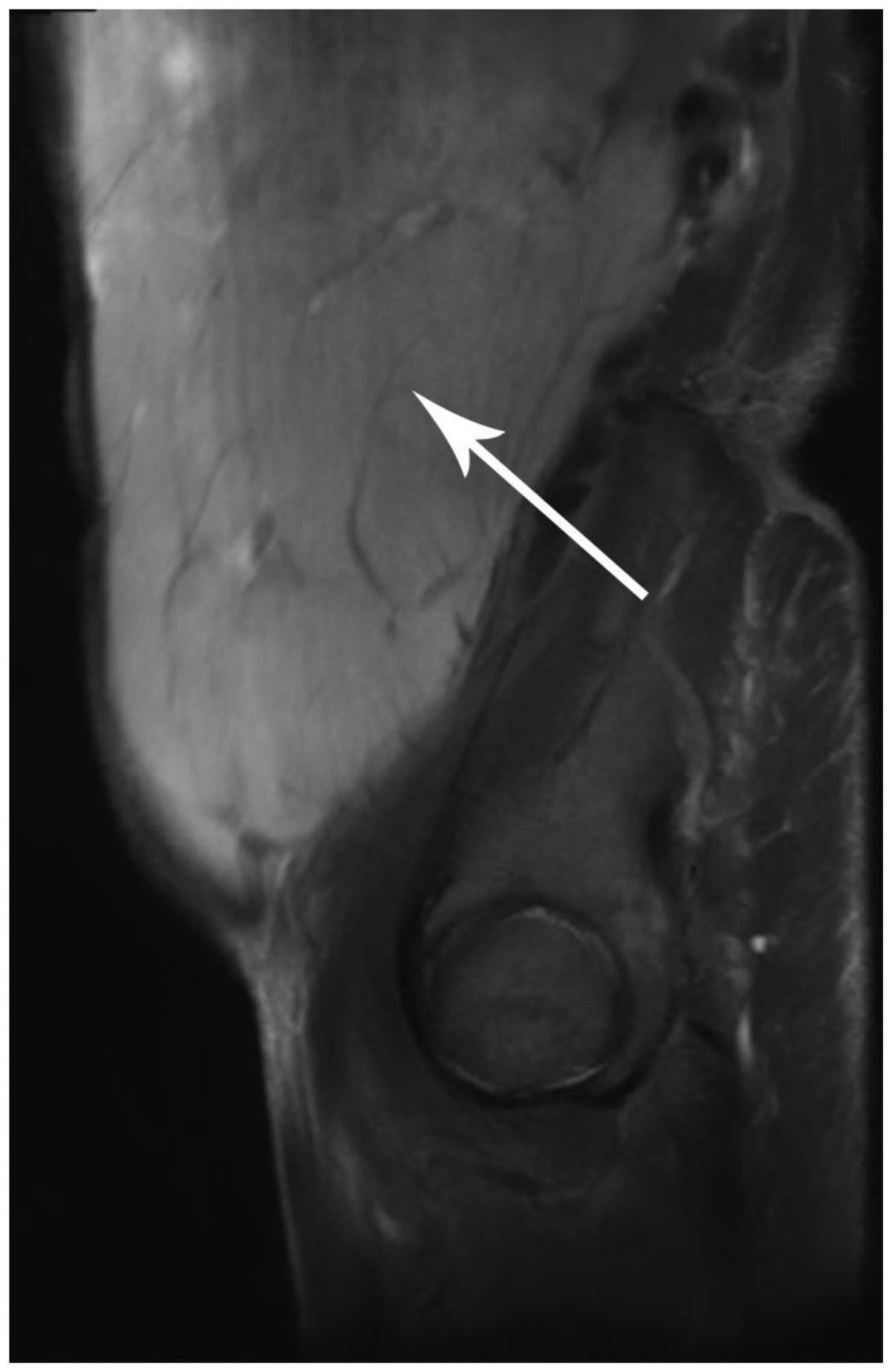
|
1
|
Novelli M, Rossi S, Rodriguez-Justo M,
Taniere P, Seddon B, Toffolatti L, Sartor C, Hogendoorn PC, Sciot
R, Van Glabbeke M, et al: DOG1 and CD117 are the antibodies of
choice in the diagnosis of gastrointestinal stromal tumours.
Histopathology. 57:259–270. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Su YY, Chiang NJ, Wu CC and Chen LT:
Primary gastrointestinal stromal tumor of the liver in an anorectal
melanoma survivor: A case report. Oncol Lett. 10:2366–2370.
2015.PubMed/NCBI
|
|
3
|
Bár T, Sankot J, Adamová Z and Mičulka P:
Extraintestinal GIST - case report. Rozhl Chir. 94:383–386.
2015.(In Czech). PubMed/NCBI
|
|
4
|
Suryawanshi KH, Patil TB, Damle RP, Dravid
NV and Surana A: Gastrointestinal stromal tumour of small intestine
presenting as a mesenteric mass. J Clin Diagn Res. 8:FD14–FD16.
2014.PubMed/NCBI
|
|
5
|
Sashidharan P, Matele A, Matele U, Al
Felahi N and Kassem KF: Gastrointestinal stromal tumors: A case
report. Oman Med J. 29:138–141. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liang X, Yu H, Zhu LH, Wang XF and Cai XJ:
Gastrointestinal stromal tumors of the duodenum: Surgical
management and survival results. World J Gastroenterol.
19:6000–6010. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Salari M, Ahadi M, Hoseini SM, Mokhtari E,
Gafarzadehgan K, Hashemian HR, Esmaeili B and Vossoughinia H:
Gastrointestinal Stromal Tumors in Northeastern Iran: 46 Cases
During 2003-2012. Middle East J Dig Dis. 7:161–165. 2015.PubMed/NCBI
|
|
8
|
Rammohan A, Sathyanesan J, Rajendran K,
Pitchaimuthu A, Perumal SK, Srinivasan U, Ramasamy R, Palaniappan R
and Govindan M: A gist of gastrointestinal stromal tumors: A
review. World J Gastrointest Oncol. 5:102–112. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Antonopoulos P, Leonardou P, Barbagiannis
N, Alexiou K, Demonakou M and Economou N: Gastrointestinal and
extragastrointestinal stromal tumors: Report of two cases and
review of the literature. Case Rep Gastroenterol. 8:61–66. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Stamatakos M, Douzinas E, Stefanaki C,
Safioleas P, Polyzou E, Levidou G and Safioleas M: Gastrointestinal
stromal tumor. World J Surg Oncol. 7:61–70. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sorour MA, Kassem MI, Ghazal A-H,
El-Riwini MT and Abu Nasr A: Gastrointestinal stromal tumors (GIST)
related emergencies. Int J Surg. 12:269–280. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao X and Yue C: Gastrointestinal stromal
tumor. J Gastrointest Oncol. 3:189–208. 2012.PubMed/NCBI
|
|
13
|
Beham AW, Schaefer I-M, Schüler P, Cameron
S and Ghadimi BM: Gastrointestinal stromal tumors. Int J Colorectal
Dis. 27:689–700. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Giuliani J and Bonetti A: The occurrence
of gastrointestinal stromal tumors and second malignancies. J
Gastrointest Cancer. 46:408–412. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gerrish ST and Smith JW: Gastrointestinal
stromal tumors - diagnosis and management: A brief review. Ochsner
J. 8:197–204. 2008.PubMed/NCBI
|
|
16
|
Dickhoff C, Leguit RJ, Slors JF, Vervenne
WL and Bemelman WA: Giant rectal gastrointestinal stromal tumors: A
report of two cases. Case Rep Gastroenterol. 2:54–69. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
de Oliveira RP, Portari Filho PE, Iglesias
AC, de Oliveira CA and Pannain VL: Comparative study of the
different degrees of risk of gastrointestinal stromal tumor. Rev
Col Bras Cir. 42:32–36. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhao WY, Xu J, Wang M, Zhang ZZ, Tu L,
Wang CJ, Cao H and Zhang ZG: Evaluation of high-risk
clinicopathological indicators in gastrointestinal stromal tumors
for prognosis and imatinib treatment outcome. BMC Gastroenterol.
14:105–113. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cai PQ, Lv XF, Tian L, Luo ZP, Mitteer RA
Jr, Fan Y and Wu YP: CT characterization of duodenal
gastrointestinal stromal tumors. AJR Am J Roentgenol. 204:988–993.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Serrano C and George S: Recent advances in
the treatment of gastrointestinal stromal tumors. Ther Adv Med
Oncol. 6:115–127. 2014. View Article : Google Scholar : PubMed/NCBI
|