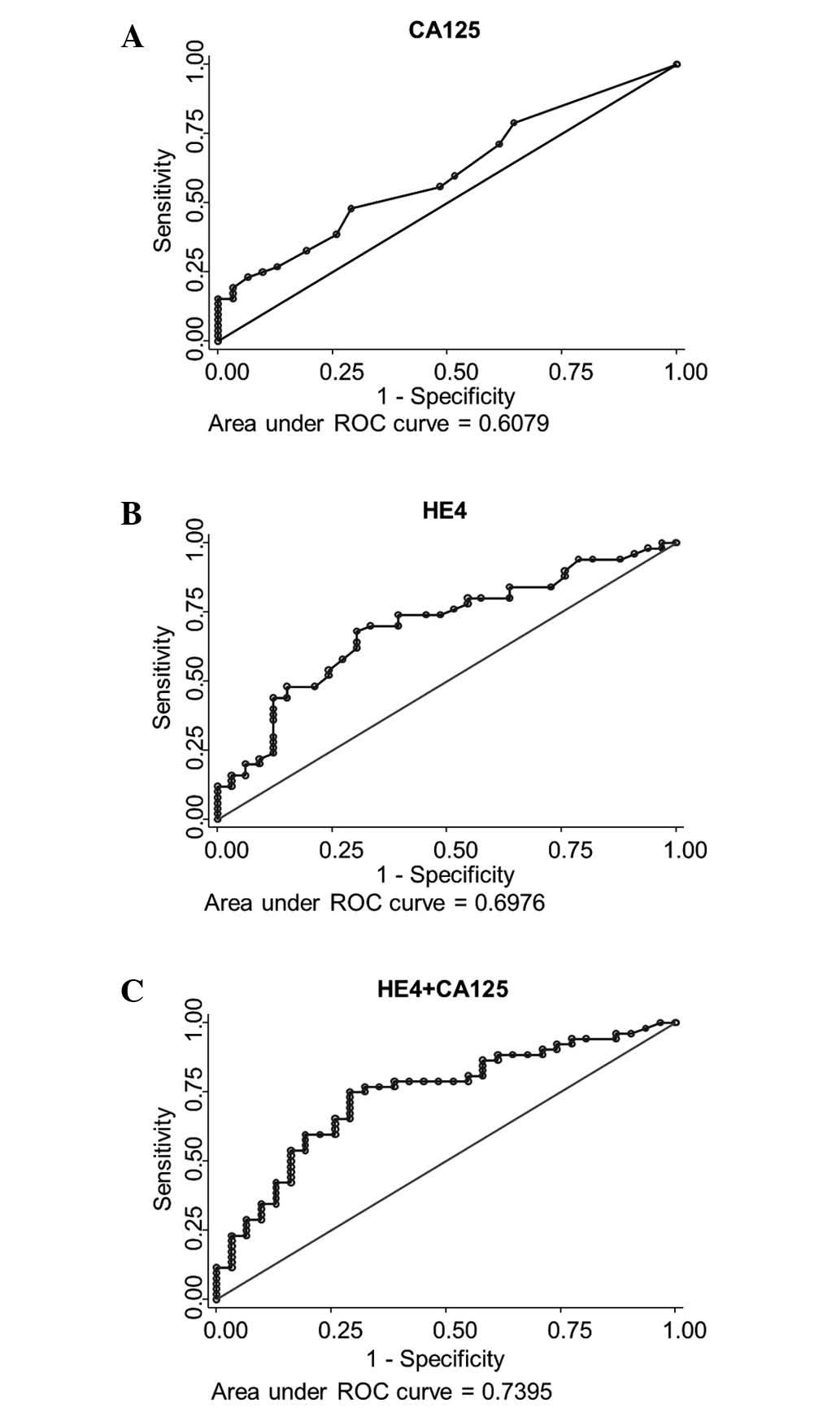
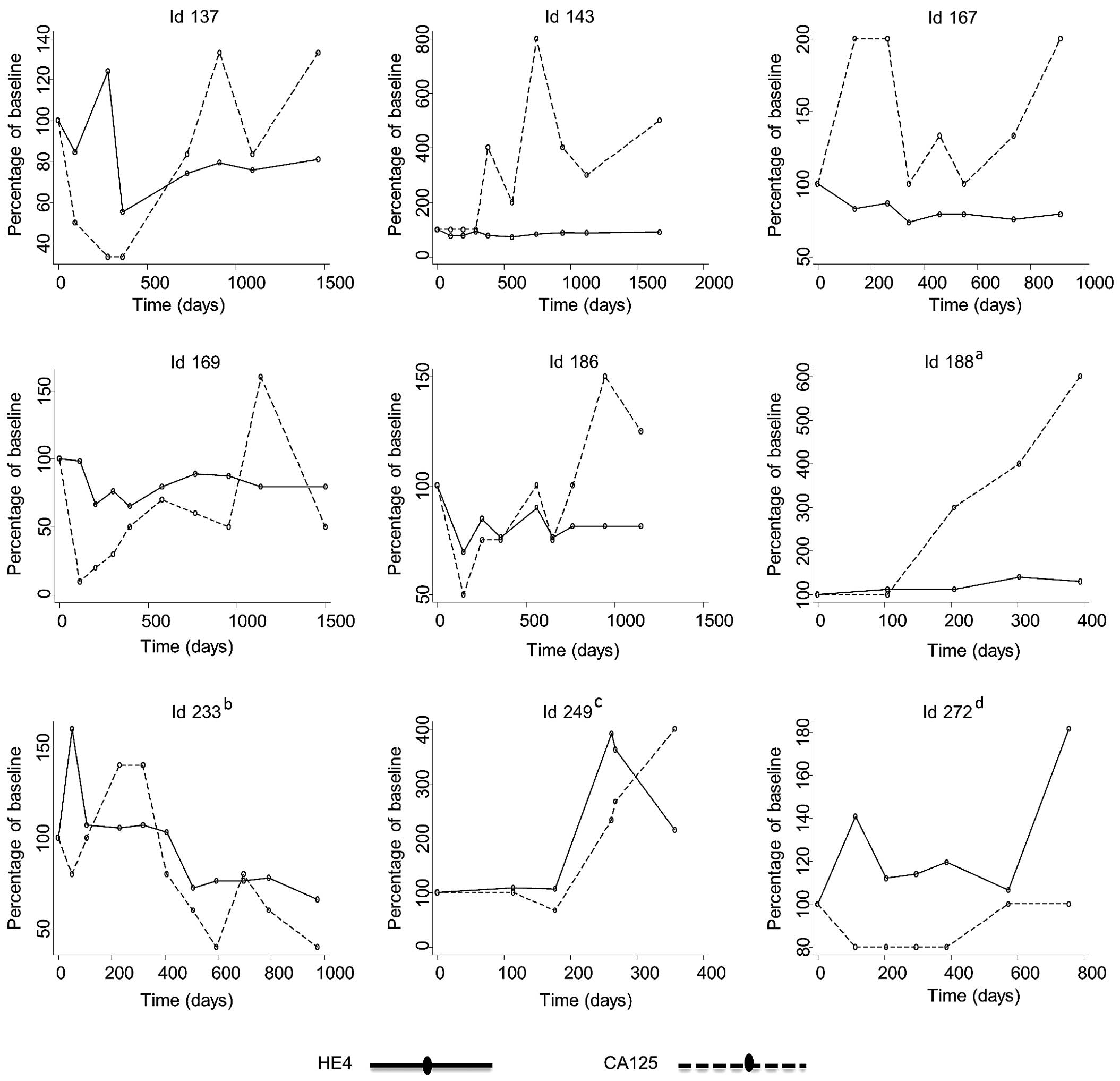
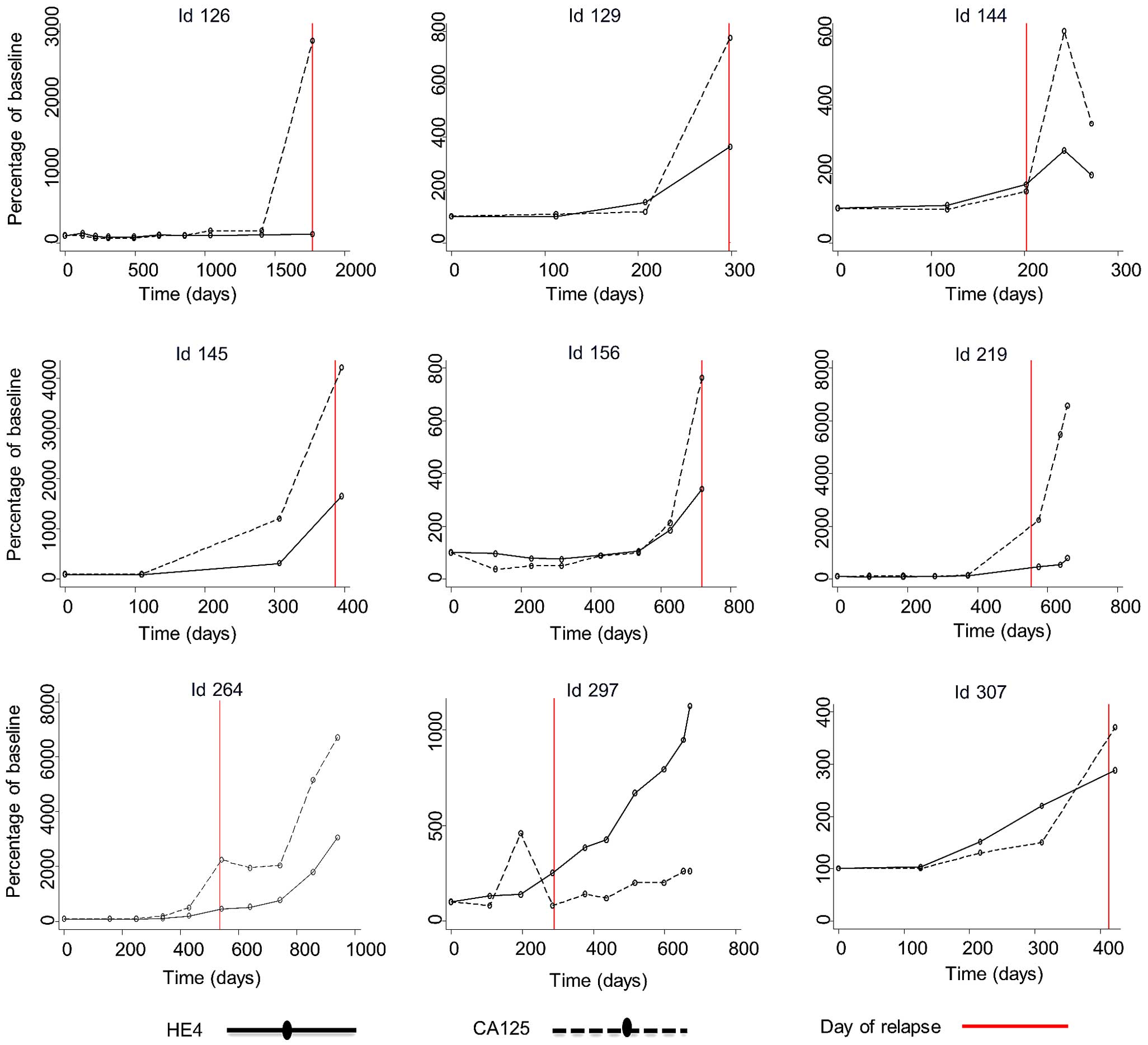
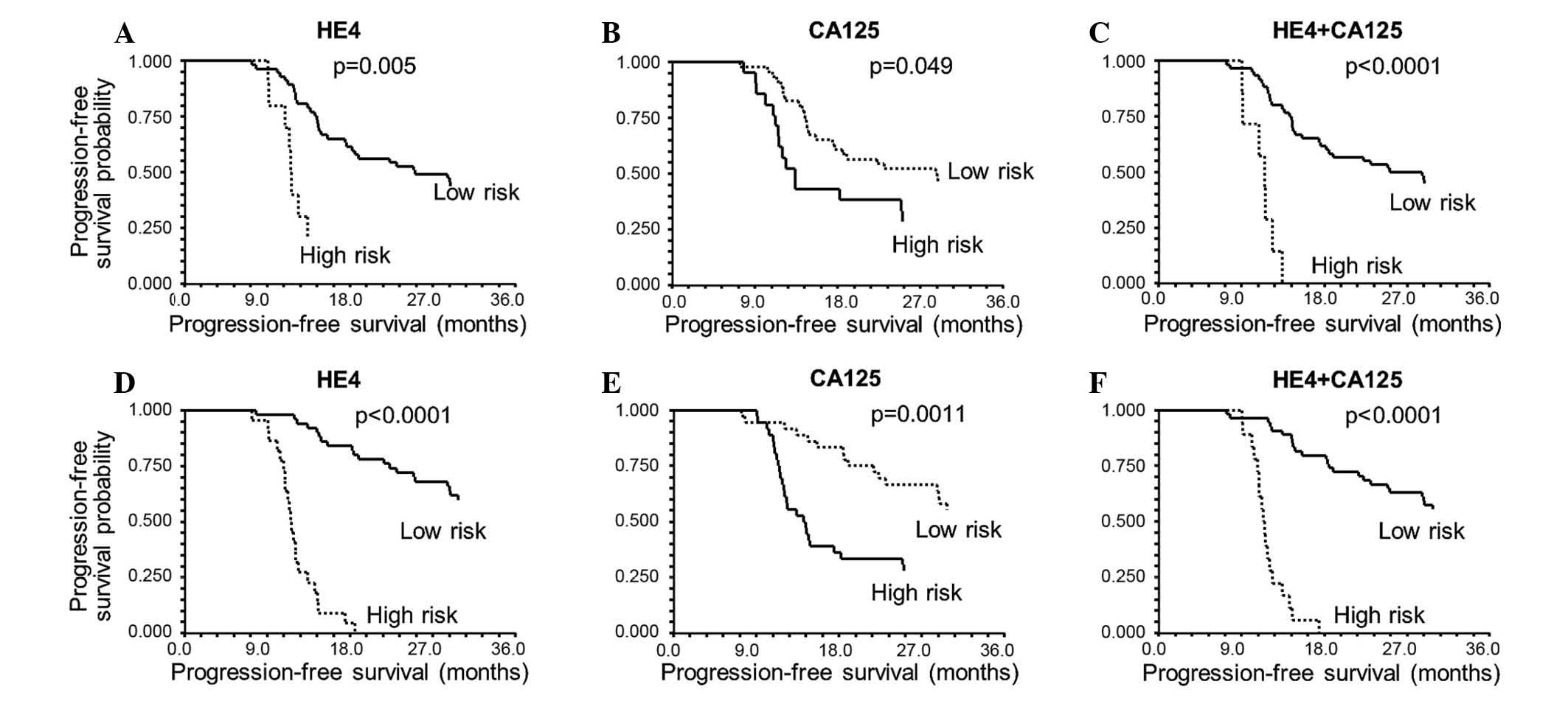
|
1
|
Siegel R, Naishadham MA and Jemal A:
Cancer statistics 2012. CA Cancer J Clin. 62:10–29. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liu PY, Alberts DS, Monk BJ, Brady M, Moon
J and Markman M: An early signal of CA-125 progression for ovarian
cancer patients receiving maintenance treatment after complete
clinical response to primary therapy. J Clin Oncol. 25:3615–3620.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vergote I, Rustin GJ, Eisenhauer EA,
Kristensen GB, Pujade-Lauraine E, Parmar MK, Friedlander M,
Jakobsen A and Vermorken JB: Re: New guidelines to evaluate the
response to treatment in solid tumors [ovarian cancer]. Gynecologic
Cancer Intergroup. J Natl Cancer Inst. 92:1534–1535. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rustin GJ, Nelstrop AE, Tuxen MK and
Lambert HE: Defining progression of ovarian carcinoma during
follow-up according to CA 125: A north thames ovary group study.
Ann Oncol. 7:361–364. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tuxen MK, Sölétormos G and Dombernowsky P:
Serum tumor marker CA 125 for monitoring ovarian cancer during
follow-up. Scand J Clin Lab Invest. 62:177–188. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rustin GJ, van der Burg ME, Griffin CL,
Guthrie D, Lamont A, Jayson GC, Kristensen G, Mediola C, Coens C,
Qian W, et al: Early versus delayed treatment of relapsed ovarian
cancer (MRC OV05/EORTC 55955): A randomised trial. Lancet.
376:1155–1163. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Harter P: DESKTOP III (AGO-OVAR OP.4): A
randomized trial evaluating cytoreductive surgery in patients with
platinum-sensitive recurrent ovarian cancer. http://www.gcig.igcs.org/ClinicalTrials.html
|
|
8
|
Fleming ND, Cass I, Walsh CS, Karlan BY
and Li AJ: CA125 surveillance increases optimal resectability at
secondary cytoreductive surgery for recurrent epithelial ovarian
cancer. Gynecol Oncol. 121:249–252. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Einhorn N, Bast RC Jr, Knapp RC, Tjernberg
B and Zurawski VR Jr: Preoperative evaluation of serum CA 125
levels in patients with primary epithelial ovarian cancer. Obstet
Gynecol. 67:414–416. 1986.PubMed/NCBI
|
|
10
|
Clauss A, Lilja H and Lundwall A: A locus
on human chromosome 20 contains several genes expressing protease
inhibitor domains with homology to whey acidic protein. Biochem J.
368:233–242. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bouchard D, Morisset D, Bourbonnais Y and
Tremblay GM: Proteins with whey-acidic-protein motifs and cancer.
Lancet Oncol. 7:167–174. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bouchard D, Morisset D, Bourbonnais Y and
Tremblay GM: Proteins with whey-acidic-protein motifs and cancer.
Lancet Oncol. 7:167–174. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Galgano MT, Hampton GM and Frierson HF Jr:
Comprehensive analysis of HE4 expression in normal and malignant
human tissues. Mod Pathol. 19:847–853. 2006.PubMed/NCBI
|
|
14
|
Drapkin R, von Horsten HH, Lin Y, Mok SC,
Crum CP, Welch WR and Hecht JL: Human epididymis protein 4 (HE4) is
a secreted glycoprotein that is overexpressed by serous and
endometrioid ovarian carcinomas. Cancer Res. 65:2162–2169. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hellström I, Raycraft J, Hayden-Ledbetter
M, Ledbetter JA, Schummer M, McIntosh M, Drescher C, Urban N and
Hellström KE: The HE4 (WFDC2) protein is a biomarker for ovarian
carcinoma. Cancer Res. 63:3695–3700. 2003.PubMed/NCBI
|
|
16
|
Hellstrom I and Hellstrom KE: SMRP and HE4
as biomarkers for ovarian carcinoma when used alone and in
combination with CA125 and/or each other. Adv Exp Med Biol.
622:15–21. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hellstrom I and Hellstrom KE: fTwo novel
biomarkers, mesothelin and HE4, for diagnosis of ovarian carcinoma.
Expert Opin Med Diagn. 5:227–240. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Montagnana M, Lippi G, Ruzzenente O,
Bresciani V, Danese E, Scevarolli S, Salvagno GL, Giudici S,
Franchi M and Guidi GC: The utility of serum human epididymis
protein 4 (HE4) in patients with a pelvic mass. J Clin Lab Anal.
23:331–335. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Moore RG, Brown AK, Miller MC, Skates S,
Allard WJ, Verch T, Steinhoff M, Messerlian G, DiSilvestro P,
Granai CO and Bast RC Jr: The use of multiple novel tumor
biomarkers for the detection of ovarian carcinoma in patients with
a pelvic mass. Gynecol Oncol. 108:402–408. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Moore RG, McMeekin DS, Brown AK,
DiSilvestro P, Miller MC, Allard WJ, Gajewski W, Kurman R, Bast RC
Jr and Skates SJ: A novel multiple marker bioassay utilizing HE4
and CA125 for the prediction of ovarian cancer in patients with a
pelvic mass. Gynecol Oncol. 112:40–46. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Steffensen KD, Waldstrøm M, Brandslund I
and Jakobsen A: Prognostic impact of prechemotherapy serum levels
of HER2, CA125, and HE4 in ovarian cancer patients. Int J Gynecol
Cancer. 21:1040–1047. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Steffensen KD, Waldstrøm M and Jakobsen A:
The relationship of platinum resistance and ERCC1 protein
expression in epithelial ovarian cancer. Int J Gynecol Cancer.
19:820–825. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
World Medical Association Declaration of
Helsinki: ethical principles for medical research involving human
subjects. JAMA. 310:2191–2194. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rustin GJ: Use of CA-125 to assess
response to new agents in ovarian cancer trials. J Clin Oncol.
21(Suppl 10): S187–S193. 2003. View Article : Google Scholar
|
|
25
|
Rustin GJ, Quinn M, Thigpen T, du Bois A,
Pujade-Lauraine E, Jakobsen A, Eisenhauer E, Sagae S, Greven K,
Vergote I, et al: Re: New guidelines to evaluate the response to
treatment in solid tumors (ovarian cancer). J Natl Cancer Inst.
96:487–488. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Havrilesky LJ, Whitehead CM, Rubatt JM,
Cheek RL, Groelke J, He Q, Malinowski DP, Fischer TJ and Berchuck
A: Evaluation of biomarker panels for early stage ovarian cancer
detection and monitoring for disease recurrence. Gynecol Oncol.
110:374–382. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Plotti F, Capriglione S, Terranova C,
Montera R, Aloisi A, Damiani P, Muzii L, Scaletta G,
Benedetti-Panici P and Angioli R: Does HE4 have a role as biomarker
in the recurrence of ovarian cancer? Tumour Biol. 33:2117–2123.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Manganaro L, Michienzi S, Vinci V,
Falzarano R, Saldari M, Granato T, Viggiani V, Frati L and Anastasi
E: Serum HE4 levels combined with CE CT imaging improve the
management of monitoring women affected by epithelial ovarian
cancer. Oncol Rep. 30:2481–2487. 2013.PubMed/NCBI
|
|
29
|
Anastasi E, Marchei GG, Viggiani V,
Gennarini G, Frati L and Reale MG: HE4: A new potential early
biomarker for the recurrence of ovarian cancer. Tumour Biol.
31:113–119. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Schummer M, Drescher C, Forrest R, Gough
S, Thorpe J, Hellström I, Hellström KE and Urban N: Evaluation of
ovarian cancer remission markers HE4, MMP7 and Mesothelin by
comparison to the established marker CA125. Gynecol Oncol.
125:65–69. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Granato T, Midulla C, Longo F, Colaprisca
B, Frati L and Anastasi E: Role of HE4, CA72.4, and CA125 in
monitoring ovarian cancer. Tumour Biol. 33:1335–1339. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Steffensen KD, Waldstrøm M, Brandslund I,
Petzold M and Jakobsen A: The prognostic and predictive value of
combined HE4 and CA-125 in ovarian cancer patients. Int J Gynecol
Cancer. 22:1474–1482. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rustin GJ, van der Burg ME, Griffin CL,
Guthrie D, Lamont A, Jayson GC, Kristensen G, Mediola C, Coens C,
Qian W, et al: Early versus delayed treatment of relapsed ovarian
cancer (MRC OV05/EORTC 55955): A randomised trial. Lancet.
376:1155–1163. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
National Cancer Institute: Cancer
statistics: SEER stat fact sheets, ovary cancer. http://seer.cancer.gov/statfacts/html/ovary.htmlAccessed.
April 14–2016
|