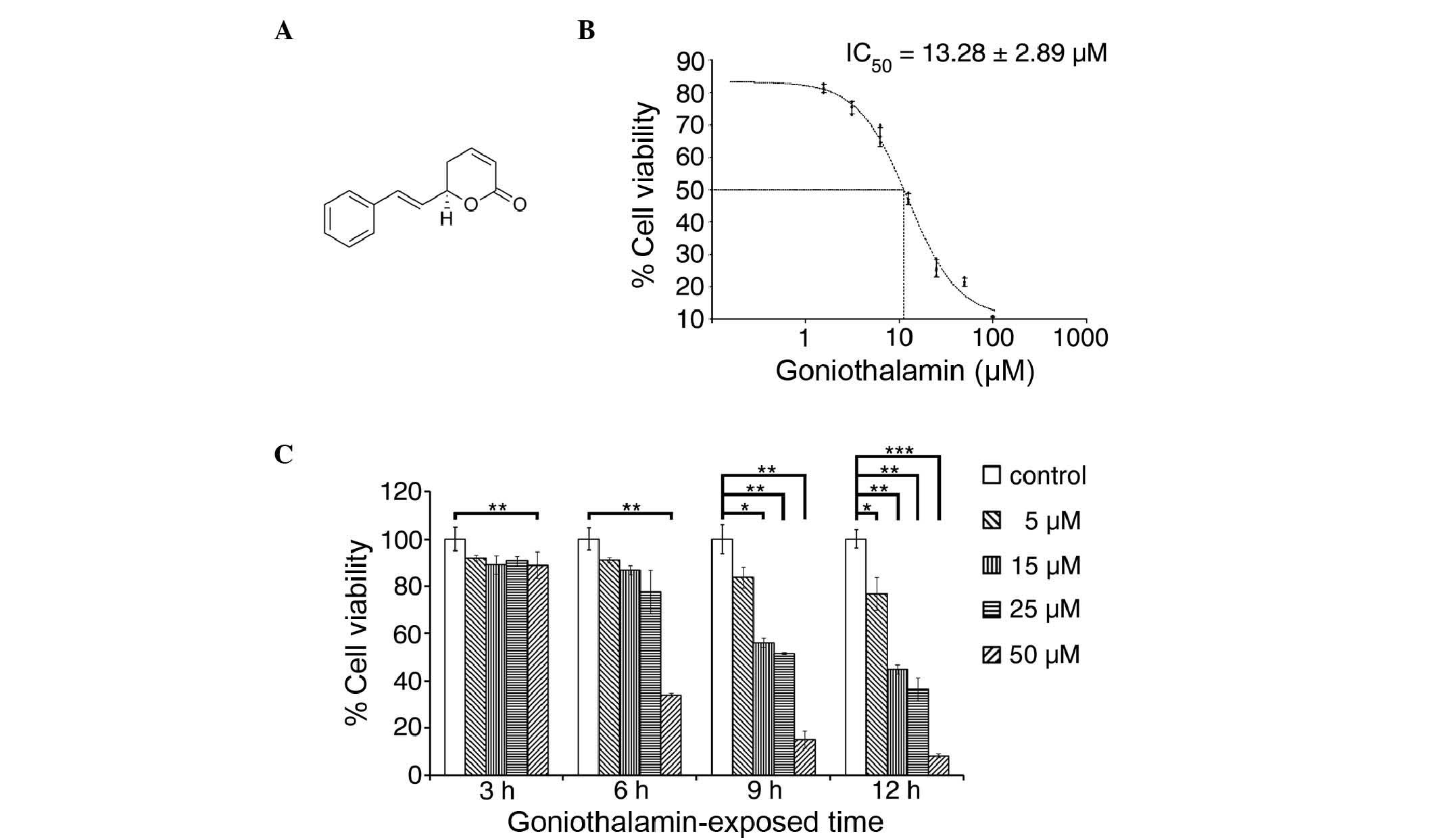
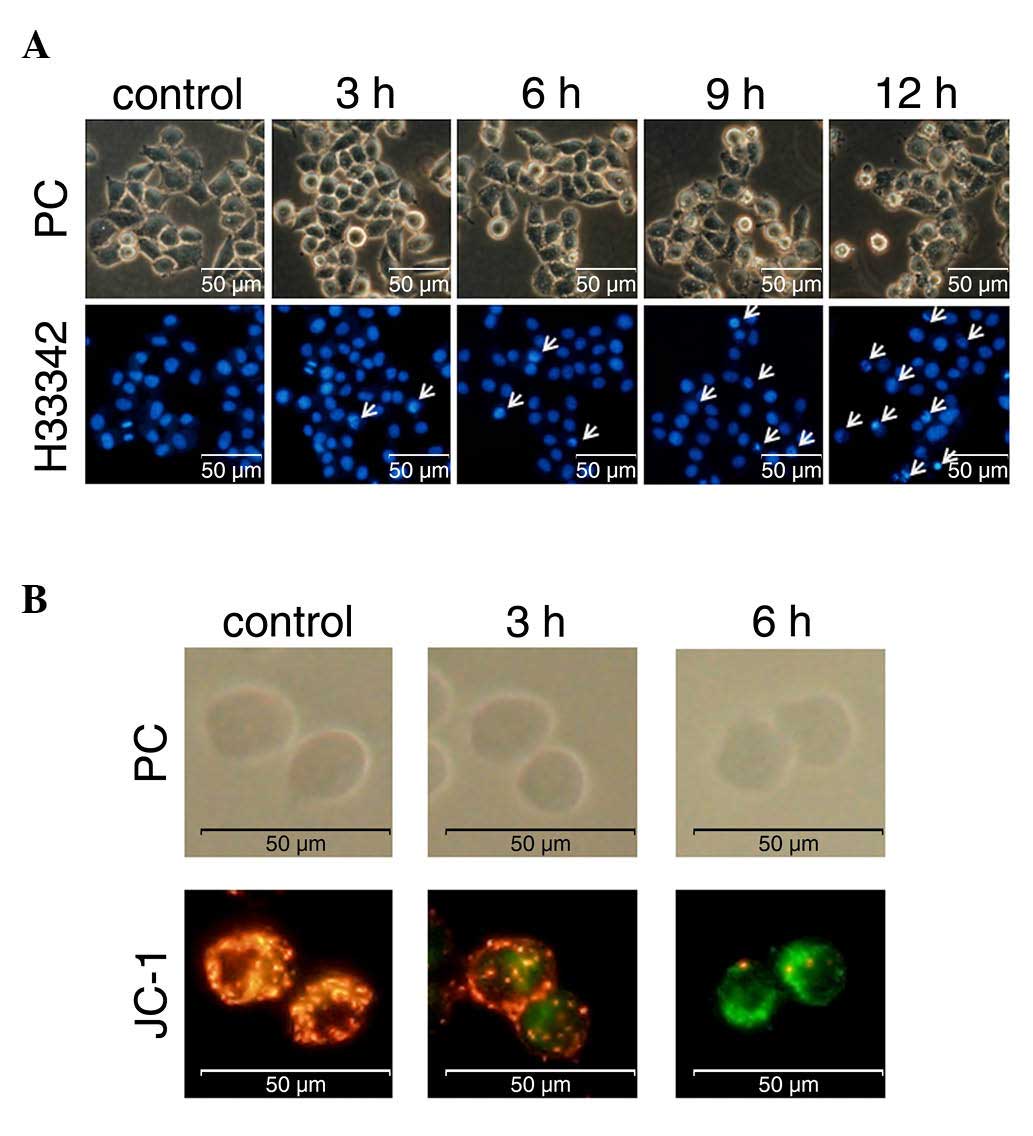
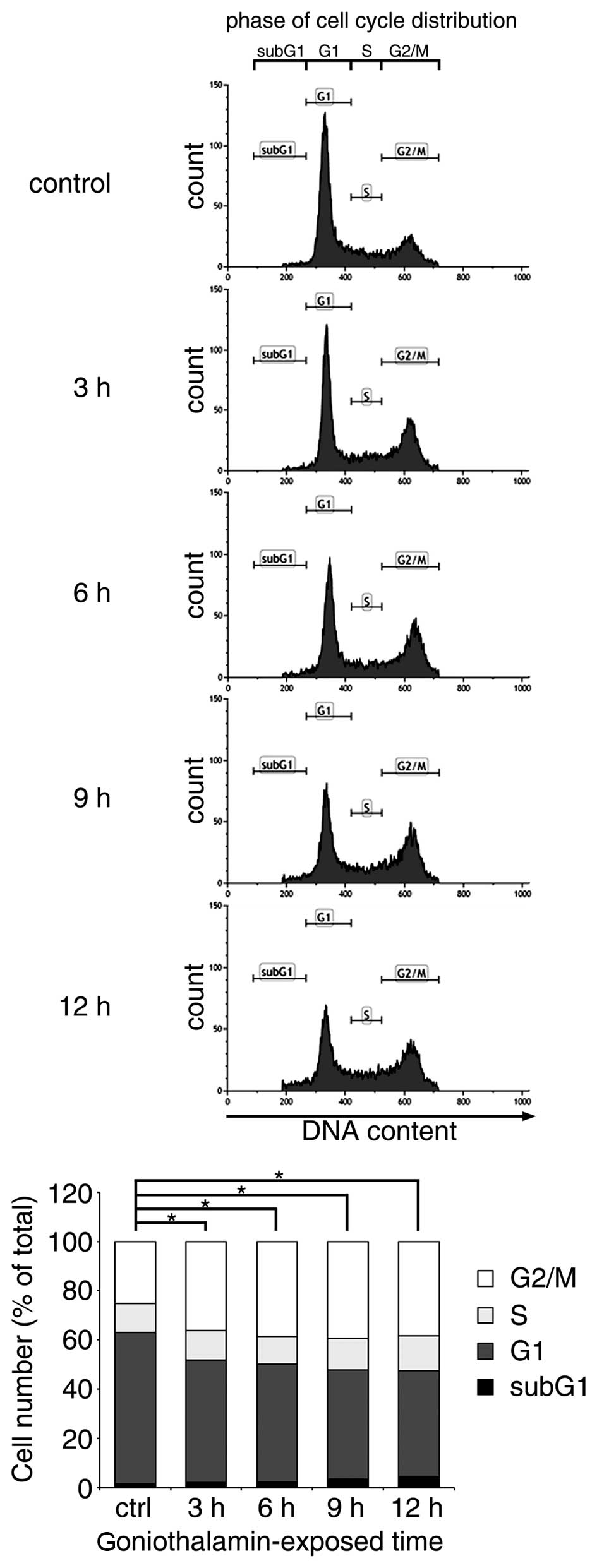
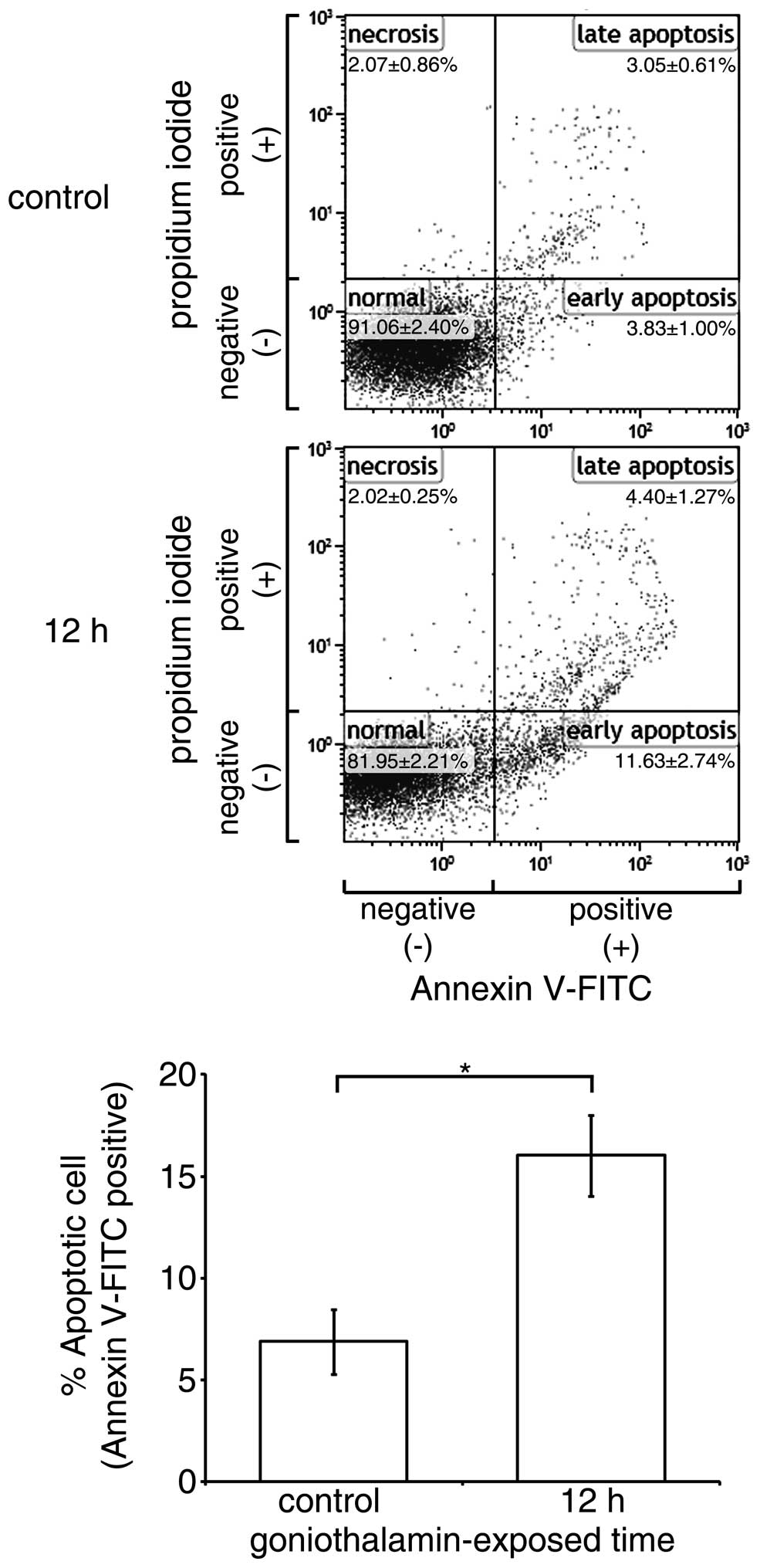
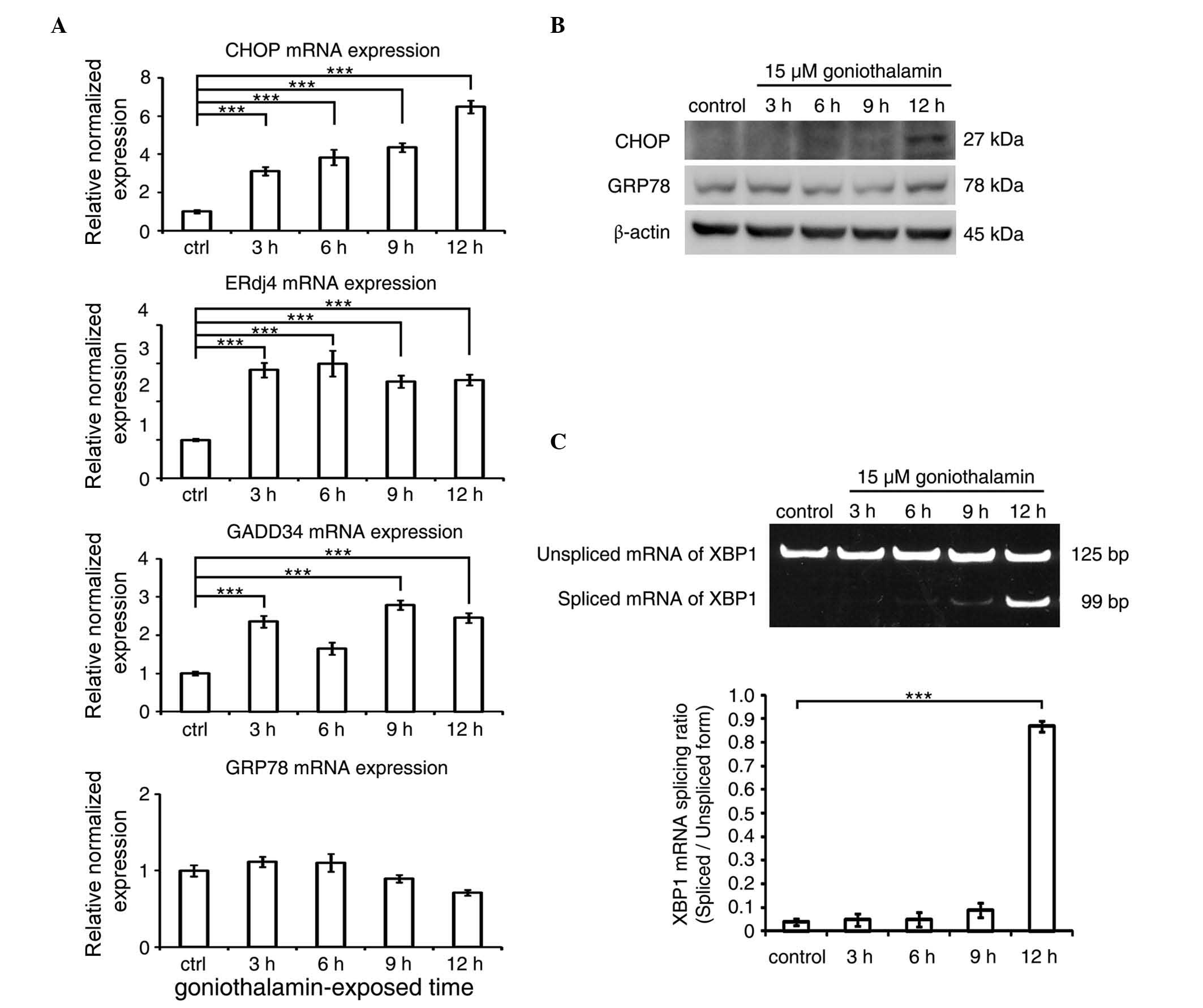
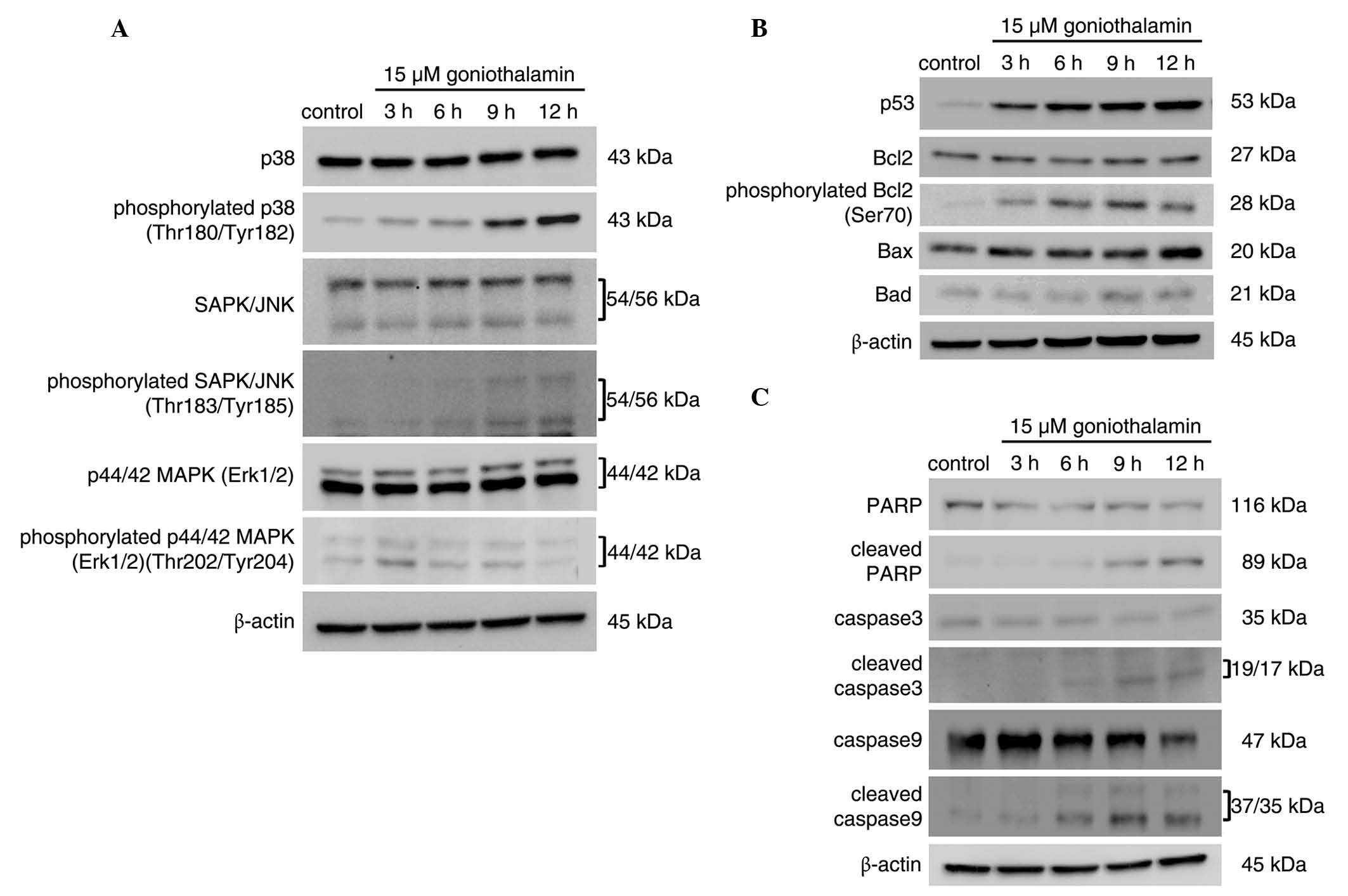
|
1
|
Mereyala HB and Joe M: Cytotoxic activity
of styryl lactones and their derivatives. Curr Med Chem Anticancer
Agents. 1:293–300. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
de Fátima Â, Modolo LV, Conegero LS, Pilli
RA, Ferreira CV, Kohn LK and de Carvalho JE: Styryl lactones and
their derivatives: Biological activities, mechanisms of action and
potential leads for drug design. Curr Med Chem. 13:3371–3384. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jewers K, Davis JR, Dougan J, Machanda AH,
Blunden G, Kyi A and Wetchapinan S: Goniothalamin and its
distribution in four goniothalamus species. Phytochemistry.
11:2025–2030. 1972. View Article : Google Scholar
|
|
4
|
Seyed MA, Jantan I and Bukhari SN:
Emerging anticancer potentials of goniothalamin and its molecular
mechanisms. Biomed Res Int. 2014:5365082014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Inayat-Hussain SH, Annuar BO, Din LB, Ali
AM and Ross D: Loss of mitochondrial transmembrane potential and
caspase-9 activation during apoptosis induced by the novel
styryl-lactone goniothalamin in HL-60 leukemia cells. Toxicol In
Vitro. 17:433–439. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rajab NF, Hamid ZA, Hassan H, Ali MA, Din
LB and Inayat-Hussain SH: Evaluation of the cytotoxic and genotoxic
effects of goniothalamin in leukemic cell lines. Environ Mutagen
Res. 27:161–164. 2005. View Article : Google Scholar
|
|
7
|
Wattanapiromsakul C, Wangsintaweekul B,
Sangprapan P, Itharat A and Keawpradub N: Goniothalamin, a
cytotoxic compound, isolated from Goniothalamus macrophyllus
(Blume) Hook. f. & Thomson var. macrophyllus. Songklanakarin
Journal of Science and Technology. 27:480–487. 2005.
|
|
8
|
de Fátima A, Kohn LK, Antônio MA, de
Carvalho E and Pilli RA: (R)-Goniothalamin: Total syntheses and
cytotoxic activity against cancer cell lines. Bioorg Med Chem.
13:2927–2933. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Alabsi AM, Ali R, Ali AM, Al-Dubai SAR,
Harun H, Abu Kasim NH and Alsalahi A: Apoptosis induction, cell
cycle arrest and in vitro anticancer activity of goniothalamin in a
cancer cell lines. Asian Pac J Cancer Prev. 13:5131–5136. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Alabsi AM, Ali R, Ali AM, Harun H,
Al-Dubai SA, Ganasegeran K, Alshagga MA, Salem SD and Abu Kasim NH:
Induction of caspase-9, biochemical assessment and morphological
changes caused by apoptosis in cancer cells treated with
goniothalamin extracted from Goniothalamus macrophyllus. Asian Pac
J Cancer Prev. 14:6273–6280. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Petsophonsakul P, Pompimon W and
Banjerdpongchai R: Apoptosis induction in Human leukemic
promyelocytic HL-60 and monocytic U937 cell lines by goniothalamin.
Asian Pac J Cancer Prev. 14:2885–2889. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xu C, Bailly-Maitre B and Reed JC:
Endoplasmic reticulum stress: Cell life and death decisions. J Clin
Invest. 115:2656–2664. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Szegezdi E, Logue SE, Gorman AM and Samali
A: Mediators of endoplasmic reticulum stress-induced apoptosis.
EMBO Rep. 7:880–885. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Elmore S: Apoptosis: A review of
programmed cell death. Toxicol Pathol. 35:495–516. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nikoletopoulou V, Markaki M, Palikaras K
and Tavernarakis N: Crosstalk between apoptosis, necrosis and
autophagy. Biochim Biophys Acta. 1833:3448–3459. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Win S, Than TA, Fernandez-Checa JC and
Kaplowitz N: JNK interaction with Sab mediates ER stress induced
inhibition of mitochondrial respiration and cell death. Cell Death
Dis. 5:e9892014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yuan L, Wang J, Xiao H, Wu W, Wang Y and
Liu X: MAPK signaling pathways regulate mitochondrial-mediated
apoptosis induced by isoorientin in human hepatoblastoma cancer
cells. Food Chem Toxicol. 53:62–68. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sui X, Kong N, Ye L, Han W, Zhou J, Zhang
Q, He C and Pan H: p38 and JNK MAPK pathways control the balance of
apoptosis and autophagy in response to chemotherapeutic agents.
Cancer Lett. 344:174–179. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Saxena U, Sauvaget C and Sankaranarayanan
R: Evidence-based screening, early diagnosis and treatment strategy
of cervical cancer for national policy in low- resource countries:
Example of India. Asian Pac J Cancer Prev. 13:1699–1703. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Landry JJ, Pyl PT, Rausch T, Zichner T,
Tekkedil MM, Stütz AM, Jauch A, Aiyar RS, Pau G, Delhomme N, et al:
The genomic and transcriptomic landscape of a HeLa cell line. G3
(Bethesda). 3:1213–1224. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Denizot F and Lang R: Rapid colorimetric
assay for cell growth and survival: Modifications to the
tetrazolium dye procedure giving improved sensitivity and
reliability. J Immunol Methods. 89:271–277. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Oberhammer FA, Hochegger K, Fröschl G,
Tiefenbacher R and Pavelka M: Chromatin condensation during
apoptosis is accompanied by degradation of Lamin A+B, without
enhanced activation of cdc2 kinase. J Cell Biol. 126:827–837. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Krishan A: Rapid flow cytofluorometric
analysis of mammalian cell cycle by propidium iodide staining. J
Cell Biol. 66:188–193. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Perelman A, Wachtel C, Cohen M, Haupt S,
Shapiro H and Tzur A: JC-1: Alternative excitation wavelengths
facilitate mitochondrial membrane potential cytometry. Cell Death
Dis. 22:e4302012. View Article : Google Scholar
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2-ΔΔCT method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Taylor SC and Posch A: The design of a
quantitative western blot experiment. Biomed Res Int.
2014:3615902014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bradford MM: A rapid and sensitive method
for the quantitation of microgram quantities of protein utilizing
the principle of protein-dye binding. Anal Biochem. 72:248–254.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shankar S, Kumar D and Srivastava RK:
Epigenetic modifications by dietary phytochemicals: Implications
for personalized nutrition. Pharmacol Ther. 138:1–17. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yen CY, Chiu CC, Haung RW, Yeh CC, Huang
KJ, Chang KF, Hseu YC, Chang FR, Chang HW and Wu YC:
Antiproliferative effects of goniothalamin on Ca9-22 oral cancer
cells through apoptosis, DNA damage and ROS induction. Mutat Res.
747:253–258. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen WY, Wu CC, Lan YH, Chang FR, Teng CM
and Wu YC: Goniothalamin induces cell cycle-specific apoptosis by
modulating the redox status in MDA-MB-231 cells. Eur J Pharmacol.
522:20–29. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Thornton TM and Rincon M: Non-classical
p38 map kinase functions: Cell cycle checkpoints and survival. Int
J Biol Sci. 5:44–51. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Fadok VA, Bratton DL, Frasch SC, Warner ML
and Henson PM: The role of phosphatidylserine in recognition of
apoptotic cells by phagocytes. Cell Death Diff. 5:551–562. 1998.
View Article : Google Scholar
|
|
35
|
Denecker G, Dooms H, Van Loo G, Vercammen
D, Grooten J, Fiers W, Declercq W and Vandenabeele P: Phosphatidyl
serine exposure during apoptosis precedes release of cytochrome c
and decrease in mitochondrial transmembrane potential. FEBS Lett.
465:47–52. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rello S, Stockert JC, Moreno V, Gámez A,
Pacheco M, Juarranz A, Cañete M and Villanueva A: Morphological
criteria to distinguish cell death induced by apoptotic and
necrotic treatments. Apoptosis. 10:201–208. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hanshaw RG and Smith BD: New reagents for
phosphatidylserine recognition and detection of apoptosis. Bioorg
Med Chem. 13:5035–5042. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Al-Qubaisi M, Rosli R, Subramani T, Omar
AR, Yeap SK, Ali AM and Alitheen NB: Goniothalamin selectively
induces apoptosis on human hepatoblastoma cells through caspase-3
activation. Nat Prod Res. 27:2216–2218. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Luo X, Budihardio I, Zou H, Slaughter C
and Wang X: Bid, a Bcl-2 interacting protein, mediates cytochrome c
release from mitochondria in response to activation of cell surface
death receptors. Cell. 94:481–490. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yen HK, Fauzi AR, Din LB, McKelvey-Martin
VJ, Meng CK, Inayat-Hussain SH and Rajab NF: Involvement of
Seladin-1 in goniothalamin-induced apoptosis in urinary bladder
cancer cells. BMC Complement Altern Med. 14:2952014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Boatright KM and Salvesen GS: Mechanisms
of caspase activation. Curr Opin Cell Biol. 15:725–731. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Riedl SJ and Shi Y: Molecular mechanisms
of caspase regulation during apoptosis. Nat Rev Mol Cell Biol.
5:897–907. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Pop C, Timmer J, Sperandio S and Salvesen
GS: The apoptosome activates caspase-9 by dimerization. Mol Cell.
22:269–275. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lee H, Park M, Choi B, Oh E, Song M, Lee
J, Kim C, Lim BU and Park HJ: Endoplasmic reticulum stress-induced
JNK activation is a critical event leading to mitochondria-mediated
cell death caused by β-lapachone treatment. PLoS One. 6:e215332011.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chen Y and Brandizzi F: IRE1: ER stress
sensor and cell fate executor. Trends Cell Biol. 23:547–555. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Selimovic D, Ahmad M, El-Khattouti A,
Hannig M, Haïkel Y and Hassan M: Apoptosis-related protein-2
triggers melanoma cell death by a mechanism including both
endoplasmic reticulum stress and mitochondrial dysregulation.
Carcinogenesis. 32:1268–1278. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Urano F, Wang X, Bertolotti A, Zhang Y,
Chung P, Harding HP and Ron D: Coupling of stress in the ER to
activation of JNK protein kinases by transmembrane protein kinase
IRE1. Science. 287:664–666. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wada T and Penninger JM: Mitogen-activated
protein kinases in apoptosis regulation. Oncogene. 23:2838–2849.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Novoa I, Zeng H, Harding HP and Ron D:
Feedback inhibition of the unfolded protein response by
GADD34-mediated dephosphorylation of eIF2alpha. J Cell Biol.
153:1011–1022. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Shinjo S, Mizotani Y, Tashiro E and Imoto
M: Comparative analysis of the expression patterns of UPR-target
genes caused by UPR-inducing compounds. Biosci Biotechnol Biochem.
77:729–735. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Annis MG, Yethon JA, Leber B and Andrews
DW: There is more to life and death than mitochondria: Bcl-2
proteins at the endoplasmic reticulum. Biochim Biophys Acta.
1644:115–123. 2004. View Article : Google Scholar : PubMed/NCBI
|