Introduction
Hepatocellular carcinoma (HCC) is one of the most
common types of malignant neoplasms worldwide, causing ~700,000
mortalities annually (1). Overall,
half of these cases and mortalities were identified to occur in
China, and were attributed to the high prevalence of hepatitis B
virus (HBV) infections and liver cirrhosis (2,3). As a
potentially curative treatment for liver cirrhosis and cancer,
liver transplantation (LT) is widely used to treat selected
patients with early-stage HCC, and it has the greatest benefit
among all the established therapeutic options (4,5). However,
the frequent recurrence and high metastasis rates of HCC hinders
positive outcomes for patients. Due to the multiple genetic
alterations that are responsible for the progression of HCC
(6), exploration of the molecular
mechanism of HCC is important for disease diagnosis, treatment and
prediction of outcomes.
MicroRNAs (miRNAs/miR) are a class of small,
non-coding RNA molecules (containing ~22 nucleotides) identified in
plants, animals and certain viruses. They function in RNA silencing
and the post-transcriptional regulation of gene expression
(7,8).
Increasing evidence supports a marked association between cancer
and miRNAs (9). It is well documented
that aberrant miRNA expression serves a significant role in
carcinogenesis and cancer development (10). miRNAs may regulate the biological
behaviors of cancer, including proliferation, apoptosis and
invasion. Therefore, exploration of miRNAs may assist in
identifying novel diagnostic or prognostic markers and treatment
targets.
The Milan criteria (11) for identifying candidate patients with
HCC for LT have been accepted worldwide. However, they ignore the
molecular biomarker expression patterns in HCC and have been
criticized for being too restrictive (12). Our prior studies aimed to explore more
inclusive criteria for identifying LT candidates within the
patients with HCC population (12,13). The
noncoding RNA molecules are potentially valuable markers that may
assist in predicting HCC recurrence following LT, their aberrant
expression was significantly associated with increased risk of
recurrence (14). MicroRNA-424
(miR-424) was previously demonstrated to inhibit the proliferation
and metastasis of HCC (15,16). In the present study, the expression
pattern of miR-424 in HCC tissues from patients who received
transplant therapy, and its association with clinicopathological
parameters were investigated, and the utility of miR-424 in
predicting tumor recurrence in patients with HCC following LT was
explored. The functional implications of miR-424 in HCC were also
explored using in vitro and in vivo assays.
Materials and methods
Patient samples
A total of 121 patients with HCC, who were treated
with LT from January 2003 to December 2010 at The First Affiliated
Hospital, Zhejiang University School of Medicine (Zhejiang, China),
were enrolled in the present study. The inclusion criteria were as
previously described (17). Ages of
patients ranged from 41 to 73 years old and the male-female ratio
was 112/9. All patients were HBV-surface antigen-positive
(HBsAg+) and none were hepatitis C virus (HCV)-positive.
The present study was approved by the Medical Ethics Committee of
The First Affiliated Hospital of Zhejiang University, and written
informed consent was obtained from all patients. The patients were
diagnosed with HCC either prior or subsequent to transplantation
via histopathological diagnosis. The complete clinical and
laboratory data were available prior to surgery and during
follow-up. The follow-up course and diagnostic criteria of
recurrence have been previously described (18). The distribution of clinicopathological
data in the study cohort is summarized in Table I. Specimens of cancer/noncancerous
tissues and clinical information were available from these patients
subsequent to obtaining written informed consent.
 | Table I.Association between miR-424 expression
and the clinicopathological features of patients with human
hepatocellular carcinoma. |
Table I.
Association between miR-424 expression
and the clinicopathological features of patients with human
hepatocellular carcinoma.
|
| Tumor miR-424
expression |
|
|---|
|
|
|
|
|---|
| Variables | Low | High | P-valuea |
|---|
| Age, years |
| ≤50 | 30 | 33 | 0.522 |
|
>50 | 31 | 27 |
|
| Sex |
| Male | 55 | 57 | 0.311 |
|
Female | 6 | 3 |
|
| Portal vein tumor
thrombi |
|
Absent | 39 | 41 | 0.609 |
|
Present | 22 | 19 |
|
| Preoperative
α-fetoprotein level, ng/ml |
| ≤400 | 27 | 29 | 0.683 |
|
>400 | 34 | 31 |
|
| Histopathological
grading |
| Well +
moderately | 33 | 33 | 0.921 |
|
Poorly | 28 | 27 |
|
| Tumor size, cm |
| ≤5 | 30 | 40 | 0.051 |
|
>5 | 31 | 20 |
|
| Tumor number |
|
Single | 21 | 30 | 0.083 |
|
Multiple | 40 | 30 |
|
Postoperative follow-up
The surviving patients were followed up at the
outpatient clinic. The median follow-up duration was 25.12 months.
Tumor recurrence was monitored by the α-fetoprotein (AFP) levels,
ultrasonography, chest X-ray and emission computed tomography every
3 months for the first 2 years, and semiannually thereafter.
Imaging techniques, either intrahepatically or extrahepatically
(lymph nodes and distant metastases) were used to diagnose
recurrence. However, a simple increase in the AFP did not
conclusively indicate recurrence.
Cell lines and chemical reagents
The normal liver cell line, LO2; liver cancer cell
lines, SMMC-7721, Huh-7, HepG2, Bel-7402, and SK-HEP-1; and
metastatic human HCC cell line, HCCLM3, were purchased from the
American Type Culture Collection (Manassas, VA, USA), the Shanghai
Institute of Cell Biology (Shanghai, China) and the Liver Cancer
Institute of Fudan University (Shanghai, China). LO2, SMMC-7721 and
BEL-7402 cells were cultured in RMPI 1640 (Biological Industries,
Kibbutz Beit-Haemek, Israel); Huh-7, HepG2, SK-HEP-1 and HCCLM3
cells were maintained in Dulbecco's modified eagle's medium (DMEM;
Biological Industries). All cells were incubated at 37°C in a
humidified environment containing 5% CO2.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) analysis
TRIzol® (Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) and miScript II RT kits
(Qiagen, Inc., Valencia, CA, USA) were used to extract RNA from
LO2, SMMC-7721, Huh-7, HepG2, Bel-7402 and SK-HEP-1 cell lines.
Following incubation for 60 min at 37°C, the reverse transcription
mix was placed in a water bath at 95°C and then transferred to
refrigerator at 4°C. qPCR was conducted using the miScript
SYBR-Green kit (Qiagen, Inc.) on an Applied Biosystems ABI 7700
System (Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol. miR-424 primers were purchased from
Qiagen, Inc. GAPDH was used as internal control, with sequences as
follows: Sense, AGGTCGGAGTCAACGGATTTG and anti-sense,
GTGATGGCATGGACTGTGGT59. For miR-424 detection, U6 snRNA was used as
the endogenous control. Each experiment was performed in three
replicates. The 2−ΔΔCq method was used to analyze the
PCR data (19).
Cell model construction and
infection
SK-HEP-1 (highest miR-424 expression) and Huh-7
cells (last but one low miR-424 expression, selected as they were
more suitable for tumorgenicity) were infected with lentiviral
vectors carrying a miRNA mimic/inhibitor or nonsense controls (NC2)
(GenePharma Co., Shanghai, China) with polybrene, according to the
manufacturer's protocol. The sequences were as follows: miR-424
inhibitor-1, 5′ CAGCAGCAATTCATGTTTTGAA3′; miR-424 inhibitor-2, 5′
TTCAAAACATGAATTGCTGCTG3′; NC2, 5′TTCTCCGAACGTGTCACGT3′.
Analysis of cell viability, migration
and invasion
Cell viability of the aforementioned transfected HCC
cells (SK-HEP-1 and Huh-7) was determined using the CCK-8 assay
(Dojindo Molecular Technologies, Inc., Kumamoto, Japan), according
to the manufacturer's protocol. Cells (5×103) were
seeded into the wells of a 96-well plate. Subsequent to the cells
being incubated at 37°C for different time periods (24, 48, 72 and
96 h), DMEM medium was replaced with 100 µl DMEM medium
supplemented with 10 µl CCK-8 reagent. The cells were incubated for
2 h at 37°C. Finally, the optical density was measured at 450
nm.
Transwell invasion and migration experiments were
conducted as previously described (20). Briefly, the polycarbonate membrane of
the upper chamber was coated with Matrigel for invasion
experiments. A total of 72 h after incubation, SK-HEP-1 and Huh-7
cells with corresponding virus transfection were collected for
Transwell experiments. Cells (2×104) were seeded into the upper
chambers, which contained DMEM medium without fetal bovine serum
(Biological Industries). DMEM medium with 15% FBS was added to the
lower compartments. After 24 h (for migration experiments) or 48 h
(for invasion experiments), the cells on the lower surface were
fixed in 95% methanol for 10 min at room temperature and stained
for 20 min with 0.1% crystal violet at room temperature. The cells
were counted and images were captured using digital microscopy
(magnification, ×100). A total of 3 fields per filter were counted
in each group. The experiments were performed in triplicate.
Xenograft model analysis
To investigate the anti-proliferative effect of
miR-424 on HCC cells in vivo, a model with nude mice bearing
HCC cell xenografts was established: A total of fourteen
five-week-old male athymic nude mice with a median weight of 14 g
(Vital River Laboratory Animal Technology Co., Ltd., Beijing,
China) were maintained under pathogen-free conditions and were
provided with ad libitum access to sterilized food and water
at 26–28°C and were exposed to light for 12 h every day. Huh-7
cells (5×106) were infected with a miR-424 mimic or
nonsense control, injected subcutaneously into the right armpit,
near the chest, of each nude mouse. Seven mice were used in each
group. Six weeks following the injection, the mice were sacrificed
by removal of the cervical spine. The maximum tumor volume obtained
in the mice was 1.8 cm3. Tumor volume=1/2 (length ×
width2).
Statistical analysis
The experimental data are presented as the mean ±
standard deviation or frequency. Continuous data were analyzed
using unpaired Student's t-test to assess differences between the
two groups. Categorical data were analyzed using the χ2
test. The recurrence-free survival probability was analyzed with
the Kaplan-Meier method, and a log-rank test was used to estimate
the differences between groups. Independent prognostic indicators
were determined in the multivariate analysis using Cox's
proportional hazards model. The expression correlation was analyzed
by a two-tailed linear Spearman's rank test. All statistical
analyses were performed using SPSS v.16.0 (SPSS, Inc., Chicago, IL,
USA) and GraphPad Prism 5.0 (GraphPad Software, La Jolla, CA, USA).
P<0.05 was considered to indicate a statistically significant
difference.
Results
miR-424 expression in liver cancer
cell lines and the tissues of patients with HCC
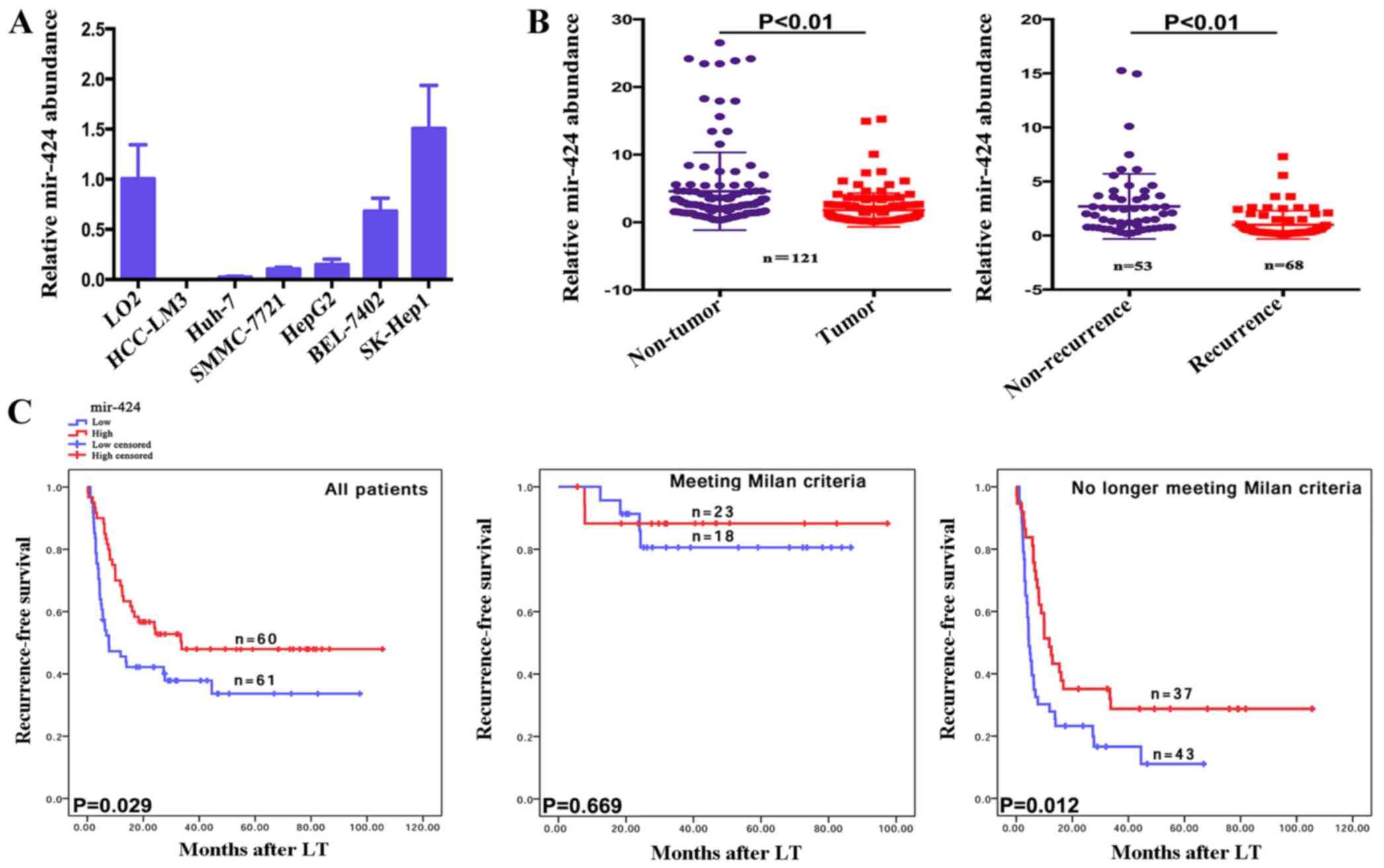
The expression levels of miR-424 in a human normal
liver cell line, LO2, and six liver cancer cell lines were measured
using RT-qPCR. Of the 6 liver cancer cell lines, 5 (HCC-LM3, Huh-7,
SMMC-7721, HepG2 and BEL-7402) expressed lower levels of miR-424
compared with the normal liver cell line (Fig. 1A). Additionally, the relative
expression levels of miR-424 in tumors and adjacent non-tumorous
tissues from 121 patients with HCC were measured. The tumors
expressed significantly lower levels of miR-424 compared with the
adjacent non-tumorous tissues. Furthermore, recurrent tumors
expressed lower miR-424 levels compared with non-recurrent tumors
(P<0.01; Fig. 1B).
Low mir-424 expression is associated
with early tumor recurrence in patients with HCC following LT
To explore the utility of miR-424 as a promising
molecular marker for predicting the prognosis of patients with HCC,
the recurrence-free survival times between 121 patients with HCC
who expressed high or low expression levels of mir-424 were
compared, based on extensive clinical follow-up data. Firstly,
mir-424 expression did not correlate with any, single
clinicopathological characteristic in the 121 patients, including
the age, sex, portal vein tumor thrombi, AFP, histopathological
grade, tumor size or tumor number, when stratified by expression
level (high or low) (Table I).
Furthermore, the univariate recurrence-free survival analysis
demonstrated that the tumor size, preoperative AFP level, PPTV,
Milan criteria (11) and mir-424
expression serve as risk factors for tumor recurrence following LT
(Table II). In addition, COX
multivariate analysis revealed that for patients who no longer met
the Milan criteria, a tumor size of >5 cm and high mir-424
expression levels were independent risk factors for tumor
recurrence following LT (Table
III).
 | Table II.Univariate analyses of predictors of
recurrence in patients with hepatocellular carcinoma following
liver transplantation. |
Table II.
Univariate analyses of predictors of
recurrence in patients with hepatocellular carcinoma following
liver transplantation.
|
| Tumor
recurrence |
|
|---|
|
|
|
|
|---|
| Variables | Negative | Positive |
P-valuea |
|---|
| Tumor size, cm |
| ≤5 | 45 | 25 | <0.001 |
|
>5 | 8 | 43 |
|
| Preoperative
α-fetoprotein level, ng/ml |
|
≤400 | 32 | 24 | 0.009 |
|
>400 | 21 | 44 |
|
| Portal vein tumor
thrombi |
|
Absent | 44 | 36 | <0.001 |
|
Present | 9 | 32 |
|
| Milan
criteriab |
| Within
criteria | 30 | 11 | <0.001 |
| Beyond
criteria | 12 | 68 |
|
| miR-424
expression |
|
Low | 34 | 7 | <0.001 |
|
High | 19 | 61 |
|
| Meeting Milan
criteria, miR-424 expression |
|
Low | 15 | 3 | 0.669 |
|
High | 19 | 4 |
|
| No longer meeting
Milan criteria, miR-424 expression |
|
Low | 8 | 35 | 0.012 |
|
High | 11 | 26 |
|
 | Table III.microRNA-424 expression in
hepatocellular carcinoma is an independent predictive factor for
recurrence in patients following liver transplantation. |
Table III.
microRNA-424 expression in
hepatocellular carcinoma is an independent predictive factor for
recurrence in patients following liver transplantation.
|
| Cumulative
recurrence |
|---|
|
|
|
|---|
| Variables | Hazard ratio (95%
CI) |
P-valuea |
|---|
| Milan criteria |
| No
longer meeting vs. meeting | 3.743
(1.439–9.738) | 0.007 |
| Tumor size, cm |
| >5
vs. ≤5 | 2.158
(1.205–3.864) | 0.010 |
| microRNA-424
expression |
| High
vs. low | 0.593
(0.363–0.969) | 0.037 |
The overexpression of miR-424 predicted earlier
recurrence times in patients with HCC who underwent LT (P=0.029;
Fig. 1C). The results of the present
study revealed that the Milan criteria effectively predicted the
risk of tumor recurrence [Table
III; no longer meeting vs. meeting, hazard ratio (HR)=3.743;
P=0.007] in patients following LT, which is consistent with
previous studies (16). Notably, in
patients who met the Milan criteria, additional classification
based on miR-424 expression did not result in significant
differences in the recurrence-free survival times (P=0.669;
Fig. 1C). However, patients who no
longer met the Milan criteria and who exhibited increased miR-424
expression levels experienced earlier tumor recurrence following LT
(P=0.012, Fig. 1C). Taken together,
these clinical data demonstrate that miR-424 expression is an
important indicator for predicting tumor recurrence in patients
with HCC following LT (P=0.029), which may also be valid in
patients who no longer meet the Milan criteria (P=0.012).
Effects of miR-424 expression on the
proliferation, migration and invasiveness of HCC cells
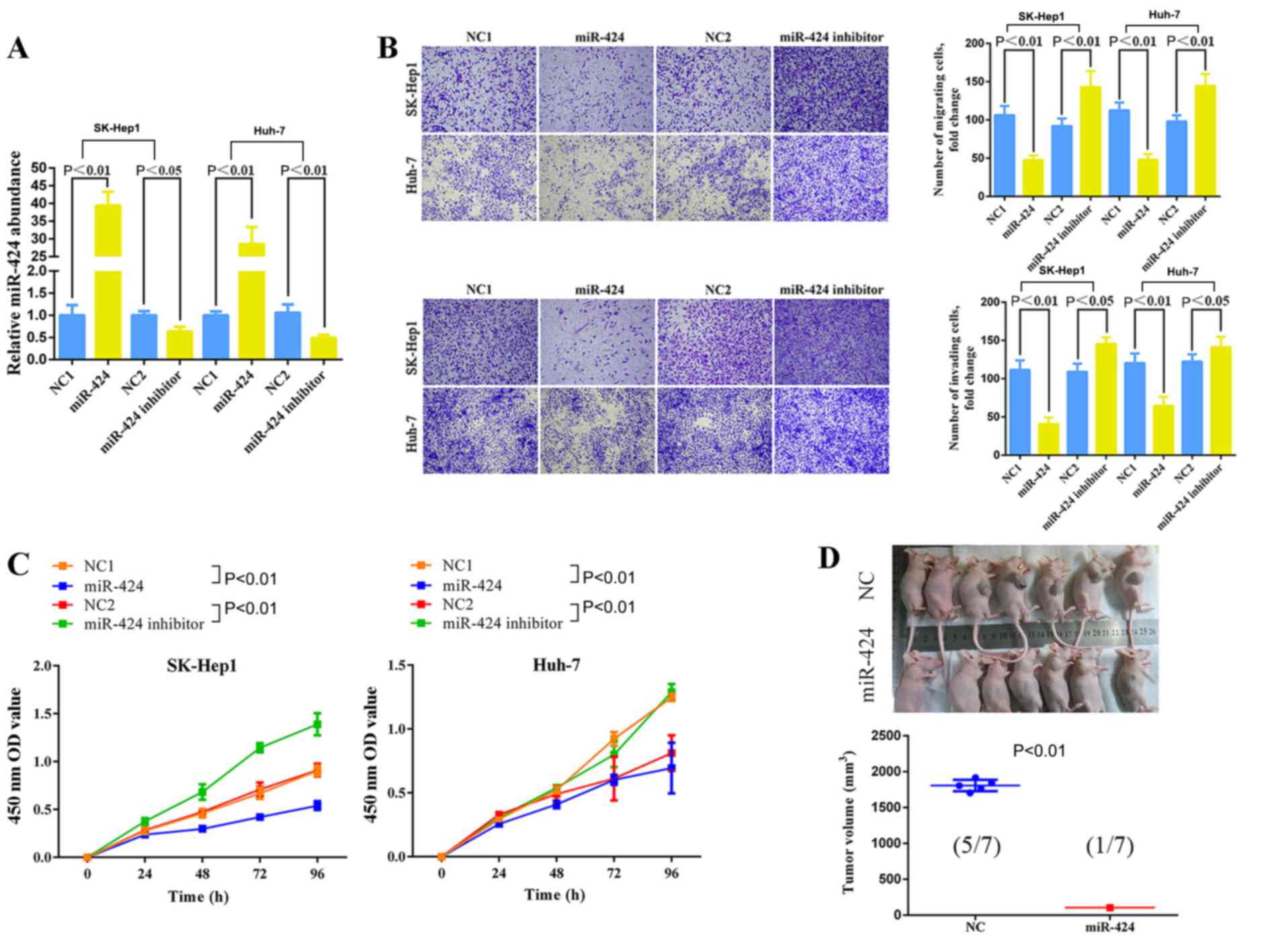
SK-HEP-1 and Huh-7 cells were treated with an
miR-424 mimic or miR-424 inhibitor. Consequently, miR-424
expression was effectively upregulated or downregulated (Fig. 2A). The Transwell assay, CCK-8 assay
and an HCC xenograft model demonstrated that the upregulation of
miR-424 expression significantly reduced the migration,
invasiveness (Fig. 2B), proliferation
(Fig. 2C) of HCC cells, and also
inhibited the tumor volume in nude mice (Fig. 2D). The downregulation of miR-424 in
HCC cells significantly promoted the migration, invasion and
proliferation of hepatocellular carcinoma cells (Fig. 2B and C).
Discussion
LT is a potentially curative treatment for HCC.
Despite the development of diagnosis and treatment protocols for
HCC in previous decades, the prognosis of HCC remains poor. HCC
frequently recurs following surgery, which is primarily
attributable to the presence of microscopic extrahepatic metastatic
foci prior to LT (21). This
recurrence remains the major obstacle for improving the long-term
survival rates of patients with HCC (22).
The Milan criteria are frequently used in patient
selection for LT. However, the Milan criteria that ignore the
molecular biomarker expression patterns have been criticized for
being too restrictive, and patients with tumors beyond the Milan
criteria may also have a satisfactory prognosis (12). The present study considers the lack of
biological molecular markers to be a limitation of the Milan
criteria. Biological molecules that reflect tumor characteristics
or immunity status are valuable for addition to the LT criteria for
patients with HCC, which is recurrently evaluated at The First
Affiliated Hospital, Zhejiang University School of Medicine
(12–14,17).
The results of the present study revealed that in
patients who are beyond the Milan criteria, those with increased
miR-424 expression experienced a longer recurrence-free time.
Patients with HCC beyond the Milan criteria, who exhibited a high
miR-424 expression level in tumor tissues may be recommended as
candidates for LT, thus potentially significantly increasing the
number of patients with HCC who are eligible for LT. However, it
appears too difficult to use a single molecular marker to
efficiently diagnose HCC or predict recurrence (23,24), which
is also a limitation of the present study. However, the advance of
high-throughput sequencing technology would promote the
establishment of a diagnostic or prognostic model of multiple
molecular biomarkers. Future studies may also evaluate additional
molecular markers and patient samples to establish a prognostic
model of recurrence and mortality in patients with HCC following
LT, and explore improved prognostic criterion for LT in patients
with HCC. The present study, along with our previous studies
(14,17), provides valuable insight for the
establishment of such a model. A preoperative detection of miR-424
expression in liver biopsy specimens and circulating blood cells
should be performed in future studies.
Additionally, the biological role of miR-424 in HCC
cell proliferation and invasion was explored with in vitro
assays. Knockdown of miR-424 promoted the invasion and
proliferation of the HCC cell lines SK-HEP-1 and Huh-7.
Upregulation of miR-424 significantly reduced the invasive
capability and suppressed the growth of HCC cells in vitro
and in vivo. The results of the functional assays conducted
in the present study are consistent with those of prior studies
(15), and theoretically support the
clinical data concerning the role of miR-424 in predicting the
prognosis of patients with HCC following LT. However, the precise
mechanisms of miR-424-induced HCC cell invasion and proliferation
regulation remain unclear.
In conclusion, the present study presents miR-424 as
a valuable biomarker for predicting recurrence in patients with HCC
following LT. These results are important for establishing
additional molecular markers that may be used in a prognostic model
for recurrence and mortality in patients with HCC following LT. The
limitation of the present study is that additional exploration is
required to determine the exact mechanisms by which miR-424
regulates malignant biological behavior in HCC cells.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Natural
Science Foundation of Zhejiang Province (grant no. LY15H160021),
the National Natural Science Foundation (grant nos. 81372425;
81572954), and the Foundation for Innovative Research Groups of the
National Natural Science Foundation of China (grant no.
81421062).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LW, LZ and SZ designed the study and wrote the
manuscript. FY, BL and XC collected and analyzed the data. SY, HX
and FZ performed the cell assays and animal experiments.
Ethics approval and consent to
participate
The present study was approved by the Medical Ethics
Committee of The First Affiliated Hospital of Zhejiang University,
and written informed consent was obtained from all patients.
Consent for publication
Written informed consent was obtained from all
patients.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
El-Serag HB and Rudolph KL: Hepatocellular
carcinoma: Epidemiology and molecular carcinogenesis.
Gastroenterology. 132:2557–2576. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dutkowski P, Linecker M, DeOliveira ML,
Mullhaupt B and Clavien PA: Challenges to liver transplantation and
strategies to improve outcomes. Gastroenterology. 148:307–323.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ramesh H: Resection for hepatocellular
carcinoma. J Clin Exp Hepatol. 4 Suppl 3:S90–S96. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Arzumanyan A, Reis HM and Feitelson MA:
Pathogenic mechanisms in HBV- and HCV-associated hepatocellular
carcinoma. Nat Rev Cancer. 13:123–135. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ambros V: The functions of animal
microRNAs. Nature. 431:350–355. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ivkovic Catela T, Voss G, Cornella H and
Ceder Y: microRNAs as cancer therapeutics: A step closer to
clinical application. Cancer Lett. 407:113–122. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hayes J, Peruzzi PP and Lawler S:
microRNAs in cancer: Biomarkers, functions and therapy. Trends Mol
Med. 20:460–469. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mazzaferro V, Regalia E, Doci R, Andreola
S, Pulvirenti A, Bozzetti F, Montalto F, Ammatuna M, Morabito A and
Gennari L: Liver transplantation for the treatment of small
hepatocellular carcinomas in patients with cirrhosis. N Engl J Med.
334:693–699. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xu X, Lu D, Ling Q, Wei X, Wu J, Zhou L,
Yan S, Wu L, Geng L, Ke Q, et al: Liver transplantation for
hepatocellular carcinoma beyond the Milan criteria. Gut.
65:1035–1041. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zheng SS, Xu X, Wu J, Chen J, Wang WL,
Zhang M, Liang TB and Wu LM: Liver transplantation for
hepatocellular carcinoma: Hangzhou experiences. Transplantation.
85:1726–1732. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang Z, Zhou L, Wu LM, Lai MC, Xie HY,
Zhang F and Zheng SS: Overexpression of long non-coding RNA HOTAIR
predicts tumor recurrence in hepatocellular carcinoma patients
following liver transplantation. Ann Surg Oncol. 18:1243–1250.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yang H, Zheng W, Shuai X, Chang RM, Yu L,
Fang F and Yang LY: microRNA-424 inhibits Akt3/E2F3 axis and tumor
growth in hepatocellular carcinoma. Oncotarget. 6:27736–27750.
2015.PubMed/NCBI
|
|
16
|
Zhang Y, Li T, Guo P, Kang J, Wei Q, Jia
X, Zhao W, Huai W, Qiu Y, Sun L and Han L: miR-424-5p reversed
epithelial-mesenchymal transition of anchorage-independent HCC
cells by directly targeting ICAT and suppressed HCC progression.
Sci Rep. 4:62482014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang F, Wu LM, Zhou L, Chen QX, Xie HY,
Feng XW and Zheng SS: Predictive value of expression and promoter
hypermethylation of XAF1 in hepatitis B virus-associated
hepatocellular carcinoma treated with transplantation. Ann Surg
Oncol. 15:3494–3502. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu LM, Zhang F, Xie HY, Xu X, Chen QX, Yin
SY, Liu XC, Zhou L, Xu XB, Sun YL and Zheng SS: MMP2 promoter
polymorphism (C-1306T) and risk of recurrence in patients with
hepatocellular carcinoma after transplantation. Clin Genet.
73:273–278. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lai MC, Yang Z, Zhou L, Zhu QQ, Xie HY,
Zhang F, Wu LM, Chen LM and Zheng SS: Long non-coding RNA MALAT-1
overexpression predicts tumor recurrence of hepatocellular
carcinoma after liver transplantation. Med Oncol. 29:1810–1816.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kaiser J: The cancer stem cell gamble.
Science. 347:226–229. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zimmerman MA, Ghobrial RM, Tong MJ, Hiatt
JR, Cameron AM, Hong J and Busuttil RW: Recurrence of
hepatocellular carcinoma following liver transplantation: A review
of preoperative and postoperative prognostic indicators. Arch Surg.
143:182–188. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Schutte K, Schulz C, Link A and
Malfertheiner P: Current biomarkers for hepatocellular carcinoma:
Surveillance, diagnosis and prediction of prognosis. World J
Hepatol. 7:139–149. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kim JH, Sohn BH, Lee HS, Kim SB, Yoo JE,
Park YY, Jeong W, Lee SS, Park ES and Kaseb A: Genomic predictors
for recurrence patterns of hepatocellular carcinoma: Model
derivation and validation. PLoS Med. 11:e10017702014. View Article : Google Scholar : PubMed/NCBI
|