Introduction
Prostate cancer (PCa) is the most commonly diagnosed
malignancy and causes over 30,000 fatalities each year in men in
the USA (1). Advanced PCa is
routinely treated by androgen deprivation therapy (ADT); however,
it can eventually progress to castration-resistant prostate cancer
(CRPC) (2). Recently developed ADT
drugs including enzalutamide and abiraterone acetate can improve
survival (3), but the overall
prognosis of patients with CRPC remains poor (4). Therefore, more effective and less toxic
agents are required for CRPC therapy.
Epithelial-to-mesenchymal transition (EMT) is a
tightly regulated biological process in which epithelial cells
acquire mesenchymal characteristics and upregulate the expression
of mesenchymal markers, and is associated with malignant
progression (5). Cancer cells that
have undergone EMT via activation of the nuclear factor (NF)-κB,
Wnt and Hedgehog signaling pathways acquire stem cell-like traits,
including the capacity for self-renewal and differentiation
(6,7).
Cancer stem cells (CSCs) form spheres in non-adherent cultures and
express Nanog, Octamer-binding protein 4 (OCT4) and cluster of
differentiation 44 (CD44) (8). An
EMT-like state and CSC features are associated with metastasis and
resistance to ADT (9,10), and are thus ideal therapeutic targets
for CRPC treatment.
Ethyl pyruvate (EP) is a stable aliphatic ester
derived from pyruvic acid that has been demonstrated to suppress
inflammation, mitigate redox-mediated cellular damage, and exert
immunoregulatory and neuroprotective effects (11,12). It
also has anti-tumor effects in many types of cancer, including
gallbladder cancer, lung adenocarcinoma and hepatic tumors, among
others (13–18). However, the molecular basis for these
effects is not well understood and, to the best of our knowledge,
they have not been investigated in the context of PCa.
To address these issues, in the current study the
antitumor effects of EP were investigated in PCa cells lines and a
mouse xenograft model. The results indicated that EP may block
tumor growth, migration and EMT, and stimulate apoptosis, and may
thus be a promising treatment modality for PCa.
Materials and methods
Cell lines
The human PCa cell lines PC3 and CWR22RV1 were
purchased from the Cell Bank of the Chinese Academy of Sciences
(Shanghai, China). The cells were cultured as previously described
(19).
Reagents and materials
The following antibodies were used in the present
study: Rabbit polyclonal anti-E-cadherin (cat. no. 0874-1-AP,
1:500), -β-actin (cat. no. 60008-1-Ig), -tubulin (cat. no.
11224-1-AP), -vimentin (cat. no. 10366-1-AP) and -p21 (cat. no.
10355-1-AP) (1:1,000; all from ProteinTech, Rosemont, IL, USA);
rabbit anti-cyclin D1 (cat. no. 2978T), -cyclin-dependent kinase
(CDK)4 (cat. no. 12790T), -poly (ADP-ribose) polymerase (PARP)
(cat. no. 9532T), -AKT (cat. no. 4691T) and -p65 (cat. no. 8242S)
(all 1:1,000); rabbit anti-caspase 3 (cat. no. 9662S), -cleaved
caspase-3 (9664T), -cleaved PARP (cat. no. 5625T), -phosphorylated
(p)-AKT (SER473) (cat. no. 4060T) and -p-p65 (cat. no. 3033T) (all
1:500; all from Cell Signaling Technology, Inc., Danvers, MA, USA);
and mouse anti-histone-3 (cat. no. ab1791, 1:1,000; Abcam,
Cambridge, UK). EP and human recombinant tumor necrosis factor
(TNF)-α were obtained from Shanghai Macklin Biochemical Co., Ltd.,
(Shanghai, China) and Sigma-Aldrich; Merck KGaA, (Darmstadt,
Germany), respectively.
Cell viability test
PC3 and CWR22RV1 cells (5×103/well) were
seeded in 96-well plates and grown until they had adhered to the
plate. They were then treated with 5, 10, 15 or 20 mM EP for 24, 48
or 72 h. Cell viability was evaluated with a Cell Counting Kit
(CCK)-8 (Dojindo Molecular Technologies, Inc., Kumamoto, Japan)
according to the manufacturer's instructions. Briefly, cells were
incubated for 2 h in the CCK-8 solution (10 µl/per well) and the
absorbance was read at 490 nm.
Colony formation assay
The effect of EP on PCa cell growth was evaluated
with a colony formation assay. PC3 cells (500/well) and CWR22RV1
cells (800/well) were seeded in a 6-well plate (Corning Inc.,
Corning, NY, USA) and cultured for 2 weeks. The cells were fixed
with methanol and stained with Giemsa. Colonies containing at least
50 cells were counted.
Wound healing assay
PCa cells (1.5×106/per well) were seeded
in a 6-well plate and incubated overnight until they reached 75–90%
confluence. The cell monolayer was scratched with a 10-µl plastic
pipette tip. After washing twice with PBS to remove cell debris,
Roswell Park Memorial Institute 1640 medium containing 1% fetal
bovine serum (FBS) and 10 or 15 mM EP or PBS was added. After 24 h,
the cells were photographed and the migrated distance was measured
with ImageJ software (National Institutes of Health, Bethesda, MD,
USA).
Transwell invasion assay
Cells were resuspended in serum-free 1640 medium
containing 10 or 15 mM EP or PBS. Cells (5×104/200 µl)
were added to the upper compartment of a Transwell chamber
containing Matrigel. The lower compartment was filled with 600 µl
medium containing 20% FBS. After incubation for 12 h, a cotton swab
was used to wipe the top chamber and remove the non-migratory
cells, and migrated cells attached to the bottom side of the
chamber were fixed with methanol and stained with Giemsa.
Cell cycle analysis
A Cell Cycle Staining kit (Hangzhou MultiSciences
(Lianke) Biotech, Co., Ltd., Shanghai, China) was used to evaluate
cell cycle distribution according to the manufacturer's
instructions. Briefly, cells were incubated with 15 mM EP for 48 h,
dissociated by trypsinization (0.25% trypin + 0.02% EDTA), and
concentrated by centrifugation (2,000 rpm, 5 min). Following
washing with PBS, the cells were resuspended in 1 ml DNA staining
solution and 10 µl permeabilization solution, then incubated at
room temperature in the dark for 30 min. DNA content was determined
by flow cytometry on a FACSCalibur instrument (BD Biosciences,
Franklin Lakes, NJ, USA).
Sphere formation assay
The stemness potential of EP-treated PCa cells was
evaluated with a sphere formation assay as previously described
(20).
Nuclear extract preparation
PC3 and CWR22RV1 cells were pretreated with 15 mM EP
for 6 h and then stimulated with 10 ng/ml TNF-α for 2 h. Total cell
lysate was separated into cytoplasmic and nuclear components using
a Nuclear-Cytosol Extraction kit (Nanjing KeyGen Biotech Co., Ltd.,
Nanjing, China).
Western blot analysis
The total cell lysate of PCa cells treated with 15
mM EP was analyzed by sodium dodecyl sulfate polyacrylamide gel
electrophoresis (SDS-PAGE) on a 4–12% gel prepared with an SDS-PAGE
Gel Preparation kit (Nanjing KeyGen Biotech Co., Ltd.), with 30 µg
protein loaded in each well. The proteins were transferred to a
0.45-µm polyvinylidene difluoride membrane that was probed with the
indicated antibodies.
Evaluation of the antitumor potential
of EP in vivo
Male BALB/C nu/nu mice (6 weeks old) were purchased
from the Laboratory Animal Center of Southern Medical University. A
total of 5×106 PC3 cells were resuspended in PBS and
injected into the right upper limb of each mouse. When subcutaneous
tumors had formed after approximately 7 days, 10 mice were randomly
allocated into control or treatment groups and intraperitoneally
injected once daily for 2 weeks with 50 mg/kg EP or saline,
respectively. Procedures involving animals were approved by the
Institutional Animal Care and Use Committee of Southern Medical
University.
Terminal deoxynucleotidyl transferase
dUTP nick end labeling (TUNEL) assays
Formalin-fixed paraffin-embedded Tumor tissues from
the saline-treated group and the EP-treated groups were analyzed
with TUNEL assays. At first, Tissue slice were deparaffinized with
xylene and dehydrated with ethanol. And then, the slices were
rinsed twice with PBS and treated with proteinase K (15 µg/ml in 10
mM Tris/HCl, pH 7.4–8.0) for 15min-20 min at 37°C. Endogenous
peroxidases were blocked with 3% hydrogen peroxide in methanol at
room temperature for 10 min. The tissue sections were then analyzed
with an in situ Cell Death Detection kit, POD (Nanjing
KeyGen Biotech Co., Ltd.,), in accordance with the manufacturer's
instructions. The reaction was visualized with fluorescence
microscopy.
Statistical analysis
Data were statistically calculated using two-tailed
Student's t-tests (two groups) or one-way ANOVA followed by the
least significant difference post-hoc test (for more than two
groups). P<0.05 was considered to indicate a statistically
significant difference. Value are presented as the means ± standard
deviation (SD) by GraphPad Prism software (GraphPad Software, Inc.,
La Jolla, CA, USA).
Results
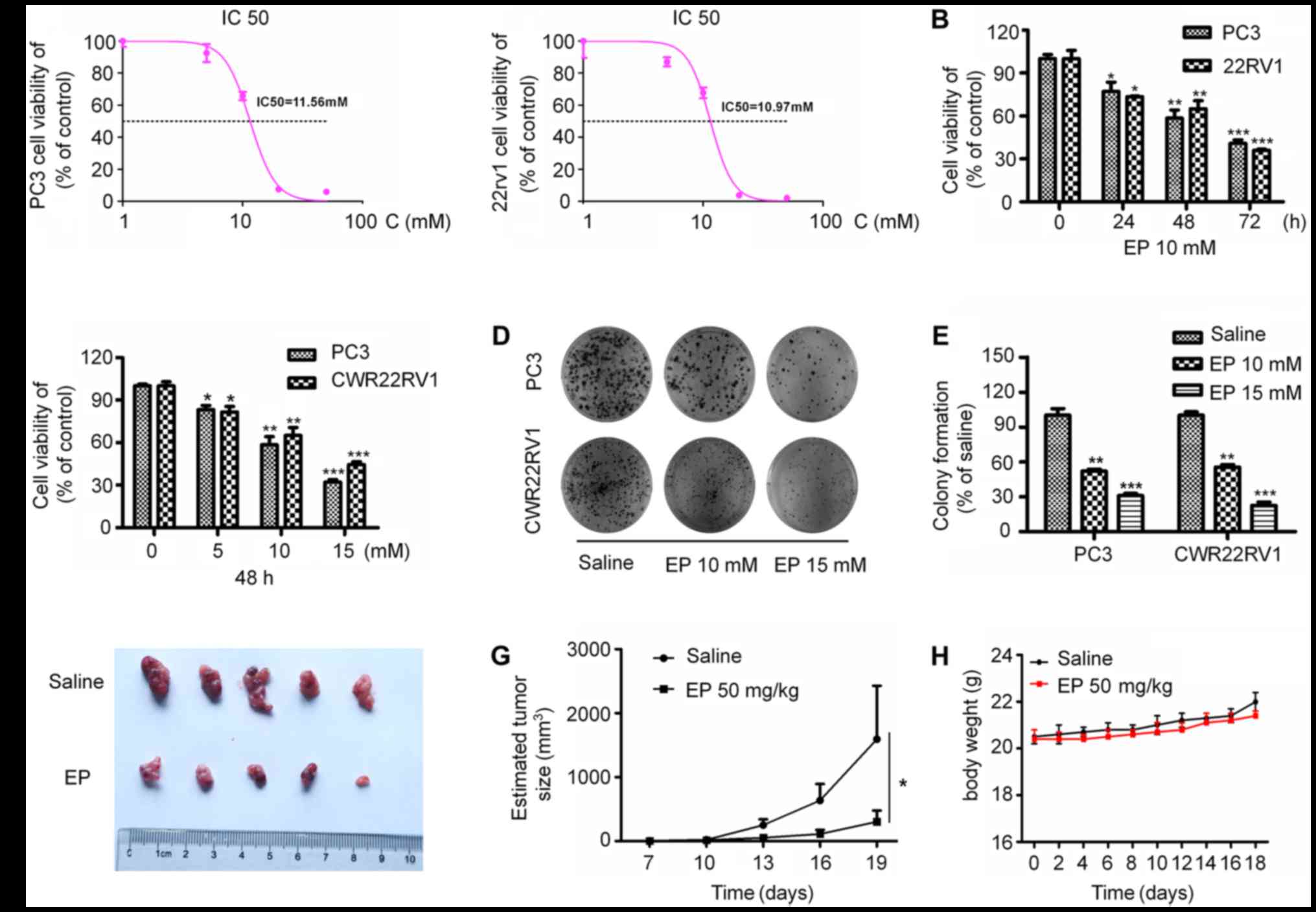
EP inhibits the proliferation of human
PCa cells in vitro and abolishes the tumor-forming capacity of PCa
cells in vivo
To investigate the effects of EP on the
proliferation of PCa cells, PC3 and CWR22RV1 cells were treated
with different concentrations of EP for various times. EP show
cytotoxic potential against the PC3 and 22Rv1 with IC50 values of
11.56 and 10.93 mM, respectively (Fig.
1A). EP treatment suppressed the proliferation of both cell
lines in a concentration- and time-dependent manner (P<0.05;
Fig. 1B and C). Furthermore, EP
markedly inhibited the colony formation capacities of the cells
(P<0.05; Fig. 1D and E). It was
next investigated whether EP can suppress tumor growth in
vivo. PC3 cells were injected into the upper limbs of male nude
mice, with EP administered after tumors had formed. Treatment with
50 mg/kg EP for 2 weeks significantly reduced tumor growth relative
to vehicle-treated controls, resulting in smaller tumors
(P<0.05; Fig. 1F and G) by the end
of the experimental period (average volume: 303.4±176.6 vs.
1,594.6±834.7 mm3). Correspondingly, the EP treatment
was well tolerated and did not result in any significant in
vivo toxicity (Fig. 1H).
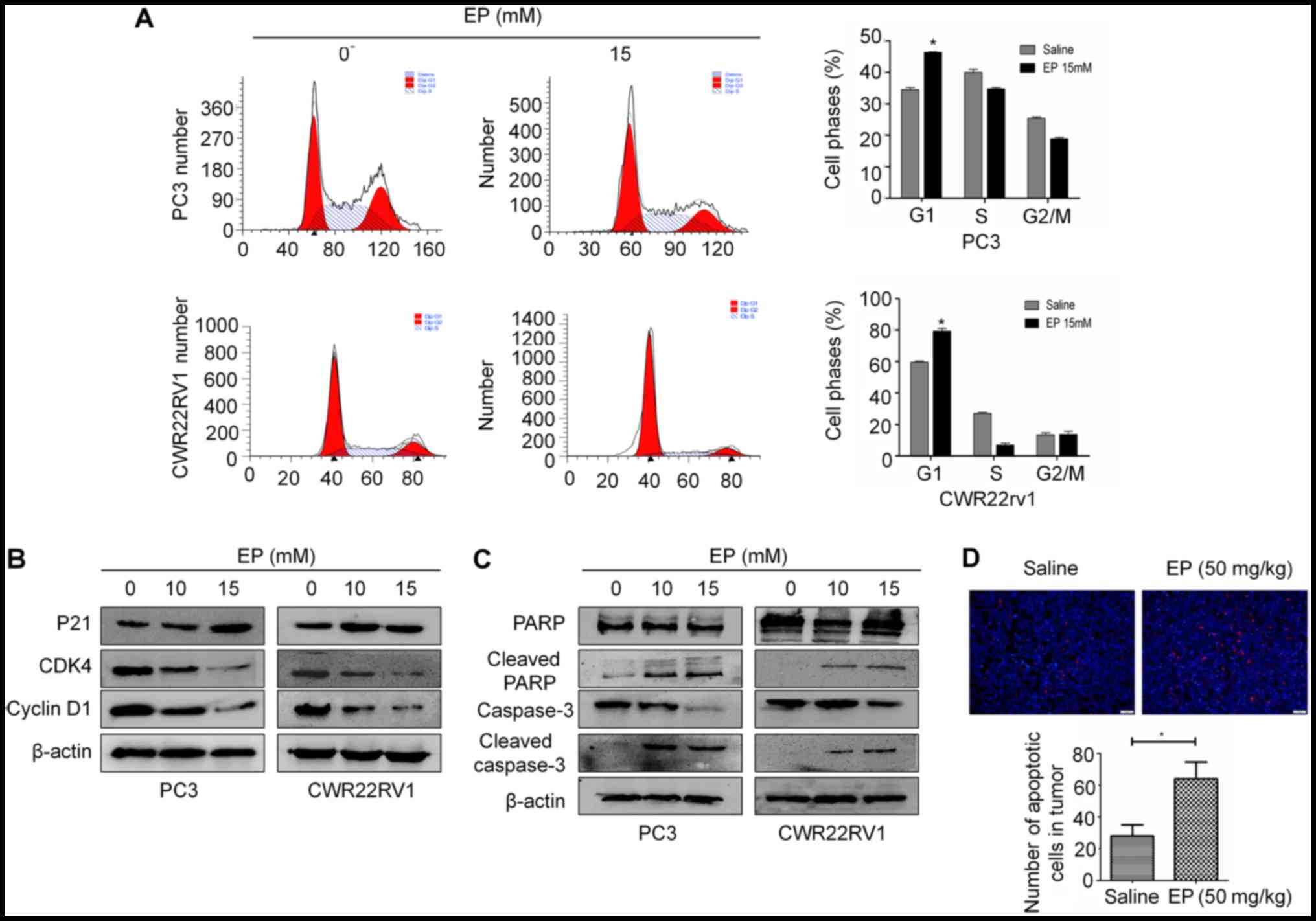
EP induces G1 arrest and
apoptosis
To investigate whether the anti-proliferative
effects of EP on PCa cells were associated with changes in cell
cycle progression, PC3 and CWR22RV1 cells were treated with 15 mM
EP for 48 h and cell cycle distribution was analyzed by flow
cytometry. The G1 fraction was markedly increased following
treatment with EP relative to the negative control group (Fig. 2A). Similar results were obtained in
CWR22RV1 cells (Fig. 2A). A western
blot analysis revealed that the treatment with EP decreased the
expression of cyclin D1 and CDK4 and increased that of p21
(Fig. 2B).
EP has been established to induce apoptosis in many
cancer cell lines (15,18). The levels of caspase-3 and PARP were
therefore examined in PCa cells by western blotting. The cleaved
forms of PARP and caspase-3 were increased in both CWR22RV1 and PC3
cells following treatment with 10 or 15 mM EP for 48 h (Fig. 2C). In vivo, TUNEL staining also
demonstrated an increase in tumor cell apoptosis after treated with
EP (P<0.05; Fig. 2D).
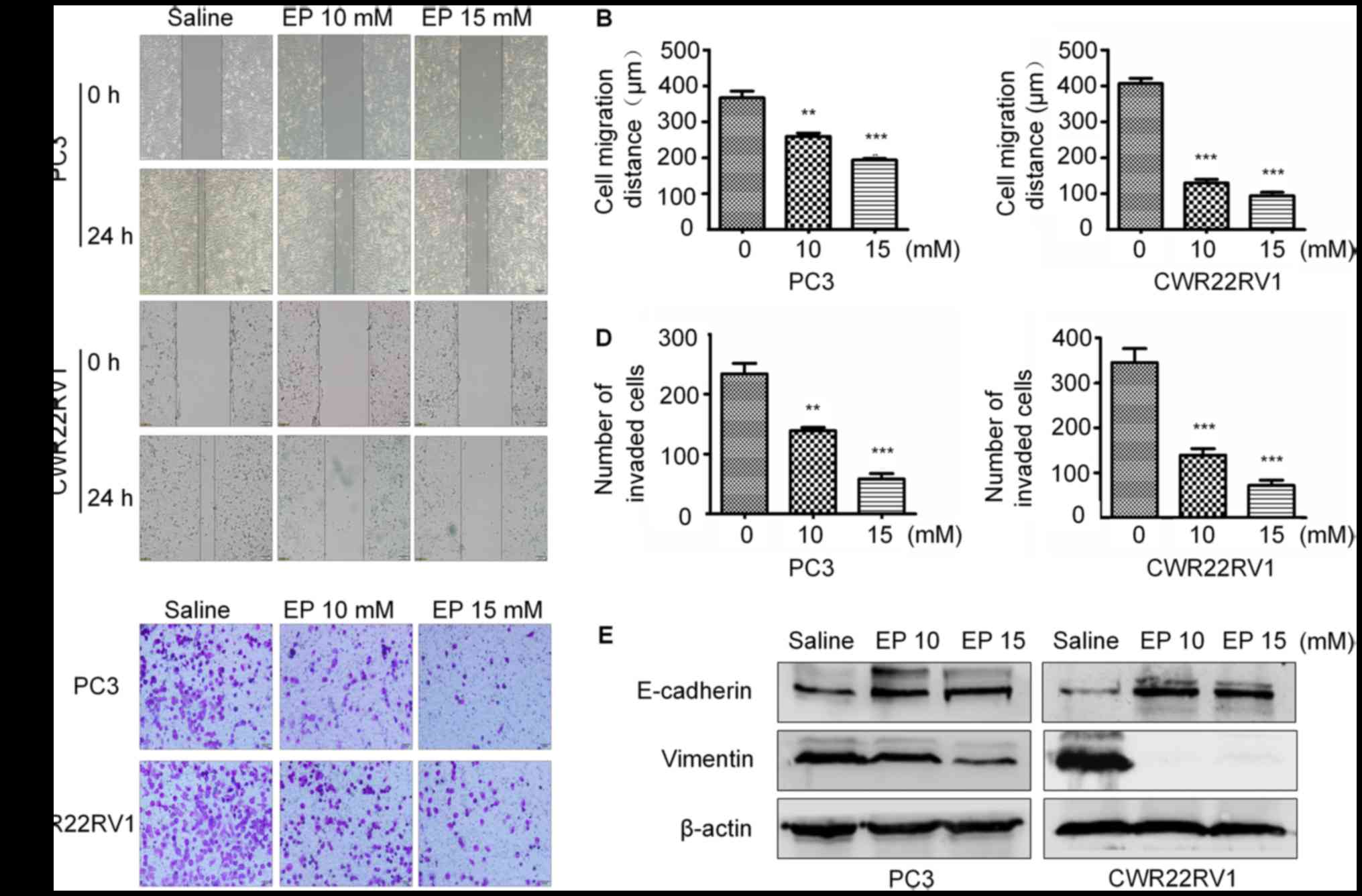
EP inhibits the migration and
suppresses the metastatic potential of PCa cells
To determine whether EP inhibits PCa cell motility,
a wound-healing assay was performed. EP reduced the migratory
capacities of both CWR22RV1 and PC3 cells in a
concentration-dependent manner (Fig. 3A
and B). Furthermore, the results of a Transwell invasion assay
revealed that there were fewer invading cells in the treatment
group as compared with the control group (Fig. 3C and D).
EMT serves a key role in cancer metastasis and
invasion (21,22). The expression of epithelial markers
including E-cadherin and mesenchymal markers including vimentin
were therefore examined in PC3 and CWR22RV1 cells treated with EP.
The level of E-cadherin was upregulated, whereas that of vimentin
was downregulated, in the treatment group relative to the control,
suggesting that EP prevents metastasis and invasion by blocking EMT
(Fig. 3E).
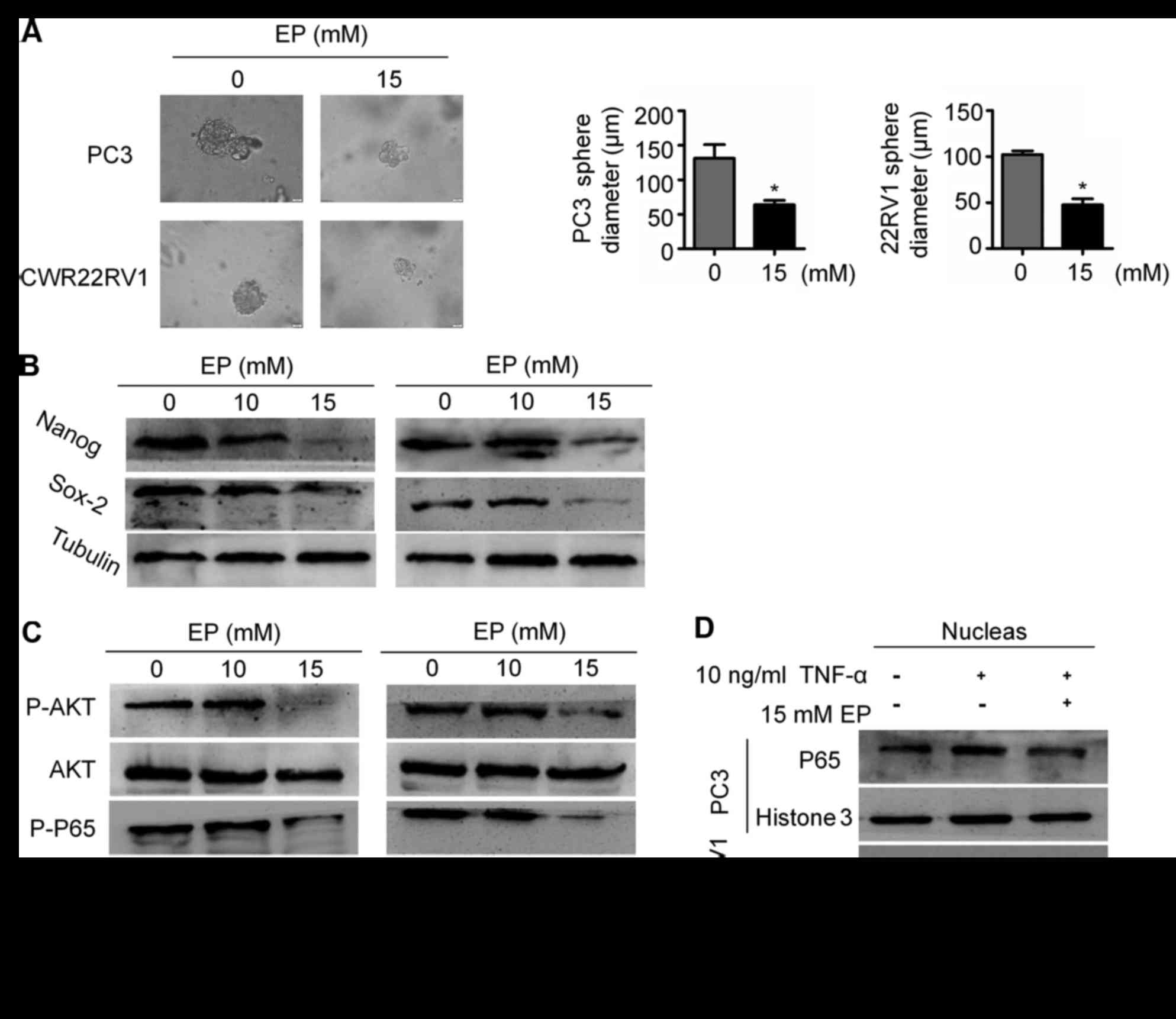
EP suppresses CSC stemness and
AKT/NF-κB signaling
Given that EMT is associated with stemness, the
effects of EP on CSCs were examined. The results of a sphere
formation assay demonstrated that EP suppressed the stem cell
characteristics of PC3 and CWR22RV1 cells: The diameter of tumor
spheres was smaller in the EP treatment group than in the control
group (P<0.05; Fig. 4A), which was
associated with the downregulation of the stemness markers Nanog
and (sex determining region Y)-box 2 (Sox2; Fig. 4B).
Aberrant activation of NF-κB signaling in PCa has
been associated with metastatic progression (23,24). NF-κB
signaling serves an important role in EMT and the maintenance of
CSC stemness (25,26). It was observed here that EP treatment
reduced p65 phosphorylation (Fig.
4C), which is required for NF-κB/p65 nuclear translocation.
Accordingly, EP blocked NF-κB nuclear accumulation induced by TNF-α
(Fig. 4D). To clarify the mechanism
underlying the inhibition of NF-κB signaling by EP, AKT expression
and phosphorylation was evaluated by western blotting. EP treatment
decreased the levels of p-AKT in both PC3 and CWR22RV1 cells
(Fig. 4C). These results suggest that
EP suppresses EMT in part by targeting the AKT/NF-κB pathway.
Discussion
The antitumor effects of EP have been reported;
however, they have not been previously investigated in the context
of PCa. The results of the present study indicate that EP inhibits
the proliferation of PCa cells by inducing cell cycle arrest and
apoptosis and suppressing tumor growth in vivo, consistent
with previous reports on gallbladder cancer cells and
hepatocellular carcinoma (16,18).
Additionally, it was observed that EP targets EMT and CSCs through
negative regulation of AKT/NF-κB signaling.
The loss of epithelial phenotypes such as spindle
morphology and intercellular adhesion, and the acquisition of
mesenchymal characteristics including high migration and invasion
capacities and lower cell-extracellular adhesion are two major
events that occur when epithelial cells undergo EMT (7). Studies demonstrate that EMT is
associated with CRPC (27,28). Song et al (29) identified that castration may cause
EMT, evidenced by decreased expression of epithelial markers
(including E-cadherin) and increased protein levels of mesenchymal
markers (including N-cadherin, Slug, Zeb1 and Twist1), in both
human LuCaP35 prostate cancer xenograft tumors and normal mouse
prostate tissue following androgen deprivation, And similar changes
have also been identified in human samples undergoing ADT (29). EMT is driven by EMT-inducing
transcription factors (including Snail, Slug, Zeb1, Zeb2 and
Twist), some of which have been reported to participate in the
development of CRPC (7). Zeb1
expression in CRPC is higher than in androgen-sensitive prostate
cancer and may be a reliable indicator of poor prognosis in
patients (30). In addition, stable
overexpression of Twist in a prostate cancer cell line served a
fundamental role in the formation and progression of CRPC by
mediating EMT and CSCs (27). Shiota
et al (31) reported that
castration-induced oxidative stress may promote androgen receptor
(AR) overexpression through Twist1 overexpression, which could
result in a gain of castration resistance. Furthermore,
facilitation of castration resistance by Slug in prostate cancer
has been reported by Wu et al (32). Slug, another transcription factor
driver of EMT, can not only augment the expression of AR but also
enhance AR transcriptional activities with or without androgen as a
novel coactivator for AR (32).
Self-renewal and differentiation into diverse tumor cells are
unique characteristics of CSCs, which have been hypothesized to be
a subpopulation of tumor cells that drive treatment resistance in
prostate cancer due to growth independence from androgen
stimulation (33). CSC biomarkers,
including Nanog, Sox2, Nkx3.1, PSA-/LO and Bmi-1, have been applied
to identify and isolate CSCs in solid tumors including prostate
cancer (34). A number of studies
have observed that these biomarkers associated with CSCs may serve
a central role in the progression of CRPC (35). Overexpression of Nanog facilitates the
self-renewal and tumor sphere-formation ability of prostate cancer
cells and promotes castration-resistant tumor growth in an
androgen-deprived environment (36,37).
Furthermore, Rybak et al (38)
demonstrated that Sox2 is critically associated with the
self-renewal and tumor progression of human prostate cancer. In
addition, Sox2 has also been demonstrated as a factor associated
with castration-resistant tumor growth and to be negatively
regulated by the AR signaling axis (39). Overall, these aforementioned studies
suggest that both EMT and CSCs are responsible for progression and
treatment resistance in prostate cancer. Accordingly, treatment
capable of reversing EMT phenotypes or suppressing CSCs may become
a viable alternative in the future for CRPC therapy. In the current
study, it was indicated that EP effectively reversed the EMT
phenotype in the CRPC cell lines 22RV1 and PC3, as evidenced by
decreased expression of vimentin and an increased protein level of
E-cadherin. Furthermore, it was demonstrated that the ability of
tumor-sphere formation in CRPC cells was suppressed following EP
treatment. Notably, the biomarkers of CSCs, including Nanog and
Sox2, decreased in CRPC cells following EP treatment, which
indicates that EP may suppress the CSC subpopulation in prostate
cancer. These results indicate that EP may be an alternative
therapy for future CRPC treatment by targeting the EMT phenotype
and CSCs.
The NF-κB family, an important class of
transcriptional regulators, comprises five members including RelA
(p65), RelB, c-Rel, p50/p105 (NF-κB1) and p52/p100 (NF-κB2). NF-κB
binds to inhibitor κB (IκB) protein in the cytoplasm in an inactive
state. In a pathological state, the IκB kinase (IKK) complex is
activated and subsequently induces the phosphorylation of IκB,
which leads to the degradation of IκB and NF-κB translocation to
nucleus (40). Increasing results
indicate that the NF-κB transcription factor family is a crucial
mediator of EMT and CSCs (25,26).
Certain studies have demonstrated that NF-κB may bind to promoters
of genes associated with EMT and CSCs, including Snail, Slug and
Twist, to increase transcription (25,41). Ozes
et al (42) reported that AKT
was involved in the activation of NF-κB by mediating the
phosphorylation of IKKA which is responsible for the activation of
its downstream target IκB. Certain reports indicate that EP
inhibits the phosphorylation of AKT (18) and suppresses the DNA-binding activity
of NF-κB via direct modification of p65 at Cys 38 (43). In the current study, it was observed
that EP could inhibit the phosphorylation of both AKT and p65; in
addition, nuclear translocation of p65 induced by TNF-α was blocked
by EP. These results indicate that EP suppresses EMT and CSCs by
negatively regulating the AKT/NF-κB pathway.
Furthermore, the AKT/NF-κB signaling pathway might
drive the progression of CRPC by another mechanism besides the
induction of EMT or CSCs. Activation of NF-κB mediated by PI3K/AKT
increases the expression of AR via NF-κB binding to the AR promoter
(44). CRPC, previously defined as
hormone-refractory prostate cancer, remains to be considered as
androgen-dependent (45), which
indicates that targeting AR remains an effective therapeutic
strategy for CRPC. The present data demonstrated that EP suppressed
the phosphorylation of AKT and p65; however, whether the effect of
EP on AR signaling axis via regulation of the AKT/NF-κB pathway is
unclear and worthy of further examination.
A phase II multicenter double-blind
placebo-controlled study has demonstrated that EP administration in
patients undergoing higher-risk cardiac surgery has no side effects
(46). In addition, another study
indicated that EP exerted a potent effect on leukemia cells, while
being safe for normal blood cells (14). Therefore, taken together the available
data on EP suggest that it may be an effective and safe therapeutic
agent for the treatment of prostate cancer.
Acknowledgements
Not applicable.
Funding
This work was supported by the Science and
Technology Planning Projects of Guangdong Province (grant nos.
2013B051000050, 2014A020212538, 2014A020212663 and 2016A020215175);
Natural Science Foundation of Guangdong Province (grant nos.
2016A030313583 and 508312335027); Medical Scientific Research
Foundation of Guangdong Province (grant no. A2016555); Science and
Technology Planning Project of Guangzhou (grant no. 201704020070);
and Outstanding Youths Development Scheme of Nanfang Hospital,
Southern Medical University (grant no. 2015J005); National
undergraduate innovation training program funded projects of
Southern Medical University (grant no. 201612121005).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
This study was conceived and supervised by SCZ and
HQ. Experiments were carried out by BH and DJL. Data analysis was
conducted by CW and FPS. XLS provided technical support for western
blotting. ZCG, YZY, TX, JJX, SL and YML assisted with
experiments.
Ethics approval and consent to
participate
Procedures involving animals were approved by the
Institutional Animal Care and Use Committee of Southern Medical
University.
Pantient consent for publication
Not applicable.
Competing interests
All authors have declared that they have no
competing interests.
Glossary
Abbreviations
Abbreviations:
|
ADT
|
androgen-deprivation therapy
|
|
PCA
|
prostate cancer
|
|
CRPC
|
castration-resistant prostate
cancer
|
|
EMT
|
epithelial-mesenchymal transition
|
|
EP
|
ethyl pyruvate
|
|
TNF
|
tumor necrosis factor
|
|
CDK
|
cyclin-dependent kinase
|
|
PARP
|
poly(ADP-ribose) polymerase
|
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2018. CA Cancer J Clin. 68:7–30. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shafi AA, Yen AE and Weigel NL: Androgen
receptors in hormone-dependent and castration-resistant prostate
cancer. Pharmacol Ther. 140:223–238. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Loriot Y, Bianchini D, Ileana E, Sandhu S,
Patrikidou A, Pezaro C, Albiges L, Attard G, Fizazi K, De Bono JS
and Massard C: Antitumour activity of abiraterone acetate against
metastatic castration-resistant prostate cancer progressing after
docetaxel and enzalutamide (MDV3100). Ann Oncol. 24:1807–1812.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schrader AJ, Boegemann M, Ohlmann CH,
Schnoeller TJ, Krabbe LM, Hajili T, Jentzmik F, Stoeckle M,
Schrader M, Herrmann E and Cronauer MV: Enzalutamide in
castration-resistant prostate cancer patients progressing after
docetaxel and abiraterone. Eur Urol. 65:30–36. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chaffer CL, Juan San BP, Lim E and
Weinberg RA: Emt, cell plasticity and metastasis. Cancer Metastasis
Rev. 35:645–654. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang D, Plukker J and Coppes RP: Cancer
stem cells with increased metastatic potential as a therapeutic
target for esophageal cancer. Semin Cancer Biol. 44:60–66. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jaworska D, Król W and Szliszka E:
Prostate cancer stem cells: Research advances. Int J Mol Sci.
16:27433–27449. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bitting RL, Schaeffer D, Somarelli JA,
Garcia-Blanco MA and Armstrong AJ: The role of epithelial
plasticity in prostate cancer dissemination and treatment
resistance. Cancer Metastasis Rev. 33:441–468. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen X, Li Q, Liu X, Liu C, Liu R, Rycaj
K, Zhang D, Liu B, Jeter C, Calhoun-Davis T, et al: Defining a
population of stem-like human prostate cancer cells that can
generate and propagate castration-resistant prostate cancer. Clin
Cancer Res. 22:4505–4516. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen W, Lian J, Ye JJ, Mo QF, Qin J, Hong
GL, Chen LW, Zhi SC, Zhao GJ and Lu ZQ: Ethyl pyruvate reverses
development of Pseudomonas aeruginosa pneumonia during
sepsis-induced immunosuppression. Int Immunopharmacol. 52:61–69.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fink MP: Ethyl pyruvate: A novel
anti-inflammatory agent. J Intern Med. 261:349–362. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pellegrini L, Xue J, Larson D, Pastorino
S, Jube S, Forest KH, Saad-Jube ZS, Napolitano A, Pagano I, Negi
VS, et al: HMGB1 targeting by ethyl pyruvate suppresses malignant
phenotype of human mesothelioma. Oncotarget. 8:22649–22661. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Birkenmeier G, Hemdan NY, Kurz S, Bigl M,
Pieroh P, Debebe T, Buchold M, Thieme R, Wichmann G and Dehghani F:
Ethyl pyruvate combats human leukemia cells but spares normal blood
cells. PLoS One. 11:e01615712016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liang X, Chavez AR, Schapiro NE, Loughran
P, Thorne SH, Amoscato AA, Zeh HJ, Beer-Stolz D, Lotze MT and de
Vera ME: Ethyl pyruvate administration inhibits hepatic tumor
growth. J Leukoc Biol. 86:599–607. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li ML, Wang XF, Tan ZJ, Dong P, Gu J, Lu
JH, Wu XS, Zhang L, Ding QC, Wu WG, et al: Ethyl pyruvate
administration suppresses growth and invasion of gallbladder cancer
cells via downregulation of HMGB1-RAGE axis. Int J Immunopathol
Pharmacol. 25:955–965. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Park SY, Yi EY, Jung M, Lee YM and Kim YJ:
Ethyl pyruvate, an anti-inflammatory agent, inhibits tumor
angiogenesis through inhibition of the NF-κB signaling pathway.
Cancer Lett. 303:150–154. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cheng P, Dai W, Wang F, Lu J, Shen M, Chen
K, Li J, Zhang Y, Wang C, Yang J, et al: Ethyl pyruvate inhibits
proliferation and induces apoptosis of hepatocellular carcinoma via
regulation of the HMGB1-RAGE and AKT pathways. Biochem Biophys Res
Commun. 443:1162–1168. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lv D, Wu H, Xing R, Shu F, Lei B, Lei C,
Zhou X, Wan B, Yang Y, Zhong L, et al: HnRNP-L mediates bladder
cancer progression by inhibiting apoptotic signaling and enhancing
MAPK signaling pathways. Oncotarget. 8:13586–13599. 2017.PubMed/NCBI
|
|
20
|
Zhong D, Zhang HJ, Jiang YD, Wu P, Qi H,
Cai C, Zheng SB and Dang Q: Saikosaponin-d: A potential
chemotherapeutics in castration resistant prostate cancer by
suppressing cancer metastases and cancer stem cell phenotypes.
Biochem Biophys Res Commun. 474:722–729. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ye X and Weinberg RA:
Epithelial-mesenchymal plasticity: A central regulator of cancer
progression. Trends Cell Biol. 25:675–686. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Karlsson MC, Gonzalez SF, Welin J and Fuxe
J: Epithelial-mesenchymal transition in cancer metastasis through
the lymphatic system. Mol Oncol. 11:781–791. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lessard L, Karakiewicz PI, Bellon-Gagnon
P, Alam-Fahmy M, Ismail HA, Mes-Masson AM and Saad F: Nuclear
localization of nuclear factor-kappaB p65 in primary prostate
tumors is highly predictive of pelvic lymph node metastases. Clin
Cancer Res. 12:5741–5745. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ismail HA, Lessard L, Mes-Masson AM and
Saad F: Expression of NF-kappaB in prostate cancer lymph node
metastases. Prostate. 58:308–313. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Min C, Eddy SF, Sherr DH and Sonenshein
GE: NF-kappaB and epithelial to mesenchymal transition of cancer. J
Cell Biochem. 104:733–744. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rajasekhar VK, Studer L, Gerald W, Socci
ND and Scher HI: Tumour-initiating stem-like cells in human
prostate cancer exhibit increased NF-κB signalling. Nat Commun.
2:1622011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nakazawa M and Kyprianou N:
Epithelial-mesenchymal-transition regulators in prostate cancer:
Androgens and beyond. J Steroid Biochem Mol Biol. 166:84–90. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li P, Yang R and Gao WQ: Contributions of
epithelial-mesenchymal transition and cancer stem cells to the
development of castration resistance of prostate cancer. Mol
Cancer. 13:552014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sun Y, Wang BE, Leong KG, Yue P, Li L,
Jhunjhunwala S, Chen D, Seo K, Modrusan Z, Gao WQ, et al: Androgen
deprivation causes epithelial-mesenchymal transition in the
prostate: Implications for androgen-deprivation therapy. Cancer
Res. 72:527–536. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Figiel S, Vasseur C, Bruyere F, Rozet F,
Maheo K and Fromont G: Clinical significance of
epithelial-mesenchymal transition markers in prostate cancer. Hum
Pathol. 61:26–32. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shiota M, Yokomizo A, Tada Y, Inokuchi J,
Kashiwagi E, Masubuchi D, Eto M, Uchiumi T and Naito S: Castration
resistance of prostate cancer cells caused by castration-induced
oxidative stress through Twist1 and androgen receptor
overexpression. Oncogene. 29:237–250. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wu K, Gore C, Yang L, Fazli L, Gleave M,
Pong RC, Xiao G, Zhang L, Yun EJ, Tseng SF, et al: Slug, a unique
androgen-regulated transcription factor, coordinates androgen
receptor to facilitate castration resistance in prostate cancer.
Mol Endocrinol. 26:1496–1507. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ni J, Cozzi P, Hao J, Duan W, Graham P,
Kearsley J and Li Y: Cancer stem cells in prostate cancer
chemoresistance. Curr Cancer Drug Targets. 14:225–240. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yun EJ, Zhou J, Lin CJ, Hernandez E, Fazli
L, Gleave M and Hsieh JT: Targeting cancer stem cells in
castration-resistant prostate cancer. Clin Cancer Res. 22:670–679.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang X, Kruithof-de Julio M, Economides
KD, Walker D, Yu H, Halili MV, Hu YP, Price SM, Abate-Shen C and
Shen MM: A luminal epithelial stem cell that is a cell of origin
for prostate cancer. Nature. 461:495–500. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jeter CR, Liu B, Liu X, Chen X, Liu C,
Calhoun-Davis T, Repass J, Zaehres H, Shen JJ and Tang DG: NANOG
promotes cancer stem cell characteristics and prostate cancer
resistance to androgen deprivation. Oncogene. 30:3833–3845. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jeter CR, Badeaux M, Choy G, Chandra D,
Patrawala L, Liu C, Calhoun-Davis T, Zaehres H, Daley GQ and Tang
DG: Functional evidence that the self-renewal gene NANOG regulates
human tumor development. Stem Cells. 27:993–1005. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rybak AP and Tang D: SOX2 plays a critical
role in EGFR-mediated self-renewal of human prostate cancer
stem-like cells. Cell Signal. 25:2734–2742. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kregel S, Kiriluk KJ, Rosen AM, Cai Y,
Reyes EE, Otto KB, Tom W, Paner GP, Szmulewitz RZ and Griend Vander
DJ: Sox2 is an androgen receptor-repressed gene that promotes
castration-resistant prostate cancer. PLoS One. 8:e537012013.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Taniguchi K and Karin M: NF-κB,
inflammation, immunity and cancer: Coming of age. Nat Rev Immunol.
18:309–324. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kotiyal S and Bhattacharya S: Breast
cancer stem cells, emt and therapeutic targets. Biochem Biophys Res
Commun. 453:112–116. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ozes ON, Mayo LD, Gustin JA, Pfeffer SR,
Pfeffer LM and Donner DB: NF-kappaB activation by tumour necrosis
factor requires the Akt serine-threonine kinase. Nature. 401:82–85.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Han Y, Englert JA, Yang R, Delude RL and
Fink MP: Ethyl pyruvate inhibits nuclear factor-kappaB-dependent
signaling by directly targeting p65. J Pharmacol Exp Ther.
312:1097–1105. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lee SO, Lou W, Nadiminty N, Lin X and Gao
AC: Requirement for NF-(kappa)B in interleukin-4-induced androgen
receptor activation in prostate cancer cells. Prostate. 64:160–167.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Penning TM: Mechanisms of drug resistance
that target the androgen axis in castration resistant prostate
cancer (CRPC). J Steroid Biochem Mol Biol. 153:105–113. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Bennett-Guerrero E, Swaminathan M, Grigore
AM, Roach GW, Aberle LG, Johnston JM and Fink MP: A phase II
multicenter double-blind placebo-controlled study of ethyl pyruvate
in high-risk patients undergoing cardiac surgery with
cardiopulmonary bypass. J Cardiothorac Vasc Anesth. 23:324–329.
2009. View Article : Google Scholar : PubMed/NCBI
|