Introduction
Breast carcinoma (BC) is one of the most common
types of malignancy and is the second leading cause of cancer
associated-mortalities among women (1). As in other lower or middle-income
countries, BC is common in China, and its incidence is increasing.
The disease occurs at a younger median age for Chinese women
compared with western Caucasian women (2), and without a nationwide screening
program, patients may be unaware of the importance of regular
examination, which may lead to difficulty in treating patients. BC
has a diverse and complex range of biological subtypes, which
possess various unique clinical, pathological and molecular
features (3). Based on the
expression status of estrogen receptor (ER), progesterone receptor
(PR) and receptor tyrosine-protein kinase erbB-2 (HER2), the
molecular classification may offer better prognostic prediction and
therapeutic outcomes (4).
Members of the Suppressor of variegation, Enhancer
of Zeste, Trithorax (SET) and Myeloid-Nervy-DEAF1 (MYND)
domain-containing protein (SMYD) family represent a group of
proteins that contain the conserved SET and MYND domains (5). In the last decade, the SMYD protein
family has attracted increasing interest due to its essential role
in the development of the heart and muscle, and its potential role
in the development of cancer (6).
There are five members of the SMYD family, SMYD1, SMYD2, SMYD3,
SMYD4 and SMYD5, and they all exhibit various biological functions,
including developmental regulation and cancer (7). SMYD3 has been identified to be
overexpressed in >15 types of cancer, and its overexpression may
promote the proliferation of cancer cells (8). SMYD2 and SMYD4 expression has
additionally been associated with cancer progression. SMYD2 was
overexpressed in patients with leukemia with a poor prognosis
(9), whereas, SMYD4 served as a
tumor suppressor in BC (10).
Advances in microarray technology have advanced DNA
and RNA research, and has become an indispensable part of genomics
(11). In the present study,
thousands of gene expression or copy number analyses published
online were collectively analyzed to investigate the expression of
the SMYD family members in these databases to determine their
clinical value in BC.
Materials and methods
ONCOMINE data-mining analysis
ONCOMINE (www.oncomine.org) is an online cancer database, which
was used to analyze the transcriptional alteration in different
types of cancer (12). A comparison
of the expression levels of the SMYD family members was made
between clinical cancer samples and normal controls.
Breast Cancer Gene-Expression Miner
v4.1 (bcGenExMiner v4.1)
bcGenExMiner v4.1
(bcgenex.centregauducheau.fr/BC-GEM) is an online analysis tool,
which contains 36 genomic datasets, with accompanying notes,
(updated in December 2017) and was used to analyze the association
between the mRNA expression levels of SMYDs and clinical
parameters, including ER, PR and HER2 (13,14).
Kaplan-Meier Plotter
Kaplan-Meier curves were created using the
Kaplan-Meier Plotter (www.kmplot.com) to analyze the prognostic value of
SMYD expression. Information on 3,951 clinical patients with BC
were obtained from the website (15). Patients with BC were divided into two
groups (high and low) based on the median expression level of SYMDs
to evaluate the relapse-free survival (RFS) rate, according to the
Kaplan-Meier curve. The JetSet best probe (16) set was selected if the target gene had
more than one probe set.
The Cancer Genome Atlas data (TCGA)
and cBioPortal
TCGA (cancergenome.nih.gov/) is a database, which collects
the sequencing and pathological data of samples from 30 different
types of cancer (17). cBioPortal
(www.cbioportal.org) is a
multi-functional visualization and analysis toolset, which may be
used to anaylze gene expression of the cancer samples. The gene
expression and pathological information of 1,108 cases were used
from the BC dataset in cBioPortal for further analysis of
expression of the SMYD family members (18,19).
Statistical analysis
For the Elston-Ellis modification of the
Scarff-Bloom-Richardson (SBR) grading system (20), significance was determined using a
Welch's test with a post-hoc Dunnett-Tukey-Kramer's tests for
pairwise comparison. The prognostic value of the SMYDs was
calculated using a univariate Cox analysis (21) to generate the Forest plots. For the
ONCOMINE datasets, a Student's t-test (22) was used to analyze the datasets.
P<0.05 or a 1.5 fold-change was considered to indicate a
statistically significant difference. Welch's test,
Dunnett-Tukey-Kramer's test and univariate Cox analysis were
performed using bcGenExMiner v4.1, and Student's t-test was
performed using ONCOMINE.
Results
Transcriptional expression levels of
SMYDs in patients with BC
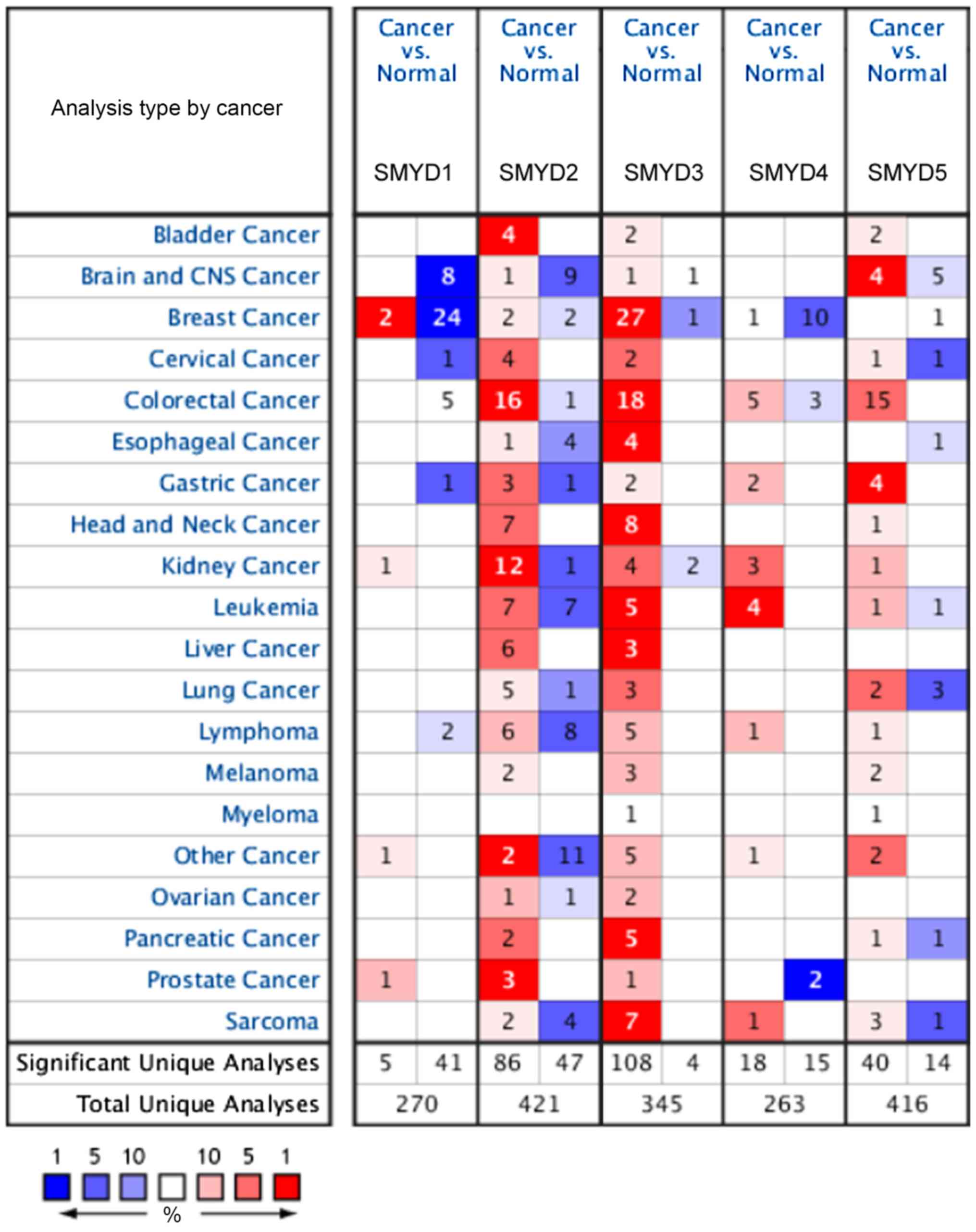
Using the datasets acquired from ONCOMINE, the mRNA
expression levels of the SMYD family members in the BC samples were
compared with the control samples (Fig.
1). The analysis demonstrated that SMYD3 was significantly
upregulated in patients with BC in 27 databases. SMYD1 and SMYD4
were significantly downregulated in 24 and 10 of the datasets,
respectively. In one dataset, Curtis et al (23), SMYD1 was identified to be
downregulated in a number of different types of BC (medullary BC
with a fold change of −2.12; ductal BC in situ with a fold
change of −1.854; invasive BC with a fold change of −1.962;
invasive lobular BC with a fold change of −1.903; and invasive
ductal and invasive lobular BC with a fold change of −1.891; with a
total fold change of −2.075 in all types of BC collectively)
compared with expression in the normal tissue samples (Table I). By contrast, SMYD3 was upregulated
in a number of types of BC (medullary BC with a fold change of
2.006; invasive ductal BC with a fold change of 2.526; invasive BC
with a fold change of 2.342; invasive lobular BC with a fold change
of 2.522; invasive ductal and invasive lobular BC with a fold
change of 2.748; with a total fold change of 2.344 across all types
of BC collectively) (18). The
upregulation of SMYD2 in invasive ductal and invasive lobular BC
(fold change of 1.449) and invasive ductal BC (fold change of
1.339) and the downregulation of SMYD4 in invasive BC (fold change
of −1.807), invasive ductal and lobular BC (fold change of −1.889),
and invasive ductal BC (fold change of −1.737) were identified in
the TCGA dataset (Table I). No
significant differences were identified for SMYD5 expression
between the BC tissues and comparative normal tissues in either of
the datasets (data not shown).
 | Table I.Different types of BC are associated
with different alterations of SMYD expression (ONCOMINE
database). |
Table I.
Different types of BC are associated
with different alterations of SMYD expression (ONCOMINE
database).
| A, SMYD1 |
|---|
|
|---|
| Author, year | Type of BC vs.
breast | Fold change | P-value | t-test | (Refs.) |
|---|
| Curtis et
al, 2012 | Medullary BC | −2.12 |
5.60×10−24 | −11.703 | (23) |
|
| Ductal BC in
situ | −1.854 | 0.00000152 | −6.339 |
|
|
| Invasive BC | −1.962 |
7.34×10−8 | −6.389 |
|
|
| Invasive lobular
BC | −1.903 |
1.96×10−19 | −9.805 |
|
|
| Invasive ductal and
invasive lobular BC | −1.891 |
2.34×10−16 | −8.743 |
|
|
| Total BC | −2.075 |
4.43×10−08 | −7.198 |
|
|
| B,
SMYD2 |
|
| Author,
year | Type of BC vs.
breast | Fold
change | P-value | t-test | (Refs.) |
|
| TCGA, accessed
2018 | Invasive ductal and
invasive lobular BC | 1.449 | 0.016 | 5.381 | – |
|
| Invasive lobular
BC | 1.339 |
2.54×10−23 | 14.652 |
|
|
| C,
SMYD3 |
|
| Author,
year | Type of BC vs.
breast | Fold
change | P-value | t-test | (Refs.) |
|
| Curtis et
al, 2012 | Medullary BC | 2.006 |
3.42×10−8 | 6.841 | (23) |
|
| Invasive ductal
BC | 2.526 |
2.05×10−100 | 35.609 |
|
|
| Invasive BC | 2.342 | 0.00000216 | 6.129 |
|
|
| Invasive lobular
BC | 2.522 |
2.41×10−54 | 20.460 |
|
|
| Invasive ductal and
invasive lobular BC | 2.748 |
4.23×10−35 | 17.426 |
|
|
| Total BC | 2.344 | 0.00000443 | 6.808 |
|
|
| D,
SMYD4 |
|
| Author,
year | Type of BC vs.
breast | Fold
change | P-value | t-test | (Refs.) |
|
| TCGA, accessed
2018 | Invasive BC | −1.807 |
3.02×10−23 | −12.039 | – |
|
| Invasive ductal and
lobular BC | −1.889 | 0.0000791 | −10.652 |
|
|
| Invasive ductal
BC | −1.737 |
3.30×10−24 | −14.280 |
|
mRNA expression levels of the SMYD
family members are significantly associated with various
clinicopathological parameters of patients with BC
The mRNA expression levels of SMYDs between groups
of patients according to different clinical parameters were
compared using bcGenExMiner and the results are presented in
Table II. SMYD3 was determined to
be upregulated in patients with BC over the age of 51 Patients with
BC with a positive nodal status presented increased SMYD5 mRNA
expression levels compared with patients who had a negative nodal
status.
 | Table II.Comparison of SMYD expression between
patients with breast cancer and different clinicopathological
parameters. |
Table II.
Comparison of SMYD expression between
patients with breast cancer and different clinicopathological
parameters.
|
| SMYD1 mRNA | SMYD2 mRNA | SMYD3 mRNA | SMYD4 mRNA | SMYD5 mRNA |
|---|
|
|
|
|
|
|
|
|---|
| Variable | Number of
patientsa | Expression | P-value | Number of
patientsa | Expression | P-value | Number of
patientsa | Expression | P-value | Number of
patientsa | Expression | P-value | Number of
patientsa | Expression | P-value |
|---|
| Age |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
≤51 | 865 | – | 0.1362 | 1,361 | – | 0.0832 | 1,329 | – | 0.0198 | 645 | – | 0.8210 | 1,28 | – | 0.8668 |
|
>51 | 1,078 | – |
| 2,142 | – |
| 2,099 | ↑ |
| 1,386 | – |
| 2,007 | – |
|
| Nodal status |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| − | 1,295 | – | 0.1642 | 2,447 | – | 0.6918 | 2,401 | – | 0.3768 | 989 | – | 0.9131 | 2,322 | – | <0.0001 |
| + | 1,116 | – |
| 1,509 | – |
| 1,480 | – |
| 880 | – |
| 1,418 | ↑ |
|
| ER, IHC |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| − | 872 | – | 0.0832 | 1,525 | – | <0.0001 | 1,452 | – | <0.0001 | 752 | – | 0.0967 | 1,443 | – | 0.0004 |
| + | 2,206 | – |
| 3,923 | ↓ |
| 3,767 | ↑ |
| 2,121 | – |
| 3,789 | ↓ |
|
| PR, IHC |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| − | 438 | – | 0.7915 | 946 | – | <0.0001 | 913 | – | <0.0001 | 509 | – | 0.2489 | 946 | – | <0.0001 |
| + | 770 | – |
| 1,439 | ↓ |
| 1,402 | ↑ |
| 935 | – |
| 1,439 | ↓ |
|
| HER2, IHC |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| − | 619 | – | 0.4522 | 1,409 | – | 0.0653 | 1,409 | – | 0.584 | 497 | – | 0.0004 | 1,409 | – | 0.0341 |
| + | 157 | – |
| 201 | – |
| 201 | – |
| 134 | ↓ |
| 201 | ↑ |
|
| Triple-negative
status |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Not | 2,324 | – | 0.9177 | 4,099 | – | <0.0001 | 3,938 | – | <0.0001 | 2,241 | – | 0.2133 | 3,965 | – | 0.0005 |
|
TNBC | 197 | – |
| 347 | ↑ |
| 374 | ↓ |
| 128 | – |
| 347 | ↑ |
|
SMYD2/3/5 were identified to be associated with ER
and PR status. SMYD2/5 exhibited decreased expression levels in
patients with ER and PR positive BC, whereas, SMYD3 demonstrated
increased expression levels in patients with ER and PR positive BC.
The transcriptional expression level of SMYD4 was decreased and the
expression levels of SMYD5 were increased in patients with HER2
positive BC. Triple-negative BC (TNBC) is an aggressive form of BC
where ER, PR and HER2 expression is absent. The mRNA expression
levels of SMYD2/5 in TNBC were increased, whereas, the expression
of SMYD3 was decreased in patients with TNBC.
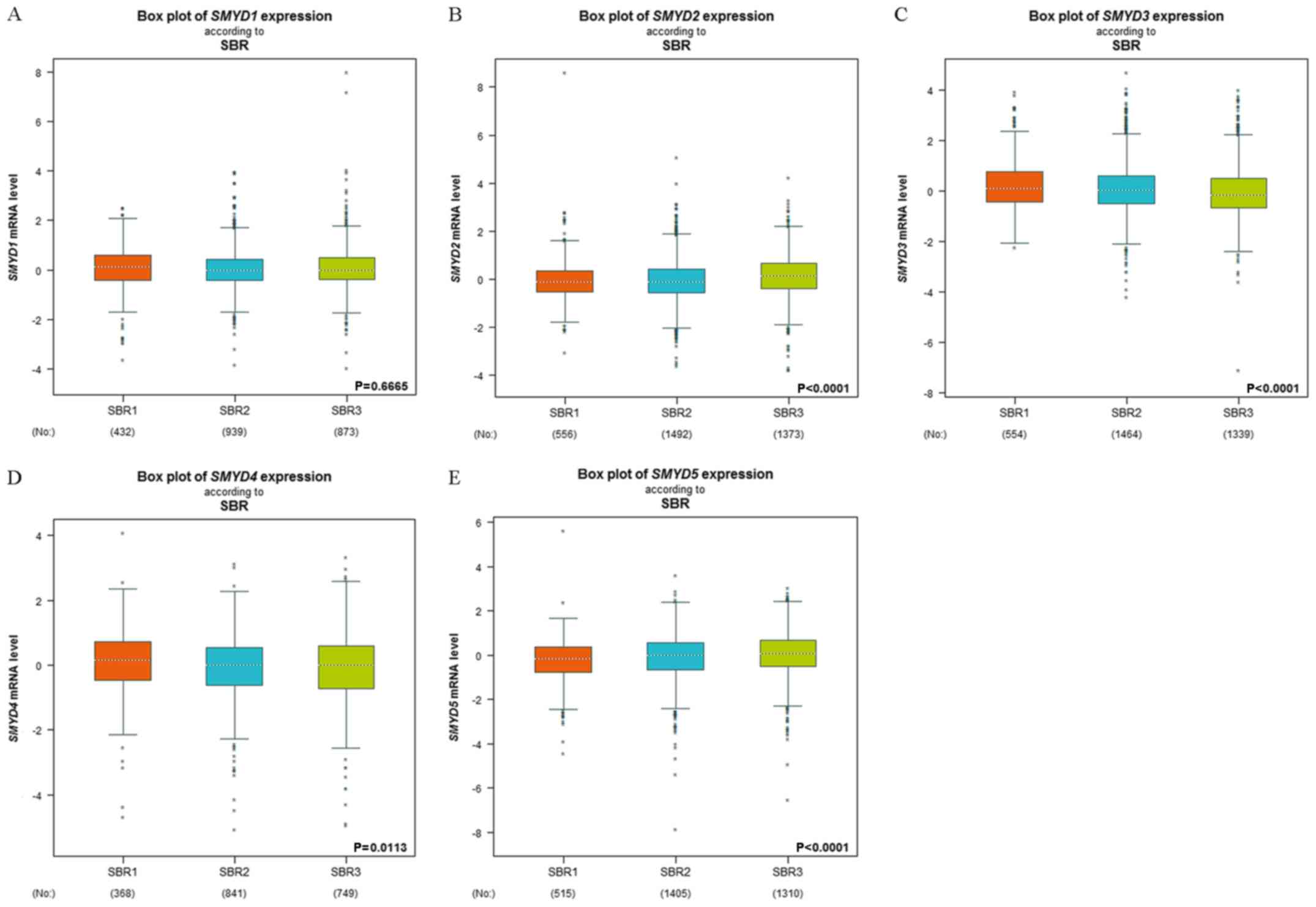
According to the SBR grading status criterion, it
was demonstrated that the expression of all the SMYDs, with the
exception of SMYD1, was associated with the SBR grade (Fig. 2). Decreased SMYD3 expression and
increased SMYD5 expression suggested advanced SBR grade, whereas,
SMYD2/4 exhibited a global significant difference with P<0.05.
No statistical significance of these SMYDs was observed using the
Dunnett-Tukey-Kramer's test of each group in association with the
SBR grade status (SBR1 vs. SBR2, SBR1 vs. SBR3 and SBR2 vs. SBR3;
Table III).
 | Table III.Dunnett-Tukey-Kramer's test for
pairwise comparison in SBR criterion. SBR1, SBR2 and SBR3 means the
SBR grade value. |
Table III.
Dunnett-Tukey-Kramer's test for
pairwise comparison in SBR criterion. SBR1, SBR2 and SBR3 means the
SBR grade value.
| mRNA | Pairwise comparison
of SBR | mRNA
expressiona | P-value |
|---|
| SMYD2 | SBR1 vs. SBR2 | SBR1=SBR2 | >0.10 |
|
| SBR1 vs. SBR3 | SBR1<SBR3 | <0.0001 |
|
| SBR2 vs. SBR3 | SBR2<SBR3 | <0.0001 |
| SMYD3 | SBR1 vs. SBR2 | SBR1>SBR2 | <0.05 |
|
| SBR1 vs. SBR3 | SBR1>SBR3 | <0.0001 |
|
| SBR2 vs. SBR3 | SBR2>SBR3 | <0.001 |
| SMYD4 | SBR1 vs. SBR2 | SBR1>SBR2 | <0.05 |
|
| SBR1 vs. SBR3 | SBR1>SBR3 | <0.05 |
|
| SBR2 vs. SBR3 | SBR2=SBR3 | >0.10 |
| SMYD5 | SBR1 vs. SBR2 | SBR1<SBR2 | <0.05 |
|
| SBR1 vs. SBR3 | SBR1<SBR3 | <0.0001 |
|
| SBR2 vs. SBR3 | SBR2<SBR3 | <0.001 |
Increased SMYD1/4 and decreased
SMYD2/5 mRNA expression levels are associated with good RFS of
patients with BC
The RFS of patients with BC was associated with the
mRNA expression levels of SMYD1/2/4/5, which was identified by the
Kaplan-Meier curve and a log-rank test (P<0.05) and stated below
the plot is the number of patients that were at risk (Fig. 3). The patients with BC with increased
SMYD1/4 mRNA expression levels or decreased SMYD2/5 mRNA expression
levels were predicted to indicate good RFS.
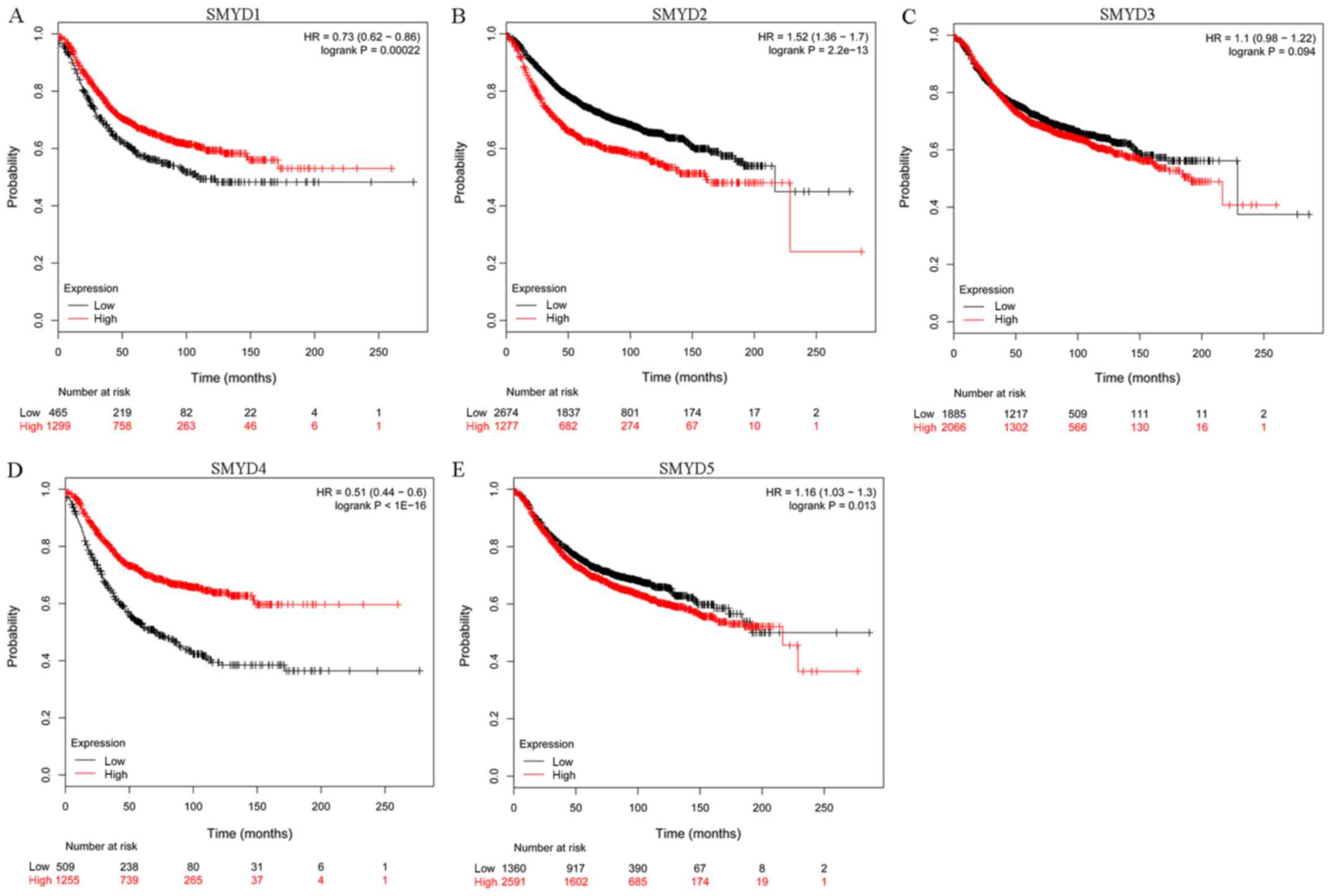 | Figure 3.Association between the mRNA
expression levels of the SMYD family members and the prognostic
value in relapse free survival of patients with BC. (A) SMYD1
(228406_at; HR, 0.73; 95% CI, 0.62–0.86; P=0.00022). (B) SMYD2
(212922_s_at; HR, 1.52; 95%CI, 1.36–1.7; P=2.2×10−13).
(C) SMYD3 (218788_s_at; HR, 1.1; 95% CI, 0.98–1.22; P=0.094). (D)
SMYD4 (229175_at; HR, 0.51; 95% CI, 0.44–0.6;
P=1×10−16). (E) SMYD5 (209516_at; HR, 1.16; 95% CI,
1.03–1.3; P=0.013). SMYD, Suppressor of variegation, Enhancer of
Zeste, Trithorax and Myeloid-Nervy-DEAF1 domain-containing; HR,
hazard ratio; CI, confidence interval. |
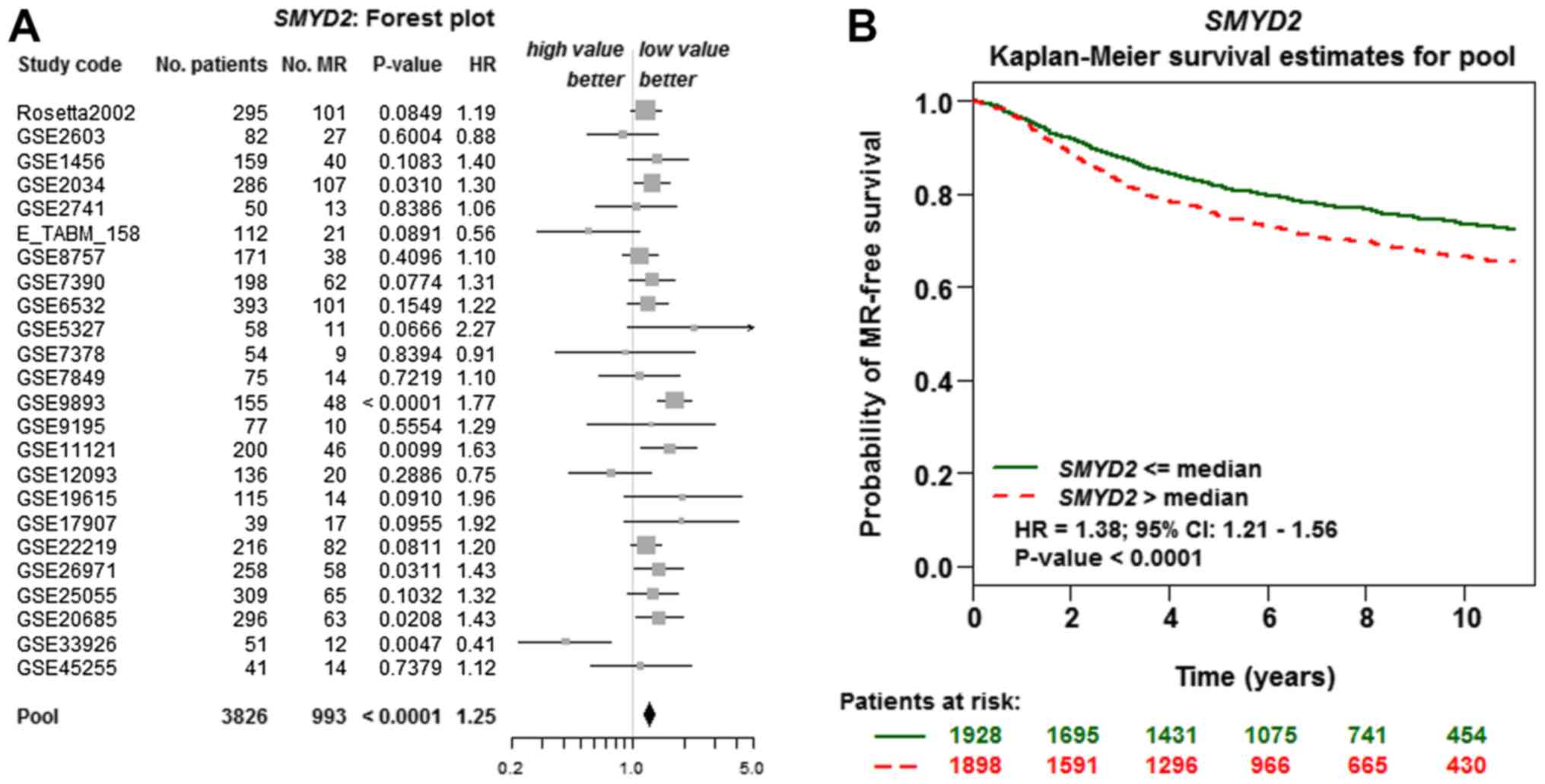
Prognostic analysis of bcGenExMiner demonstrated the
relevance between the mRNA expression levels of SMYD2 and the
metastatic RFS (MRFS) in patients with BC was further analyzed by
dataset (Table IV). An increased
risk of metastatic relapse [hazard ratio (HR) =1.25; 95% confidence
interval (CI) 1.17–1.34; P<0.0001] was observed in the patients
with increased SMYD2 expression. Similarly, patients with increased
SMYD2 expression had a decreased MRFS (HR =1.38; 95% CI 1.21–1.56;
P<0.0001; Fig. 4). The expression
of SMYD1 (HR, 0.91; 95% CI 0.77–1.09; P=0.3084), SMYD3 (HR, 0.94;
95% CI 0.83–1.07; P=0.3514), SMYD4 (HR, 0.84; 95% CI 0.69–1.02;
P=0.0798) and SMYD5 (HR, 1.10; 95% CI 0.97–1.26; P=0.1505)
exhibited no association with MRFS of patients with BC (data not
shown).
 | Table IV.Univariate Cox analysis of SMYD2. |
Table IV.
Univariate Cox analysis of SMYD2.
|
| Population and
event criteria | SMYD2 univariate
Cox analysis |
|
|
|---|
|
|
|
|
|
|
|---|
| No. | Nodal status | ER status | MR status | P-value | HR | 95% CI | Good prognostic RNA
expression level | Number of
patients | Number of
events |
|---|
| 1 | Nm | ERm | MR | <0.0001 | 1.25 | 1.17–1.34 | ↓ | 3,826 | 993 |
| 2 | Nm | ER+ | MR | <0.0001 | 1.25 | 1.15–1.36 | ↓ | 2,757 | 658 |
| 3 | N- | ERm | MR | <0.0001 | 1.29 | 1.15–1.44 | ↓ | 1,887 | 454 |
| 4 | N- | ER+ | MR | 0.0056 | 1.22 | 1.06–1.41 | ↓ | 1,389 | 312 |
| 5 | N- | ER- | MR | 0.0221 | 1.27 | 1.03–1.56 | ↓ | 479 | 140 |
| 6 | N+ | ERm | MR | 0.0291 | 1.14 | 1.01–1.29 | ↓ | 980 | 322 |
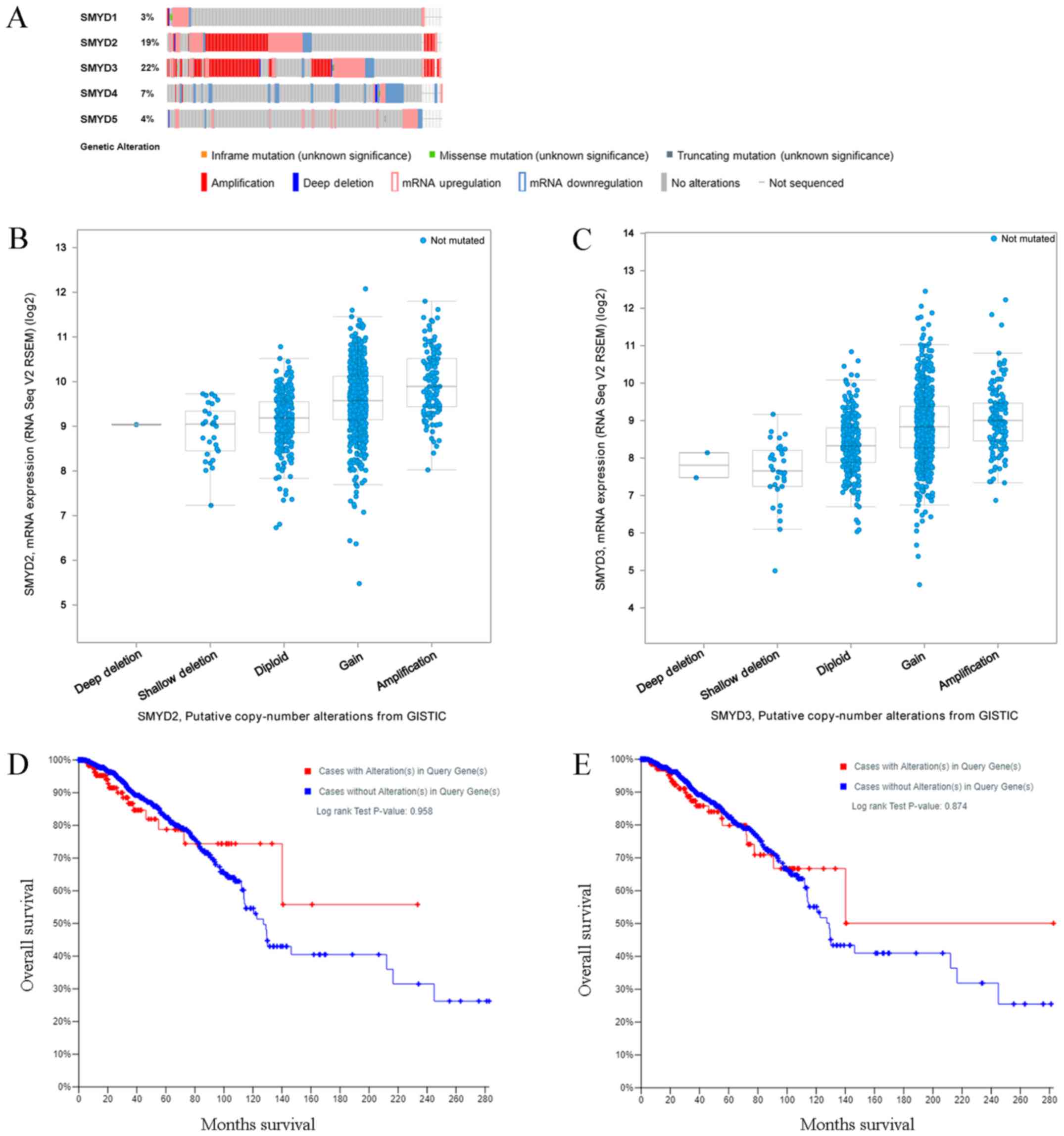
SMYD factors are significantly altered
in patients with BC
A total of 365 samples out of 1,108 (37%; data not
shown) with invasive BC had altered expression levels of at least
one of the SMYDs (3% of samples with altered expression of SMYD1,
19% of samples with altered expression of SMYD2, 22% of samples
with altered expression of SMYD3, 7% of samples with altered
expression of SMYD4 and 4% of samples with altered expression of
SMYD5; Fig. 5A). SMYD2/3 was
increased in a portion of patients with BC. SMYD1/5 mRNA
upregulation and SMYD4 mRNA downregulation were observed in certain
patients with BC. SMYD members were rarely mutated in patients with
BC; however, the copy numbers of SMYD2/3 were positively associated
with their mRNA expression levels (Fig.
5B and C). Patients with SMYD2/3 amplification had increased
expression levels of SMYD2/3 compared with patients where increased
SMYD2/3 amplification was not observed. However, no significant
association was identified between the copy-number alternations of
SMYD2/3 and the overall survival of patients with BC (SMYD2,
P=0.958; SMYD3, P=0.874; Fig. 5D and
E).
Discussion
The SMYD proteins are abundantly expressed in
cardiac and skeletal muscle, and were initially hypothesized to
serve as epigenetic regulators for myogenesis and cardiomyocyte
differentiation (8). SMYD proteins
have attracted increasing interest in research following the
identification of a potential role in the development of cancer,
suggesting that the SMYD proteins may be potential therapeutic
targets (24). To the best of the
authors' knowledge, the present study is the first to analyze the
mRNA expression levels and prognostic value of the SMYD family
members in BC.
SMYD1, the first member of the SMYD family, has
traditionally been known to function in skeletal muscle and
myocardium development during the embryonic period (5). Targeted SMYD1 deletion in mice disrupts
the maturation of cardiomyocytes and formation of the right
ventricle (25). Heparin binding
growth factors (HDGF) possess mitogenic and angiogenic activities,
and its expression is increased in the developing heart, tumor cell
lines and certain normal tissues (26). HDGF has been demonstrated to repress
the expression of SMYD1 in G-7 myoblast cells (27). HDGF is overexpressed in a number of
types of human cancer, including hepatocellular carcinoma (28) and BC (29). The interaction of SMYD1 and HDGF
suggests that SMYD1 may function in cancer development. In the
present study, the mRNA expression levels of SMYD1 were decreased
in patients with BC compared with normal breast tissue. Patients
with BC with increased SMYD1 expression levels exhibited good
RFS.
Numerous previous studies have demonstrated the
involvement of SMYD2 in various cancer types. For example, SMYD2
has been demonstrated to promote TNBC progression (30). In the present study, patients with BC
who exhibited decreased SMYD2 expression levels demonstrated
improved RFS. SMYD2 represses the activity of cellular tumor
antigen p53 by SMYD2-mediated methylation (31). SMYD2 may additionally downregulate
the phosphatase and tensin (PTEN) pathway through the methylation
of PTEN (32). These previous
studies examined the important role of SMYD2 in cancer. In the
present study, SMYD2 was significantly increased in patients with
BC and those with metastatic relapse. Decreased SMYD2 mRNA
expression levels were associated with good RFS and, thus, raises
the possibility that SMYD2 serves as a diagnostic biomarker in BC
diagnosis and as a prognostic indicator for patients with BC with
metastatic relapse.
SMYD3 is considered an oncogene in a number of
different types of cancer. SMYD3 is regulated by
β-catenin/transcription factor-4 transcription apparatus in gastric
cancer (33). SMYD3 mediated
methylation of mitogen-activated protein kinase (MAPK) kinase
kinase 2, which stimulated MAPK signaling and promoted the
formation of Ras-driven carcinomas (34). SMYD3 may additionally influence
distinct oncogenic processes by serving as a gene-specific
transcriptional regulator (35).
SMYD3 has been demonstrated to promote the development of BC
(36,37) and is associated with familial BC
(38). In the present study, it was
identified that SMYD3 was amplified in patients with BC; however,
it was not associated with the RFS of patients with BC.
Additionally, it was observed that advanced SBR grade was
associated with decreased SMYD3 mRNA expression levels. Therefore,
SMYD3 may be used as a biomarker for diagnosis of BC and as an
indicator of the SBR grade of BC tissue.
Less is known regarding the role of SMYD4 in cancer
development. Hu et al (10)
demonstrated that SMYD4 may serve as a tumor suppressor gene in BC.
However, to the best of the authors' knowledge, there is no
information regarding an association between SMYD4 and the
prognosis of patients with BC. In the present study, increased
SMYD4 mRNA expression levels were associated with good RFS in
patients with BC. As a result, it is possible that SMYD4 may serve
as a good prognostic indicator.
Similar to SMYD4, there are comparatively fewer
studies investigating the role of SMYD5 in cancer development. At
present, previous studies on SMYD5 focused on its role in embryonic
stem (ES) cells. SMYD5 primarily serves a role in the
differentiation of ES cells (39,40).
However, the depletion of SMYD5 in human colon and lung cancer
cells resulted in increased tumor growth and the upregulation of
genes associated with colon and lung cancer (39). In the present study, it was
demonstrated that the SMYD5 mRNA expression levels were decreased
in patients with ER/PR-positive BC compared with those with
ER/PR-negative BC and increased in patients with HER2-positive BC
and TNBC. Increased SMYD5 mRNA expression levels were associated
with advanced SBR grade. Therefore, SMYD5 may serve as a potential
oncogene in BC.
In conclusion, the SMYD family may function in the
development of BC. Previous studies investigating the functions of
the SMYD family members in cancer are rare and the mechanisms
regarding the differential expression pattern of its family members
in BC remain unclear. In the present study, the expression of SMYDs
was systemically analyzed to evaluate their clinical and prognostic
value in BC. The present findings suggested that SMYD2/3 may serve
as potential diagnostic biomarkers and therapeutic targets for BC.
In addition, the present data suggested that SMYD4 may serve as a
potential prognostic marker of survival in patients with BC. The
increases in SMYD1/5 mRNA expression levels observed in the present
data suggested a potential role for SMYD1/5 in the development of
BC.
Acknowledgements
The authors would like to acknowledge Mr Yanfeng Liu
(Qilu Hospital of Shandong University, Jinan, China) and Professor
Zhaojian Liu (Institute of Cell Biology, Shandong University School
of Basic Medicine, Jinan, China) for editing the manuscript and
assisting with data analyses.
Funding
The present study was supported by The National
Natural Science Foundation of China (grant no. 81571367), The
Shandong Scientific and Technological Research Program (grant nos.
2016GSF201082, 2017GSF218021 and 2018GSF118191; China) and Shandong
Natural Science Foundation (grant no. ZR2014HQ043; China).
Availability of data and materials
The datasets used and analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JS, BJ and ZL designed and conceived the study. YL
wrote the manuscript. QC, JY, ZJ and HZ reviewed the manuscript for
important intellectual content YL, QC and JY assisted with
acquisition of data. ZJ and HZ were involved in the analysis and
interpretation of data. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
DeSantis C, Siegel R, Bandi P and Jemal A:
Breast cancer statistics, 2011. CA Cancer J Clin. 61:409–418. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fan L, Strasser-Weippl K, Li JJ, St LJ,
Finkelstein DM, Yu KD, Chen WQ, Shao ZM and Goss PE: Breast cancer
in China. Lancet Oncol. 15:e279–e289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2015. CA Cancer J Clin. 65:5–29. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Onitilo AA, Engel JM, Greenlee RT and
Mukesh BN: Breast cancer subtypes based on ER/PR and Her2
expression: Comparison of clinicopathologic features and survival.
Clin Med Res. 7:4–13. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Du SJ, Tan X and Zhang J: SMYD proteins:
Key regulators in skeletal and cardiac muscle development and
function. Anat Rec (Hoboken). 297:1650–1662. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Doughan M, Spellmon N, Li C and Yang Z:
SMYD proteins in immunity: Dawning of a new era. AIMS Biophys.
3:450–455. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Leinhart K and Brown M: SET/MYND lysine
methyltransferases regulate gene transcription and protein
activity. Genes (Basel). 2:210–218. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Spellmon N, Holcomb J, Trescott L,
Sirinupong N and Yang Z: Structure and function of SET and MYND
domain-containing proteins. Int J Mol Sci. 16:1406–1428. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sakamoto LH, Andrade RV, Felipe MS,
Motoyama AB and Pittella SF: SMYD2 is highly expressed in pediatric
acute lymphoblastic leukemia and constitutes a bad prognostic
factor. Leuk Res. 38:496–502. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hu L, Zhu YT, Qi C and Zhu YJ:
Identification of Smyd4 as a potential tumor suppressor gene
involved in breast cancer development. Cancer Res. 69:4067–4072.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sealfon SC and Chu TT: RNA and DNA
microarrays. Methods Mol Biol. 671:3–34. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rhodes DR, Yu J, Shanker K, Deshpande N,
Varambally R, Ghosh D, Barrette T, Pandey A and Chinnaiyan AM:
ONCOMINE: A cancer microarray database and integrated data-mining
platform. Neoplasia. 6:1–6. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jézéquel P, Campone M, Gouraud W,
Guérin-Charbonnel C, Leux C, Ricolleau G and Campion L:
bc-GenExMiner: An easy-to-use online platform for gene prognostic
analyses in breast cancer. Breast Cancer Res Treat. 131:765–775.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jézéquel P, Frénel JS, Campion L,
Guérin-Charbonnel C, Gouraud W, Ricolleau G and Campone M:
bc-GenExMiner 3.0: New mining module computes breast cancer gene
expression correlation analyses. Database (Oxford).
2013:bas0602013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Györffy B, Lanczky A, Eklund AC, Denkert
C, Budczies J, Li Q and Szallasi Z: An online survival analysis
tool to rapidly assess the effect of 22,277 genes on breast cancer
prognosis using microarray data of 1,809 patients. Breast Cancer
Res Treat. 123:725–731. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li Q, Birkbak NJ, Gyorffy B, Szallasi Z
and Eklund AC: Jetset: Selecting the optimal microarray probe set
to represent a gene. BMC Bioinformatics. 12:4742011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cancer Genome Atlas Network, .
Comprehensive molecular portraits of human breast tumours. Nature.
490:61–70. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gao J, Aksoy BA, Dogrusoz U, Dresdner G,
Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, et al:
Integrative analysis of complex cancer genomics and clinical
profiles using the cBioPortal. Sci Signal. 6:2013. View Article : Google Scholar
|
|
19
|
Cerami E, Gao J, Dogrusoz U, Gross BE,
Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, et
al: The cBio cancer genomics portal: An open platform for exploring
multidimensional cancer genomics data. Cancer Discov. 2:401–404.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Martinazzi M, Zampatti C, Crivelli F,
Zampieri A and Martinazzi S: Scarff-bloom-richardson
histoprognostic grading correlates with the immunohistochemical
expressions of genomic alterations in infiltrating ductal
carcinomas (nos) of the breast. Oncol Rep. 1:1087–1091.
1994.PubMed/NCBI
|
|
21
|
Nicolai P, Redaelli de Zinis LO, Tomenzoli
D, Barezzani MG, Bertoni F, Bignardi M and Antonelli AR: Prognostic
determinants in supraglottic carcinoma: Univariate and Cox
regression analysis. Head Neck. 19:323–34. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hsu CL and Lee WC: Detecting
differentially expressed genes in heterogeneous diseases using half
student's t-test. Int J Epidemiol. 39:1597–1604. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Curtis C, Shah SP, Chin SF, Turashvili G,
Rueda OM, Dunning MJ, Speed D, Lynch AG, Samarajiwa S, Yuan Y, et
al: The genomic and transcriptomic architecture of 2,000 breast
tumours reveals novel subgroups. Nature. 486:346–352. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gottlieb PD, Pierce SA, Sims RJ, Yamagishi
H, Weihe EK, Harriss JV, Maika SD, Kuziel WA, King HL, Olson EN, et
al: Bop encodes a muscle-restricted protein containing MYND and SET
domains and is essential for cardiac differentiation and
morphogenesis. Nat Genet. 31:25–32. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Everett AD: Identification, cloning, and
developmental expression of hepatoma-derived growth factor in the
developing rat heart. Dev Dyn. 222:450–458. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Al-Shar'i NA and Alnabulsi SM: Explaining
the autoinhibition of the SMYD enzyme family: A theoretical study.
J Mol Graph Model. 68:147–157. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang J and Everett AD: Hepatoma-derived
growth factor represses SET and MYND domain containing 1 gene
expression through interaction with C-terminal binding protein. J
Mol Biol. 386:938–950. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hu TH, Huang CC, Liu LF, Lin PR, Liu SY,
Chang HW, Changchien CS, Lee CM, Chuang JH and Tai MH: Expression
of hepatoma-derived growth factor in hepatocellular carcinoma.
Cancer. 98:1444–1456. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen SC, Kung ML, Hu TH, Chen HY, Wu JC,
Kuo HM, Tsai HE, Lin YW, Wen ZH, Liu JK, et al: Hepatoma-derived
growth factor regulates breast cancer cell invasion by modulating
epithelial-mesenchymal transition. J Pathol. 228:158–169. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li LX, Zhou JX, Calvet JP, Godwin AK,
Jensen RA and Li X: Lysine methyltransferase SMYD2 promotes triple
negative breast cancer progression. Cell Death Dis. 9:3262018.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Huang J, Perez-Burgos L, Placek BJ,
Sengupta R, Richter M, Dorsey JA, Kubicek S, Opravil S, Jenuwein T
and Berger SL: Repression of p53 activity by Smyd2-mediated
methylation. Nature. 444:629–632. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Nakakido M, Deng Z, Suzuki T, Dohmae N,
Nakamura Y and Hamamoto R: Dysregulation of AKT pathway by
SMYD2-mediated lysine methylation on PTEN. Neoplasia. 17:367–373.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang T, Wu H, Liu S, Lei Z, Qin Z, Wen L,
Liu K, Wang X, Guo Y, Liu Q, et al: SMYD3 controls a Wnt-responsive
epigenetic switch for ASCL2 activation and cancer stem cell
maintenance. Cancer Lett. 430:11–24. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mazur PK, Reynoird N, Khatri P, Jansen PW,
Wilkinson AW, Liu S, Barbash O, Van Aller GS, Huddleston M, Dhanak
D, et al: SMYD3 links lysine methylation of MAP3K2 to Ras-driven
cancer. Nature. 510:283–287. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kim JM, Kim K, Schmidt T, Punj V, Tucker
H, Rice JC, Ulmer TS and An W: Cooperation between SMYD3 and PC4
drives a distinct transcriptional program in cancer cells. Nucleic
Acids Res. 43:8868–8883. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tsai CH, Chen YJ, Yu CJ, Tzeng SR, Wu IC,
Kuo WH, Lin MC, Chan NL, Wu KJ and Teng SC: SMYD3-Mediated H2A.Z.1
Methylation promotes cell cycle and cancer proliferation. Cancer
Res. 76:6043–6053. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hamamoto R, Silva FP, Tsuge M, Nishidate
T, Katagiri T, Nakamura Y and Furukawa Y: Enhanced SMYD3 expression
is essential for the growth of breast cancer cells. Cancer Sci.
97:113–118. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Frank B, Hemminki K, Wappenschmidt B,
Klaes R, Meindl A, Schmutzler RK, Bugert P, Untch M, Bartram CR and
Burwinkel B: Variable number of tandem repeats polymorphism in the
SMYD3 promoter region and the risk of familial breast cancer. Int J
Cancer. 118:2917–2918. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kidder BL, He R, Wangsa D, Padilla-Nash
HM, Bernardo MM, Sheng S, Ried T and Zhao K: SMYD5 Controls
heterochromatin and chromosome integrity during embryonic stem cell
differentiation. Cancer Res. 77:6729–6745. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kidder BL, Hu G, Cui K and Zhao K: SMYD5
regulates H4K20me3-marked heterochromatin to safeguard ES cell
self-renewal and prevent spurious differentiation. Epigenetics
Chromatin. 10:82017. View Article : Google Scholar : PubMed/NCBI
|