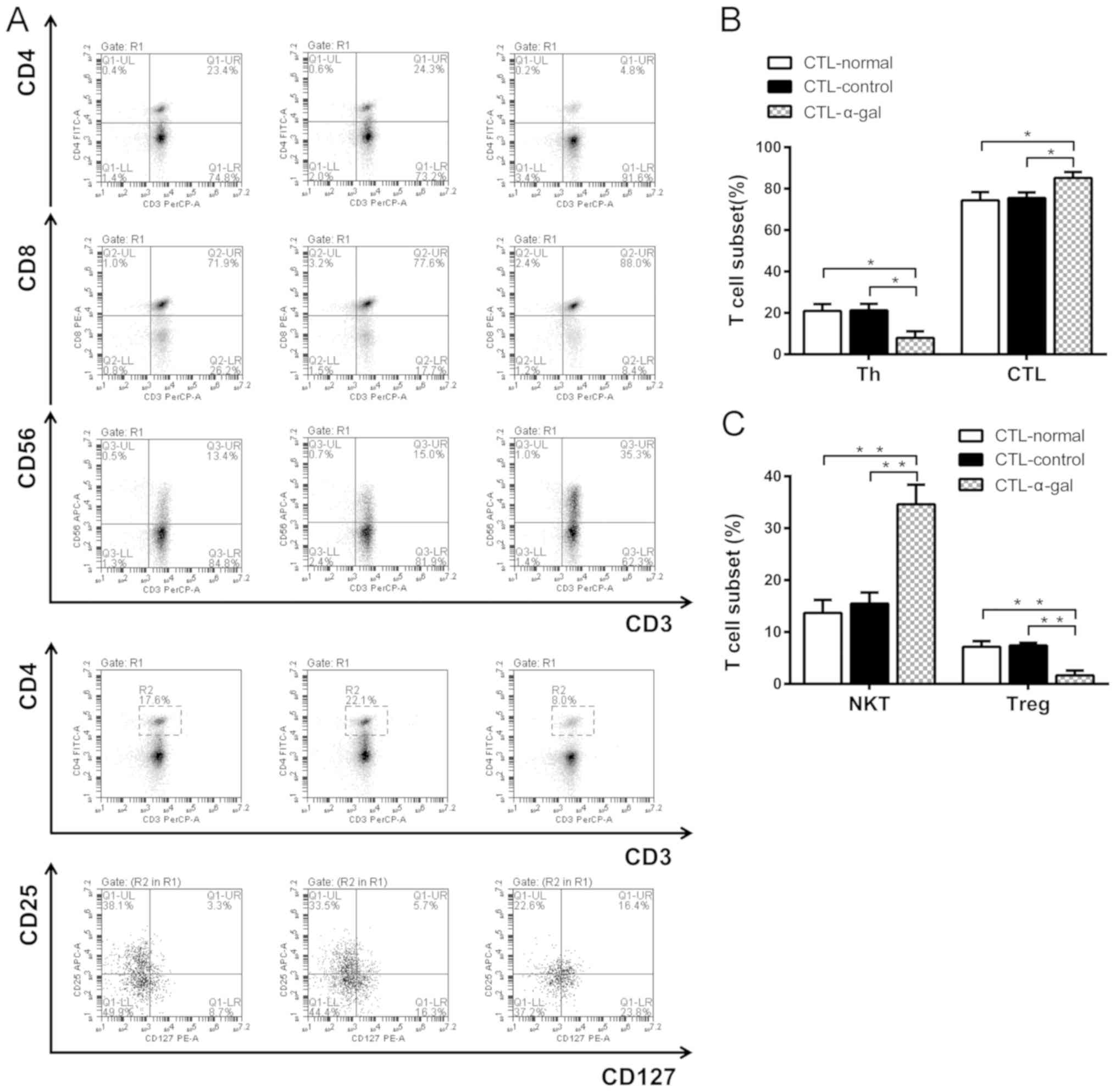
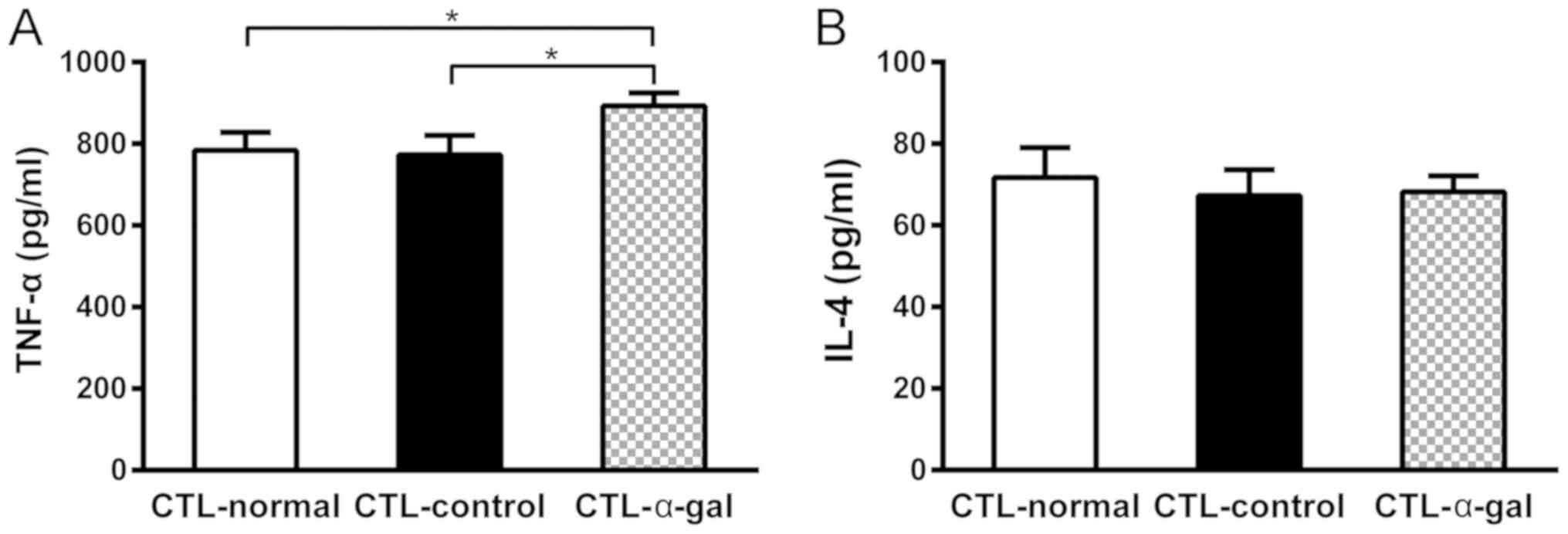
|
1
|
Adam R and Vinet E: Regional treatment of
metastasis: Surgery of colorectal liver metastases. Ann Oncol. 15
(Suppl 4):iv103–iv106. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ramos A and Hemann MT: Drugs, bugs, and
cancer: Fusobacterium nucleatum promotes chemoresistance in
colorectal cancer. Cell. 170:411–413. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Khan K, Wale A, Brown G and Chau I:
Colorectal cancer with liver metastases: Neoadjuvant chemotherapy,
surgical resection first or palliation alone? World J
Gastroenterol. 20:12391–12406. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Castro JE and Kipps TJ: Adoptive cellular
therapy for chronic lymphocytic leukemia and B cell malignancies.
CARs and more. Best Pract Res Clin Haematol. 29:15–29. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Schubert ML, Hückelhoven A, Hoffmann JM,
Schmitt A, Wuchter P, Sellner L, Hofmann S, Ho AD, Dreger P and
Schmitt M: Chimeric antigen receptor T cell therapy targeting
CD19-positive leukemia andlymphoma in the context of stem cell
transplantation. Hum Gene Ther. 27:758–771. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Reissfelder C, Stamova S, Gossmann C,
Braun M, Bonertz A, Walliczek U, Grimm M, Rahbari NN, Koch M,
Saadati M, et al: Tumor-specific cytotoxic T lymphocyte activity
determines colorectal cancer patient prognosis. J Clin Invest.
125:739–751. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhu H, Yang X, Li J, Ren Y, Zhang T, Zhang
C, Zhang J, Li J and Pang Y: Immune response, safety, and survival
and quality of life outcomes for advanced colorectal cancer
patients treated with dendritic cell vaccine and cytokine-induced
killer cell therapy. Biomed Res Int. 2014:6038712014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chou J, Voong LN, Mortales CL, Towlerton
AM, Pollack SM, Chen X, Yee C, Robbins PF and Warren EH: Epigenetic
modulation to enable antigen-specific T-cell therapy of colorectal
cancer. J Immunother. 35:131–141. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gao D, Li C, Xie X, Zhao P, Wei X, Sun W,
Liu HC, Alexandrou AT, Jones J, Zhao R and Li JJ: Autologous tumor
lysate-pulsed dendritic cell immunotherapy with cytokine-induced
killer cells improves survival in gastric and colorectal cancer
patients. PLoS One. 9:e938862014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Baratin M, Kayibanda M, Ziol M, Romieu R,
Briand JP, Guiller JG and Viguier M: Amino acid modifications in
the wild type sequence p53 232–240 overcome the poor immunogenicity
of this self tumour epitope. J Pept Sci. 8:327–334. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kalinski P: Dendritic cells in
immunotherapy of established cancer: Roles of signals 1, 2, 3 and
4. Curr Opin Investig Drugs. 10:526–535. 2009.PubMed/NCBI
|
|
12
|
Iorgulescu JB, Braun D, Oliveira G, Keskin
DB and Wu CJ: Acquired mechanisms of immune escape in cancer
following immunotherapy. Genome Med. 10:872018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Galili U: Autologous tumor vaccines
processed to express alpha-gal epitopes: A practical approach to
immunotherapy in cancer. Cancer Immunol Immunother. 53:935–945.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Galili U, Clark MR, Shohet SB, Buehler J
and Macher BA: Evolutionary relationship between the natural
anti-Gal antibody and the Gal alpha 1----3Gal epitope in primates.
Proc Natl Acad Sci USA. 84:1369–1373. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Galili U: Anti-Gal: An abundant human
natural antibody of multiple pathogeneses and clinical benefits.
Immunology. 140:1–11. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Abdel-Motal UM, Wigglesworth K and Galili
U: Mechanism for increased immunogenicity of vaccines that form in
vivo immune complexes with the natural anti-Gal antibody. Vaccine.
27:3072–3082. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Huai G, Qi P, Yang H and Wang Y:
Characteristics of α-Gal epitope, anti-Gal antibody, α1,3
galactosyltransferase and its clinical exploitation (Review). Int J
Mol Med. 37:11–20. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Galili U: Evolution of alpha
1,3galactosyltransferase and of the alpha-Gal epitope. Subcell
Biochem. 32:1–23. 1999.PubMed/NCBI
|
|
19
|
Yao X, Dong Z, Zhang Q, Wang Q and Lai D:
Epithelial ovarian cancer stem-like cells expressing α-gal epitopes
increase the immunogenicity of tumor associated antigens. BMC
Cancer. 15:9562015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yan Y, Li S, Jia T, Du X, Xu Y, Zhao Y, Li
L, Liang K, Liang W, Sun H and Li R: Combined therapy with CTL
cells and oncolytic adenovirus expressing IL-15-induced enhanced
antitumor activity. Tumour Biol. 36:4535–4543. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Carluccio S, Delbue S, Signorini L, Setola
E, Bagliani A, Della Valle A, Galli A, Ferrante P and Bregni M:
Generation of tumor-specific cytotoxic T-lymphocytes from the
peripheral blood of colorectal cancer patients for adoptive T-cell
transfer. J Cell Physiol. 230:1457–1465. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hunyadi J, András C, Szabó I, Szántó J,
Szluha K, Sipka S, Kovács P, Kiss A, Szegedi G, Altorjay I, et al:
Autologous dendritic cell based adoptive immunotherapy of patients
with colorectal cancer-A phase I–II study. Pathol Oncol Res.
20:357–365. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yoshida Y, Naito M, Yamada T, Aisu N,
Daibo K, Mera T, Tanaka T, Naito K, Yasumoto K, Kamigaki T, et al:
Adoptive chemoimmunotherapy using activated αβ T cells for stage IV
colorectal cancer. Anticancer Res. 36:3741–3746. 2016.PubMed/NCBI
|
|
25
|
Yoshida Y, Naito M, Yamada T, Aisu N,
Kojima D, Mera T, Tanaka T, Naito K, Yasumoto K, Kamigaki T, et al:
Clinical study on the medical value of combination therapy
involving adoptive immunotherapy and chemotherapy for stage IV
colorectal cancer (COMVI study). Anticancer Res. 37:3941–3946.
2017.PubMed/NCBI
|
|
26
|
Wang GZ, Tang XD, Lü MH, Gao JH, Liang GP,
Li N, Li CZ, Wu YY, Chen L, Cao YL, et al: Multiple antigenic
peptides of human heparanase elicit a much more potent immune
response against tumors. Cancer Prev Res (Phila). 4:1285–1295.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Shimizu H, Ito H, Kimura F, Togawa A,
Yoshidome H, Ohtsuka M, Kato A, Nukui Y and Miyazaki M: Decreased
cell-mediated immune status in colorectal cancer patients with
hepatic metastasis. Hepatogastroenterology. 52:1106–1109.
2005.PubMed/NCBI
|
|
28
|
Kwak Y, Koh J, Kim DW, Kang SB, Kim WH and
Lee HS: Immunoscore encompassing CD3+ and CD8+ T cell densities in
distant metastasis is a robust prognostic marker for advanced
colorectal cancer. Oncotarget. 7:81778–81790. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ho NI, Huis In't Veld LGM, Raaijmakers TK
and Adema GJ: Adjuvants enhancing cross-presentation by dendritic
cells: The key to more effective vaccines? Front Immunol.
9:28742018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ugur M and Mueller SN: T cell and
dendritic cell interactions in lymphoid organs: More than just
being in the right place at the right time. Immunol Rev.
289:115–128. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chu WM: Tumor necrosis factor. Cancer
Lett. 328:222–225. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Foged C, Hansen J and Agger EM: License to
kill: Formulation requirements for optimal priming of CD8(+) CTL
responses with particulate vaccine delivery systems. Eur J Pharm
Sci. 45:482–491. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gagnon SJ, Wang Z, Turner R, Damirjian M
and Biddison WE: MHC recognition by hapten-specific
HLA-A2-restricted CD8+ CTL. J Immunol. 171:2233–2241. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Guzmán-Flores JM and Portales-Pérez DP:
Mechanisms of suppression of regulatory T-cells (Treg). Gac Med
Mex. 149:630–638. 2013.(In Spanish). PubMed/NCBI
|
|
35
|
Schmidt A, Oberle N and Krammer PH:
Molecular mechanisms of treg-mediated T cell suppression. Front
Immunol. 3:512012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tian Y, Yuan C, Ma D, Zhang Y, Liu Y,
Zhang W, Hou F and Cui B: IL-21 and IL-12 inhibit differentiation
of Treg and TH17 cells and enhance cytotoxicity of peripheral blood
mononuclear cells in patients with cervical cancer. Int J Gynecol
Cancer. 21:1672–1678. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fujii S, Shimizu K, Okamoto Y, Kunii N,
Nakayama T, Motohashi S and Taniguchi M: NKT cells as an ideal
anti-tumor immunotherapeutic. Front Immunol. 4:4092013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Robertson FC, Berzofsky JA and Terabe M:
NKT cell networks in the regulation of tumor immunity. Front
Immunol. 5:5432014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Taniguchi M, Harada M, Dashtsoodol N and
Kojo S: Discovery of NKT cells and development of NKT cell-targeted
anti-tumor immunotherapy. Proc Jpn Acad Ser B Phys Biol Sci.
91:292–304. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Furukawa K, Tanemura M, Miyoshi E, Eguchi
H, Nagano H, Matsunami K, Nagaoka S, Yamada D, Asaoka T, Noda T, et
al: A practical approach to pancreatic cancer immunotherapy using
resected tumor lysate vaccines processed to express α-gal epitopes.
PLoS One. 12:e01849012017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xue D, Liang Y, Duan S, He J, Su J, Zhu J,
Hu N, Liu J, Zhao Y and Lu X: Enhanced anti-tumor immunity against
breast cancer induced by whole tumor cell vaccines genetically
modified expressing α-Gal epitopes. Oncol Rep. 36:2843–2851. 2016.
View Article : Google Scholar : PubMed/NCBI
|