Introduction
Epithelial ovarian cancer (EOC) is a common
malignant tumor found in the female reproductive system (1). Given its deep anatomical location, easy
recurrence, easy metastasis and high susceptibility to drug
resistance, EOC is the most common cause of mortality among
gynecologic malignancies in China (2). EOC is mainly treated by surgery
supplemented with chemotherapy, radiotherapy and biotherapy
(3). However, the majority of
patients are diagnosed at an advanced stage, easily exhibiting
chemotherapy resistance. Consequently, chemotherapy effect is
reduced, and the 5-year survival rate is low (25–30%) (4,5).
Multidrug resistance (MDR) is one of the major reasons for the
failure of EOC chemotherapy during the advanced stage (6). This drug resistance is associated with
the molecular activity and expression of drug pumps, abnormal pH of
tumor cell, DNA damage repair ability, drug detoxification,
apoptotic pathway and methylation of a number of genes, including
BRAC1, BRAC2, MLH1 and FBX032 (7–10). MDR
may be divided into primary drug resistance, that exists prior to
chemotherapy treatment, and acquired drug resistance, which
develops during chemotherapy (11).
MDR can reduce the concentration of chemotherapeutic drugs in tumor
cells and, therefore, the sensitivity of these tumor cells to drugs
(12). In addition, the changes in
methylation state of tumor cells can decrease the overall
methylation state or increase the local methylation, therefore,
causing tumor cells to resist chemotherapeutic drugs (13). The multidrug-resistance 1 (MDR1) gene
product P glycoprotein is an efflux pump that actively transports
substrates. In addition, lung resistance protein is a small
subcellular structure located at cytoplasmic vaults that may be in
charge of the subsequent exocytosis of agents from the cell.
Furthermore, glutathione S-transferase-P1 is a phase II metabolic
enzyme that protects cells from anticancer drug-induced injury. All
aforementioned genes are involved in MDR (14). Therefore, the present study aims to
investigate the expression and the role of the methylation state of
MDR in different ovarian tissues of EOC, and to analyze the
association between the methylation state of MDR and the
clinicopathological features of EOC. In addition, the presents
study aims to provide a theoretical basis for predicting individual
responsiveness to chemotherapy and prognosis, and improving
treatment.
Materials and methods
Patients and specimens
A total of fresh specimens (57 cases of PEOC, 34
cases of borderline adenoma and 21 cases of benign adenoma) were
collected between March 2009 and July 2011 in Taihe Hospital
(Shiyan, China). The average age of patients was between 35 and 71
years. Histologic cell types were as follows: 30 cases of serous
adenocarcinoma, 20 cases of mucinous adenocarcinoma and 7 cases of
endometrioid carcinoma in PEOC; 20 cases of serous adenocarcinoma,
10 cases of mucinous adenocarcinoma and 4 cases of endometrioid
carcinoma in borderline adenoma; 13 cases of serous adenocarcinoma,
7 cases of mucinous adenocarcinoma and 1 cases of endometrioid
carcinoma in benign adenoma. Clinical staging using the standards
established by the International Federation of Obstetricians and
Gynecologists (15) identified the
following number of cases for each stage: Stage I, 25 cases; stage
II, 19 cases; and stage III, 13 cases. Of these cases, 12 were well
differentiated, 20 were moderately differentiated and 25 were
poorly differentiated carcinomas. Histological diagnosis was based
on the histological typing system of the World Health Organization
(16) and the stage of disease was
determined according to the International Federation of Gynecology
and Obstetrics staging system (17).
The diagnosis was confirmed by at least two experienced
pathologists at the Department of Pathology of Taihe Hospital in a
blinded manner. All patients had not received chemotherapy and
radiation therapy prior to sample collection. Samples were stored
in liquid nitrogen for future use. The integrity of clinical data
was maintained, and the patients were followed up between 6 and 60
months. In accordance with the Declaration of Helsinki, the present
study was approved by the Ethics Committee of Taihe Hospital
(Shiyan, China), and written informed consent were obtained from
all patients or their families.
Cell culture and zebularine
treatment
Ovarian cancer cell line, A2780 and A2780/DDP
(platinum-resistant), were provided by the Fourth Military Medical
University (Xi'an, China) and cultivated at 37°C with 5%
CO2 in RPMI-1640 medium for 48 h, containing 10% fetal
bovine serum (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA,
USA), 1.5% L-glutamine and 1% penicillin/streptomycin. Two to four
generations of well-grown cells were obtained for testing, and the
remaining cells were preserved in liquid nitrogen. A2780/DDP were
treated with various doses (0.0, 0.2 and 0.5 mM) of zebularine, a
DNA Methyl Transferase inhibitor that acts similarly to 5-aza-dC,
for 48 h at 37°C and were subsequently collected for cell
apoptosis. A total of 1×106 cells were collected and
washed with PBS three times. The collected cells were incubated
with 5 µl propidium iodide (PI; Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany) and 5 µl fluorescein isothiocyanate (FITC;
Annexin V-FITC/PI kit; cat. no. E606336; Sangon Biotech Co., Ltd.,
Shanghai, China) for 1 h at 37°C.
Cell apoptosis was detected using flow cytometry
(Beckman Coulter, Inc., Brea, CA, USA) and cell Modifit software
(version 3.1; Verity Software House, Inc., Topsham, ME, USA). All
experiments were repeated six times.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA were extracted from tissues and cells
using TRIzol® Reagent (Thermo Fisher Scientific, Inc.),
according to the manufacturer's protocols. The absorbance of RNA
was determined at a wavelength of 260 and 280 nm with NanoDrop-2000
(Thermo Fisher Scientific, Inc.). cDNA was synthesized from RNA
with Reverse Transcription system (Promega Corporation, Madison,
WI, USA) and was used immediately or stored at −80°C until use. The
primers are presented in Table I.
PCR amplification system (Sangon Biotech Co., Ltd.) included the
following: Mg2+ 2.4 µl, 5′ and 3′ primer 2 µl, 2 mmol/l
dNTP 1.5 µl, 10X SYBR-Green I 1 µl, Taq 0.3 µl, 10× Buffer 3 µl,
cDNA5 µl, with sterile water total volume filled 30 µl. The
thermocycling conditions were as follows: 95°C denaturation for 5
min, 94°C for 30 sec, 60°C for 30 sec, 72°C for 1 min with 35
cycles. Dissociation curve analysis was performed at the end point
of the PCR cycles. GSTP1, LRP and MDR1 expression levels were
normalized to GAPDH in each sample, and were determined using the
2−∆∆Cq method (18).
 | Table I.Primers sequences of gene expression
and methylation. |
Table I.
Primers sequences of gene expression
and methylation.
| Gene | Forward
(5′-3′) | Reverse
(5′-3′) | Product (bp) |
|---|
| MDR1 |
CCCATCATTGCAATAGCAGG |
TGTTCAAACTTCTGCTCCTGA | 158 |
| LRP |
GTCTTCGGGCCTGAGCTGGTGTCG |
CTTGGCCGTCTCTTGGGGGTCCTT | 240 |
| GSTP1 |
CCAGAACCAGGGAGGCAAGA |
GAGGCGCCCCACATATGCT | 325 |
| RASSF1A |
GGCGTCGTGCGCAAAGGCC | GGG
TGGCTTCTTGCTGGAGGG | 330 |
| GAPDH |
GAAGGTGAAGGTCGGAGTC |
GAAGATGGTGATGGGATTTC | 226 |
| Methylation |
GTGTTAACGCGTTGCGTATC |
AACCCCGCGAACTAAAAACGA | 93 |
| Un-methylation |
TTTGGTTGGAGTGTGTTAATGTG |
CAAACCCCACAAACTAAAAACAA | 105 |
Western blot analysis
Total protein of cells and tissues was extracted
using the Total Protein Extraction kit (Sangon Biotech Co., Ltd.)
and concentrations were determined with NanoDrop-2000 (NanoDrop;
Thermo Fisher Scientific, Inc., Wilmington, DE, USA). A total 10 µg
protein was loaded onto 10% SDS-PAGE for electrophoresis and
transferred to polyvinylidene difluoride membranes. Following
transfer, protein blots were blocked with 5% non-fat dry
milk-TBS-0.1% Tween-20 for 2 h at room temperature and subsequently
washed three times with TBS-0.1% Tween-20 for 10 min, and incubated
overnight at 4°C with primary polyclonal antibodies against P-GP
(1:1,000 dilution; cat. no. MAB-0237; Maxim-Bio Ltd., Fuzhou,
China), LRP (1:1,000 dilution; cat. no. MAB-0319; Maxim-Bio Ltd.)
and GSTP1 (1:1,000 dilution; cat. no. MAB-0583; Maxim-Bio Ltd.),
according to the manufacturer's protocols. Signals were detected
following incubation for 2 h at room temperature in horseradish
peroxidase-conjugated secondary antibody (1:1,000 dilution; cat.
no. KIT-C10; Maxim-Bio Ltd.). Protein bands were detected using an
enhanced chemiluminescence kit (GE Healthcare, Chicago, IL, USA).
The optical density of the bands was determined using ImageJ 2×
software (version 2.1.4.7; National Institutes of Health, Bethesda,
MD, USA). GAPDH (1:1,000 dilution; cat. no. MAB-97166; Cell
Signaling Technology, Inc., Danvers, MA, USA) acted as the
reference protein for the loading control.
DNA isolation and bisulfite conversion
of target DNA
Tissue DNA was extracted through proteinase K
treatment, phenol/chloroform/isoamylalcohol extraction and ethanol
precipitation in sequence. Bisulfite conversion was carried out
with modified Herman's method (19).
The conversion was based on the principle that DNA, treated with
bisulfite, would result in the conversion of unmethylated cytosine
residues into uracil. Methylated cytosine residues, on the other
hand, would remain unchanged. Therefore, the DNA sequences of
methylated and unmethylated genomic regions following bisulfite
conversion would differ and would be distinguished by
sequence-specific PCR primers. The bisulfite conversion of target
DNA was stored at −20°C prior to methylation-specific PCR (MSP) for
2 months.
MSP
The primers that were used for MSP of the Ras
association domain family member 1 (RASSF1A) gene are presented in
Table I. PCR amplification system
included: Mg2+ 2.4 µl, 0.8 µl each of the 5′ and 3′
primer, Taq 0.3 µl, dNTP 1.6 µl, 10X buffer 2 µl, bisulfite
conversion of target DNA 0.5 µl, total volume filled to 20 µl with
sterile water. The thermocycling conditions were as follows: 94°C
denaturation for 5 min, 94°C for 30 sec, 60°C for 45 sec and 72°C
for 1 min with 35 cycles. Additionally, a final extension for 5 min
at 70°C was performed. The reaction products were separated
electrophoretically on a 2% agarose gel and stained with ethidium
bromide for further confirmation of the PCR products.
Statistical analysis
Statistical calculations were performed using SPSS
software 16.0 (SPSS, Inc., Chicago, IL, USA), and P<0.05 was
considered to indicate a statistically significant difference. The
measured data were expressed as the mean ± standard deviation.
One-way analysis of variance followed by Dunnett's T3 post hoc
test, two-tailed χ2 test and multivariate logistic
regression analysis were used to compare difference among groups.
Kaplan-Meier analysis was used to plot survival curves and the
survival curves were compared using the two-sided data log-rank
method.
Results
mRNA/protein expression of MDR1
(P-gp), LRP, GSTP1, RASSF1A and methylation frequency of RASSF1A in
all samples
The mRNA expression of MDR1, LRP and GSTP1 was
detected in 57 cases of PEOC, 34 cases of borderline adenoma and 21
cases of benign adenoma by RT-qPCR. The resistance genes/proteins
MDR1 (P-gp), LRP and GSTP1 were highly expressed in PEOC compared
with in borderline adenoma, and the resistance gene positive
expression rates were 56.1, 78.9 and 57.9%, while the protein
positive expression rates were 52.6, 70.2 and 56.1%, respectively.
In borderline adenoma, their positive expression rates were 26.5,
35.3 and 26.5%, while the protein positive expression rates were
23.5, 29.4 and 23.5%, respectively (data not shown). However, in
benign adenoma, the resistance genes/proteins were either poorly
expressed or not expressed at all (Fig.
1A-C). The expression levels of the resistance genes/proteins
were significantly higher in PEOC tissues/borderline adenoma
tissues compared with benign adenoma tissues (P<0.001).
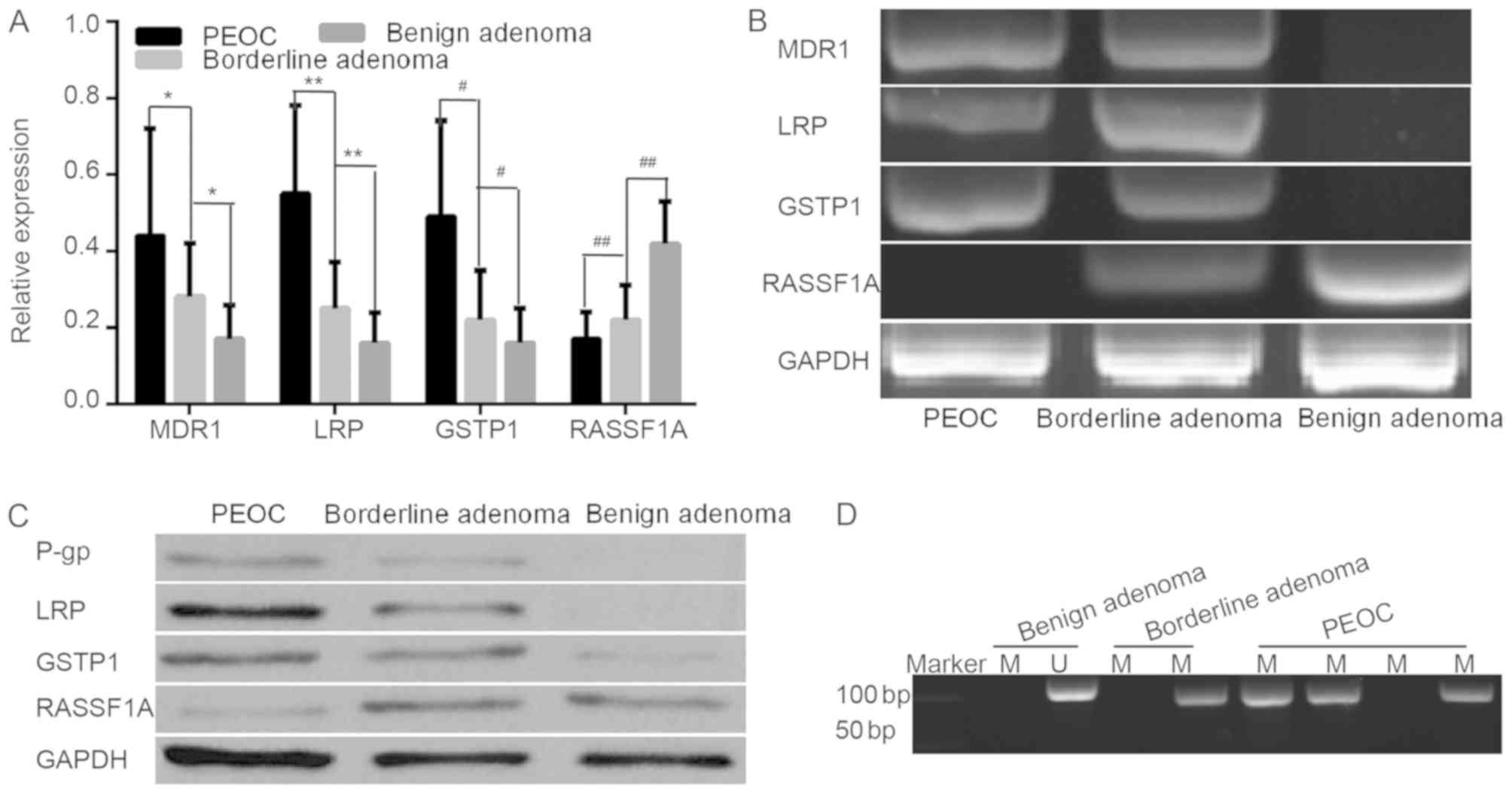 | Figure 1.The mRNA/protein expression of MDR1
(P-gp), LRP, GSTP1, RASSF1A and methylation frequency of RASSF1A in
all samples. (A) mRNA relative expression levels of resistance
genes in PEOC, benign adenoma and borderline adenoma. (B) Different
gene expressions in PEOC, borderline adenoma and benign adenoma.
(C) Different protein expressions in PEOC, borderline adenoma and
benign adenoma. (D) Representation of methylated and unmethylated
products of RASSF1A in all sample. *P<0.01, **P<0.01,
#P<0.01, ##P<0.01. M, methylated; U,
un-methylated; MDR1, multidrug resistance 1; LRP, lung resistance
protein; GSTP1, placental glutathione S-transferase-P1; RASSF1A,
Ras association domain family member; P-gp, product P glycoprotein;
PEOC, primary epithelial ovarian cancer. |
The methylation status of RASSF1A was analyzed using
MSP assay in all samples. Methylation products were indicated with
33/57 (57.9%), 13/34 (38.2%) and 3/21 (14.3%) in PEOC, borderline
adenoma and benign adenoma, respectively. Hypermethylation and low
expression of RASSF1A gene was detected in all cancer tissues
(Fig. 1D).
mRNA expression, gene methylation and
survival time in different clinicopathological data of PEOC
The expression of resistance genes was significantly
higher in well and moderately differentiated carcinoma compared
with poorly differentiated carcinomas (P<0.05; Table II). In addition, the difference in
other clinicopathological factors, including age, pathological
type, and clinical stage was not statistically significant. The
survival time was higher in well and moderately differentiated
carcinoma compared with poorly differentiated carcinomas, however
not statistically significant. The association between the
expression levels of the resistance genes/gene methylation and the
clinicopathological features are presented in Table II.
 | Table II.Association between expression of
gene, methylation, survival time and clinicopathological parameters
in primary epithelial ovarian cancer. |
Table II.
Association between expression of
gene, methylation, survival time and clinicopathological parameters
in primary epithelial ovarian cancer.
| Study groups | n | Multi-drug
resistance gene | Lung resistance
protein | Placental
glutathione S-transferase-P1 | Methylation
Positive % | Survival time |
|---|
| Age (years) |
|
<50 | 19 | 0.45±0.31 | 0.61±0.25 | 0.50±0.24 | 47.4 (9/19) | 41.0±19.1 |
|
≥50 | 38 | 0.44±0.27 | 0.52±0.22 | 0.43±0.26 | 63.2 (24/38) | 39.2±16.5 |
| Histologic
types |
| Serous
adenocarcinoma | 30 | 0.50±0.27 | 0.61±0.26 | 0.54±0.21 | 53.3 (16/30) | 41.3±18.3 |
|
Mucinous + Endometrioid | 27 | 0.37±0.28 | 0.50±0.19 | 0.43±0.28 | 63.0 (17/27) | 38.2±16.3 |
| Stage |
|
I+II | 25 | 0.49±0.29 | 0.62±0.24 | 0.54±0.23 | 40.0
(10/25)b | 40.2±18.4 |
|
III+IV | 32 | 0.40±0.26 | 0.51±0.22 | 0.45±0.27 | 71.9 (23/32) | 39.5±16.6 |
|
Differentiation |
|
Well+Moderately | 32 |
0.51±0.26a |
0.61±0.25a |
0.56±0.23a | 43.8
(14/32)c | 41.6±18.1 |
|
Poorly | 25 | 0.35±0.28 | 0.48±0.18 | 0.41±0.27 | 72.0 (18/25) | 37.5±16.3 |
mRNA/protein expression of MDR1
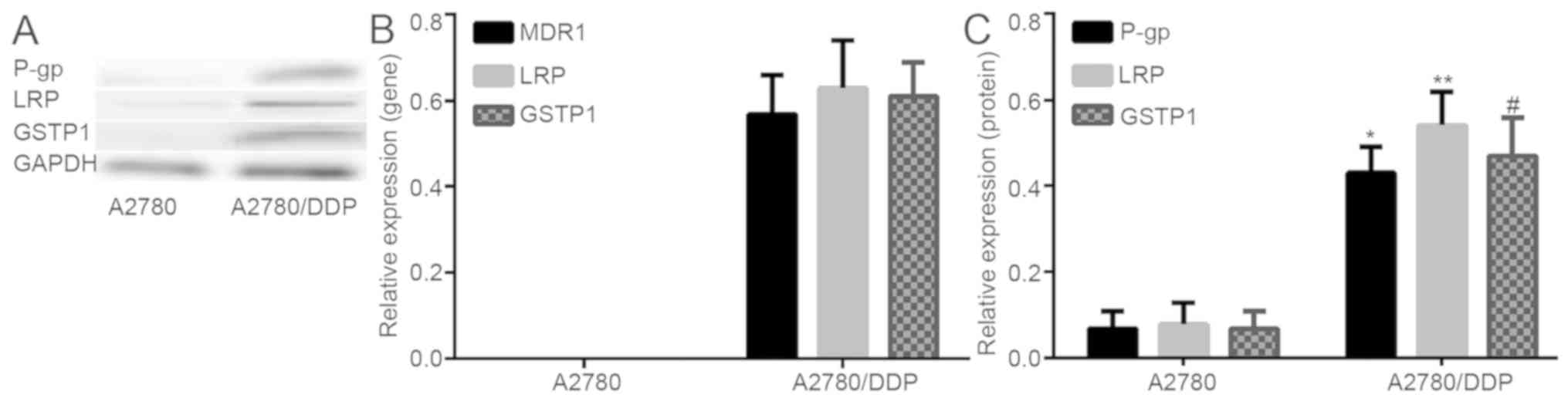
(P-gp), LRP and GSTP1 in A2780 and A2780/DDP
To study the role of methylation in the acquired
drug-resistance of ovarian cancer, the changes of drug-resistance
gene/protein expression in A2780 and A2780/DDP cells was examined.
It was indicated that the resistance genes/protein were either
poorly expressed or not expressed in A2780 cell lines, however
highly expressed in A2780/DDP. The difference indicated to be
statistically significant (Fig. 2).
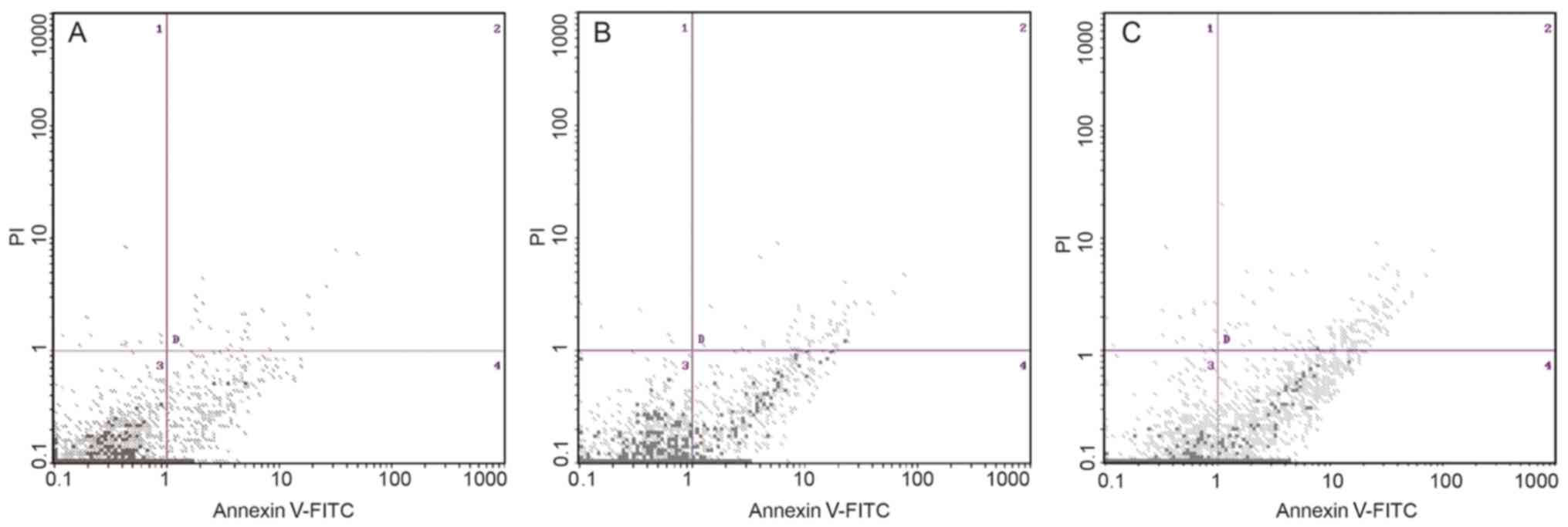
In addition, following further treatment of A2780/DDP with
different concentrations of de-methylation reagents, including
zebularine, the expression levels of drug resistance genes/proteins
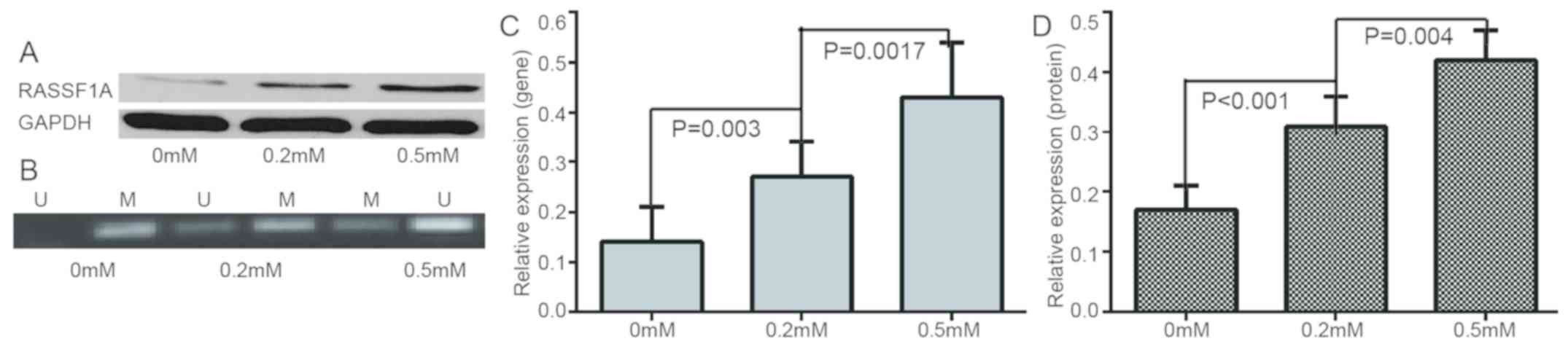
were decreased and apoptotic rate was increased (Figs. 3 and 4). RASSF1A gene methylation was weakened
and unmethylation was increased. Furthermore, the expression level
of RASSF1A gene/protein expression gradually increased (Fig. 5).
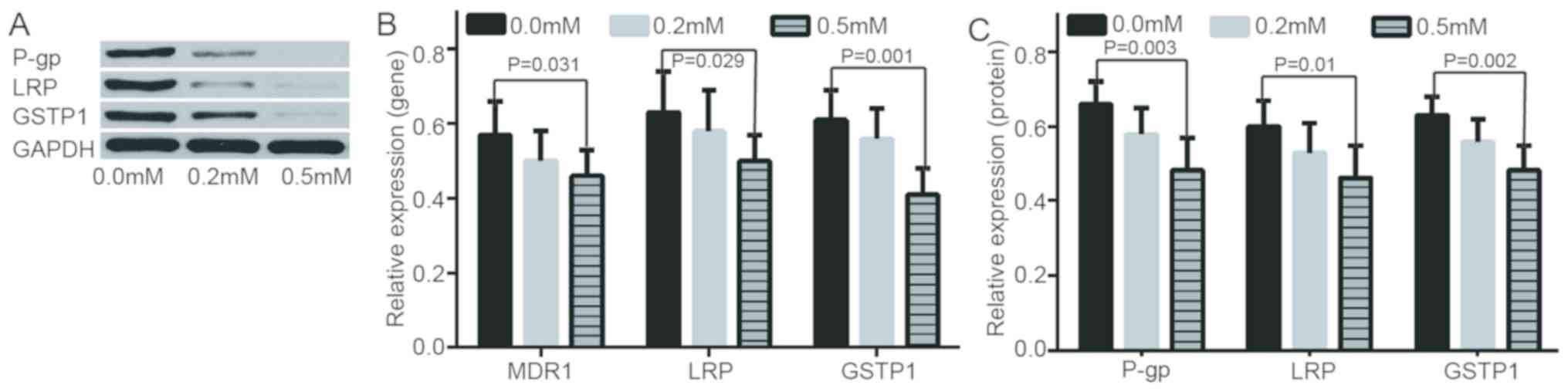 | Figure 3.Evaluation of expression of resistance
genes/proteins at 0.0 mM, 0.2 mM, 0.5 mM of zebularine. (A)
Different protein expressions in LRP, GSTP1 and P-gp at different
concentrations of zebularine. (B) mRNA relative expressions of
MDR1, GSTP1 and P-gp gene at 0.0 mM, 0.2 mM, 0.5 m zebularine. (C)
Relative expressions of LRP, GSTP1 and P-gp protein at different
concentrations of zebularine. MDR1, multidrug resistance 1; LRP,
lung resistance protein; GSTP1, placental glutathione
S-transferase-P1; P-gp, product P glycoprotein. |
Clinicopathology, gene expression
level, gene methylation and survival rate of patients with
PEOC
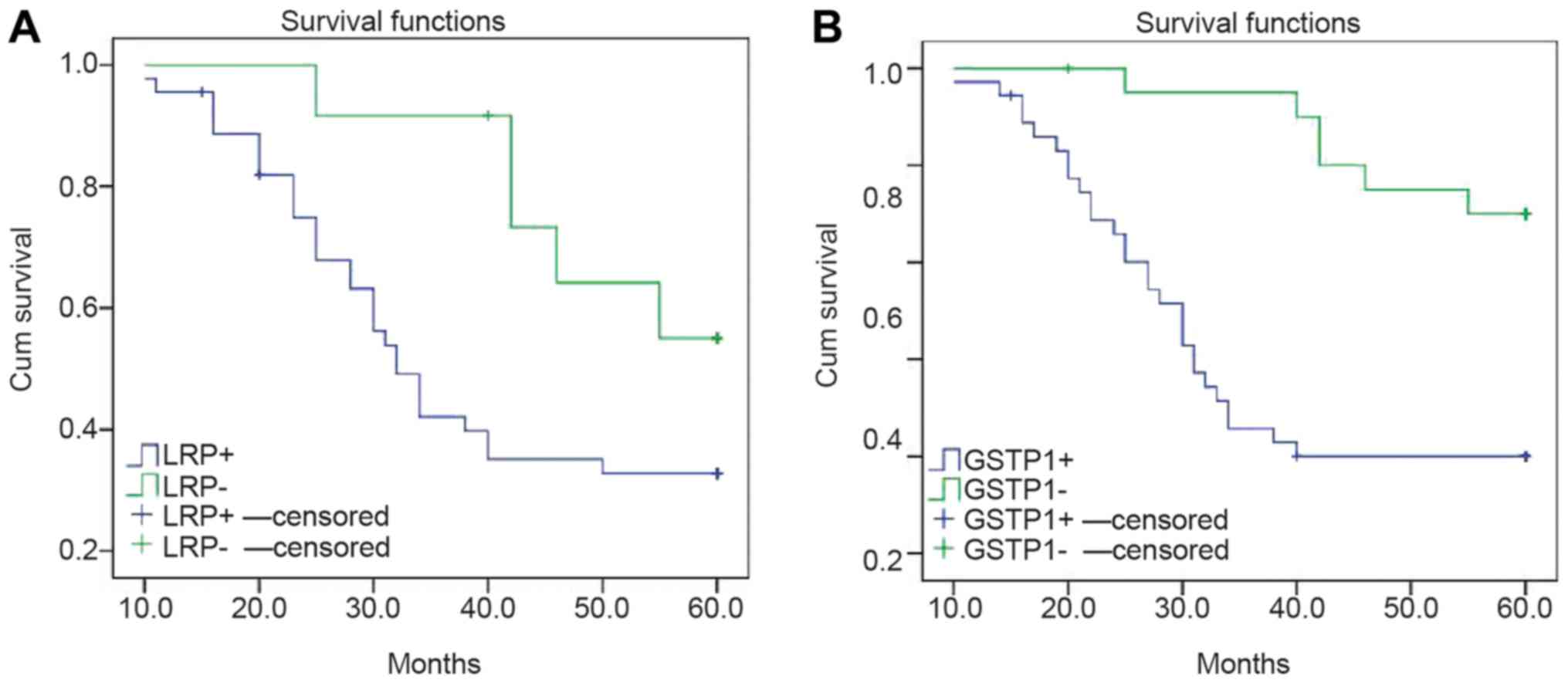
Risk modeling and multivariate logistic analysis
indicated that the prognostic factors of EOC were associated with
high expression levels of LRP and GSTP1 (Table III), however not with age, stage,
degree of differentiation, RASSF1A methylation and MDR1 expression
(data not shown). The cases were categorized into positive and
negative groups, according to the expression levels of LRP and
GSTP1. The survival curve of the negative group, non-expression of
LRP and GSTP1, was significantly higher compared with the positive
group (P<0.05; Fig. 6).
 | Table III.Multivariate logistics analysis on
influencing factors for primary epithelial ovarian cancer
prognosis. |
Table III.
Multivariate logistics analysis on
influencing factors for primary epithelial ovarian cancer
prognosis.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
|
Characteristics | B | Exp (B) | P-value | B | Exp (B) | P-value |
|---|
| Lung resistance
protein positive vs. negative | 2.72 | 0.065 |
0.003 |
|
|
|
| Placental
glutathione S-transferase-P1 positive vs. negative | 3.06 | 0.047 | <0.001 | 1.58 | 4.84 | 0.001 |
Discussion
Tumor cells may develop MDR to various chemotherapy
regimens, such as paclitaxel and carboplatin or docetaxel and
carboplatin (9). MDR resistance
reduces the sensitivity of tumor cells to chemotherapy drugs,
leading to the failure of ovarian cancer treatment and disease
progression. MDR was identified as an important cause leading to
failure of cancer treatment (9).
The MDR1 gene encodes the glycoprotein drug
transporter P-gp. Elevated MDR1 genes or P-gp protein have been
reported to be associated with drug resistance (20). MDR1/P-gp expression is mainly
mediated by alkylating agents (21).
LRP serves an important role in various tumors, including gastric
cancer and non-small cell lung cancer, with drug resistance
(22,23). Its increased expression can mainly
make tumor cells resistant to cisplatin (24). The aberrant methylation of RASSF1A
gene serves an important role in the occurrence and development of
various tumors, including bladder cancer and thyroid cancer
(25,26), and is associated with drug resistance
and tumor prognosis (27,28). The purpose of the present study was
to detect the expression of drug resistance genes and aberrant
methylation of RASSF1A in different types of ovarian tissues and to
investigate their role in the primary resistance of ovarian
cancer.
This study indicated that the expression levels of
resistance genes MDR1, LRP and GSTP1 in epithelial ovarian cancer
were significantly higher compared with those in borderline adenoma
and benign adenoma. Furthermore, all patients had not received any
prior chemotherapy and radiation therapy. This phenomenon suggested
that primary drug resistance was associated with PEOC.
Co-expression of three genes/proteins in cancer tissues, suggesting
the expression of drug resistance genes, is indicated to be
regulated by certain factors, such as DNA methylation, commonly
causing ovarian cancer MDR (9).
The expression level of the drug resistance genes
was higher in well and moderately differentiated carcinomas
compared with poorly differentiated tissues. This finding suggested
that high degree of cancer differentiation indicates strong
resistance to chemotherapeutic drugs. In addition, an association
was indicated to exist between RASSF1A methylation state and PEOC
differentiation degree. Choi et al (29) reported that the abnormal methylation
of RASSF1A gene indicated a gradual increasing trend from benign
tumor to cancer. This result indicated that abnormal methylation
can gradually accumulate and finally result in PEOC occurrence. In
addition, methylation changes commonly occur prior to solid tumors.
This change also participates in the occurrence and development of
malignant epithelial ovarian tumor (9). In addition, RASSF1A gene can assist the
paclitaxel chemotherapeutic drugs in interfering with the spindle
polymerization in normal cases (28). The loss of RASSF1A protein expression
can cause the drug resistance to paclitaxel in PEOC cell, however,
the activation of RASSF1A expression through demethylation can
recover the paclitaxel sensitivity (29). This result suggests that RASSF1A
methylation may be associated with PEOC drug resistance (30). In the present study, RASSF1A
methylation was not a prognostic risk factor, suggesting that MDR
is a complex process of multigene, multistep, and the cross action
of multifactor.
Zebularine is a DNA methyl transferase inhibitor,
acting similarly to 5-aza-dC, however is more specific and
therefore, less toxic compared with 5-AzaC (31). In the present study, zebularine
treatment induced cell apoptosis at doses of 0.5 mM in A2780/DDP,
implying that zebularine-induced A2780/DDP cell growth inhibition
was due to cell apoptosis. However, with the increase of zebularine
concentration, the genes/protein expression decreased. This result
implied that zebularine is an important antiproliferative agent
against A2780/DDP. With increasing concentration of zebularine, the
level of methylation decreased and the level of un-methylation
increased. This finding indicated that zebularine could reverse the
methylation state of the RASSF1A gene in A2780/DDP. RT-qPCR and
western blot analysis also confirmed that zebularine may increase
the expression of RASSF1A gene at mRNA and protein expression
levels. Low expression caused by hypermethylation of RASSF1A gene
may serve an important role in cancer-acquired resistance in
PEOC.
High expression of resistance genes is the main
cause of tumor cell resistance to chemotherapy. Hou et al
(32) reported that the drug
resistance of P-gp was proportionate to its high expression in PEOC
drug-resistant strain. Curcumin can reduce P-gp biosynthesis and
inhibit P-gp biological activity to enhance the cytotoxicity of
drugs to tumor cell and reduce the drug resistance of tumor cells.
Furthermore, Cao et al (33)
demonstrated that LRP expression was reduced subsequent to curcumin
acting on the drug-resistant hepatoma cells. Chemotherapy drugs
significantly increased the intracellular concentration and
toxicity, therefore, reversing drug resistance (33). Wang et al (34) indicated that the TGFBI methylation
level of the cell line is higher compared with that of sensitive
cells. Following 5-aza-dc treatment, the TGFBI mRNA and protein
expression levels in drug resistant cell line increased
significantly. This result implied that the re-expression of TGFBI
can reverse paclitaxel drug resistance (34). Therefore, reduced expression of
drug-resistant genes is likely to reverse the drug resistance.
Further studies on reversing solid tumor drug resistance are
required to provide an experimental basis.
Yang et al (11) reported that the MDR1 expression in
patients with cervical cancer of the negative-group survival curves
was higher compared with that of the positive group. In the present
study, high LRP and GSTP1 mRNA expression were associated with the
survival rates of patients with PEOC. The expression of LRP and
GSTP1 in the patients of the negative-group survival curves was
higher compared with the patients in the positive group. This
result suggested that the expression of LRP and GSTP1 in patients
with PEOC is a high-risk prognostic factor. In addition, Chiang
et al (35) reported that
patients with zinc finger MYND-type containing 10 (BLU) methylation
exhibited short progression free survival (PFS) and overall
survival time. BLU can also enhance paclitaxel to induce the
apoptosis of EOC cells subsequent to recovering its expression
(35). BLU methylation may be an
indicator in the evaluation of EOC prognosis. This result reveals
that the change in methylation state of drug-resistant genes is
also likely to reverse the drug resistance (35).
In conclusion, high expression of resistance genes
and methylation of RASSF1A gene may be one of the important
mechanisms of primary ovarian cancer MDR. The expression and
methylation detection of resistance genes may provide novel
guidance to predict the effect of ovarian cancer chemotherapy and
to assist the chemotherapy regimen. However, due to cost, a number
of experiments were not carried out, including cell transfection
and experiments on animals. In subsequent experiments, the aim is
to provide further studies on the mechanism of drug resistance.
Acknowledgements
Not applicable.
Funding
The present study was funded by Taihe Hospital
Projects (grant nos. 2016JZ26, 2017JJXM044 and 2014JJXM019).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
BG and DL conceived and designed the study. FY, WC
and YL performed the experiments. RL and XH analyzed the data. BG
wrote the manuscript.
Ethics approval and consent to
participate
In accordance with the Declaration of Helsinki, the
present study was approved by the Ethics Committee of Taihe
Hospital (Shiyan, China), and written informed consent was obtained
from all patients or their families.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lheureux S, Gourley C, Vergote I and Oza
AM: Epithelial ovarian cancer. Lancet. 393:1240–1253. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Marcus CS, Maxwell GL, Darcy KM, Hamilton
CA and McGuire WP: Current approaches and challenges in managing
and monitoring treatment response in ovarian cancer. J Cancer.
5:25–30. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ledermann JA, Raja FA, Fotopoulou C,
Gonzalez-Martin A, Colombo N and Sessa C; ESMO Guidelines Working
Group, : Newly diagnosed and relapsed epithelial ovarian carcinoma:
ESMO Clinical Practice Guidelines for diagnosis, treatment and
follow-up. Ann Oncol. 24:vi24–vi32. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Asgari Z, Rouholamin S, Hosseini R,
Sepidarkish M, Hafizi L and Javaheri A: Comparing ovarian reserve
after laparoscopic excision of endometriotic cysts and hemostasis
achieved either by bipolar coagulation or suturing: A randomized
clinical trial. Arch Gynecol Obstet. 293:1015–1022. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Qin L, Qiu H, Zhang M, Zhang F, Yang H,
Yang L, Jia L, Qin K, Jia L, Dou X, et al: Soluble CD40 ligands
sensitize the epithelial ovarian cancer cells to cisplatin
treatment. Biomed Pharmacother. 79:166–175. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang F, Gao B, Chen W, Du E, Liang Y, Hu X
and Yang X: Expression of resistance gene and prognosis of
chemotherapy in primary epithelial ovarian cancer. Medicine
(Baltimore). 97:e123642018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Matei D, Fang F, Shen C, Schilder J,
Arnold A, Zeng Y, Berry WA, Huang T and Nephew KP: Epigenetic
resensitization to platinum in ovarian cancer. Cancer Res.
72:2197–2205. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yin F, Liu X, Li D, Wang Q, Zhang W and Li
L: Tumor suppressor genes associated with drug resistance in
ovarian cancer (review). Oncol Rep. 30:3–10. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chaudhry P, Srinivasan R and Patel FD:
Utility of gene promoter methylation in prediction of response to
platinum-based chemotherapy in epithelial ovarian cancer (EOC).
Cancer Invest. 27:877–884. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang F, Gao B, Li R, Li W, Chen W, Yu Z
and Zhang J: Expression levels of resistant genes affect cervical
cancer prognosis. Mol Med Rep. 15:2802–2806. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Koh I, Hinoi T, Sentani K, Hirata E,
Nosaka S, Niitsu H, Miguchi M, Adachi T, Yasui W, Ohdan H and Kudo
Y: Regulation of multidrug resistance 1 expression by CDX2 in
ovarian mucinous adenocarcinoma. Cancer Med. 5:1546–1555. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Balch C, Huang TH, Brown R and Nephew KP:
The epigenetics of ovarian cancer drug resistance and
resensitization. Am J Obstet Gynecol. 191:1552–1572. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lu C, Shan Z, Li C and Yang L: MiR-129
regulates cisplatin-resistance in human gastric cancer cells by
targeting P-gp. Biomed Pharmacother. 86:450–456. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Javadi S, Ganeshan DM, Qayyum A, Iyer RB
and Bhosale P: Ovarian cancer, the revised FIGO staging system and
the role of imaging. AJR Am J Roentgenol. 206:1351–1360. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kurman RJ, Carcangiu ML, Herrington CS and
Young RH: WHO Classification of Tumours of Female Reproductive
Organs. (4th). International Agency for Research on Cancer.
3072014.
|
|
17
|
Prat J; FIGO Committee on Gynecologic
Oncology, : Staging classification for cancer of the ovary,
fallopian tube, and peritoneum. Int J Gynaecol Obstet. 124:1–5.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Herman JG, Graff JR, Myöhänen S, Nelkin BD
and Baylin SB: Methylation-specific PCR: A novel PCR assay for
methylation status of CpG islands. Proc Natl Acad Sci USA.
93:9821–9826. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yuan Z, Shi X, Qiu Y, Jia T, Yuan X, Zou
Y, Liu C, Yu H, Yuan Y, He X, et al: Reversal of P-gp-mediated
multidrug resistance in colon cancer by cinobufagin. Oncol Rep.
37:1815–1825. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Stewart DJ: Tumor and host factors that
may limit efficacy of chemotherapy in non-small cell and small cell
lung cancer. Crit Rev Oncol Hematol. 75:173–264. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang KG, Qin CY, Wang HQ, Wang JX and
Wang QM: The effect of TRAIL on the expression of multidrug
resistant genes MDR1, LRP and GST-π in drug-resistant gastric
cancer cell SGC7901/VCR. Hepatogastroenterology. 59:2672–2676.
2012.PubMed/NCBI
|
|
23
|
Wei H, Lu W, Li M, Zhang Q and Lu S:
Concomitance of P-gp/LRP expression with EGFR mutations in exons 19
and 21 in non-small cell lung cancers. Yonsei Med J. 57:50–57.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jiao JW and Wen F: Tanshinone IIA acts via
p38 MAPK to induce apoptosis and the down-regulation of ERCC1 and
lung-resistance protein in cisplatin-resistant ovarian cancer
cells. Oncol Rep. 25:781–788. 2011.PubMed/NCBI
|
|
25
|
Zhan L, Zhang B, Tan Y, Yang C, Huang C,
Wu Q, Zhang Y, Chen X, Zhou M and Shu A: Quantitative assessment of
the relationship between RASSF1A gene promoter methylation and
bladder cancer (PRISMA). Medicine (Baltimore). 96:e60972017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shou F, Xu F, Li G, Zhao Z, Mao Y, Yang F,
Wang H and Guo H: RASSF1A promoter methylation is associated with
increased risk of thyroid cancer: A meta-analysis. Onco Targets
Ther. 10:247–257. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Vos MD, Martinez A, Elam C, Dallol A,
Taylor BJ, Latif F and Clark GJ: A role for the RASSF1A tumor
suppressor in the regulation of tubulin polymerization and genomic
stability. Cancer Res. 64:4244–4250. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kassler S, Donninger H, Birrer MJ and
Clark GJ: RASSF1A and the taxol response in ovarian cancer. Mol
Biol Int. 2012:2632672012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Choi YL, Kang SY, Shin YK, Choi JS, Kim
SH, Lee SJ, Bae DS and Ahn G: Aberrant hypermethylation of RASSF1A
promoter in ovarian borderline tumors and carcinomas. Virchows
Arch. 448:331–336. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Pronina IV, Loginov VI, Kholdyrev DS,
Kazubskaia TP and Braga ÉA: Alterations of expression level of
RASSFIA gene in primary epithelial tumors of various locations. Mol
Biol (Mosk). 46:260–268. 2012.(In Russian). View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Balch C, Yan P, Craft T, Young S, Skalnik
DG, Huang TH and Nephew KP: Antimitogenic and chemosensitizing
effects of the methylation inhibitor zebularine in ovarian cancer.
Mol Cancer Ther. 4:1505–1514. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hou XL, Takahashi K, Tanaka K, Tougou K,
Qiu F, Komatsu K, Takahashi K and Azuma J: Curcuma drugs and
curcumin regulate the expression and function of P-gp in Caco-2
cells in completely opposite ways. Int J Pharm. 358:224–229. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cao SQ, Li P, Yin TY and Yang SL: Curcumin
reverses multi-drugresistance of human hepatocellular carcinoma
bel7402/5-FU cells. World Chin J Digestol. 20:135–139. 2012.
|
|
34
|
Wang N, Zhang H, Yao Q, Wang Y, Dai S and
Yang X: TGFBI promoter hypermethylation correlating with paclitaxel
chemoresistance in ovarian cancer. J Exp Clin Cancer Res. 31:62012.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chiang YC, Chang MC, Chen PJ, Wu MM, Hsieh
CY, Cheng WF and Chen CA: Epigenetic silencing of BLU through
interfering apoptosis results in chemoresistance and poor prognosis
of ovarian serous carcinoma patients. Endocr Relat Cancer.
20:213–227. 2013. View Article : Google Scholar : PubMed/NCBI
|