Introduction
Urinary bladder cancer (BCa) is the ninth most
common cancer worldwide. It is the fifth most common malignancy in
males and seventeenth most common in females with the global
standardized incidence rate being 9/100,000 in males and 2/100,000
in females (1) Transitional cell
carcinoma (TCC) is the predominant histologic type of BCa in the
United States and Europe accounting for 90% of cases (2). Approximately 75% of BCas are diagnosed
as superficial, confined to mucosa (Ta, TIS) and submucosa (T1)
while 25% are muscle-invasive (T2-T4) (3). BCa remains a highly prevalent and
lethal malignancy. The optimal treatment selection depends on early
diagnosis as well as accurate staging and grading. Gender
differences in BCa have been proposed to result from the influence
of sex hormones (4–7). The search for cancer biomarkers in
blood and urine is worthy of intense attention due to patients'
comfort and ease of sampling. In general the expectation in respect
to biomarkers is for their sensitivity and specificity to be as
high as possible. Biomarkers have come to play an important role in
routine clinical practice. They are seen as potential tools for the
detection of and for the prediction of recurrence and progression
of bladder carcinoma. Currently there is a need for new prognostic
molecular biomarkers that can help clinicians to identify patients
requiring early, aggressive treatment.
There are a dozen urinary biomarkers for BCa
including the BCa stem-cell marker CD44 (8), the nuclear matrix protein 22 (NMP22)
(9), the fibroblast growth factor
receptor 3 (FGFR3) (9), the
BCa-Specific Antigen-1 (BLCA-1) (10), cathepsin D (11), podoplanin (12), Cystatin C (13), BLCA-4, CYFRA 21-1 and Survivin
(14). Several of them such as
BLCA-1 (10), podoplanin (12) and Cystatin C (13) were also tested as blood serum
biomarkers for BCa. Until now, however, none of the known urinary
or serum biosensors exhibit satisfactory sensitivity and
selectivity for successful diagnosis of BCa. The serum BLCA-1
marker is characterized by 74% sensitivity and 69% selectivity
(10), the podoplanin serum
biosensor shows 72% sensitivity and 69% selectivity (12) while Cystatin C displays 87%
sensitivity and 92% selectivity (13). That is the reason that further
research into finding new biomarkers for BCa is required. A recent
publication of Wu et al (15)
shows a high expression of aromatase in stroma associated with BCa
and preliminary investigations confirmed that aromatase deserves
our attention as a potential BCa biomarker.
Aromatase (known also as CYP19A1) is a key enzyme in
the process of catalysis of androgens to estrogens. Raised levels
of aromatase may result in an intramural microenvironment with
increased estrogen production (15).
Nguyen has reported that aromatase expression is correlated with
bladder tumor pathological stage as well as poor survivability and
has shown that the estrogen receptor may have a promoting role
during tumorigenesis (16).
Aromatase is an enzymatic complex responsible for
the biosynthesis of estrogens from androgens occurring in all
speleids. This complex contains two different proteins:
NADPH-cytochrome P450 reductase and cytochrome P450arom and is
bound to the membrane of the endoplasmic reticulum of
estrogen-producing cells via the N-terminal of the short,
hydrophobic transmembrane domain (17). In humans aromatase is expressed in a
number of cells such as the ovarian granular cells, the placental
syncytiotrophoblast cells, Leydig cells, in skin fibroblasts and in
many areas of the brain, including the hypothalamus, the
hippocampus or the amygdala. Aromatase is also expressed in human
adipose tissue. Increased expression of aromatase is critical in
the pathology of such diseases as breast cancer, endometriosis or
hypogonadism (18) while lack of or
reduced activity of aromatase may cause reduced functioning of
brain neurons and support the development of such diseases as
Alzheimer's disease or Parkinson's disease. It has been
scientifically proven that aromatase inhibitors (anastrozole,
letrozole or exemestane) can be successfully used as drugs for
hormone receptor breast cancer (19).
Aromatase activity can be measured through several
methods. The enzyme-linked immunosorbent assay (ELISA) (20) has so far only been applied in tests
for potential aromatase inhibitors. Other methods which deserve a
mention include the mammalian cell bioassay and fluorescence
substrate assay. Frequently the semi-quantitative
immunohistochemical analysis is used, a method which additionally
shows the location of aromatase in the tissue being
investigated.
The concentration of aromatase in biological samples
can also be measured using biosensors with surface plasmon
resonance imaging (SPRi) detection with specific antibodies and
inhibitors sensitive to Cytochrom P450arom used as receptors
(11). Surface Plasmon Resonance
Imaging (SPRi) is a sensitive, ‘label-free’ technique that can
measure interactions between enzyme and inhibitor or
antibody-antigen, which, in turn, can become a basis for the
development of sensitive sensors for the determination of
biologically active species (21,22).
SPRI biosensors can be used to study interactions in various
biological systems containing proteins, oligonucleotides,
oligosaccharides, lipids, phages, particles and virus cells
(23) as well as for quantitative
analysis (24,25). Recently this technique has gained
significance in the resolution of various clinical problems.
The aim of this work was to investigate aromatase as
a potential plasma BCa biomarker. The recently developed SPRi
biosensor (11) selective for
aromatase was applied. The method exhibits a linear response range
of 0.3–5 ng/ml, an LOD of 0.09 ng/l and an LOQ of 0.3 ng/ml. The
precision (RSD) is 1%, and the recoveries of spikes in natural
samples are within the range 98–103%. Plasma samples corresponding
to different stages of BCa were collected. Healthy donors' plasma
samples were used as controls. There is no information concerning
aromatase concentration in human blood but two other papers
reported aromatase activity in human serum (26,27).
Materials and methods
Reagents
Aromatase peptide and rabbit polyclonal antibody
specific for aromatase (Lucerna-Chem AG, www.lucerna-chem.ch), cysteamine hydrochloride,
N-ethyl-N′-(3-dimethylaminopropyl) carbodiimide (EDC; Sigma
Steinheim), N-Hydroxysuccinimide (NHS) (Aldrich) were used. HBS-ES
solution pH=7.4 (0.01 M HEPES, 0.15 M sodium chloride, 0.005% Tween
20, 3 mM EDTA), photopolimer ELPEMER SD 2054, hydrophobic
protective paint SD 2368 UV SG-DG (Peters), Phosphate Buffered
Saline (PBS) pH=7.4, carbonate buffer pH=8.5 (BIOMED) were used as
received. Aqueous solutions were prepared with MilliQ water
(Simplicity®MILLIPORE).
Patients
The samples were obtained from patients with TCC
seeking treatment at the J. Sniadecki Provincial Hospital of
Białystok (Białystok, Poland). The subjects were divided into two
groups: malignant and control. Plasma samples of patients with
initial (after cystoscopy) or confirmed (prior transurethral
resection of bladder tumor-TURBT) diagnosis of BCa were obtained.
Individuals diagnosed with additional malignant disease or
endometriosis were excluded.
The control group included 18 healthy volunteers
from the Blood Donor Centre of Białystok, Poland.
Cancer diagnosis was determined through a
histological examination of tumor specimens obtained from
transurethral resection or cystectomy. In the end the malignant
group consisted of 78 patients with confirmed TCC. The clinical
parameters including stage, grade, size, tendency to reoccur,
pattern of growth and multifocal nature were determined. Patient
clinical characteristics have been presented in Table I.
 | Table I.Demographic and clinicopathological
characteristics of patients. |
Table I.
Demographic and clinicopathological
characteristics of patients.
| Variable | Range | No. of
patients |
|---|
| Age (years) | ≤65 | 34 |
|
| >65 | 44 |
| Sex | Female | 23 |
|
| Male | 55 |
| Tumor stage | Superficial
(Ta+T1) | 34 |
|
| Invasive
(T2+T3+T4) | 44 |
| Tumor grade | Low grade | 37 |
|
| High grade | 41 |
| Tumor size
(mm) | ≤30 | 38 |
|
| >30 | 40 |
| Recurrence | Primary | 41 |
|
| Recurrent | 29 |
| Multiplicity | Single | 44 |
|
| Multiply | 34 |
Approval (R-I-002/409/2014) for this study was
obtained from the Bioethics Committee of the Medical University of
Białystok (Białystok, Poland) with a written informed consent
obtained from all the patients and donors.
Preparation of biological samples
Blood samples were obtained from patients' cubital
vein. Plasma was prepared according to standard procedures. Plasma
samples were frozen immediately and kept at −80°C. For the
determination of aromatase concentration the prepared plasma
samples were diluted tenfold with phosphate buffered saline
(PBS).
Procedure determination of concentrations
with SPRi biosensor
Chip preparation
Gold chips were manufactured as described in a
previous paper (28,29). The gold surface of the chip was
covered with a photopolymer and hydrophobic paint, a procedure
described in a previous paper (28,29).
9×12 free gold surfaces were obtained. Through the use of this
chip, nine different solutions can be simultaneously measured
without mixing the tested solutions. Twelve single SPRi
measurements can be performed from one solution.
Antibody immobilization
Chips were rinsed with ethanol and water and dried
under a stream of argon. They were then immersed in 20 mM
cysteamine ethanolic solution for at least 2 h after which they
were again rinsed with ethanol and water and dried under a stream
of argon. The next step was the immobilization of the receptor.
Antibody solution in a PBS buffer (20 ng/ml) was activated with NHS
(50 mM) and EDC (200 mM) in a carbonate buffer (pH=8.5)
environment. Then the activated receptor was placed on thiol
(cysteamine) modified surface and incubated at 37°C for 1 h.
SPRi measurements
SPRI measurements for the protein biosensor array
were performed as described previously (29). The signal was measured twice on the
basis of registered images, after the immobilization of the
antibody and then after the interaction with aromatase. Plasma
samples were placed directly on the prepared biosensor for 10 min
to allow an interaction with the receptor. The volume of the sample
applied to each measuring field was 3 µl. After this time the
biosensor was washed with water to remove unbound molecules from
the surface. The SPRi technique used during the study measured the
signal at a constant angle of light. Two images were recorded: The
first image reflects the immobilization of the antibody and the
second image shows the interaction of aromatase with the sample
containing the analyte. The SPRI signal, which is proportional to
the conjugated biomolecules, was obtained through the subtraction
of the signal before and after the interaction with the biomolecule
for each site separately.
Statistical analysis
All results are given as the mean ± standard error
of the mean (SEM), calculated on the basis of 12 repeated
measurements. Statistical analyses were performed using Student's
t-test and P<0.05 was considered to indicate a statistically
significant difference. The receiver operating characteristic (ROC)
curves with optimal cut-off points were calculated. Sensitivity,
specificity, positive and negative predictive values were specified
for cut-off points. All statistical analyses were performed using
PQStat 1.6.4 Software.
Results
Changes in aromatase
concentration
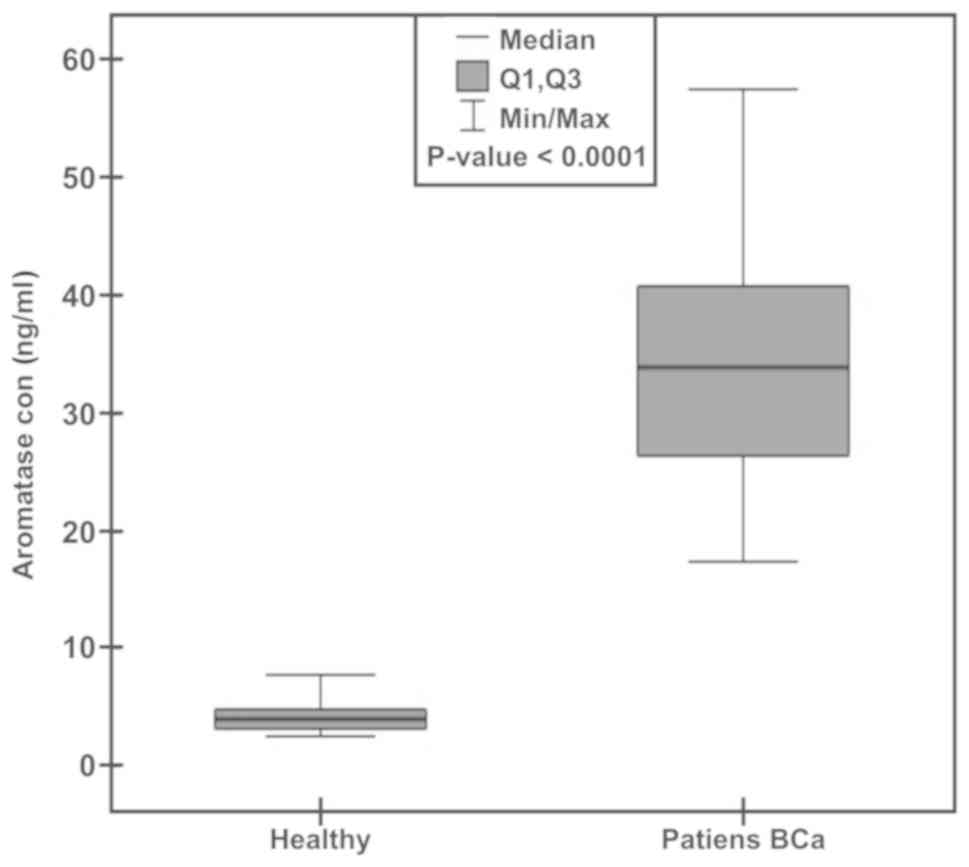
A significant difference in plasma concentration of
aromatase was observed between patients with BCa and healthy
subjects. The aromatase concentration in plasma of BCa patients and
healthy donors ranged from 17.41–57.44 and 2.59–7.74 ng/ml
respectively. The median value for healthy individuals reached 3.99
(an average of 4.3) while that of BCa patients amounted to 33.82
(an average of 33.98) (Fig. 1). This
difference is highly statistically significant, with a P-value
lower than 0.0001.
Aromatase in terms of
clinicopathological parameters of BCa
The aromatase concentration results were analyzed in
terms of different cancer parameters with the following factors
being considered: recurrence of the tumor, tumor stage, tumor
grade, size and multiply of the tumor. Table II shows the plasma concentration of
the aromatase in correlation to clinicopathological
characteristics.
 | Table II.Diagnostic characteristics of plasma
aromatase concentration ratios compared with various
clinocopathological parameters. |
Table II.
Diagnostic characteristics of plasma
aromatase concentration ratios compared with various
clinocopathological parameters.
|
|
| Concentration of
aromatase [ng/ml] |
|---|
|
|
|
|
|---|
| Parameters | Range | Median | P-value |
|---|
|
Primary/recurrent |
| Primary
(n=41) | 19.62–57.44 | 34.06 | 0.14 |
|
Recurrent (n=29) | 17.41–56.81 | 33.17 |
|
| Multiplicity |
| Single
(n=44) | 20.64–57.44 | 35.55 | 0.11 |
|
Multiply (n=34) | 17.41–46.78 | 30.05 |
|
| Stage |
|
Non-muscle invasive (n=34)
(Ta+T1) | 17.41–46.78 | 29.83 | 0.01 |
| Muscle
invasive (n=44) (T2+T3=T4) | 20.64–57.44 | 38.64 |
|
| Grade |
|
Low-grade (37) | 19.62–57.44 | 30.8 | 0.20 |
|
High-grade (41) | 17.41–56.81 | 34.01 |
|
| Size (mm) |
| ≤30
(n=38) | 20.64–46.75 | 35.71 | 0.17 |
| >30
(n=40) | 17.41–57.44 | 32.53 |
|
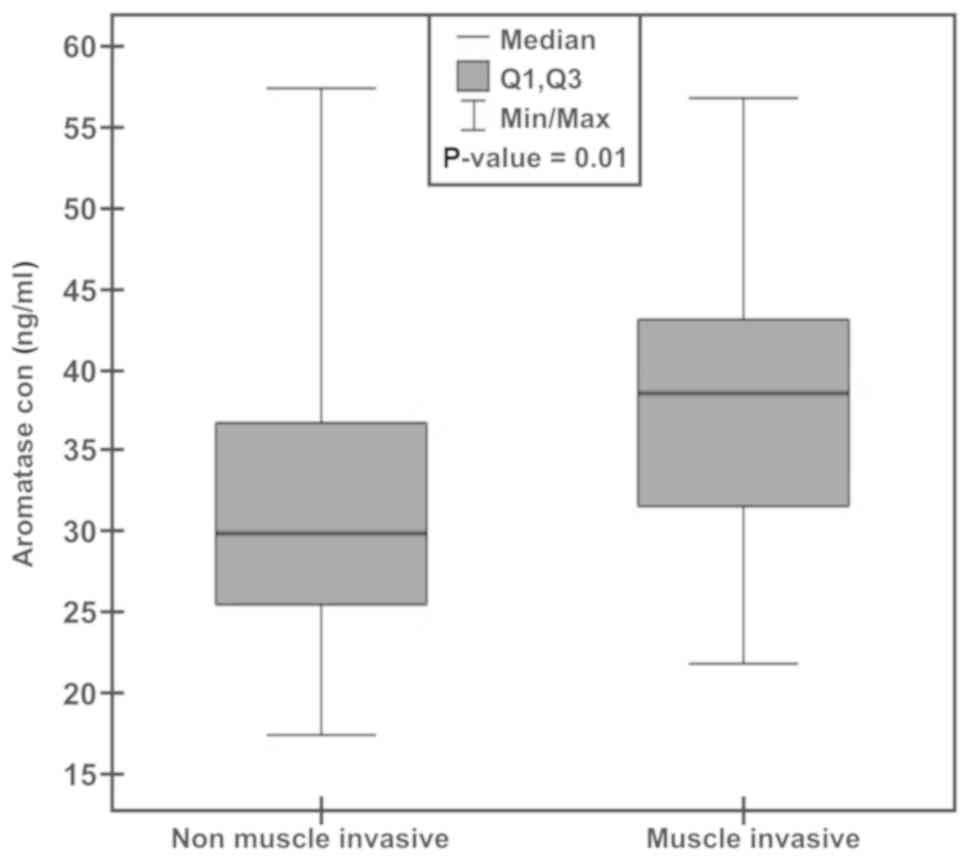
The aromatase concentration in plasma of non
muscle-invasive and muscle-invasive patients fit within the range
of 17.41–46.78 and 20.64–57.44 ng/ml, respectively (Fig. 2). The median for non muscle-invasive
patients was 29.83 ng/ml (an average of 31.19) and that of
muscle-invasive BCa patients was calculated at 38.64 ng/ml (an
average of 37.53). This difference is statistically significant,
with a P-value of 0.01.
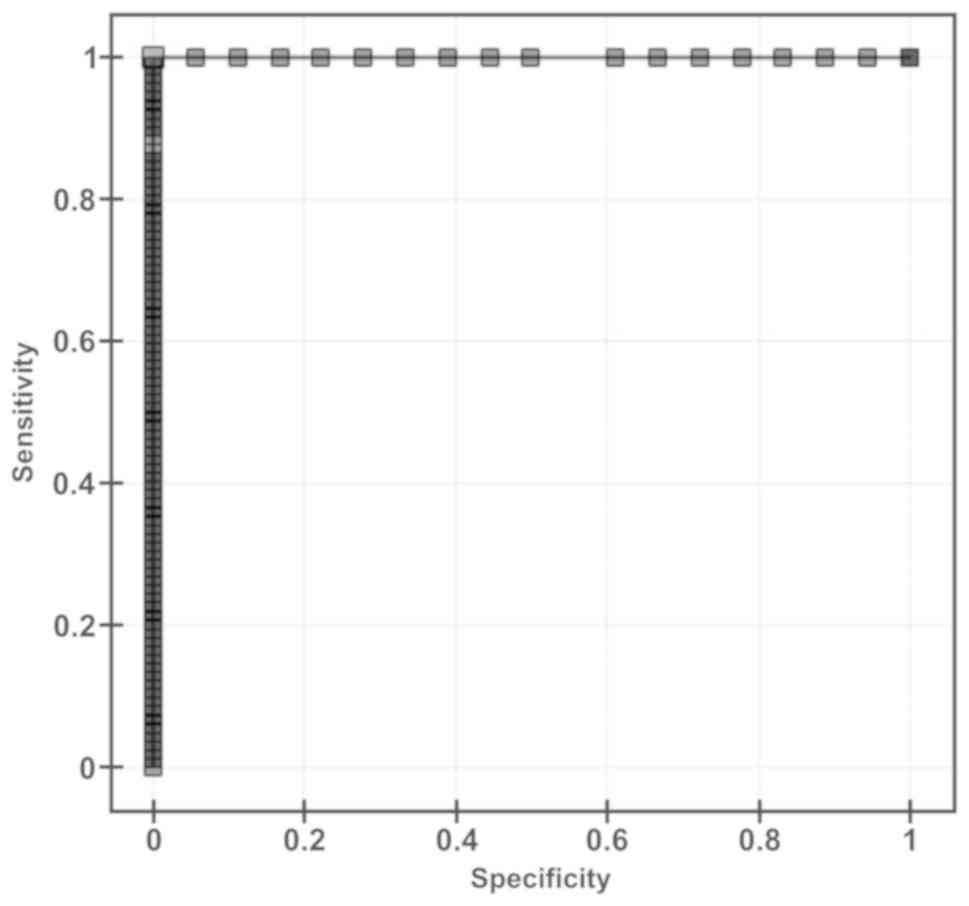
ROC analysis
ROC curve analysis was performed and demonstrated
that blood plasma aromatase levels could be used to distinguish
patients with bladder tumor from healthy individuals through the
area under the curve of 1 (P=0.0001). Specificity, sensitivity,
positive predictive value (PPV) and negative predictive value (NPV)
was estimated at 100% (Fig. 3).
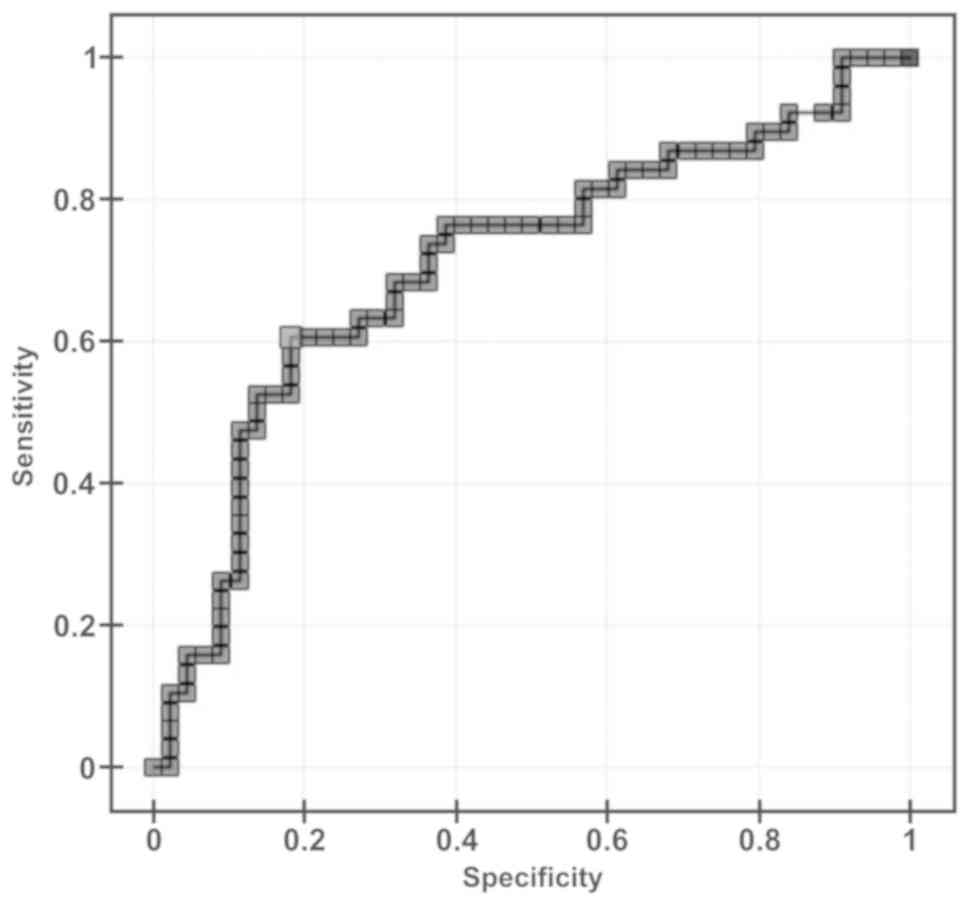
Furthermore, the level of plasma aromatase was able to distinguish
patients with muscle invasive tumors from patients with non
muscle–invasive tumors through the area under the curve of 0.7
(P=0.0012). Other parameter values were as follows:
sensitivity-60%, specificity 81%, PPV 74% and NPV 70% (Fig. 4).
Discussion
The obtained results are surprisingly good. The
values for the average and the median for aromatase plasma
concentration for patients with BCa are almost 10 times higher than
the corresponding values for healthy individuals. The gap between
the lowest result for BCa patients (17.41 ng/ml) and the highest
result from the set of results for healthy individuals (7.74 ng/ml)
is almost 10 ng/ml and there are no overlapping results within
these two groups (Fig. 1). The
difference is highly statistically significant, with a P-value
lower than 0.0001. Thus, plasma aromatase as the BCa marker
exhibits 100% sensitivity and 100% selectivity and the AUC of the
ROC curve is 1.0 (Fig. 3). The
cut-off value concentration calculated from the ROC curves is equal
to 17.41 ng/ml. Results above this value should be considered as
positive for TCC. This type of result is rare not only for BCa
markers but also in general. The other studied potential blood BCa
markers (BLCA-1, podoplanin, cystatin C) exhibited 72–87%
sensitivity and 69–92% specificity (10,12,13). In
terms of sensitivity and selectivity plasma aromatase also produces
better results than urinary BCa markers.
It is certainly true that further studies into
aromatase as a BCa marker may discover problems with the use of
this substance as a marker. Particularly the range of aromatase
blood concentration for individuals not suffering from BCa may vary
due to different factors such as the difference in aromatase
activity due to coronary arterial disease (27).
Certainly, significantly more non-cancer blood
samples are needed to test the real potential of aromatase as a
marker, including a decision curve analysis. This is a limitation
to the present study. The authors hope that this paper will
encourage other groups to carry out such investigations. However,
the obtained results are a very good starting point for the
recommendation of aromatase as a BCa marker. An introduction of
plasma aromatase as a BCa marker may significantly improve early
diagnosis of this disease. It is worth mentioning that the use of
the level of aromatase concentration in blood (plasma) for this
purpose presented in this paper is the first such data ever
published.
The plasma aromatase concentration may also be
useful in the determination of muscle-invasiveness of BCa.
Statistically, muscle-invasive tumors have significantly higher
concentrations of aromatase (P-value=0.01) in comparison to
non-muscle invasive cancers with their sensitivity reaching 60% and
their specificity being 81%. Thus, the results for non-muscle and
for muscle invasive cancer partly overlap (Fig. 2) but the higher the aromatase
concentration the higher the probability of muscle invasiveness of
BCa. The cut-off value concentration calculated from the ROC curve
is equal to 37.31 ng/ml. Patients with aromatase concentrations
above this value should be considered as having bladder invasive
TCC, subject to a sensitivity of 60% and a specificity of 81%.
Aromatase expression was found to be high in
epithelial and stroma tissue of BCa and was associated with worse
overall survival rate (15). These
results are consistent with those reported in this paper. High
aromatase expression was found in endometrial cancer but
conflicting findings were presented for prostate cancer where
aromatase overexpression demonstrates a protective effect (30). This suggests that sex hormones have a
different influence on particular cancers' biology (31).
Generally, hormones may play an important role in
BCa. Epidemiological differences between males and females
(3) suggest potential involvement of
sex hormones in tumorigenesis and progression of BCa (32). Men are 3 to 4 times more often
afflicted by BCa (2) even after
chemical influence and cigarette smoking are taken into
consideration (33). Females are
diagnosed more frequently with a more progressive stage of the
disease and display poorer treatment results after cystectomy and a
higher risk of disease reoccurrence in comparison to males, a fact
that may be explained by earlier invasion and progression.
Moreover, it has been proven that estrogen, estrogen receptor,
testosterone and dihydrotestosteron (34–36) as
well as androgen receptor (37) are
involved in BCa development. It has been reported that loss of
androgen receptor expression was correlated with the progression of
BCa stage (5). Interdependence
between hormone receptors and aromatase observed in case of breast
cancer (38,39) may also be a significant factor in BCa
development.
According to Kirma et al (39) aromatase may directly contribute to
tumorigenesis by paracrine elevation of estrogen what leads to the
induction of genes involved in cell cycle and down-regulation of
tumor suppressor genes altering their mutual relationships and
increasing cellular proliferation. This is consistent with the
findings of Tekmal and Santen (40)
who observed that local estrogen synthesis via aromatase in breast
tissue could be important in the initiation of breast cancer as
well as for its progression.
Thus, apart from advancing the proposal of a new BCa
biomarker our results may also introduce a new factor in
understanding BCa biology. It should be pointed out that this paper
provides initial data concerning aromatase concentration in blood
plasma, although the enzyme may be present in various tissues,
including adipose.
Finally, it is worth stressing the significance of
the introduction of the SPRi technique into the set of tools used
in the investigation of cancer biomarkers. A limited number of
reliable methods available for the determination of cancer
biomarkers in body fluids is a significant barrier in the faster
development of so called liquid biopsy. The SPRi technique is
label-free, relatively inexpensive, operationally simple,
sufficiently accurate and precise and the results of this paper
have confirmed its potential within this area.
In conclusion, plasma aromatase may serve as an
excellent biomarker of BCa with 100% sensitivity, 100% selectivity
and an AUC value of the ROC curve equal to 1.0. Additionally,
levels of plasma aromatase distinguish between muscle-invasive and
non muscle-invasive BCa with a sensitivity of 60% and a specificity
of 81%. The present study also determined the concentration of
aromatase present in the plasma of healthy individuals and patients
with BCa.
Acknowledgements
Not applicable.
Funding
The present study received financial support from
the Polish Ministry of Science and Higher Education under subsidy
for maintaining the research potential of the Faculty of Biol-Chem,
University of Białystok (grant no. 162/2018).
Availability of data and materials
The datasets used and/or analysed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
TG, EG, ZL conceived the current study. TG, EG and
BS designed the current study. BS performed the experiments. TG
collected plasma samples. TG, EG, BS, RK, PL and ZL acquired,
analyzed and interpreted the data, drafted the manuscript and
approved the final manuscript for publication. TG, EG, RK, ZL, BS
and PL agree to be accountable for all aspects of the work in
ensuring that questions related to the accuracy or integrity of any
part of the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
Ethical approval for the current study was obtained
from the Bioethics Committee of the Medical University of Białystok
(Białystok, Poland; approval no. R-I-002/409/2014). Written
informed consent was obtained from all patients and donors.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Siegel R, Ma J and Zou Z: Cancer
statistics, 2014. Cancer J Clinic. 64:9–29. 2014. View Article : Google Scholar
|
|
3
|
Burger M, Catto JW, Dalbagni G, Grossman
HB, Herr H, Karakiewicz P, Kassouf W, Kiemeney LA, La Vecchia C,
Shariat S and Lotan Y: Epidemiology and risk factors of urothelial
bladder cancer. Eur Urol. 63:234–241. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Imada S, Akaza H, Ami Y, Koiso K, Ideyama
Y and Takenaka T: Promoting effects and mechanisms of action of
androgen in bladder carcinogenesis in male rats. Eur Urol.
31:360–364. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Boorjian S, Ugras S, Mongan NP, Gudas LJ,
You X, Tickoo SK and Scherr DS: Androgen receptor expression is
inversely correlated with pathologic tumor stage in bladder cancer.
Urology. 64:383–388. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Miyamoto H, Yao JL, Chaux A, Zheng Y, Hsu
I, Izumi K, Chang C, Messing EM, Netto GJ and Yeh S: Expression of
androgen and oestrogen receptors and its prognostic significance in
urothelial neoplasm of the urinary bladder. BJU Int. 109:1716–1726.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kauffman EC, Robinson BD, Downes M,
Marcinkiewicz K, Vourganti S, Scherr DS, Gudas LJ and Mongan NP:
Estrogen receptor-β expression and pharmacological targeting in
bladder cancer. Oncol Rep. 30:131–138. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Azevedo R, Soares J, Gaiteiro C, Peixoto
A, Lima L, Ferreira D, Relvas-Santos M, Fernandes E, Tavares A,
Cotton S, et al: Glycan affinity magnetic nanoplatforms for urinary
glycobiomarkers discovery in bladder cancer. Talanta. 184:347–355.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bangma CH, Loeb S, Busstra M, Zhu X, El
Bouazzaoui S, Refos J, Van Der Keur KA, Tjin S, Franken CG, Van
Leenders GJ, et al: Outcomes of a bladder cancer screening program
using home hematuria testing and molecular markers. Eur Urol.
64:41–47. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang Z, Li H, Chi Q, Qui Y, Li X and Xin
L: Clinical significance of serological and urological levels of
bladder cancer-specific antigen-1 (BLCA-1) in bladder cancer. Med
Sci Monit. 24:3882–3887. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gorodkiewicz E, Sankiewicz A and Laudanski
P: Surface plasmon resonance imaging biosensors for aromatase based
on a potent inhibitor and a specific antibody: Sensor development
and application for biological material. Cent Eur J Chem.
12:557–567. 2014. View Article : Google Scholar
|
|
12
|
Sankiewicz A, Guszcz T, Mena-Hortelano R,
Zukowski K and Gorodkiewicz E: Podoplanin serum and urine
concentration in transitional bladder cancer. Cancer Biomark.
16:343–350. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tokarzewicz A, Guszcz T, Onopiuk A,
Kozlowski R and Gorodkiewicz E: Utility of cystatin C as a
potential bladder tumour biomarker confirmed by surface plasmon
resonance technique. Indian J Med Res. 21:846–850. 2018.
|
|
14
|
Vrooman OP and Witjes JA: Urinary markers
in bladder cancer. Eur Urol. 53:909–916. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wu S, Ye J, Wang Z, Lin SX, Lu M, Liang Y,
Zhu X, Olumi AF, Zhong W and Wu CL: Expression of aromatase in
tumor related stroma is associated with human bladder cancer
progression. Cancer Biol Ther. 19:175–180. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nguyen DP, O'Malley P, Al Hussein Al
Awamlh B, Furrer MA, Mongan NP, Robinson BD, Wang GJ and Scherr DS:
Association of aromatase with bladder cancer stage and long-term
survival: New insights into the hormonal paradigm in bladder
cancer. Clin Genitourin Canc. 15:256–262.e1. 2017. View Article : Google Scholar
|
|
17
|
Carreau S, Lambard S, Delaland C,
Denis-Galeraud I, Bilinska B and Bourguiba S: Aromatase expression
and role of estrogens in male gonad: A review. Reprod Biol
Endocrin. 1:352003. View Article : Google Scholar
|
|
18
|
Miedlich SU, Karamooz N and Hammes SR:
Aromatase deficiency in a male patient-case report and review of
the literature. Bone. 93:181–186. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Miller WR: Aromatase inhibitors: Mechanism
of action and role in the treatment of breast cancer. Semin Oncol.
30 (4 Suppl 14):S3–S11. 2003. View Article : Google Scholar
|
|
20
|
Satoh K, Nonaka R, Ishikawa F, Ogata A and
Nagai F: In vitro screening assay for detecting aromatase activity
using rat ovarian microsomes and estrone ELISA. Biol Pharm Bull.
31:357–362. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gorodkiewicz E: The surface plasmon
resonance imaging sensor for papain based on immobilized cystatin.
Protein Pept Lett. 14:443–445. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gorodkiewicz E, Guszcz T,
Roszkowska-Jakimiec W and Kozlowski R: Cathepsin D serum and urine
concentration in superficial and invasive transitional bladder
cancer as determined by surface plasmon resonance imaging. Oncol
Let. 8:1323–1327. 2014. View Article : Google Scholar
|
|
23
|
Thevenot DR, Toth K, Durst RA and Wilson
GS: Electrochemical biosensors: Recommended definition and
classification. Pure Appl Chem. 71:2333–2348. 1999. View Article : Google Scholar
|
|
24
|
Laudanski P, Gorodkiewicz E, Ramotowska B,
Charkiewicz R, Kuzmicki M and Szamotowicz J: Determination of
cathepsins B, D and G concentration in eutopic proliferative
endometrium of women with endometriosis by the surface plasmon
resonance imaging (SPRI) technique. Eur J Obstet Gynecol Reprod
Bio. 169:80–83. 2013. View Article : Google Scholar
|
|
25
|
Gorodkiewicz E and Luszczyn J: Surface
plasmon resonance imaging (SPRI) sensor for cystatin determination
based on immobilized papain. Protein Pept Lett. 18:23–29. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Dhefer IH, Abbas SAR and Ahmed NSH:
Polymorphism of cytochrome p450, superfamily19, polypeptide 1 gene
and related to aromatase enzyme activity in acromegaly Iraqi
patients. Asian J Pharm Clin Res. 10:228–232. 2017. View Article : Google Scholar
|
|
27
|
Meng Y, Adi D, Wu Y, Wang Y, Abudoukelimu
M, Huang D, Ma X, Liu C, Wang T, Liu F, et al: CYP19A1
polymorphisms associated with coronary artery disease and
circulating sex hormone levels in a Chinese population. Oncotarget.
8:97101–97113. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gorodkiewicz E: Surface plasmon resonance
imaging sensor for cathepsin determination based on immobilized
cystatin. Protein Pept Lett. 16:1379–1385. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gorodkiewicz E, Regulska E and
Roszkowska-Jakimiec W: Determination of the active form
concentration of cathepsins D and B by SPRI biosensors. J Lab
Diagn. 46:107–109. 2010.
|
|
30
|
Grindstad T, Skjefstad K, Andersen S, Ness
N, Nordby Y, Al-Saad S, Fismen S, Donnem T, Khanehkenari MR, Busund
LT, et al: Estrogen receptors α and β and aromatase as independent
predictors for prostate cancer outcome. Sci Rep. 6:331142016.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chang C, Lee SO, Yeh S and Chang TM:
Androgen receptor (AR) differential roles in hormone-related tumors
including prostate, bladder, kidney, lung, breast and liver.
Oncogene. 33:3225–3234. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Guilherme G, Gakis G, Smith CL and Fahmy
O: Effects of androgen and estrogen receptor signaling pathways on
bladder cancer initiation and progression. Bladder Cancer.
2:127–137. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Castelao JE, Yuan JM, Skipper PL,
Tannenbaum SR, Dado-Dominguez M, Crowder JS, Ross RK and Yu MC:
Gender-and smoking-related bladder cancer risk. J Natl Cancer Inst.
93:538–545. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Shen SS, Smith CL, Hsieh JT, Yu J, Kim IY,
Jian W, Sonpavde G, Ayala GE, Younes M and Lerner SP: Expression of
estrogen receptors-alpha and -beta in bladder cancer cell lines and
human bladder tumor tissue. Cancer. 106:2610–2616. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kawahara T, Ide H, Kashiwagi E,
El-Shishtawy KA, Li Y, Reis LO, Zheng Y and Miyamoto H:
Enzalutamide inhibits androgen receptor-positive bladder cancer
cell growth. Urol Oncol. 34:432.e15–e23. 2016. View Article : Google Scholar
|
|
36
|
Liu X, Cheng X, Liu X, He L, Zhang W, Wang
Y, Sun W and Ji Z: Investigation of the urinary metabolic
variations and the application in bladder cancer biomarker
discovery. Int J Cancer. 143:408–418. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Miyamoto H, Yang Z, Chen YT, Ishiguro H,
Uemura H, Kubota Y, Nagashima Y, Chang YJ, Hu YC, Tsai MY, et al:
Promotion of bladder cancer development and progression by androgen
receptor signals. J Natl Cancer Inst. 99:558–568. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kinoshita Y and Chen S: Induction of
aromatase (CYP19) expression in breast cancer cells through a
nongenomic action of estrogen receptor alpha. Cancer Res.
63:3546–3555. 2003.PubMed/NCBI
|
|
39
|
Kirma N, Gill K, Mandava U and Tekmal RR:
Overexpression of aromatase leads to hyperplasia and changes in the
expression of genes involved in apoptosis, cell cycle, growth, and
tumor suppressor functions in the mammary glands of transgenic
mice. Cancer Res. 61:1910–1918. 2001.PubMed/NCBI
|
|
40
|
Tekmal RR and Santen RJ: ‘Local estrogen
production: Is aromatase an oncogene?’Manni A: Contemporary
Endocrinology: Endocrinology of Breast Cancer. Humana Press;
Totowa, NY: pp. 79–89. 1999
|