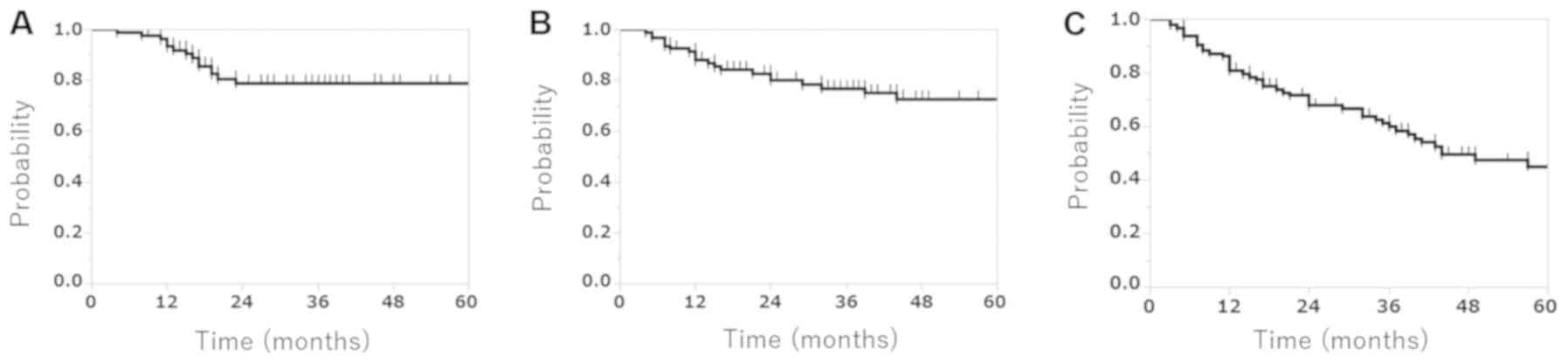
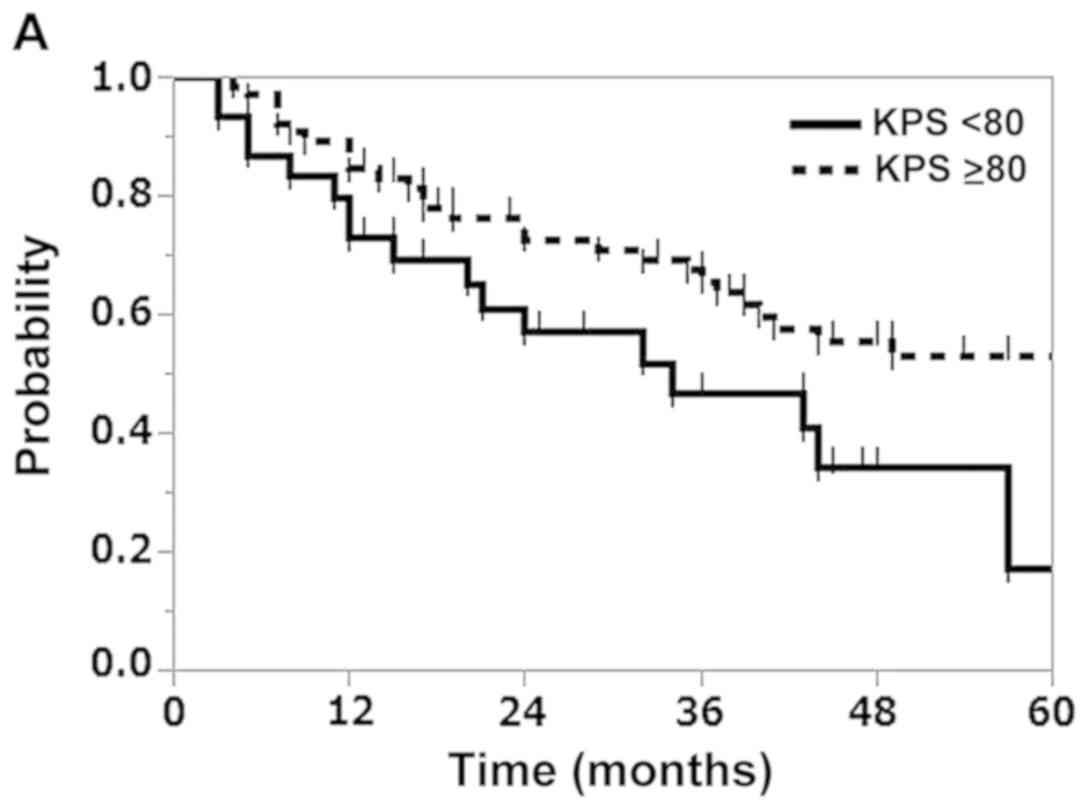
|
1
|
Blomgren H, Lax I, Näslund I and Svanström
R: Stereotactic high dose fraction radiation therapy of
extracranial tumors using an accelerator. Clinical experience of
the first thirty-one patients. Acta Oncol. 34:861–870. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Uematsu M, Shioda A, Suda A, Fukui T,
Ozeki Y, Hama Y, Wong JR and Kusano S: Computed tomography-guided
frameless stereotactic radiotherapy for stage I non-small cell lung
cancer: A 5-year experience. Int J Radiat Oncol Biol Phys.
51:666–670. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nagata Y, Takayama K, Matsuo Y, Norihisa
Y, Mizowaki T, Sakamoto T, Sakamoto M, Mitsumori M, Shibuya K,
Araki N, et al: Clinical outcomes of a phase I/II study of 48 Gy of
stereotactic body radiotherapy in 4 fractions for primary lung
cancer using a stereotactic body frame. Int J Radiat Oncol Biol
Phys. 63:1427–1431. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Onimaru R, Shirato H, Shimizu S, Kitamura
K, Xu B, Fukumoto S, Chang TC, Fujita K, Oita M, Miyasaka K, et al:
Tolerance of organs at risk in small-volume, hypofractionated,
image-guided radiotherapy for primary and metastatic lung cancers.
Int J Radiat Oncol Biol Phys. 56:126–135. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Timmerman R, Paulus R, Galvin J, Michalski
J, Straube W, Bradley J, Fakiris A, Bezjak A, Videtic G, Johnstone
D, et al: Stereotactic body radiation therapy for inoperable early
stage lung cancer. JAMA. 303:1070–1076. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wulf J, Haedinger U, Oppitz U, Thiele W,
Mueller G and Flentje M: Stereotactic radiotherapy for primary lung
cancer and pulmonary metastases: A noninvasive treatment approach
in medically inoperable patients. Int J Radiat Oncol Biol Phys.
60:186–196. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Xia T, Li H, Sun Q, Wang Y, Fan N, Yu Y,
Li P and Chang JY: Promising clinical outcome of stereotactic body
radiation therapy for patients with inoperable Stage I/II
non-small-cell lung cancer. Int J Radiat Oncol Biol Phys.
66:117–125. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Baumann P, Nyman J, Hoyer M, Wennberg B,
Gagliardi G, Lax I, Drugge N, Ekberg L, Friesland S, Johansson K-A,
et al: Outcome in a prospective phase II trial of medically
inoperable stage I non-small-cell lung cancer patients treated with
stereotactic body radiotherapy. J Clin Oncol. 27:3290–3296. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nagata Y, Hiraoka M, Shibata T, Onishi H,
Kokubo M, Karasawa K, Shioyama Y, Onimaru R, Kozuka T, Kunieda E,
et al: Prospective trial of stereotactic body radiation therapy for
both operable and inoperable T1N0M0 non-small cell lung cancer:
Japan Clinical Oncology Group Study JCOG0403. Int J Radiat Oncol
Biol Phys. 93:989–996. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nagata Y and Kimura T: Stereotactic body
radiotherapy (SBRT) for Stage I lung cancer. Jpn J Clin Oncol.
48:405–409. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Palma D, Lagerwaard F, Rodrigues G,
Haasbeek C and Senan S: Curative treatment of Stage I
non-small-cell lung cancer in patients with severe COPD:
Stereotactic radiotherapy outcomes and systematic review. Int J
Radiat Oncol Biol Phys. 82:1149–1156. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen H, Senan S, Nossent EJ, Boldt RG,
Warner A, Palma DA and Louie AV: Treatment-related toxicity in
patients with early-stage non-small cell lung cancer and coexisting
interstitial lung disease: A systematic review. Int J Radiat Oncol
Biol Phys. 98:622–631. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
International Union Against Cancer: TNM
Classification of Malignant Tumours. Sobin LH, Gospodarowicz MK and
Wittekind C: 7th. Wiley-Blackwell Inc.; New York, NY: 2009
|
|
14
|
Péus D, Newcomb N and Hofer S: Appraisal
of the Karnofsky Performance Status and proposal of a simple
algorithmic system for its evaluation. BMC Med Inform Decis Mak.
13:722013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
U.S. Department of Health and Human
Services, National Institutes of Health National Cancer Institute,
. Common Terminology Criteria for Adverse Events (CTCAE). Version
4.0. https://www.eortc.be/services/doc/ctc/CTCAE_4.03_2010-06-14_QuickReference_5×7.pdfMay
28–2009
|
|
16
|
Guckenberger M, Kestin LL, Hope AJ,
Belderbos J, Werner-Wasik M, Yan D, Sonke J-J, Bissonnette JP,
Wilbert J, Xiao Y, et al: Is there a lower limit of pretreatment
pulmonary function for safe and effective stereotactic body
radiotherapy for early-stage non-small cell lung cancer? J Thorac
Oncol. 7:542–551. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Henderson M, McGarry R, Yiannoutsos C,
Fakiris A, Hoopes D, Williams M and Timmerman R: Baseline pulmonary
function as a predictor for survival and decline in pulmonary
function over time in patients undergoing stereotactic body
radiotherapy for the treatment of stage I non-small-cell lung
cancer. Int J Radiat Oncol Biol Phys. 72:404–409. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Stone B, Mangona VS, Johnson MD, Ye H and
Grills IS: Changes in Pulmonary Function Following Image-Guided
Stereotactic Lung Radiotherapy: Neither Lower Baseline Nor
Post-SBRT Pulmonary Function Are Associated with Worse Overall
Survival. J Thorac Oncol. 10:1762–1769. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shibamoto Y, Hashizume C, Baba F, Ayakawa
S, Manabe Y, Nagai A, Miyakawa A, Murai T, Iwata H, Mori Y, et al:
Stereotactic body radiotherapy using a radiobiology-based regimen
for stage I nonsmall cell lung cancer: A multicenter study. Cancer.
118:2078–2084. 2012. View Article : Google Scholar : PubMed/NCBI
|