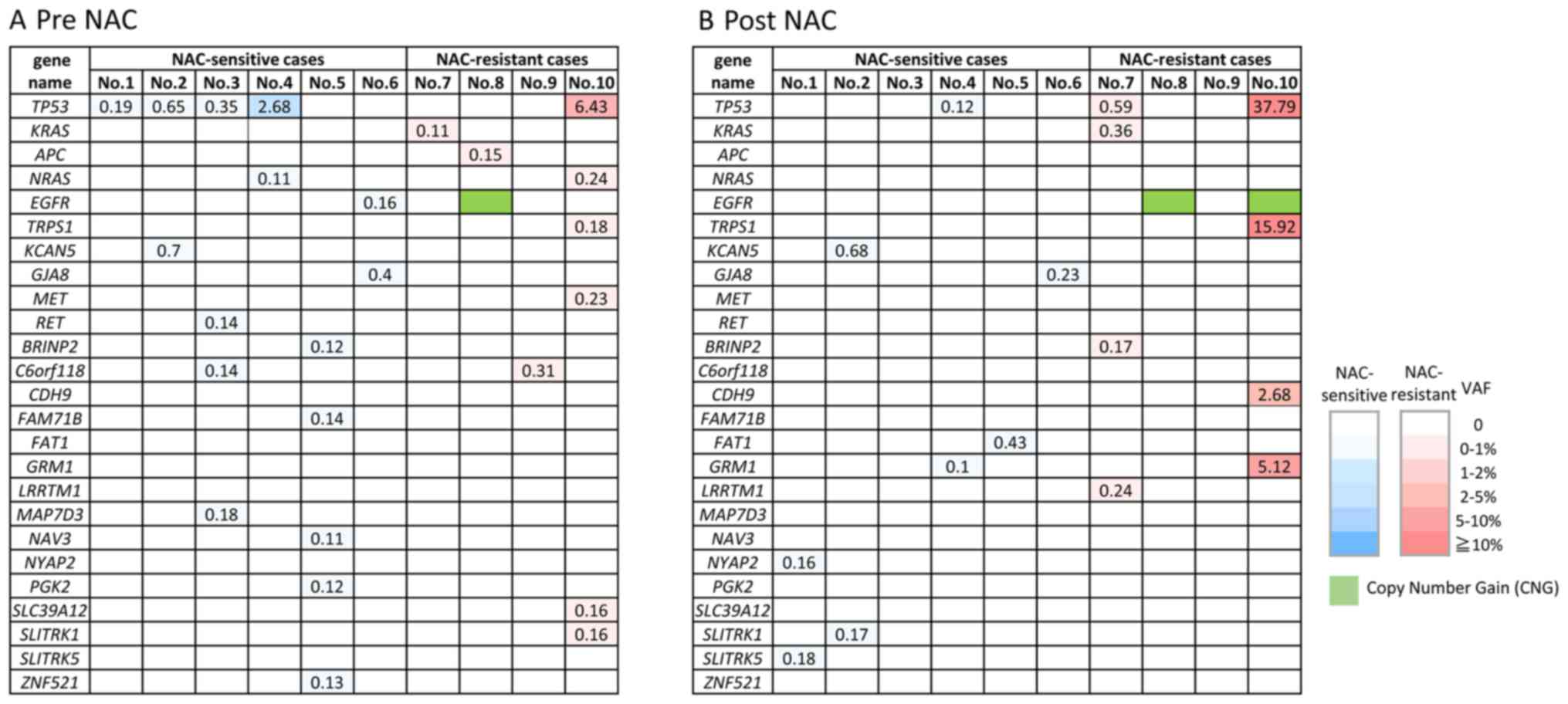
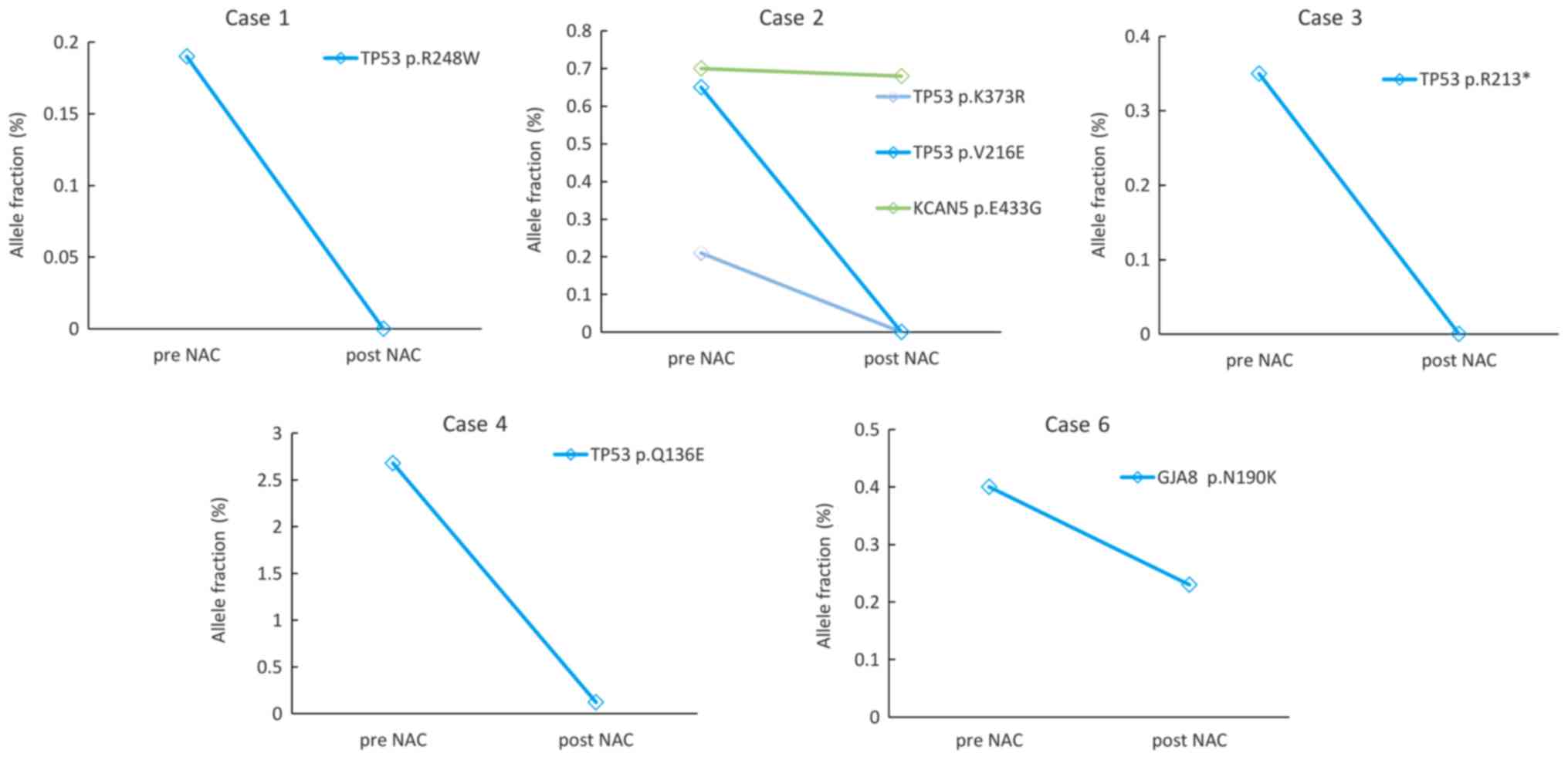
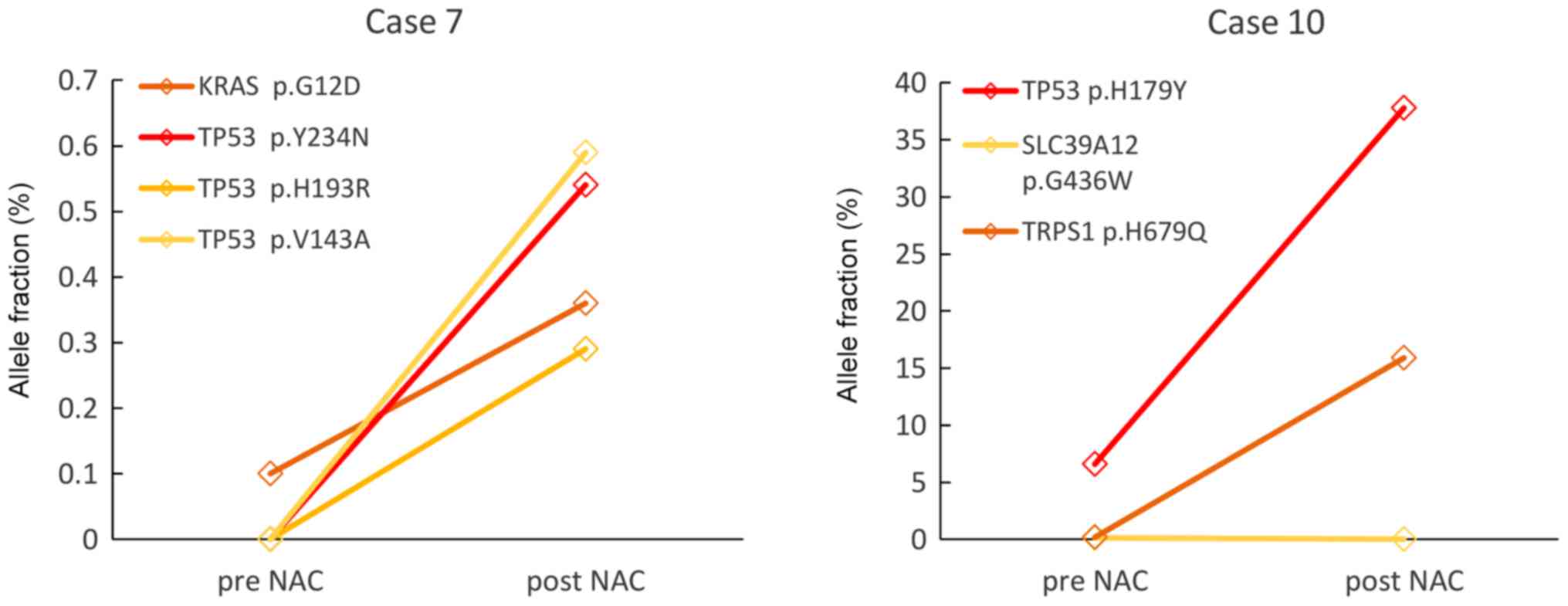
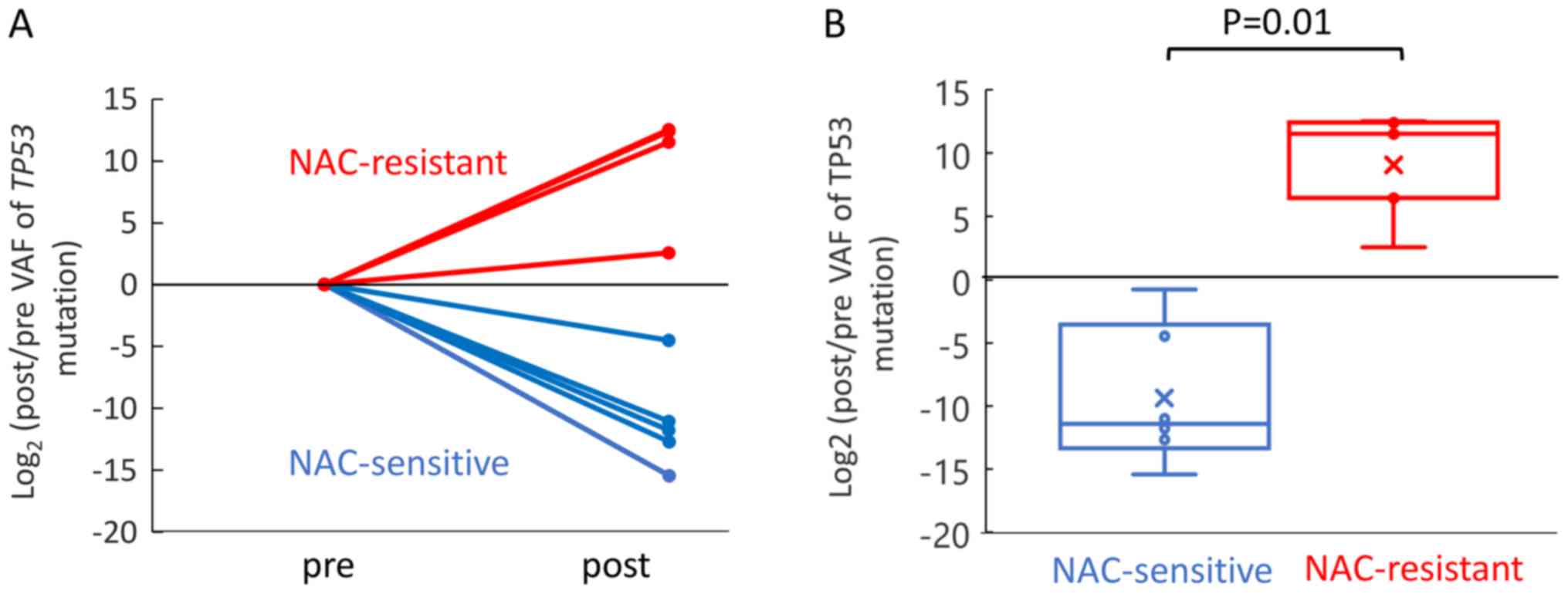
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chi DS, Eisenhauer EL, Zivanovic O, Sonoda
Y, Abu-Rustum NR, Levine DA, Guile MW, Bristow RE, Aghajanian C and
Barakat RR: Improved progression-free and overall survival in
advanced ovarian cancer as a result of a change in surgical
paradigm. Gynecol Oncol. 114:26–31. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fagotti A, Ferrandina G, Vizzielli G,
Fanfani F, Gallotta V, Chiantera V, Costantini B, Margariti PA,
Gueli Alletti S, Cosentino F, et al: Phase III randomised clinical
trial comparing primary surgery versus neoadjuvant chemotherapy in
advanced epithelial ovarian cancer with high tumour load (SCORPION
trial): Final analysis of peri-operative outcome. Eur J Cancer.
59:22–33. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kehoe S, Hook J, Nankivell M, Jayson GC,
Kitchener H, Lopes T, Luesley D, Perren T, Bannoo S, Mascarenhas M,
et al: Primary chemotherapy versus primary surgery for newly
diagnosed advanced ovarian cancer (CHORUS): An open-label,
randomised, controlled, non-inferiority trial. Lancet. 386:249–257.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vergote I, Tropé CG, Amant F, Kristensen
GB, Ehlen T, Johnson N, Verheijen RH, van der Burg ME, Lacave AJ,
Panici PB, et al: Neoadjuvant chemotherapy or primary surgery in
stage IIIC or IV ovarian cancer. N Engl J Med. 363:943–953. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pelissier A, Bonneau C, Chéreau E, de La
Motte Rouge T, Fourchotte V, Daraï E and Rouzier R: CA125 kinetic
parameters predict optimal cytoreduction in patients with advanced
epithelial ovarian cancer treated with neoadjuvant chemotherapy.
Gynecol Oncol. 135:542–546. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rodriguez N, Rauh-Hain JA, Shoni M,
Berkowitz RS, Muto MG, Feltmate C, Schorge JO, Del Carmen MG,
Matulonis UA and Horowitz NS: Changes in serum CA-125 can predict
optimal cytoreduction to no gross residual disease in patients with
advanced stage ovarian cancer treated with neoadjuvant
chemotherapy. Gynecol Oncol. 125:362–366. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zeng J, Yin J, Song X, Jin Y, Li Y and Pan
L: Reduction of CA125 levels during neoadjuvant chemotherapy can
predict cytoreduction to no visible residual disease in patients
with advanced epithelial ovarian cancer, primary carcinoma of
fallopian tube and peritoneal carcinoma. J Cancer. 7:2327–2332.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Diaz LA Jr and Bardelli A: Liquid
biopsies: Genotyping circulating tumor DNA. J Clin Oncol.
32:579–586. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Diehl F, Schmidt K, Choti MA, Romans K,
Goodman S, Li M, Thornton K, Agrawal N, Sokoll L, Szabo SA, et al:
Circulating mutant DNA to assess tumor dynamics. Nat Med.
14:985–990. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vogelstein B and Kinzler KW: Digital PCR.
Proc Natl Acad Sci USA. 96:9236–9241. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bettegowda C, Sausen M, Leary RJ, Kinde I,
Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B, Alani RM, et al:
Detection of circulating tumor DNA in early- and late-stage human
malignancies. Sci Transl Med. 6:224ra242014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Narayan A, Carriero NJ, Gettinger SN,
Kluytenaar J, Kozak KR, Yock TI, Muscato NE, Ugarelli P, Decker RH
and Patel AA: Ultrasensitive measurement of hotspot mutations in
tumor DNA in blood using error-suppressed multiplexed deep
sequencing. Cancer Res. 72:3492–3498. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Newman AM, Bratman SV, To J, Wynne JF,
Eclov NC, Modlin LA, Liu CL, Neal JW, Wakelee HA, Merritt RE, et
al: An ultrasensitive method for quantitating circulating tumor DNA
with broad patient coverage. Nat Med. 20:548–554. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Newman AM, Lovejoy AF, Klass DM, Kurtz DM,
Chabon JJ, Scherer F, Stehr H, Liu CL, Bratman SV, Say C, et al:
Integrated digital error suppression for improved detection of
circulating tumor DNA. Nat Biotechnol. 34:547–555. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Scherer F, Kurtz DM, Newman AM, Stehr H,
Craig AF, Esfahani MS, Lovejoy AF, Chabon JJ, Klass DM, Liu CL, et
al: Distinct biological subtypes and patterns of genome evolution
in lymphoma revealed by circulating tumor DNA. Sci Transl Med.
8:364ra1552016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Przybyl J, Chabon JJ, Spans L, Ganjoo KN,
Vennam S, Newman AM, Forgó E, Varma S, Zhu S, Debiec-Rychter M, et
al: Combination approach for detecting different types of
alterations in circulating tumor DNA in leiomyosarcoma. Clin Cancer
Res. 24:2688–2699. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Iwahashi N, Sakai K, Noguchi T, Yahata T,
Toujima S, Nishio K and Ino K: A comprehensive gene mutation
analysis of liquid biopsy samples from patients with metastatic
colorectal cancer to the ovary: A case report. Oncol Lett.
16:6431–6436. 2018.PubMed/NCBI
|
|
19
|
Iwahashi N, Sakai K, Noguchi T, Yahata T,
Matsukawa H, Toujima S, Nishio K and Ino K: Liquid biopsy-based
comprehensive gene mutation profiling for gynecological cancer
using cancer personalized profiling by deep sequencing. Sci Rep.
9:104262019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Otsubo K, Sakai K, Takeshita M, Harada D,
Azuma K, Ota K, Akamatsu H, Goto K, Horiike A, Kurata T, et al:
Genetic profiling of non-small cell lung cancer at development of
resistance to first- or second-generation EGFR-TKIs by CAPP-Seq
analysis of circulating tumor DNA. Oncologist. 24:1022–1026. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rizvi H, Sanchez-Vega F, La K, Chatila W,
Jonsson P, Halpenny D, Plodkowski A, Long N, Sauter JL, Rekhtman N,
et al: Molecular determinants of response to anti-programmed cell
death (PD)-1 and anti-programmed death-ligand 1 (PD-L1) blockade in
patients with non-small-cell lung cancer profiled with targeted
next-generation sequencing. J Clin Oncol. 36:633–641. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gandara DR, Paul SM, Kowanetz M,
Schleifman E, Zou W, Li Y, Rittmeyer A, Fehrenbacher L, Otto G,
Malboeuf C, et al: Blood-based tumor mutational burden as a
predictor of clinical benefit in non-small-cell lung cancer
patients treated with atezolizumab. Nat Med. 24:1441–1448. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Birkbak NJ, Kochupurakkal B, Izarzugaza
JM, Eklund AC, Li Y, Liu J, Szallasi Z, Matulonis UA, Richardson
AL, Iglehart JD and Wang ZC: Tumor mutation burden forecasts
outcome in ovarian cancer with BRCA1 or BRCA2 mutations. PLoS One.
8:e800232013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Narahara M, Higasa K, Nakamura S, Tabara
Y, Kawaguchi T, Ishii M, Matsubara K, Matsuda F and Yamada R:
Large-scale East-Asian eQTL mapping reveals novel candidate genes
for LD mapping and the genomic landscape of transcriptional effects
of sequence variants. PLoS One. 9:e1009242014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cancer Genome Atlas Research Network, .
Integrated genomic analyses of ovarian carcinoma. Nature.
474:609–615. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Capizzi E, Gabusi E, Grigioni AD, De Iaco
P, Rosati M, Zamagni C and Fiorentino M: Quantification of free
plasma DNA before and after chemotherapy in patients with advanced
epithelial ovarian cancer. Diagn Mol Pathol. 17:34–38.
2008.PubMed/NCBI
|
|
27
|
Gu XH, Wu NW and Liu XS: Application of
serum DNA quantification in the diagnosis of epithelial ovarian
cancer. Matern Child Health Care China. 24:1413–1415. 2009.
|
|
28
|
Holdenrieder S, Stieber P, Bodenmuller H,
Busch M, Fertig G, Fürst H, Schalhorn A, Schmeller N, Untch M and
Seidel D: Nucleosomes in serum of patients with benign and
malignant diseases. Int J Cancer. 95:114–120. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kamat AA, Baldwin M, Urbauer D, Dang D,
Han LY, Godwin A, Karlan BY, Simpson JL, Gershenson DM, Coleman RL,
et al: Plasma cell-free DNA in ovarian cancer: An independent
prognostic biomarker. Cancer. 116:1918–1925. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhou Q, Li W, Leng B, Zheng W, He Z, Zuo M
and Chen A: Circulating cell Free DNA as the diagnostic marker for
ovarian cancer: A systematic review and meta-analysis. PLoS One.
11:e01554952016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Barbosa A, Peixoto A, Pinto P, Pinheiro M
and Teixeira MR: Potential clinical applications of circulating
cell-free DNA in ovarian cancer patients. Expert Rev Mol Med.
20:e62018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Swisher EM, Wollan M, Mahtani SM, Willner
JB, Garcia R, Goff BA and King MC: Tumor-specific p53 sequences in
blood and peritoneal fluid of women with epithelial ovarian cancer.
Am J Obstet Gynecol. 193:662–667. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kim YM, Lee SW, Lee YJ, Lee HY, Lee JE and
Choi EK: Prospective study of the efficacy and utility of TP53
mutations in circulating tumor DNA as a non-invasive biomarker of
treatment response monitoring in patients with high-grade serous
ovarian carcinoma. J Gynecol Oncol. 30:e322019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Pereira E, Camacho-Vanegas O, Anand S,
Sebra R, Catalina Camacho S, Garnar-Wortzel L, Nair N, Moshier E,
Wooten M, Uzilov A, et al: Personalized circulating tumor DNA
biomarkers dynamically predict treatment response and survival in
gynecologic cancers. PLoS One. 10:e01457542015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lafky JM, Wilken JA, Baron AT and Maihle
NJ: Clinical implications of the ErbB/epidermal growth factor (EGF)
receptor family and its ligands in ovarian cancer. Biochim Biophys
Acta. 1785:232–265. 2008.PubMed/NCBI
|
|
36
|
Lassus H, Sihto H, Leminen A, Joensuu H,
Isola J, Nupponen NN and Butzow R: Gene amplification, mutation,
and protein expression of EGFR and mutations of ERBB2 in serous
ovarian carcinoma. J Mol Med (Berl). 84:671–681. 2006. View Article : Google Scholar : PubMed/NCBI
|