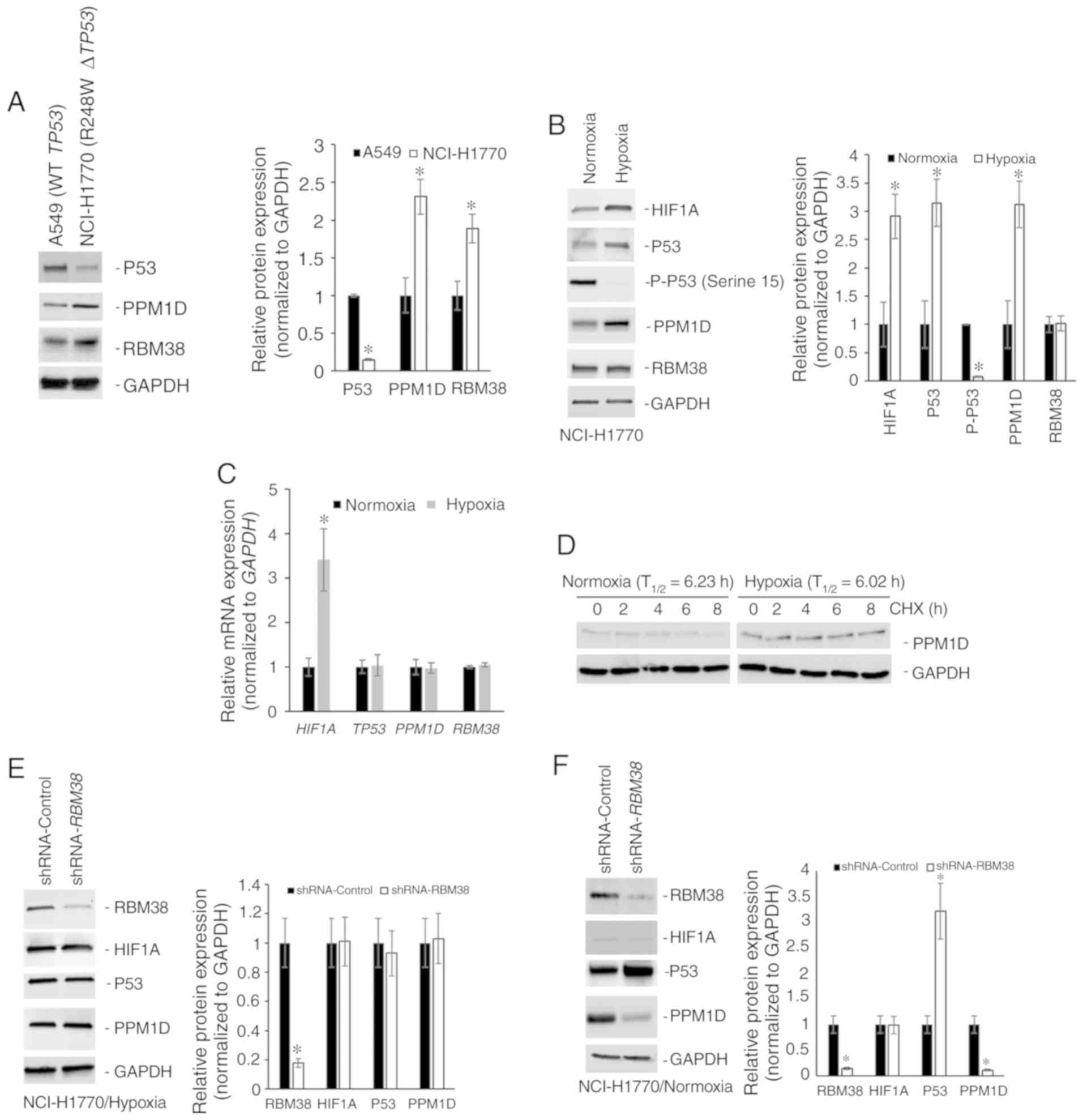
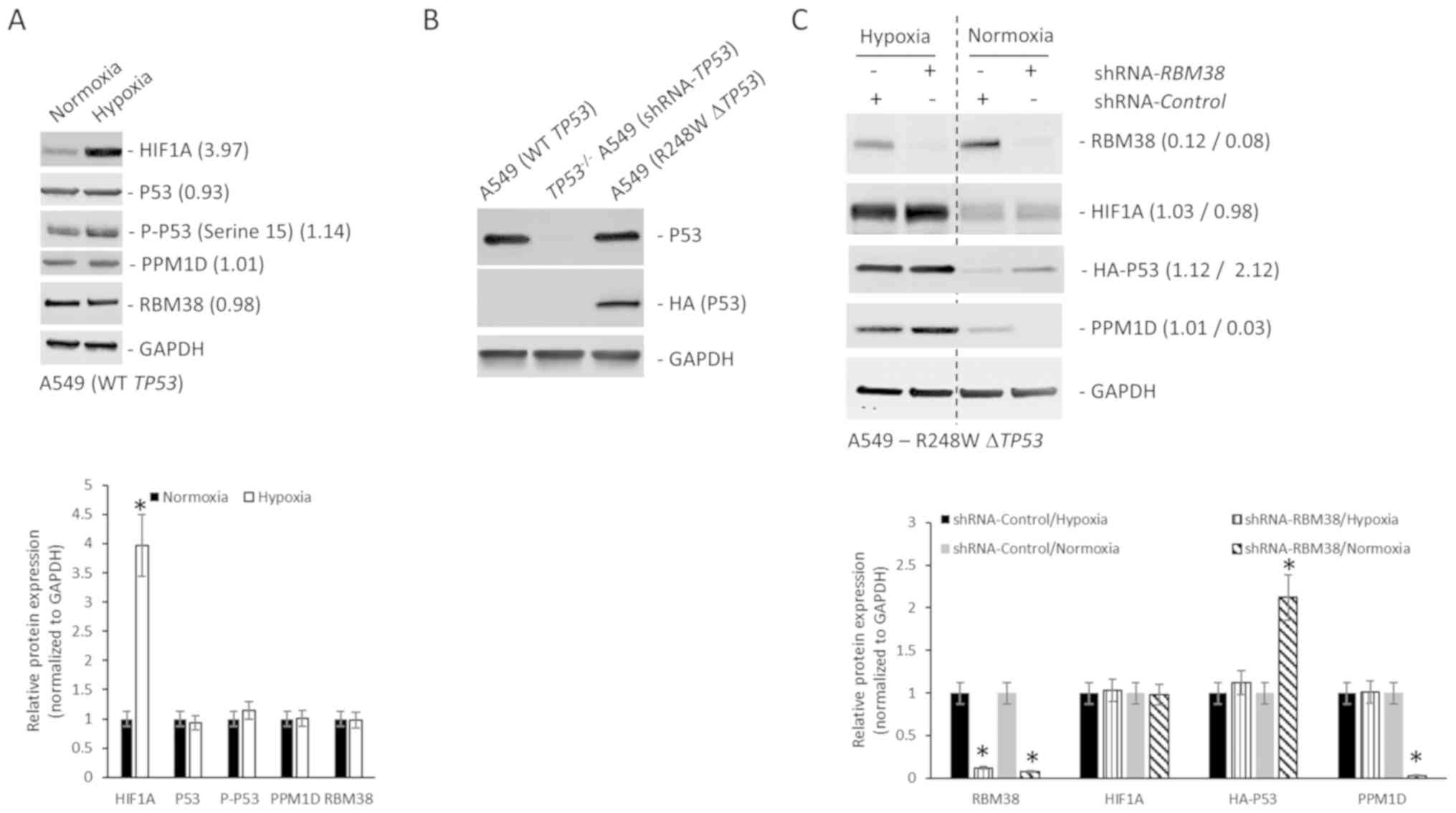
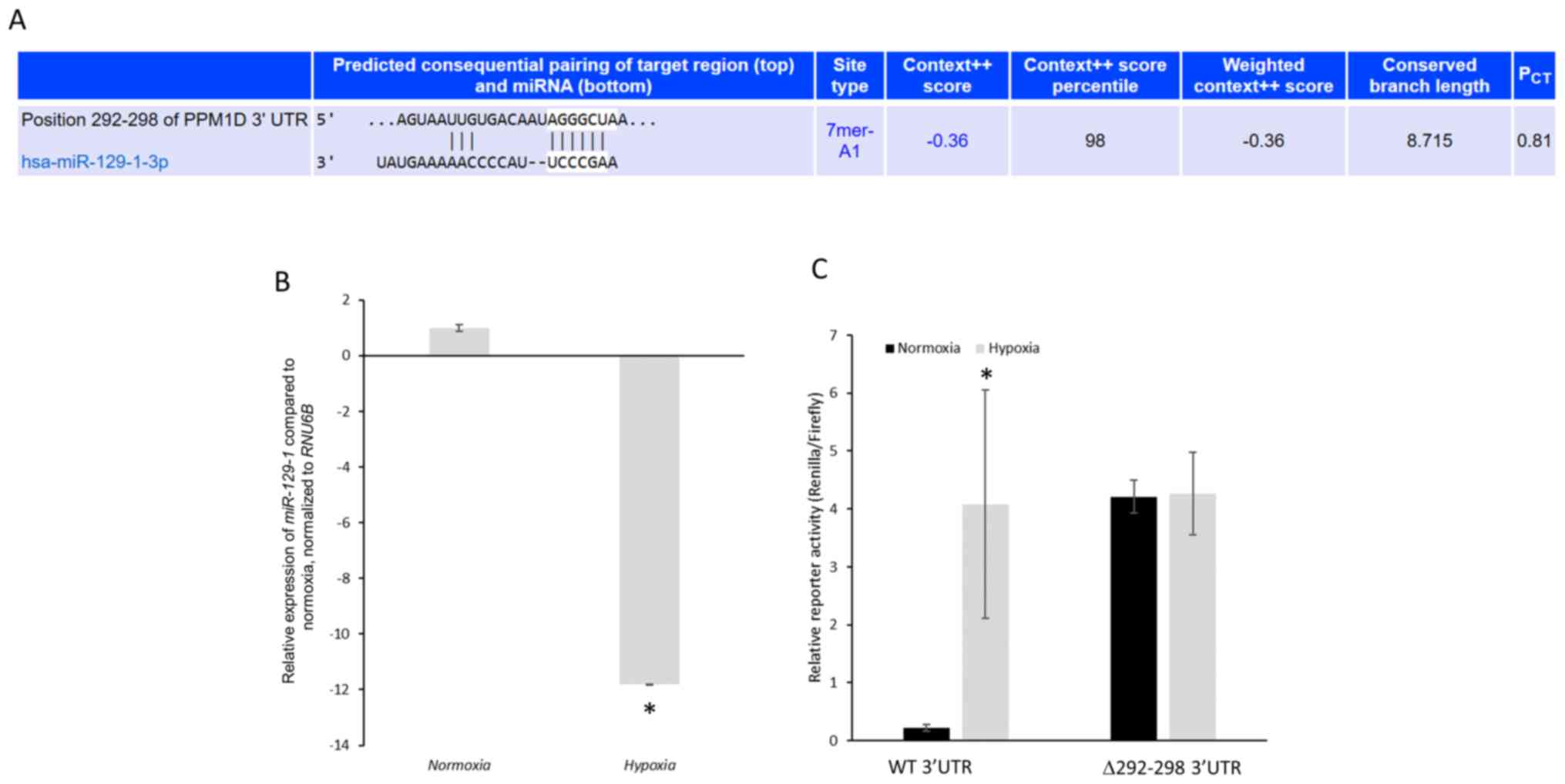
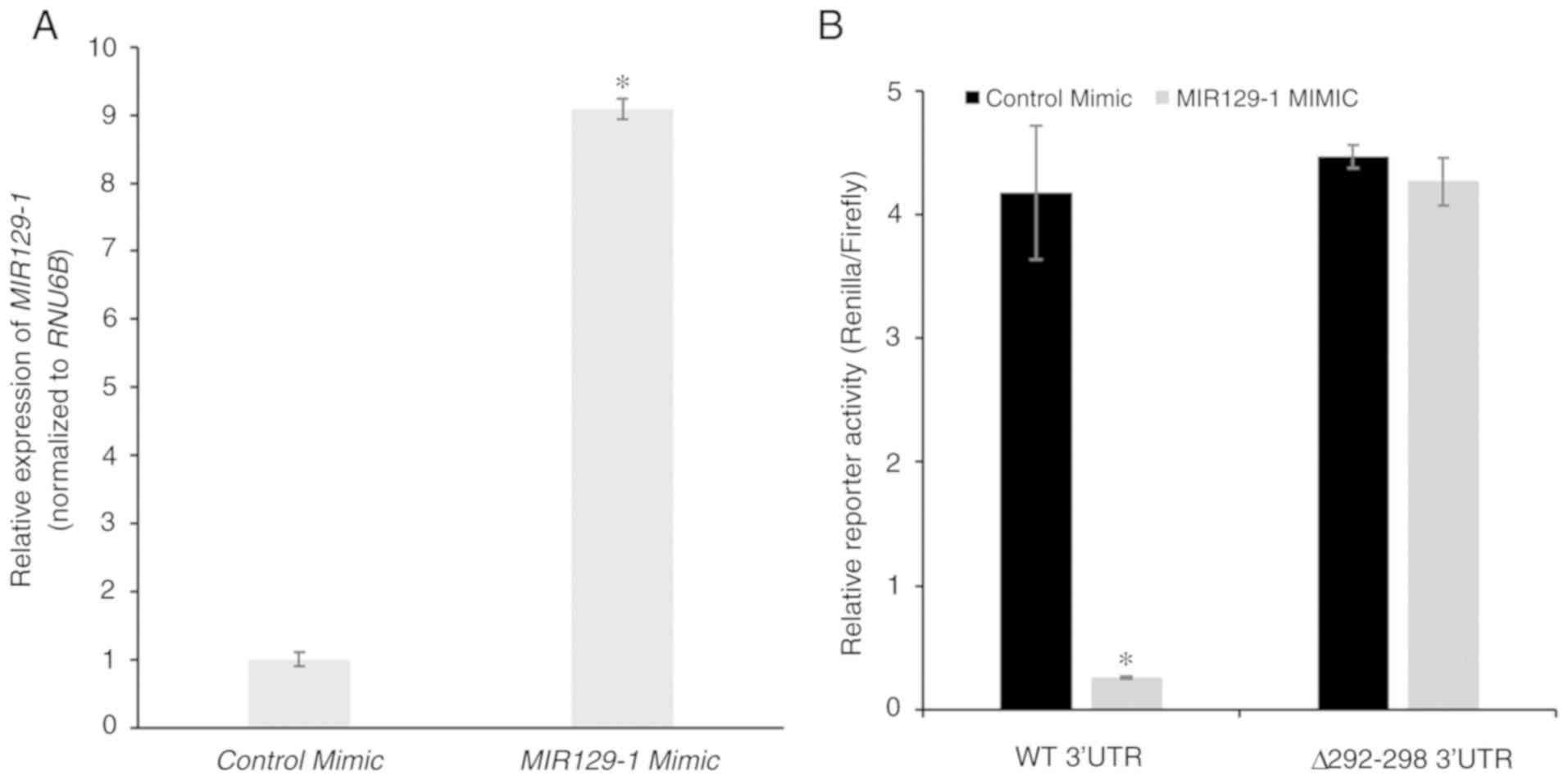
|
1
|
Deng W, Li J, Dorrah K, Jimenez-Tapia D,
Arriaga B, Hao Q, Cao W, Gao Z, Vadgama J and Wu Y: The role of
PPM1D in cancer and advances in studies of its inhibitors. Biomed
Pharmacother. 125:1099562020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fiscella M, Zhang HL, Fan SJ, Sakaguchi K,
Shen SF, Mercer WE, Vande Woude GF, O'Connor PM and Appella E:
Wip1, a novel human protein phosphatase that is induced in response
to ionizing radiation in a p53-dependent manner. Proc Natl Acad Sci
USA. 94:6048–6053. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li J, Yang Y, Peng Y, Austin RJ, van
Eyndhoven WG, Nguyen KC, Gabriele T, McCurrach ME, Marks JR, Hoey
T, et al: Oncogenic properties of PPM1D located within a breast
cancer amplification epicenter at 17q23. Nat Genet. 31:133–134.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nannenga B, Lu X, Dumble M, Van Maanen M,
Nguyen TA, Sutton R, Kumar TR and Donehower LA: Augmented cancer
resistance and DNA damage response phenotypes in PPM1D null mice.
Mol Carcinog. 45:594–604. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tan DS, Lambros MB, Rayter S, Natrajan R,
Vatcheva R, Gao Q, Marchiò C, Geyer FC, Savage K, Parry S, et al:
PPM1D is a potential therapeutic target in ovarian clear cell
carcinomas. Clin Cancer Res. 15:2269–2280. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lu X, Ma O, Nguyen TA, Jones SN, Oren M
and Donehower LA: The Wip1 phosphatase acts as a gatekeeper in the
p53-Mdm2 autoregulatory loop. Cancer Cell. 12:342–354. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lu X, Nannenga B and Donehower LA: PPM1D
dephosphorylates Chk1 and p53 and abrogates cell cycle checkpoints.
Genes Dev. 19:1162–1174. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang M, Xu E, Zhang J and Chen X: PPM1D
phosphatase, a target of p53 and RBM38 RNA-binding protein,
inhibits p53 mRNA translation via dephosphorylation of RBM38.
Oncogene. 34:5900–5911. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang J, Cho SJ, Shu L, Yan W, Guerrero T,
Kent M, Skorupski K, Chen H and Chen X: Translational repression of
p53 by RNPC1, a p53 target overexpressed in lymphomas. Genes Dev.
25:1528–1543. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang M, Zhang J, Chen XL, Cho SJ and Chen
XB: Glycogen synthase kinase 3 promotes p53 mRNA translation via
phosphorylation of RNPC1. Genes Dev. 27:2246–2258. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shu L, Yan W and Chen X: RNPC1, an
RNA-binding protein and a target of the p53 family, is required for
maintaining the stability of the basal and stress-induced p21
transcript. Genes Dev. 20:2961–2972. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Feldstein O, Ben-Hamo R, Bashari D, Efroni
S and Ginsberg D: RBM38 is a direct transcriptional target of E2F1
that limits E2F1-induced proliferation. Mol Cancer Res.
10:1169–1177. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Heinicke LA, Nabet B, Shen S, Jiang P, van
Zalen S, Cieply B, Russell JE, Xing Y and Carstens RP: The RNA
binding protein RBM38 (RNPC1) regulates splicing during late
erythroid differentiation. PLoS One. 8:e780312013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Warzecha CC, Sato TK, Nabet B, Hogenesch
JB and Carstens RP: ESRP1 and ESRP2 are epithelial
cell-type-specific regulators of FGFR2 splicing. Mol Cell.
33:591–601. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Olivier M, Eeles R, Hollstein M, Khan MA,
Harris CC and Hainaut P: The IARC TP53 database: New online
mutation analysis and recommendations to users. Hum Mutat.
19:607–614. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Freed-Pastor WA and Prives C: Mutant p53:
One name, many proteins. Genes Dev. 26:1268–1286. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang J, Xu E, Ren C, Yang HJ, Zhang Y,
Sun W, Kong X, Zhang W, Chen M, Huang E and Chen X: Genetic
ablation of Rbm38 promotes lymphomagenesis in the context of mutant
p53 by downregulating PTEN. Cancer Res. 78:1511–1521. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cho SJ, Teng IF, Zhang M, Yin T, Jung YS,
Zhang J and Chen X: Hypoxia-inducible factor 1 alpha is regulated
by RBM38, a RNA-binding protein and a p53 family target, via mRNA
translation. Oncotarget. 6:305–316. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Amelio I, Mancini M, Petrova V, Cairns RA,
Vikhreva P, Nicolai S, Marini A, Antonov AA, Le Quesne J, Baena
Acevedo JD, et al: P53 mutants cooperate with HIF-1 in
transcriptional regulation of extracellular matrix components to
promote tumor progression. Proc Natl Acad Sci USA.
115:E10869–E10878. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang WC: MicroRNAs tune oxidative stress
in cancer therapeutic tolerance and resistance. Int J Mol Sci.
20:E60942019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yu X, Song H, Xia T, Han S, Xiao B, Luo L,
Xi Y and Guo J: Growth inhibitory effects of three miR-129 family
members on gastric cancer. Gene. 532:87–93. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bandres E, Agirre X, Bitarte N, Ramirez N,
Zarate R, Roman-Gomez J, Prosper F and Garcia-Foncillas J:
Epigenetic regulation of microRNA expression in colorectal cancer.
Int J Cancer. 125:2737–2743. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen X, Hu H, Guan X, Xiong G, Wang Y,
Wang K, Li J, Xu X, Yang K and Bai Y: CpG island methylation status
of miRNAs in esophageal squamous cell carcinoma. Int J Cancer.
130:1607–1613. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Dyrskjot L, Ostenfeld MS, Bramsen JB,
Silahtaroglu AN, Lamy P, Ramanathan R, Fristrup N, Jensen JL,
Andersen CL, Zieger K, et al: Genomic profiling of microRNAs in
bladder cancer: MiR-129 is associated with poor outcome and
promotes cell death in vitro. Cancer Res. 69:4851–4860. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schmaltz C, Hardenbergh PH, Wells A and
Fisher DE: Regulation of proliferation-survival decisions during
tumor cell hypoxia. Mol Cell Biol. 18:2845–2854. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gao J, Aksoy BA, Dogrusoz U, Dresdner G,
Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, et al:
Integrative analysis of complex cancer genomics and clinical
profiles using the cBioPortal. Science Signal. 6:112013. View Article : Google Scholar
|
|
28
|
Unberath P, Knell C, Prokosch HU and
Christoph J: Developing new analysis functions for a translational
research platform: Extending the cBioPortal for cancer genomics.
Stud Health Technol Inform. 258:46–50. 2019.PubMed/NCBI
|
|
29
|
Barretina J, Caponigro G, Stransky N,
Venkatesan K, Margolin AA, Kim S, Wilson CJ, Lehár J, Kryukov GV,
Sonkin D, et al: The cancer cell line encyclopedia enables
predictive modelling of anticancer drug sensitivity. Nature.
483:603–607. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Reinhold WC, Sunshine M, Liu H, Varma S,
Kohn KW, Morris J, Doroshow J and Pommier Y: CellMiner: A web-based
suite of genomic and pharmacologic tools to explore transcript and
drug patterns in the NCI-60 cell line set. Cancer Res.
72:3499–3511. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rizvi H, Sanchez-Vega F, La K, Chatila W,
Jonsson P, Halpenny D, Plodkowski A, Long N, Sauter JL, Rekhtman N,
et al: Molecular determinants of response to anti-programmed cell
death (PD)-1 and anti-programmed death-ligand 1 (PD-L1) blockade in
patients with non-small-cell lung cancer profiled with targeted
next-generation sequencing. J Clin Oncol. 36:633–641. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jamal-Hanjani M, Wilson GA, McGranahan N,
Birkbak NJ, Watkins TBK, Veeriah S, Shafi S, Johnson DH, Mitter R,
Rosenthal R, et al: Tracking the evolution of non-small-cell lung
cancer. N Engl J Med. 376:2109–2121. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Vavalà T, Monica V, Lo Iacono M, Mele T,
Busso S, Righi L, Papotti M, Scagliotti GV and Novello S: Precision
medicine in age-specific non-small-cell-lung-cancer patients:
Integrating biomolecular results into clinical practice-A new
approach to improve personalized translational research. Lung
Cancer. 107:84–90. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rizvi NA, Hellmann MD, Snyder A, Kvistborg
P, Makarov V, Havel JJ, Lee W, Yuan J, Wong P, Ho TS, et al: Cancer
immunology. Mutational landscape determines sensitivity to PD-1
blockade in non-small cell lung cancer. Science. 348:124–128. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Campbell JD, Alexandrov A, Kim J, Wala J,
Berger AH, Pedamallu CS, Shukla SA, Guo G, Brooks AN, Murray BA, et
al: Distinct patterns of somatic genome alterations in lung
adenocarcinomas and squamous cell carcinomas. Nat Genet.
48:607–616. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Agarwal V, Bell GW, Nam J and Bartel DP:
Predicting effective microRNA target sites in mammalian mRNAs.
Elife. 4:e050052015. View Article : Google Scholar
|
|
37
|
Li J, Wang H, Ke H and Ni S: MiR-129
regulates MMP9 to control metastasis of non-small cell lung cancer.
Tumour Biol. 36:5785–5790. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wu J, Qian J, Li C, Kwok L, Cheng F, Liu
P, Perdomo C, Kotton D, Vaziri C, Anderlind C, et al: MiR-129
regulates cell proliferation by downregulating Cdk6 expression.
Cell Cycle. 9:1809–1818. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wampfler J, Federzoni EA, Torbett BE, Fey
MF and Tschan MP: The RNA binding proteins RBM38 and DND1 are
repressed in AML and have a novel function in APL differentiation.
Leuk Res. 41:96–102. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Leveille N, Elkon R, Davalos V, Manoharan
V, Hollingworth D, Oude Vrielink J, le Sage C, Melo CA, Horlings
HM, Wesseling J, et al: Selective inhibition of microRNA
accessibility by RBM38 is required for p53 activity. Nat Commun.
2:513–523. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kakkad S, Krishnamachary B, Jacob D,
Pacheco-Torres J, Goggins E, Bharti SK, Penet MF and Bhujwalla ZM:
Molecular and functional imaging insights into the role of hypoxia
in cancer aggression. Cancer Metastasis Rev. 38:51–64. 2019.
View Article : Google Scholar : PubMed/NCBI
|