Introduction
Cervical cancer is one of the most common malignant
tumors in women worldwide and in China (1), 80% of which is made up of the cervical
squamous cell carcinoma (2). In
2018, there were 106,430 new cases and 47,739 deaths due to
cervical cancer in China (3). The
average morbidity and mortality are on the increase with annual
rates of 8.7% and 8.1%, respectively (4). However, precursor lesions with regards
to the diagnosis of cervical cancer could reduce its incidence and
improve the patient's quality of life.
Effective early screening technology that detects
precursor lesions includes the Pap smear and the ThinPrep Cytologic
Test (TCT). Previous reports showed that the TCT has higher
sensitivity (5), and is one of the
most common cervical cancer screening methods (6). The diagnosis from TCT samples is mainly
dependent on a pathologist's interpretation under a microscope,
according to the Bethesda report system (7). This system defines six categories of
cervical squamous cells: Negative for intraepithelial lesion or
malignancy (NILM), atypical squamous cells-unclear meaning
(ASC-US), atypical squamous cells-do not rule out high-grade
squamous intraepithelial lesions (ASC-H), low-grade squamous
intraepithelial lesions (LSIL), high-grade squamous intraepithelial
lesions (HSIL), and squamous cell carcinoma (SCC). The first
category, NILM, indicates the presence of normal cells, while the
remaining five categories indicate the presence of abnormal cells.
The degree of morphological abnormality is the key to defining the
lesion grade, including the shape, size, and boundary of the
nucleus and the characteristics of the chromatin. However, accurate
diagnosis of cervical squamous cells by pathologists is
time-consuming and subjective, with the classification of samples
influenced by cellular complexity and the pathologist's previous
experience interpreting TCT samples.
Advances in artificial intelligence (AI) technology,
especially the deep convolution neural networks (DCNN), has
obtained great success in medical research, including skin lesion
classification (8) and acute
lymphoblastic leukemia diagnosis (9). The DCNN often contains several layers
with convolutional kernels, similar to a mathematical model that
contains numerous parameters; thus, a large amount of data is
needed to train kernels. Unlike traditional algorithms (10), DCNN is not limited to features that
are extracted based on the experience of expertise. Instead, it
uses the original images as input, automatically extracts a large
number of hierarchical features, and finally generates a diagnosis
as an output. Many researchers have focused on digital cervical
pathology, where DCNN is used to provide a diagnosis of cervical
cancer (11–16). Abdulkerim et al (11) and Chankong et al (12) segmented the cervical cells for
diagnosis. Zhang et al (13)
and Wu et al (14) classified
the cervical lesion grades in single cell images. Tian et al
(15) and Liu et al (16) simultaneously detected a lesion
location and classified the lesion degree in whole pathological
images without prior cell segmentation or cropping.
Although previous studies have advanced the use of
DCNN in cervical cancer screening, the studies face three major
limitations. First, those studies usually focused on PAP-smear
images (11–14). This early-screening method has been
largely replaced by TCT-staining, but researchers have not yet
examined the use of DCNN to diagnose cervical cancer from TCT
images. Second, to improve and evaluate the comprehensiveness and
robustness of DCNN models, multi-center datasets, which includes
data from different levels of hospitals, are needed. However,
previous studies have only included data from one center (14–15).
Third, in order to analyze the results of the AI diagnoses,
researchers should compare the results with those from the manual
interpretation completed by pathologists. However, few studies have
generated such comparisons (17).
The aim of the present study was to address the
limitations of previous studies that have examined the utility of
AI in cervical cancer detection by building a TCT dataset from
samples collected from different hospitals and creating an
automated pipeline for the normal and abnormal cervical squamous
cell classification using VGG16, which is a popular DCNN model. The
results were compared with the classifications provided by two
different pathologists.
Materials and methods
TCT image collection
TCT specimens were obtained from a total of 82
patients in four hospitals located in the city of Chongqing, China
(Table I).
 | Table I.Characteristics of the TCT specimens
used to form the dataset. |
Table I.
Characteristics of the TCT specimens
used to form the dataset.
| Variables | Daping
Hospitala (n=38) | Armed Police General
Hospital (n=10) | Hi-Tech People's
Hospital (n=16) | Du Deling Clinic
(n=18) |
|---|
| Age (years) | 43 (20–66) | 48 (36–62) | 42 (25–57) | 54 (34–56) |
| Cells | 1,184 | 776 | 1,115 | 215 |
| Training | 977 | 625 | 889 | 178 |
| Testing | 207 | 151 | 226 | 37 |
The inclusion criteria in this study were: i)
Patients aged ≥18 years, ii) satisfactory TCT specimens that
contained ≥2,000 squamous epithelial cells with clear morphologies,
and iii) the diagnostic results of the TCT had to be one of the
precursor lesions subtypes or cervical cancer. This study was
approved by the Ethics Committee of the Army Military Medical
University (approval no. KY201774) and was conducted in accordance
with the Helsinki Declaration. The study was explained to all the
patients, and oral informed consent was obtained from them for
their sample to be used for scientific research by phone when
images were collected. Written patient consent was not required,
according to the guidance of the ethics committee.
A total of 1736 abnormal cells and 1554 normal cell
samples were obtained, which were randomly divided into training
(n=2669) and test (n=621) datasets at a ratio of 4:1. All images
were in the BMP file format.
Data preprocessing
TCT specimens were digitized to images using a
microscope with a 40× objective lens (model: VS120, Brand: Olympus)
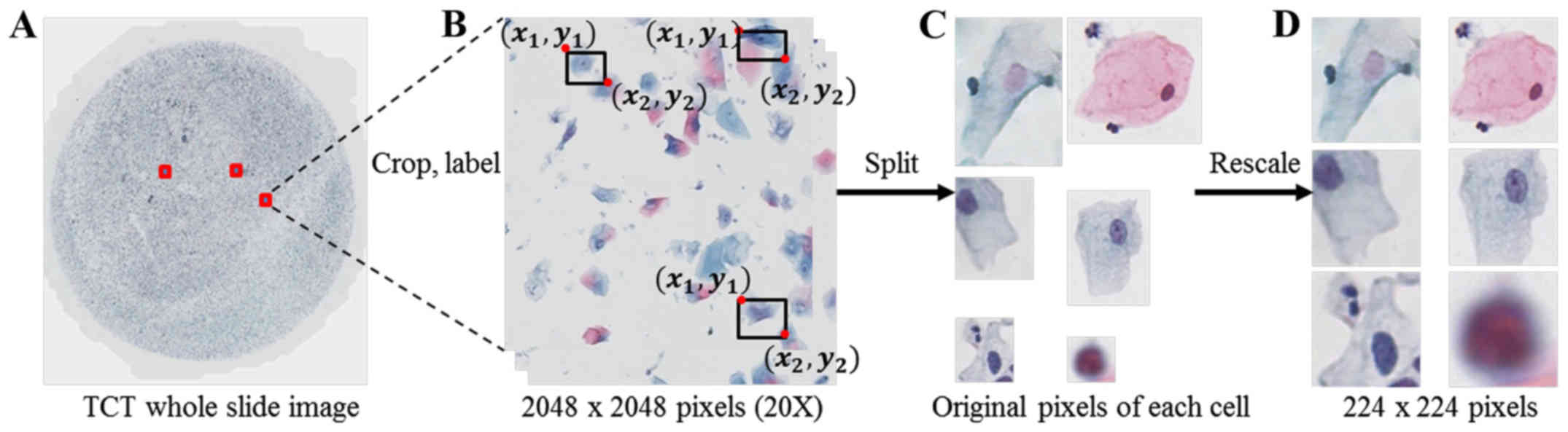
and cut into 2048×2048 pixels for labeling (Fig. 1). Two clinical pathologists
independently labelled the squamous epithelial cells with bounding
boxes as NILM, ASC-US, LSIL, ASC-H, HSIL, and SCC using LabelImg
software, which is a graphical image annotation tool (https://github.com/tzutalin/labelImg),
according to the Bethesda reporting system. After labeling, all
data were split into single-cell images according to the labeled
coordinates of bounding boxes and rescaled to 224×224 pixels
according to the shortest side (Fig.
1). To reduce the inter- and intra-observer variability, an
annotated review was conducted of the original pixel images of the
single cells one month following the initial labeling.
In order to prevent the problem of over-fitting,
where the model may not accurately predict additional data, data
augmentation on the training set using image flipping was
performed. Data normalization was completed to ease the redundant
image differences caused by the different environments and staining
workflow of multiple hospitals (18).
Development of the DCNN model
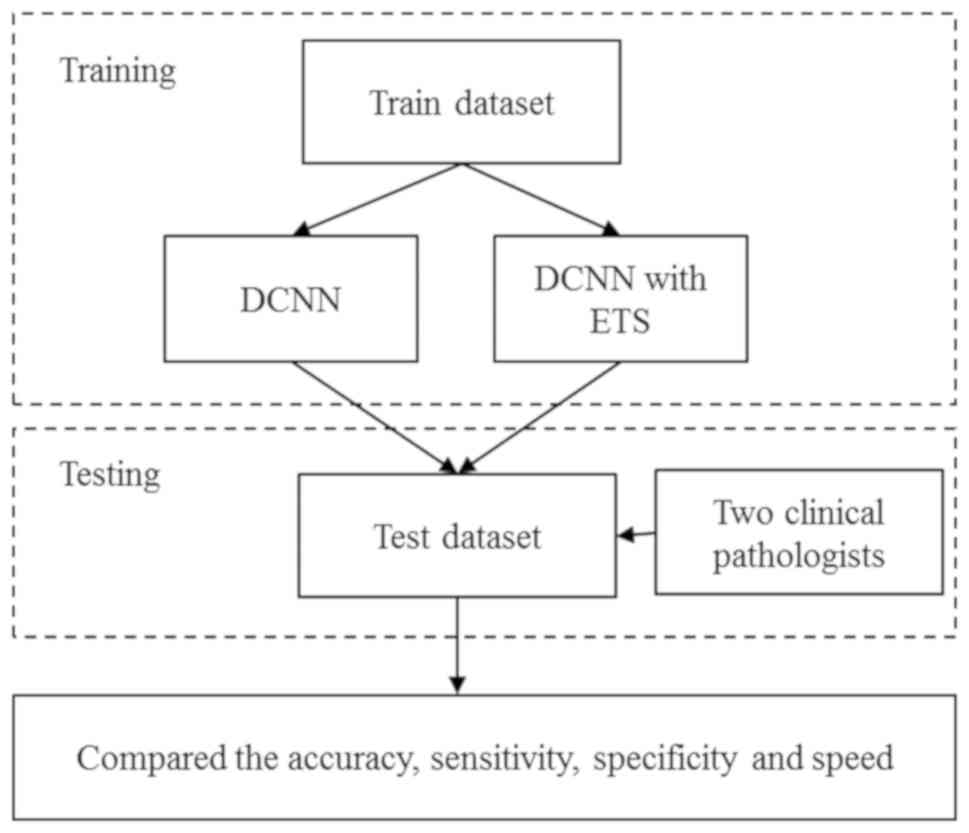
The development of the DCNN model followed a
protocol of training and then testing (Fig. 2). The results of the model were
compared with that independently obtained by two experienced
pathologists. An Intel Core I7-7800X 3.50GHz * 12 with four Titan X
GPUs using Python 3.5 (https://www.python.org) and the TensorFlow (https://www.tensorflow.org) library for all
experiments were used. The number of training iterations was set to
2,000, and the initial learning rate was set to 0.01. The optimizer
was selected via the Batch Gradient Descent.
Through previous literature and preliminary
experiments, the VGG16 model demonstrated a balance in framework,
accuracy, computational efficiency and proven performance in the
medical field, and was chosen for our experiments (19–21). The
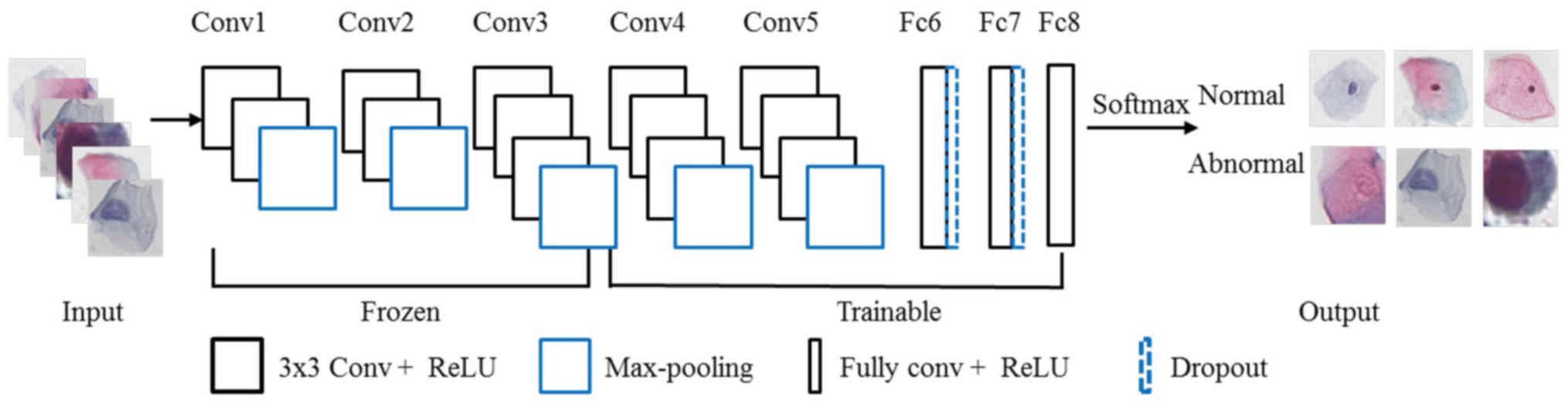
model comprises 16 layers, including 13 convolutional layers with
3×3 convolution kernels and 3 fully connected layers. At the same
time, there are also five max-pooling layers for feature
dimensionality reduction and two drop-out layers to prevent
over-fitting. First, the original single-cell images were input
into the network and represented by a matrix. Then, the
convolutional layer and the max-pooling layer extracted the deep
features and mapped them onto a hidden layer. The fully connected
layers distributed the features onto the sample tag space. Finally,
the last fully connected layer with a softmax classifier was used
to predict the classification probability (Fig. 3). In our experiment, the activation
functions were measured as Rectified Linear Units (ReLU).
An ensemble training strategy (ETS) was proposed
based on 5-fold cross-validation and was employed in our training
stage. In our strategy, the training set was randomly divided into
five equal parts, 4/5 for training and the remaining 1/5 for
validation. This division was repeated five times, and the previous
model was used as a pre-training model for the next iteration each
time. The last repeated model parameter indicated the final
result.
Transfer learning was used to improve the
generalizability of the model (22).
The dataset was used to fine-tune the DCNN model that was trained
on the ImageNet dataset (23). To
reduce computational complexity, the first seven layers of the DCNN
network were frozen, and the remaining nine layers were trainable
in the training stage.
We further improved the model generalizability and
prevented over-fitting with early-stopping (24,25). The
classical stochastic gradient descent optimizer was selected, and
the learning rate changed adaptively with the introduction of a
variable factor G, defined as:
G:=(Lti-Lvi)2ddLVi
where, Lit indicates the loss
value at the ith iteration in the training stage, and
Liv stands for the loss value in the
ith iteration in the validation stage. When G is
greater than a threshold (set to 5 in our experiment), the learning
rate is adjusted to 0.1 times the current learning rate.
Evaluation
The performance of the proposed DCNN model was
evaluated by measuring its accuracy, precision, sensitivity,
F1 values, and speed. Accuracy was defined as the
proportion of the correct classified samples of the total samples,
and precision was defined as the ratio of the positive sample that
was correctly classified. Sensitivity was expressed as the ratio of
negative samples, which were correctly classified as the total
negative samples. F1 reflected the performance of the
model and is a composite index that reflects both sensitivity and
specificity. Speed was defined as the average time that one cell
was identified, and it was calculated to evaluate the efficiency of
the DCNN model. In addition, the independent component analysis
(ICA), which is a popular method to represent multivariate data
(26), was used to visualize the
features of cells in the test dataset before and after
classification by the DCNN model (27).
Two experienced pathologists (pathologist 1 and 2,
12-years and 3-years working experience, respectively) were invited
to classify the squamous epithelial cells independently, and their
performance was compared with that of the DCNN models.
Statistical analysis
The comparison of the classification accuracy
between the DCNN models and the pathologists was conducted by the
paired sample t-tests. The statistical analyses were performed
using SPSS software (version 16.0). P<0.05 was considered to
indicate a statistically significant difference.
Results
DCNN models and classification
accuracy
The accuracy, precision, sensitivity, and
F1 of the DCNN model with ETS achieved slightly better
performance in identifying lesion cells in the test dataset than
those of the model without ETS and the pathologists (Table II). The training strategy improved
the accuracy by 1.29%, the precision by 1.22%, the sensitivity by
1.12%, and the F1 value by 1.32%. Compared with
Pathologist 1, the DCNN model with ETS had 0.80% lower accuracy,
1.13% lower precision, 0.80% lower sensitivity, and 0.86% lower
F1 value. Compared with Pathologist 2, the DCNN model
with ETS had 0.33% higher accuracy, 0.85% higher precision, 1.47%
higher F1 value, but 0.5% lower sensitivity. When the
accuracy was examined by lesion subtype, the differences between
the accuracy of the models and that of each pathologist were not
significant (Table II; DCNN without
ETS vs. DCNN with ETS: P0=0.387>0.05, DCNN with ETS
vs. Pathologist 1: P1=0.771>0.05, DCNN with ETS vs.
Pathologist 2: P2=0.489>0.05). The accuracy rate of
the DCNN model with ETS varied by lesion subtype, with ASC-H and
HSIL having the highest accuracies (100%) and ASC-US having the
lowest (75%; Table III).
 | Table II.Performance of the DCNN models and the
pathologists on the test dataset. |
Table II.
Performance of the DCNN models and the
pathologists on the test dataset.
| Variables | DCNN without ETS | DCNN with ETS | Pathologist 1 | Pathologist 2 |
|---|
| Accuracy (%) | 96.78 | 98.07 | 98.87 | 97.74 |
| Precision (%) | 96.69 | 97.91 | 99.10 | 97.06 |
| Sensitivity
(%) | 96.89 | 98.01 | 98.81 | 98.51 |
| F1
(%) | 96.77 | 98.09 | 98.95 | 96.62 |
| Speed (sec) | 0.0768 | 0.0732 | 0.4638 | 0.4992 |
 | Table III.Accuracy of the model with ETS for
different lesion subtype classifications. |
Table III.
Accuracy of the model with ETS for
different lesion subtype classifications.
|
| Normal | Abnormal |
|---|
|
|
|
|
|---|
| Variables | NILM | ASC-US | ASC-H | LSIL | HSIL | SCC |
|---|
| Test dataset | 285 | 24 | 2 | 92 | 211 | 7 |
|
Correct-classified | 282 | 18 | 2 | 90 | 211 | 6 |
|
Misclassification | 3 | 6 | 0 | 2 | 0 | 1 |
| Accuracy (%) | 98.95 | 75 | 100 | 97.83 | 100 | 85.71 |
DCNN models and classification
efficiency
In the classification efficiency, the results showed
that the DCNN model with ETS achieved the fastest speed (0.0732
sec; Table II), and it was 0.0036
sec faster compared with that without ETS, 0.3906 sec faster
compared with Pathologist 1, and 0.4260 sec faster compared with
Pathologist 2 (Table II). The
classification speed of the DCNN models was almost six times faster
than that of the pathologists.
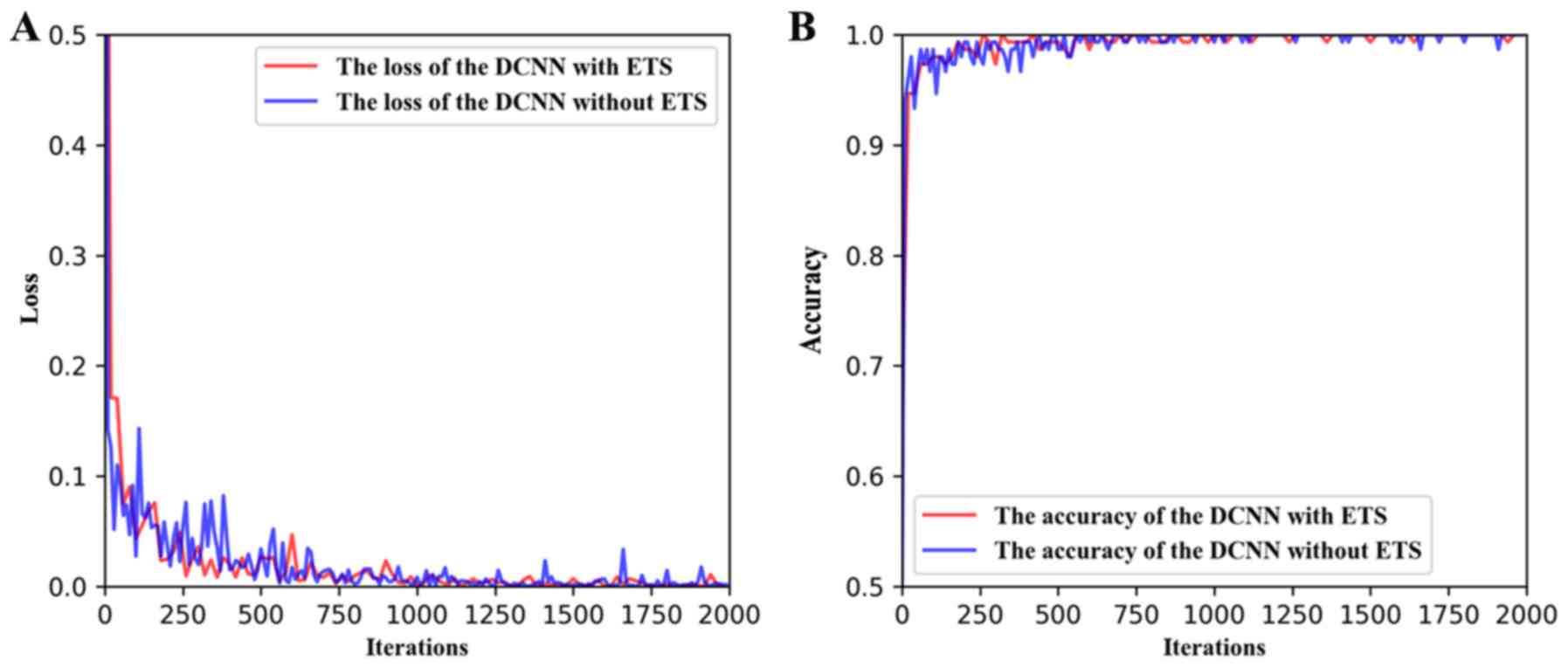
We visualized the loss and accuracy values during
the training stage of the DCNN models. The final loss values were
both <0.1, and the accuracy values were >0.9 after several
iterations (Fig. 4). However, the
values for the DCNN with ETS were always lower and smoother than
that without ETS, indicating that the DCNN model with the ETS had a
more stable performance (Fig.
4).
DCNN model and ICA
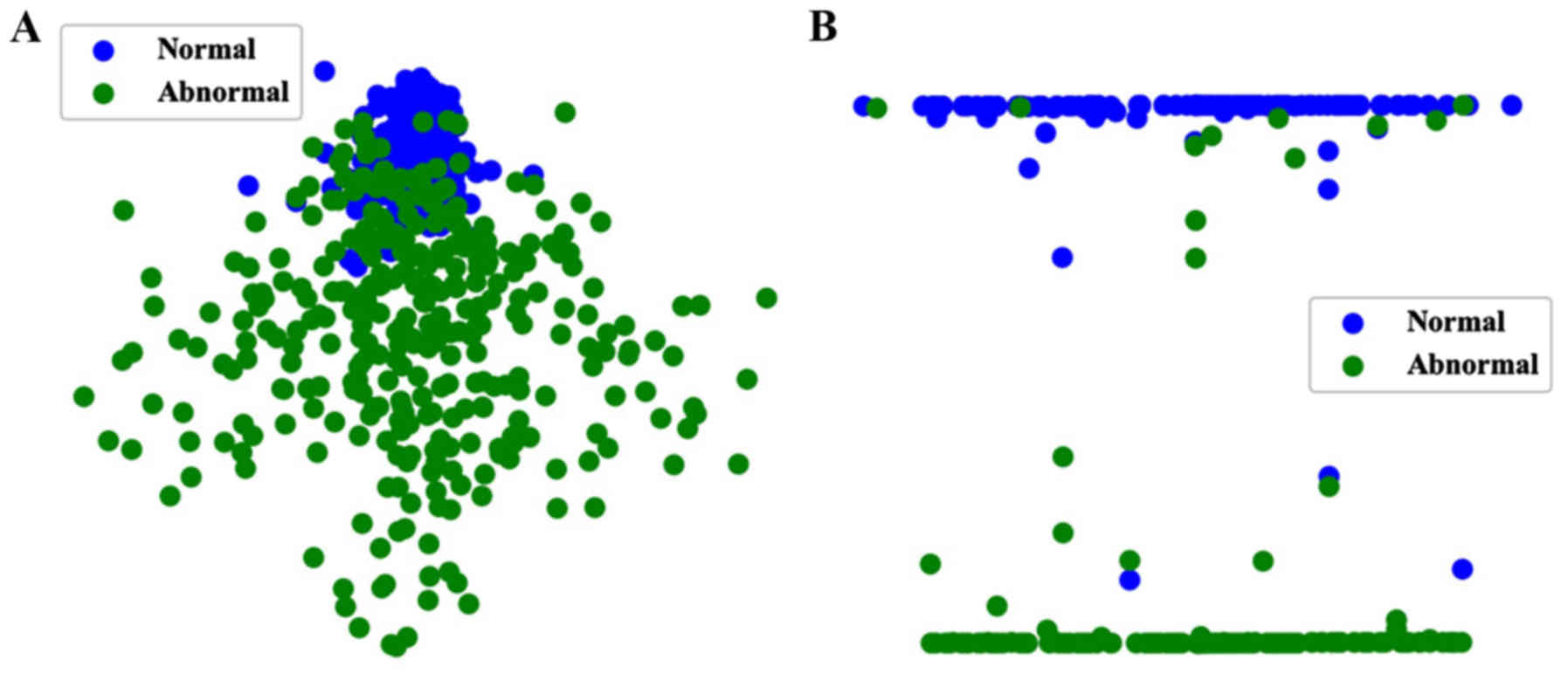
Visualization of the test dataset using ICA revealed
that the normal and abnormal cells were separated into two distinct
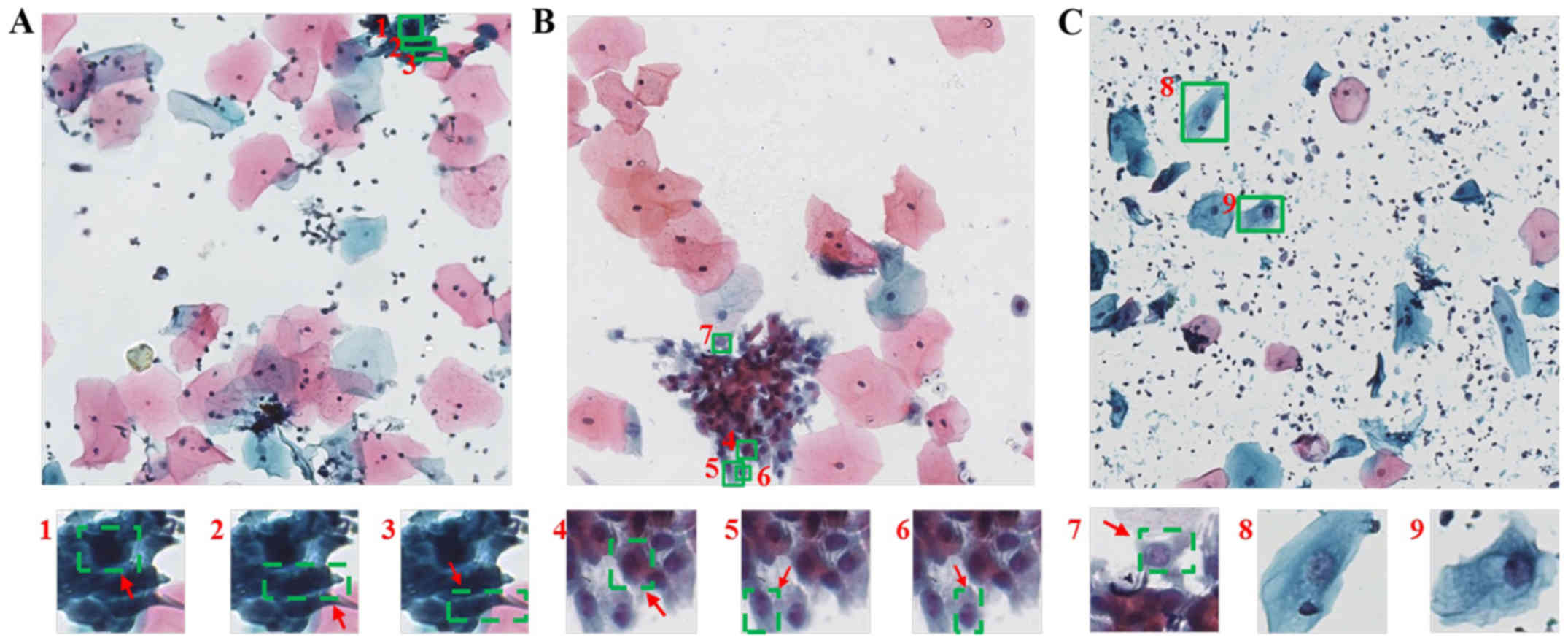
classes after identification with the DCNN model with ETS (Fig. 5). Simultaneously, we randomly showed
some cells which were classified correctly and put into 2048×2048
images (Fig. 6). It indicated that
the DCNN model could classify the cells which had unclear nuclei,
even in the cell cluster.
Discussion
In the present study, we developed a DCNN model
based on VGG16 for the automated classification of cervical
squamous cells that accurately differentiated between normal cells
and lesion cells in a multi-center TCT dataset. Compared to
classifications independently provided by two experienced
pathologists, the AI system achieved similar classification
accuracy with improved efficiency.
Our datasets were composed of many TCT images that
originated from four different hospitals that differed in their
trauma levels and TCT machines. These sources of variation provided
diversity within the dataset that was used to train and optimize
the DCNN algorithm, thereby improving the generalizability of this
model.
Relative to the large-scale dataset, like the
millions of ImageNet datasets (23),
required for the development of most DCNN models in the training
stage, our dataset is small. Therefore, we employed ETS based on
5-fold cross-validation, which improved the accuracy and speed
compared with the traditional DCNN model. At the same time, in
terms of model performance, the loss value obtained by the DCNN
model with ETS could be fitted better and faster than the model
without ETS. Even though the inclusion of ETS did not significantly
improve the accuracy, the incremental increase in accuracy may
still be clinically important. Future investigations will continue
to verify the classification effect of this method for future
studies.
The classification accuracy of the DCNN model with
ETS was 98.07% for our multi-center dataset. The ICA analysis found
that the characteristics of normal squamous epithelial cells before
classification were more concentrated, and the characteristics of
abnormal cells were scattered and polymorphic. This observation may
be mainly due to the presence of many subtypes in our test dataset,
including ASC-US, LSIL, ASC-H, HSIL, and SCC. The characteristics
of some abnormal cells are very similar to each other. Meanwhile,
the features of normal and abnormal cervical squamous epithelial
cells classified by the DCNN model were clearly distinguished,
which indicates that the DCNN could accurately classify cervical
squamous epithelial cells with fine-grained characteristics.
The DCNN model with ETS differed in classification
accuracy among lesion subtypes. SCC is the most direct evidence for
the diagnosis of cervical cancer (7). Compared with other precancerous cells,
SCC contains significant nuclear atypia, yet classifications with
our model only achieved 85.71% accuracy. This lower accuracy may be
due to the significantly smaller number of SCC cells than the
number of other classes in our training dataset (n=39), which is
called class-imbalance problem. The low classification accuracy for
ASC-US, 75%, may be due to the high similarity of features between
ASC-US, NSIL, and LSIL. In fact, even experienced pathologists can
experience difficulty distinguishing among these subtypes.
Observation of the cells which were correctly classified
demonstrated that even though the cells were isolated from cell
clusters, were small, had difficulty in distinguishing deep nuclear
staining, or had unclear nuclei, the model could still accurately
classify these samples.
The DCNN model developed here provides a similar
classification performance to that of the pathologists, but with a
highly improved classification efficiency. This result indicates
that our model could assist pathologists with the interpretation of
TCT specimens and reduce their workload. In the future, this highly
efficient automated method may be widely used in clinical settings
to overcome the subjective and time-consuming nature of the manual
diagnosis.
Researchers have previously proposed DCNN models to
classify samples of cervical cells. Zhang et al (13), used a DCNN to classify normal and
abnormal cervical cells and found accuracies of 98.3 and 98.6% for
the two public datasets, Herlev dataset and HEMLBC dataset,
respectively. However, the testing time for one cervical cell
averaged 3.5 sec. Wu et al (14), used a DCNN to classify keratinizing
squamous, non-keratinizing squamous, and basaloid squamous cells in
a single-center dataset, and achieved an accuracy of 93.33% (speed
was not reported). While the accuracies reported in these two
studies were high, the authors did not externally validate the
results by comparing classification with those provided by
pathologists. Furthermore, the diagnosis efficiency reported by
Zhang et al (13) did not
satisfy the need of the clinic.
In summary, our study focused on developing an
accurate and efficient automated method that could be used in
clinics to assist pathologists in interpreting TCT
early-screenings. There are four main limitations of the method
employed in the present study: i) We used images of single cells
that were extracted from whole slide images in advance. Future
studies should focus on detecting the cervical squamous lesion
cells in whole slide images without any prior segmentation or
splits. ii) Our study only included a binary classification of
cervical squamous cells. Future studies should focus on a
multi-classification model for cervical squamous cells. iii) The
small size of our dataset and the class-imbalance problem have a
negative effect on the results, thus more data should be collected
from clinics, especially on ASC-US, ASC-H, and SCC cells in the
future. iv) Our data was collected from the southwestern region of
China, and this geographically limited dataset may limit the
generalizability of the resulting model. Future studies should
focus on collecting data globally.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Key
Research and Development Plan (grant no. 2016YFC0106403), the
National Natural Science Foundation of China (grant nos. 31771324
and 61701506), the Military Medical Research Program Youth
Development Project (grant nos. 16QNP100 and 2018XLC3023) and the
Graduate Education and Teaching Reform Project of Chongqing (grant
no. yjg183144).
Availability of data and materials
The datasets analyzed during the present study are
not publicly available as they are part of an ongoing study.
Authors' contributions
LL conceived the study, analyzed the data, performed
the statistical analysis and drafted the initial manuscript. LL and
JX created the DCNN model and analyzed the data. YW and QM
collected the TCT dataset and performed pathological analysis. LT
and YW were involved in project development and critically reviewed
the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the Army Military Medical University (Chongqing,
China; approval no. KY201774). The study was explained to all the
patients, and oral informed consent was obtained from them for
their sample to be used for scientific research by phone when
images were collected. Written patient consent was not required,
according to the guidance of the ethics committee.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Small W Jr, Bacon MA, Bajaj A, Chuang LT,
Fisher BJ, Harkenrider MM, Jhingran A, Kitchener HC, Mileshkin LR,
Viswanathan AN and Gaffney DK: Cervical cancer: A global health
crisis. Cancer. 123:2404–2412. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Globocan, . 2018.China factsheet.
July
18–2020
|
|
4
|
Cheng WQ, Zheng QS, Zhang SW, Zeng HM, Zuo
TT, Jia MM, Xia CF, Zhou XN and He J: Analysis of incidence and
death of malignant tumors in China in 2012. China Cancer. 25:1–8.
2016.
|
|
5
|
Chacho MS, Mattie ME and Schwartz PE:
Cytohistologic correlation rates between conventional Papanicolaou
smears and ThinPrep cervical cytology: A comparison. Cancer.
99:135–140. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Behtash N and Mehrdad N: Cervical cancer:
screening and prevention. Asian Pac J Cancer Prev. 7:683–686.
2006.PubMed/NCBI
|
|
7
|
Ritu N and Wilbur D: The Bethesda System
for reporting cervical cytology: Definitions, criteria, and
explanatory notes. Springer International Publishing; Berlin: 2015,
PubMed/NCBI
|
|
8
|
Xie Y, Zhang J, Xia Y and Shen C: A mutual
bootstrapping model for automated skin lesion segmentation and
classification. IEEE Trans Med Imaging. 39:2482–2493. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gehlot S, Gupta A and Gupta R:
SDCT-AuxNet°: DCT augmented stain deconvolutional CNN with
auxiliary classifier for cancer diagnosis. Med Image Anal.
61:1016612020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Murugan A, Nair SAH and Kumar KPS:
Detection of skin cancer using SVM, random forest and kNN
classifiers. J Med Syst. 43:2692019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Abdulkerim C, Abdülkadir A, Aslı Ü,
Nurullah Ç, Gökhan B, Ilknur T, Aslı Ç, Behçet UT and Lütfiye DA:
Segmentation of precursor lesions in cervical cancer using
convolutional neural networks. 25th Signal Processing
and Communications Applications Conference (SIU). 2017.
|
|
12
|
Chankong T, Theera-Umpon N and
Auephanwiriyakul S: Automatic cervical cell segmentation and
classification in Pap smears. Comput Methods Programs Biomed.
113:539–556. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang L, Lu L, Nogues I, Summers RM, Liu S
and Yao J: DeepPap: Deep convolutional networks for cervical cell
classification. IEEE J Biomed Health Inform. 21:1633–1643. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wu M, Yan C, Liu H, Liu Q and Yin Y:
Automatic classification of cervical cancer from cytological images
by using convolutional neural network. Biosci Rep.
38:BSR201817692018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tian Y, Yang L, Wang W, Zhang J, Tang Q,
Ji M, Yu Y, Li Y, Yang H and Qian A: Computer-aided detection of
squamous carcinoma of the cervix in whole slide images. arXiv
1905.10959. 2019.
|
|
16
|
Liu L, Wang Y, Wu D, Zhai Y, Tan L and
Xiao J: Multi-task learning for pathomorphology recognition of
squamous intraepithelial lesion in Thinprep cytologic test. ISICDM.
10:73–77. 2018.
|
|
17
|
Miyagi Y, Takehara K, Nagayasu Y and
Miyake T: Application of deep learning to the classification of
uterine cervical squamous epithelial lesion from colposcopy images
combined with HPV types. Oncol Lett. 19:1602–1610. 2020.PubMed/NCBI
|
|
18
|
Weiss S, Xu ZZ, Shyamal P, Amir A,
Bittinger K, Gonzalez A, Lozupone C, Zaneveld JR, Vázquez-Baeza Y,
Birmingham A, et al: Normalization and microbial differential
abundance strategies depend upon data characteristics. Microbiome.
5:272017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Simonyan K and Zisserman A: Very deep
convolutional networks for large-scale image recognition. arXiv:
1409.1556. 2014.
|
|
20
|
Saikia AR, Bora K, Mahanta LB and Das AK:
Comparative assessment of CNN architectures for classification of
breast FNAC images. Tissue Cell. 57:8–14. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sharma S and Mehra R: Conventional machine
learning and deep learning approach for multi-classification of
breast cancer histopathology images-a comparative insight. J Digit
Imaging. 33:632–654. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Pan SJ and Yang Q: A survey on transfer
learning. IEEE Trans Knowledge Data Eng. 10:1345–1359. 2010.
View Article : Google Scholar
|
|
23
|
Deng J, Dong W, Socher R, Li LJ, Li K and
Li FF: ImageNet: A large-scale hierarchical image database. IEEE
Conference on Computer Vision and Pattern Recognition. 248–255.
2009.
|
|
24
|
Takase T, Oyama S and Kurihara M:
Effective neural network training with adaptive learning rate based
on training loss. Neural Netw. 101:68–78. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mo Z, Wang Q, Tian S, Yan R, Geng J, Yao Z
and Lu Q: Evaluating treatment via flexibility of dynamic MRI
community structures in depression. J Southeast Univ. 33:273–276.
2017.
|
|
26
|
Hyvärinen A and Oja E: Independent
component analysis: Algorithms and applications. Neural Netw.
13:411–430. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Taqi AM, Awad A, Al-Azzo F and Milanova M:
The impact of multi-optimizers and data augmentation on TensorFlow
convolutional neural network performance. IEEE Conference on
Multimedia Information Processing Retrieval. 140–145. 2018.
|