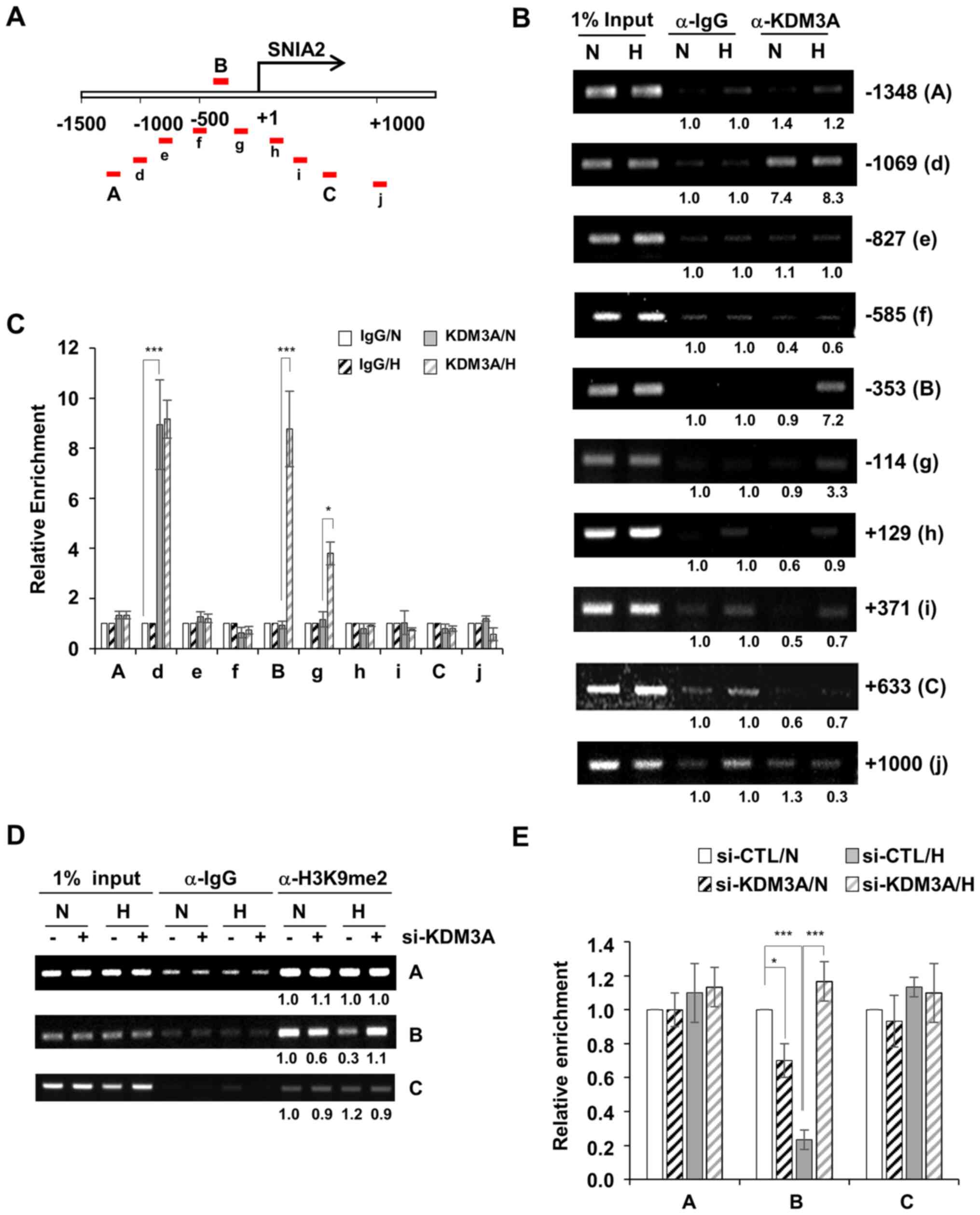
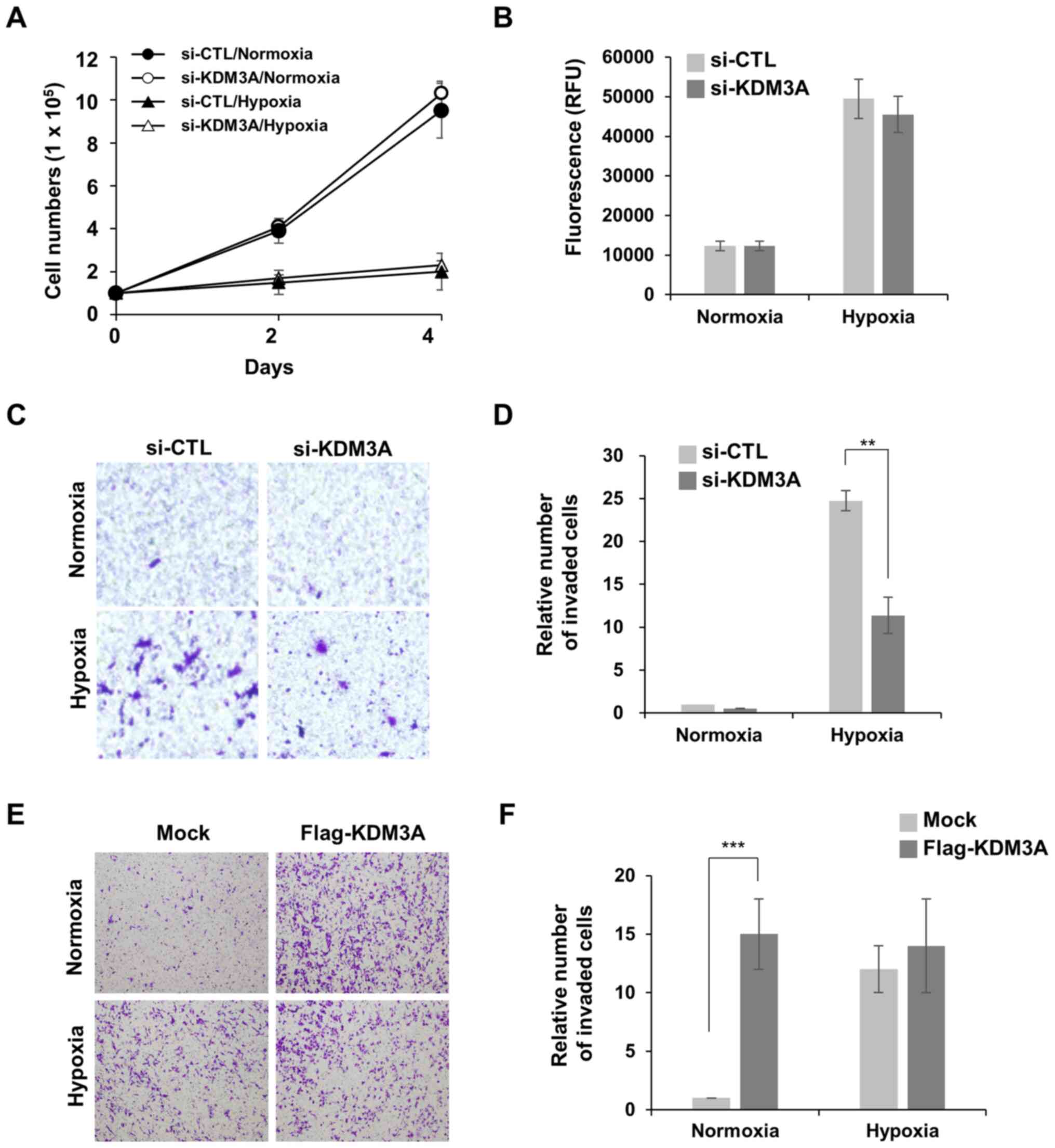
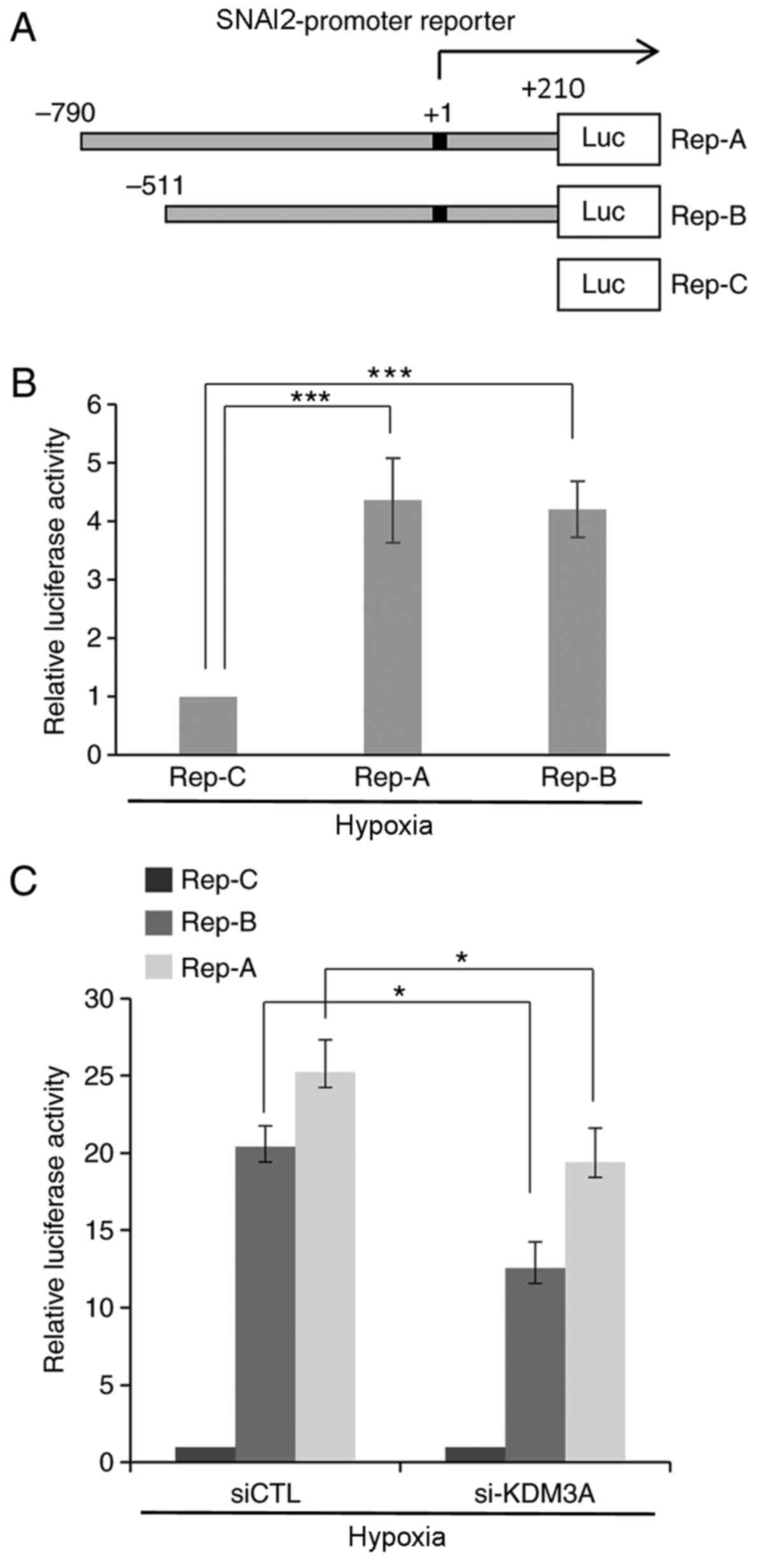
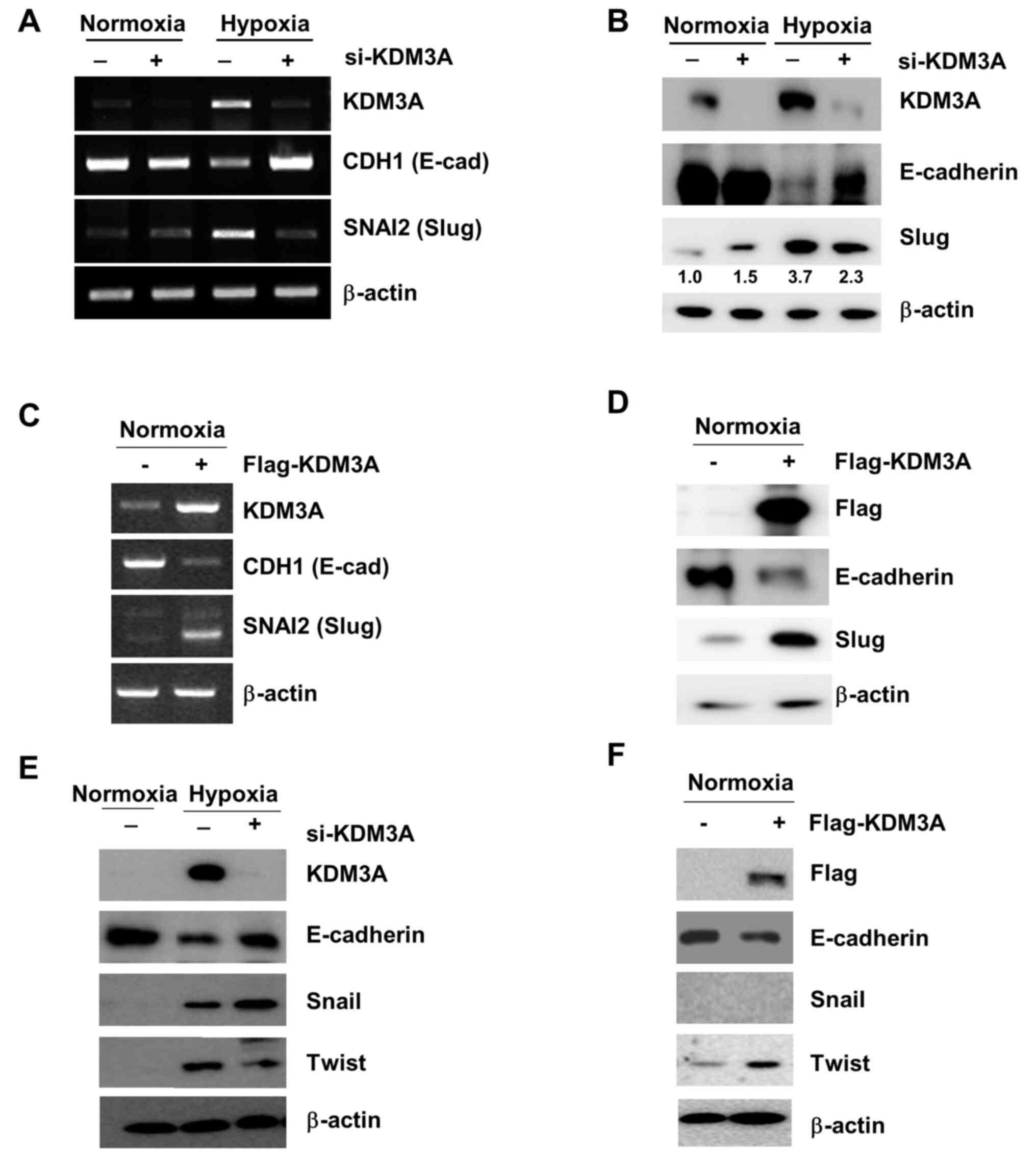
|
1
|
Rankin EB, Nam JM and Giaccia AJ: Hypoxia:
Signaling the Metastatic Cascade. Trends Cancer. 2:295–304. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Eales KL, Hollinshead KE and Tennant DA:
Hypoxia and metabolic adaptation of cancer cells. Oncogenesis.
5:e1902016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chouaib S, Messai Y, Couve S, Escudier B,
Hasmim M and Noman MZ: Hypoxia promotes tumor growth in linking
angiogenesis to immune escape. Front Immunol. 3:212012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Semenza GL: Hypoxia-inducible factors in
physiology and medicine. Cell. 148:399–408. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xia X, Lemieux ME, Li W, Carroll JS, Brown
M, Liu XS and Kung AL: Integrative analysis of HIF binding and
transactivation reveals its role in maintaining histone methylation
homeostasis. Proc Natl Acad Sci USA. 106:4260–4265. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hancock RL, Dunne K, Walport LJ, Flashman
E and Kawamura A: Epigenetic regulation by histone demethylases in
hypoxia. Epigenomics. 7:791–811. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Luo W, Chang R, Zhong J, Pandey A and
Semenza GL: Histone demethylase JMJD2C is a coactivator for
hypoxia-inducible factor 1 that is required for breast cancer
progression. Proc Natl Acad Sci USA. 109:E3367–E3376. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mimura I, Nangaku M, Kanki Y, Tsutsumi S,
Inoue T, Kohro T, Yamamoto S, Fujita T, Shimamura T, Suehiro J, et
al: Dynamic change of chromatin conformation in response to hypoxia
enhances the expression of GLUT3 (SLC2A3) by cooperative
interaction of hypoxia-inducible factor 1 and KDM3A. Mol Cell Biol.
32:3018–3032. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee HY, Yang EG and Park H: Hypoxia
enhances the expression of prostate-specific antigen by modifying
the quantity and catalytic activity of Jumonji C domain-containing
histone demethylases. Carcinogenesis. 34:2706–2715. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Park SJ, Kim JG, Son TG, Yi JM, Kim ND,
Yang K and Heo K: The histone demethylase JMJD1A regulates
adrenomedullin-mediated cell proliferation in hepatocellular
carcinoma under hypoxia. Biochem Biophys Res Commun. 434:722–727.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ueda J, Ho JC, Lee KL, Kitajima S, Yang H,
Sun W, Fukuhara N, Zaiden N, Chan SL, Tachibana M, et al: The
hypoxia-inducible epigenetic regulators Jmjd1a and G9a provide a
mechanistic link between angiogenesis and tumor growth. Mol Cell
Biol. 34:3702–3720. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Favaro E, Lord S, Harris AL and Buffa FM:
Gene expression and hypoxia in breast cancer. Genome Med. 3:552011.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Serrano-Gomez SJ, Maziveyi M and Alahari
SK: Regulation of epithelial-mesenchymal transition through
epigenetic and post-translational modifications. Mol Cancer.
15:182016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jiang J, Tang YL and Liang XH: EMT: A new
vision of hypoxia promoting cancer progression. Cancer Biol Ther.
11:714–723. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Flavahan WA, Gaskell E and Bernstein BE:
Epigenetic plasticity and the hallmarks of cancer. Science.
357:eaal23802017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Perez-Perri JI, Acevedo JM and Wappner P:
Epigenetics: New questions on the response to hypoxia. Int J Mol
Sci. 12:4705–4721. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chakraborty D, Cui W, Rosario GX, Scott
RL, Dhakal P, Renaud SJ, Tachibana M, Rumi MA, Mason CW, Krieg AJ,
et al: HIF-KDM3A-MMP12 regulatory circuit ensures trophoblast
plasticity and placental adaptations to hypoxia. Proc Natl Acad Sci
USA. 113:E7212–E7221. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wan W, Peng K, Li M, Qin L, Tong Z, Yan J,
Shen B and Yu C: Histone demethylase JMJD1A promotes urinary
bladder cancer progression by enhancing glycolysis through
coactivation of hypoxia inducible factor 1α. Oncogene.
36:3868–3877. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ramadoss S, Guo G and Wang CY: Lysine
demethylase KDM3A regulates breast cancer cell invasion and
apoptosis by targeting histone and the non-histone protein p53.
Oncogene. 36:47–59. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wade MA, Jones D, Wilson L, Stockley J,
Coffey K, Robson CN and Gaughan L: The histone demethylase enzyme
KDM3A is a key estrogen receptor regulator in breast cancer.
Nucleic Acids Res. 43:196–207. 2015. View Article : Google Scholar : PubMed/NCBI
|