Introduction
Worldwide, lung cancer is the most common cause of
cancer-associated mortality, accounting for 11.6% of all new cancer
cases and 18.4% of all cancer-associated mortalities in 2018
(1). The high mortality rate of this
disease is attributable to the diagnosis of the majority of
patients at an incurable stage (2).
Non-small cell lung cancer (NSCLC) accounts for >80% of all
diagnosed lung cancer cases (3).
Platinum-based chemotherapy, one of the primary agents used for
treatment of NSCLC for the last two decades, has only resulted in a
modest improvement in overall survival (4). Despite the development of drugs that
target the growth of tumors, the overall survival rate of patients
with NSCLC has remained low (5).
Thus, identifying novel prognostic factors may assist clinical
decisions and patient prognosis.
Coiled-coil domain-containing (CCDC) proteins
participate in a variety of regulatory functions associated with
their highly versatile coiled-coil motif (6). A range of CCDC proteins have been
demonstrated to be associated with the development and progression
of various malignant tumors, such as papillary thyroid carcinoma,
breast tumor, bladder cancer and esophageal squamous cell carcinoma
(7–10). The tumor-suppressive role of CCDC68
in tumors was initially reported in colorectal adenocarcinoma
(11), which was also subsequently
demonstrated in pancreatic ductal adenocarcinoma (PDAC) (12). However, the role of CCDC68 in NSCLC
has not been extensively studied. Thus, the present study aimed to
assess the expression of CCDC68 in lung cancer cell lines and
determine the effects of CCDC68 knockdown.
Materials and methods
Cell culture and treatment
All cell lines were purchased from The Cell Bank of
Type Culture Collection of the Chinese Academy of Sciences and
cultured at 37°C with 5% CO2 in a humidified incubator.
A549 cells were maintained in DME/F12 medium (Sigma-Aldrich; Merck
KGaA) supplemented with 10% FBS (Gibco; Thermo Fisher Scientific,
Inc.) and 1% penicillin-streptomycin (HyClone; Cytiva). NCI-H1299,
NCI-H1975 and 95-D cells were all cultured in DMEM (Gibco; Thermo
Fisher Scientific, Inc.) supplemented with 10% FBS and 1%
penicillin-streptomycin.
Tissue samples
A total of 15 paired NSCLC and normal lung tissues
were collected after resection from Xi'an Chest Hospital between
June 2018 to February 2019. The patients included 9 males and 6
females, with a mean age of 56 years (range, 38–71). The distance
between normal lung tissue and lung cancer tissue was >3 cm. The
tissues were frozen in −80°C. The patients who participated in this
study did not undergo any treatment before surgery. Written
informed consent was obtained from each participant, and the study
was approved by the Ethics Committee of Xi'an Chest Hospital (no.
2019-S0014).
Lentiviral vector transfection
Short hairpin (sh)RNAs were constructed using a
linearized GV115 lentiviral vector. Escherichia coli strain
DH5α was used to amplify the lentiviral vector and auxiliary
packaging vector plasmid (all purchased from Shanghai GeneChem Co.,
Ltd.). The plasmids were then transfected into 293T cells and
cultured in DMEM medium containing 10% FBS. After 48–72 h in
culture, the virus was harvested. The sequence of the CCDC68 shRNA
and the shCtrl were 5′-GAAGCCCAGAATAAAGAACTA-3′ and
5′-TTCTCCGAACGTGTCACGT-3′, respectively. The shCtrl lentivirus was
termed psc3741 and the CCDC-shRNA lentivirus was termed
LVpGCSIL-004PSC50502-1. For lentiviral transfection, NSCLC cells
were plated in 6-well plates (1×105 cell/well) and the
shCtrl or the CCDC68-shRNA lentivirus was added according to the
multiplicity of infection (MOI=5), using Lipofectamine®
2000 transfection reagent (cat. no. 11668019; Thermo Fisher
Scientific, Inc.). Cultured cells were inspected at 72 h
post-transfection with a fluorescence microscope (Olympus
Corporation) at ×100 magnification.
Tissue immunohistochemistry
Paraffin-embedded paired NSCLC and normal lung
tissues were cut into 4-µm sections and dried at 60°C overnight.
The sections were dewaxed in dimethylbenzene solution twice at room
temperature for 10 min and rehydrated through graded ethanol to
water, followed by antigen retrieval and penetration in 0.5% Triton
X-100 in PBS at room temperature for 20 min. Endogenous peroxidase
was blocked with 3% hydrogen peroxide at room temperature for 10
min, washed with PBS for 3 min three times, blocked at room
temperature for 30 min with normal goat serum (Abcam) and incubated
with the rabbit anti-CCDC68 primary antibody (1:400; cat. no.
ab97815; Abcam) overnight at 4°C. Subsequently, Goat anti-rabbit
IgG antibody (1:2,000; cat. no. 7074; CST Biological Reagents Co.,
Ltd.) was added, then the sections were incubated at room
temperature for 1 h and washed with PBS. 3′3′-Diaminobenxidine
(Beyotime Institute of Biotechnology) was used to develop the color
for 3 min at room temperature. Slides were counterstained with
hematoxylin for 5 min at room temperature, mounted after rinsing
with water and observed using a light microscopy (Olympus
Corporation).
RNA extraction and reverse
transcription-quantitative (RT-q)PCR
Total RNA of transfected A549 and NCI-H1299 cells
was extracted using SuperfecTRI (Shanghai Pufei Biotechnology Co.,
Ltd.) according to the manufacturer's protocol. An ultraviolet
spectrophotometer was used to measure the concentration of
extracted RNA. A total of 2 µg RNA and 2 µl (0.5 µg/µl) reverse
transcription primer (Bulge-Loop™ miRNA qPCR Primer Set; cat. no.
MQP-0102; Guangzhou RiboBio Co., Ltd.) were reverse-transcribed
using an M-MLV Reverse Transcriptase kit (cat. no. M1705; Promega
Corporation) and dNTPs (cat. no. U1240; Promega Corporation)
according to the manufacturer's protocol. SYBR® Master
mix Real-Time PCR system (Takara Bio, Inc.) was used to perform
qPCR in 12-µl reactions with the following reaction conditions:
Pre-denaturation for 30 sec at 95°C; 40 cycles of 5 sec at 95°C and
30 sec at 60°C; and dissociation for 15 sec at 95°C, 30 sec at 60°C
and 15 sec at 95°C. The sequences of the primers (Shanghai GeneChem
Co., Ltd.) were as follows: CCDC68 forward,
5′-CCTTGTATGAGTCTACGTCCGC-3′ and reverse,
5′-ATCTGGGTCCTGATCTTTTGC−3′; GAPDH forward,
5′-TGACTTCAACAGCGACACCCA−3′ and reverse,
5′-CACCCTGTTGCTGTAGCCAAA−3′. GAPDH was used as the loading control.
The relative gene expression levels were measured and compared
using the 2−ΔΔCq method (13).
Cell proliferation assay
A549 and NCI-H1299 transfected cells were digested
during the logarithmic growth phase using trypsin. The resuspended
cells were seeded in a 96-well plate (2×103 cells/well)
in 100 µl supplemented DME/F12 medium or DMEM and incubated
overnight in 5% CO2 incubator at 37°C. The number of
cells was continuously counted using a Celigo Imaging Cytometer
(Nexcelom Bioscience, Ltd.) for 5 days. To obtain the cell
proliferation ratio curve, the cell count value of each group of
cells at each time point was normalized to the first day of the
respective cell group.
Cell viability assay
A549 and NCI-H1299 transfected cells were seeded
into a 96-well culture plate at a density of 2,000 cells/well and
allowed to grow to subconfluence. 3-(4,5)-dimethylthiahiazo(−z-y1)-3,
5-di-phenytetrazoliumromide (MTT; Genview PTY, Ltd.) incubation was
performed according to the manufacturer's instructions. A total of
0.5 mg/ml MTT was added to each well and incubated for 4 h at 37°C,
followed by the addition of 100 µl DMSO (Shanghai Shiyi Chemical
Reagent Co., Ltd.). After 10 min of shaking, the optical density
(OD) of the cells was measured at 490 nm using a microplate reader
(Tecan Group, Ltd.). The mean count of three repetitions for 5
consecutive days in each group was used for analysis. The cell
viability curve was obtained by normalizing the OD values of each
time point to the first day of readings.
Apoptosis assay
A549 and NCI-H1299 transfected cells were passaged
on day 3 post-transfection and harvested when confluence reached
70%. Cells were digested with trypsin and resuspended with DME/F12
medium or DMEM, centrifuged at 1,300 × g for 5 min at room
temperature and washed twice in PBS. Cells were then washed in
pre-cooled D-Hanks solution (pH, 7.2–7.4; Beijing Solarbio Science
& Technology Co., Ltd.) at 4°C. The cell apoptosis kit (cat.
no. V35113; Thermo Fisher Scientific, Inc.) was then used to
perform the next steps, where 1X binding buffer was added to wash
cells, following by centrifugation at 1,300 × g for 3 min at room
temperature. Cells were then resuspended in 1X binding buffer and
stained by adding 10 µl Annexin V-allophycocyanin (Thermo Fisher
Scientific, Inc.) at room temperature in the dark for 15 min.
Subsequently, 1X binding buffer was added based on the cell volume,
and apoptosis was analyzed using a BD FACS Calibur flow cytometer
(Accuri™ C6 Plus; BD Biosciences).
Western blotting
Transfected A549 and NCI-H1299 cells were lysed
using a lysis buffer (100 mM Tris-HCl, pH 6.8; 100 mM DTT; 2% SDS;
10% glycerin; Beijing Solarbio Science & Technology Co., Ltd.),
and the protein concentration was determined using a BCA Protein
Assay kit (Beyotime Institute of Biotechnology). A total of 45 µg
protein from the cell lysates were resolved on a 10% SDS gel using
SDS-PAGE (Bio-Rad Laboratories, Inc.) and transferred to a PVDF
membrane (EMD Millipore). The membrane was blocked in TBS + 0.05%
Tween-20 solution containing 5% skimmed milk at 4°C overnight and
incubated with the primary antibodies rabbit anti-CCDC68 (1:300;
cat. no. ab97815; Abcam) and mouse anti-GAPDH (1:2,000; cat. no.
sc-32233; Santa Cruz Biotechnology, Inc.) primary antibodies
overnight at 4°C. Subsequently, the membrane was incubated with
goat anti-rabbit IgG (1:2,000; cat. no. 7074) or horse anti-mouse
IgG (1:2,000; cat. no. 7076; both from CST Biological Reagents Co.,
Ltd.) secondary antibody for 1.5 h at room temperature. An ECL
Western Blotting Substrate kit (Thermo Fisher Scientific, Inc.) was
used to visualize the protein bands.
Statistical analysis
Data are presented as the mean ± standard deviation.
Statistical analysis was performed using SPSS version 19.0 software
(IBM Corp.). Differences between two groups were compared using a
paired Student's t-test. P<0.05 was considered to indicate a
statistically significant difference.
Results
CCDC68 is highly expressed in NSCLC
tissues and cell lines
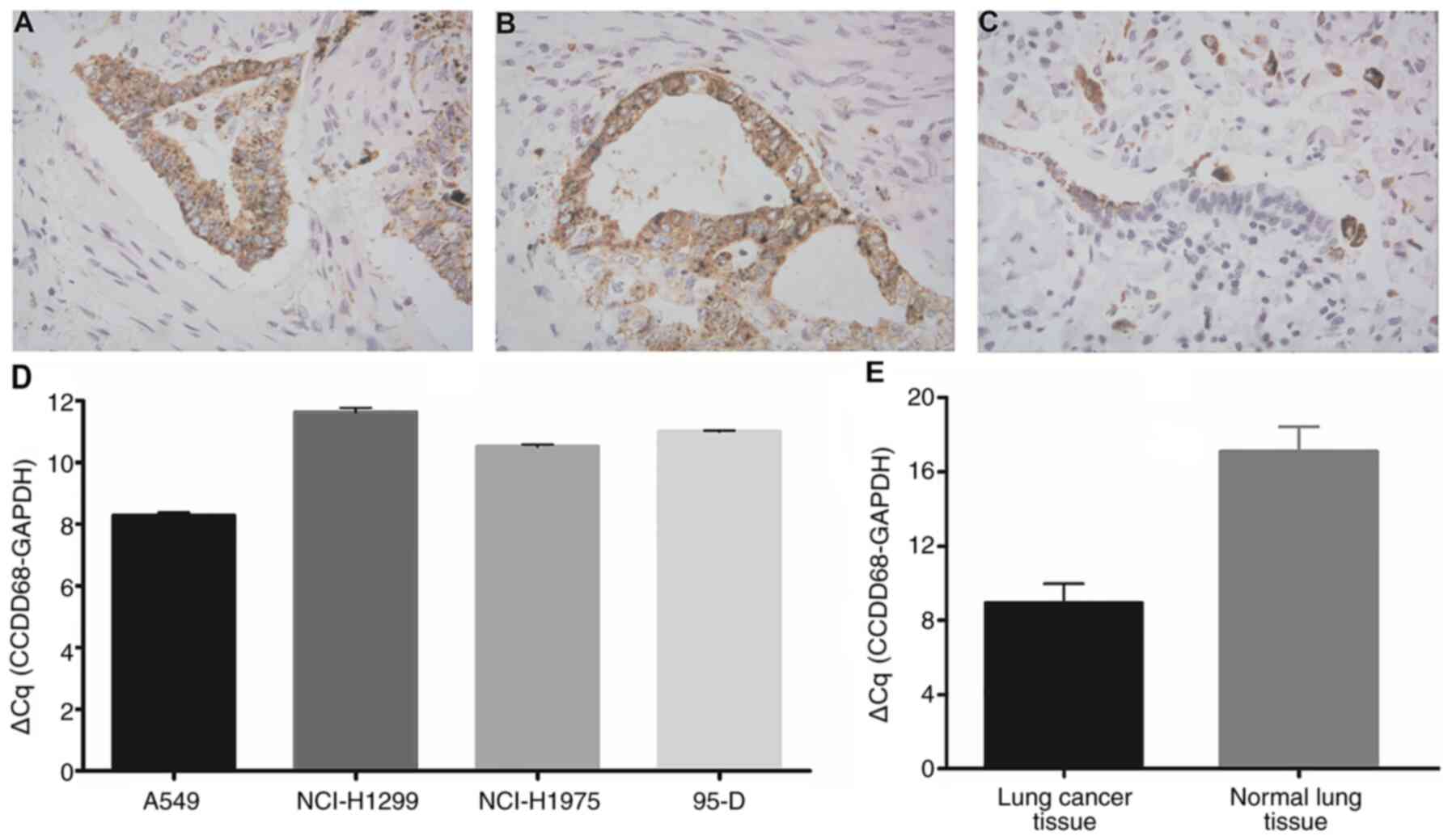
Immunohistochemistry analysis of tissues from
patients with NSCLC demonstrated that CCDC68 was strongly expressed
in the cytoplasm in lung cancer tissues (Fig. 1A and B). In normal lung tissues, weak
expression of CCDC68 was observed in bronchial columnar epithelial
and alveolar epithelial cells (Fig.
1C). CCDC68 mRNA expression levels were determined in A549,
H1299, H1975 and 95-D NSCLC cell lines, as well as in lung cancer
and normal lung tissues using RT-qPCR. The results demonstrated
that CCDC68 mRNA expression was high in all tested cell lines
(Fig. 1D) and in lung cancer tissue
(Fig. 1E). The four cell lines used
are all human NSCLC cell lines. A549, NCI-H1975 and 95-D are
derived from primary lung tissue, A549 is a commonly used and
mature gene knockout cell line. Unlike the other cell lines,
NCI-H1299 cells are derived from a lymph node metastasis, and these
cells partially lack p53 protein homogeneity (14). The simultaneous use of A549 and
NCI-H1299 cell lines in the present study can increase the
conviction of the experiments.
CCDC68 knockdown efficiency in
non-small-cell lung cancer cell lines
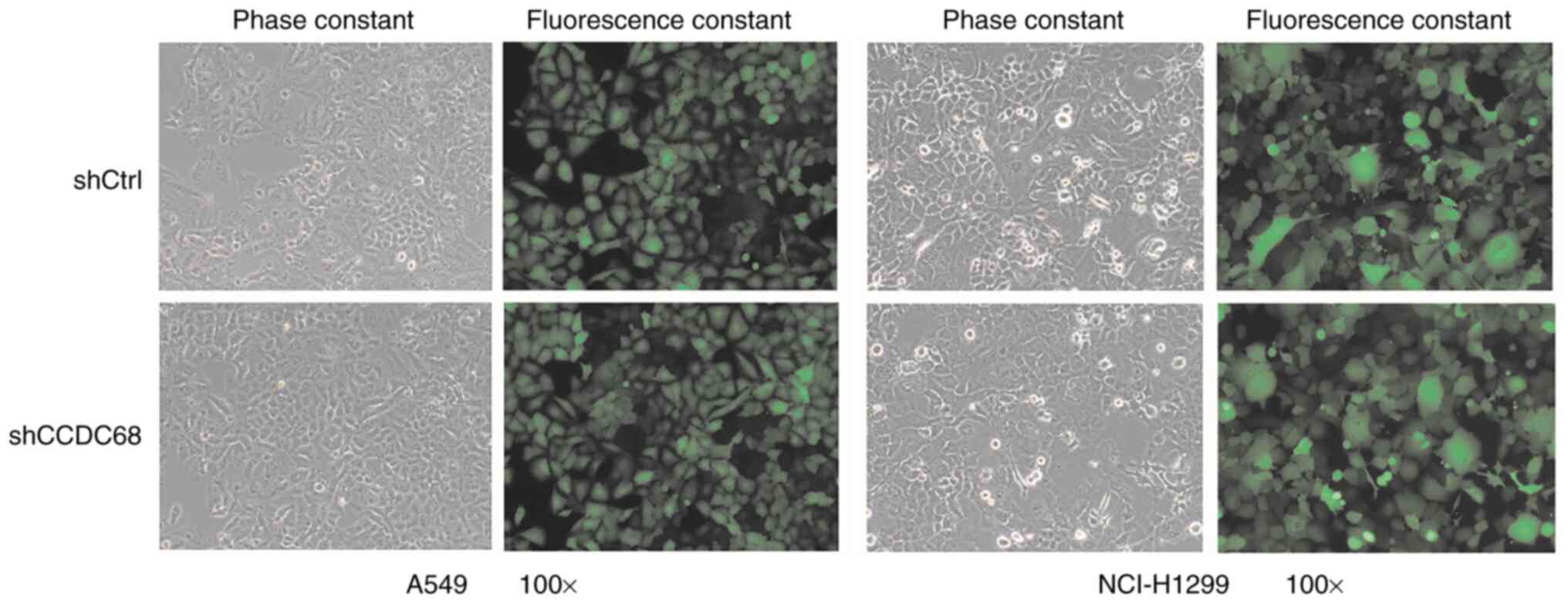
The shCCDC68 lentivirus was transfected into A549
and H1299 cells to knock down the expression of CCDC68.
Fluorescence microscopy was used to observe the transfection
efficiency. The cells transfected with shCCDC68 and shCtrl were
considered the experimental and the negative control group,
respectively. The percentage of infected cells was >80%, and
cell morphology was normal 3 days post-transfection in both groups
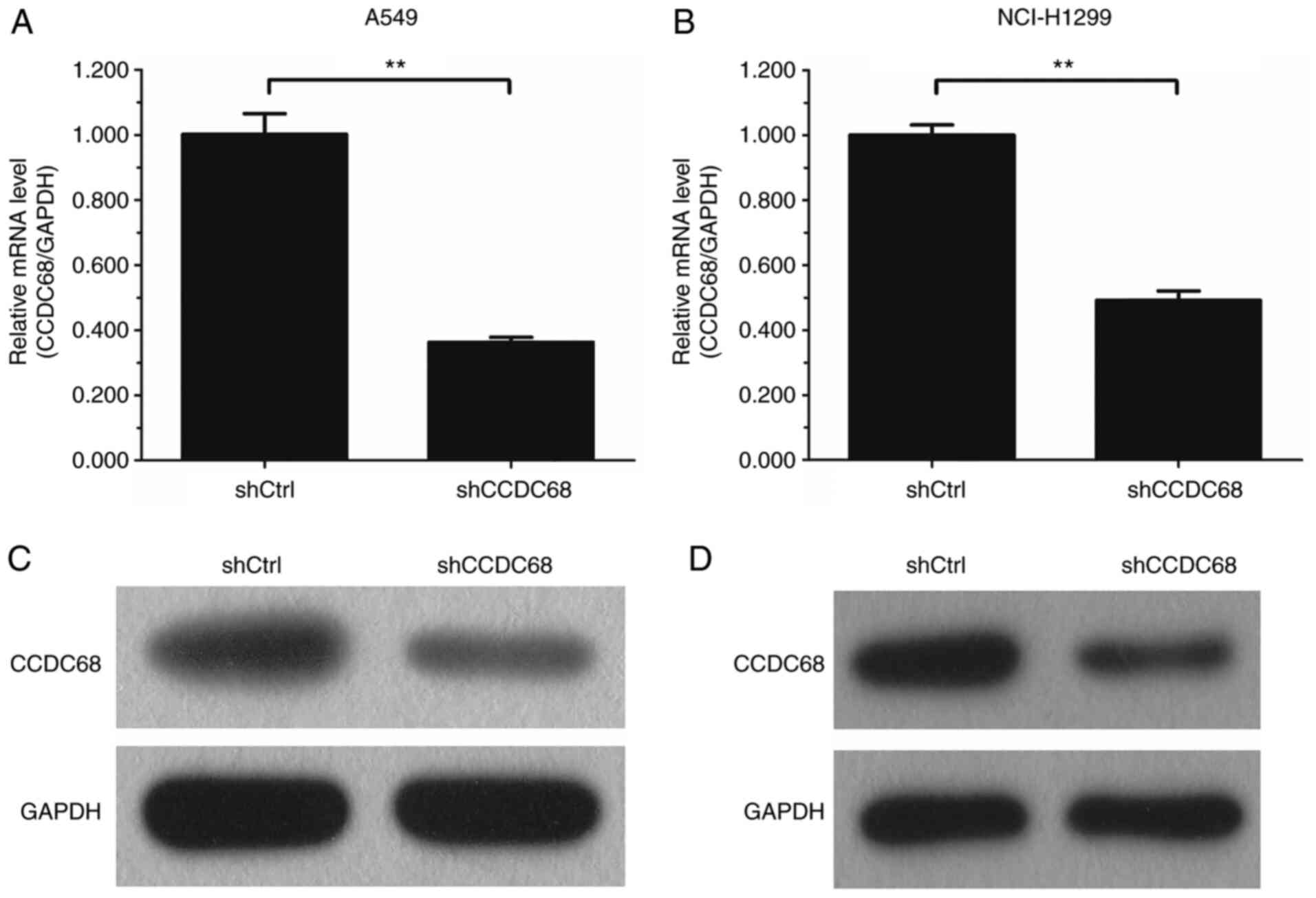
(Fig. 2). To further verify the
efficiency of CCDC68 knockdown, the protein and mRNA expression
levels of CCDC68 were assessed in transfected cells. The results
demonstrated that compared with that in the shCtrl group, CCDC68
expression was decreased in A549 and H1299 cells at the mRNA and
protein level following transfection with shCCDC68 (Fig. 3)
Knockdown of CCDC68 reduces cell
proliferation
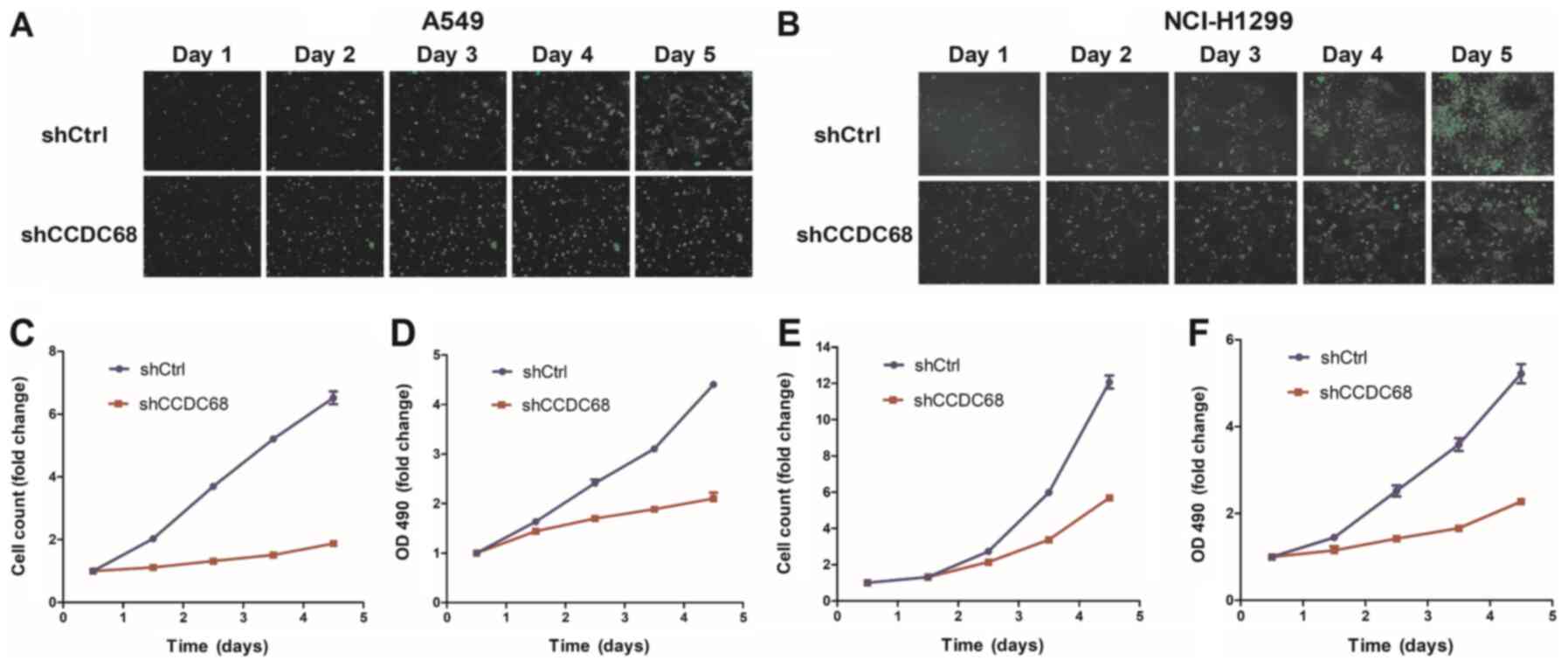
The effects of CCDC68 on NSCLC cell proliferation
was determined using a Celigo Imaging Cytometer and MTT assay. The
results demonstrated that over 5 days, the proliferation of cells
observed with a fluorescence microscope in the shCtrl group was
higher compared with that of cells in the shCCDC68 group in A549
and H1299 cells (Fig. 4A and B),
which was consistent with the results observed for the cell
viability curve (Fig. 4C and E). The
MTT viability assay demonstrated that the viability of A549 and
H1299 cells in the shCCDC68 group was significantly lower compared
with that in the respective shCtrl groups (Fig. 4D and F). Therefore, CCDC68 may be
associated with the proliferative capacity of NSCLC cells.
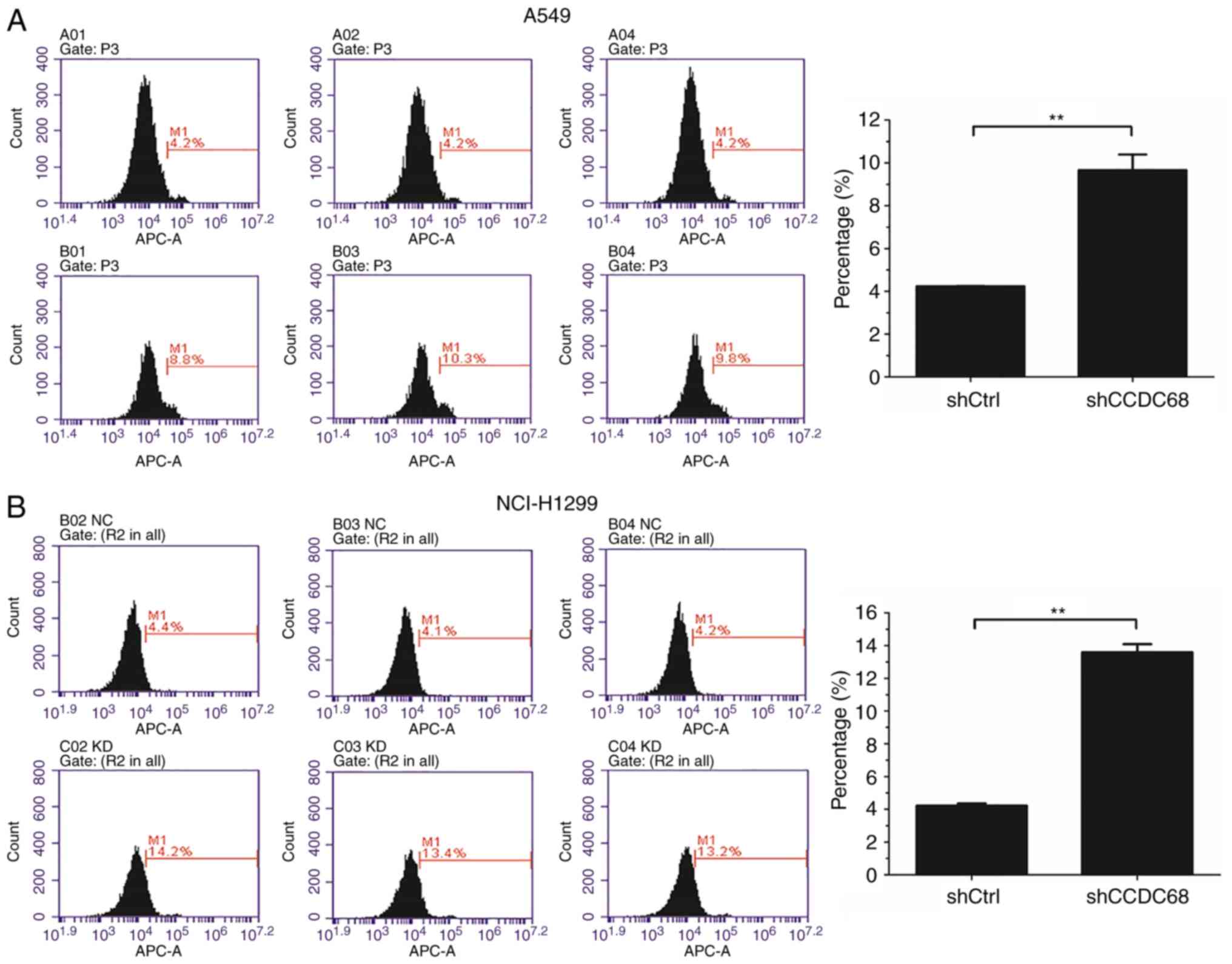
Effects of CCDC68 on apoptosis
Flow cytometry was used to analyze the effects of
CCDC68 on apoptosis in NSCLC cells following CCDC68 knockdown. At 3
days post-shRNA lentiviral infection, the proportion of apoptotic
cells was higher in the CCDC68-knockdown A549 and H1299 cells
compared with that in the respective shCtrl groups (Fig. 5). These results demonstrated that
CCDC68 knockdown induced apoptosis in NSCLC cells.
Discussion
During the past two decades, there has been a focus
on identifying potential biomarkers for the diagnosis and treatment
of malignant tumors, such as DNA methylation (15), growth factors and protein kinases
(16). Research on lung cancer is
more prominent (17), and specific
driver mutations, such as those in epidermic growth factor
receptor, anaplastic lymphoma kinase and v-ros avian UR2 sarcoma
virus oncogene homolog 1, can be used to identify patients that are
sensitive to certain inhibitors, such as gefitinib, crizotinib, and
others (18–20). Tyrosine kinase inhibitors (TKIs) have
significantly improved overall response rates and progression-free
survival compared with standard chemotherapeutic regimens for
subgroups of patients carrying specific gene mutations; however,
TKIs do not improve overall survival in all patients with NSCLC
(21). Therefore, identifying novel
candidate targets and exploring their underlying mechanisms may
result in the identification of more suitable targets.
All proteins of the CCDC family contain coiled-coil
structures and this motif exerts a series of biological functions,
such as cell division, regulation of gene expression, membrane
fusion and drug delivery or extrusion (22,23).
Several studies have demonstrated that CCDC proteins are abnormally
expressed in various types of tumors and are associated with tumor
migration, invasion and metastasis (24,25);
CCDC67 and CCDC8 may serve as novel potential therapeutic targets
or biomarkers for the diagnosis and prognosis of specific malignant
tumors (7,8), and CCDC34 can function as both,
therapeutic targets and biomarkers (9,10).
Previous studies have explored the role of CCDC proteins in lung
cancer; for example, low levels of CCDC19 are associated with an
unfavorable outcome in patients with lung squamous cell carcinoma
(26) and with NSCLC pathogenesis
(27). CCDC6 may represent a
predictive biomarker of resistance to conventional single-mode
therapy and yield insight on tumor sensitivity to poly(ADP-ribose)
polymerase (PARP) inhibitors in NSCLC (28). CCDC106 is highly expressed in NSCLC
tissues and cell lines, and its expression levels are associated
with an advanced TNM stage, lymph node metastasis and poor overall
survival in patients with NSCLC (29). As a member of the CCDC family
proteins, CCDC68, also termed cutaneous T cell lymphoid antigen
se57-1, has a molecular weight for 39 kDa and is located on
chromosome 18q21.2 (12). Studies on
CCDC68 in malignant tumors are rare, it has previously been
demonstrated in colorectal adenocarcinoma and PDAC as a
tumor-suppressive gene (11,12). However, the role of CCDC68 in lung
cancer has not been extensively studied.
The present exploratory study found that CCDC68 was
highly expressed in lung cancer tissues compared with normal lung
tissues, which appeared to be inconsistent with the effects of
CCDC68 in other malignant tumors. To clarify the role of CCDC68 in
lung cancer, the present study preliminarily explored the
biological functions of CCDC68 in NSCLC cell lines. The results of
the in vitro experiments demonstrated that CCDC68 was
expressed in NSCLC cell lines. The high expression levels of CCDC68
highlighted its potential involvement in the proliferation of NSCLC
cells. RNAi technology is widely used for gene silencing to explore
protein function, and for the treatment of malignant tumors in
in vitro and in vivo models of various types of
cancer (30,31). In the present study, the expression
of CCDC68 was knocked down in A549 and H1299 cells, and the effects
of CCDC68 knockdown on cell proliferation and apoptosis were
assessed. The results demonstrated that CCDC68 knockdown reduced
the proliferation and increased apoptosis in A549 and H1299 cells.
These results suggested that CCDC68 increased tumorigenesis in
vitro in NSCLC cells. This result contradicts previous studies,
which suggested that CCDC68 may function as a tumor suppressor. For
example, in patients with colorectal cancer, the CCDC68 copy number
was decreased and its expression was downregulated compared with
normal tissues (11). Additionally,
~60% of PDAC cases exhibited lost/reduced CCDC68 protein expression
compared with normal pancreas specimens of healthy patients. CCDC68
was expressed significantly in normal duct epithelium and
well-differentiated PDAC; thus, CCDC68 serves as a tumor suppressor
in PDAC (12). Studies on the
effects and mechanisms of CCDC68 in malignant tumors are relatively
lacking, and these results should be further verified.
CCDCs have been demonstrated to serve different
roles in malignant tumors, and researchers have tried to explore
their underlying mechanisms, including those in lung cancer. In
lung cancer, CCDC106 enhances the expression of the cell
cycle-regulating proteins cyclin A2 and cyclin B1 and promotes A549
and H1299 cell proliferation, which is dependent on the AKT
signaling pathway (29). In breast
and cervical cancer, protein kinase CK2-mediated CCDC106
phosphorylation is required for p53 degradation; the
CK2/CCDC106/p53 signaling axis may be a novel therapeutic target
(32). CCDC19 significantly reduces
the proliferation and cell cycle progression of NSCLC cells in
vitro via the PI3K/AKT/c-Jun signaling pathway (27), and serves as a potential tumor
suppressor by modulating the cell cycle and the MAPK signaling
pathway in nasopharyngeal carcinoma (33). CCDC6 acts as a novel biomarker for
the clinical use of PARP1 inhibitors in malignant pleural
mesothelioma (34) and high-grade
urothelial bladder cancer (35) in
addition to lung cancer (36). CCDC6
fuses with proto-oncogenes, which are associated with the
occurrence and development of various types of malignant tumors,
such as ovarian epithelial tumor, breast cancer, NSCLC and others
(37). Therefore, CCDC6 may serve as
a potential therapeutic target for patients with these tumors
(36). CCDC34 is upregulated in
bladder cancer (9), esophageal
squamous cell carcinoma (ESCC) (10)
and cervical cancer (38) compared
with adjacent normal tissues. Knockdown of CCDC34 expression
significantly reduces the phosphorylation of the MAPK family
proteins and the signal transduction factor AKT (9,39).
Additionally, a positive association between CCDC34 with VEGF and
MVD expression has been identified, highlighting the potential
mechanism by which CCDC34 promotes tumorigenesis (10,38).
Together, the present and previous studies suggest that the
functions of various CCDCs in malignant tumors are based on the
specific CCDC and the specific type of cancer, and the mechanisms
of CCDCs in lung cancer appear to vary. CCDC68 is a novel centriole
subdistal appendage (SDA) component required for hierarchical SDA
assembly in human cells and is involved in centrosome microtubule
anchorin. The functions of CCDC68 in microtubule anchoring are
mainly mediated by CEP170, and loss of CCDC68 affects microtubule
nucleation at the early stage of microtubule regrowth (40). The mechanism of CCDC68 in cancer has
not yet been fully elucidated and since CCDC68 exhibits differing
effects dependent on the type of cancer, the role and mechanism of
CCDC68 in NSCLC requires further investigation.
In conclusion, the present study preliminarily
determined the expression and explored the role of CCDC68 in lung
cancer cell lines. The results demonstrated that CCDC68 was
expressed in human NSCLC cell lines, and in vitro knockdown
of CCDC68 expression reduced the proliferation and increased
apoptosis in A549 and H1299 cells. Future studies should examine
the expression of CCDC68 in lung cancer tissues and its effects in
in vivo animal models of lung cancer. CCDC68 may serve as a
potential candidate biomarker for detection of malignant
transformation of lung cancer. Whether CCDC68 may also serve as a
therapeutic target requires further study.
Acknowledgements
Not applicable.
Funding
This study was supported by Xi'an Chest Hospital and
the Xi'an Science and Technology Bureau's ‘Science and Technology
+’ Action Plan-Medical Research Project (grant no.
201805093YX1SF27).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JWL, TH and JD conceived and designed the study.
JWL, JD and ZJL performed the experiments. YF and JLX analyzed
data. JWL and TH wrote and edited the manuscript. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Balata H, Fong KM, Hendriks LE, Lam S,
Ostroff JS, Peled N, Wu N and Aggarwal C: Prevention and early
detection for NSCLC: Advances in Thoracic Oncology 2018. J Thorac
Oncol. 14:1513–1527. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rodriguez-Lara V, Hernandez-Martinez JM
and Arrieta O: Influence of estrogen in non-small cell lung cancer
and its clinical implications. J Thorac Dis. 10:482–497. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schiller JH, Harrington D, Belani CP,
Langer C, Sandler A, Krook J, Zhu J and Johnson DH; Eastern
Cooperative Oncology Group, : Comparison of four chemotherapy
regimens for advanced non-small-cell lung cancer. N Engl J Med.
346:92–98. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li HD and Liu SL: Molecular targeted
therapy for non-small cell lung cancer: The reality in China and
coping strategy. Prac J Med Pharm. 35:373–379. 2018.
|
|
6
|
Burkhard P and Stetefeld J: Coiled coils:
A highly versatile protein folding motif. Trends Cell Biol.
11:82–88. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yin DT, Xu J, Lei M, Li H, Wang Y, Liu Z,
Zhou Y and Xing M: Characterization of the novel tumor-suppressor
gene CCDC67 in papillary thyroid carcinoma. Oncotarget.
7:5830–5841. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pangeni RP, Channathodiyil P, Huen DS,
Eagles LW, Johal BK, Pasha D, Hadjistephanou N, Nevell O, Davies
CL, Adewumi AI, et al: The GALNT9, BNC1 and CCDC8 genes are
frequently epigenetically dysregulated in breast tumours that
metastasise to the brain. Clin Epigenetics. 7:572015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gong Y, Qiu W, Ning X, Yang X, Liu L, Wang
Z, Lin J, Li X and Guo Y: CCDC34 is up-regulated in bladder cancer
and regulates bladder cancer cell proliferation, apoptosis and
migration. Oncotarget. 6:25856–25867. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hu DD, Li PC, He YF, Jia W and Hu B:
Overexpression of coiled-coil domain-containing protein 34 (CCDC34)
and its correlation with angiogenesis in esophageal squamous cell
carcinoma. Med Sci Monit. 24:698–705. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sheffer M, Bacolod MD, Zuk O, Giardina SF,
Pincas H, Barany F, Paty PB, Gerald WL, Notterman DA and Domany E:
Association of survival and disease progression with chromosomal
instability: A genomic exploration of colorectal cancer. Proc Natl
Acad Sci USA. 106:7131–7136. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Radulovich N, Leung L, Ibrahimov E, Navab
R, Sakashita S, Zhu CQ, Kaufman E, Lockwood WW, Thu KL, Fedyshyn Y,
et al: Coiled-coil domain containing 68 (CCDC68) demonstrates a
tumor-suppressive role in pancreatic ductal adenocarcinoma.
Oncogene. 34:4238–4247. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gazdar AF, Girard L, Lockwood WW, Lam WL
and Minna JD: Lung cancer cell lines as tools for biomedical
discovery and research. J Natl Cancer Inst. 102:1310–1321. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pan Y, Liu G, Zhou F, Su B and Li Y: DNA
methylation profiles in cancer diagnosis and therapeutics. Clin Exp
Med. 18:1–14. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Suurmeijer AJH, Kao YC and Antonescu CR:
New advances in the molecular classification of pediatric
mesenchymal tumors. Genes Chromosomes Cancer. 58:100–110. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kalemkerian GP, Narula N, Kennedy EB,
Biermann WA, Donington J, Leighl NB, Lew M, Pantelas J, Ramalingam
SS, Reck M, et al: Molecular testing guideline for the selection of
patients with lung cancer for treatment with targeted tyrosine
kinase inhibitors: American Society of Clinical Oncology
endorsement of the College of American Pathologists/International
Association for the Study of Lung Cancer/Association for Molecular
Pathology clinical practice guideline update. J Clin Oncol.
36:911–919. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Roeper J and Griesinger F: Epidermal
growth factor receptor tyrosine kinase inhibitors in advanced
nonsmall cell lung cancer: What is the preferred first-line
therapy? Curr Opin Oncol. 31:1–7. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pirker R and Filipits M: From crizotinib
to lorlatinib: Continuous improvement in precision treatment of
ALK-positive non-small cell lung cancer. ESMO Open. 4:e0005482019.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu S, Yang H, Jang Y, Zhang T, Yan R and
Zhang J: Evolution strategy of ROS1 kinase inhibitors for use in
cancer therapy. Future Med Chem. 10:1705–1720. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rocco D, Della Gravara L and Battiloro C:
The role of combination chemo-immunotherapy in advanced non-small
cell lung cancer. Expert Rev Anticancer Ther. 19:561–568. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mcfarlane AA, Orriss GL and Stetefeld J:
The use of coiled-coil proteins in drug delivery systems. Eur J
Pharmacol. 625:101–107. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Frezzo JA and Montclare JK: Exploring the
potential of engineered coiled-coil protein microfibers in drug
delivery. Ther Deliv. 6:643–646. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhong J, Zhao M, Luo Q, Ma Y, Liu J, Wang
J, Yang M, Yuan X, Sang J and Huang C: CCDC134 is down-regulated in
gastric cancer and its silencing promotes cell migration and
invasion of GES-1 and AGS cells via the MAPK pathway. Mol Cell
Biochem. 372:1–8. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tsolakis AV, Grimelius L and Islam MS:
Expression of the coiled coil domain containing protein 116 in the
pancreatic islets and endocrine pancreatic tumors. Islets.
4:349–353. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang Y, Liu Z, Luo R and Xie Y: Decreased
CCDC19 is correlated with unfavorable outcome in lung squamous cell
carcinoma. Int J Clin Exp Pathol. 11:802–807. 2018.PubMed/NCBI
|
|
27
|
Liu Z, Mai C, Yang H, Zhen Y, Yu X, Hua S,
Wu Q, Jiang Q, Zhang Y, Song X and Fang W: Candidate tumour
suppressor CCDC19 regulates miR-184 direct targeting of C-Myc
thereby suppressing cell growth in non-small cell lung cancers. J
Cell Mol Med. 18:1667–1679. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cerrato A, Morra F, Di Domenico I and
Celetti A: NSCLC mutated isoforms of CCDC6 affect the intracellular
distribution of the wild type protein promoting cisplatinum
resistance and PARP inhibitors sensitivity in lung cancer cells.
Cancers (Basel). 12:442019. View Article : Google Scholar
|
|
29
|
Zhang X, Zheng Q, Wang C, Zhou H, Jiang G,
Miao Y, Zhang Y, Liu Y, Li Q, Qiu X and Wang E: CCDC106 promotes
non-small cell lung cancer cell proliferation. Oncotarget.
8:26662–26670. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Dana H, Chalbatani GM, Mahmoodzadeh H,
Karimloo R, Rezaiean O, Moradzadeh A, Mehmandoost N, Moazzen F,
Mazraeh A, Marmari V, et al: Molecular mechanisms and biological
functions of siRNA. Int J Biomed Sci. 13:48–57. 2017.PubMed/NCBI
|
|
31
|
Jain S, Pathak K and Vaidya A: Molecular
therapy using siRNA: Recent trends and advances of multi target
inhibition of cancer growth. Int J Biol Macromol. 116:880–892.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ning Y, Wang C, Liu X, Du Y, Liu S, Liu K,
Zhou J and Zhou C: CK2-mediated CCDC106 phosphorylation is required
for p53 degradation in cancer progression. J Exp Clin Cancer Res.
38:1312019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu Z, Li X, He X, Jiang Q, Xie S, Yu X,
Zhen Y, Xiao G, Yao K and Fang W: Decreased expression of updated
NESG1 in nasopharyngeal carcinoma: Its potential role and
preliminarily functional mechanism. Int J Cancer. 128:2562–2571.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Morra F, Merolla F, D'Abbiero D, Ilardi G,
Campione S, Monaco R, Guggino G, Ambrosio F, Staibano S, Cerrato A,
et al: Analysis of CCDC6 as a novel biomarker for the clinical use
of PARP1 inhibitors in malignant pleural mesothelioma. Lung Cancer.
135:56–65. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Morra F, Merolla F, Criscuolo D, Insabato
L, Giannella R, Ilardi G, Cerrato A, Visconti R, Staibano S and
Celetti A: CCDC6 and USP7 expression levels suggest novel treatment
options in high-grade urothelial bladder cancer. J Exp Clin Cancer
Res. 8:902019. View Article : Google Scholar
|
|
36
|
Cerrato A, Visconti R and Celetti A: The
rationale for druggability of CCDC6- tyrosine kinase fusions in
lung cancer. Mol Cancer. 17:462018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Laxmi A, Gupta P and Gupta J: CCDC6, a
gene product in fusion with different protoncogenes, as a potential
chemotherapeutic target. Cancer Biomark. 24:383–393. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu LB, Huang J, Zhong JP, Ye GL, Xue L,
Zhou MH, Huang G and Li SJ: High expression of CCDC34 is associated
with poor survival in cervical cancer patients. Med Sci Monit.
24:8383–8390. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lei YY, Wang WJ, Mei JH and Wang CL:
Mitogen-activated protein kinase signal transduction in solid
tumors. Asian Pac J Cancer Prev. 15:8539–8548. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Huang N, Xia Y, Zhang D, Wang S, Bao Y, He
R, Teng J and Chen J: Hierarchical assembly of centriole subdistal
appendages via centrosome binding proteins CCDC120 and CCDC68. Nat
Commun. 8:150572017. View Article : Google Scholar : PubMed/NCBI
|