Introduction
Osteosarcoma is a common type of primary bone
cancer, with an incidence of 4–5 per million individuals and a
60–70% 5-year survival rate (1). In
recent years, increasing research has been conducted to develop
clinical therapeutic strategies for osteosarcoma, including
chemotherapy, radiotherapy and surgery; however, the survival rate
of patients with osteosarcoma remains low (2). Therefore, identifying the molecular
mechanisms underlying the occurrence and progression of
osteosarcoma is important for the development of effective
therapeutic agents.
MicroRNAs (miRNAs/miRs) are small, endogenous
non-coding RNAs that contain 20–25 nucleotides. By
post-transcriptionally regulating the silencing of genes in the
3′-untranslated region (3′-UTR), miRNAs are involved in biological
processes, including tumor occurrence, growth and progression
(3). miR-216 is downregulated in
numerous types of tumors, which suggests that miR-216 may serve as
a promising prognostic indicator (4,5). By
knocking down syndecan binding protein in breast cancer or Cyclin
T2 in gastric cancer, miR-216 effectively inhibits cancer cell
proliferation and migration (6,7). By
targeting FoxM1, miR-216 suppresses proliferation, migration and
invasion in osteosarcoma cells, as well as in liver and cervical
cancer cells (8–10). In addition, miR-216 is closely linked
to improved outcomes in patients with cervical cancer (10).
Thiostrepton (TST) is a natural cyclic oligopeptide
antibiotic of the thiopeptide class, which is derived from
streptomyces (11). TST was first
studied in breast cancer as a FoxM1 and proteasome inhibitor in
2008 (12). In vivo and in
vitro evidence has demonstrated the significant anticancer
effects of TST (11). By targeting
FoxM1 protein and the proteasome, TST induces cell apoptosis in
ovarian and lung cancer, as well as in other types of tumor cells
(13–15). As a novel anticancer target, TST has
been extensively studied in tumor research (11). Additionally, by targeting FoxM1, TST
is involved in the progression of Ewing's sarcoma and
synoviosarcoma (16–18). The present study investigated FoxM1
as a common target of TST and miR-216b, and the co-influences of
TST and miR-216b on osteosarcoma cell behaviors.
Materials and methods
Cell culture
Human osteosarcoma cell lines (U2OS, MG63 and
Saos-2) and the human osteoblast cell line (hFOB1.19) were
purchased from The Cell Bank of Type Culture Collection of the
Chinese Academy of Sciences. Cells were cultured in Dulbecco's
Modified Eagle's medium (Gibco, Thermo Fisher Scientific, Inc.)
supplemented with 10% fetal bovine serum (Gibco, Thermo Fisher
Scientific, Inc.), 100 U/ml penicillin and 100 µg/ml streptomycin
with 5% CO2 at 37°C. Cells in the logarithmic growth
phase were used for subsequent experiments.
Cell transfection
Cells were seeded (5×105 cells/well) into
6-well plates. Subsequently, cells were transfected with miR-216b
mimic (Shanghai GenePharma Co., Ltd.) or mimic-negative control
(NC) (Shanghai GenePharma Co., Ltd.). The miR-216b mimic sequence
was 5′-AAAUCUCUGCAGGCAAAUGUGA-3′, and the mimic-NC sequence was
5′-UUCUCCGAACGUGUCACGUTT-3′. miR-216b mimic or mimic-NC were
transfected at a final concentration of 50 nM using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.). Following culture for 6 h at 37°C, the medium
was replaced. At 48 h post-transfection, cells were used for
subsequent experiments.
RNA extraction and reverse
transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from cells using
TRIzol® (Thermo Fisher Scientific, Inc.). Total RNA was
reverse transcribed into cDNA using the M-MLV reverse transcription
kit (cat. no. RR047A; Takara Bio, Inc.). Relative miR-216b
expression was determined using the miRNA Real-Time PCR Assay kit
(Aidlab Biotechnologies, Ltd.). Relative FoxM1 expression was
determined using SYBR Premix Ex Taq™ II with Tli RNaseH (cat. no.
RR820A, Takara Bio, Inc.) and an ABI PRISM 7500 system (Thermo
Fisher Scientific, Inc.). The following primers were used for qPCR:
miR-216b forward, 5′-AAAUCUCUGCAGGCAAAUGUGA-3′ and reverse,
5′-ACAUUUGCCUCCAGAGAUUUUU-3′; β-actin forward,
5′-AAACTGGAACGGTGAAGGTG-3′ and reverse, 5′-AGTGGGGTGGCTTTTAGGAT-3′;
FoxM1 forward, 5′-TGCCCAGCAGTCTCTTACCT-3′ and reverse,
5′-CTACCCACCTTCTGGCAGTC-3′; and U6 forward, 5′-CTCGCTTCGGCAGCACA-3′
and reverse, 5′-AACGCTTCACGAATTTGCGT-3′. miRNA and mRNA expression
levels were quantified using the 2−ΔΔCq method (19) and normalized to the internal
reference genes U6 and β-actin, respectively.
MTT assay
The MTT assay was performed to assess cell
cytotoxicity. Transfected cells were seeded (3×103
cells/well) into 96-well plates and cultured at 37°C overnight.
Subsequently, cells were treated with different concentrations (1,
2, 3 and 5 µM) of TST (cat. no. MB13332; Meilunbio) at 37°C for 48
h. Cells were incubated with 5 mg/ml MTT solution at 37°C for 4 h.
Subsequently, DMSO was added to dissolve the formazan crystals. The
optical density (OD) of each well was measured at a wavelength of
490 nm. Cell cytotoxicity (%) = (average OD490 of the
control group-average OD490 of the experimental
group)/(average OD490 of the control group-average
OD490 of the blank group) ×100.
Western blotting
Cells were washed twice in cold PBS and total
protein was extracted using RIPA lysate (cat. no. P0013C; Beyotime
Institute of Biotechnology) containing protease inhibitor (Merck
KGaA). Total protein was quantified using the bicinchoninic acid
protein assay kit (cat. no. P0012S; Beyotime Institute of
Biotechnology). Proteins (40 µg) were separated via 10% SDS-PAGE
and transferred onto PVDF membranes, which were blocked in 5% skim
milk at room temperature for 1 h. The membranes were incubated the
following primary antibodies at 4°C: Anti-FoxM1 (cat. no. A2493;
ABclonal Biotech Co., Ltd.), anti-Bax (cat. no. A0207; ABclonal
Biotech Co., Ltd.), anti-Bcl-2 (cat. no. 2870; Cell Signaling
Technology, Inc.) and anti-tubulin (cat. no. 2144; Cell Signaling
Technology). Subsequently, the membranes were incubated with a
horseradish peroxidase-conjugated secondary antibody (cat. no.
A0208; Beyotime Institute of Biotechnology) at room temperature for
1 h. Protein bands were visualized using ECL plus reagent (Thermo
Fisher Scientific, Inc.) and a Chemo XRS+ luminometer (Bio-Rad
Laboratories). Protein expression levels were quantified using
Quantity One software (Bio-Rad Laboratories) with tubulin as the
loading control.
Renilla luciferase activity
The binding sites between FoxM1 and miR-216b were
predicted using TargetScan software (version 7.2.0; www.TargetScan.org/vert_72). The 1.8 kb binding
sequences in the 3′UTR of FoxM1 were cloned into the pmiR-GLO
vector (Promega Corporation) to construct FoxM1 3′-UTR wild-type
(wt). Using the GeneTailer site-directed mutagenesis kit
(Invitrogen; Thermo Fisher Scientific, Inc.), binding sequences
were mutated to generate FoxM1 3′-UTR mutant (mut). U2OS and MG63
cells were seeded into 96-well plates until the confluence reached
70%, and then the cells were co-transfected by
Lipofectamine® 2000 with FoxM1 3′-UTR wt or FoxM1 3′-UTR
mut and miR-216b mimic or mimic-NC according to the protocol.
Following transfection for 48 h, relative luciferase activity was
measured using the Dual-Luciferase Reporter assay kit (Promega
Corporation).
Apoptosis analysis (early and late
apoptosis)
At 48 h post-transfection/TST treatment, cells were
washed twice with cold PBS. Cell apoptosis was detected using the
Annexin APC/7AAD kit (Nanjing KeyGen Biotech Co., Ltd.) according
to the manufacturer's protocol. Annexin V-positive cells were
defined as apoptotic cells. Apoptotic cells were analyzing using a
FACSCalibur flow cytometer (BD Biosciences) and CellQuest Pro
software (version 5.1; BD Biosciences).
Statistical analysis
Statistical analyses were performed using SPSS
software (version 17.0; IBM Corp.). Data are expressed as the mean
± standard deviation. All data conformed to normal distribution.
Comparisons among multiple groups were analyzed using one-way ANOVA
followed by Tukey's or Bonferroni's post hoc test. P<0.05 was
considered to indicate a statistically significant difference. All
experiments were performed in triplicate and repeated three
times.
Results
Opposite expression patterns of FoxM1
and miR-216b in osteosarcoma cells
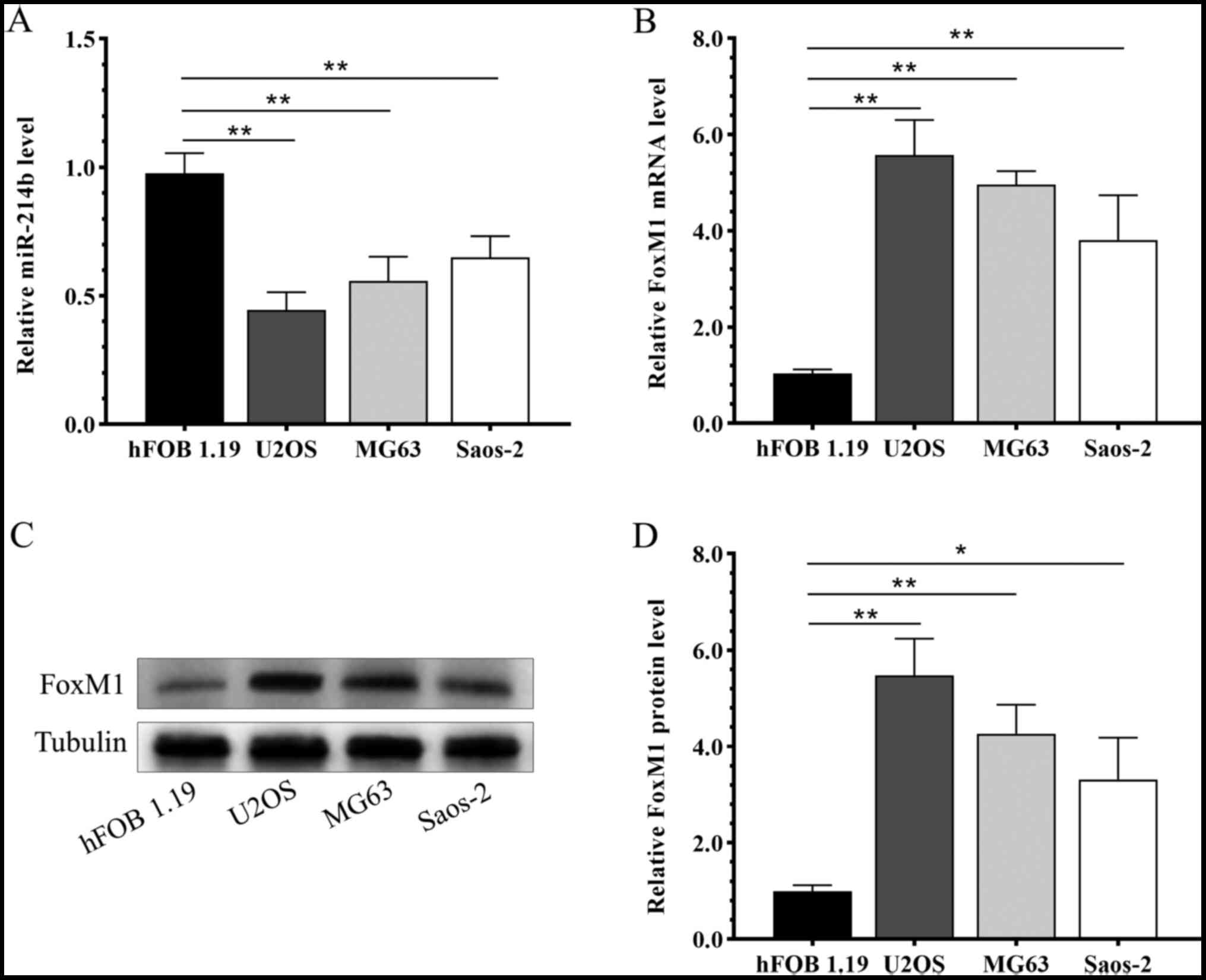
Compared with the human osteoblast cell line
hFOB1.19, miR-216b was significantly downregulated in osteosarcoma
cell lines U2OS, MG63 and Saos-2 (Fig.
1A). By contrast, the mRNA (Fig.
1B) and protein (Fig. 1C and D)
expression levels of FoxM1 were significantly upregulated in the
osteosarcoma cell lines U2OS, MG63 and Saos-2 compared with the
hFOB1.19 cell line.
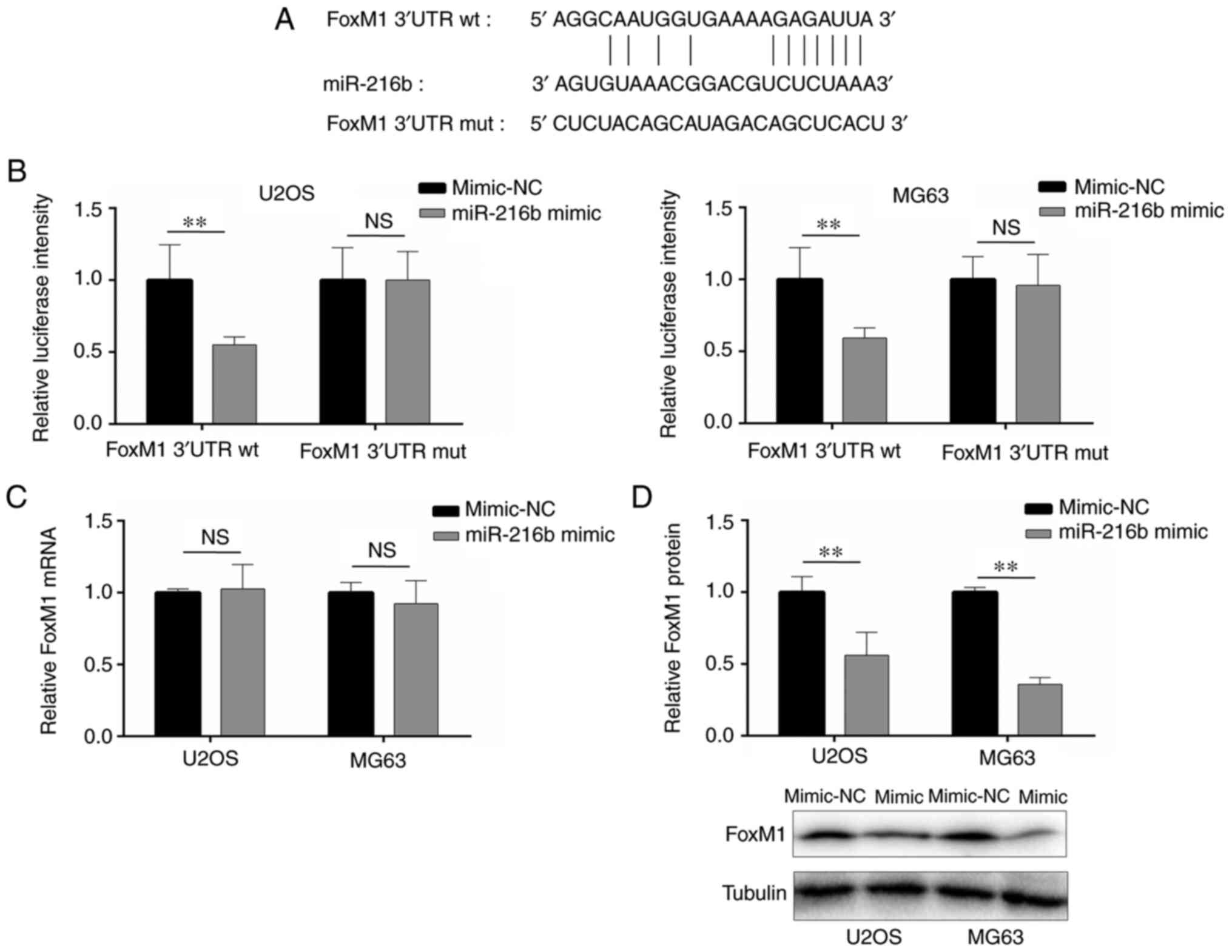
miR-216b targets the 3′-UTR of
FoxM1
Based on a previous report (6) and online prediction, the results
indicated that miR-216b bound to the 3′-UTR of FoxM1 (Fig. 2A). Compared with the mimic-NC group,
miR-216b overexpression significantly decreased the luciferase
activity of FoxM1 3′-UTR wt, whereas miR-216b overexpression did
not significantly alter the luciferase activity of FoxM1 3′-UTR mut
(Fig. 2B), indicating that miR-216b
bound to the 3′-UTR of FoxM1. Additionally, the regulatory effects
of miR-216b on FoxM1 expression were examined via RT-qPCR and
western blotting. FoxM1 mRNA expression levels were not
significantly altered by miR-216b overexpression compared with the
mimic-NC group (Fig. 2C). However,
miR-216b overexpression significantly decreased FoxM1 protein
expression levels in U2OS and MG63 cells compared with the mimic-NC
group (Fig. 2D).
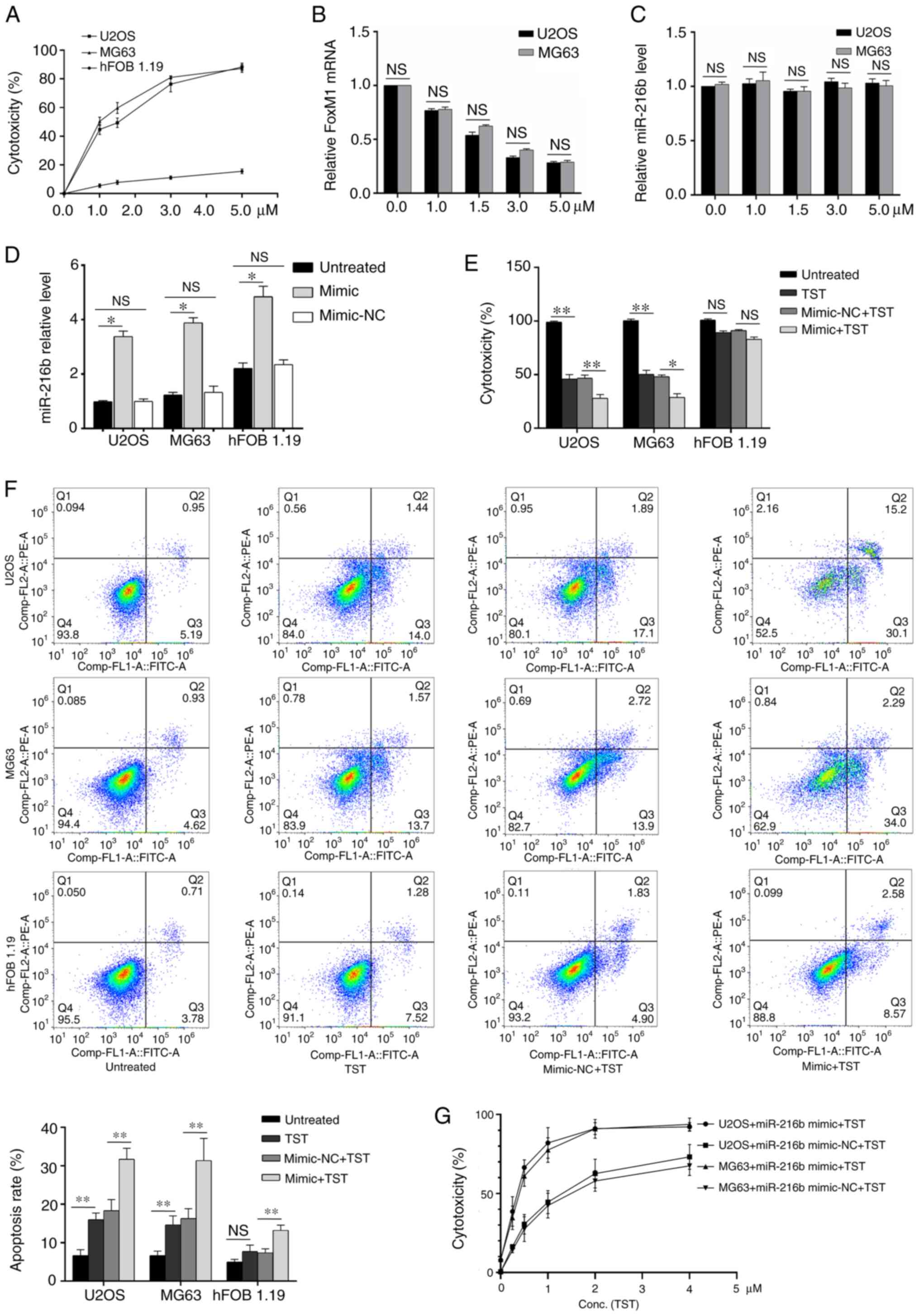
Effects of TST and miR-216b on
osteosarcoma cell cytotoxicity and apoptosis
TST increased U2OS and MG63 cell cytotoxicity in a
dose-dependent manner (Fig. 3A).
FoxM1 mRNA expression levels were decreased by TST treatment in
U2OS and MG63 cells in a dose-dependent manner compared with the
control group (0 µM TST; Fig. 3B).
By contrast, compared with the control group (0 µM TST), miR-216b
expression levels were not notably altered by TST treatment
(Fig. 3C). miR-216b overexpression
significantly increased miR-126b expression levels in U2OS, MG63
and hFOB1.19 cells compared with the untreated cells (Fig. 3D). Treatment with 1 µM TST
significantly decreased osteosarcoma cell cytotoxicity (Fig. 3E) and significantly increased
osteosarcoma cell apoptosis (Fig.
3F) compared with untreated osteosarcoma cells. miR-216b
overexpression enhanced the regulatory effects of TST on
osteosarcoma cell cytotoxicity and apoptosis (Fig. 3E and F). Subsequently, osteosarcoma
cells were treated with miR-126 mimic and different concentrations
of TST. The results indicated that there was a notable synergistic
effect between miR-216b and TST on osteosarcoma cell cytotoxicity
(Fig. 3G).
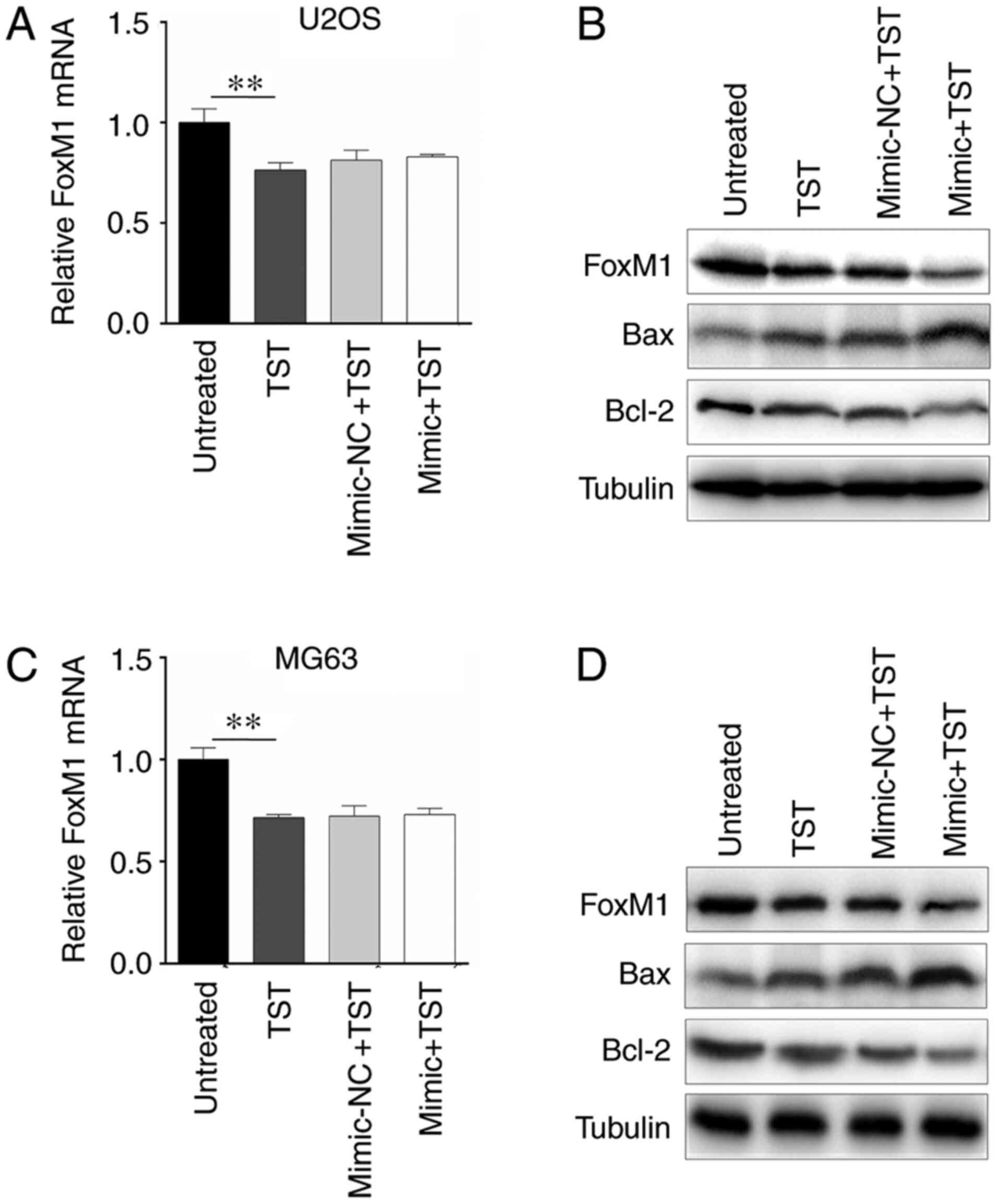
miR-216b and TST co-regulate
apoptosis-associated genes in osteosarcoma cells
The results indicated that 1 µM TST notably
downregulated the mRNA and protein expression levels of FoxM1 in
U2OS cells compared with untreated cells (Fig. 4A and B). However, miR-216b
overexpression only amplified the inhibitory effect of TST on FoxM1
protein expression (Fig. 4B). In
addition, protein expression levels of apoptosis-associated genes
Bax and Bcl-2 were detected. The western blotting results indicated
that TST treatment markedly increased Bax expression and decreased
Bcl-2 expression in U2OS cells compared with untreated cells
(Fig. 4B); however, TST-induced
alterations to Bax and Bcl-2 expression were enhanced by miR-216b
overexpression. Similar results were obtained in MG63 cells
(Fig. 4C and D).
Discussion
As a cancerous bone tumor, osteosarcoma primarily
affects the metaphysis of long bones, especially the regions around
the knee, and it is highly prevalent in children and teenagers
(1). Currently, the therapeutic
efficacy of osteosarcoma treatment strategies is not ideal.
Limb-preserving surgery is widely used for the treatment of
osteosarcoma; however, even with radical amputation, 10% of
patients experience local relapse and 40–50% suffer from lung
metastases (20–22). Moreover, large bone defects caused by
surgery do not heal easily, which seriously affects the limb
function of affected patients (1).
Therefore, understanding the pathogenesis and etiology of
osteosarcoma is important for the development of highly specific
and less toxic targeted drugs. Osteosarcoma progression is
complicated, involving multiple signaling pathways, vital factors,
and regulatory miRNAs and their target genes (23,24).
FoxM1 is part of the forkhead box family of
transcription factors, containing a winged helix in the DNA-binding
region that consists of 100 amino acids (25). FoxM1 expression is upregulated in
numerous different human tumor types, which indicates its vital
involvement in tumor progression (25,26). In
the prognostic landscape of >18,000 genes across 39 human cancer
types, the FoxM1 regulatory network is the main prognostic
indicator for poor prognosis, in cancers such as lung cancer, colon
cancer, liver cancer, head and neck cancer, brain cancer and others
(27). Nevertheless, the exact
mechanisms underlying the anticancer functions of FoxM1 are not
completely understood. One potential theory suggested that FoxM1
transcription activation enhances tumorigenicity, including
promoting tumor cell proliferation (28). Another theory proposed that FoxM1
supports different carcinogenetic signaling pathways, including the
Wnt, SMAD3 and NF-κB signaling pathways, by interacting with other
proteins (29). A relevant study
indicated that FoxM1 is upregulated in osteosarcoma cells and
triggers cell proliferation, migration and invasion (8). FoxM1 knockdown via RNA interference
induces tumor cell proliferation and anchors independent growth
(30). Meanwhile, FoxM1 silencing
can inhibit tumor cell migration, invasion and tumorigenesis
(28). Based on the aforementioned
studies, it has been suggested that FoxM1 may serve as a novel
osteosarcoma drug target. The present study indicated that FoxM1
expression was upregulated in osteosarcoma cell lines compared with
the human osteoblast cell line hFOB1.19.
The diagnostic and prognostic potential of miRNAs
for tumors has recently been reported, with a number of miRNAs
being used a tumor biomarkers and targeted drugs (31). Moreover, miRNAs are involved in
osteosarcoma-associated signaling pathway regulation, which
provides a novel strategy for identifying the molecular mechanisms
underlying osteosarcoma (32). The
miRNAs and drug resistance relationships in osteosarcoma have been
identified, which further broadens the knowledge of the structure
and functions of miRNAs in the disease (33). It has been predicted that miRNAs will
reverse chemotherapy resistance in future analyses (34). miR-216b is a member of the miR-216
cluster and is located on human chromosome 2p16.1 (35). A previous study demonstrated the
close relationship between miR-216b and different types of human
cancer such as breast cancer, cervical cancer and colorectal cancer
(35). miR-216b inhibits
proliferation and promotes apoptosis in pancreatic cancer and clear
cell renal cell carcinoma by downregulating KRAS (36,37). In
colorectal cancer, miR-216b suppresses tumors by activating the
JAK2/STAT3 signaling pathway (38).
miR-216b is downregulated in non-small cell lung cancer (NSCLC)
tissues and cell lines. Multivariate Cox regression analysis
indicated that miR-216b is an independent prognostic factor for
patients with NSCLC (39). By
targeting FoxM1 and SOX9, miR-216b regulates NSCLC proliferation
and invasion (39,40). miR-216b is also downregulated in
breast cancer samples, which suppresses breast cancer cell
proliferation by targeting the purinergic receptor P2X7 (41). A recent study reported that the
miR-216b/FoxM1 axis inhibits osteosarcoma proliferation, migration
and metastasis (8). In the present
study, miR-216b was downregulated in osteosarcoma cell lines
compared with the hFOB1.19 cell line, and the results indicated
that miR-216b regulated FoxM1 protein expression by binding to the
3′UTR.
TST has been widely applied in the clinic as a
natural cyclic oligopeptide antibiotic of the thiopeptide class,
which is derived from streptomyces (11). TST exerts a specific targeted
inhibitory effect on FoxM1, but whether or not FoxM1 can be
downregulated by TST poses little impact on the activity of other
transcription factors (42–44). Previous studies have demonstrated
that TST can inhibit proliferation and induce apoptosis in
different types of malignant tumors by downregulating FoxM1,
thereby indicating that TST displays strong anticancer activity
(45,46) TST has a limited toxic effect on
normal cells, and in vivo experiments also verified that TST
treatment at an effective anticancer concentration presents few
toxic effects (46–48). The combined treatment of TST,
cisplatin and Bortezomib significantly improves the therapeutic
sensitivity of breast cancer (49,50).
Therefore, the aforementioned studies indicated that TST may serve
as a potential anticancer treatment by targeting FoxM1. Previous
studies have proposed that TST, as a FoxM1 inhibitor, may serve as
a novel therapeutic agent for Ewing's sarcoma (17,18). The
results of the present study indicated that TST reduced
osteosarcoma cell cytotoxicity and negatively regulated FoxM1 mRNA
expression levels.
In the present study, since FoxM1 is the common
target of TST and miR-216b, osteosarcoma cells were treated with
different TST concentrations and subsequently, miR-216b expression
levels were detected. miR-216b expression levels were not
significantly altered by TST compared with the control group. The
present study also indicated that 1 µM TST displayed an inhibitory
effect on osteosarcoma cytotoxicity. Subsequently, miR-216b mimic-
or mimic-NC-transfected osteosarcoma cells were treated with 1 µM
TST. Cytotoxicity was decreased by TST treatment in
miR-216b-overexpression osteosarcoma cells compared with the
control group. The combination of miR-216b mimic and TST indicated
that there was a notable synergistic effect between miR-216b and
TST. Furthermore, TST significantly decreased FoxM1 mRNA expression
levels, which were not altered by miR-216b overexpression, compared
with the control group. However, miR-216b synergistically
downregulated FoxM1 protein expression levels in TST-treated
osteosarcoma cells. It has been suggested that TST and miR-216b
independently target FoxM1, which combined with the results of the
present study indicated that TST and miR-216b may display
synergistic effects on osteosarcoma progression via FoxM1.
Jin et al (51) reported that TST treatment
downregulated FoxM1, matrix metallopeptidase 9 and Bcl-2 expression
in a dose-dependent manner in endometriosis rat samples. By
downregulating cyclin D1 and cyclin E1, TST stops cell cycle
progression at the early S phase; moreover, TST suppresses
laryngeal squamous cell carcinoma (LSCC) DNA synthesis in a dose-
and time-dependent manner (52).
Similarly, TST induces LSCC apoptosis in a dose- and time-dependent
manner via cytochrome C release, Bcl-2 downregulation, and
upregulation of Bax, p53, cleaved Caspase-9, cleaved Caspase-3 and
cleaved poly(ADP-ribose) polymerase 1 (52). The present study assessed cell
apoptosis by detecting Bax and Bcl-2 expression levels. miR-216b
overexpression combined with TST treatment increased Bax expression
and decreased Bcl-2 expression compared with the control group,
thereby promoting osteosarcoma cell apoptosis.
Collectively, to the best of our knowledge, the
present study indicated for the first time that miR-216b was
downregulated in osteosarcoma cells compared with hFOB1.19 cells,
and its expression was not altered by TST treatment. Moreover, the
results indicated that TST and miR-216b targeted FoxM1,
synergistically inhibiting cytotoxicity and stimulating apoptosis
in osteosarcoma cells. Therefore, the combined used of TST and
miR-216b may serve as a promising therapeutic strategy for
osteosarcoma.
Acknowledgements
Not applicable.
Funding
The present study was supported by Shanghai
Municipal Health Commission (grant no. 201740160) and the Chongming
District of Shanghai Science and Technology Project (grant no.
CKY2017-18).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XC and JG substantially contributed to the
conception and designed the work. WX and JS mainly completed the
acquisition, analysis and interpretation of data for the work. HL,
YL and XL were also involved in the acquisition, analysis and
interpretation of data for the work. HL and YL drafted the work and
revised it critically for important intellectual content. XL agreed
to be accountable for the work in ensuring that questions related
to the integrity of any part of the work are appropriately
investigated and resolved. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wu J, Sun H, Li J, Guo Y, Zhang K, Lang C,
Zou C and Ma H: Increased survival of patients aged 0–29 years with
osteosarcoma: A period analysis, 1984–2013. Cancer Med.
7:3652–3661. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hattinger CM, Patrizio MP, Magagnoli F,
Luppi S and Serra M: An update on emerging drugs in osteosarcoma:
Towards tailored therapies? Expert Opin Emerg Drug. 24:153–171.
2019. View Article : Google Scholar
|
|
3
|
Inui M, Martello G and Piccolo S: MicroRNA
control of signal transduction. Nat Rev Mol Cell Biol. 11:252–263.
2010. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li Q, Wang M, Wang N, Wang J, Qi L and Mao
P: Downregulation of microRNA-216b contributes to glioma cell
growth and migration by promoting AEG-1-mediated signaling. Biomed
Pharmacother. 104:420–426. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Azevedo-Pouly AC, Sutaria DS, Jiang J,
Elgamal OA, Amari F, Allard D, Grippo PJ, Coppola V and Schmittgen
TD: miR-216 and miR-217 expression is reduced in transgenic mouse
models of pancreatic adenocarcinoma, knockout of miR-216/miR-217
host gene is embryonic lethal. Funct Integr Genomics. 17:203–212.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jana S, Sengupta S, Biswas S, Chatterjee
A, Roy H and Bhattacharyya A: miR-216b suppresses breast cancer
growth and metastasis by targeting SDCBP. Biochem Biophys Res
Commun. 482:126–133. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen X, Zhang L, Song Q and Chen Z:
MicroRNA-216b regulates cell proliferation, invasion and cycle
progression via interaction with cyclin T2 in gastric cancer.
Anticancer Drugs. 31:623–631. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang W, Guo Z, Yu H and Fan L: miR-216b
inhibits osteosarcoma cell proliferation, migration, and invasion
by targeting Forkhead Box M1. J Cell Biochem. 120:5435–5443. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zheng WW, Zhou J, Zhang CH and Liu XS:
MicroRNA-216b is downregulated in hepatocellular carcinoma and
inhibits HepG2 cell growth by targeting Forkhead box protein M1.
Eur Rev Med Pharmacol Sci. 20:2541–2550. 2016.PubMed/NCBI
|
|
10
|
He S, Liao B, Deng Y, Su C, Tuo J, Liu J,
Yao S and Xu L: miR-216b inhibits cell proliferation by targeting
FOXM1 in cervical cancer cells and is associated with better
prognosis. BMC Cancer. 17:6732017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sandu C, Ngounou Wetie AG, Darie CC and
Steller H: Thiostrepton, a natural compound that triggers heat
shock response and apoptosis in human cancer cells: A proteomics
investigation. Adv Exp Med Biol. 806:443–451. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kwok JM, Myatt SS, Marson CM, Coombes RC,
Constantinidou D and Lam EW: Thiostrepton selectively targets
breast cancer cells through inhibition of forkhead box M1
expression. Mol Cancer Ther. 7:2022–2032. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kongsema M, Wongkhieo S, Khongkow M, Lam
EW, Boonnoy P, Vongsangnak W and Wong-Ekkabut J: Molecular
mechanism of Forkhead box M1 inhibition by thiostrepton in breast
cancer cells. Oncol Rep. 42:953–962. 2019.PubMed/NCBI
|
|
14
|
Huang TH, Wu ATH, Cheng TS, Lin KT, Lai
CJ, Hsieh HW, Chang PM, Wu CW, Huang CF and Chen KY: In silico
identification of thiostrepton as an inhibitor of cancer stem cell
growth and an enhancer for chemotherapy in non-small-cell lung
cancer. J Cell Mol Med. 23:8184–8195. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kalathil D, Prasad M, Chelladurai M, John
S and Nair AS: Thiostrepton degrades mutant p53 by eliciting an
autophagic response in SW480 cells. J Cell Physiol. 233:6938–6950.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Maekawa A, Kohashi K, Kuda M, Iura K,
Ishii T, Endo M, Nakatsura T, Iwamoto Y and Oda Y: Prognostic
significance of FOXM1 expression and antitumor effect of FOXM1
inhibition in synovial sarcomas. BMC Cancer. 16:5112016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Christensen L, Joo J, Lee S, Wai D, Triche
TJ and May WA: FOXM1 is an oncogenic mediator in Ewing Sarcoma.
PLoS One. 8:e545562013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sengupta A, Rahman M, Mateo-Lozano S,
Tirado OM and Notario V: The dual inhibitory effect of thiostrepton
on FoxM1 and EWS/FLI1 provides a novel therapeutic option for
Ewing's sarcoma. Int J Oncol. 43:803–812. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Palmerini E, Torricelli E, Cascinu S,
Pierini M, De Paolis M, Donati D, Cesari M, Longhi A, Abate M,
Paioli A, et al: Is there a role for chemotherapy after local
relapse in high-grade osteosarcoma? Pediatr Blood Cancer.
66:e277922019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li W and Zhang S: Survival of patients
with primary osteosarcoma and lung metastases. J BUON.
23:1500–1504. 2018.PubMed/NCBI
|
|
22
|
Grimer RJ, Taminiau AM and Cannon SR;
Surgical Subcommitte of the European Osteosarcoma Intergroup, :
Surgical outcomes in osteosarcoma. J Bone Joint Surg Br.
84:395–400. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang JY, Yang Y, Ma Y, Wang F, Xue A, Zhu
J, Yang H, Chen Q, Chen M, Ye L, et al: Potential regulatory role
of lncRNA-miRNA-mRNA axis in osteosarcoma. Biomed Pharmacother.
121:1096272020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rickel K, Fang F and Tao J: Molecular
genetics of osteosarcoma. Bone. 102:69–79. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Laoukili J, Stahl M and Medema RH: FoxM1:
At the crossroads of ageing and cancer. Biochim Biophys Acta.
1775:92–102. 2007.PubMed/NCBI
|
|
26
|
Pilarsky C, Wenzig M, Specht T, Saeger HD
and Grutzmann R: Identification and validation of commonly
overexpressed genes in solid tumors by comparison of microarray
data. Neoplasia. 6:744–750. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gentles AJ, Newman AM, Liu CL, Bratman SV,
Feng W, Kim D, Nair VS, Xu Y, Khuong A, Hoang CD, et al: The
prognostic landscape of genes and infiltrating immune cells across
human cancers. Nat Med. 21:938–945. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Halasi M and Gartel AL: FOX(M1) news-it is
cancer. Mol Cancer Ther. 12:245–254. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gartel AL: FOXM1 in cancer: Interactions
and vulnerabilities. Cancer Res. 77:3135–3139. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bhat UG, Jagadeeswaran R, Halasi M and
Gartel AL: Nucleophosmin interacts with FOXM1 and modulates the
level and localization of FOXM1 in human cancer cells. J Biol Chem.
286:41425–41433. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Umeh-Garcia M, Simion C, Ho PY, Batra N,
Berg AL, Carraway KL, Yu A and Sweeney C: A Novel bioengineered
miR-127 prodrug suppresses the growth and metastatic potential of
triple-negative breast cancer cells. Cancer Res. 80:418–429. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gong HL, Tao Y, Mao XZ, Song DY, You D and
Ni JD: MicroRNA-29a suppresses the invasion and migration of
osteosarcoma cells by regulating the SOCS1/NF-κB signalling pathway
through negatively targeting DNMT3B. Int J Mol Med. 44:1219–1232.
2019.PubMed/NCBI
|
|
33
|
Patil SL, Palat A, Pan Y, Rajapakshe K,
Mirchandani R, Bondesson M, Yustein JT, Coarfa C and Gunaratne PH:
MicroRNA-509-3p inhibits cellular migration, invasion, and
proliferation, and sensitizes osteosarcoma to cisplatin. Sci Rep.
9:190892019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen D, Liu D and Chen Z: Potential
therapeutic implications of miRNAs in osteosarcoma chemotherapy.
Tumour Biol. 39:10104283177057622017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jana S, Krishna M, Singhal J, Horne D,
Awasthi S, Salgia R and Singhal SS: Therapeutic targeting of
miRNA-216b in cancer. Cancer Lett. 484:16–28. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wu X, Chen W, Cai H, Hu J, Wu B, Jiang Y,
Chen X, Sun D and An Y: miR-216b inhibits pancreatic cancer cell
progression and promotes apoptosis by down-regulating KRAS. Arch
Med Sci. 14:1321–1332. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang Y, Dong D, Jiang S, Zhang E, Zheng W,
Mao L, Li W, Zhou J, Fan L, Cheng R, et al: miR-216b
post-transcriptionally downregulates oncogene KRAS and inhibits
cell proliferation and invasion in clear cell renal cell carcinoma.
Cell Physiol Biochem. 49:1755–1765. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen X, Liu X, He B, Pan Y, Sun H, Xu T,
Hu X and Wang S: miR-216b functions as a tumor suppressor by
targeting HMGB1-mediated JAK2/STAT3 signaling way in colorectal
cancer. Am J Cancer Res. 7:2051–2069. 2017.PubMed/NCBI
|
|
39
|
Wang L, Wang Y, Du X, Yao Y, Wang L and
Jia Y: miR-216b suppresses cell proliferation, migration, invasion,
and epithelial-mesenchymal transition by regulating FOXM1
expression in human non-small cell lung cancer. Onco Targets Ther.
12:2999–3009. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liu S, Dong H, Dai H, Liu D and Wang Z:
MicroRNA-216b regulated proliferation and invasion of non-small
cell lung cancer by targeting SOX9. Oncol Lett. 15:10077–10083.
2018.PubMed/NCBI
|
|
41
|
Zheng L, Zhang X, Yang F, Zhu J, Zhou P,
Yu F, Hou L, Xiao L, He Q and Wang B: Regulation of the P2X7R by
microRNA-216b in human breast cancer. Biochem Biophys Res Commun.
452:197–204. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bhat UG, Halasi M and Gartel AL: FoxM1 is
a general target for proteasome inhibitors. PLoS One. 4:e65932009.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hegde NS, Sanders DA, Rodriguez R and
Balasubramanian S: The transcription factor FOXM1 is a cellular
target of the natural product thiostrepton. Nat Chem. 3:725–731.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Gartel AL: Thiostrepton, proteasome
inhibitors and FOXM1. Cell Cycle. 10:4341–4342. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Koo CY, Muir KW and Lam EW: FOXM1: From
cancer initiation to progression and treatment. Biochim Biophys
Acta. 1819:28–37. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ahmed M, Uddin S, Hussain AR, Alyan A,
Jehan Z, Al-Dayel F, Al-Nuaim A, Al-Sobhi S, Amin T, Bavi P and
Al-Kuraya KS: FoxM1 and its association with matrix
metalloproteinases (MMP) signaling pathway in papillary thyroid
carcinoma. J Clin Endocrinol Metab. 97:E1–E13. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Halasi M, Schraufnagel DP and Gartel AL:
Wild-type p53 protects normal cells against apoptosis induced by
thiostrepton. Cell Cycle. 8:2850–2851. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang M and Gartel AL: Micelle-encapsulated
thiostrepton as an effective nanomedicine for inhibiting tumor
growth and for suppressing FOXM1 in human xenografts. Mol Cancer
Res. 10:2287–2297. 2011.
|
|
49
|
Kwok JM, Peck B, Monteiro LJ, Schwenen HD,
Millour J, Coombes RC, Myatt SS and Lam EW: FOXM1 confers acquired
cisplatin resistance in breast cancer cells. Mol Cancer Res.
8:24–34. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wang M and Gartel AL: Combination with
bortezomib enhances the antitumor effects of
nanoparticle-encapsulated thiostrepton. Cancer Biol Ther.
13:184–189. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Jin P, Chen X, Yu G, Li Z, Zhang Q and
Zhang JV: The clinical and experimental research on the treatment
of endometriosis with thiostrepton. Anticancer Agents Med Chem.
19:323–329. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Jiang L, Wu X, Wang P, Wen T, Yu C, Wei L
and Chen H: Targeting FoxM1 by thiostrepton inhibits growth and
induces apoptosis of laryngeal squamous cell carcinoma. J Cancer
Res Clin Oncol. 141:971–981. 2015. View Article : Google Scholar : PubMed/NCBI
|