Introduction
Glioblastoma (GBM) is the most common and severe
type of human glioma, with an incidence rate of 3.19 cases/100,000
individuals/year globally, as reported in 2017 (1). Glioma is a primary malignant tumor
occurring in the brain, the incidence rate of which is the highest
among tumors of the central nervous system (2,3). GBM is
known as grade IV glioma, with characteristics of high malignancy,
high recurrence rate and very low 5-year survival rate (4–5%)
(4,5). One of the reasons why GBM is difficult
to cure is that the tumor tissue mainly grows infiltratively; the
tumor cells migrate to the surrounding brain tissue during the
process of proliferation, binding to the dura mater or directly
penetrating into deep brain structures, such as the ventricles
(6–8). The molecular mechanisms of GBM cell
proliferation and invasion are crucial, and remain to be fully
elucidated.
Some important molecules have been found to be
involved in the proliferation and invasion of GBM cells, such as
cell adhesion molecule 1 (CADM1) (9–11). CADM1
has been identified as a tumor suppressor in a variety of tumors,
including lung, prostate, liver, pancreas and breast cancer
(12). Studies have indicated that
CADM1 is able to activate STAT3 and the PI3K/Akt signaling pathway,
and thus regulates the proliferation of tumor cells, including lung
cancer cells and squamous cell carcinoma cells (13,14). In
GBM-associated research, it has been demonstrated that CADM1
expression is decreased in patients with GBM and in GBM cell lines,
and CADM1 overexpression inhibits the proliferation of GBM cells
(9,11). These findings indicate that CADM1 is
an effective suppressor of GBM proliferation.
MicroRNAs (miRNAs/miRs) are a type of non-coding
RNAs, which form RNA-induced silencing complexes by binding to the
mRNA of target genes, and thus inhibiting their translation
(15,16). Therefore, by regulating the
expression levels of oncogenes and tumor suppressors, miRNAs are
able to regulate the development and pathogenesis of a variety of
tumors, including breast, prostate and pancreatic cancer (17). In addition, a number of studies have
indicated that the expression levels of some miRNAs are abnormal
during the occurrence of GBM (18–20).
Therefore, miRNAs have been widely investigated in tumor detection
and treatment, including GBM. For instance, the expression levels
of miR-21 and miR-10b are upregulated in GBM, while those of
miR-15b, miR-137 and miR-124 are downregulated in GBM;
additionally, manipulating the levels of these miRNAs can regulate
some of the pathological characteristics of GBM, including cell
migration, invasion, proliferation and apoptosis (21–24).
Among the cancer-associated miRNAs, miR-15a-5p has been reported to
be involved in the proliferation and invasion of a variety of tumor
cells, including non-small cell lung cancer, colorectal
adenocarcinoma and endometrial cancer cells (25–27).
However, its role in GBM has not yet been elucidated. Therefore,
the present study aimed to investigate the role of miR-15a-5p in
GBM.
Materials and methods
Antibodies and reagents
The antibodies used in the present were as follows:
CADM1 (1:1,000; cat. no. ABT66; EMD Millipore), Akt (1:1,000; cat.
no. 9272; Cell Signaling Technology, Inc.), phosphorylated (p)-Akt
(1:1,000; cat. no. 4060; Cell Signaling Technology, Inc.) and GAPDH
(1:1,000; cat. no. HPA061280; EMD Millipore) primary antibodies,
and goat anti-rabbit (1:3,000; cat. no. 7074; Cell Signaling
Technology, Inc.) secondary antibodies.
The reagents used were as follows: Fetal bovine
serum (FBS; Thermo Fisher Scientific, Inc.), Dulbecco's modified
Eagle's medium (DMEM; Thermo Fisher Scientific, Inc.),
penicillin-streptomycin (Thermo Fisher Scientific, Inc.),
Lipofectamine® 2000 (Thermo Fisher Scientific, Inc.),
TRIzol® (Thermo Fisher Scientific, Inc.), the
PrimeScript qRT Reagent kit (Takara Bio, Inc.), SYBR-Green Mix
(Roche Diagnostics), the Dual-Luciferase Assay System (Promega
Corporation), Cell Counting Kit-8 (CCK-8; Dojindo Molecular
Technologies, Inc.), protease inhibitor cocktail (Sigma-Aldrich;
Merck KGaA), RIPA buffer (Roche Diagnostics), BSA (Beijing Solarbio
Science & Technology Co., Ltd.) and Premixed Luminata™ Western
HRP substrates for stronger signals (EMD Millipore).
Cell culture and transfection
The T98G cell line was obtained from the American
Type Culture Collection. Human brain normal astroglia HEB cells
were purchased from Ningbo Mingzhou Biotechnology Co., Ltd. (cat.
no. MZ-0831). The cells were cultured in DMEM containing 10% FBS
and 1% penicillin-streptomycin, and incubated in a 37°C incubator
containing 5% CO2.
Small RNAs [50 nM miR-negative control (NC) mimic or
miR-15a-5p mimic; 100 nM miR-NC inhibitor or miR-15a-5p inhibitor]
and small interfering (si)RNAs (50 nM siNC, siCADM1-1 and
siCADM1-2) were transfected into the cells using Lipofectamine
2000. After 48 h of incubation, the cells were used for subsequent
experiments. The non-targeting scrambled miR-NC mimic and miR-NC
inhibitor, miR-15a-5p inhibitor and miR-15a-5p mimic were obtained
from Shanghai GenePharma Co., Ltd., while siCADM1 and siNC were
purchased from Shanghai Tuoran Biological Technology Co., Ltd.
Their sequences were as follows: miR-NC inhibitor,
5′-CAGUACUUUUGUGUAGUACAA-3′; miR-15a-5p inhibitor,
5′-CACAAACCAUUAUGUGCUGCUA-3′; miR-NC mimic,
5′-UUCUCCGAACGUGUCACGUTT-3′; miR-15a-5p mimic sense,
5′-UAGCAGCACAUAAUGGUUUGUG-3′ and antisense,
5′-CACAAACCAUUAUGUGCUGCUA-3′; siNC sense,
5′-UUCUCCGAACGUGUCACGUdTdT-3′ and antisense,
5′-ACGUGACACGUUCGGAGAAdTdT-3′; siCADM1-1 sense,
5′-CCAACCUGUUCAUCAAUAAdTdT-3′ and antisense,
5′-UUAUUGAUGAACAGGUUGGdTdT-3′; and siCADM1-2 sense,
5′-UCAGGUGGUUCAAAGGGAAdTdT-3′ and antisense,
5′-UUCCCUUUGAACCACCUGAdTdT-3′.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from the cultured T98G or
HEB cells using TRIzol reagent, and then reverse transcribed into
cDNA at 37°C for 15 min and 85°C for 5 sec using the PrimeScript
qRT Reagent kit according to the manufacturer's protocol. The
SYBR-Green Mix kit was used for the RT-qPCR experiments on an ABI
Prism 7500 system (Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. The thermocycling conditions were as
follows: 95°C for 3 min, followed by 40 cycles of 95°C for 10 sec,
60°C for 15 sec and 72°C for 31 sec. The expression levels of
miRNAs and mRNAs were analyzed using the 2−∆∆Cq method
(28), with U6 and GAPDH as the
internal control for miRNAs and mRNAs, respectively. The primer
sequences were as follows: miR-15a-5p forward,
5′-TAGCAGCACATAATGGTTTGTG-3′ and reverse, 5′-CTCAACTGGTGTCGTGGA-3′;
U6 forward, 5′-CTCGCTTCGGCAGCACA-3′ and reverse,
5′-AACGCTTCACGAATTTGCGT-3′; CADM1 forward,
5′-CCACAGGTGATGGGCAGAAT-3′ and reverse, 5′-TTCCTGTGGGGGATCGGTAT-3′;
GAPDH forward, 5′-GAGAAGGCTGGGGCTCATTT-3′ and reverse,
5′-AGTGATGGCATGGACTGTGG-3′.
Transwell assay
T98G cells (5×104) were seeded in
serum-free DMEM in the upper chamber of a Matrigel-precoated
Transwell chamber (Corning, Inc.), while the lower chamber was
filled with DMEM with 10% FBS. Following incubation at 37°C for 24
h, the upper chamber was removed, the polycarbonate membrane was
inverted and cells on top of the membrane were removed using a
cotton swab. The invading cells were fixed with 4% paraformaldehyde
for 1 h at room temperature and stained with 0.25% crystal violet
for 1 h at room temperature. The invading cells were counted based
on five field digital images taken at a magnification of ×100 using
an Olympus IX51 light microscope (Olympus Corporation).
CCK-8 assay
CCK-8 assay was used to detect the cell
proliferative ability. T98G cells transfected with miR-NC
inhibitor, miR-15a-5p inhibitor, siNC or siCADM1 were seeded (1,000
cells/well) in 96-well plates and incubated at 37°C for different
periods of time (24, 48 or 72 h). Prior to CCK-8 detection, 100 µl
fresh DMEM containing 10 µl CCK-8 reagent were added to each well
according to the manufacturer's protocol and the cells were
cultured for 4 h at 37°C. The OD values of the different groups of
cells were measured at 450 nm using a spectrophotometer (BioTek
Instruments, Inc.).
Dual-luciferase reporter assay
The candidate target genes for miR-15a-5p were
screened using TargetScan (http://www.targetscan.org) (29). The 3′-untranslated region (3′-UTR)
sequences of wild-type (WT) CADM1 were obtained from T98G cell
cDNA, and inserted into pGL3-luciferase reporter plasmids (Promega
Corporation) using KpnI and XhoI restriction sites to
obtain pGL3-CADM1-3′-UTR-WT. Two-point mutations were introduced
into CADM1 3′-UTR using a site-directed mutagenesis kit (Agilent
Technologies, Inc.), in order to construct pGL3-CADM1-3′-UTR-mutant
(Mut). The plasmids were then co-transfected with 50 nM miR-15a-5p
mimic or NC mimic into T98G cells using Lipofectamine 2000. After
incubation at 37°C for 48 h, the luciferase intensity was detected
using the Dual-Luciferase Assay System (Promega Corporation).
Renilla luciferase activity was used as the reference.
Western blot analysis
T98G cells were washed with PBS, collected in test
tubes by centrifugation at 1,000 × g at 4°C for 5 min, and lysed
with RIPA buffer containing protease inhibitor. Protein
concentration was determined using a BCA kit (Beyotime Institute of
Biotechnology). The SDS loading buffer was mixed with the protein
samples. Proteins (15 µg/lane) were separated via 8% SDS-PAGE and
transferred to a PVDF membrane, which was blocked with 5% BSA at
room temperature for 2 h in TBS-Tween 20 (0.5%; TBST), then
incubated with primary antibodies overnight at 4°C, followed by
HRP-conjugated secondary antibodies at room temperature for 1 h.
After washing thoroughly with TBST, the HRP signals were detected
with chemical HRP substrate. The intensity of positive protein
bands was measured using ImageJ software version 1.50 (National
Institutes of Health), and the average value of the control group
was normalized to 1. GAPDH served as the reference protein.
Statistical analysis
All data were analyzed using GraphPad prism 6.0
(GraphPad Software, Inc.) and are expressed as the mean ± SEM. Each
experiment was repeated ≥3 times. Unpaired student's t-test was
used to compare differences between 2 groups. One-way ANOVA
followed by Tukey's post hoc test was used to compare differences
among ≥3 groups. P<0.05 was considered to indicate a
statistically significant difference.
Results
miR-15a-5p expression is upregulated
in T98G cells
To examine whether miR-15a-5p expression is normal
in GBM, RT-qPCR was performed to detect miR-15a-5p expression in
T98G and HEB cells. T98G is a widely used human GBM cell line,
while HEB is a type of human brain normal astroglia cell line. As
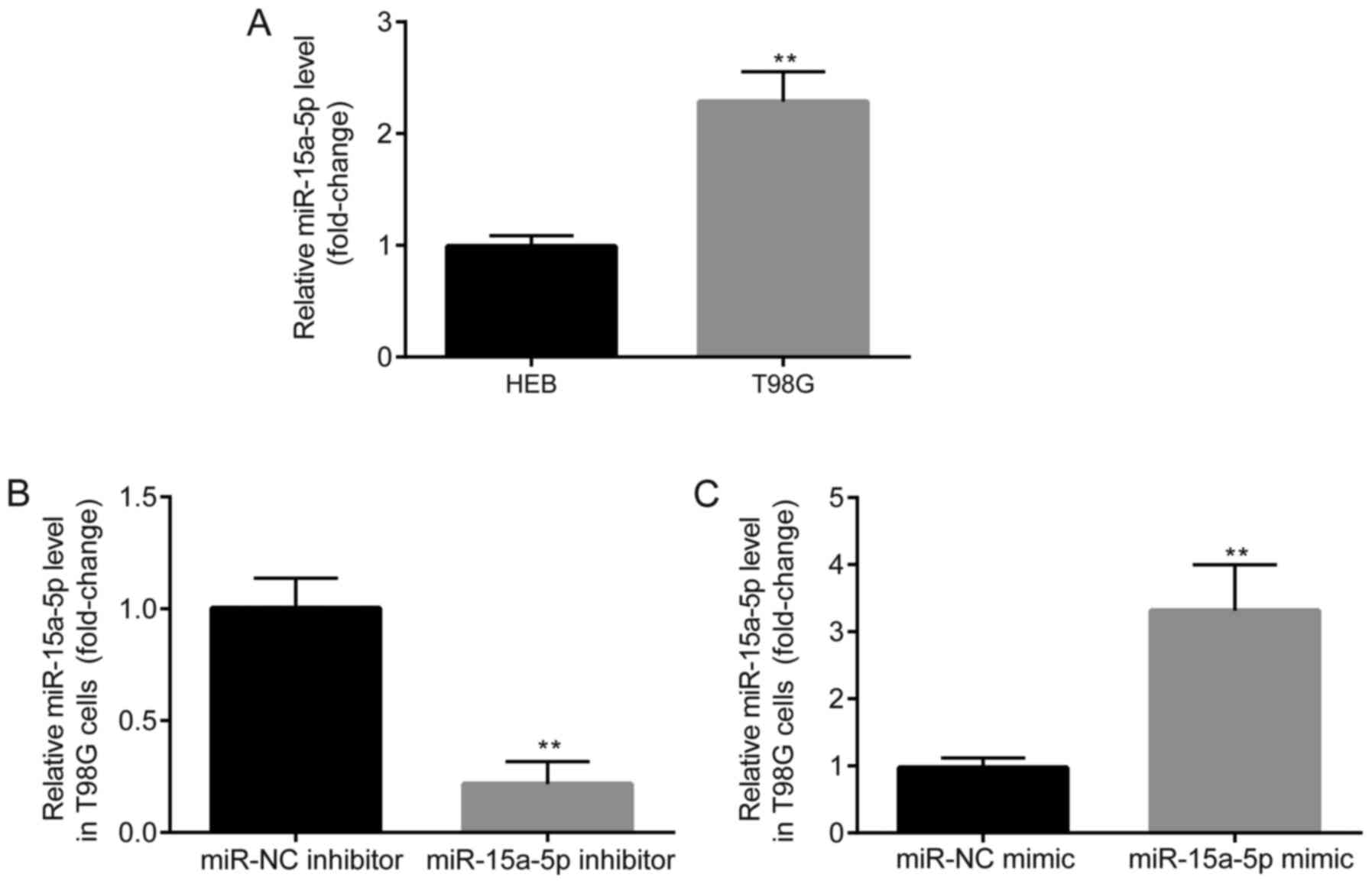
shown by the results presented in Fig.
1A, miR-15a-5p expression was significantly higher in T98G
cells compared with that in HEB cells, which indicated that
miR-15a-5p expression was upregulated in GBM cells. To further
examine the roles of miR-15a-5p in GBM, miR-15a-5p inhibitor and
miR-15a-5p mimic were designed to inhibit or enhance miR-15a-5p
expression, respectively (Fig. 1B and
C).
miR-15a-5p promotes T98G cell
proliferation and invasion
In order to verify whether miR-15a-5p participates
in GBM cell proliferation and invasion, CCK-8 and Transwell assays
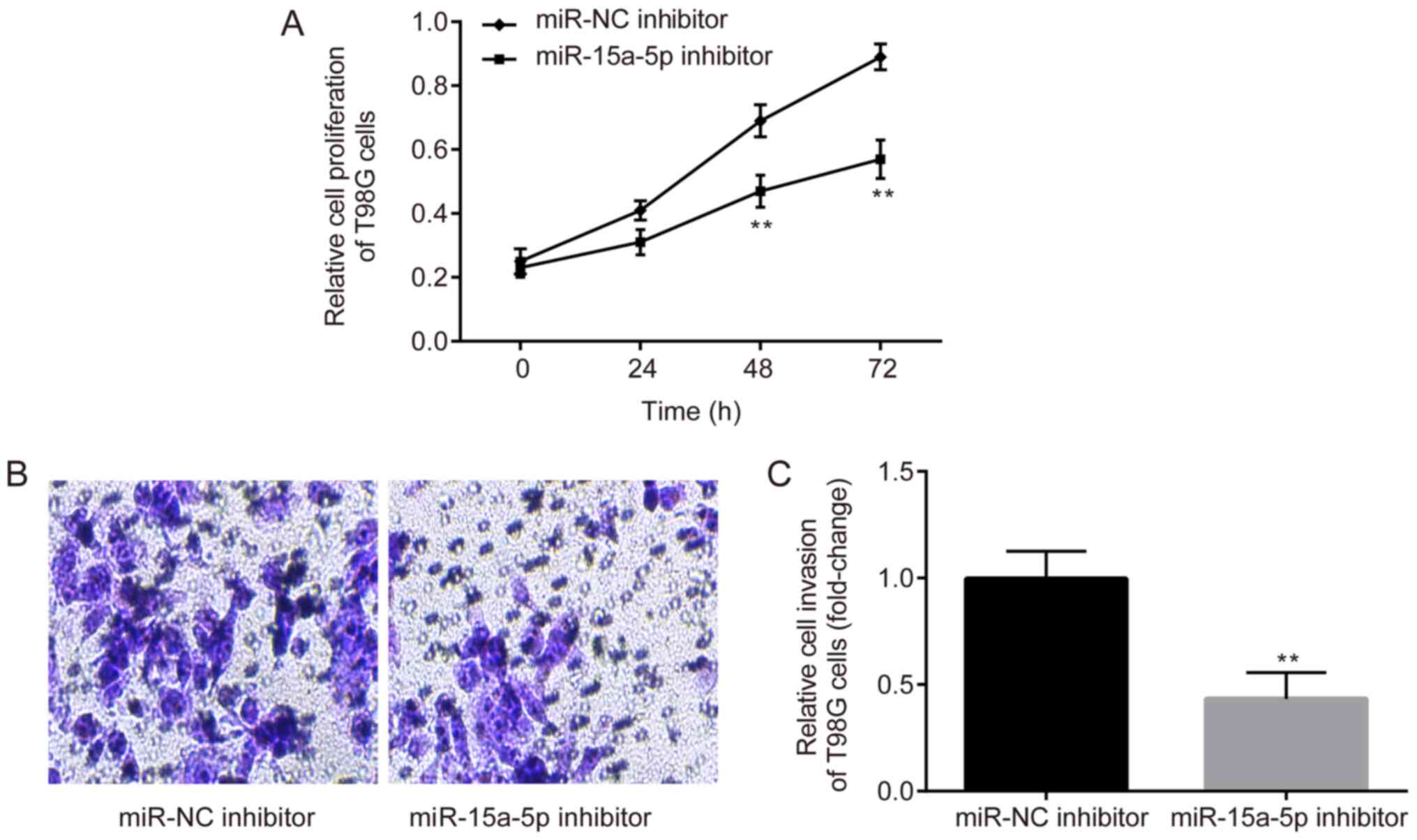
were performed. As shown in Fig. 2A,
the results of CCK-8 assay revealed that the proliferation of T98G
cells was significantly suppressed by the miR-15a-5p inhibitor
compared with the NC inhibitor. Similarly, as shown by Transwell
assay, which was used to measure the cell invasive ability,
miR-15a-5p inhibitor effectively restrained the invasion of T98G
cells (Fig. 2B and C). These
experiments indicated that miR-15a-5p was able to promote the
proliferation and invasion of GBM cells.
CADM1 is regulated by miR-15a-5p
In order to determine the mechanisms through which
miR-15a-5p regulates cell proliferation and invasion, the candidate
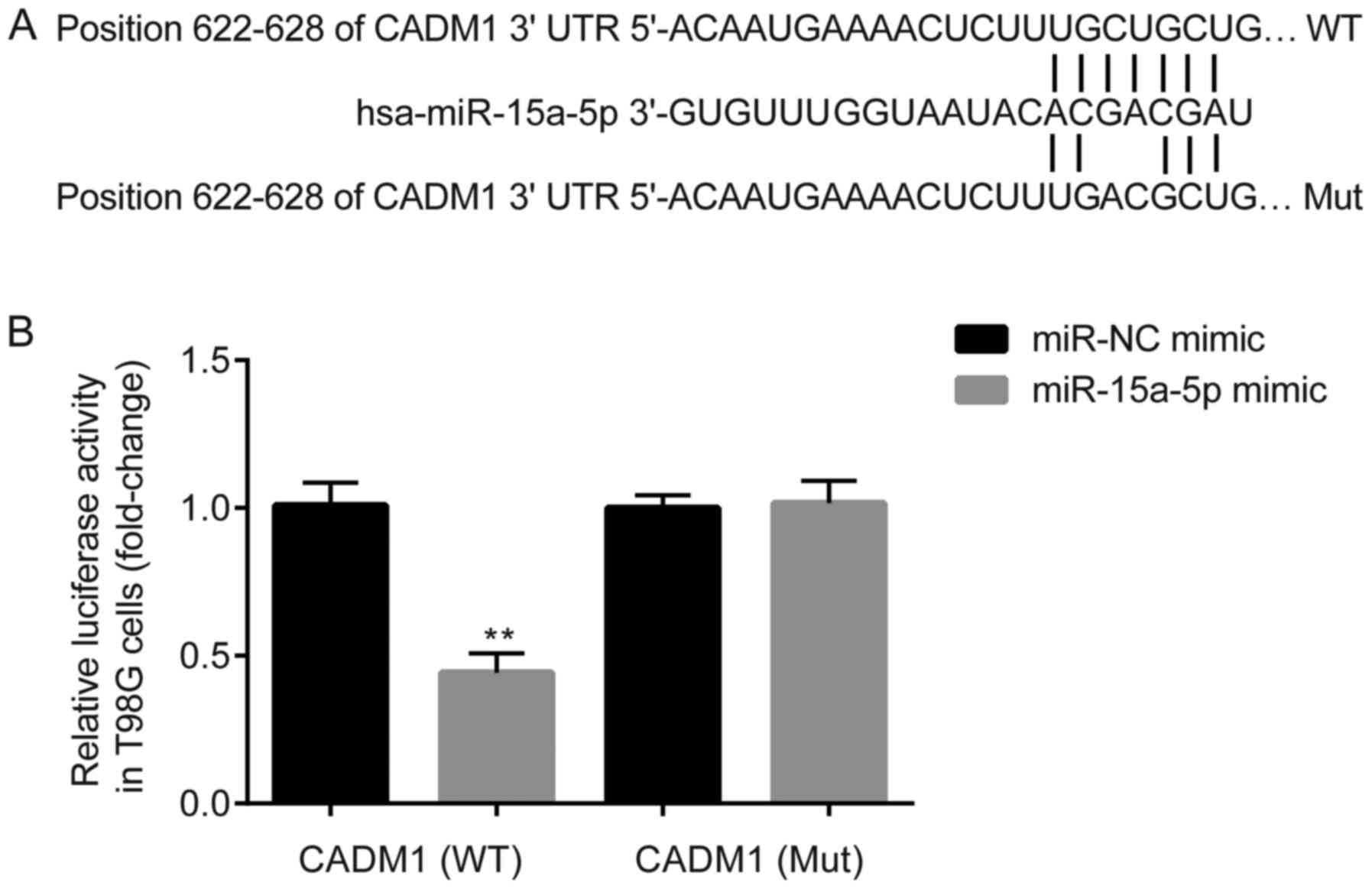
target genes of miR-15a-5p were screened using TargetScan. The
results revealed that the 3′-UTR of CADM1 may contain a targeting
site of miR-15a-5p (Fig. 3A). For
further verification, a normal CADM1 3′-UTR sequence (WT) and a
mutated one (Mut) were designed, and each sequence was inserted
into the luciferase reporter plasmid for dual-luciferase activity
assay. As shown by the results presented in Fig. 3B, in the T98G cells transfected with
the WT reporter plasmid, the luciferase activity in the miR-15a-5p
mimic group was significantly lower than that in the NC mimic
group. On the other hand, no significant difference was observed
between the two groups transfected with the Mut plasmid.
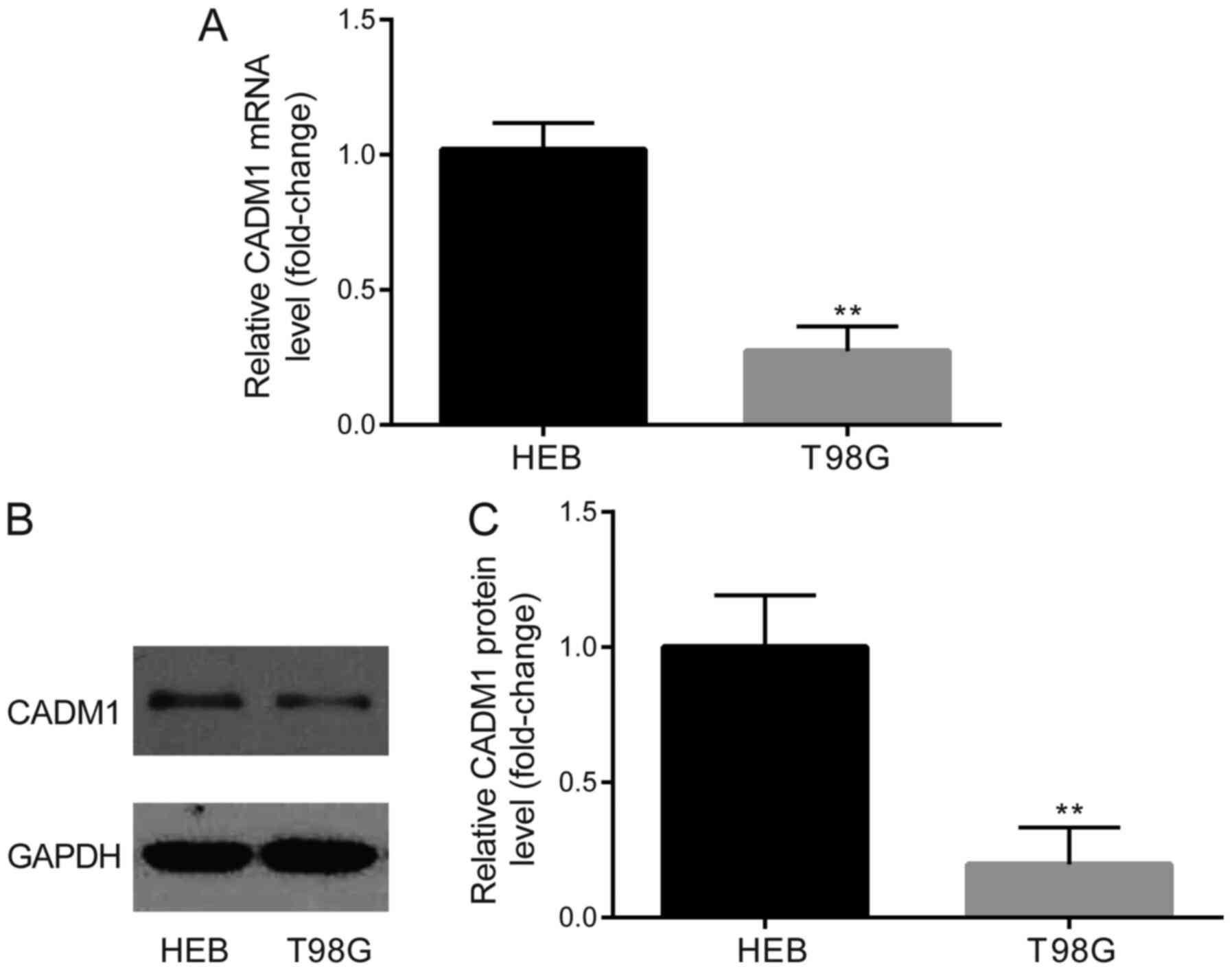
Subsequently, the present study determined whether
CADM1 expression was altered in GBM cells. RT-qPCR and western blot
analysis were performed to detect the expression levels of CADM1 in
T98G and HEB cells. The results revealed that both the mRNA and
protein levels of CADM1 were downregulated in T98G cells compared
with those in HEB cells (Fig. 4A-C).
These findings were consistent with the upregulated miR-15a-5p
phenotype observed in T98G cells.
Inhibiting CADM1 attenuates the
effects of miR-15a-5p on cell proliferation and invasion
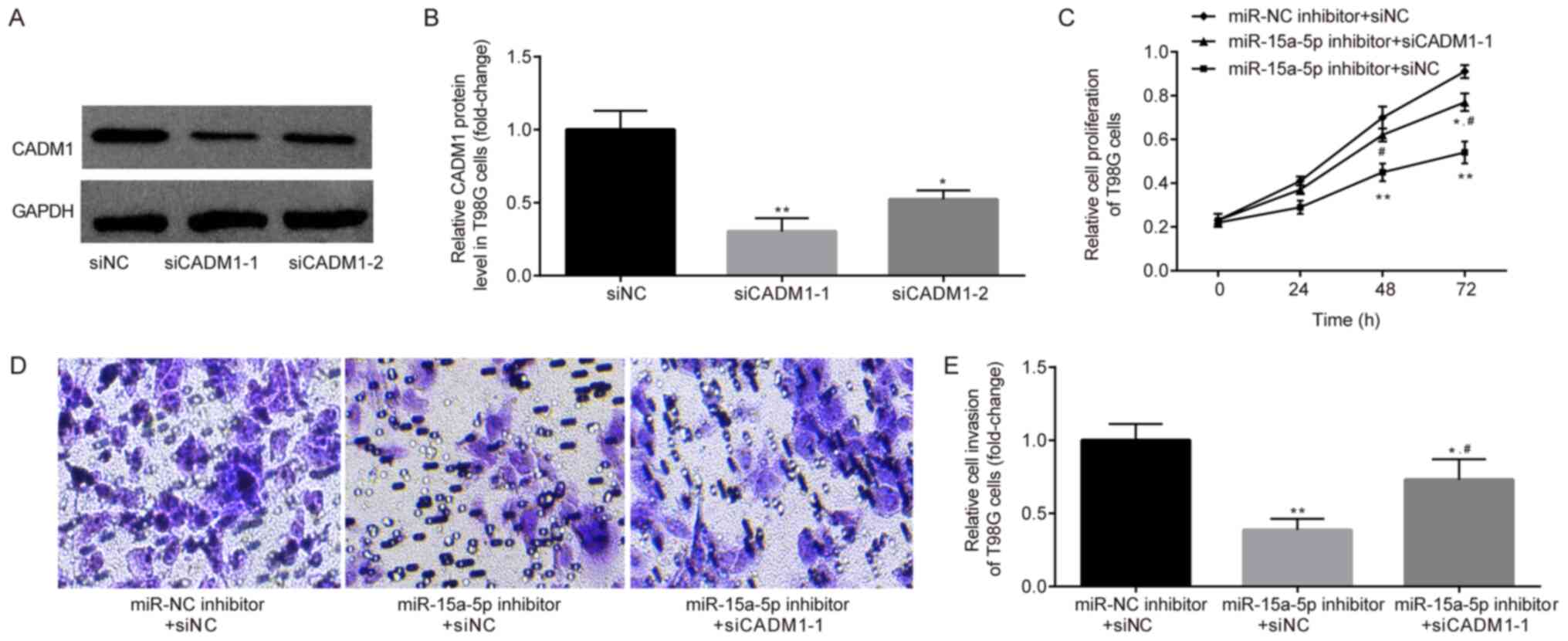
To examine whether CADM1 was involved in
miR-15a-5p-induced cell proliferation and invasion, two CADM1
siRNAs (Fig. 5A and B) were
designed, with siCADM-1 resulting more effective and being
therefore selected for further research. Subsequently, siCADM1-1
and miR-15a-5p inhibitor were co-transfected into the T98G cells
for CCK-8 and Transwell assays. As shown in Fig. 5C-E, miR-15a-5p inhibitor
significantly suppressed T98G cell proliferation and invasion, and
these phenotypes were attenuated when the cells were co-transfected
with siCADM1-1. These results suggested that miR-15a-5p promoted
T98G cell proliferation and invasion by targeting CADM1.
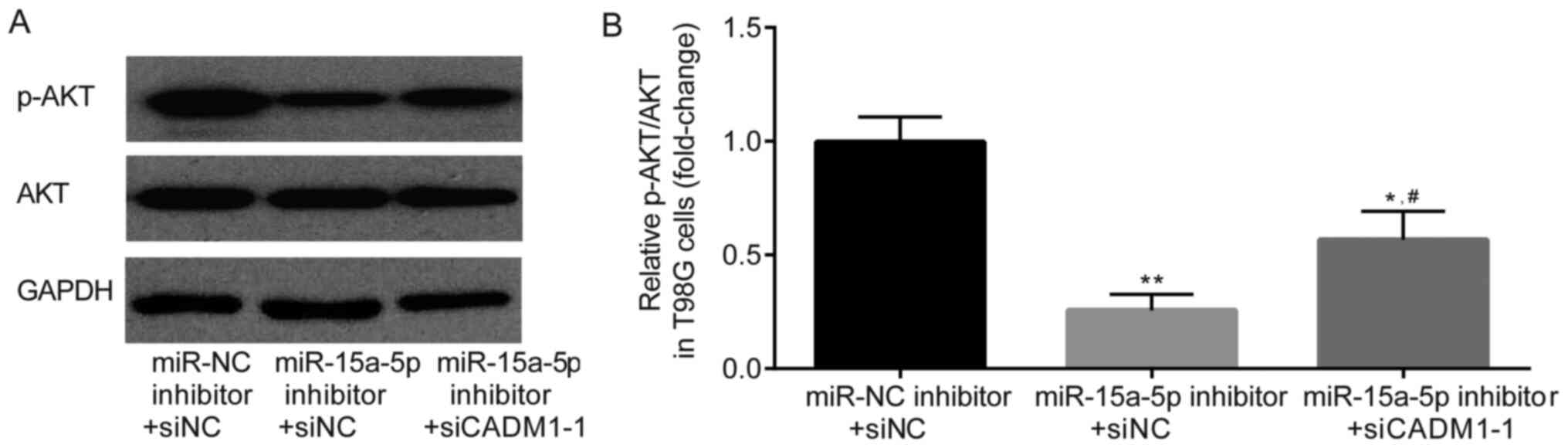
Effects of miR-15a-5p and CADM1 on Akt
activity
Akt, also known as protein kinase B, is an important
protein regulating cell proliferation and has been found to be
overactivated in GBM tissues and cells (30,31).
p-Akt is the activated form, and has been demonstrated to be
regulated by CADM1 (13).
Consequently, the present study investigated whether miR-15a-5p and
CADM1 were able to regulate Akt phosphorylation in T98G cells. As
shown in Fig. 6A and B, the
phosphorylation levels of Akt were decreased using the miR-15a-5p
inhibitor, and this effect was partially reversed by siCADM1-1.
Discussion
The present study identified a novel mechanism
through which miRNAs may promote GBM cell proliferation and
invasion. The data demonstrated that miR-15a-5p expression was
higher in the GBM T98G cell line than in an immortal control cell
line. Introducing miR-15a-5p inhibitor into T98G cells effectively
inhibited Akt phosphorylation and suppressed T98G cell
proliferation and invasion. The cell adhesion protein CADM1 was
found to be regulated by miR-15a-5p, and inhibiting CADM1 in the
T98G cells attenuated the effects induced by the miR-15a-5p
inhibitor.
One of the key findings of the present study was
that miR-15a-5p expression in T98G cells was significantly
increased compared with that in normal cells, and subsequent
experiments revealed that miR-15a-5p inhibition suppressed cell
proliferation and invasion, indicating that miR-15a-5p was able to
promote cancer cell invasion. Therefore, in GBM cells, the abnormal
expression levels of miR-15a-5p further aggravated the cancer
pathological process. In addition, miR-15a-5p expression has been
evaluated in other types of tumors, including non-small cell lung
cancer and colorectal adenocarcinoma, in which it has been found to
promote cancer cell proliferation (25,26).
However, a previous study has reported that miR-15a-5p expression
is decreased in endometrial carcinoma (27), suggesting that the role of miR-15a-5p
may differ in various types of cancer.
The present study identified that CADM1 was one of
the candidate target genes of miR-15a-5p using TargetScan, and this
finding was confirmed using the dual-luciferase activity assay. The
current results demonstrated that only the miR-15a-5p mimic, but
not the NC mimic or Mut CADM1 3′-UTR, suppressed luciferase
activity. This strongly suggested that CADM1 may be targeted by
miR-15a-5p. In addition, subsequent experiments revealed a
functional association between miR-15a-5p and CADM1. In previous
studies, CADM1 has been demonstrated to interact with FERM and PDZ
domain containing protein and inhibit cancer cell proliferation of
squamous cell carcinoma and non-small cell lung cancer (14,32). In
GBM-associated studies, it has been demonstrated that CADM1
expression in GBM cell lines and patients with GBM are
significantly decreased (9). These
findings are consistent with the results of the present study,
suggesting that CADM1 may be a reliable and stable tumor
suppressor.
In addition to CADM1, there are several other target
genes of miR-15a-5p. It has been reported that miR-15a-5p regulates
the proliferation of endometrial mesenchymal stem cells by
inhibiting VEGFA (33), and can also
inhibit the growth of uterine leiomyoma cells by inhibiting the
Wnt3 protein through the Wnt/β-catenin signaling pathway (27). These results suggest that miR-15a-5p
may be able to regulate cell cycle through different signaling
pathways. CADM1 inhibition alone may not be sufficient to reverse
all the effects induced by the miR-15a-5p inhibitor, which was one
of the reasons that the knockdown of CADM1 expression only
partially reversed the effects of the miR-15a-5p inhibitor on T98G
cell proliferation and invasion. Another reason for this partial
reversal effect may be that a small amount of CADM1 is sufficient
to inhibit cancer cell proliferation and invasion, since siCADM1
could not completely prevent all CADM1 mRNA translation.
The PI3K/Akt signaling pathway is closely associated
with the proliferation and invasion of a number of tumor cells,
including GBM and non-small cell lung cancer cells (30,31,34). The
present study firstly demonstrated that miR-15a-5p was able to
inhibit Akt activity in GBM cells, which was consistent with the
results obtained in other cancer systems, including gastric and
ovarian cancer (35,36). Additionally, it has been demonstrated
that Akt participates in other physiological processes of tumor
cells, such as autophagy, aging and apoptosis (37–39),
suggesting that miR-15a-5p may also serve a role in these
functions.
In conclusion, the present study demonstrated that
miR-15a-5p promoted GBM cell proliferation and invasion by
targeting CADM1. The present findings revealed a novel candidate
mechanism through which miRNAs may participate in the development
of GBM and provided a potential target for the treatment of
GBM.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets generated and/or analyzed during the
current study are not publicly available due to their association
with another ongoing study, but are available from the
corresponding author on reasonable request.
Authors' contributions
FK, XL, SL, DS and WL conducted the experiments and
data analysis. MS designed the study and wrote the article. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Batash R, Asna N, Schaffer P, Francis N
and Schaffer M: Glioblastoma multiforme, diagnosis and treatment;
recent literature review. Curr Med Chem. 24:3002–3009. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ricard D, Idbaih A, Ducray F, Lahutte M,
Hoang-Xuan K and Delattre JY: Primary brain tumours in adults.
Lancet. 379:1984–1996. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dalessio A, Proietti G, Sica G and
Scicchitano BM: Pathological and molecular features of glioblastoma
and its peritumoral tissue. Cancers (Basel). 11:4692019. View Article : Google Scholar
|
|
4
|
Louis DN, Perry A, Reifenberger G, von
Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD,
Kleihues P and Ellison DW: The 2016 World health organization
classification of tumors of the central nervous system: A summary.
Acta Neuropathol. 131:803–820. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Carlsson SK, Brothers SP and Wahlestedt D:
Emerging treatment strategies for glioblastoma multiforme. EMBO Mol
Med. 6:1359–1370. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Aldape KD, Brindle KM, Chesler L, Chopra
R, Gajjar A, Gilbert MR, Gottardo N, Gutmann DH, Hargrave D,
Holland EC, et al: Challenges to curing primary brain tumours. Nat
Rev Clin Oncol. 16:509–520. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Brandao M, Simon T, Critchley G and Giamas
G: Astrocytes, the rising stars of the glioblastoma
microenvironment. Glia. 67:779–790. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Osti D, De Bene MD, Rappa G, Santos M,
Matafora V, Richichi C, Faletti S, Beznoussenko GV, Mironov A,
Bachi A, et al: Clinical significance of extracellular vesicles in
plasma from glioblastoma patients. Clin Cancer Res. 25:266–276.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Isobe T, Hisamori S, Hogan DJ, Zabala M,
Hendrickson DG, Dalerba P, Cai S, Scheeren F, Kuo AH, Sikandar SS,
et al: miR-142 regulates the tumorigenicity of human breast cancer
stem cells through the canonical WNT signaling pathway. Elife.
3:e019772014. View Article : Google Scholar
|
|
10
|
Ghosh D, Ulasov IV, Chen L, Harkins LE,
Wallenborg K, Hothi P, Rostad S, Hood L and Cobbs CS:
TGFβ-responsive HMOX1 expression is associated with stemness and
invasion in glioblastoma multiforme. Stem Cells. 34:2276–2289.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang X, Li W, Kang Y, Zhang J and Yuan H:
SynCAM, a novel putative tumor suppressor, suppresses growth and
invasiveness of glioblastoma. Mol Biol Rep. 40:5469–5475. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Murakami Y: Involvement of a cell adhesion
molecule, TSLC1/IGSF4, in human oncogenesis. Cancer Sci.
96:543–552. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kikuchi S, Iwai M, Sakurai-Yageta M,
Tsuboi Y, Ito T, Maruyama T, Tsuda H, Kanai Y, Onizuka M, Sato Y
and Murakami Y: Expression of a splicing variant of the CADM1
specific to small cell lung cancer. Cancer Sci. 103:1051–1057.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Vallath S, Sage EK, Kolluri KK, Lourenco
SN, Teixeira VS, Chimalapati S, George PJ, Janes SM and Giangreco
A: CADM1 inhibits squamous cell carcinoma progression by reducing
STAT3 activity. Sci Rep. 6:240062016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gregory RI, Chendrimada TP and Shiekhattar
R: MicroRNA biogenesis: Isolation and characterization of the
microprocessor complex. Methods Mol Biol. 342:33–47.
2006.PubMed/NCBI
|
|
16
|
Krol J, Loedige I and Filipowicz W: The
widespread regulation of microRNA biogenesis, function and decay.
Nat Rev Genet. 11:597–610. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lee YS and Dutta A: MicroRNAs in cancer.
Annu Rev Pathol. 4:199–227. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dong Z and Cui H: Epigenetic modulation of
metabolism in glioblastoma. Semin Cancer Biol. 57:45–51. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Candido S, Lupo G, Pennisi M, Basile MS,
Anfuso CD, Petralia MC, Gattuso G, Vivarelli S, Spandidos DA, Libra
M and Falzone L: The analysis of miRNA expression profiling
datasets reveals inverse microRNA patterns in glioblastoma and
Alzheimer's disease. Oncol Rep. 42:911–922. 2019.PubMed/NCBI
|
|
20
|
Ye X, Wei W, Zhang Z, He C, Yang R, Zhang
J, Wu Z, Huang Q and Jiang Q: Identification of microRNAs
associated with glioma diagnosis and prognosis. Oncotarget.
8:26394–26403. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Guessous F, Alvaradovelez M, Marcinkiewicz
L, Zhang Y, Kim J, Heister S, Kefas B, Godlewski J, Schiff D, Purow
B and Abounader R: Oncogenic effects of miR-10b in glioblastoma
stem cells. J Neurooncol. 112:153–163. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang J, Liu H, Tian L, Wang F, Han L,
Zhang W and Bai YA: miR-15b inhibits the progression of
glioblastoma cells through targeting insulin-like growth factor
receptor 1. Horm Cancer. 8:49–57. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shi L, Chen J, Yang J, Pan T, Zhang S and
Wang Z: MiR-21 protected human glioblastoma U87MG cells from
chemotherapeutic drug temozolomide induced apoptosis by decreasing
Bax/Bcl-2 ratio and caspase-3 activity. Brain Res. 1352:255–264.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Silber J, Lim DA, Petritsch C, Persson AI,
Maunakea AK, Yu M, Vandenberg SR, Ginzinger DG, James CD, Costello
JF, et al: miR-124 and miR-137 inhibit proliferation of
glioblastoma multiforme cells and induce differentiation of brain
tumor stem cells. BMC Med. 6:142008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ergun S, Güney S, Temiz E, Petrovic N and
Gunes S: Significance of miR-15a-5p and CNKSR3 as novel prognostic
biomarkers in non-small cell lung cancer. Anticancer Agents Med
Chem. 18:1695–1701. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kontos CK, Tsiakanikas P, Avgeris M,
Papadopoulos IN and Scorilas A: miR-15a-5p, a novel prognostic
biomarker, predicting recurrent colorectal adenocarcinoma. Mol
Diagn Ther. 21:453–464. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang ZM, Wan XH, Sang GY, Zhao JD and Wang
DM: miR-15a-5p suppresses endometrial cancer cell growth via
Wnt/β-catenin signaling pathway by inhibiting WNT3A. Eur Rev Med
Pharmacol Sci. 21:4810–4818. 2017.PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
29
|
Lewis BP, Burge CB and Bartel DP:
Conserved seed pairing, often flanked by adenosines, indicates that
thousands of human genes are microRNA targets. Cell. 120:15–20.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Karar J and Maity A: PI3K/AKT/mTOR pathway
in angiogenesis. Front Mol Neurosci. 4:512011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li X, Wu C, Chen N, Gu H, Yen A, Cao L,
Wang E and Wang L: PI3K/Akt/mTOR signaling pathway and targeted
therapy for glioblastoma. Oncotarget. 7:33440–33450. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mao X, Seidlitz E, Ghosh K, Murakami Y and
Ghosh HP: The cytoplasmic domain is critical to the tumor
suppressor activity of TSLC1 in non-small cell lung cancer. Cancer
Res. 63:7979–7985. 2003.PubMed/NCBI
|
|
33
|
Chen H and Tian Y: MiR-15a-5p regulates
viability and matrix degradation of human osteoarthritis
chondrocytes via targeting VEGFA. Biosci Trends. 10:482–488. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vivanco I and Sawyers CL: The
phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat Rev
Cancer. 2:489–501. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Pang K, Song J, Bai Z and Zhang Z:
miR-15a-5p targets PHLPP2 in gastric cancer cells to modulate
platinum resistance and is a suitable serum biomarker for
oxaliplatin resistance. Neoplasma. 67:1114–1121. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang K, Zhong W, Li WP, Chen ZJ and Zhang
C: miR-15a-5p levels correlate with poor ovarian response in human
follicular fluid. Reproduction. 154:483–496. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Franke TF, Hornik CP, Segev L, Shostak GA
and Sugimoto C: PI3K/Akt and apoptosis: Size matters. Oncogene.
22:8983–8998. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mirzaa GM, Campbell CD, Solovieff N, Goold
C, Jansen LA, Menon S, Timms AE, Conti V, Biag JD, Adams C, et al:
Association of MTOR mutations with developmental brain disorders,
including megalencephaly, focal cortical dysplasia, and pigmentary
mosaicism. JAMA Neurol. 73:836–845. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yang J, Pi C and Wang G: Inhibition of
PI3K/Akt/mTOR pathway by apigenin induces apoptosis and autophagy
in hepatocellular carcinoma cells. Biomed Pharmacother.
103:699–707. 2018. View Article : Google Scholar : PubMed/NCBI
|