|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2018. CA Cancer J Clin. 68:7–30. 2018. View Article : Google Scholar
|
|
2
|
Doubeni CA, Doubeni AR and Myers AE:
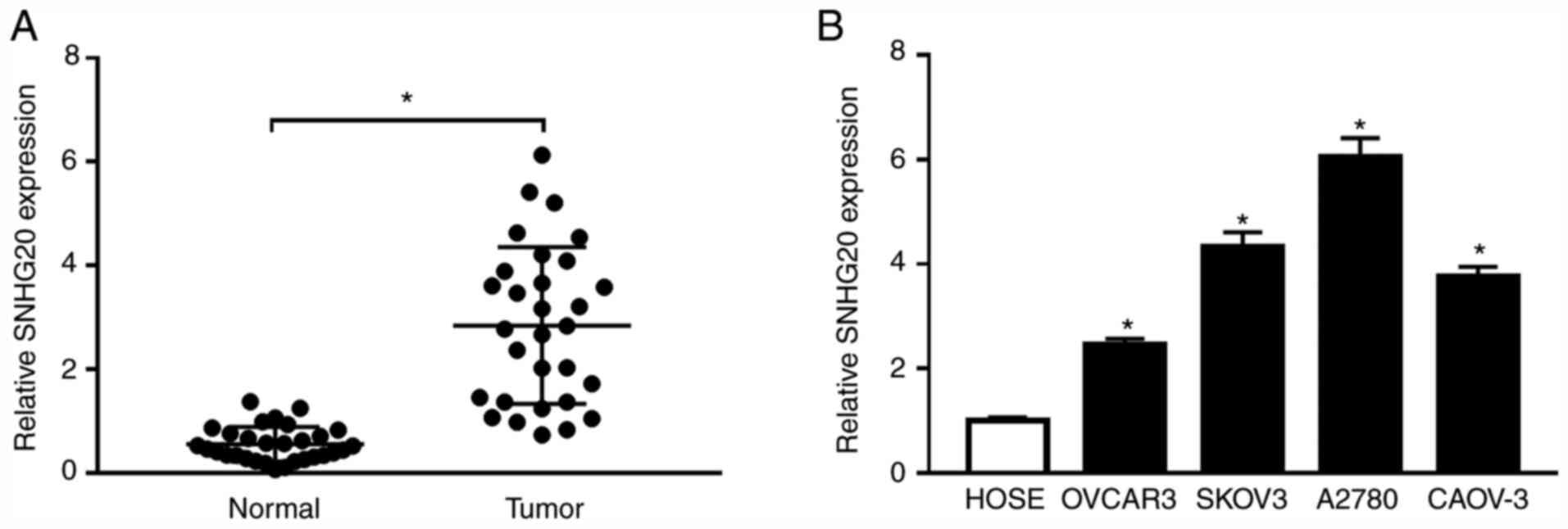
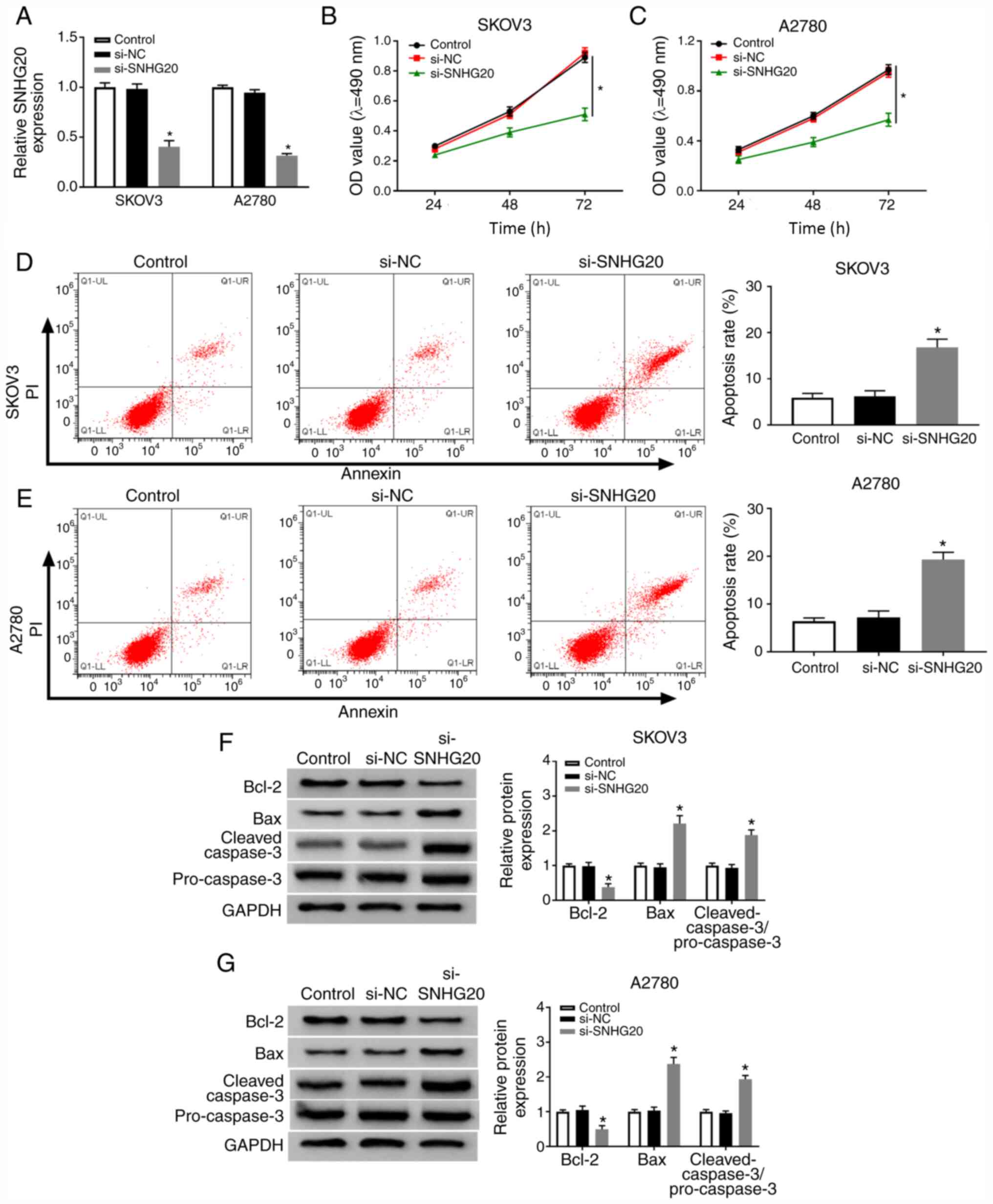
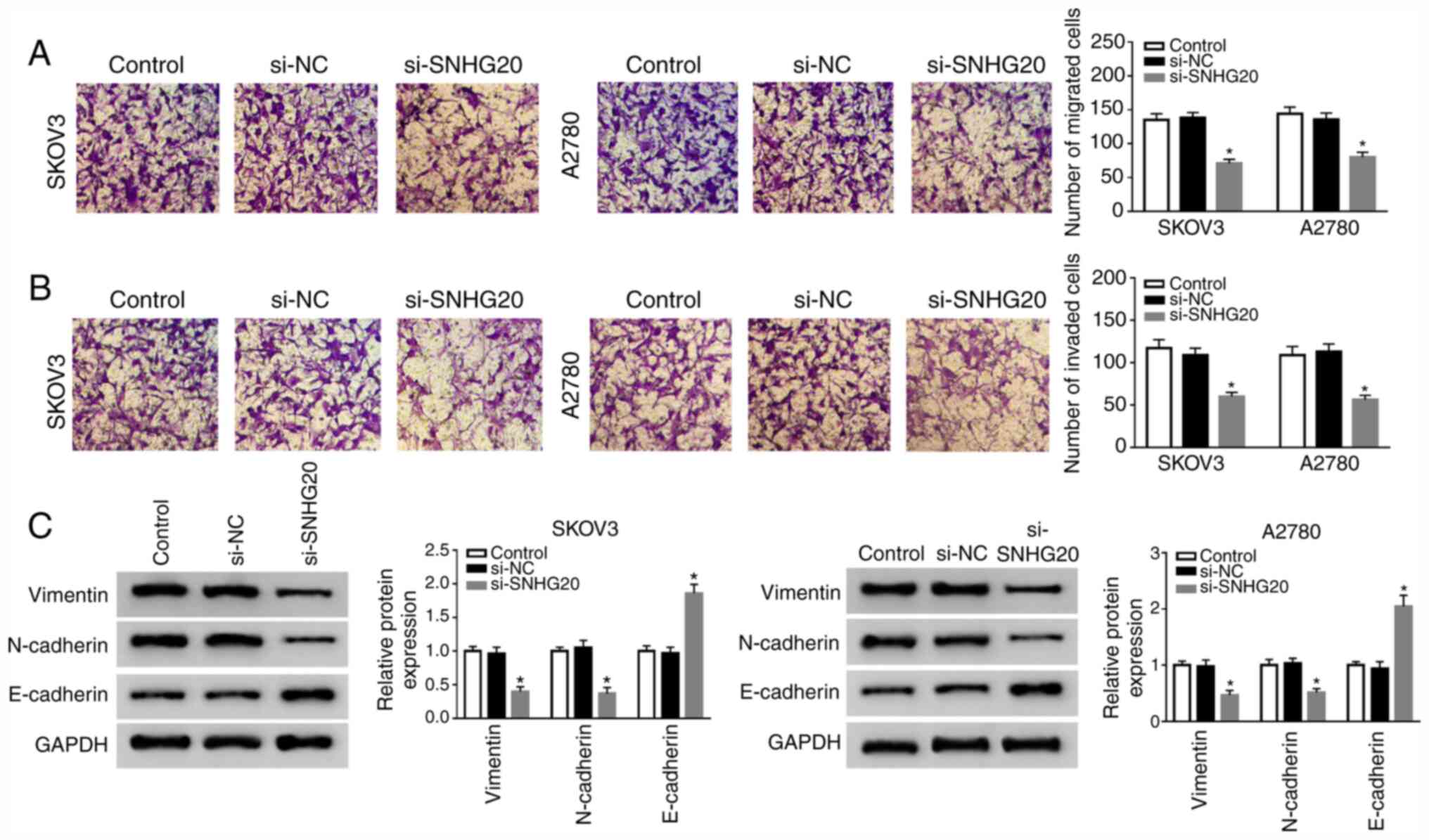
Diagnosis and management of ovarian cancer. Am Fam Physician.
93:937–944. 2016.
|
|
3
|
Xue Y, Ma G, Gu D, Zhu L, Hua Q, Du M, Chu
H, Tong N, Chen J, Zhang Z and Wang M: Genome-wide analysis of long
noncoding RNA signature in human colorectal cancer. Gene.
556:227–234. 2015. View Article : Google Scholar
|
|
4
|
Xiao J, Lai H, Wei SH, Ye ZS, Gong FS and
Chen LC: lncRNA HOTAIR promotes gastric cancer proliferation and
metastasis via targeting miR-126 to active CXCR4 and RhoA signaling
pathway. Cancer Med. 8:6768–6779. 2019. View Article : Google Scholar
|
|
5
|
Liang H, Su X, Wu Q, Shan H, Lv L, Yu T,
Zhao X, Sun J, Yang R, Zhang L, et al: LncRNA promotes ischemic
myocardial injury by regulating autophagy through targeting.
Autophagy undefined. 1–15. 2019.
|
|
6
|
Li Q, Zhang J, Su DM, Guan LN, Mu WH, Yu
M, Ma X and Yang RJ: lncRNA TUG1 modulates proliferation,
apoptosis, invasion, and angiogenesis via targeting miR-29b in
trophoblast cells. Hum Genomics. 13:502019. View Article : Google Scholar
|
|
7
|
Terashima M, Ishimura A, Wanna-Udom S and
Suzuki T: MEG8 long noncoding RNA contributes to epigenetic
progression of the epithelial-mesenchymal transition of lung and
pancreatic cancer cells. J Biol Chem. 293:18016–18030. 2018.
View Article : Google Scholar
|
|
8
|
Lu YM, Wang Y, Liu SQ, Zhou MY and Guo YR:
Profile and validation of dysregulated long noncoding RNAs and
mRNAs in ovarian cancer. Oncol Rep. 40:2964–2976. 2018.
|
|
9
|
Zhang D, Cao C, Liu L and Wu D:
Up-regulation of LncRNA SNHG20 predicts poor prognosis in
hepatocellular carcinoma. J Cancer. 7:608–617. 2016. View Article : Google Scholar
|
|
10
|
He S, Zhao Y, Wang X, Deng Y, Wan Z, Yao S
and Shen H: Up-regulation of long non-coding RNA SNHG20 promotes
ovarian cancer progression via Wnt/β-catenin signaling. Biosci Rep.
38:BSR201706812018. View Article : Google Scholar
|
|
11
|
Trionfini P and Benigni A: MicroRNAs as
master regulators of glomerular function in health and disease. J
Am Soc Nephrol. 28:1686–1696. 2017. View Article : Google Scholar
|
|
12
|
Wang Y, Kim S and Kim IM: Regulation of
metastasis by microRNAs in ovarian cancer. Front Oncol. 4:1432014.
View Article : Google Scholar
|
|
13
|
Sun J, Qiao Y, Song T and Wang H: MiR-495
suppresses cell proliferation by directly targeting HMGA2 in lung
cancer. Mol Med Rep. 19:1463–1470. 2019.
|
|
14
|
Liu C, Cai L and Li H: miR185 regulates
the growth of osteosarcoma cells via targeting Hexokinase 2. Mol
Med Rep. 20:2774–2782. 2019.
|
|
15
|
Luo Y, Hao T, Zhang J, Zhang M, Sun P and
Wu L: MicroRNA-592 suppresses the malignant phenotypes of thyroid
cancer by regulating lncRNA NEAT1 and downregulating NOVA1. Int J
Mol Med. 44:1172–1182. 2019.
|
|
16
|
Wang C, Zhang W, Xing S, Wang Z, Wang J
and Qu J: MiR-342-3p inhibits cell migration and invasion through
suppressing forkhead box protein Q1 in ovarian carcinoma.
Anticancer Drugs. 30:917–924. 2019. View Article : Google Scholar
|
|
17
|
Zhang Y, Shi B, Chen J, Hu L and Zhao C:
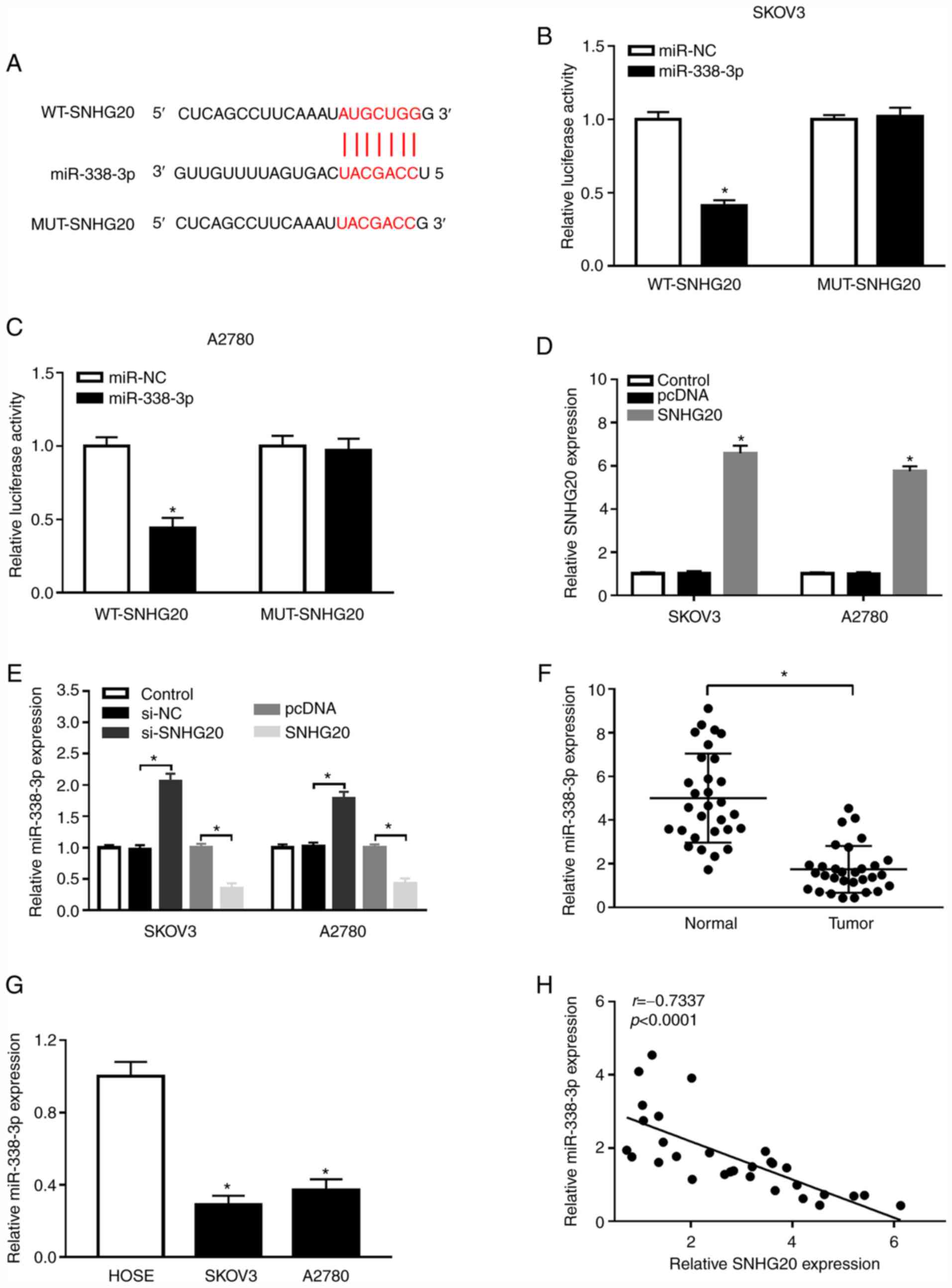
MiR-338-3p targets pyruvate kinase M2 and affects cell
proliferation and metabolism of ovarian cancer. Am J Transl Res.
8:3266–3273. 2016.
|
|
18
|
Liu X, Wen J, Wang H and Wang Y: Long
non-coding RNA LINC00460 promotes epithelial ovarian cancer
progression by regulating microRNA-338-3p. Biomed Pharmacother.
108:1022–1028. 2018. View Article : Google Scholar
|
|
19
|
Yip KW and Reed JC: Bcl-2 family proteins
and cancer. Oncogene. 27:6398–6406. 2008. View Article : Google Scholar
|
|
20
|
Zhang S, Zhang M, Jing Y, Yin X, Ma P,
Zhang Z, Wang X, Di W and Zhuang G: Deubiquitinase USP13 dictates
MCL1 stability and sensitivity to BH3 mimetic inhibitors. Nat
Commun. 9:2152018. View Article : Google Scholar
|
|
21
|
Su J, Ruan S, Dai S, Mi J, Chen W and
Jiang S: NF1 regulates apoptosis in ovarian cancer cells by
targeting MCL1 via miR-142-5p. Pharmacogenomics. 20:155–165. 2019.
View Article : Google Scholar
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
23
|
Sun Q, Li Q and Xie F: LncRNA-MALAT1
regulates proliferation and apoptosis of ovarian cancer cells by
targeting miR-503-5p. Onco Targets Ther. 12:6297–6307. 2019.
View Article : Google Scholar
|
|
24
|
Wang Y, Huang Y, Liu H, Su D, Luo F and
Zhou F: Long noncoding RNA CDKN2B-AS1 interacts with miR-411-3p to
regulate ovarian cancer in vitro and in vivo through
HIF-1a/VEGF/P38 pathway. Biochem Biophys Res Commun. 514:44–50.
2019. View Article : Google Scholar
|
|
25
|
You Q, Shi HY, Gong CF, Tian XY and Li S:
Long non-coding RNA DLX6-AS1 acts as an oncogene by targeting
miR-613 in ovarian cancer. Eur Rev Med Pharmacol Sci. 23:6429–6435.
2019.
|
|
26
|
Wu X, Xiao Y, Zhou Y, Zhou Z and Yan W:
lncRNA SNHG20 promotes prostate cancer migration and invasion via
targeting the miR-6516-5p/SCGB2A1 axis. Am J Transl Res.
11:5162–5169. 2019.
|
|
27
|
Liu J, Cheng LG and Li HG: LncRNA SNHG20
promoted the proliferation of glioma cells via sponging miR-4486 to
regulate the MDM2-p53 pathway. Eur Rev Med Pharmacol Sci.
23:5323–5331. 2019.
|
|
28
|
Gao XF, He HQ, Zhu XB, Xie SL and Cao Y:
LncRNA SNHG20 promotes tumorigenesis and cancer stemness in
glioblastoma via activating PI3K/Akt/mTOR signaling pathway.
Neoplasma. 66:532–542. 2019. View Article : Google Scholar
|
|
29
|
Sun C, Sun Y and Zhang E: Long non-coding
RNA SNHG20 promotes nasopharyngeal carcinoma cell migration and
invasion by upregulating TGF-β1. Exp Ther Med. 16:4967–4974.
2018.
|
|
30
|
Zhao W, Ma X, Liu L, Chen Q, Liu Z, Zhang
Z, Ma S, Wang Z, Li H, Wang Z and Wu J: SNHG20: A vital lncRNA in
multiple human cancers. J Cell Physiol. Jan 15–2019.(Epub ahead of
print).
|
|
31
|
Wang D, Dai J, Hou S and Qian Y: LncRNA
SNHG20 predicts a poor prognosis and promotes cell progression in
epithelial ovarian cancer. Biosci Rep. 39:BSR201821862019.
View Article : Google Scholar
|
|
32
|
Chen Z, Chen X, Chen P, Yu S, Nie F, Lu B,
Zhang T, Zhou Y, Chen Q, Wei C, et al: Long non-coding RNA SNHG20
promotes non-small cell lung cancer cell proliferation and
migration by epigenetically silencing of P21 expression. Cell Death
Dis. 8:e30922017. View Article : Google Scholar
|
|
33
|
Liu J, Liu L, Wan JX and Song Y: Long
noncoding RNA SNHG20 promotes gastric cancer progression by
inhibiting p21 expression and regulating the GSK-3β/β-catenin
signaling pathway. Oncotarget. 8:80700–80708. 2017. View Article : Google Scholar
|
|
34
|
Guo LP, Zhang ZJ, Li RT, Li HY and Cui YQ:
Influences of LncRNA SNHG20 on proliferation and apoptosis of
glioma cells through regulating the PTEN/PI3K/AKT signaling
pathway. Eur Rev Med Pharmacol Sci. 23:253–261. 2019.
|
|
35
|
Guo H, Yang S, Li S, Yan M, Li L and Zhang
H: LncRNA SNHG20 promotes cell proliferation and invasion via
miR-140-5p-ADAM10 axis in cervical cancer. Biomed Pharmacother.
102:749–757. 2018. View Article : Google Scholar
|
|
36
|
Sun F, Liang W, Tang K, Hong M and Qian J:
Profiling the lncRNA-miRNA-mRNA ceRNA network to reveal potential
crosstalk between inflammatory bowel disease and colorectal cancer.
PeerJ. 7:e74512019. View Article : Google Scholar
|
|
37
|
Wang X, Lan Z, He J, Lai Q, Yao X, Li Q,
Liu Y, Lai H, Gu C, Yan Q, et al: LncRNA SNHG6 promotes
chemoresistance through ULK1-induced autophagy by sponging
miR-26a-5p in colorectal cancer cells. Cancer Cell Int. 19:2342019.
View Article : Google Scholar
|
|
38
|
Liu X, Zhu Q, Guo Y, Xiao Z, Hu L and Xu
Q: LncRNA LINC00689 promotes the growth, metastasis and glycolysis
of glioma cells by targeting miR-338-3p/PKM2 axis. Biomed
Pharmacother. 117:1090692019. View Article : Google Scholar
|
|
39
|
Li M, Bian Z, Jin G, Zhang J, Yao S, Feng
Y, Wang X, Yin Y, Fei B, You Q and Huang Z: LncRNA-SNHG15 enhances
cell proliferation in colorectal cancer by inhibiting miR-338-3p.
Cancer Med. 8:2404–2413. 2019. View Article : Google Scholar
|
|
40
|
Zhang R, Shi H, Ren F, Liu Z, Ji P, Zhang
W and Wang W: Down-regulation of miR-338-3p and Up-regulation of
MACC1 indicated poor prognosis of epithelial ovarian cancer
patients. J Cancer. 10:1385–1392. 2019. View Article : Google Scholar
|
|
41
|
Huang Y, Luo H, Li F, Yang Y, Ou G, Ye X
and Li N: LINC00152 down-regulated miR-193a-3p to enhance MCL1
expression and promote gastric cancer cells proliferation. Biosci
Rep. 38:BSR201716072018. View Article : Google Scholar
|
|
42
|
Suzuki J, Nakajima W, Suzuki H, Asano Y
and Tanaka N: Chaperone-mediated autophagy promotes lung cancer
cell survival through selective stabilization of the pro-survival
protein, MCL1. Biochem Biophys Res Commun. 482:1334–1340. 2017.
View Article : Google Scholar
|
|
43
|
Zhou C, Li G, Zhou J, Han N, Liu Z and Yin
J: miR-107 activates ATR/Chk1 pathway and suppress cervical cancer
invasion by targeting MCL1. PLoS One. 9:e1118602014. View Article : Google Scholar
|
|
44
|
Xu W, Li Z, Zhu X, Xu R and Xu Y: miR-29
family inhibits resistance to methotrexate and promotes cell
apoptosis by targeting COL3A1 and MCL1 in osteosarcoma. Med Sci
Monit. 24:8812–8821. 2018. View Article : Google Scholar
|