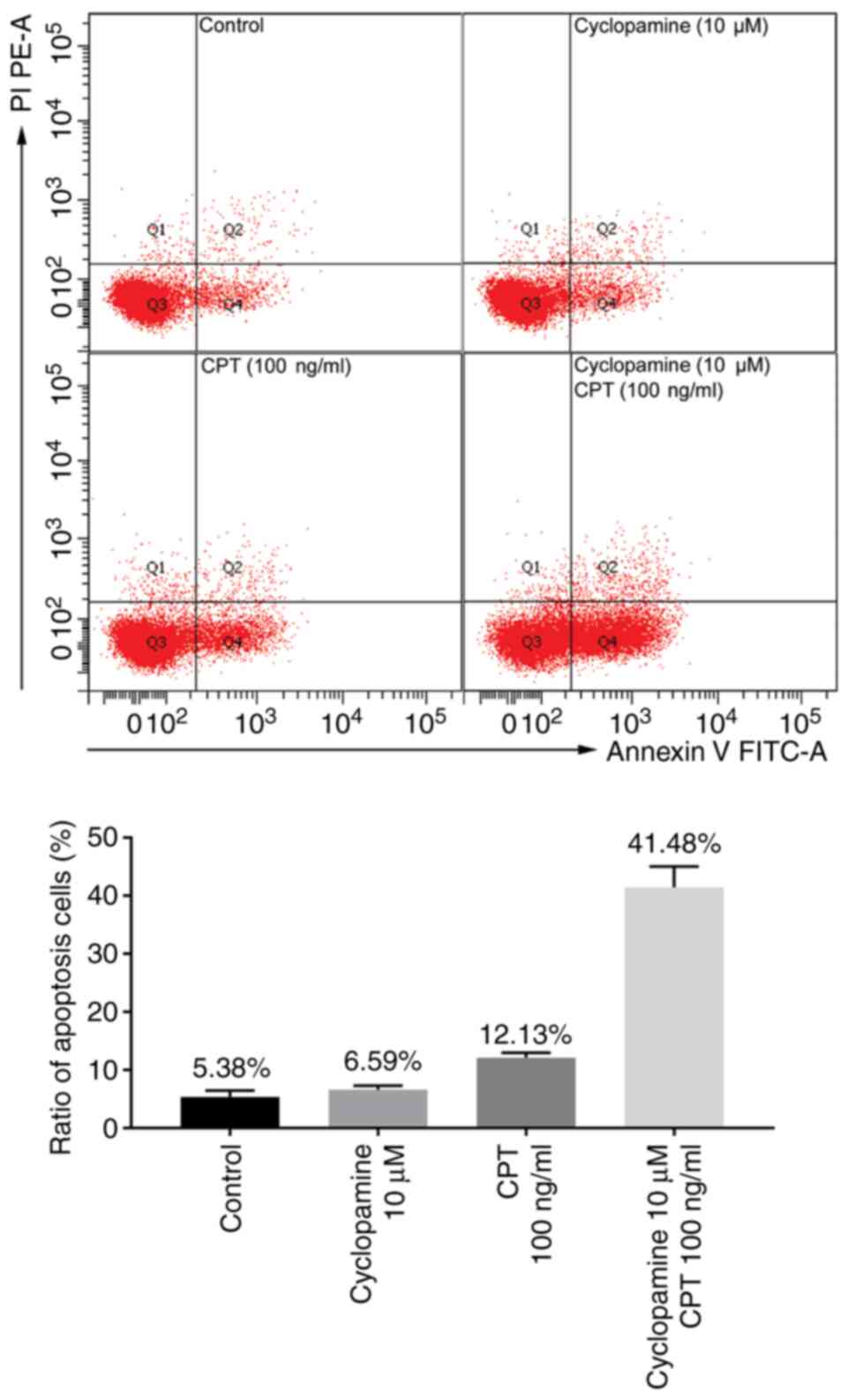
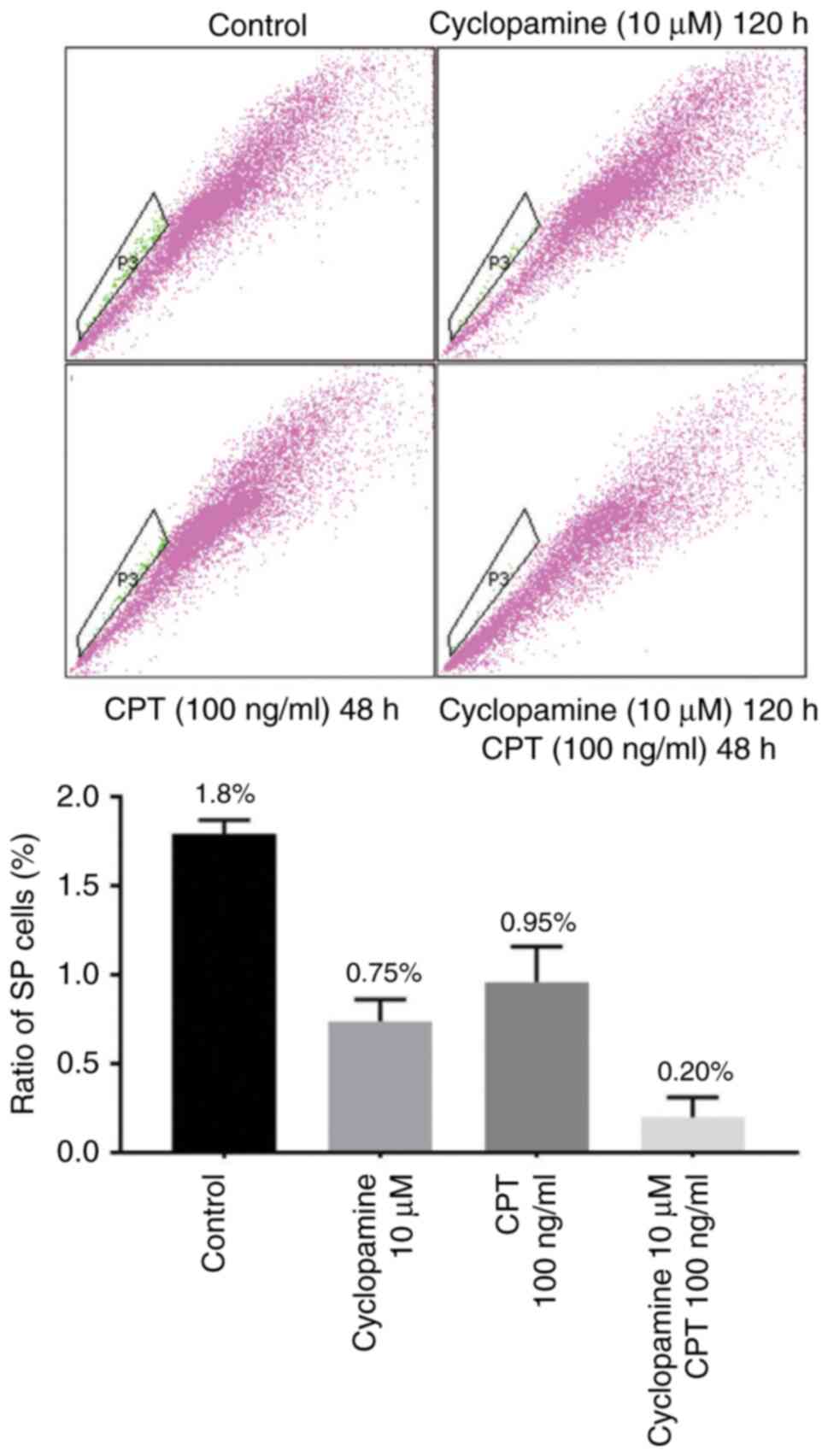
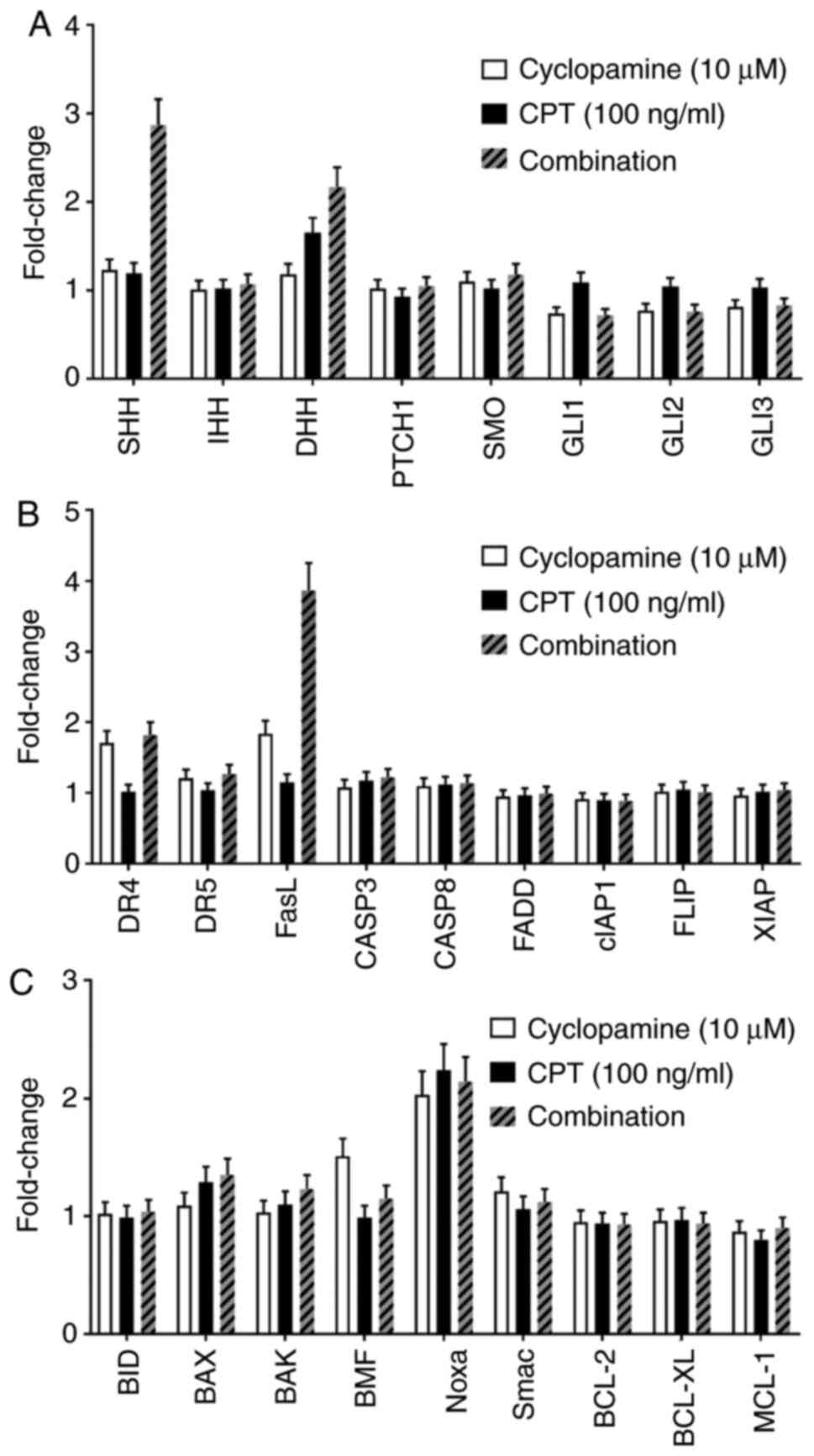
|
1
|
Martin T and Huff CA: Multiple myeloma:
Current advances and future directions. Clin Lymphoma Myeloma Leuk.
19:255–263. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ziogas DC, Dimopoulos MA and Kastritis E:
Prognostic factors for multiple myeloma in the era of novel
therapies. Expert Rev Hematol. 11:863–879. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ishida T: Therapeutic antibodies for
multiple myeloma. Jpn J Clin Oncol. 48:957–963. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Aljama MA, Sidiqi MH and Dingli D: Therapy
for relapsed multiple myeloma. Panminerva Med. 60:174–184. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gazitt Y: TRAIL is a potent inducer of
apoptosis in myeloma cells derived from multiple myeloma patients
and is not cytotoxic to hematopoietic stem cells. Leukemia.
13:1817–1824. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fang F, Wang AP and Yang SF: Antitumor
activity of a novel recombinant mutant human tumor necrosis
factor-related apoptosis-inducing ligand. Acta Pharmacol Sin.
26:1373–1381. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Geng C, Hou J, Zhao Y, Ke X, Wang Z, Qiu
L, Xi H, Wang F, Wei N, Liu Y, et al: A multicenter, open-label
phase II study of recombinant CPT (Circularly Permuted TRAIL) plus
thalidomide in patients with relapsed and refractory multiple
myeloma. Am J Hematol. 89:1037–1042. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Leng Y, Qiu L, Hou J, Zhao Y, Zhang X,
Yang S, Xi H, Huang Z, Pan L and Chen W: Phase II open-label study
of recombinant circularly permuted TRAIL as a single-agent
treatment for relapsed or refractory multiple myeloma. Chin J
Cancer. 35:862016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fan C, Wang Y, Liu Z, Sun Y, Wang X, Wei G
and Wei J: Metformin exerts anticancer effects through the
inhibition of the Sonic hedgehog signaling pathway in breast
cancer. Int J Mol Med. 36:204–214. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gonnissen A, Isebaert S and Haustermans K:
Hedgehog signaling in prostate cancer and its therapeutic
implication. Int J Mol Sci. 14:13979–14007. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang R, Mondal G, Wen D and Mahato RI:
Combination therapy of paclitaxel and cyclopamine polymer-drug
conjugates to treat advanced prostate cancer. Nanomedicine.
13:391–401. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Iovine V, Mori M, Calcaterra A, Berardozzi
S and Botta B: One hundred faces of cyclopamine. Curr Pharm Des.
22:1658–1681. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hassanzadeh A, Farshdousti Hagh M, Alivand
MR, Akbari AAM, Shams Asenjan K, Saraei R and Solali S:
Down-regulation of intracellular anti-apoptotic proteins,
particularly c-FLIP by therapeutic agents; the novel view to
overcome resistance to TRAIL. J Cell Physiol. 233:6470–6485. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yuan X, Gajan A, Chu Q, Xiong H, Wu K and
Wu GS: Developing TRAIL/TRAIL death receptor-based cancer
therapies. Cancer Metastasis Rev. 37:733–748. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Testa U: TRAIL/TRAIL-R in hematologic
malignancies. J Cell Biochem. 110:21–34. 2010.PubMed/NCBI
|
|
17
|
Buckle CH, Neville-Webbe HL, Croucher PI
and Lawson MA: Targeting RANK/RANKL in the treatment of solid
tumours and myeloma. Curr Pharm Des. 16:1272–1283. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Arhoma A, Chantry AD, Haywood-Small SL and
Cross NA: SAHA-induced TRAIL-sensitisation of multiple myeloma
cells is enhanced in 3D cell culture. Exp Cell Res. 360:226–235.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Leng Y, Hou J, Jin J, Zhang M, Ke X, Jiang
B, Pan L, Yang L, Zhou F, Wang J, et al: Circularly permuted TRAIL
plus thalidomide and dexamethasone versus thalidomide and
dexamethasone for relapsed/refractory multiple myeloma: A phase 2
study. Cancer Chemother Pharmacol. 79:1141–1149. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gao M, Kong Y, Yang G, Gao L and Shi J:
Multiple myeloma cancer stem cells. Oncotarget. 7:35466–35477.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shi F, Li M, Wang J, Wu D, Pan M, Guo M
and Dou J: Induction of multiple myeloma cancer stem cell apoptosis
using conjugated anti-ABCG2 antibody with epirubicin-loaded
microbubbles. Stem Cell Res Ther. 9:1442018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shi F, Li M, Wu S, Yang F, Di W, Pan M,
Zhao F, Luo S, Gu N and Dou J: Enhancing the anti-multiple myeloma
efficiency in a cancer stem cell xenograft model by conjugating the
ABCG2 antibody with microbubbles for a targeted delivery of
ultrasound mediated epirubicin. Biochem Pharmacol. 132:18–28. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu Z, Xu J, He J, Zheng Y, Li H, Lu Y,
Qian J, Lin P, Weber DM, Yang J and Yi Q: A critical role of
autocrine sonic hedgehog signaling in human CD138+ myeloma cell
survival and drug resistance. Blood. 124:2061–2071. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang J, Li Y, Sun W, Liu J and Chen W:
Synergistic effects of rmhTRAIL and 17-AAG on the proliferation and
apoptosis of multiple myeloma cells. Hematology. 23:620–625. 2018.
View Article : Google Scholar : PubMed/NCBI
|