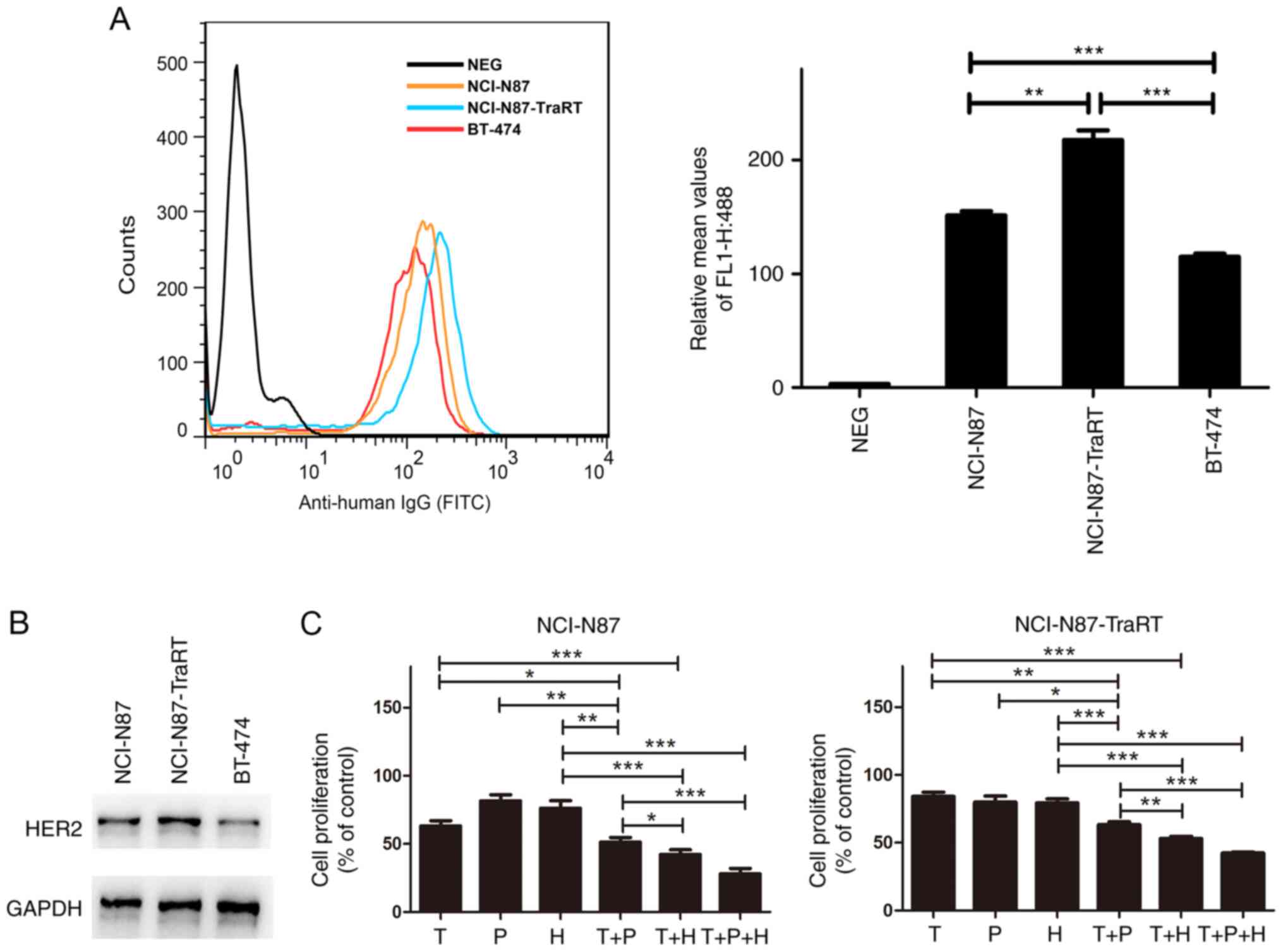
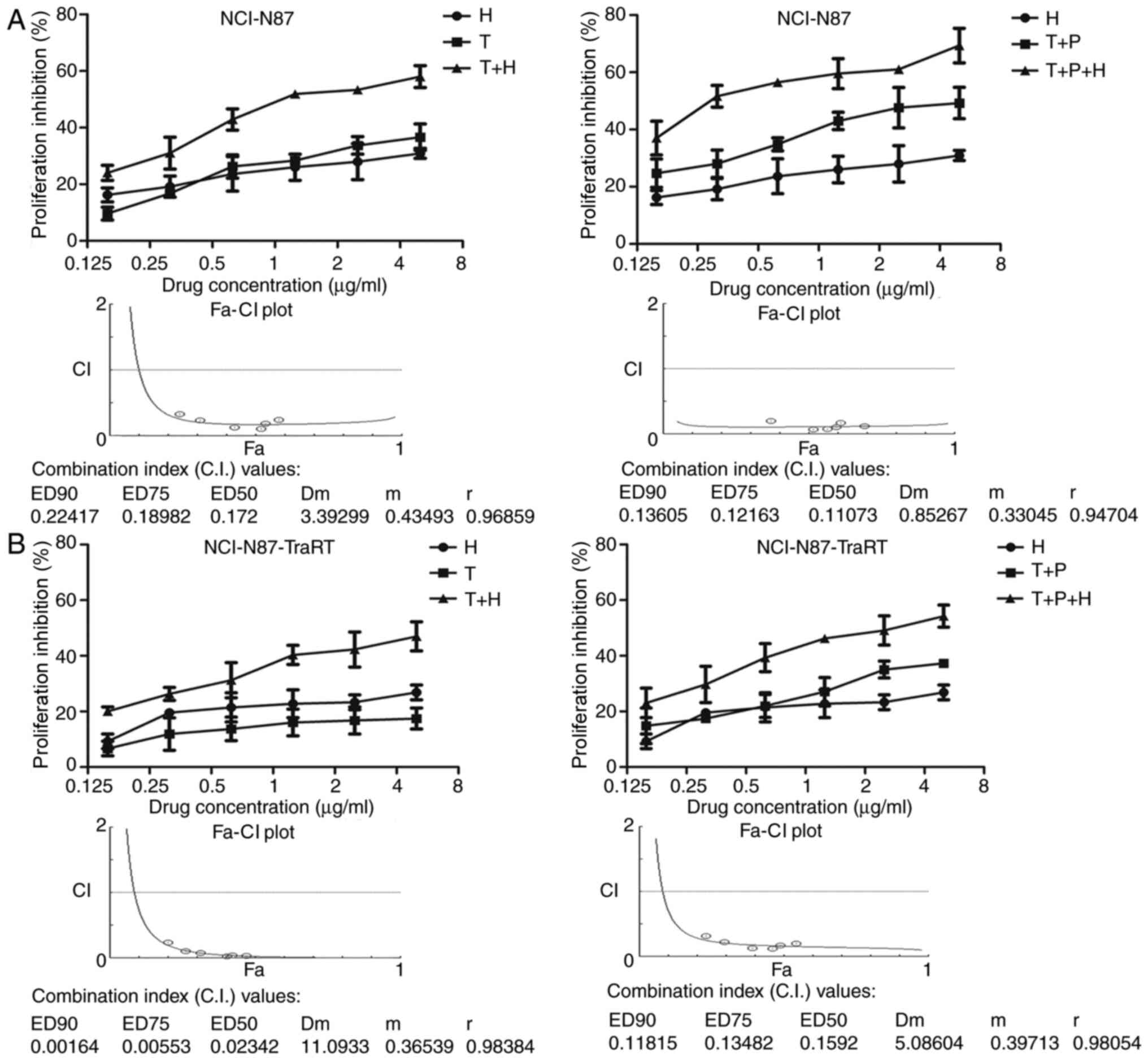
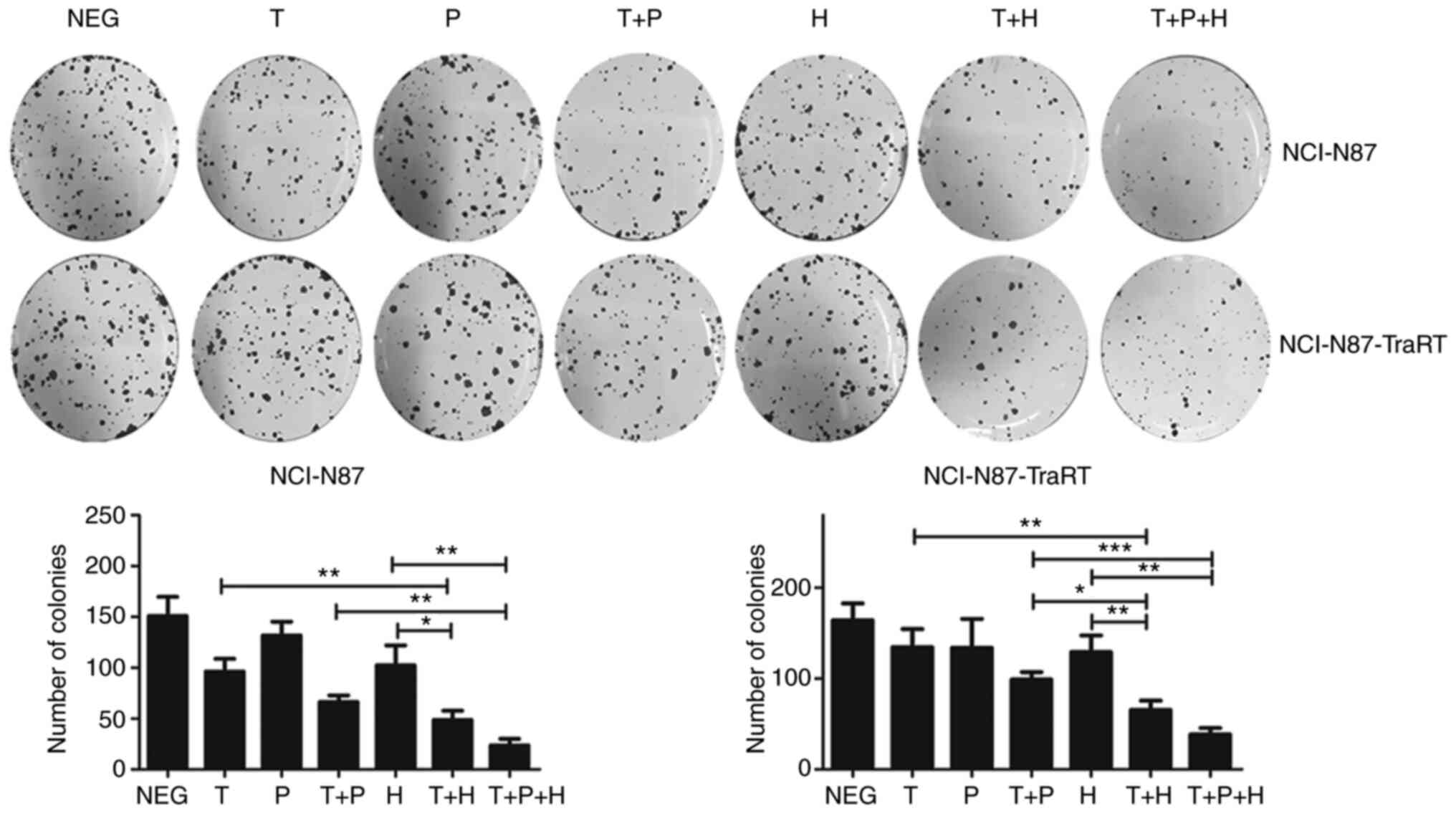
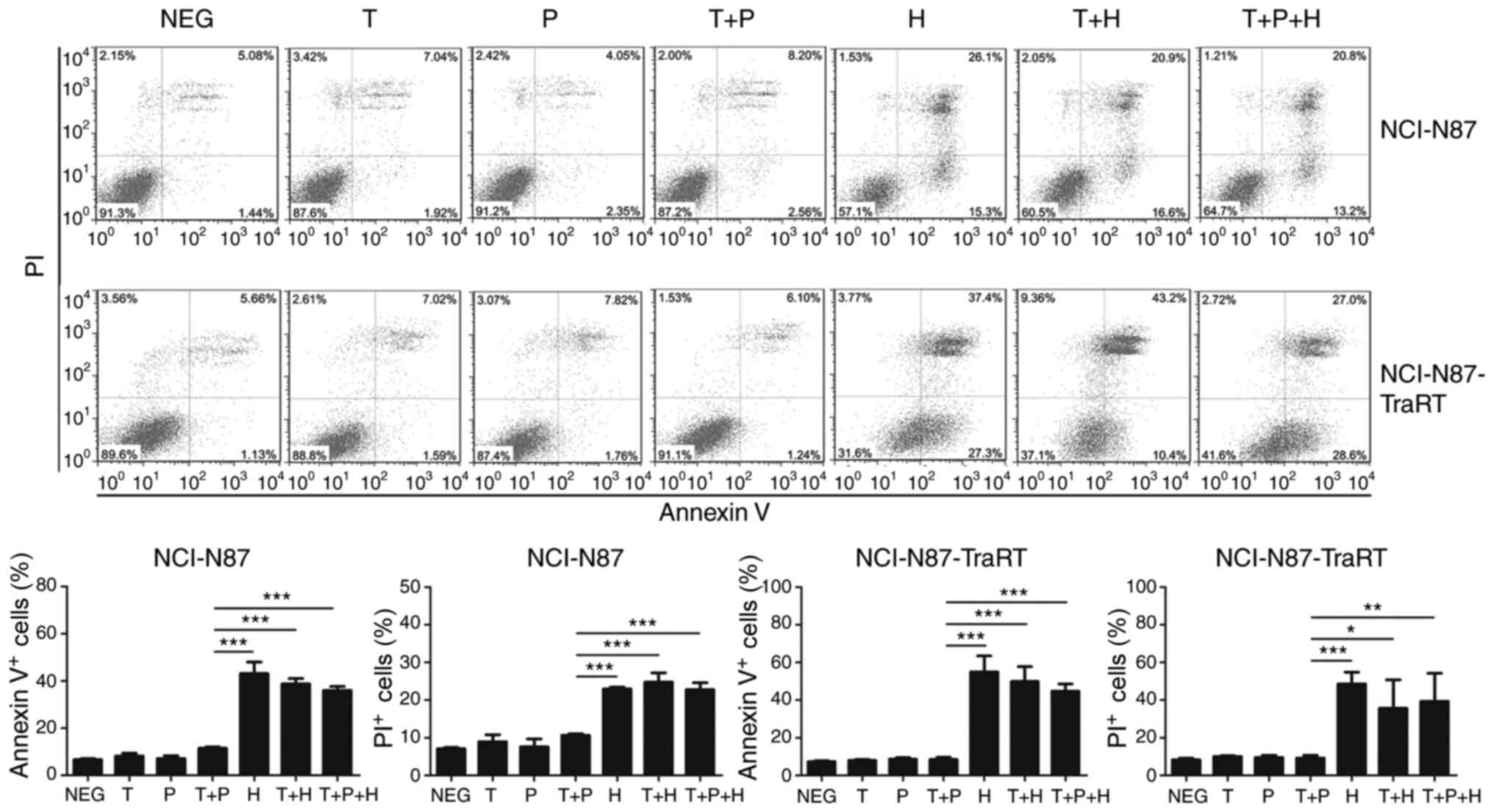
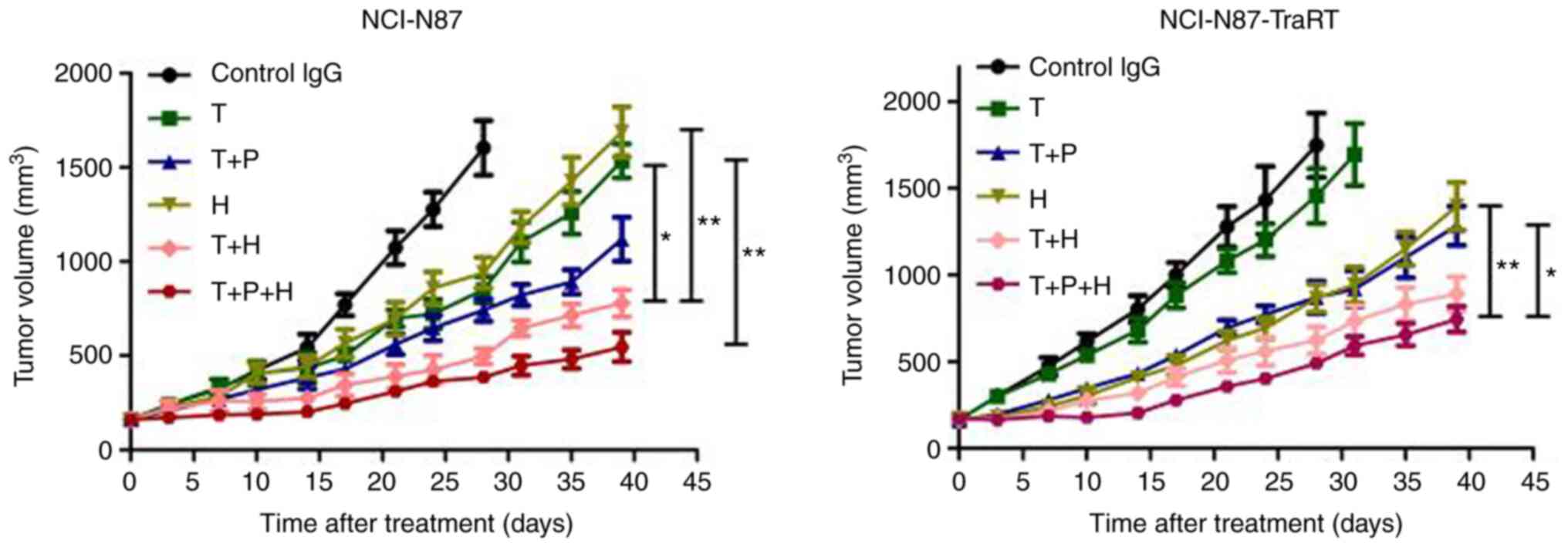
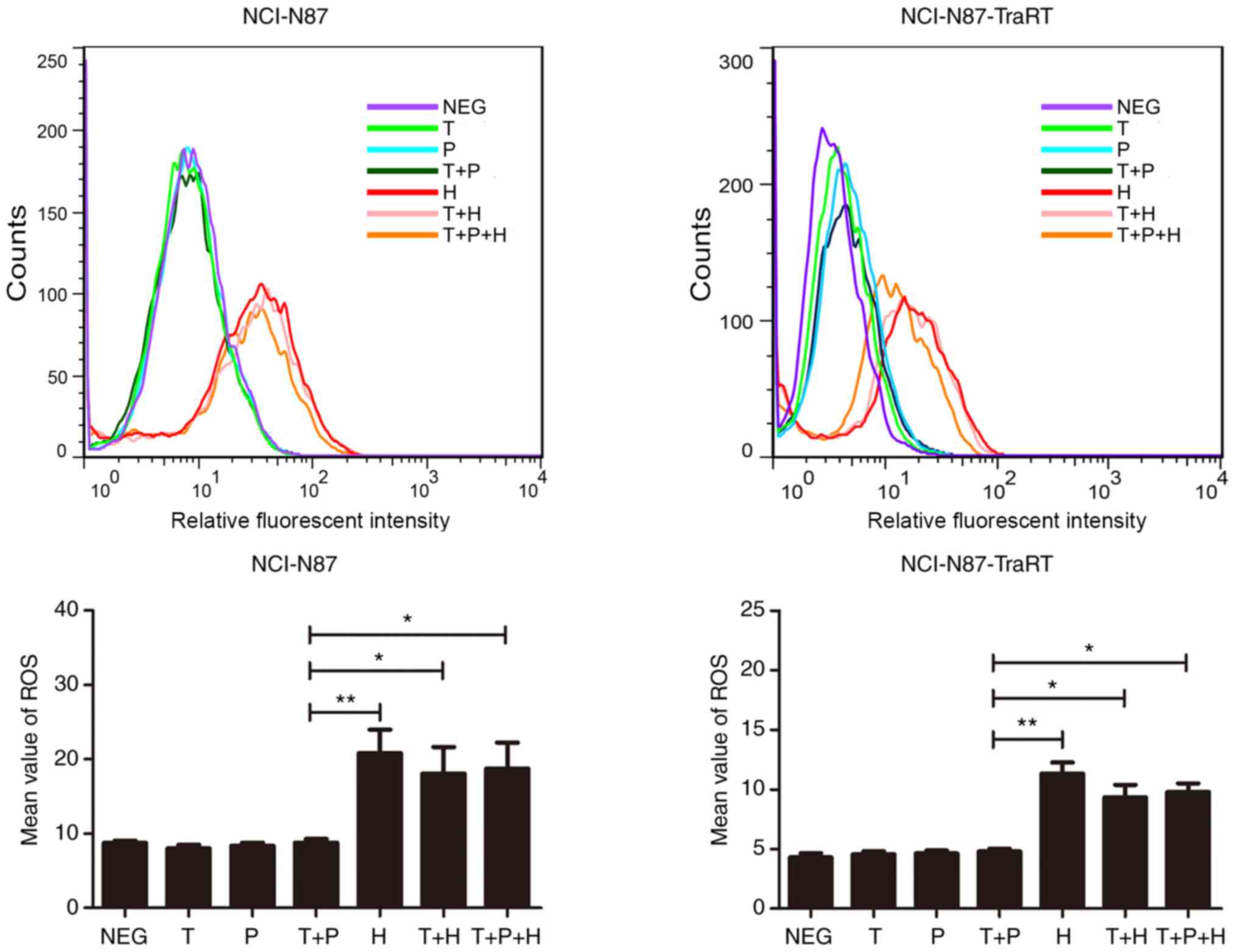
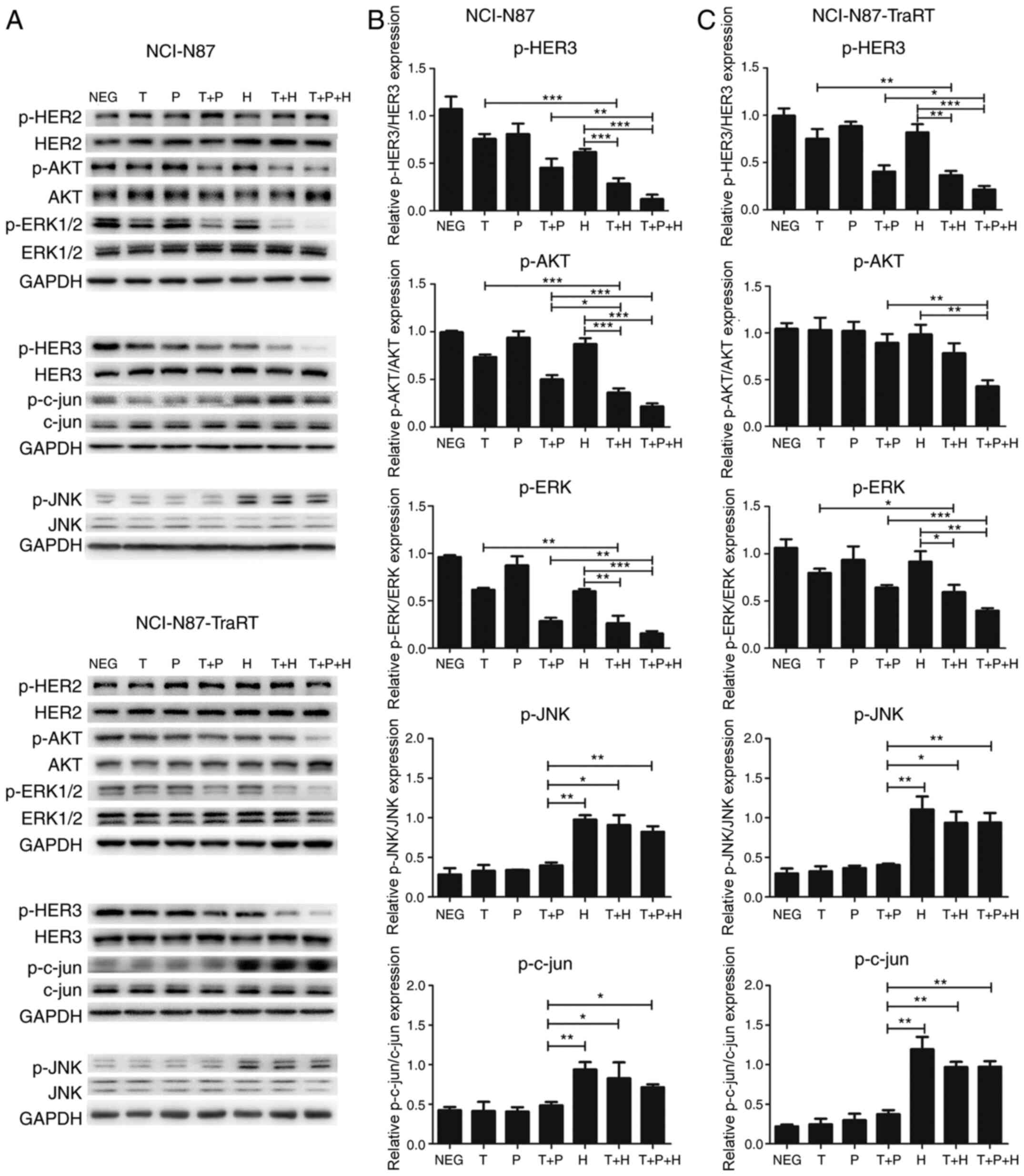
|
1
|
Slamon DJ, Clark GM, Wong SG, Levin WJ,
Ullrich A and McGuire WL: Human breast cancer: Correlation of
relapse and survival with amplification of the HER-2/neu oncogene.
Science. 235:177–182. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Slamon DJ, Godolphin W, Jones LA, Holt JA,
Wong SG, Keith DE, Levin WJ, Stuart SG, Udove J, Ullrich A and
Press MF: Studies of the HER-2/neu proto-oncogene in human breast
and ovarian cancer. Science. 244:707–712. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Thiel A and Ristimaki A: Targeted therapy
in gastric cancer. APMIS. 123:365–372. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bang YJ, Van Cutsem E, Feyereislova A,
Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T,
et al: Trastuzumab in combination with chemotherapy versus
chemotherapy alone for treatment of HER2-positive advanced gastric
or gastro-oesophageal junction cancer (ToGA): A phase 3,
open-label, randomised controlled trial. Lancet. 376:687–697. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Smyth EC, Verheij M, Allum W, Cunningham
D, Cervantes A and Arnold D: Gastric cancer: ESMO clinical practice
guidelines for diagnosis, treatment and follow-up. Ann Oncol.
27:v38–v49. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vogel CL, Cobleigh MA, Tripathy D, Gutheil
JC, Harris LN, Fehrenbacher L, Slamon DJ, Murphy M, Novotny WF,
Burchmore M, et al: Efficacy and safety of trastuzumab as a single
agent in first-line treatment of HER2-overexpressing metastatic
breast cancer. J Clin Oncol. 20:719–726. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nahta R and Esteva FJ: HER2 therapy:
Molecular mechanisms of trastuzumab resistance. Breast Cancer Res.
8:2152006. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nahta R, Yu D, Hung MC, Hortobagyi GN and
Esteva FJ: Mechanisms of disease: Understanding resistance to
HER2-targeted therapy in human breast cancer. Nat Clin Pract Oncol.
3:269–280. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Baselga J, Gelmon KA, Verma S, Wardley A,
Conte P, Miles D, Bianchi G, Cortes J, McNally VA, Ross GA, et al:
Phase II trial of pertuzumab and trastuzumab in patients with human
epidermal growth factor receptor 2-positive metastatic breast
cancer that progressed during prior trastuzumab therapy. J Clin
Oncol. 28:1138–1144. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cortes J, Fumoleau P, Bianchi GV, Petrella
TM, Gelmon K, Pivot X, Verma S, Albanell J, Conte P, Lluch A, et
al: Pertuzumab monotherapy after trastuzumab-based treatment and
subsequent reintroduction of trastuzumab: Activity and tolerability
in patients with advanced human epidermal growth factor receptor
2-positive breast cancer. J Clin Oncol. 30:1594–1600. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Blackwell KL, Burstein HJ, Storniolo AM,
Rugo H, Sledge G, Koehler M, Ellis C, Casey M, Vukelja S, Bischoff
J, et al: Randomized study of Lapatinib alone or in combination
with trastuzumab in women with ErbB2-positive,
trastuzumab-refractory metastatic breast cancer. J Clin Oncol.
28:1124–1130. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
von Minckwitz G, du Bois A, Schmidt M,
Maass N, Cufer T, de Jongh FE, Maartense E, Zielinski C, Kaufmann
M, Bauer W, et al: Trastuzumab beyond progression in human
epidermal growth factor receptor 2-positive advanced breast cancer:
A german breast group 26/breast international group 03–05 study. J
Clin Oncol. 27:1999–2006. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang C, Wang L, Yu X, Zhang Y, Meng Y,
Wang H, Yang Y, Gao J, Wei H, Zhao J, et al: Combating acquired
resistance to trastuzumab by an anti-ErbB2 fully human antibody.
Oncotarget. 8:42742–42751. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Agus DB, Akita RW, Fox WD, Lewis GD,
Higgins B, Pisacane PI, Lofgren JA, Tindell C, Evans DP, Maiese K,
et al: Targeting ligand-activated ErbB2 signaling inhibits breast
and prostate tumor growth. Cancer Cell. 2:127–137. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Junttila TT, Akita RW, Parsons K, Fields
C, Lewis Phillips GD, Friedman LS, Sampath D and Sliwkowski MX:
Ligand-independent HER2/HER3/PI3K complex is disrupted by
trastuzumab and is effectively inhibited by the PI3K inhibitor
GDC-0941. Cancer Cell. 15:429–440. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nahta R, Hung MC and Esteva FJ: The
HER-2-targeting antibodies trastuzumab and pertuzumab
synergistically inhibit the survival of breast cancer cells. Cancer
Res. 64:2343–2346. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Scheuer W, Friess T, Burtscher H,
Bossenmaier B, Endl J and Hasmann M: Strongly enhanced antitumor
activity of trastuzumab and pertuzumab combination treatment on
HER2-positive human xenograft tumor models. Cancer Res.
69:9330–9336. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lu Q, Wang L, Zhang Y, Yu X, Wang C, Wang
H, Yang Y, Chong X, Xia T, Meng Y, et al: An anti-ErbB2 fully human
antibody circumvents trastuzumab resistance. Oncotarget.
7:67129–76141. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li B, Meng Y, Zheng L, Zhang X, Tong Q,
Tan W, Hu S, Li H, Chen Y, Song J, et al: Bispecific antibody to
ErbB2 overcomes trastuzumab resistance through comprehensive
blockade of ErbB2 heterodimerization. Cancer Res. 73:6471–6483.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Adams CW, Allison DE, Flagella K, Presta
L, Clarke J, Dybdal N, McKeever K and Sliwkowski MX: Humanization
of a recombinant monoclonal antibody to produce a therapeutic HER
dimerization inhibitor, pertuzumab. Cancer Immunol Immunother.
55:717–727. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li B, Shi S, Qian W, Zhao L, Zhang D, Hou
S, Zheng L, Dai J, Zhao J, Wang H and Guo Y: Development of novel
tetravalent anti-CD20 antibodies with potent antitumor activity.
Cancer Res. 68:2400–2408. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li B, Zhao L, Guo H, Wang C, Zhang X, Wu
L, Chen L, Tong Q, Qian W, Wang H and Guo Y: Characterization of a
rituximab variant with potent antitumor activity against
rituximab-resistant B-cell lymphoma. Blood. 114:5007–5015. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang X, Chen J, Weng Z, Li Q, Zhao L, Yu
N, Deng L, Xu W, Yang Y, Zhu Z, et al: A new anti-HER2 antibody
that enhances the anti-tumor efficacy of trastuzumab and pertuzumab
with a distinct mechanism of action. Mol Immunol. 119:48–58. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chou TC: Drug combination studies and
their synergy quantification using the Chou-Talalay method. Cancer
Res. 70:440–446. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yoriko YK, Sei S, Naoki H and Kaori F:
Enhanced antitumor activity of trastuzumab emtansine (T-DM1) in
combination with pertuzumab in a HER2-positive gastric cancer
model. Oncol Rep. 30:1087–1093. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Stanley A, Ashrafi GH, Seddon AM and
Modjtahedi H: Synergistic effects of various Her inhibitors in
combination with IGF-1R, C-MET and Src targeting agents in breast
cancer cell lines. Sci Rep. 7:39642017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sampera A, Sánchez-Martín FJ, Arpí O, Visa
L, Iglesias M, Menéndez S, Gaye É, Dalmases A, Clavé S,
Gelabert-Baldrich M, et al: HER-family ligands promote acquired
resistance to trastuzumab in gastric cancer. Mol Cancer Ther.
18:2135–2145. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Derakhshani A, Rezaei Z, Safarpour H,
Sabri M, Mir A, Sanati MA, Vahidian F, Gholamiyan Moghadam A,
Aghadoukht A, Hajiasgharzadeh K and Baradaran B: Overcoming
trastuzumab resistance in HER2-positive breast cancer using
combination therapy. J Cell Physiol. 235:3142–3156. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ritter CA, Perez-Torres M, Rinehart C,
Guix M, Dugger T, Engelman JA and Arteaga CL: Human breast cancer
cells selected for resistance to trastuzumab in vivo overexpress
epidermal growth factor receptor and ErbB ligands and remain
dependent on the ErbB receptor network. Clin Cancer Res.
13:4909–4919. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kumar R: ErbB-dependent signaling as a
determinant of trastuzumab resistance. Clin Cancer Res.
13:4657–4659. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shitara K, Hara H, Yoshikawa T, Fujitani
K, Nishina T, Hosokawa A, Asakawa T, Kawakami S and Muro K:
Pertuzumab plus trastuzumab and chemotherapy for Japanese patients
with HER2-positive metastatic gastric or gastroesophageal junction
cancer: A subgroup analysis of the JACOB trial. Int J Clin Oncol.
25:301–311. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tabernero J, Hoff PM, Shen L, Ohtsu A,
Shah MA, Cheng K, Song C, Wu H, Eng-Wong J, Kim K and Kang YK:
Pertuzumab plus trastuzumab and chemotherapy for HER2-positive
metastatic gastric or gastro-oesophageal junction cancer (JACOB):
Final analysis of a double-blind, randomised, placebo-controlled
phase 3 study. Lancet Oncol. 19:1372–1384. 2018. View Article : Google Scholar : PubMed/NCBI
|