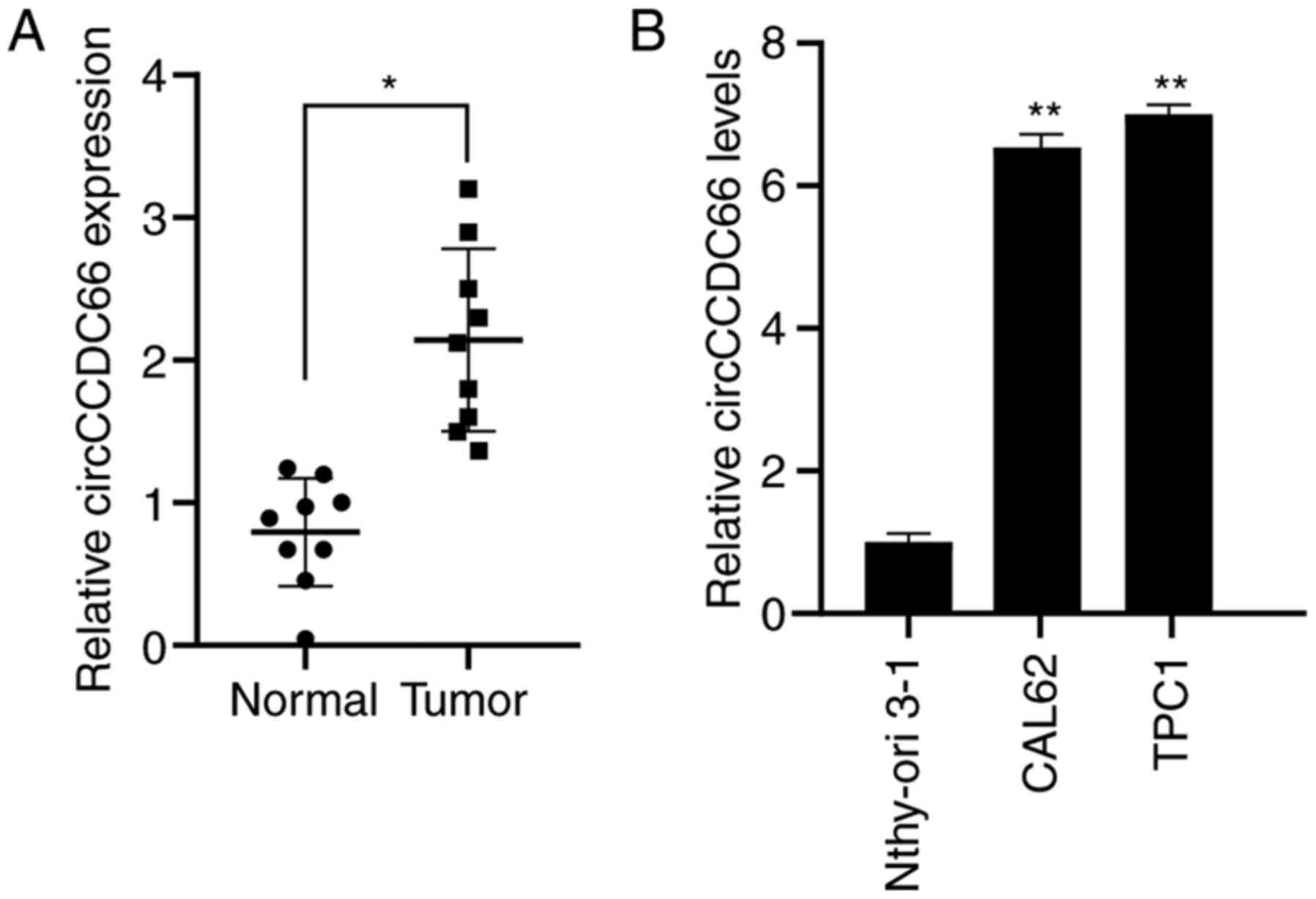
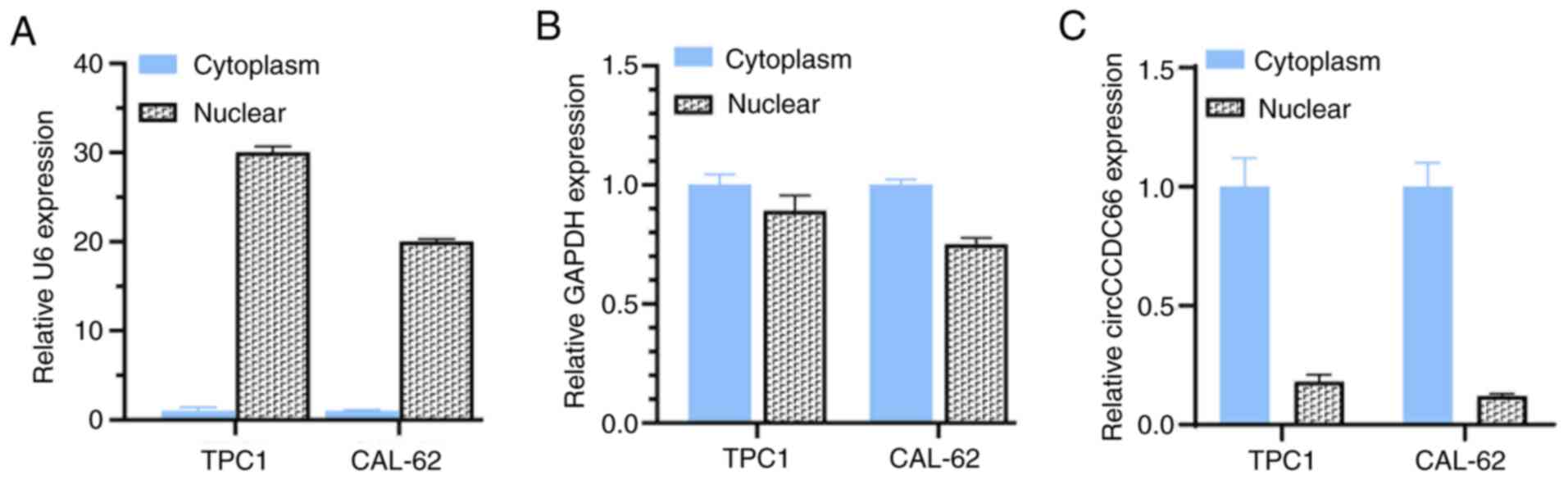
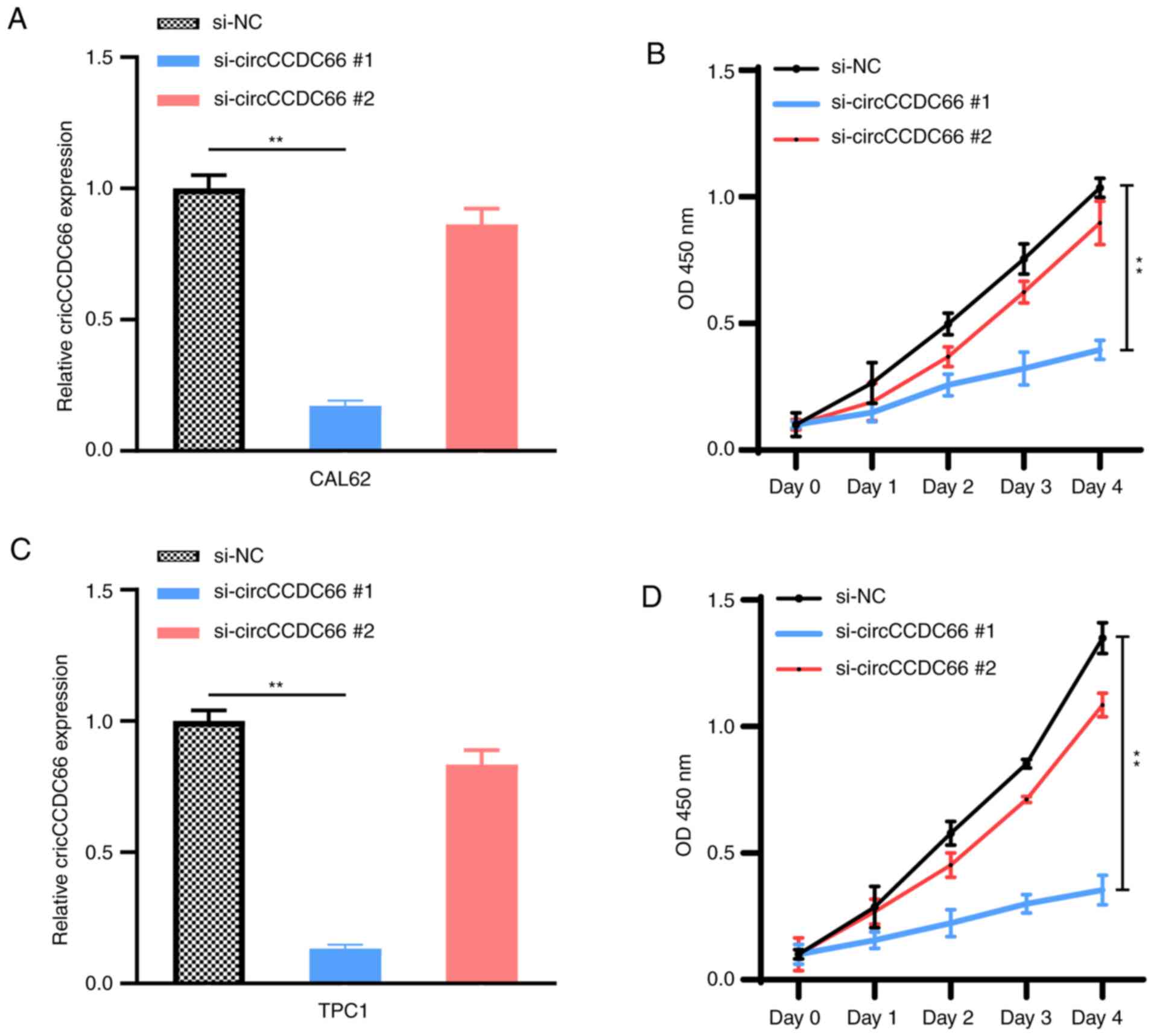
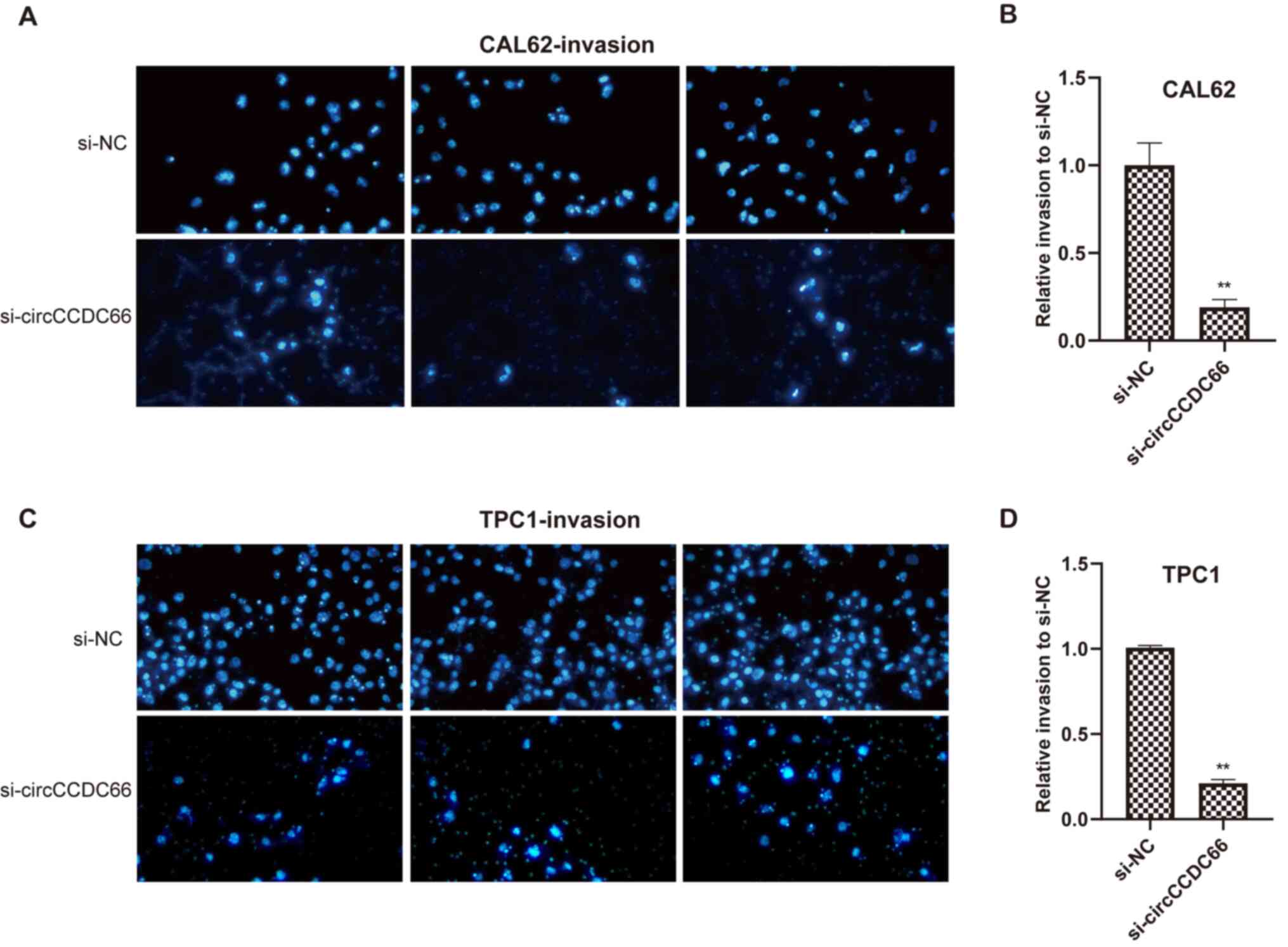
|
1
|
Zhang JW, Long KR, Wang X, LI MZ and Ma
JD: The research advance of circular RNA. Chin J Anim Veterinary
Sci,. 11:2151–2158. 2016.(In Chinese).
|
|
2
|
Cai H, Li Y, Niringiyumukiza JD, Su P and
Xiang W: Circular RNA involvement in aging: An emerging player with
great potential. Mech Ageing Dev. 178:16–24. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lu Q, Liu T, Feng H, Yang R, Zhao X, Chen
W, Jiang B, Qin H, Guo X, Liu M, et al: Circular RNA circSLC8A1
acts as a sponge of miR-130b/miR-494 in suppressing bladder cancer
progression via regulating PTEN. Mol Cancer. 18:1112019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Xu JZ, Shao CC, Wang XJ, Zhao X, Chen JQ,
Ouyang YX, Feng J, Zhang F, Huang WH, Ying Q, et al: circTADA2As
suppress breast cancer progression and metastasis via targeting
miR-203a-3p/SOCS3 axis. Cell Death Dis. 10:1752019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lu J, Wang YH, Yoon C, Huang XY, Xu Y, Xie
JW, Wang JB, Lin JX, Chen QY, Cao LL, et al: Circular RNA
circ-RanGAP1 regulates VEGFA expression by targeting miR-877-3p to
facilitate gastric cancer invasion and metastasis. Cancer Lett.
471:38–48. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gaffo E, Boldrin E, Dal Molin A, Bresolin
S, Bonizzato A, Trentin L, Frasson C, Debatin KM, Meyer LH, Te
Kronnie G and Bortoluzzi S: Circular RNA differential expression in
blood cell populations and exploration of circRNA deregulation in
pediatric acute lymphoblastic leukemia. Sci Rep. 9:146702019.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Xiong X, Zhu H and Chen X: Low expression
of long noncoding RNA CASC2 indicates a poor prognosis and promotes
tumorigenesis in thyroid carcinoma. Biomed Pharmacother.
93:391–397. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li S, Li Z, Guo F, Qin X, Liu B, Lei Z,
Song Z, Sun L, Zhang HT, You J and Zhou Q: MiR-223 regulates
migration and invasion by targeting Artemin in human esophageal
carcinoma. J Biomed Sci. 18:242011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Du WW, Yang W, Liu E, Yang Z, Dhaliwal P
and Yang BB: Foxo3 circular RNA retards cell cycle progression via
forming ternary complexes with p21 and CDK2. Nucleic Acids Res.
44:2846–2858. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Durante C, Grani G, Lamartina L, Filetti
S, Mandel SJ and Cooper DS: The diagnosis and management of Thyroid
Nodules: A review. JAMA. 319:914–924. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cong S, Fang LW, Bao HL, Feng YJ, Wang N,
Yin P, Li YC, Duan XN and Zhou MG: (Disease burden of thyroid
cancer in the Chinese population, in 1990 and 2013). Zhonghua Liu
Xing Bing Xue Za Zhi. 37:773–777. 2016.PubMed/NCBI
|
|
12
|
Schmidt B and Davies L: The rising
incidence of thyroid cancer: Contributions from healthcare practice
and biologic risk factors. Management of Differentiated Thyroid
Cancer. Springer; Cham: pp. 1–13. 2017, View Article : Google Scholar
|
|
13
|
Murugan AK, Dong J, Xie J and Xing M: MEK1
mutations, but not ERK2 mutations, occur in melanomas and colon
carcinomas, but none in thyroid carcinomas. Cell Cycle.
8:2122–2124. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gao Y, Wang J, Zheng Y, Zhang J, Chen S
and Zhao F: Comprehensive identification of internal structure and
alternative splicing events in circular RNAs. Nat Commun.
7:120602016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wei H, Pan L, Tao D and Li R: Circular RNA
circZFR contributes to papillary thyroid cancer cell proliferation
and invasion by sponging miR-1261 and facilitating C8orf4
expression. Biochem Biophys Res Commun. 503:56–61. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hsiao KY, Lin YC, Gupta SK, Chang N, Yen
L, Sun HS and Tsai SJ: Noncoding effects of circular RNA CCDC66
promote colon cancer growth and metastasis. Cancer Res.
77:2339–2350. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hong W, Yu S, Zhuang Y, Zhang Q, Wang J
and Gao X: SRCIN1 regulated by circCCDC66/miR-211 is upregulated
and promotes cell proliferation in non-small-cell lung cancer.
Biomed Res Int. 2020:53076412020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xiang D, Li Y and Lin Y: Circular RNA
circCCDC66 contributes to malignant phenotype of osteosarcoma by
Sponging miR-338-3p to upregulate the expression of PTP1B. Biomed
Res Int. 2020:46371092020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang M, Chen B, Ru Z and Cong L: CircRNA
circ-ITCH suppresses papillary thyroid cancer progression through
miR-22-3p/CBL/β-catenin pathway. Biochem Biophys Res Commun.
504:283–288. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu H, Liu Y, Cheng P, Wang C, Liu Y, Zhou
W, Xu Y and Ji G: CircRNA_0000392 promotes colorectal cancer
progression through the miR-193a-5p/PIK3R3/AKT axis. J Exp Clin
Cancer Res. 39:2832020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Schmittgen TD and Livak KJ: Analyzing
real-time PCR data by the comparative C(T) method. Nat Protoc.
3:1101–1108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nahm JH, Kim HM and Koo JS:
Glycolysis-related protein expression in thyroid cancer. Tumour
Biol. 39:10104283176959222017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang Y, Zhou X, Shan B, Han J, Wang F, Fan
X, Lv Y, Chang L and Liu W: Downregulation of microRNA-33a promotes
cyclin-dependent kinase 6, cyclin D1 and PIM1 expression and
gastric cancer cell proliferation. Mol Med Rep. 12:6491–6500. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kim Y, Williams KC, Gavin CT, Jardine E,
Chambers AF and Leong HS: Quantification of cancer cell
extravasation in vivo. Nat Protoc. 11:937–948. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang KW and Dong M: Role of circular RNAs
in gastric cancer: Recent advances and prospects. World J
Gastrointest Oncol. 11:459–469. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cui S, Qian Z, Chen Y, Li L, Li P and Ding
H: Screening of up- and downregulation of circRNAs in HBV-related
hepatocellular carcinoma by microarray. Oncol Lett. 15:423–432.
2018.PubMed/NCBI
|
|
27
|
Chen LL, Zhang ZJ, Yi ZB and Li JJ:
MicroRNA-211-5p suppresses tumour cell proliferation, invasion,
migration and metastasis in triple-negative breast cancer by
directly targeting SETBP1. Br J Cancer. 117:78–88. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang K, Jin W, Jin P, Fei X, Wang X and
Chen X: MiR-211-5p suppresses metastatic behavior by targeting
SNAI1 in Renal cancer. Mol Cancer Res. 15:448–456. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jiang G, Wen L, Deng W, Jian Z and Zheng
H: Regulatory role of miR-211-5p in hepatocellular carcinoma
metastasis by targeting ZEB2. Biomed Pharmacother. 90:806–812.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shan C, Kang HB, Elf S, Xie J, Gu TL,
Aguiar M, Lonning S, Hitosugi T, Chung TW, Arellano M, et al:
Tyr-94 phosphorylation inhibits pyruvate dehydrogenase phosphatase
1 and promotes tumor growth. J Biol Chem. 289:21413–21422. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sui W, Shi Z, Xue W, Ou M, Zhu Y, Chen J,
Lin H, Liu F and Dai Y: Circular RNA and gene expression profiles
in gastric cancer based on microarray chip technology. Oncol Rep.
37:1804–1814. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sun S, Zhao M, Han Y, Juanzi W, Peng W and
Liu J: PDK4 mRNA Expression in Breast Cancer and Its Relationship
with Prognosis. Cancer Res Prev Treat. 45:73–76. 2018.
|
|
33
|
Mazar J, Richardson A, Qi F, Lee B, Duran
A, Govindarajan S, Shelley J, Brill LM, Li JL, Han X, Moscat J and
Perera RJ: MicroRNA-211 modulates energy metabolism in human
melanoma cells by destabilizing HIF1-α and downregulating PDK4.
AACR. 74:S9782014.
|