Introduction
The vitamin D receptor (VDR), as a member of the
nuclear receptor superfamily, is essential for initiating the
intranuclear signaling pathways that are activated by the active
metabolite of vitamin D. 1,25-dihydroxyvitamin [1,25(OH)2D3] is the
biologically active form of vitamin D3, which directly or
indirectly controls hundreds of genes in a cell- and
tissue-specific manner (1,2). It is widely accepted that 1,25(OH)2D3
plays a critical role in the metabolism of calcium; however, little
is known about the role of other biological functions of
1,25(OH)2D3, such as the inhibition of cell proliferation,
induction of differentiation, cell cycle and apoptosis regulation,
suppression of angiogenesis and participation in inflammatory
processes. Epidemiological evidence has revealed an association
between vitamin D status and the risk of developing cancer
(3,4). Different studies also indicated that
1,25(OH)2D3 has a potent anticancer effect in vitro and
in vivo (5–7). Furthermore, it was shown in a previous
study that VDR expression was markedly decreased when the tumor
disease stage advanced (8).
Although, to the best of our knowledge, the molecular mechanisms of
the anticancer activity of 1,25(OH)2D3 have not yet been fully
elucidated, the ability of 1,25(OH)2D3 to inhibit cancer
development is considered to be mediated by a combination of
different pathways. In view of its potent antiproliferative and
pro-differentiation action, and the presence of the VDR in a large
variety of myeloid leukemia cells, 1,25(OH)2D3 could be considered
a promising drug to treat myeloid malignancies (9).
It is well known that arsenic trioxide
(As2O3) has been successfully used to treat
acute promyelocytic leukemia (APL) in traditional medicine
(10). Recently, it has been
approved by the US Food and Drug Administration for the treatment
of relapsed/refractory APL (11).
Studies have revealed that As2O3 exerts
proapoptotic effects, not only in APL cells, but also in other
hematopoietic malignancies and solid tumors (12,13). The
underlying mechanisms of the antitumor activity of
As2O3 have been associated with the induction
of tumor apoptosis and inhibition of cell proliferation by
promoting the production of reactive oxygen species (14). However, whether 1,25(OH)2D3 and
As2O3 exert synergistic effects on the
proliferation and differentiation of leukemia cells remains
unknown.
In the present study, the antitumor effect of
1,25(OH)2D3 combined with As2O3 was
investigated, and the underlying molecular mechanisms were
determined using a K562 cell line established from human chronic
myelogenous leukemia cells in blast crisis.
Materials and methods
Cell line, cell culture and
reagents
The K562 cell line was purchased from The Cell Bank
of Type Culture Collection of The Chinese Academy of Sciences.
Cells were cultured in RPMI-1640 medium (Thermo Fisher Scientific,
Inc.) supplemented with 10% fetal bovine serum (FBS; Cytiva), 100
µg/ml penicillin, 10 µg/ml streptomycin, and 2 mmol/l L-glutamine.
Cells were maintained in log phase growth at 37°C in a humidified
atmosphere containing 5% CO2.
1,25(OH)2D3 (Merck KGaA), also
known as calcitriol, was dissolved in 100% ethanol at a
concentration of 5×10−4 mol/l as a stock solution and
stored at 20°C for use in the following experiments. For all
experiments, dilutions of the stock solution were made in RPMI-1640
medium without FBS. The maximum concentration of ethanol in the
culture did not exceed 0.1%. As2O3 was
purchased from Heilongjiang Harbin Yida Pharmaceutical Co.,
Ltd.
Optimal drug concentration screening
and evaluation
Inhibition rate of the cell proliferation in K562
cells was measured using a
3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium,
inner salt/phenazine ethosulfate (MTS/PES) assay. The cytotoxic
effects of 1,25(OH)2D3 on K562 cells were
examined by treating 4,000 cells/well with 0.1% ethanol and 0, 10,
50, 100 or 200 nM 1,25(OH)2D3 for 72 h (15), and then analyzed using a MTS/PES
assay with the absorbance measured at a wavelength of 490 nm. For
arsenic trioxide, no concentration gradient experiment was
performed; the optimal concentration of 2.5 µM was obtained from
the literature (16).
Cell proliferation assay
Cell proliferation was determined in 96-well plates
using an MTS/PES assay, according to the manufacturer's
instructions. Briefly, following treatment with 100 nM
1,25(OH)2D3 and 2.5 µM
As2O3, alone or combined, K562 cells were
seeded in 96-well plates at a density of 4,000 cells/well. The same
number of untreated cells were seeded as the control. Cultures were
set up in triplicate and maintained at 37°C in a humidified
atmosphere with 5% CO2. Following 24, 48, 72, 96 or 120
h of treatment, 10 µl MTS/PES (10 mg/ml; Promega Corporation) was
added into each well for an additional 6 h incubation. A microplate
reader was used to measure the absorbance value at 490 nm for each
well, which represented K562 cell proliferation.
Assessment of apoptosis and cell cycle
analysis
The apoptosis assay of K562 cells was performed
using an Annexin V/propidium iodide (PI) apoptosis assay kit
(BioLegend, Inc.), according to the manufacturer's instructions.
Briefly, the cells were harvested and washed twice with cold
phosphate-buffered saline (PBS). Cells were then resuspended in 1X
binding buffer at a concentration of 1×106 cells/ml. A
total of 100 µl of the solution was transferred to a 5-ml culture
tube with 5 µl Annexin V-FITC and 10 µl PI (50 mg/ml). Cells were
gently vortexed and incubated for 15 min at room temperature in the
dark. Following the addition of 400 ml of binding buffer, these
cells were analyzed by flow cytometry (FCM). Annexin V−
and PI− cells were considered viable cells, Annexin
V+ and PI− cells were considered early
apoptotic cells and Annexin V+ and PI+ cells
were considered late apoptotic cells.
Cell cycle distribution was determined by staining
DNA with PI, as previously described (17). Briefly, ~5×106 K562 cells
were collected from cultures, washed twice with PBS and fixed in
70% pre-cold ethanol at 4°C overnight. The fixed cells were then
collected and resuspended in PBS, containing 50 mg/ml PI and 100
µg/ml DNase-free RNase A. Cells were incubated for 1 h at room
temperature and then analyzed by FCM.
Reverse transcription-quantitative PCR
(RT-qPCR)
K562 cells from each experimental group were
collected and washed three times with cold PBS, and then total cell
mRNA was extracted using TRIzol® reagent (Invitrogen;
Thermo Fisher Scientific, Inc.), according to the manufacturer's
instructions. cDNA was synthesized from 2 µg total RNA using a
first-strand cDNA synthesis kit (Thermo Fisher Scientific, Inc.).
The PCR amplification protocol was as follows: Denaturation at 94°C
for 10 min, followed by 40 cycles of 94°C for 15 sec, 58°C for 30
sec and 72°C for 40 sec. The mRNA expression levels of VDR, Bcl-2,
survivin, Bax, p21, p27 and β-actin were detected using ABI 7000
(Thermo Fisher Scientific, Inc.) and Talent qPCR PreMix
(SYBR-Green) (Tiangen Biotech Co., Ltd). The relative
quantification based on the relative expression of target genes was
calculated using the 2−ΔΔCq method (18). The PCR primers were designed based on
the corresponding gene structure, and the sequences are listed in
Table I. RT-qPCR was performed in
triplicate.
 | Table I.Reverse transcription-quantitative
PCR primers. |
Table I.
Reverse transcription-quantitative
PCR primers.
| Gene | Direction | Sequence |
|---|
| VDR | Sense |
5′-AGCTGGCCCTGGCACTGACTCTGCTCT-3′ |
|
| Antisense |
5′-ATGGAAACACCTTGCTTCTTCTCCCTC-3′ |
| Bcl-2 | Sense |
5′-ATCGCCCTGTGGATGACTGAG-3′ |
|
| Antisense |
5′-CAGCCAGGAGAAATCAAACAGAGG-3′ |
| Survivin | Sense |
5′-CCCTGCCTGGCAGCCCTTTC-3′ |
|
| Antisense |
5′-CTGGCTCCCAGCCTTCCA-3′ |
| Bax | Sense |
5′-GGACGAACTGGACAGTAACATGG-3′ |
|
| Antisense |
5′-GCAAAGTAGAAAAGGGCGACAAC-3′ |
| p21 | Sense |
5′-GCAGACCAGCATGACAGATTT-3′ |
|
| Antisense |
5′-GGATTAGGGCTTCCTCTTGGA-3′ |
| p27 | Sense |
5′-CCTCTTCGGCCCGGTGGAC-3′ |
|
| Antisense |
5′-TTTGGGGAACCGTCTGAAAC-3′ |
| β-actin | Sense |
5′-TCTGGCACCACACCTTCTACAATG-3′ |
|
| Antisense |
5′-AGCACAGCCTGGATAGCAACG-3′ |
Western blot analysis
Following various treatments with 100 nM 1,25(OH)2D3
and 2.5 µM As2O3, alone or combined, K562 cells were collected and
washed three times with pre-cooled PBS. The proteins were extracted
using RIPA buffer (Thermo Fisher Scientific, Inc.) and quantified
using a bicinchoninic acid protein assay kit (Thermo Fisher
Scientific, Inc.). Proteins were separated via SDS-PAGE (15% gel)
and transferred to a polyvinylidene difluoride membrane. The
membrane was blocked with 5% non-fat milk in Tris-buffered saline
containing 0.1% Tween-20 (TBST) at 37°C for 2 h, and subsequently
incubated with the primary antibodies mouse anti-VDR (1:1,000; cat.
no. sc13133), mouse anti-Bcl-2 (1:1,000; cat. no. sc7382), mouse
anti-survivin (1:1,000; cat. no. sc17779), mouse anti-Bax (1:1,000;
cat. no. sc7480), mouse anti-p21 (1:1,000; cat. no. sc6246), mouse
anti-p27 (1:1,000; cat. no. sc1641) and mouse anti-β-actin
(1:1,000; cat. no. sc69879) (all from Santa Cruz Biotechnology,
Inc.) at 4°C overnight. After five more washes with TBST, the blots
were incubated with the horseradish peroxidase-conjugated rabbit
anti-mouse IgG (1:5,000; cat. no. ab6728; Abcam) for 1 h at room
temperature. The immunoreactive bands were visualized by enhanced
chemiluminescence (ECL) on a Kodak Image Station 4000 mm Pro system
(Kodak Corporation). The quantitative data from bands were analyzed
using the Image Tool system (version 3.0; www.bio-soft.net).
Statistical analysis
All statistical data are presented as the mean ±
standard deviation. The treatment effects among different groups
were compared using one-way analysis of variance and Bonferroni's
post hoc test. All analyses were performed using GraphPad Prism
version 5.0 (GraphPad Software, Inc.). P<0.05 was considered to
indicate a statistically significant difference.
Results
1,25(OH)2D3 exerts synergistic effects
with As2O3 on the proliferation of K562
cells
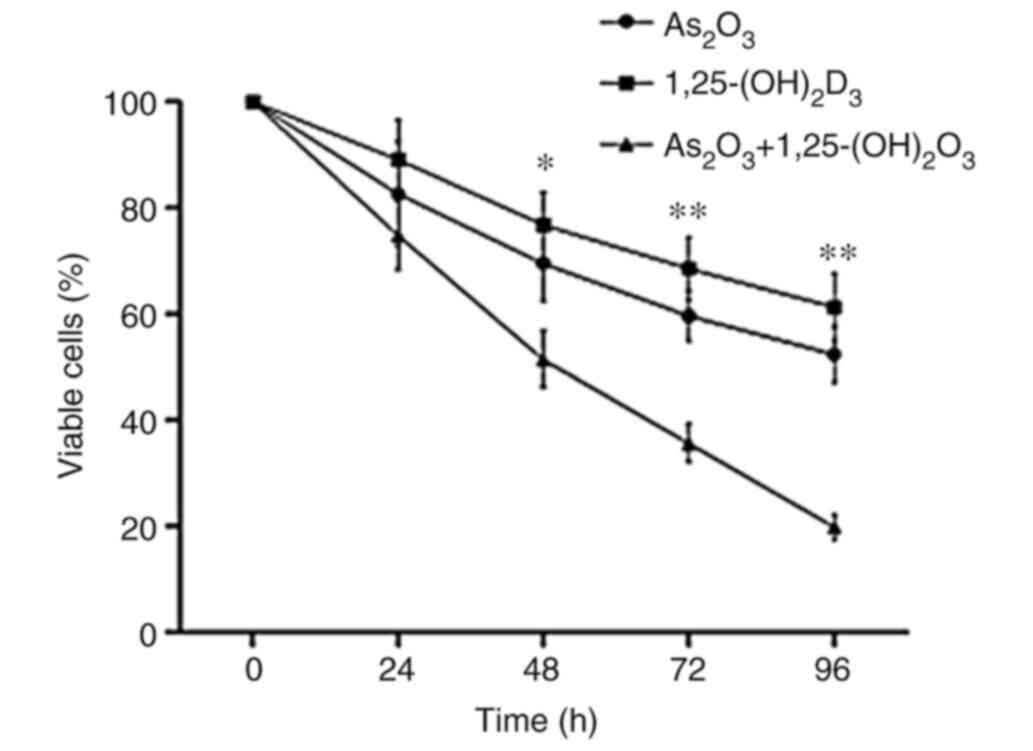
The results of the MTS/PES assay revealed that
1,25(OH)2D3 suppressed the proliferation of K562 cells in a
dose-dependent manner (Fig. 1).
Since 100 nM 1,25(OH)2D3 displayed the most significant anticancer
effect on K562 cells, that concentration was selected for
subsequent experiments. The results showed that 100 nM 1,25(OH)2D3
or 2.5 µM As2O3 alone exerted inhibitory
effects on K562 cells. Of note, the inhibition of cell
proliferation was more effective with combined treatment than with
treatment with As2O3 or 1,25(OH)2D3 alone
(Fig. 2).
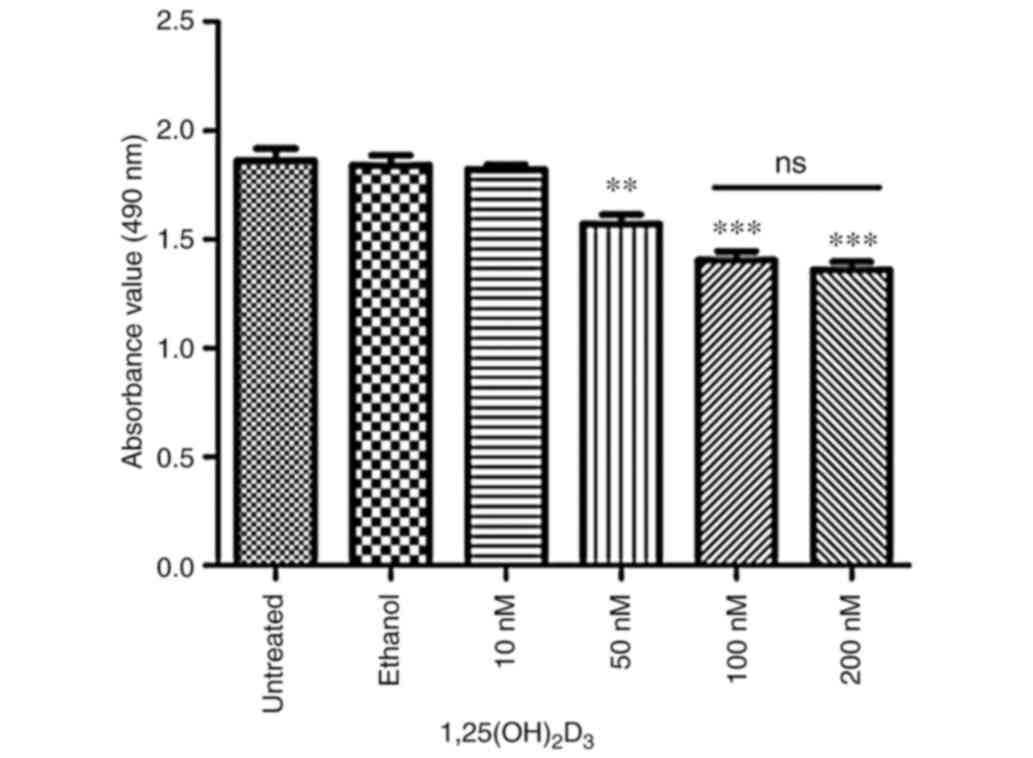 | Figure 1.Proliferation of K562 cells is
suppressed by 1,25(OH)2D3 in a dose-dependent manner. K562 cells at
an initial density of 4,000 cells/well were treated with different
concentrations of 1,25(OH)2D3 (10, 50, 100 or 200 nM) for 72 h, and
then analyzed by MTS/PES assay absorbance measured at a wavelength
of 490 nm. Untreated, without 1,25(OH)2D3 treatment; ethanol, with
0.1% ethanol treatment. Data are presented as the mean ± SD of
three independent experiments. **P<0.01 and ***P<0.001 vs.
untreated group. 1,25(OH)2D3, 1,25-dihydroxyvitamin; ns, not
significant. |
1,25(OH)2D3 enhances the
As2O3-induced apoptosis of K562 cells
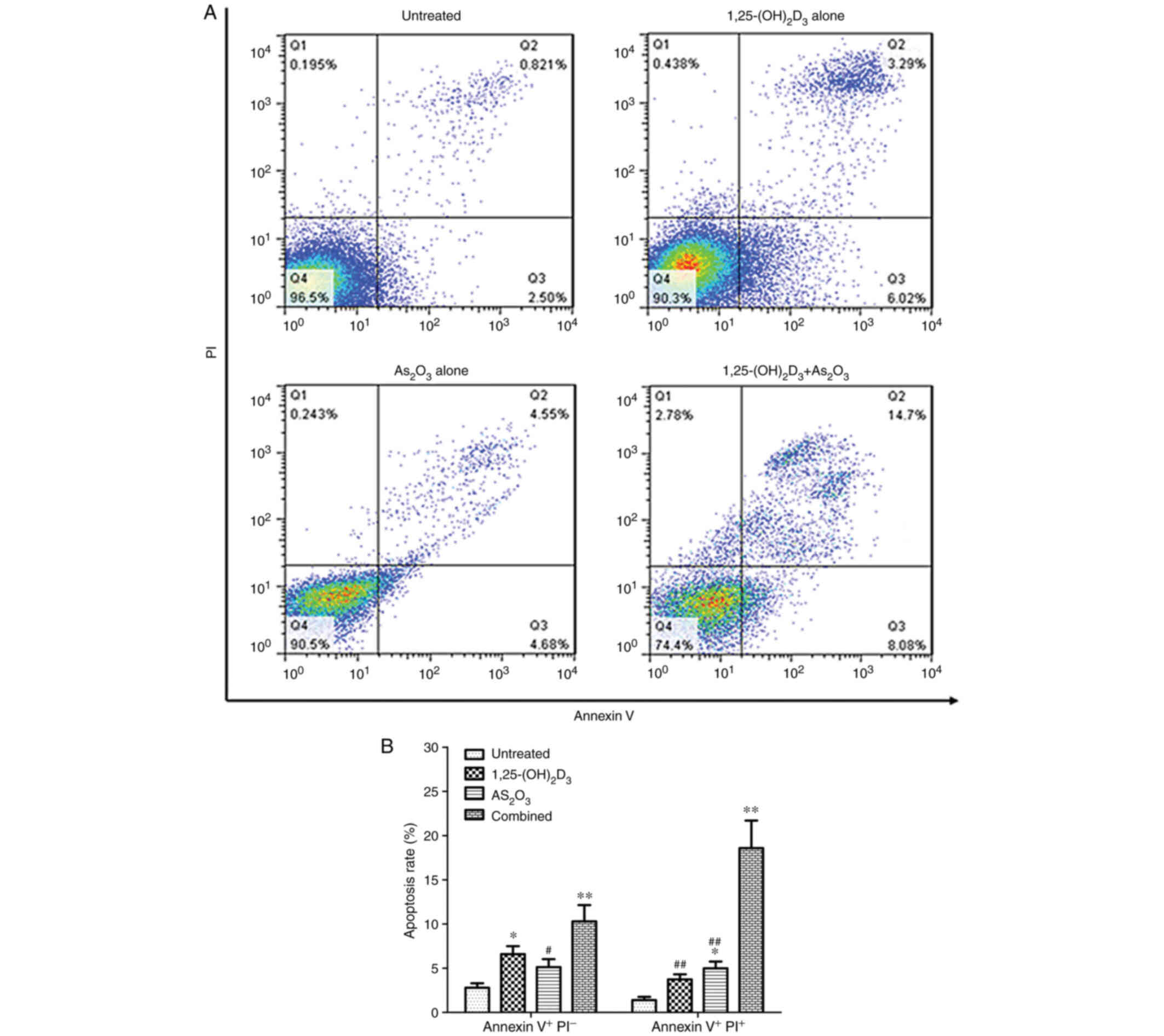
K562 cells were treated with 100 nM 1,25(OH)2D3
and/or 2.5 µM As2O3 for 72 h. Fig. 3A shows a representative example of
apoptotic cells in untreated K562 cells and cells treated with
1,25(OH)2D3, As2O3 or a combination of both.
The results showed that the percentage ratio of early and late
apoptotic cells in the combination group was significantly higher
than that in the blank control and 1,25(OH)2D3 or
As2O3 treatment groups (P<0.01; Fig. 3B).
1,25(OH)2D3 and
As2O3 induce G1 cell cycle
arrest
Cell cycle distribution was analyzed via FCM.
Fig. 4A shows a representative
example of cell cycle distribution in untreated K562 cells and
cells treated with 1,25(OH)2D3, As2O3 or a
combination of both. The results revealed that the combined
treatment markedly increased the population of cells in the
G0/G1 phase and significantly decreased the
population of cells in the S-phase compared with cells treated with
1,25(OH)2D3 or As2O3 alone. By contrast, the
percentage of cells in the G2/M phase was relatively
unaffected (Fig. 4B). These data
demonstrated that the combined treatment induced significant arrest
of cell cycle progression in the G0/G1
phase.
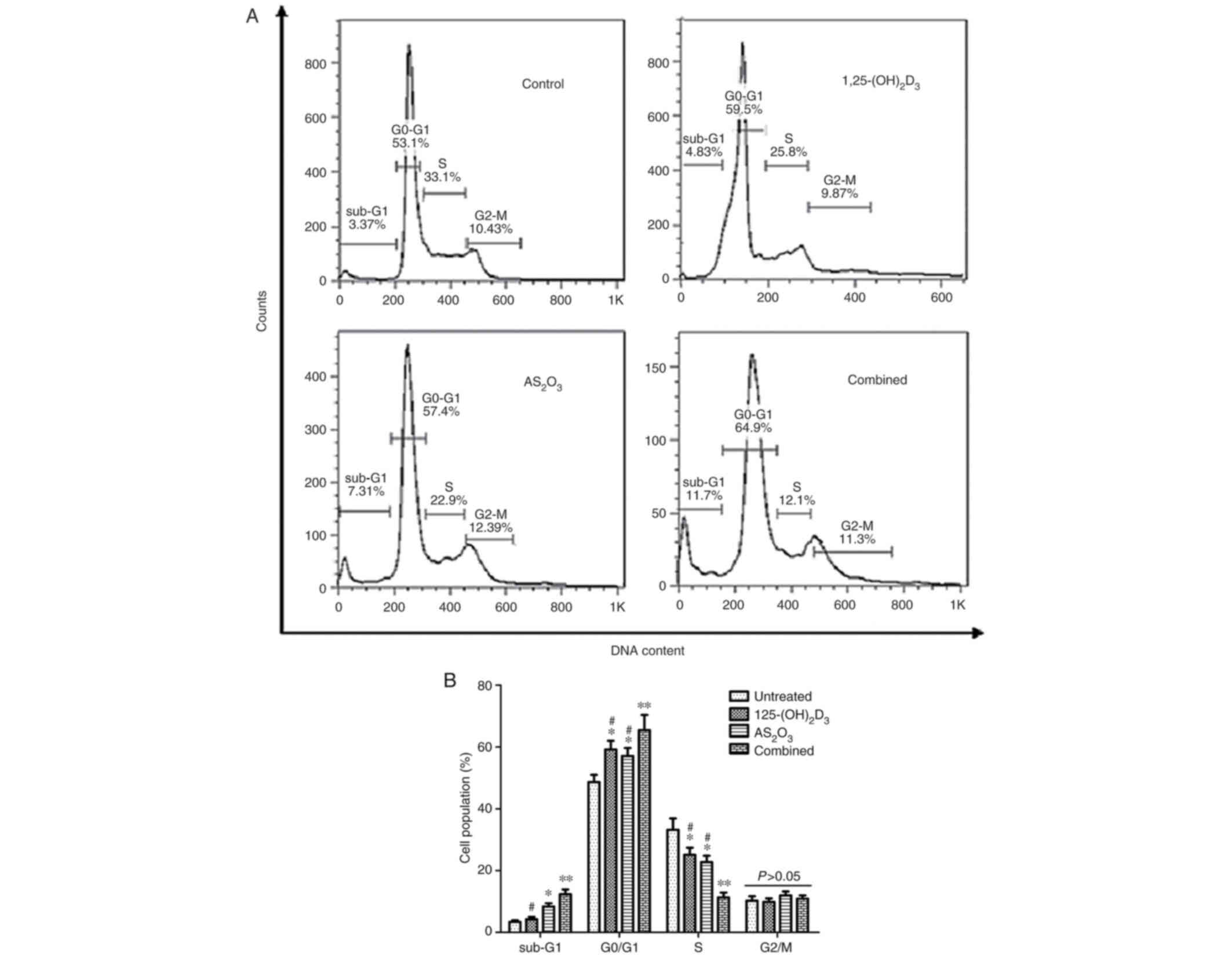 | Figure 4.G1 cell cycle arrest is
induced by 1,25(OH)2D3 and As2O3 following
treatment with 1,25(OH)2D3, As2O3 or a
combination of both for 72 h. (A) Representative example of cell
cycle distribution in untreated K562 cells and cells treated with
1,25(OH)2D3, As2O3, or the combination of
both. (B) Cell cycle distribution in each group. Data are presented
as the mean ± SD of three independent experiments. *P<0.05 and
**P<0.01 vs. control group; #P<0.05 vs. combined
treatment group. 1,25(OH)2D3, 1,25-dihydroxyvitamin;
As2O3, arsenic trioxide. |
Effects of 1,25(OH)2D3 and
As2O3 on the expression of apoptosis-related
and cycle-regulated genes and proteins
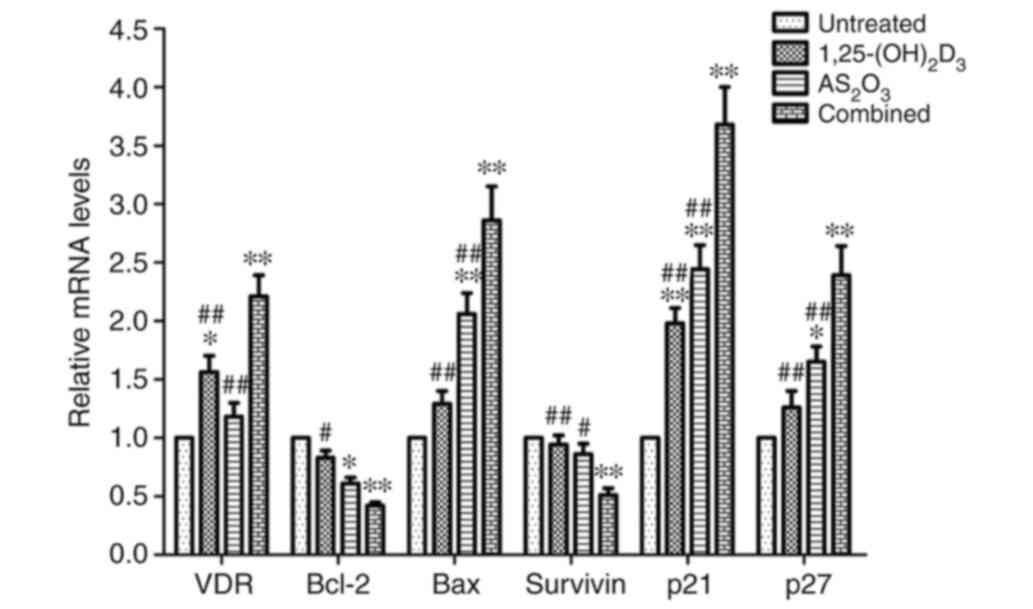
To further investigate the underlying molecular
mechanisms of cell proliferation suppression, apoptosis induction
and G0/G1 cell cycle arrest, the mRNA and
protein expression levels of VDR, Bcl-2, Bax, survivin, p21 and p27
were analyzed in K562 cells using RT-qPCR and western blot
analysis. Fig. 5 presents the
relative mRNA quantification of these genes in untreated K562 cells
and cells treated with 1,25(OH)2D3, As2O3 or
a combination of both. The results revealed that the combined
treatment with 1,25(OH)2D3 and As2O3 resulted
in a marked reduction in the levels of Bcl-2 and survivin when
compared with untreated cells and 1,25(OH)2D3 or
As2O3 alone, while the levels of the VDR,
Bax, p21 and p27 were significantly increased following combined
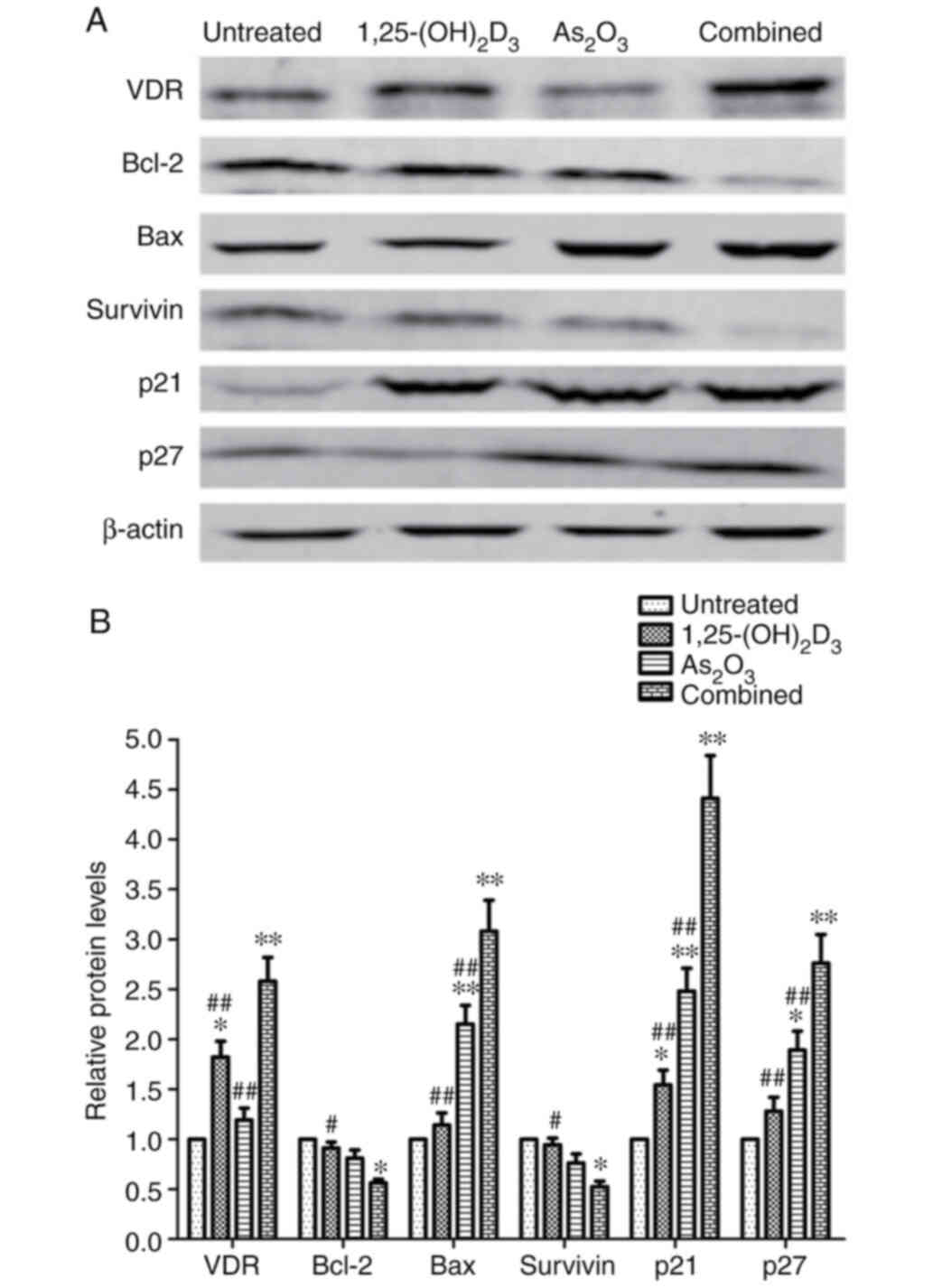
treatment. Representative immunoblots are presented in Fig. 6A. Similar to the observed mRNA
expression levels, the protein expression levels of VDR, Bax, p21
and p27 were found to be increased, while the expression levels of
Bcl-2 and survivin were found to be significantly decreased
(Fig. 6B).
Discussion
The ability of 1,25(OH)2D3 to induce apoptosis has
been demonstrated in various tumor cells. Although, to the best of
our knowledge, the mechanisms underlying the apoptotic effects have
not yet been fully elucidated, 1,25(OH)2D3 may induce cell death by
triggering the intrinsic, mitochondria-dependent pathway (6). According to cell type, 1,25(OH)2D3 can
increase the expression levels of proapoptotic factors Bax and
Bcl-2 homologous antagonist killer (Bak) and/or decrease their
anti-apoptotic equivalents Bcl-2 and B-cell lymphoma-extra large,
thus directing the cells towards apoptosis rather than towards
survival (19,20). Elucidating the precise molecular
mechanism underlying the antiproliferative action of 1,25(OH)2D3
could help identify new biomarkers for targeted treatment with
novel vitamin D analogs.
The results of the present study showed that both
1,25(OH)2D3 and As2O3 are reagents that can
effectively inhibit the proliferation of K562 leukemia cells. FCM
showed that As2O3 significantly increased the
rate of late apoptotic cells, but had no significant effect on the
rate of early apoptotic cells. By contrast, 1,25(OH)2D3 only
increased the rate of early apoptotic cells, but did not affect the
rate of late apoptotic cells. When As2O3 and
1,25(OH)2D3 were combined for the treatment of K562 cells, a
synergistic effect was observed. It is well known that apoptosis is
regulated by a series of genes. Survivin is a member of the
inhibitor of apoptosis family of proteins, which are involved in
the inhibition of apoptosis and regulation of the cell cycle
(21). Upregulation of survivin
promotes tumor progression by inhibiting both the intrinsic and
extrinsic pathways of apoptosis, altering sensitivity to antitumor
drugs or prolonging the survival of cancer stem cells (22). It has been hypothesized that survivin
can also serve as a universal tumor antigen, since it is expressed
in the majority of human hematological malignancies and has the
potential to trigger immune effector responses (23). The results of the present study
indicated that the combined treatment of 1,25(OH)2D3 and
As2O3 may significantly decrease the
expression of survivin, while 1,25(OH)2D3 and
As2O3 alone cannot affect its expression.
Therefore, blocking the function of survivin through the use of
various drugs or molecular approaches is a promising therapeutic
strategy for leukemia.
Previous studies have demonstrated that both
1,25(OH)2D3 and As2O3 can arrest the cell
cycle at the G1 phase, probably through the upregulation
of one or both of the cyclin-dependent kinase inhibitors (p21 and
p27), so as to inhibit the malignant proliferation of K562 cells
(15,16,24). In
accordance with these results, the data generated in the present
study indicated that treatment with As2O3 and
1,25(OH)2D3 alone caused clear G1/S arrest, while the
number of cells in the G2/M phase was relatively
unaffected. Further research showed that the G1/S
blockade induced by combined treatment with 1,25(OH)2D3 and
As2O3 could upregulate the protein expression
of p21 and p27, while treatment with 1,25(OH)2D3 only upregulated
the expression of p21. In addition, 1,25(OH)2D3 could increase the
expression of VDR, while As2O3 did not affect
the expression.
Due to the small amount of experimental conditions
in the present study, there are still some limitations to the
present study and some aspects that were not investigated. The
present study focused on the changes observed from traditional
apoptosis and cell cycle-related genes in regard to the underlying
anticancer molecular mechanisms. However, the effects of combined
treatment on mitochondrial dysfunctions, oxidative stress and
regulation of calcium influx are not involved. Future studies could
use a reactive oxygen species assay kit to quantify the levels of
intracellular reactive oxygen species and apply fluorescent
rhodamine derivatives.
Overall, the results of the present study provided
evidence that the addition of 1,25(OH)2D3 may increase the
therapeutic efficacy of As2O3, which may in
turn decrease adverse effects and initiate a more comprehensive
antitumor pathway. This potential method urgently requires further
investigation to elucidate the potential therapeutic benefits of a
variety of non-APL hematological malignancies.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
YLZ, JHR, XNG and JNZ performed the research,
analysis and interpretation of the data. YLZ and XNG drafted the
manuscript and gave final approval of the version to be published.
SKQ designed the study and supervised preparation of the
manuscript, and provided general support. YLZ and SKQ confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Christakos S, Dhawan P, Verstuyf A,
Verlinden L and Carmeliet G: Vitamin D: Metabolism, molecular
mechanism of action, and pleiotropic effects. Physiol Rev.
96:365–408. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Umar M, Sastry KS and Chouchane AI: Role
of vitamin D beyond the skeletal function: A review of the
molecular and clinical studies. Int J Mol Sci. 19:16182018.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Krishnan AV and Feldman D: Mechanisms of
the anti-cancer and anti-inflammatory actions of vitamin D. Annu
Rev Pharmacol Toxicol. 51:311–336. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Grant WB and Boucher BJ: Randomized
controlled trials of vitamin D and cancer incidence: A modeling
study. PLoS One. 12:e01764482017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bandera Merchan B, Morcillo S,
Martin-Nuñez G, Tinahones FJ and Macías-González M: The role of
vitamin D and VDR in carcinogenesis: Through epidemiology and basic
sciences. J Steroid Biochem Mol Biol. 167:203–218. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Picotto G, Liaudat AC, Bohl L and Tolosa
de Talamoni N: Molecular aspects of vitamin D anticancer activity.
Cancer Invest. 30:604–614. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Díaz L, Díaz-Muñoz M, García-Gaytán AC and
Méndez I: Mechanistic effects of calcitriol in cancer biology.
Nutrients. 7:5020–5050. 2015. View Article : Google Scholar
|
|
8
|
Campbell MJ and Trump DL: Vitamin D
receptor signaling and cancer. Endocrinol Metab Clin North Am.
46:1009–1038. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kulling PM, Olson KC, Olson TL, Feith DJ
and Loughran TP Jr: Vitamin D in hematological disorders and
malignancies. Eur J Haematol. 98:187–197. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lazo G, Kantarjian H, Estey E, Thomas D,
O'Brien S and Cortes J: Use of arsenic trioxide (As2O3) in the
treatment of patients with acute promyelocytic leukemia. Cancer.
97:2218–2224. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hoonjan M, Jadhav V and Bhatt P: Arsenic
trioxide: Insights into its evolution to an anticancer agent. J
Biol Inorg Chem. 23:313–329. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Abudoureyimu A and Muhemaitibake A:
Arsenic trioxide regulates gastric cancer cell apoptosis by
mediating cAMP. Eur Rev Med Pharmacol Sci. 21:612–617.
2017.PubMed/NCBI
|
|
13
|
Sadaf N, Kumar N, Ali M, Ali V, Bimal S
and Haque R: Arsenic trioxide induces apoptosis and inhibits the
growth of human liver cancer cells. Life Sci. 205:9–17. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Woo SH, Park IC, Park MJ, Lee HC, Lee SJ,
Chun YJ, Lee SH, Hong SI and Rhee CH: Arsenic trioxide induces
apoptosis through a reactive oxygen species-dependent pathway and
loss of mitochondrial membrane potential in HeLa cells. Int J
Oncol. 21:57–63. 2002.PubMed/NCBI
|
|
15
|
Kizildag S, Ates H and Kizildag S:
Treatment of K562 cells with 1,25-dihydroxyvitamin D3 induces
distinct alterations in the expression of apoptosis-related genes
BCL2, BAX, BCLXL, and p21. Ann Hematol. 89:1–7. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bae JY, Kim JW and Kim I: Low-dose
1,25-dihydroxyvitamin D(3) combined with arsenic trioxide
synergistically inhibits proliferation of acute myeloid leukemia
cells by promoting apoptosis. Oncol Rep. 30:485–491. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pozarowski P and Darzynkiewicz Z: Analysis
of cell cycle by flow cytometry. Methods Mol Biol. 281:301–311.
2004.PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Deeb KK, Trump DL and Johnson CS: Vitamin
D signalling pathways in cancer: Potential for anticancer
therapeutics. Nat Rev Cancer. 7:684–700. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Krishnan AV, Trump DL, Johnson CS and
Feldman D: The role of vitamin D in cancer prevention and
treatment. Endocrinol Metab Clin North Am. 39:401–418. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Peery RC, Liu JY and Zhang JT: Targeting
survivin for therapeutic discovery: Past, present, and future
promises. Drug Discov Today. 22:1466–1477. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mobahat M, Narendran A and Riabowol K:
Survivin as a preferential target for cancer therapy. Int J Mol
Sci. 15:2494–2516. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Friedrichs B, Siegel S, Andersen MH,
Schmitz N and Zeis M: Survivin-derived peptide epitopes and their
role for induction of antitumor immunity in hematological
malignancies. Leuk Lymphoma. 47:978–985. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li N, Guan X, Li F, Li X and Chen Y:
Vorinostat enhances chemosensitivity to arsenic trioxide in K562
cell line. Peer J. 3:e9622015. View Article : Google Scholar : PubMed/NCBI
|