Introduction
Oral squamous cell carcinoma (OSCC) is a type of
malignant tumor derived from the oral epithelium (1,2). It is
the sixth most common malignant tumor in the world (3). Resection combined with radiotherapy and
chemotherapy is currently the predominantly used treatment for
OSCC, but the prognosis is poor and serious side effects are common
(1,2,4).
Therefore, investigating the underlying molecular mechanisms of the
development of OSCC is required for identifying novel therapeutic
targets (5).
The tumor microenvironment (TME) plays an important
role in regulating the development of OSCC (6). The most abundant cells in the TME are
cancer-associated fibroblasts (CAFs), which have similar
characteristics to myofibroblasts (7). CAFs are primarily derived from stromal
fibroblasts (8). Indeed, CAFs can be
activated by signals, such as paracrine transforming growth factor
(TGF)-β, epidermal growth factor (EGF) and platelet-derived growth
factor (PDGF) from tumor cells (9–12), and
specifically express α-smooth muscle actin (α-SMA), fibroblast
activation protein-α (FAP-α), PDGF receptor (PDGFR)-β and
fibroblast specific protein (13,14).
Conversely, CAFs release a large number of cytokines to regulate
the biological behavior of tumors (15) and promote tumors by promoting
microvessel formation, interstitial remodeling, drug resistance,
immunosuppression, tumor cell proliferation, invasion and
metastasis (16).
The PDGF family plays key roles in normal embryonic
development, cell growth, cell differentiation and response to
tissue damage (17,18). PDGF-BB is one of the important
members of the PDGF family and its specific receptor is PDGFR-β
(19). The binding of PDGF-BB to its
receptor PDGFR-β triggers the formation of dimeric complexes of the
receptor, thereby activating downstream signaling molecules to
produce a series of biological effects and promote the occurrence
and development of tumors (19).
Zhang et al (20)
demonstrated that PDGF-BB induces human normal fibroblasts (NFs) to
transform into CAFs.
Long non-coding (lnc)RNA are RNA molecules 0.2–100
kb in length that lack protein-coding ability. They are key
regulators of cell growth, apoptosis, differentiation, invasion and
stem cell diversity (21). Studies
have shown that metastasis-associated lung adenocarcinoma
transcript 1, maternally expressed gene 3 and lncRNA AC132217.4 are
closely related to the development, invasion and metastasis of OSCC
(22–24). Ding et al (25) identified that lnc-CAF/interleukin-33
transforms NFs into CAFs, promoting the development of OSCC. In
addition, Zhao et al (26)
found that long intergenic non-protein coding RNA 92 in ovarian
cancer cells could maintain CAF activation through glycolysis.
Therefore, lncRNAs are important modulators between cancer cells
and CAFs, and demonstrate potential application prospects in cancer
treatment.
Nuclear factor (NF)-κB is an inducible
transcriptional regulator that manipulates the expression of
multiple inflammation and immune genes (27). It is a key signaling molecule
connecting inflammation and tumors, and has important effects on
the TME (28). Phosphorylated (p)
NF-κB enters the nucleus to induce the reorganization of the
fibroblast structure, as characterized by the increased expression
of cytoskeletal protein α-SMA and activation of fibroblasts into
CAFs (29,30). Leucine-rich adaptor protein 1-like
(LURAP1L) is a protein of the LURAP1 family that activates the
canonical NF-κB pathway (31). The
present study hypothesized that LURAP1L was the target of lncRNA
LURAP1L antisense RNA 1 (LURAP1L-AS1). Notably, the preliminary
lncRNA ChIP results indicated that expression of lncRNA LURAP1L-AS1
was upregulated during the activation of fibroblasts induced by
PDGF-BB. Therefore, it was hypothesized that PDGF-BB might
upregulate LURAP1L by promoting lncRNA LURAP1L-AS1 expression and
subsequently activating the canonical NF-κB pathway, thereby
inducing the transformation of NFs into CAFs.
Materials and methods
Cell culture and treatment
Normal human oral mucosa (p3) 500K fibroblast (hOMF)
cells were purchased from CellResearch Corporation and grown in
complete DMEM, high glucose (Gibco; Thermo Fisher Scientific, Inc.)
supplemented with 10% fetal bovine serum (cat. no 16000-04; Gibco;
Thermo Fisher Scientific, Inc.; fetal bovine serum:DMEM=1:9) and
Gibco penicillin-streptomycin solution (0.1 U/ml penicillin and 0.1
µg/ml streptomycin; Thermo Fisher Scientific, Inc.) at 37°C in
humidified air with 5% CO2.
For activation, recombinant human PDGF-BB (32) was used to stimulate the conventional
hOMF cells for 72 h before passage. After three consecutive
passages, the cells were collected for subsequent experiments.
Protein extraction and western
blotting
Total protein was extracted from cell cultures using
Cell Extraction Buffer (Thermo Fisher Scientific, Inc.) and the
Mini Protease Inhibitor Cocktail (Roche Diagnostics). The protein
concentration was determined using a BCA Protein assay kit
(Beyotime Institute of Biotechnology). Equal quantities (20 µg) of
proteins were separated using 10% SDS-PAGE and then transferred to
a PVDF membrane (EMD Millipore). The membrane was blocked in 5%
skimmed milk in TBS containing 0.1% Tween-20 (TBST, pH 7.2) for 1 h
at room temperature, incubated with an appropriate quantity of
primary antibody at 4°C overnight and washed three times with TBST
for 15 min each time and then incubated with secondary antibodies.
Immunoreactivity was revealed by chemiluminescence using a western
blot imaging system (ImageQuant LAS 4000; GE Healthcare) and the
gray value was analyzed using ImageJ software 1.41 (National
Institutes of Health). β-tubulin, β-actin and GAPDH served as
internal references. All of the samples were run in triplicate as a
minimum. The following antibodies were used in the present study:
α-SMA (cat. no. ab5694; 1:1,000), FAP-α (cat. no. ab53066; 1:500),
IKKα (cat. no. ab32041; 1:1,000), IκBα (cat. no. ab32518; 1:1,000),
NF-κB p65 (cat. no. ab16502; 1:1,000), p-NF-κBp65 (p-S536; cat. no.
ab86299; 1:1,000), GAPDH (cat. no. ab181602; 1:10,000), β-tubulin
(cat. no. ab179511; 1:1,000) and β-actin (cat. no. ab8227; 1:1,000)
were obtained from Abcam; LURAP1L (cat. no. PA5-55072; 1:1,000) was
obtained from Thermo Fisher Scientific, Inc., and PDGFR-β (cat. no.
bs-0232R; 1:1,000) and p-PDGFR-β (Tyr740; cat. no. bs-3323R;
1:1,000) were obtained from BIOSS. Goat Anti-Rabbit IgG H&L
(HRP) (cat. no. zs-2301; 1:5,000) was obtained from ZSBIO.
Immunofluorescence (IF)
For IF, cells were plated on coverslips, washed
three times with PBS, fixed in 4% paraformaldehyde for 10 min at
room temperature, permeabilized with 0.5% Triton X-100 for 15 min
and incubated with primary antibodies, anti-α-SMA, anti-FAP-α and
anti-PDGFR-β at 4°C overnight, followed by a 1-h incubation with
rhodamine-conjugated goat anti-rabbit IgG (DyLight 649; cat. no.
A23620; Abbkine Scientific Co., Ltd. and Alexa
Fluor®488, cat. no. ab150081; Abcam) at 37°C. The nuclei
were counterstained with 4′,6-diamidino-2-phenylindole (Invitrogen;
Thermo Fisher Scientific, Inc, 0.5 µg/ml) for 5 min in the dark at
room temperature. The coverslips were evaluated by fluorescence
microscopy using an 80i Eclipse microscope (Nikon Corporation).
RNA extraction
Total RNA was extracted using TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.) according to
the manufacturer's instructions. RNA quantity and quality were
measured using a NanoDrop® ND-1000 (NanoDrop
Technologies; Thermo Fisher Scientific, Inc.). RNA integrity was
assessed by standard denaturing agarose gel electrophoresis.
Microarray
An Agilent array platform (Agilent Technologies,
Inc.) was used for microarray analysis. The sample preparation and
microarray hybridization were performed according to the
manufacturer's instructions with minor modifications. Briefly, mRNA
was purified from total RNA after removal of rRNA with an
mRNA-ONLY™ Eukaryotic mRNA Isolation kit (Epicentre; Illumina,
Inc.). Then, each sample was amplified and transcribed into
fluorescent cRNA along the entire length of the transcripts without
3′bias using a random priming method (Flash RNA Labeling kit;
Arraystar, Inc.). The labeled cRNAs were hybridized onto a Human
LncRNA Microarray V4.0 (8×60k; Arraystar, Inc.) designed for 40,173
lncRNAs and 20,730 coding transcripts. The lncRNAs were carefully
constructed using public transcriptome databases, including RefSeq
(http://www.ncbi.nlm.nih.gov/refseq/),
UCSC Known Genes (http://www.biomedsearch.com/nih/UCSC-Known-Genes/16500937.html)
and GENCODE (http://www.gencodegenes.org/) as well as landmark
publications (33–35). Each transcript was accurately
identified by a specific exon or splice junction probe. Positive
probes for housekeeping genes and negative probes were printed onto
the array for hybridization quality control. After washing the
slides with Gene Expression Wash Buffer 1 (p/n 5188–5325; Agilent
Technologies, Inc.), the arrays were scanned using a G2505C scanner
(Agilent Technologies, Inc.) and the acquired array images were
analyzed with the Feature Extraction software Version 11.0.1.1
(Agilent Technologies, Inc.). Quantile normalization and subsequent
data processing were performed using the GeneSpring GX V12 1
software package (Agilent Technologies, Inc.). Microarray was
performed by Kangchen Bio-tech Inc.
Reverse transcription-quantitative PCR
(RT-qPCR) validation of lncRNAs
Total RNA (2 µg) was reverse transcribed into cDNA
using SuperScriptTM III Reverse Transcriptase (cat. no. 18080093;
Invitrogen; Thermo Fisher Scientific, Inc.). LncRNA expression was
measured by qPCR using the SYBR Premix Ex Taq (Thermo Fisher
Scientific, Inc.) with an ABI PRISM® 7000 Sequence
Detection System (Thermo Fisher Scientific, Inc.). The total
reaction volume was 10 µl, including 5 µl Master Mix (2X), 2 µl
cDNA template, 0.5 µl forward primer, 0.5 µl reverse primer (10 µM)
and 2 µl double-distilled water. The qPCR reaction was performed
with an initial denaturation step of 10 min at 95°C, then 95°C (5
sec) and 60°C (60 sec) for a total of 40 cycles, with a final
extension step at 72°C for 5 min. All experiments were performed in
triplicate and all samples were normalized to GAPDH. The median for
each triplicate was used to calculate the relative lncRNA
concentrations (∆Cq=Cq median lncRNAs-Cq median GAPDH) (36). The primer sequences used were as
follows: GAPDH forward, 5′-GGGAAACTGTGGCGTGAT-3′ and reverse,
5′-GAGTGGGTGTCGCTGTTGA-3′; LURAP1L forward,
5′-CCTCCTCAGGCAAGAGATGGT-3′ and reverse,
5′-TGCTGCCTCTGCTGGTAATG-3′; and LURAP1L-AS1 forward,
5′-GAGCGGTCAAATAGAGGATAT-3′ and reverse,
5′-ATATCCTCTATTTGACCGCTC-3′.
Bioinformatics prediction and
dual-luciferase reporter assays
The LURAP1L-AS1 potential mRNA binding sites were
predicted using network analysis (http://atlasgeneticsoncology.org). The luciferase
reporter plasmid encoding both Renilla luciferase (hRluc)
pmiR-RB-REPORT™ the control firefly luciferase (hluc+)
(both Guangzhou RiboBio Co., Ltd.) were used for all assays. The
dual-luciferase reporter assays were performed by Shanghai GeneChem
Co., Ltd. according to the method described by Fish et al
(37). The 3′-untranslated region
(UTR) of the target gene or the relevant negative control was
constructed to the back of luciferase. The lncRNA LURAP1L-AS1 was
provided and constructed into the reporter plasmid pmir-GLO
(Shanghai GeneChem Co., Ltd.; LURAP1L-AS1 mimic). The empty
pmir-GLO plasmid served as the negative control for lncRNA
LURAP1L-AS1 (mimic control). Constructed vectors (or constructed
and negative vectors) were used to co-transfect HEK293T cells
(Shanghai GeneChem Co., Ltd.) into four experimental groups:
3′UTR-NC+LURAP1L-AS1-NC; 3′UTR-NC+LURAP1L-AS1;
3′UTR-LURAP1L+LURAP1L-AS1-NC; and 3′UTR- LURAP1L+LURAP1L-AS1.
HEK293T cells were seeded into 96-well plates and transfection
solution (100 µl) containing 0.5 µl Lipofectamine 2000®
(Invitrogen; Thermo Fisher Scientific, Inc.) and 25 ng reporter
plasmids with LURAP1L-AS1 mimic (50 nM) or mimic control (50 nM)
was added. The cells were washed with phosphate buffer saline and
then fully lysed with Passive Lysis Buffer 1× (Shanghai GeneChem
Co., Ltd.) for 20 min at 4°C 24 h after transfection.
Luciferase activities were measured using the
Dual-luciferase Reporter assay kit (Promega Corporation) and Centro
XS (Titertek-Berthold). The ratio of firefly luciferase to
Renilla luciferase in the same sample pore represented the
relative expression of luciferase. For the two groups transfected
with the same luciferase plasmid, the relative expression of
luciferase in the LURAP1L-AS1-NC group was normalized to 1 and the
relative expression of luciferase in the LURAP1L-AS1 group was
normalized to that in the LURAP1L-AS1-NC group. The normalized data
of the 3′UTR-NC+LURAP1L-AS1 group and relative expression of
luciferase in 3′UTR-NC+LURAP1L and 3′UTR-LURAP1L+LURAP1L-AS1 were
compared.
Co-immunoprecipitation (co-IP)
Co-IP analysis was performed as described previously
(38). Briefly, the cultured cells
were lysed using RIPA buffer (Beijing Solarbio Science &
Technology Co., Ltd.) containing 1% protease inhibitors. The
lysates were immunoprecipitated with anti-LURAP1L or anti-IKKα for
1 h at 37°C, followed by incubation with 100 µl Protein A Agarose
(cat. no. #9863; Cell Signaling Technology, Inc.) overnight at 4°C.
The next day, the Protein A Agarose-antigen-antibody complexes were
collected by centrifugation at 12,000 × g for 2 min at 4°C and the
immunoprecipitation-HAT buffer (cat. no. 10009330-1; Aimeijie
Technology Co., Ltd.) was used to wash the complexes five times.
Western blot was used to detect bound proteins.
Knockdown and overexpression of
LURAP1L-AS1
To observe the effects of LURAP1L-AS1 knockdown on
fibroblasts activated by PDGF-BB, three different small interfering
(si)RNAs that targeted LURAP1L-AS1 RNA and a scrambled siRNA
control were provided by Shanghai GenePharma Co. Ltd. The three
siRNAs were transfected into hOMF cells using Lipofectamine 2000
according to the manufacturer's instructions. Twenty-four hours
after transfection, the LURAP1L-AS1 expression levels were measured
through RT-qPCR and it was observed that siRNA-LURAP1L-AS1
(forward, 5′-GAGCGGTCAAATAGAGGATAT-3′ and reverse,
5′-ATATCCTCTATTTGACCGCTC-3′) yielded the highest degree of
LURAP1L-AS1 silencing. Subsequently, the LURAP1L-AS1-targeting
sequence was designed, synthesized and inserted into a
SuperSilencing shRNA Expression Vector System (Shanghai GenePharma
Co. Ltd.). An unrelated sequence lentiviral vector GV248 (Shanghai
GenePharma Co. Ltd.) served as a negative control. hOMF cells were
then plated into 6-well plates and allowed to adhere for 24 h. The
lentivirus was transfected according to the manufacturer's
instructions. Stably transfected cells were selected with puromycin
(MilliporeSigma) and confirmed through fluorescence microscopy and
RT-qPCR.
The overexpression of LURAP1L-AS1 in hOMF was
achieved using a transient transfection method. The primers for
LURAP1L-AS1-targeting sequence cloning were designed and
synthesized in the present study (LURAP1L-AS1-p1 forward,
TACCGGACTCAGATCTCGAGTTAGAATGATCTAATGAAAAC and reverse,
TACCGTGACTGCAGAATTCACAAATTGAAGAATATTTATTTTAGGTTAAAATATTTTTAAG;
Shanghai Genechem Co., Ltd.). The cloned LURAP1L-AS1 was verified
by sequencing (sequencing primer forward, CGCAAATGGGCGGTAGGCGTG and
reverse, AACGCACACCGGCCTTATTC) and subsequently cloned into a GV146
overexpression vector (Shanghai Genechem Co., Ltd.). The GV146
empty vector served as a negative control, while PDGF-BB served as
a positive control. hOMF cells were transfected using Lipofectamine
2000 according to the manufacturer's instructions. After culturing
for 3–4 days, RT-qPCR was performed to confirm cell
transfection.
Statistical analysis
All experiments were repeated at least three times
and data are presented as means ± standard errors of the mean.
Graphpad Prism 5.0 (GraphPad Software Inc.) was used to perform
statistical analysis. Differences between two groups were compared
with an independent samples t-test. Differences among three or more
groups were compared with one-way analysis of variance and the
Dunnett's post hoc test. P<0.05 was considered to indicate a
statistically significant difference.
Results
Effects of PDGF-BB on fibroblast
activation
PDGF-BB (10, 20 and 30 ng/ml) was used to stimulate
conventionally cultured fibroblasts for 72 h. The cells from the
third passage were used. Western blotting showed that all the three
concentrations of PDGF-BB could upregulate the expression of α-SMA,
FAP-α, PDGFR-β and p-PDGFR-β, while the effect of PDGF-BB was in a
dose-dependent manner (Fig. 1A). IF
confirmed that PDGF-BB upregulated the expression levels of α-SMA,
FAP-α and PDGFR-β, as shown by an increased fluorescence signal
(Fig. 1B). These results indicate
that PDGF-BB could induce fibroblast activation.
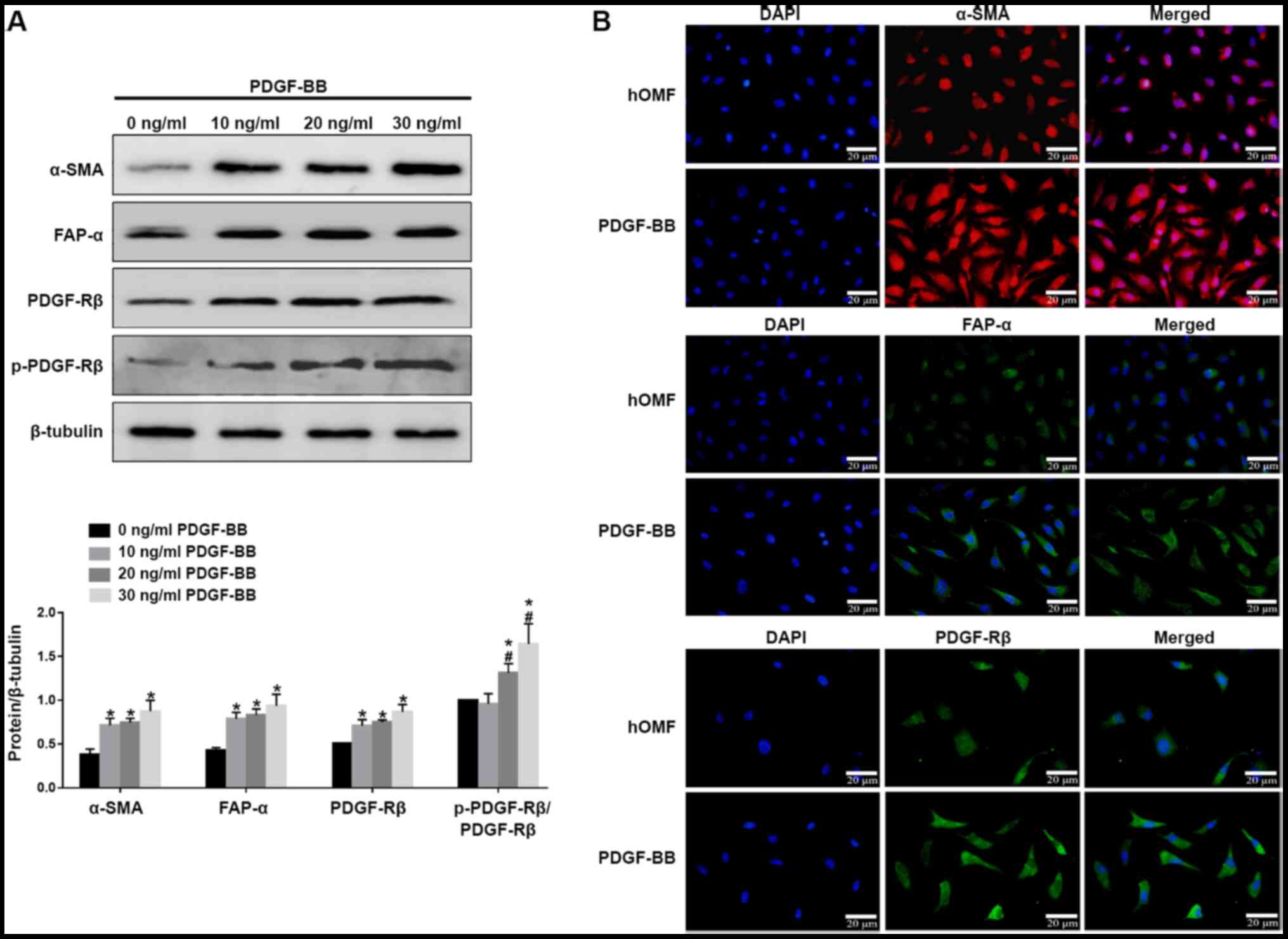 | Figure 1.PDGF-BB induced the formation of the
CAF phenotype in hOMF. (A) The cells were treated with PDGF-BB in a
dose-dependent manner (10, 20, 30 ng/ml) in 10% fetal bovine serum
for 72 h for three passages. The cells were subjected to western
blot analysis with antibodies against CAF markers α-SMA, FAP-α,
PDGFR-β and p-PDGFR-β. β-tubulin served as a loading control and
sample loading was 20 µg. (B) Fluorescence microscopy of hOMF
stained with α-SMA, FAP-α and PDGFR-β (probed with a primary and a
secondary antibody). Cells were counterstained with DAPI. Data were
expressed as means ± SEM (n=3). *P<0.05 vs. hOMF,
#P<0.05 vs. 10 ng/ml PDGF-BB. PDGF, platelet-derived
growth factor; CAF, cancer-associated fibroblast; hOMF, human oral
mucosa (p3) 500K fibroblast; α-SMA, α-smooth muscle actin; FAP-α,
fibroblast activation protein-α; PDGFR-β, platelet-derived growth
factor receptor-β; p, phosphorylated. |
Target gene prediction and
dual-luciferase reporter assay
As the aim of the present study was to identify the
lncRNA interaction with LURAP1L, a key regulator activating the
canonical NF-κB pathway, western blotting and RT-qPCR were used to
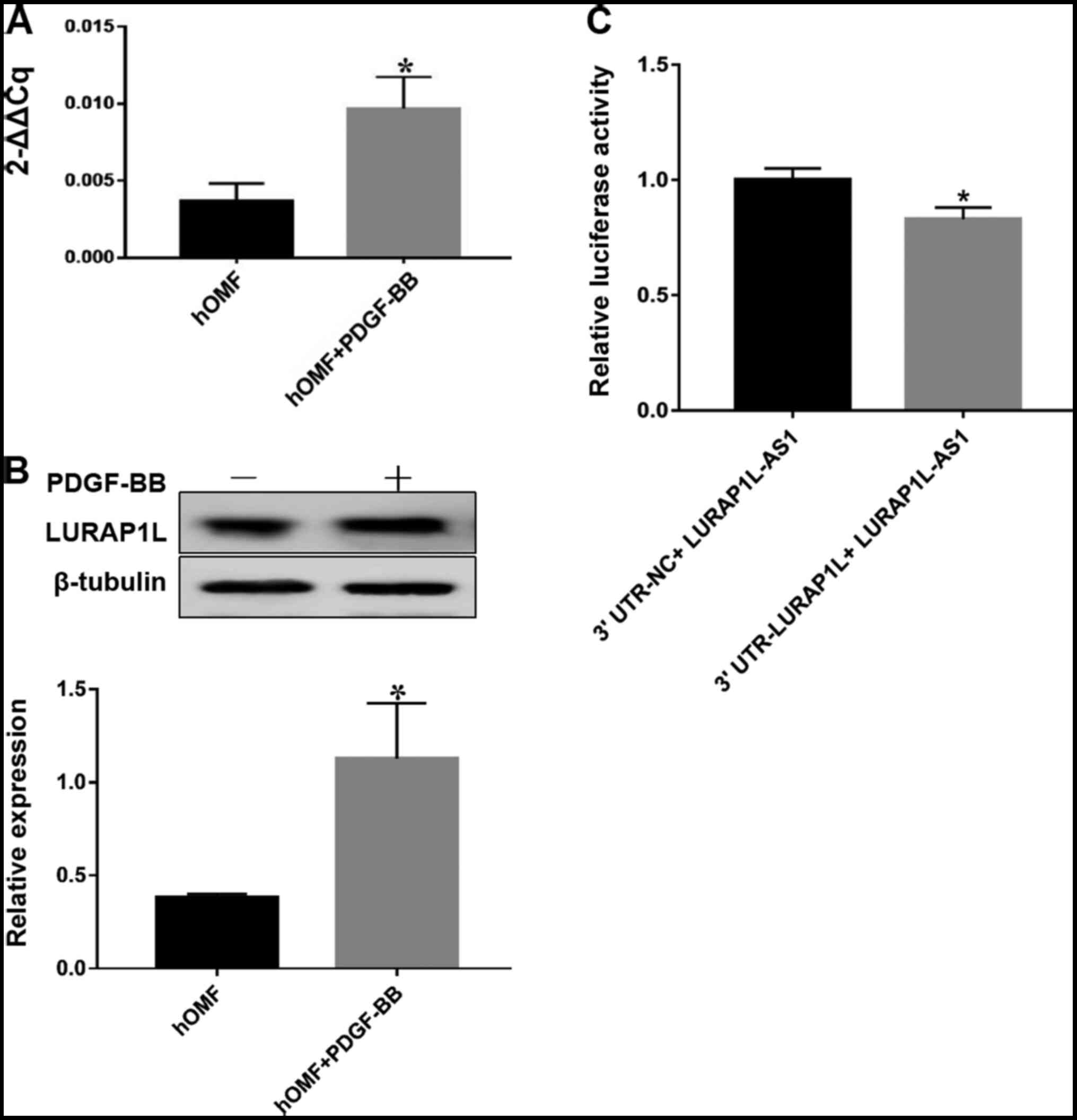
evaluate the expression of LURAP1L. The results indicated that
LURAP1L expression was upregulated by PDGF-BB (Fig. 2A and B). Next, we predicted
LURAP1L-AS1 may have an impact on the expression levels of LURAP1L,
as well as the binding sites between LURAP1L and LURAP1L-AS1 using
network analysis. The predicted results demonstrated that there was
a significant correlation between LURAP1L-AS1 and LURAP1L (Fig. S1). Furthermore, the dual-luciferase
reporter assay indicated that LURAP1L-AS1 significantly repressed
the activity of luciferase derived from RNAs containing the 3′UTR
of LURAP1L (Fig. 2C). This implies
that LURAP1L is the target gene of LURAP1L-AS1 and that PDGF-BB
induces the upregulation of LURAP1L via LURAP1L-AS1.
PDGF-BB regulates lncRNA expression in
fibroblasts, as confirmed by RT-PCR
In order to confirm the results above and the
mechanisms of activation of fibroblasts induced by PDGF-BB, an
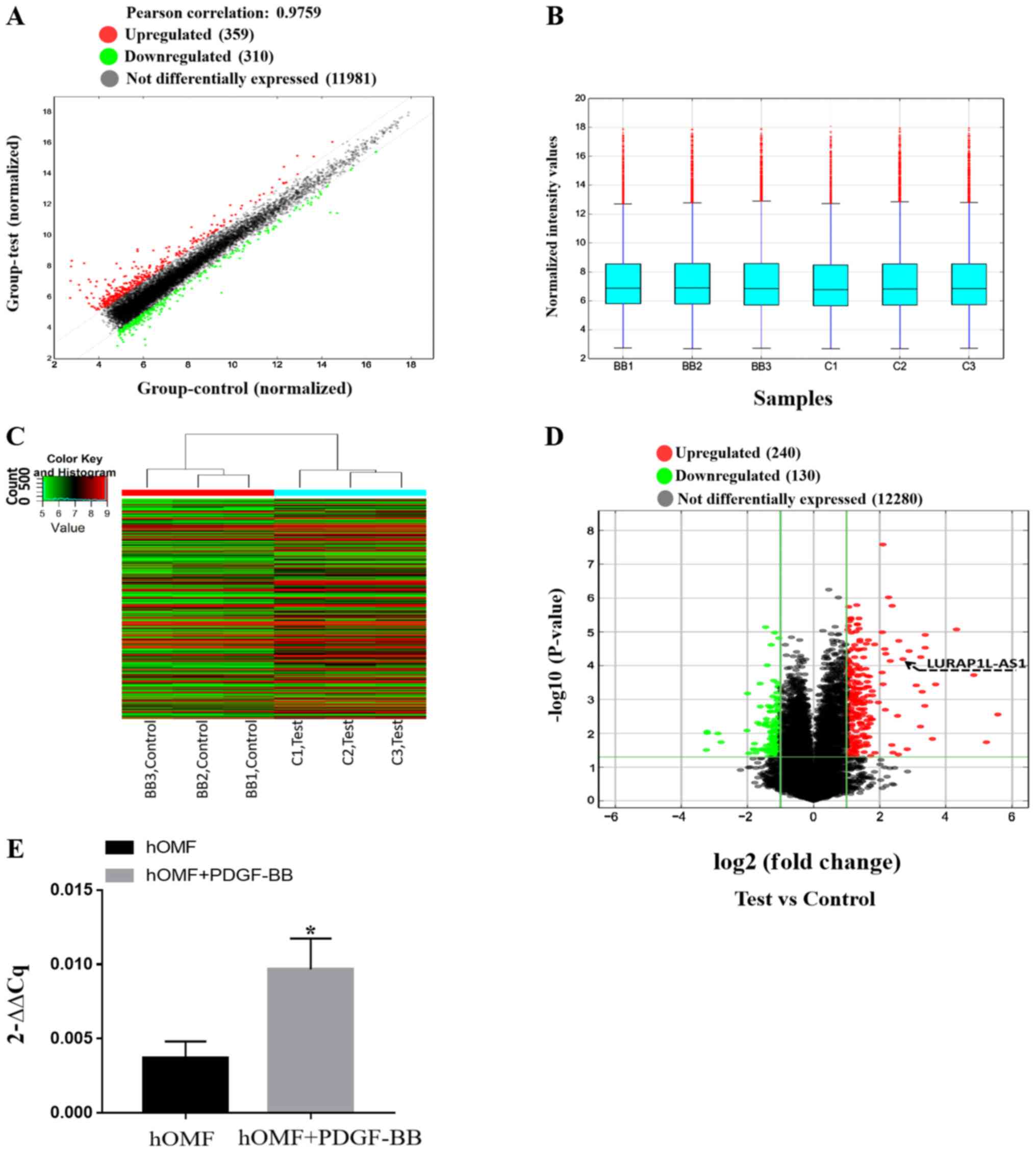
lncRNA microarray was used to screen the samples from fibroblasts
and PDGF-BB-activated fibroblasts. After normalization and data
filtering, 12,650 lncRNAs were identified. The heat map showed a
distinguishable lncRNA expression profile between the two groups
(Fig. 3A-C). Compared with the
control group, 240 lncRNAs were upregulated, and 130 were
downregulated (fold-change ≥2) as shown in Fig. 3D (P<0.05). Table I presents the lncRNAs that were up-
or downregulated by PDGF-BB (fold-change ≥5; P<0.05). Among
these, it was confirmed that LURAP1L-AS1 was upregulated, as
demonstrated by the arrow in Fig.
3D. RT-qPCR for LURAP1L-AS1 was also performed and the results
were consistent with the microarray results (P<0.05; Fig. 3E). These results indicated
significantly upregulated expression of LURAP1L-AS1 in fibroblasts
following PDGF-BB treatment and supported the prediction of the
present study that LURAP1L-AS1 activates LURAP1L in
fibroblasts.
 | Table I.Differentially expressed long
non-coding RNAs. |
Table I.
Differentially expressed long
non-coding RNAs.
| Probe name | P-value | Fold-change | LncRNA
expression | Gene symbol |
|---|
| ASHGV40002366 |
1.54×10−3 | 10.2773694 | Upregulated | LOC102724467 |
| ASHGV40016892 |
2.78×10−3 | 47.6213517 | Upregulated | RORA-AS1 |
| ASHGV40003735 |
1.69×10−6 | 5.186901 | Upregulated | SNHG8 |
| ASHGV40024576 |
2.78×10−4 | 28.7604505 | Upregulated | XLOC_013370 |
| ASHGV40035139 |
3.04×10−3 | 5.8165393 | Upregulated | LINC00886 |
| ASHGV40048333 |
6.30×10−3 | 9.386177 | Upregulated | G077644 |
| ASHGV40059227 |
5.59×10−5 | 9.4526926 | Upregulated | G090807 |
| ASHGV40010343 |
5.91×10−4 | 9.7206085 | Upregulated | G019348 |
| ASHGV40005652 |
3.70×10−5 | 7.4065884 | Upregulated | T042114 |
| ASHGV40035740 |
1.84×10−5 | 5.9734672 | Upregulated | T239636 |
| ASHGV40055486 |
8.45×10−6 | 19.9812999 | Upregulated | G090385 |
| ASHGV40033198 |
1.23×10−5 | 10.3854729 | Upregulated | RP4-756G23.5 |
| ASHGV40007819 |
3.57×10−4 | 12.9009956 | Upregulated | RP11-264E20.2 |
| ASHGV40051519 |
6.35×10−5 | 6.5042034 | Upregulated | LURAP1L-AS1 |
| ASHGV40041072 |
8.92×10−3 | 9.313185 | Downregulated | CTC-436K13.5 |
Inhibition of PDGFR-β affects LURAP1L-AS1-
LURAP1L/IKK/IκB/NF-κB signaling and CAF programming. To examine the
PDGF-BB-induced signaling pathway via PDGFR-β, a PDGFR-β tyrosine
kinase inhibitor, CP-673451 (Selleck Chemicals) was used prior to
constructing a fibroblast activation model using PDGF-BB (32). The results indicated that CP-673451
downregulated LURAP1L-AS1 and LURAP1L (Fig. 4A). In addition, western blotting
showed that CP-673451 downregulated α-SMA, FAP-α, IKKα and p-p65,
but upregulated IκBα (Fig. 4B).
These results demonstrate that PDGFR-β is involved in
LURAP1L-AS1-LURAP1L/IKK/IκB/NF-κB signaling and CAF
programming.
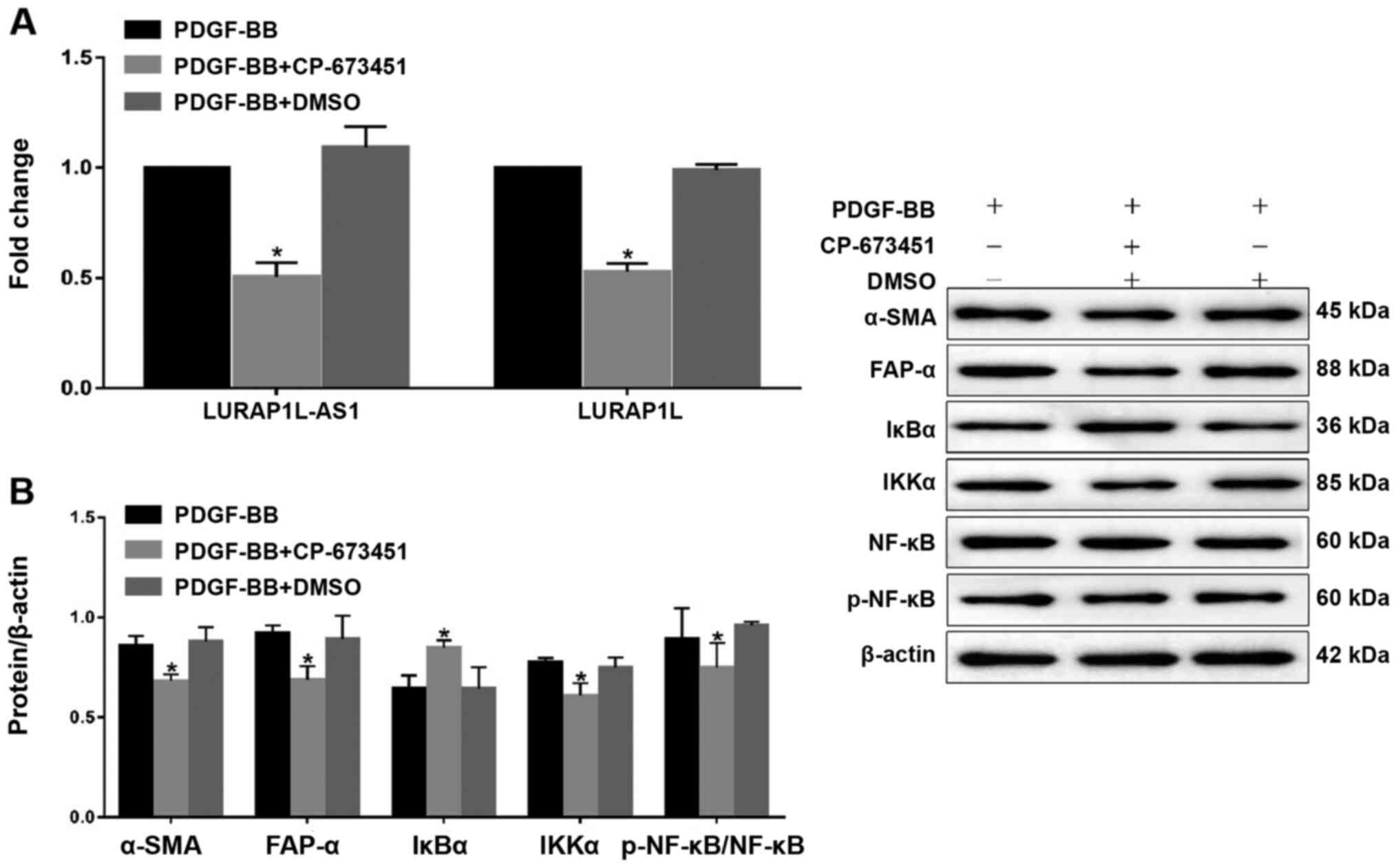 | Figure 4.Effects of the PDGFR-β inhibitor,
CP-673451 on LURAP1L-AS1-LURAP1L/IKK/IκB/NF-κB signaling and CAF
programming. (A) Reverse transcription-quantitative PCR analysis of
LURAP1L and LURAP1L-AS1 expression levels. (B) Western blot
analysis of FAP-α, α-SMA, IKKα, IκBα, NF-κB p65 and p-p65
expression following overexpression of LURAP1L-AS1 and treatment
with PDGF-BB. *P<0.05 vs. PDGF-BB. PDGF, platelet-derived growth
factor; LURAP1L, leucine-rich adaptor protein 1-like; LURAP1L-AS1,
LURAP1L antisense RNA 1; IKKα, IκB kinase α; IKK, I-κB kinase;
IκBα, nuclear factor of κ light polypeptide gene enhancer in
B-cells inhibitor α; NF-κB, nuclear factor-κB; CAF,
cancer-associated fibroblasts; p, phosphorylated; α-SMA, α-smooth
muscle actin; FAP-α, fibroblast activation protein-α. |
Knockdown and overexpression of lncRNA
LURAP1L-AS1 alter the effects of PDGF-BB on fibroblast activation
and IKKα/IκBα/p-NF-κB expression
To verify the role of LURAP1L-AS1 in PDGF-BB-induced
fibroblast activation, LURAP1L-AS1 was knocked down or
overexpressed in fibroblast cells, as confirmed by RT-qPCR
(Fig. 5A and B). The fibroblast
activation model was then established using these cells. The
results indicated that levels of the activation marker proteins
α-SMA, FAP-α, PDGFR-β and p-PDGFR-β were significantly lower after
knocking down LURAP1L-AS1 (Fig. 5C).
Furthermore, IKKα and p-p65 were downregulated, while IκBα was
upregulated by knocking down LURAP1L-AS1 (Fig. 5C; P<0.05). Contrasting results
were obtained by overexpressing LURAP1L-AS1 (Fig. 5D). The results indicated that the
LURAP1L-AS1/IKKα/IκBα/NF-κB axis might be involved in
PDGF-BB-induced fibroblast activation into CAFs. Furthermore,
LURAP1L-AS1 associated with PDGF-BB-induced fibroblast activation
and affected the NF-κB signaling pathway.
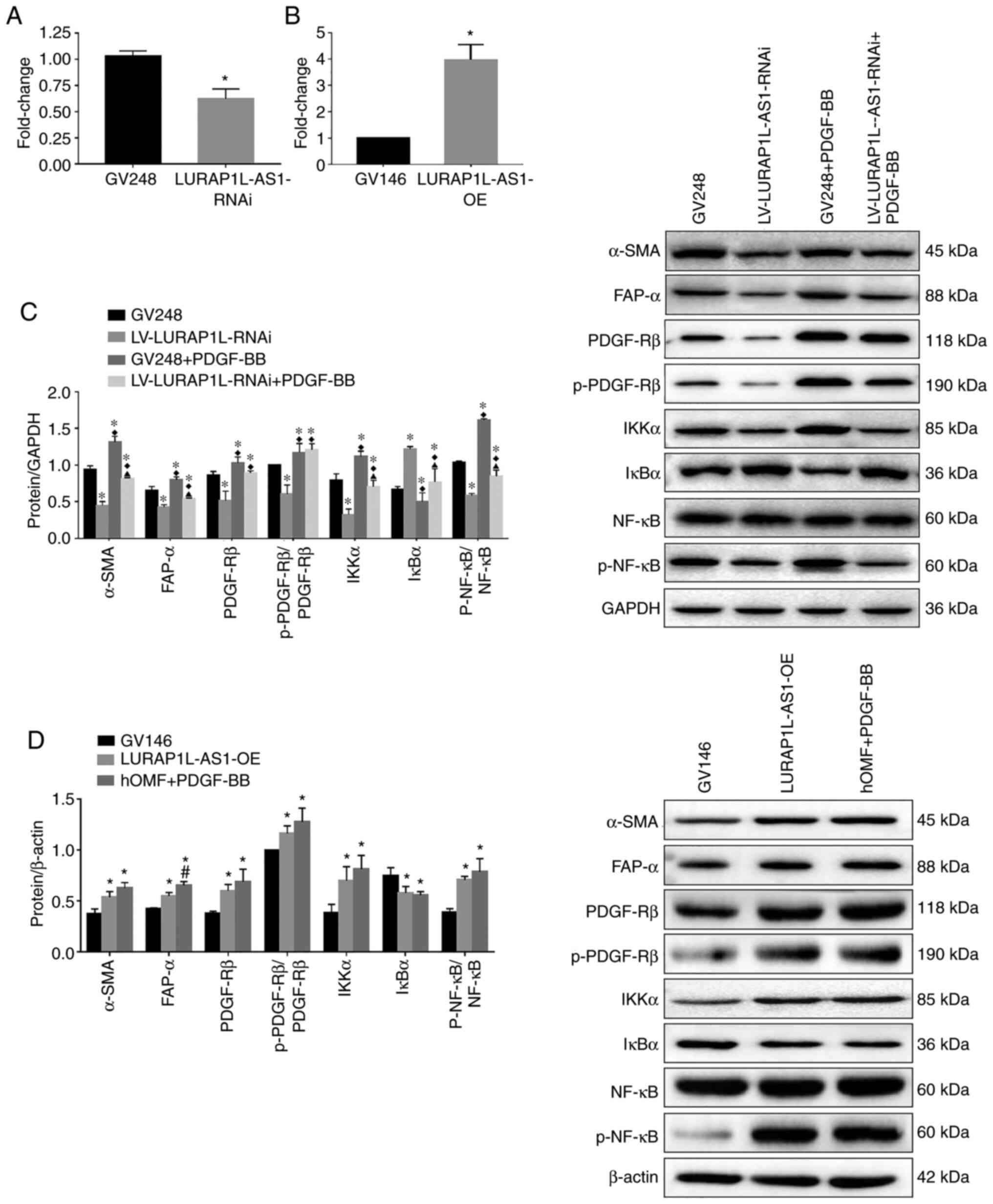 | Figure 5.Knockdown and overexpression of
lncRNA LURAP1L-AS1 alters the effects of PDGF-BB on fibroblast
activation and interaction between LURAP1L and IKKα. hOMF cells
were constructed for knocking down and overexpressing LURAP1L-AS1.
The fibroblast activation model was established using transfected
cells. Expression of lncRNA LURAP1L-AS1 in hOMF cells with
LURAP1L-AS1 (A) knocked down and (B) overexpressed. (C) Western
blot analysis of the expression of FAP-α, α-SMA, IKKα, IκBα, NF-κB
p65, and p-p65 after knocking down LURAP1L-AS1 and treatment with
PDGF-BB. *P<0.05 vs. GV248; ♦P<0.05 vs.
LURAP1L-AS1-RNAi; ▲P<0.05 vs. GV248+PDGF-BB. (D)
Western blot analysis of the expression of FAP-α, α-SMA, IKKα,
IκBα, NF-κB p65 and p-p65 after overexpressing LURAP1L-AS1 and
treatment with PDGF-BB. *P<0.05 vs. GV146; #P<0.05
vs. LURAP1L-AS1-OE. lncRNA, long non-coding RNA; OE,
overexpression; GV248, negative control; GV146, negative control;
PDGF, platelet-derived growth factor; hOMF, human oral mucosa (p3)
500K fibroblast; LURAP1L, leucine-rich adaptor protein 1-like;
LURAP1L-AS1, LURAP1L antisense RNA 1; p, phosphorylated; α-SMA,
α-smooth muscle actin; FAP-α, fibroblast activation protein-α;
IKKα, IκB kinase α; IκBα, nuclear factor of κ light polypeptide
gene enhancer in B-cells inhibitor α; RNAi, RNA interference. |
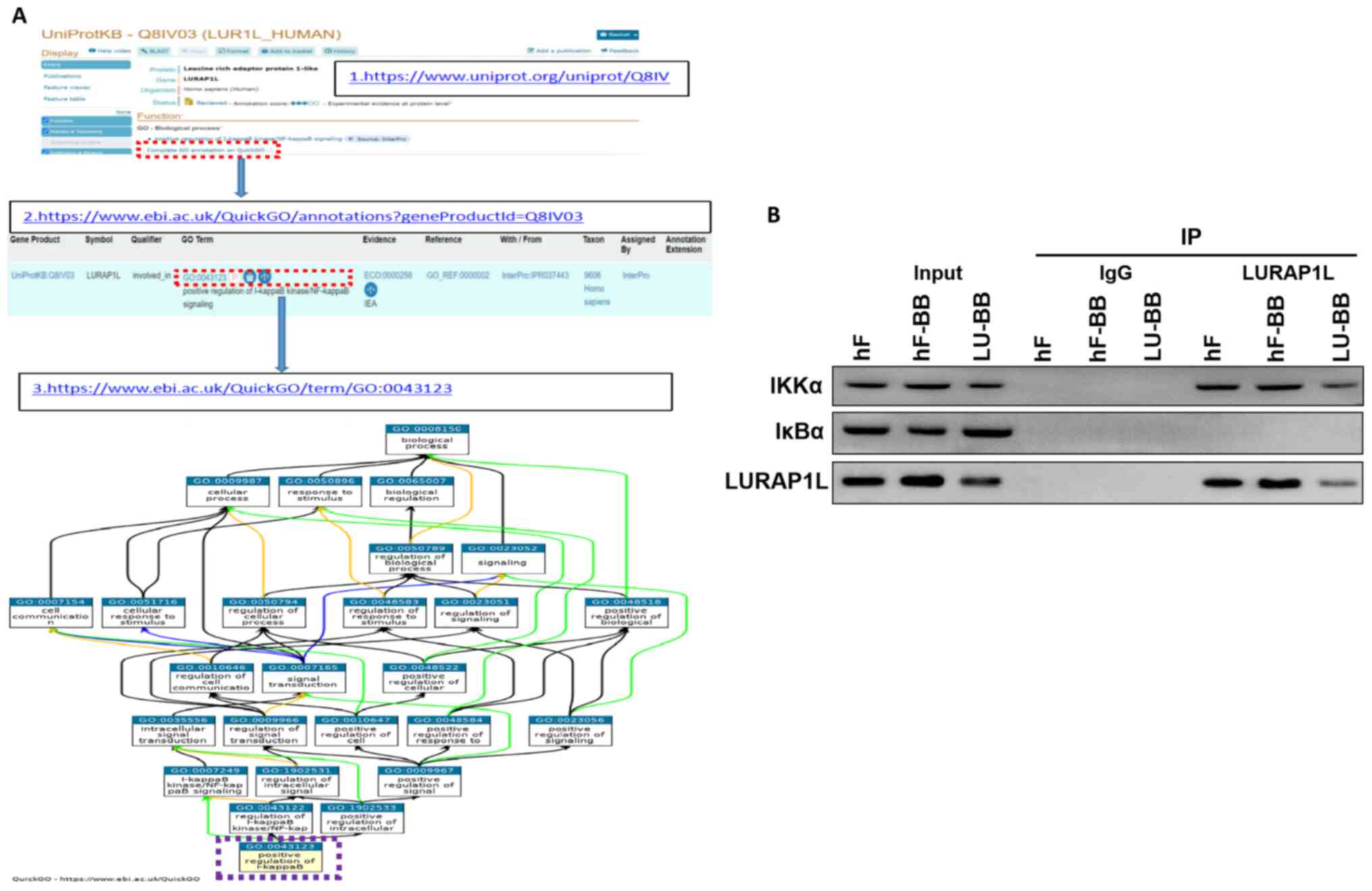
Co-IP indicates an interaction between
LURAP1L and IKKα
The results demonstrated that LURAP1L-AS1 could
affect the changes in the NF-κB signaling pathway and that LURAP1L
was a target gene of LURAP1L-AS1, but the regulatory effect of
LURAP1L on the NF-κB signaling pathway had to be verified. Through
protein-protein interaction analysis, an interaction was identified
between LURAP1L and IKKα in the NF-κB signaling pathway (Fig. 6A). Therefore, co-IP was performed for
verification. The results demonstrated that the IKKα protein was
detected in the interacting protein of LURAP1L, indicating that
LURAP1L interacts with IKKα and promotes the activation of the
NF-κB signaling pathway to affect the activation of fibroblasts
(Fig. 6B). It was also observed that
lncRNA LURAP1L-AS1 knockdown weakened the interaction between
LURAP1L and IKKα (Fig. 6B).
Discussion
CAFs are matrix fibroblasts that were first isolated
from prostate cancer tissues by Olumi et al (39) in 1999. In the present study, the
results showed that compared with the normal group, α-SMA, FAP-α,
PDGFR-β and p-PDGFR-β were significantly increased by PDGF-BB,
indicating fibroblast activation. The lncRNA microarray showed that
the expression of LURAP1L-AS1 was upregulated by PDGF-BB.
Furthermore, the present study showed that the target gene of
LURAP1L-AS1 is LURAP1L. The LURAP1L protein interacts with IKKα to
activate NF-κB, thereby promoting the activation of NFs.
CAFs are derived from the host's fibroblasts and are
induced by a variety of cytokines secreted by tumor cells, such as
TGF-β, PDGF and fibroblast growth factor (40). CAFs specifically express α-SMA, FAP-α
and PDGFR-β (13). The present study
showed that the expression of α-SMA, FAP-α, PDGFR-β and p-PDGFR-β
were upregulated in fibroblasts induced by PDGF-BB, indicating that
PDGF-BB could activate NFs into CAFs. This is consistent with the
results by Zhang et al (20).
Indeed, CAFs are mainly derived from resting fibroblasts (NFs) in
the matrix. When stimulated by external factors, NFs will activate
into CAFs with very different shapes and functions. Another
previous study showed that the overexpression of PDGF-BB and its
receptor could promote the occurrence and development of tumors and
was closely related to CAFs (41).
Aoto et al (42) and Rizvi
et al (43) suggested the
blockade of PDGFRβ as a potential strategy to prevent the formation
of CAFs, supporting the role of PDGF-BB in the formation of CAFs in
the TME, which is consistent with the present study.
CAF activation can be regulated by many factors,
such as growth factors, inflammatory factors, transcription
factors, hypoxia, reactive oxygen species and non-coding RNA
(44,45). The biological functions of lncRNA are
complex and they regulate transcription and post-transcription.
Tumorigenesis and development are closely related to tumor cell
proliferation, invasion, metastasis and recurrence. lnc003875 also
has a significant regulatory effect on CAF activation (46). The present study found that lncRNA
LURAP1L-AS1 expression was significantly upregulated during
PDGF-BB-induced fibroblast activation. Following RNA interference,
lncRNA LURAP1L-AS1 could inhibit the expression of the activation
marker proteins α-SMA, FAP-α, PDGFR-β and p-PDGFR-β, indicating
that the lncRNA LURAP1L-AS1 is involved in the process by which
PDGF-BB activates fibroblasts.
The present study showed that the LURAP1L-
AS1/LURAP1L/IKKα/IκBα/NF-κB axis might be involved in
PDGF-BB-induced fibroblast activation into CAFs. NF-κB primarily
exists in eukaryotic cells. It is a multidirectional and
multifunctional nuclear transcription factor that plays a pivotal
role in mediating intracellular signal transduction to promote
tumor cell proliferation, inhibit apoptosis, promote migration and
stimulate angiogenesis. NF-κB in tumor cells and the TME is
continuously activated, and the activation of NF-κB depends on the
decrease in the expression of IκB, which is the inhibitor of NF-κB.
The phosphorylation of IKK is closely associated with this process.
Zheng et al (47) found that
downregulation of the cell membrane protein CD146 in pancreatic
cancer stroma could also stimulate NF-κB signaling and promote CAF
activation. Liu et al (29)
showed that TNF-α could activate CAFs by enhancing the
transcriptional activity of NF-κB, thereby promoting tumorigenesis
and cancer development. Reactive oxygen species produced by tumors
induce the expression of the chloride intracellular channel 4 and
C-C motif chemokine ligand 2 in CAFs, stimulate the expression of
TGF-β1 and NF-κB and induce CAF activation (30,48). The
abovementioned studies showed that the NF-κB signaling pathway
plays an important role in the activation of CAFs.
LURAP1L is a key regulator that activates the
canonical NF-κB pathway (31). In
the present study, potential direct mRNA targets of LURAP1L-AS1
were identified through bioinformatics and LURAP1L was identified
as the target gene of LURAP1L-AS1. Additionally, its encoding
protein LURAP1L interacts with IKKα, which was demonstrated by
co-IP. Knockdown of LURAP1L-AS1 downregulated the expression levels
of IKKα and p-NF-κB and increased the expression of IκBα,
indicating that LURAP1L-AS1 may have a regulatory effect on the
NF-κB signaling pathway. Therefore, the present study showed that
the LURAP1L-AS1/LURAP1L/IKK/IκB/NF-κB axis plays an important role
in the regulation of PDGF-BB-induced fibroblast activation.
However, additional studies are required to determine the exact
mechanisms involved in this process. Furthermore, the present study
focused on LURAP1L-AS1, but other lncRNAs are also involved in CAF
activation and require investigation in the future.
Thus, the present study identified that the LURAP1L-
AS1/LURAP1L/IKK/IĸB/NF-κB axis plays an important regulatory role
in PDGF-BB-induced fibroblast activation into CAFs, indicating that
this axis might represent a potential target for the treatment of
OSCC.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant no. 81660448 and
81360401), the Special Health Technical Personnel Training program
of Yunnan, China (grant no. L-201612) and the Natural Science
Foundation of Yunnan, China (grant number 2017FE468-006).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XR and LL performed the experiments, participated in
collecting data and drafted the manuscript. YH and LB designed the
current study and confirm the authenticity of all the raw data. KL
and JW analyzed the data and performed the experiments. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Argiris A, Karamouzis MV, Raben D and
Ferris RL: Head and neck cancer. Lancet. 371:1695–1709. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wei WI and Sham JS: Nasopharyngeal
carcinoma. Lancet. 365:2041–2054. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
NCCN Clinical Practice Guidelines in
Oncology (NCCN Guidelines). Head and Neck Cancers. Version 1.2020.
National Comprehensive Cancer Network; Fort Washington: 2020
|
|
5
|
Li CC, Shen Z, Bavarian R, Yang F and
Bhattacharya A: Oral cancer: Genetics and the role of precision
medicine. Surg Oncol Clin N Am. 29:127–144. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Reina-Campos M, Moscat J and Diaz-Meco M:
Metabolism shapes the tumor microenvironment. Curr Opin Cell Biol.
48:47–53. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Brouty-Boyé D: Developmental biology of
fibroblasts and neoplastic disease. Prog Mol Subcell Biol.
40:55–77. 2005. View Article : Google Scholar
|
|
8
|
Kalluri R: The biology and function of
fibroblasts in cancer. Nat Rev Cancer. 16:582–598. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wu W, Zaal EA, Berkers CR, Lemeer S and
Heck AJR: CTGF/VEGFA-activated fibroblasts promote tumor migration
through Micro-environmental modulation. Mol Cell Proteomics.
17:1502–1514. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Abdul-Wahid A, Cydzik M, Fischer NW,
Prodeus A, Shively JE, Martel A, Alminawi S, Ghorab Z, Berinstein
NL and Gariépy J: Serum-derived carcinoembryonic antigen (CEA)
activates fibroblasts to induce a local re-modeling of the
extracellular matrix that favors the engraftment of CEA-expressing
tumor cells. Int J Cancer. 143:1963–1977. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhu Y, Zhang L, Zha H, Yang F, Hu C, Chen
L, Guo B and Zhu B: Stroma-derived Fibrinogen-like protein 2
activates cancer-associated fibroblasts to promote tumor growth in
lung cancer. Int J Biol Sci. 13:804–814. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tao L, Huang G, Song H, Chen Y and Chen L:
Cancer associated fibroblasts: An essential role in the tumor
microenvironment. Oncol Lett. 14:2611–2620. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sharon Y, Alon L, Glanz S, Servais C and
Erez N: Isolation of normal and cancer-associated fibroblasts from
fresh tissues by Fluorescence Activated Cell Sorting (FACS). J Vis
Exp. e44252013.doi: 10.3791/4425. PubMed/NCBI
|
|
14
|
Ishii G, Ochiai A and Neri S: Phenotypic
and functional heterogeneity of cancer-associated fibroblast within
the tumor microenvironment. Adv Drug Deliv Rev. 99:186–196. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ziani L, Chouaib S and Thiery J:
Alteration of the antitumor immune response by cancer-associated
fibroblasts. Front Immunol. 9:4142018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kashima H, Noma K, Ohara T, Kato T,
Katsura Y, Komoto S, Sato H, Katsube R, Ninomiya T, Tazawa H, et
al: Cancer-associated fibroblasts (CAFs) promote the lymph node
metastasis of esophageal squamous cell carcinoma. Int J Cancer.
144:828–840. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Huber L, Birk R, Rotter N, Aderhold C,
Lammert A, Jungbauer F and Kramer B: Effect of small-molecule
tyrosine kinase inhibitors on PDGF-AA/BB and PDGFRα/β expression in
SCC according to HPV16 Status. Anticancer Res. 40:825–835. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ouyang L, Zhang K, Chen J, Wang J and
Huang H: Roles of platelet-derived growth factor in vascular
calcification. J Cell Physiol. 233:2804–2814. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Heldin CH, Lennartsson J and Westermark B:
Involvement of platelet-derived growth factor ligands and receptors
in tumorigenesis. J Intern Med. 283:16–44. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang D, Wang Y, Shi Z, Liu J, Sun P, Hou
X, Zhang J, Zhao S, Zhou BP and Mi J: Metabolic reprogramming of
cancer-associated fibroblasts by IDH3α downregulation. Cell Rep.
10:1335–1348. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Maass PG, Luft FC and Bahring S: Long
non-coding RNA in health and disease. J Mol Med (Berl). 92:337–346.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chang L, Wang G, Jia T, Zhang L, Li Y, Han
Y, Zhang K, Lin G, Zhang R, Li J and Wang L: Armored long
non-coding RNA MEG3 targeting EGFR based on recombinant MS2
bacteriophage virus-like particles against hepatocellular
carcinoma. Oncotarget. 7:23988–24004. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhu M, Zhang C, Chen D, Chen S and Zheng
H: lncRNA MALAT1 potentiates the progression of tongue squamous
cell carcinoma through regulating miR-140-5p-PAK1 pathway. Onco
Targets Ther. 12:1365–1377. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kolenda T, Guglas K, Ryś M, Bogaczynska M,
Teresiak A, Bliźniak R, Łasińska I, Mackiewicz J and Lamperska KM:
Biological role of long non-coding RNA in head and neck cancers.
Rep Pract Oncol Radiother. 22:378–388. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ding L, Ren J, Zhang D, Li Y, Huang X, Hu
Q, Wang H, Song Y, Ni Y and Hou Y: A novel stromal lncRNA signature
reprograms fibroblasts to promote the growth of oral squamous cell
carcinoma via LncRNA-CAF/interleukin-33. Carcinogenesis.
39:397–406. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhao L, Ji G, Le X, Wang C, Xu L, Feng M,
Zhang Y, Yang H, Xuan Y, Yang Y, et al: Long noncoding RNA
LINC00092 acts in cancer-associated fibroblasts to drive glycolysis
and progression of ovarian cancer. Cancer Res. 77:1369–1382. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Freitas R and Fraga CAM: NF-κB-IKKβ
pathway as a target for drug development: Realities, challenges and
perspectives. Curr Drug Targets. 19:1933–1942. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Park MH and Hong JT: Roles of NF-kappaB in
cancer and inflammatory diseases and their therapeutic approaches.
Cells. 5:152016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu J, Chen S, Wang W, Ning BF, Chen F,
Shen W, Ding J, Chen W, Xie WF and Zhang X: Cancer-associated
fibroblasts promote hepatocellular carcinoma metastasis through
chemokine-activated hedgehog and TGF-β pathways. Cancer Lett.
379:49–59. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li X, Xu Q, Wu Y, Li J, Tang D, Han L and
Fan Q: A CCL2/ROS autoregulation loop is critical for
cancer-associated fibroblasts-enhanced tumor growth of oral
squamous cell carcinoma. Carcinogenesis. 35:1362–1370. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jing Z, Yuan X, Zhang J, Huang X, Zhang Z,
Liu J, Zhang M, Oyang J, Zhang Y, Zhang Z and Yang R: Chromosome 1
open reading frame 190 promotes activation of NF-κB canonical
pathway and resistance of dendritic cells to tumor-associated
inhibition in vitro. J Immunol. 185:6719–6727. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Saini H, Rahmani Eliato K, Veldhuizen J,
Zare A, Allam M, Silva C, Kratz A, Truong D, Mouneimne G, LaBaer J,
et al: The role of tumor-stroma interactions on desmoplasia and
tumorigenicity within a microengineered 3D platform. Biomaterials.
247:1199752020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Faghihi MA and Wahlestedt C: Regulatory
roles of natural antisense transcripts. Nat Rev Mol Cell Biol.
10:637–643. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ørom UA, Derrien T, Beringer M, Gumireddy
K, Gardini A, Bussotti G, Lai F, Zytnicki M, Notredame C, Huang Q,
et al: Long noncoding RNAs with enhancer-like function in human
cells. Cell. 1:46–58. 2010. View Article : Google Scholar
|
|
35
|
Cabili MN, Trapnell C, Goff L, Koziol M,
Tazon-Vega B, Regev A and Rinn JL: Integrative annotation of human
large intergenic noncoding RNAs reveals global properties and
specific subclasses. Genes Dev. 25:1915–1927. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fish JE, Santoro MM, Morton SU, Yu S, Yeh
RF, Wythe JD, Ivey KN, Bruneau BG, Stainier DY and Srivastava D:
miR-126 regulates angiogenic signaling and vascular integrity. Dev
Cell. 15:272–284. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang S, Zhu M, Wang Q, Hou Y, Li L, Weng
H, Zhao Y, Chen D, Ding H, Guo J and Li M: Alpha-fetoprotein
inhibits autophagy to promote malignant behaviour in hepatocellular
carcinoma cells by activating PI3K/AKT/mTOR signalling. Cell Death
Dis. 9:10272018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Olumi AF, Grossfeld GD, Hayward SW,
Carroll PR, Tlsty TD and Cunha GR: Carcinoma-associated fibroblasts
direct tumor progression of initiated human prostatic epithelium.
Cancer Res. 59:5002–5011. 1999.PubMed/NCBI
|
|
40
|
Lu Y, Chen R, Ma JY, Wang LP, Qiu LL, Wang
CP, Yan JC and Liu PJ: Platelet derived growth factor-BB regulates
phenotype transformation of pulmonary artery smooth muscle cells
via SIRT3 affecting glycolytic pathway. Zhonghua Xin Xue Guan Bing
Za Zhi. 47:993–999. 2019.(In Chinese). PubMed/NCBI
|
|
41
|
Gialeli C, Nikitovic D, Kletsas D,
Theocharis AD, Tzanakakis GN and Karamanos NK: PDGF/PDGFR signaling
and targeting in cancer growth and progression: Focus on tumor
microenvironment and cancer-associated fibroblasts. Curr Pharm Des.
20:2843–2848. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Aoto K, Ito K and Aoki S: Complex
formation between platelet-derived growth factor receptor beta and
transforming growth factor beta receptor regulates the
differentiation of mesenchymal stem cells into cancer-associated
fibroblasts. Oncotarget. 9:34090–34102. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Rizvi S, Mertens JC, Bronk SF, Hirsova P,
Dai H, Roberts LR, Kaufmann SH and Gores GJ: Platelet-derived
growth factor primes cancer-associated fibroblasts for apoptosis. J
Biol Chem. 289:22835–22849. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Shiga K, Hara M, Nagasaki T, Sato T,
Takahashi H and Takeyama H: Cancer-associated fibroblasts: Their
characteristics and their roles in tumor growth. Cancers (Basel).
7:2443–2458. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Primac I, Maquoi E, Blacher S, Heljasvaara
R, Van Deun J, Smeland HY, Canale A, Louis T, Stuhr L, Sounni NE,
et al: Stromal integrin α11 regulates PDGFR-β signaling and
promotes breast cancer progression. J Clin Invest. 129:4609–4628.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhang S, Tao X, Cao Q, Feng X, Wu J, Yu H,
Yu Y, Xu C and Zhao H: lnc003875/miR-363/EGR1 regulatory network in
the carcinoma -associated fibroblasts controls the angiogenesis of
human placental site trophoblastic tumor (PSTT). Exp Cell Res.
387:1117832020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zheng B, Ohuchida K, Chijiiwa Y, Zhao M,
Mizuuchi Y, Cui L, Horioka K, Ohtsuka T, Mizumoto K, Oda Y, et al:
CD146 attenuation in cancer-associated fibroblasts promotes
pancreatic cancer progression. Mol Carcinog. 55:1560–1572. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yao Q, Qu X, Yang Q, Wei M and Kong B:
CLIC4 mediates TGF-beta1-induced fibroblast-to-myofibroblast
transdifferentiation in ovarian cancer. Oncol Rep. 22:541–548.
2009.PubMed/NCBI
|
|
49
|
Binns D, Dimmer E, Huntley R, Barrell D,
O'Donovan C and Apweiler R: QuickGO: a web-based tool for Gene
Ontology searching. Bioinformatics. 25:3045–3046. 2009. View Article : Google Scholar : PubMed/NCBI
|