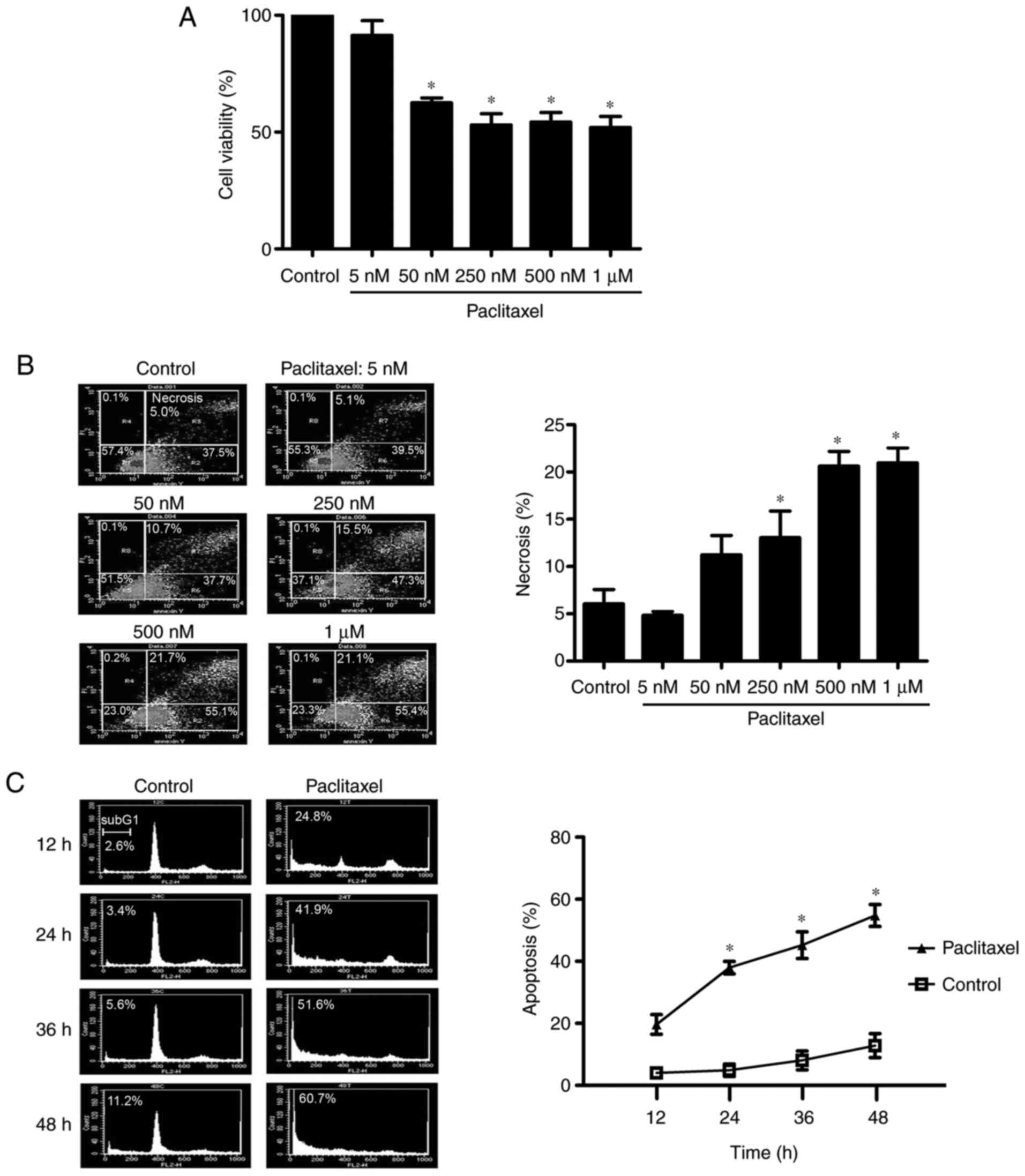
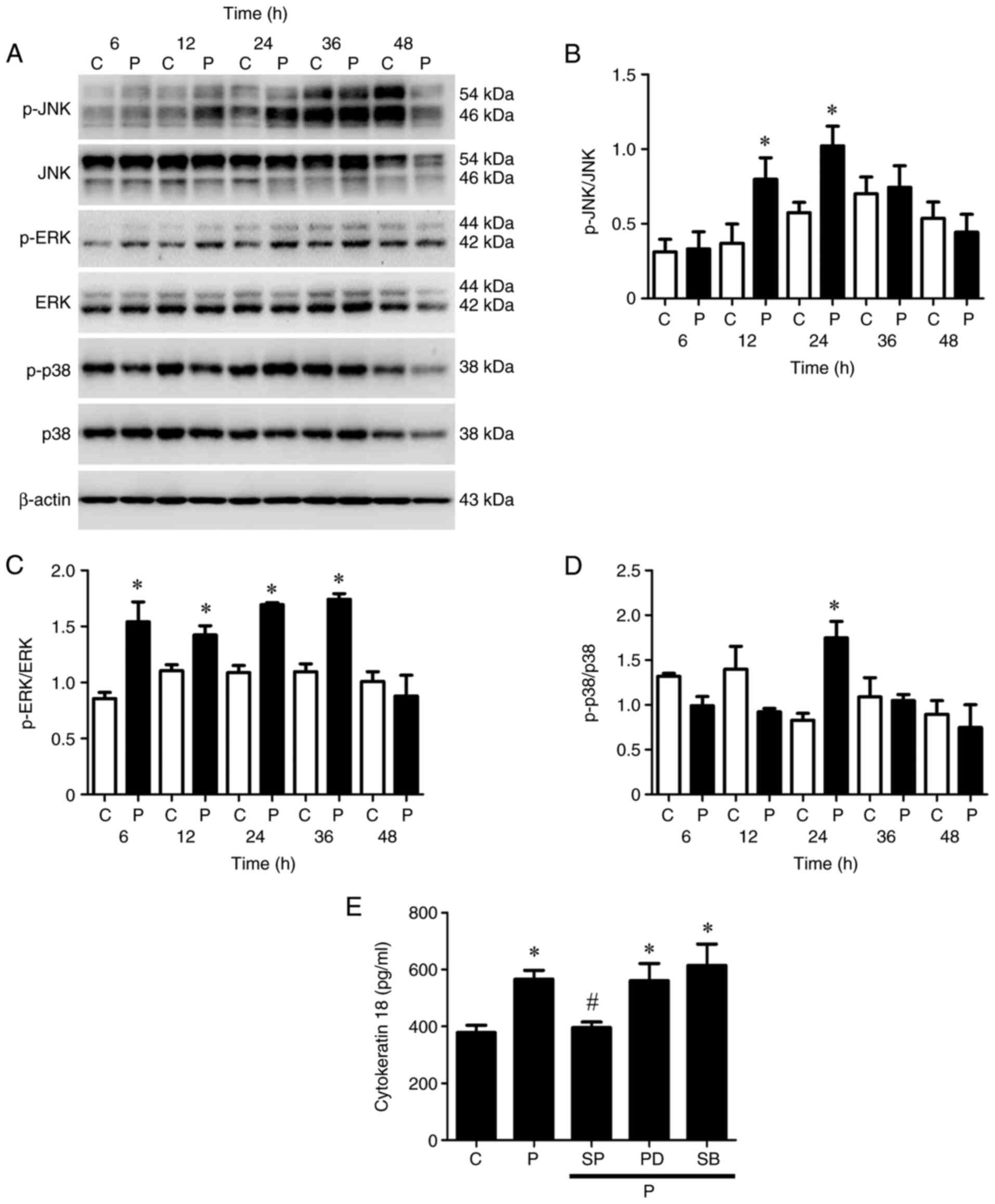
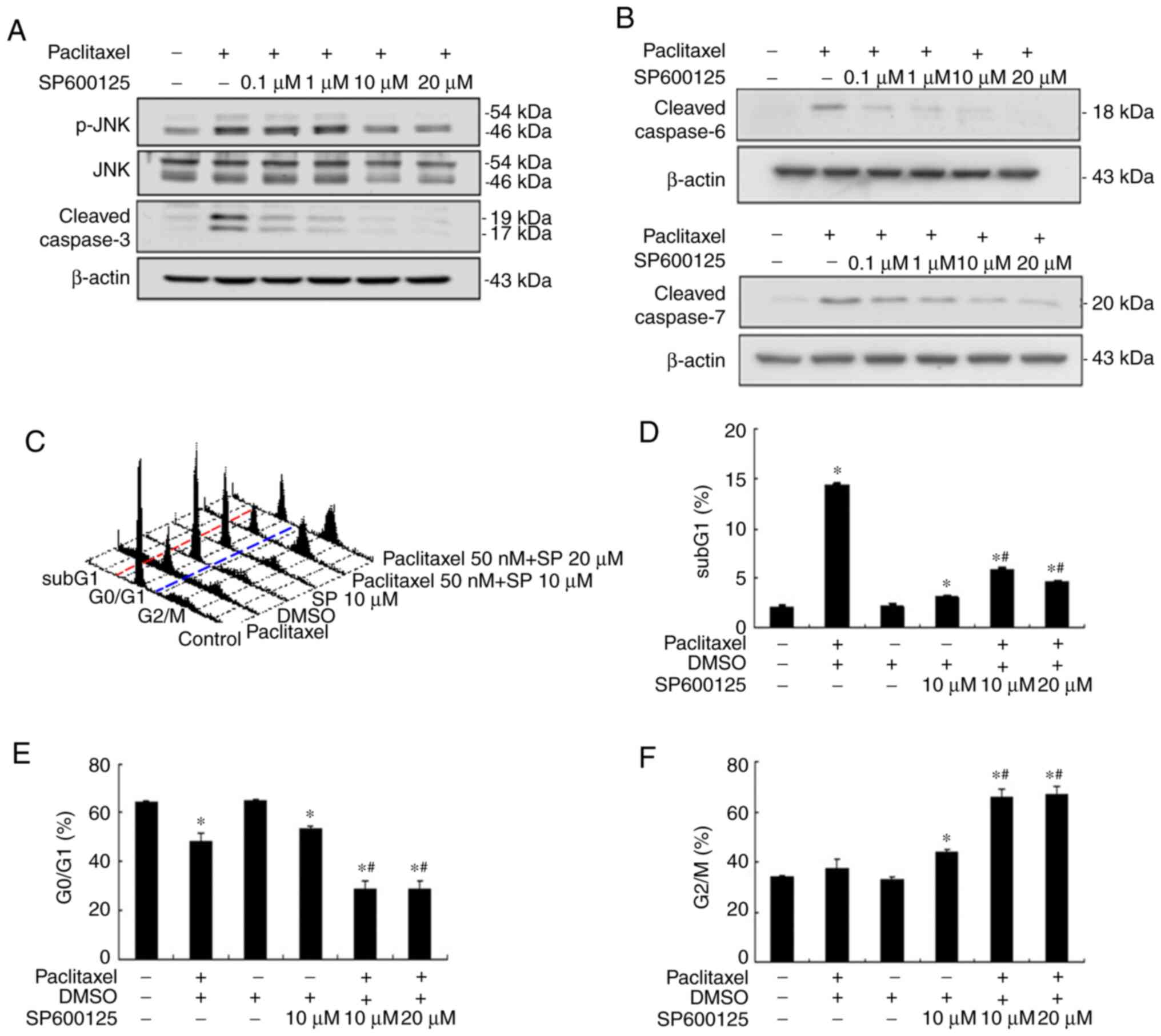
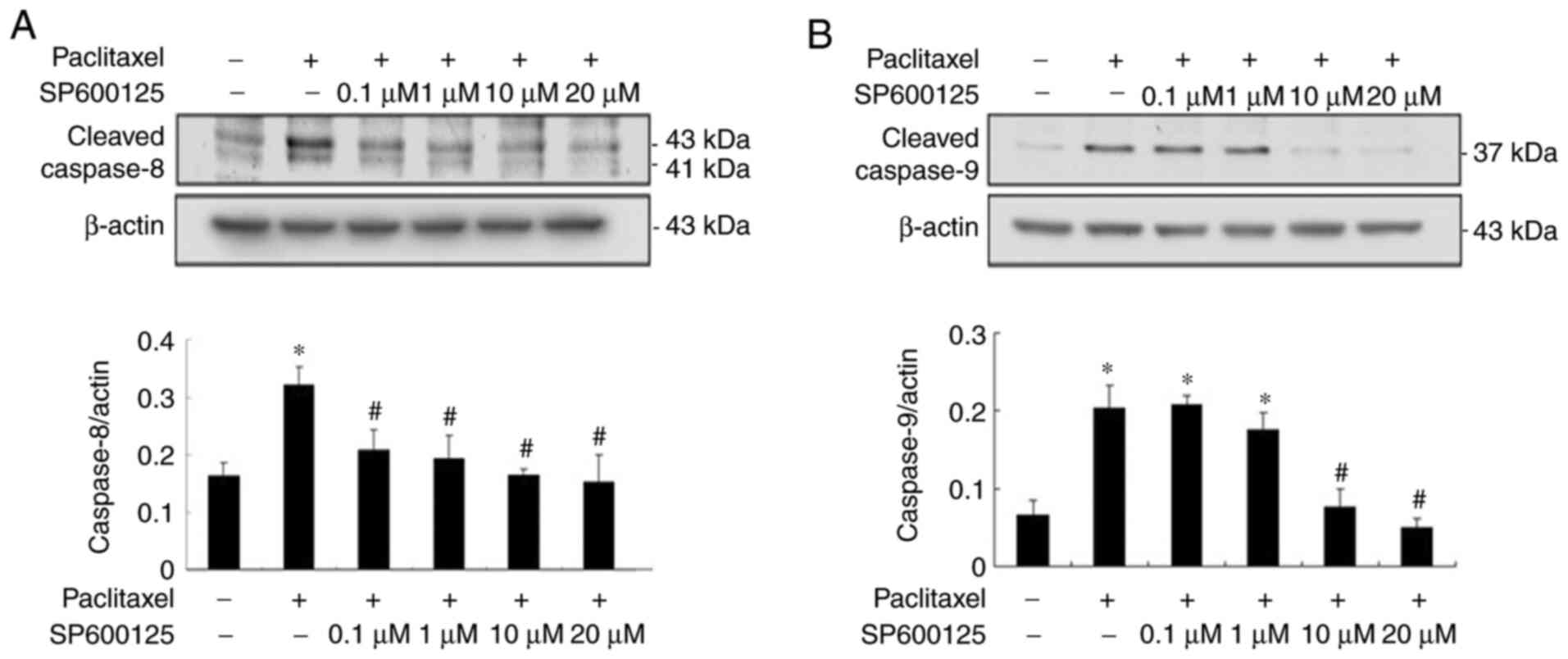
|
1
|
Rades D, Seidl D, Wollenberg B, Schild SE
and Hakim SG: Radiochemotherapy with paclitaxel for recurrent
previously irradiated squamous cell carcinoma of the head and neck.
Anticancer Res. 36:5463–5468. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Forastiere AA, Goepfert H, Maor M, Pajak
TF, Weber R, Morrison W, Glisson B, Trotti A, Ridge JA, Chao C, et
al: Concurrent chemotherapy and radiotherapy for organ preservation
in advanced laryngeal cancer. N Engl J Med. 349:2091–2098. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Psyrri A, Kwong M, DiStasio S, Lekakis L,
Kassar M, Sasaki C, Wilson LD, Haffty BG, Son YH, Ross DA, et al:
Cisplatin, fluorouracil, and leucovorin induction chemotherapy
followed by concurrent cisplatin chemoradiotherapy for organ
preservation and cure in patients with advanced head and neck
cancer: Long-term follow-up. J Clin Oncol. 22:3061–3069. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lo Nigro C, Denaro N, Merlotti A and
Merlano M: Head and neck cancer: Improving outcomes with a
multidisciplinary approach. Cancer Manag Res. 9:363–371. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Adamo V, Ferraro G, Pergolizzi S, Sergi C,
Laudani A, Settineri N, Alafaci E, Scimone A, Spano F and Spitaleri
G: Paclitaxel and cisplatin in patients with recurrent and
metastatic head and neck squamous cell carcinoma. Oral Oncol.
40:525–531. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Monnerat C, Faivre S, Temam S, Bourhis J
and Raymond E: End points for new agents in induction chemotherapy
for locally advanced head and neck cancers. Ann Oncol. 13:995–1006.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pignon JP, Bourhis J, Domenge C and
Designe L: Chemotherapy added to locoregional treatment for head
and neck squamous-cell carcinoma: Three meta-analyses of updated
individual data. MACH-NC Collaborative Group. Meta-analysis of
chemotherapy on head and neck cancer. Lancet. 355:949–955. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pendleton KP and Grandis JR:
Cisplatin-based chemotherapy options for recurrent and/or
metastatic squamous cell cancer of the head and neck. Clin Med
Insights Ther. 2013.10.4137/CMT.S10409, 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Miller KD, Siegel RL, Lin CC, Mariotto AB,
Kramer JL, Rowland JH, Stein KD, Alteri R and Jemal A: Cancer
treatment and survivorship statistics, 2016. CA Cancer J Clin.
66:271–289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rowinsky EK and Donehower RC: Paclitaxel
(taxol). N Engl J Med. 332:1004–1014. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Abu Samaan TM, Samec M, Liskova A, Kubatka
P and Busselberg D: Paclitaxel's mechanistic and clinical effects
on breast cancer. Biomolecules. 9:7892019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Habib S, Delourme J, Dhalluin X, Petyt G,
Tacelli N, Scherpereel A, Lafitte JJ and Cortot AB: Bevacizumab and
weekly paclitaxel for non-squamous non small cell lung cancer
patients: A retrospective study. Lung Cancer. 80:197–202. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Specenier P and Vermorken JB: The role of
taxanes and targeted therapies in locally advanced head and neck
cancer. Curr Opin Oncol. 19:195–201. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ferrari D, Ghi MG, Franzese C, Codeca C,
Gau M and Fayette J: The slippery role of induction chemotherapy in
head and neck cancer: Myth and reality. Front Oncol. 10:72020.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Xiao P, Ma T, Zhou C, Xu Y, Liu Y and
Zhang H: Anticancer effect of docetaxel induces apoptosis of
prostate cancer via the cofilin-1 and paxillin signaling pathway.
Mol Med Rep. 13:4079–4084. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Miller AV, Hicks MA, Nakajima W,
Richardson AC, Windle JJ and Harada H: Paclitaxel-induced apoptosis
is BAK-dependent, but BAX and BIM-independent in breast tumor. PLoS
One. 8:e606852013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Han TD, Shang DH and Tian Y: Docetaxel
enhances apoptosis and G2/M cell cycle arrest by suppressing
mitogen-activated protein kinase signaling in human renal clear
cell carcinoma. Genet Mol Res. 15:2016. View Article : Google Scholar
|
|
18
|
Pan Z, Avila A and Gollahon L: Paclitaxel
induces apoptosis in breast cancer cells through different
calcium-regulating mechanisms depending on external calcium
conditions. Int J Mol Sci. 15:2672–2694. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hu J, Zhang NA, Wang R, Huang F and Li G:
Paclitaxel induces apoptosis and reduces proliferation by targeting
epidermal growth factor receptor signaling pathway in oral cavity
squamous cell carcinoma. Oncol Lett. 10:2378–2384. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nonaka M, Ikeda H, Fujisawa A, Uehara M
and Inokuchi T: Induction of apoptosis by paclitaxel in human oral
carcinoma cells. Int J Oral Maxillofac Surg. 35:649–652. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Holsinger FC, Doan DD, Jasser SA, Swan EA,
Greenberg JS, Schiff BA, Bekele BN, Younes MN, Bucana CD, Fidler IJ
and Myers JN: Epidermal growth factor receptor blockade potentiates
apoptosis mediated by Paclitaxel and leads to prolonged survival in
a murine model of oral cancer. Clin Cancer Res. 9:3183–3189.
2003.PubMed/NCBI
|
|
22
|
Ganansia-Leymarie V, Bischoff P, Bergerat
JP and Holl V: Signal transduction pathways of taxanes-induced
apoptosis. Curr Med Chem Anticancer Agents. 3:291–306. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Huang CY, Ju DT, Chang CF, Muralidhar
Reddy P and Velmurugan BK: A review on the effects of current
chemotherapy drugs and natural agents in treating non-small cell
lung cancer. Biomedicine (Taipei). 7:232017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hsiao JR, Leu SF and Huang BM: Apoptotic
mechanism of paclitaxel-induced cell death in human head and neck
tumor cell lines. J Oral Pathol Med. 38:188–197. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cargnello M and Roux PP: Activation and
function of the MAPKs and their substrates, the MAPK-activated
protein kinases. Microbiol Mol Biol Rev. 75:50–83. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang X, Martindale JL and Holbrook NJ:
Requirement for ERK activation in cisplatin-induced apoptosis. J
Biol Chem. 275:39435–39443. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Teraishi F, Zhang L, Guo W, Dong F, Davis
JJ, Lin A and Fang B: Activation of c-Jun NH2-terminal kinase is
required for gemcitabine's cytotoxic effect in human lung cancer
H1299 cells. FEBS Lett. 579:6681–6687. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Uzu M, Sato H, Shimizu A, Shibata Y, Ueno
K and Hisaka A: Connexin 43 enhances Bax activation via JNK
activation in sunitinib-induced apoptosis in mesothelioma cells. J
Pharmacol Sci. 134:101–107. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Schrantz N, Bourgeade MF, Mouhamad S, Leca
G, Sharma S and Vazquez A: p38-mediated regulation of an
Fas-associated death domain protein-independent pathway leading to
caspase-8 activation during TGFbeta-induced apoptosis in human
Burkitt lymphoma B cells BL41. Mol Biol Cell. 12:3139–3151. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhao H, Wu S, Li H, Duan Q, Zhang Z, Shen
Q, Wang C and Yin T: ROS/KRAS/AMPK signaling contributes to
gemcitabine-induced stem-like cell properties in pancreatic cancer.
Mol Ther Oncolytics. 14:299–312. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
James D, Parone PA, Terradillos O,
Lucken-Ardjomande S, Montessuit S and Martinou JC: Mechanisms of
mitochondrial outer membrane permeabilization. Novartis Found Symp.
287:170–176; discussion 176-182. 2007.PubMed/NCBI
|
|
32
|
Gahl RF, Dwivedi P and Tjandra N: Bcl-2
proteins bid and bax form a network to permeabilize the
mitochondria at the onset of apoptosis. Cell Death Dis.
7:e24242016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tait SW and Green DR: Mitochondrial
regulation of cell death. Cold Spring Harb Perspect Biol.
5:a0087062013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li P, Zhou L, Zhao T, Liu X, Zhang P, Liu
Y, Zheng X and Li Q: Caspase-9: Structure, mechanisms and clinical
application. Oncotarget. 8:23996–24008. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
McIlwain DR, Berger T and Mak TW: Caspase
functions in cell death and disease. Cold Spring Harb Perspect
Biol. 7:a0267162015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Foo NP, Ko CL, Chu CY, Wang CY, So EC and
Huang BM: Arsenic compounds activate the MAPK and caspase pathways
to induce apoptosis in OEC-M1 gingival epidermal carcinoma. Oncol
Rep. 44:2701–2714. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Krysko DV, Vanden Berghe T, D'Herde K and
Vandenabeele P: Apoptosis and necrosis: Detection, discrimination
and phagocytosis. Methods. 44:205–221. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nicoletti I, Migliorati G, Pagliacci MC,
Grignani F and Riccardi C: A rapid and simple method for measuring
thymocyte apoptosis by propidium iodide staining and flow
cytometry. J Immunol Methods. 139:271–279. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Galluzzi L, Zamzami N, de La Motte Rouge
T, Lemaire C, Brenner C and Kroemer G: Methods for the assessment
of mitochondrial membrane permeabilization in apoptosis. Apoptosis.
12:803–813. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lowry OH, Rosebrough NJ, Farr AL and
Randall RJ: Protein measurement with the Folin phenol reagent. J
Biol Chem. 193:265–275. 1951. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yeung TK, Germond C, Chen X and Wang Z:
The mode of action of taxol: Apoptosis at low concentration and
necrosis at high concentration. Biochem Biophys Res Commun.
263:398–404. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Esteve MA, Carre M and Braguer D:
Microtubules in apoptosis induction: Are they necessary? Curr
Cancer Drug Targets. 7:713–729. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ulukaya E, Karaagac E, Ari F, Oral AY,
Adim SB, Tokullugil AH and Evrensel T: Chemotherapy increases
caspase-cleaved cytokeratin 18 in the serum of breast cancer
patients. Radiol Oncol. 45:116–122. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lu KH, Lue KH, Chou MC and Chung JG:
Paclitaxel induces apoptosis via caspase-3 activation in human
osteogenic sarcoma cells (U-2 OS). J Orthop Res. 23:988–994. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Mhaidat NM, Wang Y, Kiejda KA, Zhang XD
and Hersey P: Docetaxel-induced apoptosis in melanoma cells is
dependent on activation of caspase-2. Mol Cancer Ther. 6:752–761.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Janssen K, Pohlmann S, Janicke RU,
Schulze-Osthoff K and Fischer U: Apaf-1 and caspase-9 deficiency
prevents apoptosis in a Bax-controlled pathway and promotes
clonogenic survival during paclitaxel treatment. Blood.
110:3662–3672. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Li H, Zhu H, Xu CJ and Yuan J: Cleavage of
BID by caspase 8 mediates the mitochondrial damage in the Fas
pathway of apoptosis. Cell. 94:491–501. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Baig S, Seevasant I, Mohamad J, Mukheem A,
Huri HZ and Kamarul T: Potential of apoptotic pathway-targeted
cancer therapeutic research: Where do we stand? Cell Death Dis.
7:e20582016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Huang GC, Liu SY, Lin MH, Kuo YY and Liu
YC: The synergistic cytotoxicity of cisplatin and taxol in killing
oral squamous cell carcinoma. Jpn J Clin Oncol. 34:499–504. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Dhuriya YK and Sharma D: Necroptosis: A
regulated inflammatory mode of cell death. J Neuroinflammation.
15:1992018. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lee SY, Ju MK, Jeon HM, Jeong EK, Lee YJ,
Kim CH, Park HG, Han SI and Kang HS: Regulation of tumor
progression by programmed necrosis. Oxid Med Cell Longev.
2018:35374712018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mielke S: Individualized pharmacotherapy
with paclitaxel. Curr Opin Oncol. 19:586–589. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Eniu A, Palmieri FM and Perez EA: Weekly
administration of docetaxel and paclitaxel in metastatic or
advanced breast cancer. Oncologist. 10:665–685. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Stone AA and Chambers TC: Microtubule
inhibitors elicit differential effects on MAP kinase (JNK, ERK, and
p38) signaling pathways in human KB-3 carcinoma cells. Exp Cell
Res. 254:110–119. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Park SJ, Wu CH, Gordon JD, Zhong X, Emami
A and Safa AR: Taxol induces caspase-10-dependent apoptosis. J Biol
Chem. 279:51057–51067. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Broker LE, Kruyt FA and Giaccone G: Cell
death independent of caspases: A review. Clin Cancer Res.
11:3155–3162. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Constantinou C, Papas KA and Constantinou
AI: Caspase-independent pathways of programmed cell death: The
unraveling of new targets of cancer therapy? Curr Cancer Drug
Targets. 9:717–728. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Qu L, Liu FX, Cao XC, Xiao Q, Yang X and
Ren KQ: Activation of the apoptosis signal-regulating kinase
1/c-Jun N-terminal kinase pathway is involved in the
casticin-induced apoptosis of colon cancer cells. Exp Ther Med.
8:1494–1500. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Brockmann A, Bluwstein A, Kogel A, May S,
Marx A, Tschan MP and Brunner T: Thiazolides promote apoptosis in
colorectal tumor cells via MAP kinase-induced Bim and Puma
activation. Cell Death Dis. 6:e17782015. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Alexandre J, Batteux F, Nicco C, Chéreau
C, Laurent A, Guillevin L, Weill B and Goldwasser F: Accumulation
of hydrogen peroxide is an early and crucial step for
paclitaxel-induced cancer cell death both in vitro and in vivo. Int
J Cancer. 119:41–48. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Taniguchi T, Takahashi M, Shinohara F,
Sato T, Echigo S and Rikiishi H: Involvement of NF-kappaB and
mitochondrial pathways in docetaxel-induced apoptosis of human oral
squamous cell carcinoma. Int J Mol Med. 15:667–673. 2005.PubMed/NCBI
|
|
62
|
Zhao Q, Liu Y, Zhong J, Bi Y, Liu Y, Ren
Z, Li X, Jia J, Yu M and Yu X: Pristimerin induces apoptosis and
autophagy via activation of ROS/ASK1/JNK pathway in human breast
cancer in vitro and in vivo. Cell Death Discov. 5:1252019.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Asakuma J, Sumitomo M, Asano T, Asano T
and Hayakawa M: Selective Akt inactivation and tumor necrosis
actor-related apoptosis-inducing ligand sensitization of renal
cancer cells by low concentrations of paclitaxel. Cancer Res.
63:1365–1370. 2003.PubMed/NCBI
|
|
64
|
Swanton C, Marani M, Pardo O, Warne PH,
Kelly G, Sahai E, Elustondo F, Chang J, Temple J, Ahmed AA, et al:
Regulators of mitotic arrest and ceramide metabolism are
determinants of sensitivity to paclitaxel and other
chemotherapeutic drugs. Cancer Cell. 11:498–512. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Lu S, Natarajan SK, Mott JL, Kharbanda KK
and Harrison-Findik DD: Ceramide induces human hepcidin gene
transcription through JAK/STAT3 pathway. PLoS One. 11:e01474742016.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Villena J, Henriquez M, Torres V, Moraga
F, Díaz-Elizondo J, Arredondo C, Chiong M, Olea-Azar C, Stutzin A,
Lavandero S and Quest AF: Ceramide-induced formation of ROS and ATP
depletion trigger necrosis in lymphoid cells. Free Radic Biol Med.
44:1146–1160. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Kogot-Levin A and Saada A: Ceramide and
the mitochondrial respiratory chain. Biochimie. 100:88–94. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Yamaguchi H, Chen J, Bhalla K and Wang HG:
Regulation of Bax activation and apoptotic response to
microtubule-damaging agents by p53 transcription-dependent and
-independent pathways. J Biol Chem. 279:39431–39437. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Lee WT and Chang CW: Bax is upregulated by
p53 signal pathway in the SPE B-induced apoptosis. Mol Cell
Biochem. 343:271–279. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Henry RE, Andrysik Z, Paris R, Galbraith
MD and Espinosa JM: A DR4:tBID axis drives the p53 apoptotic
response by promoting oligomerization of poised BAX. EMBO J.
31:1266–1278. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Shen YH, Utama B, Wang J, Raveendran M,
Senthil D, Waldman WJ, Belcher JD, Vercellotti G, Martin D,
Mitchelle BM and Wang XL: Human cytomegalovirus causes endothelial
injury through the ataxia telangiectasia mutant and p53 DNA damage
signaling pathways. Circ Res. 94:1310–1317. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Aubrey BJ, Kelly GL, Janic A, Herold MJ
and Strasser A: How does p53 induce apoptosis and how does this
relate to p53-mediated tumour suppression? Cell Death Differ.
25:104–113. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Buschmann T, Potapova O, Bar-Shira A,
Ivanov VN, Fuchs SY, Henderson S, Fried VA, Minamoto T,
Alarcon-Vargas D, Pincus MR, et al: Jun NH2-terminal kinase
phosphorylation of p53 on Thr-81 is important for p53 stabilization
and transcriptional activities in response to stress. Mol Cell
Biol. 21:2743–2754. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Dong Y, Shen X, He M, Wu Z, Zheng Q, Wang
Y, Chen Y, Wu S, Cui J and Zeng Z: Activation of the JNK-c-Jun
pathway in response to irradiation facilitates Fas ligand secretion
in hepatoma cells and increases hepatocyte injury. J Exp Clin
Cancer Res. 35:1142016. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Chu R, Upreti M, Ding WX, Yin XM and
Chambers TC: Regulation of Bax by c-Jun NH2-terminal kinase and
Bcl-xL in vinblastine-induced apoptosis. Biochem Pharmacol.
78:241–248. 2009. View Article : Google Scholar : PubMed/NCBI
|