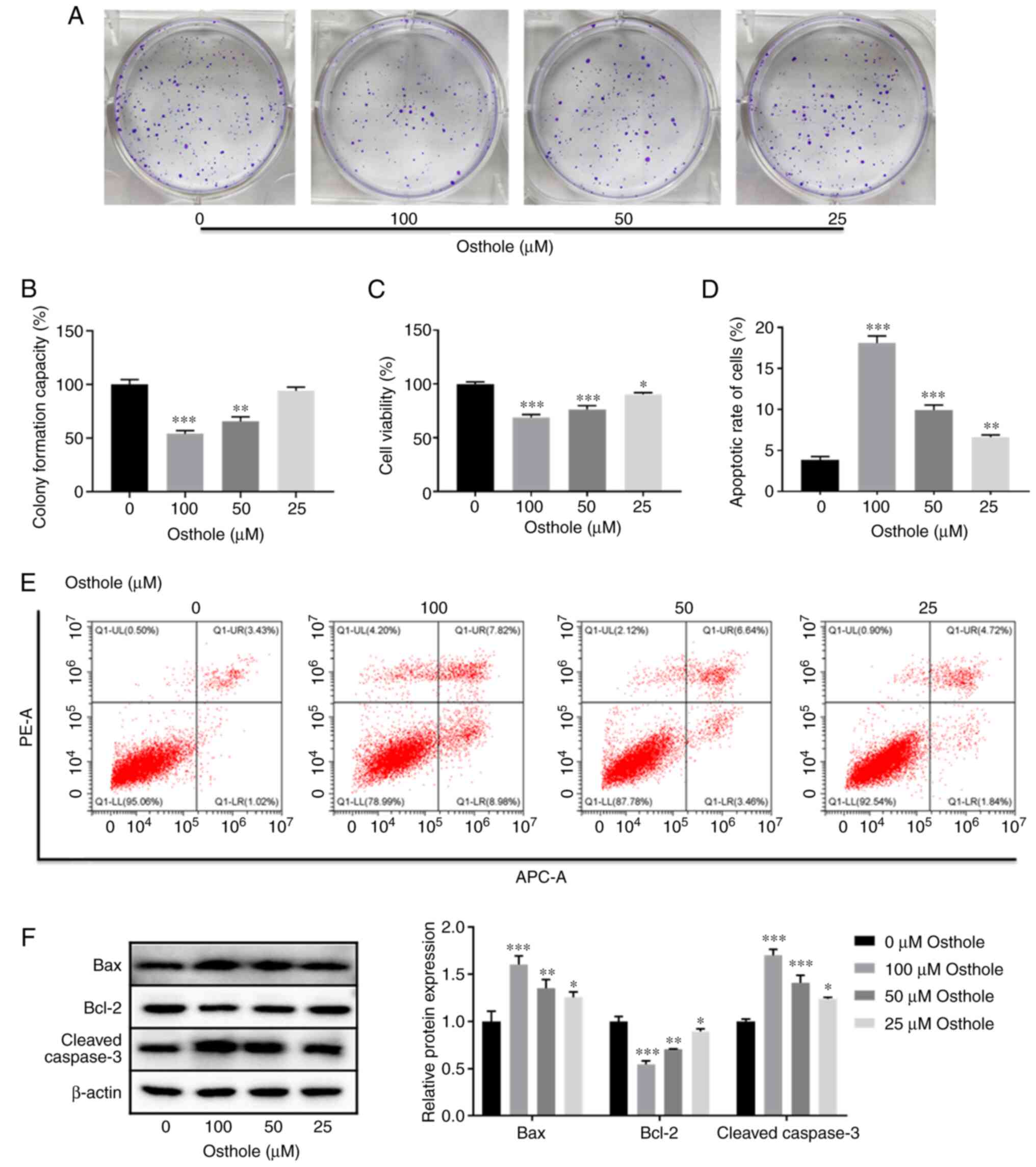
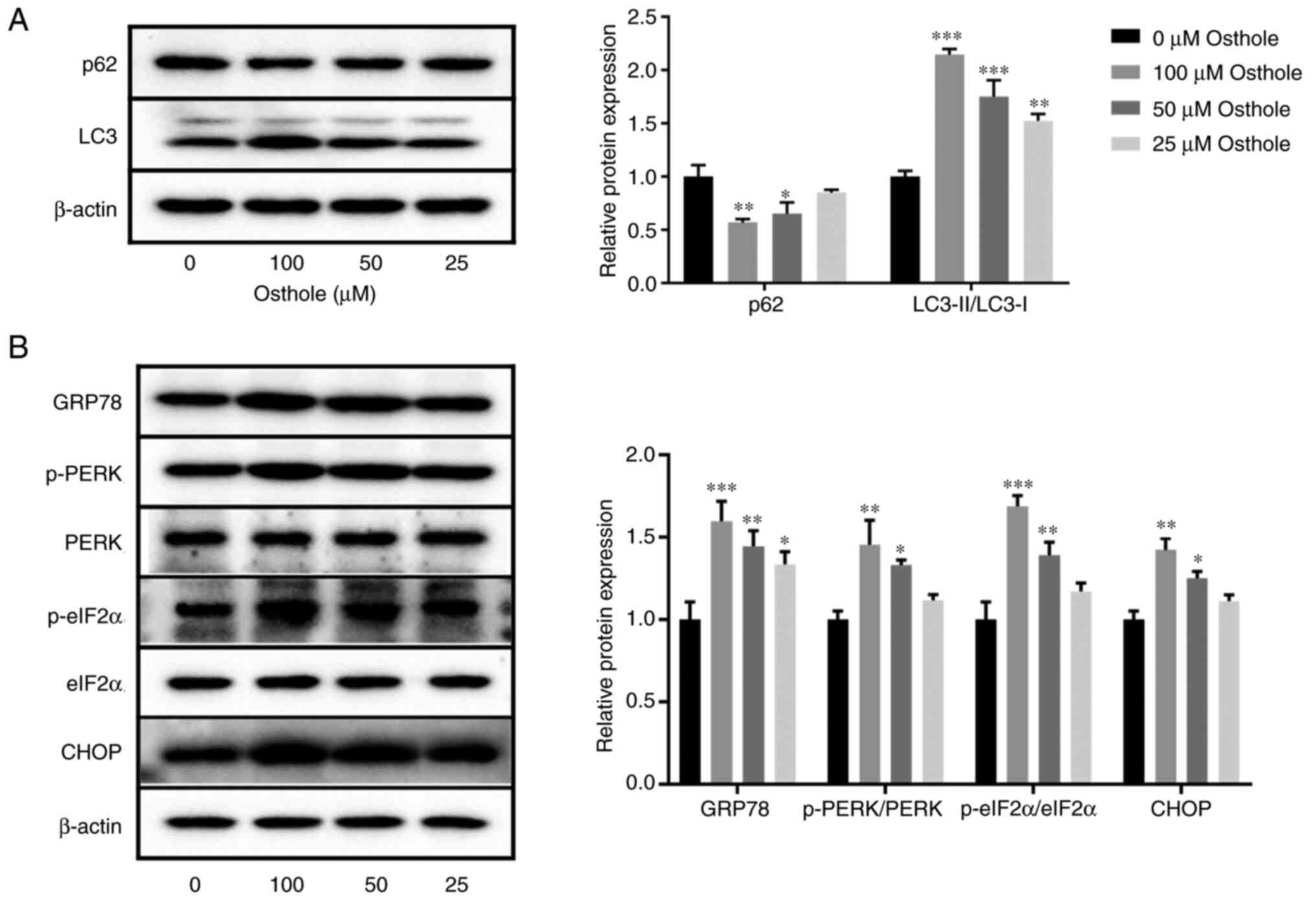
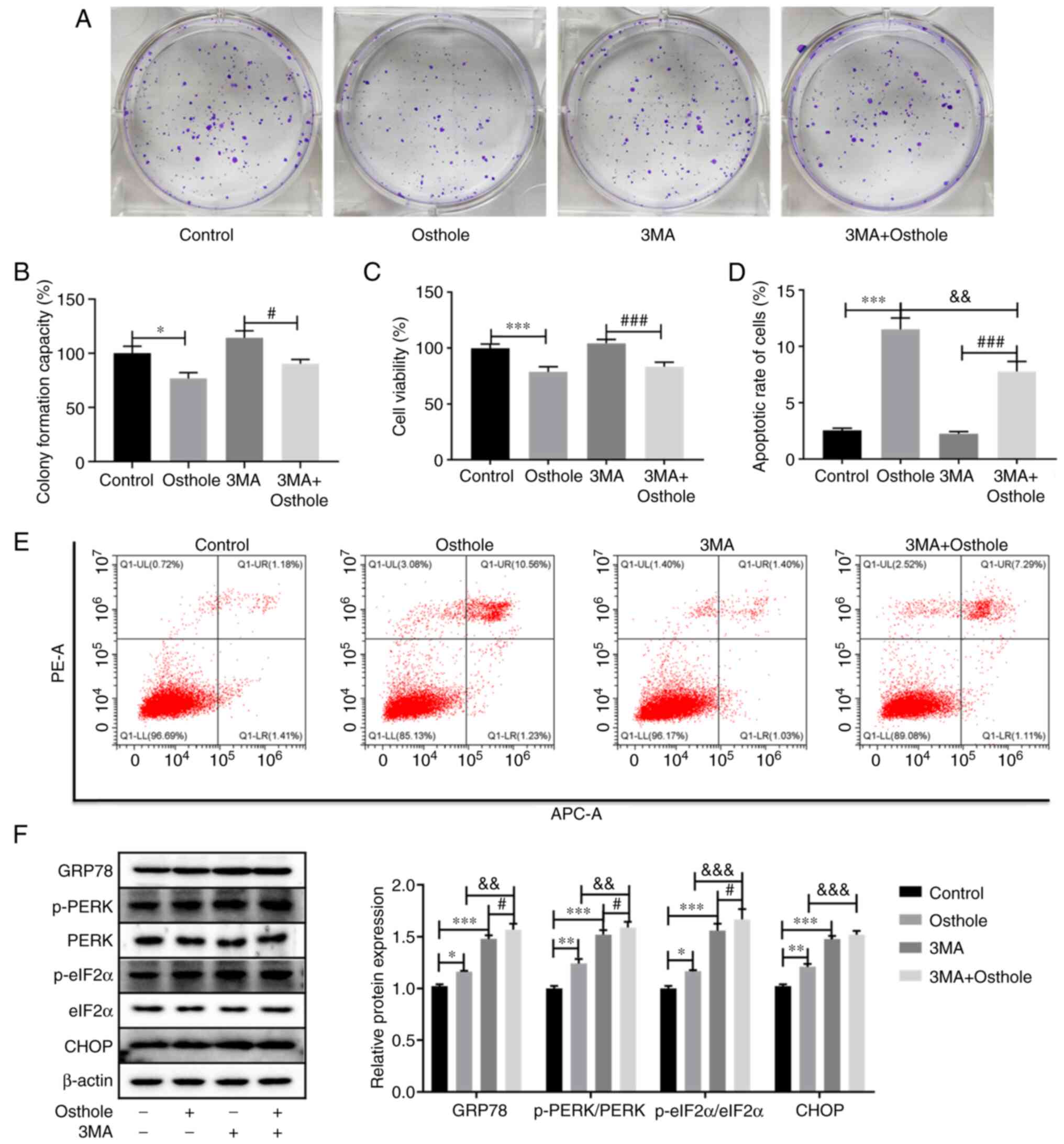
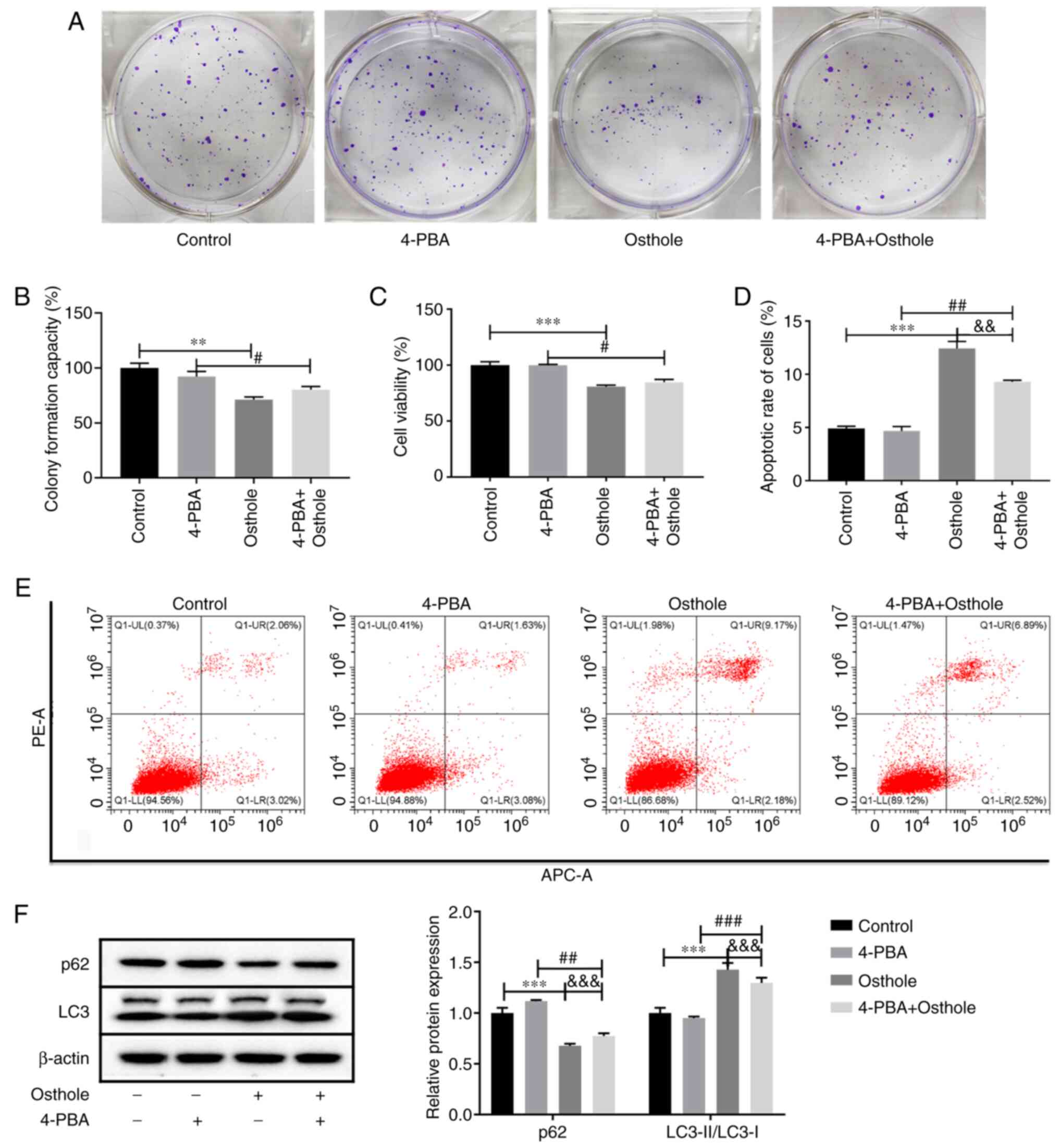
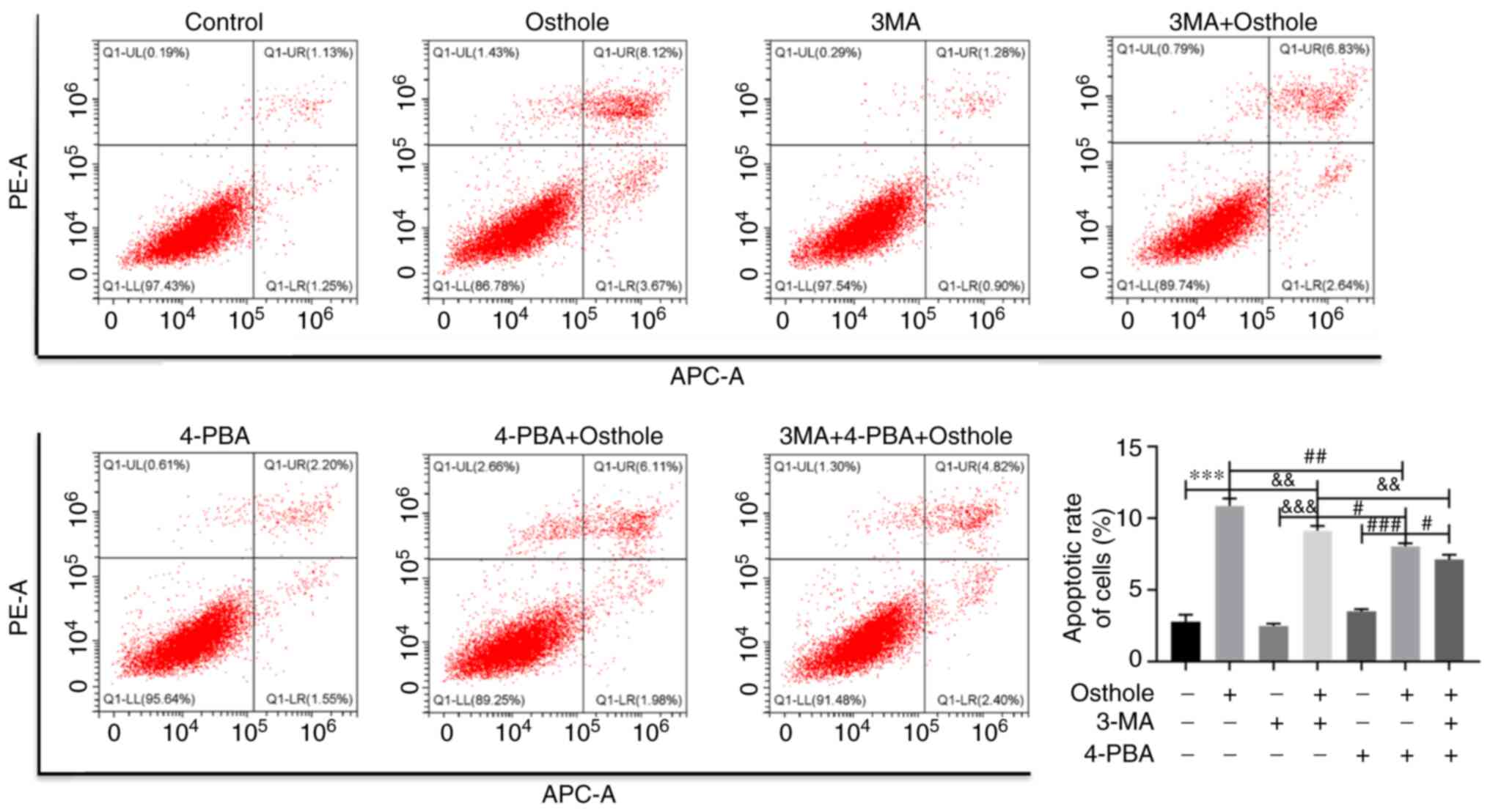
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hu T, Li Z, Gao CY and Cho CH: Mechanisms
of drug resistance in colon cancer and its therapeutic strategies.
World J Gastroenterol. 22:6876–6889. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang L, Peng Y, Shi K, Wang H, Lu J, Li Y
and Ma C: Osthole inhibits proliferation of human breast cancer
cells by inducing cell cycle arrest and apoptosis. J Biomed Res.
29:132–138. 2015.PubMed/NCBI
|
|
4
|
Tang DZ, Hou W, Zhou Q, Zhang M, Holz J,
Sheu TJ, Li TF, Cheng SD, Shi Q, Harris SE, et al: Osthole
stimulates osteoblast differentiation and bone formation by
activation of beta-catenin-BMP signaling. J Bone Miner Res.
25:1234–1245. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bao Y, Meng X, Liu F, Wang F, Yang J, Wang
H and Xie G: Protective effects of osthole against inflammation
induced by lipopolysaccharide in BV2 cells. Mol Med Rep.
17:4561–4566. 2018.PubMed/NCBI
|
|
6
|
Li Y, Li Y, Shi F, Wang L, Li L and Yang
D: Osthole attenuates right ventricular remodeling via decreased
myocardial apoptosis and inflammation in monocrotaline-induced
rats. Eur J Pharmacol. 818:525–533. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liu WB, Zhou J, Qu Y, Li X, Lu CT, Xie KL,
Sun XL and Fei Z: Neuroprotective effect of osthole on MPP+-induced
cytotoxicity in PC12 cells via inhibition of mitochondrial
dysfunction and ROS production. Neurochem Int. 57:206–215. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liang J, Zhou J, Xu Y, Huang X, Wang X,
Huang W and Li H: Osthole inhibits ovarian carcinoma cells through
LC3-mediated autophagy and GSDME-dependent pyroptosis except for
apoptosis. Eur J Pharmacol. 874:1729902020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xu XM, Zhang ML, Zhang Y and Zhao L:
Osthole induces lung cancer cell apoptosis through inhibition of
inhibitor of apoptosis family proteins. Oncol Lett. 12:3779–3784.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang SM, Tsai CF, Chen DR, Wang MY and
Yeh WL: p53 is a key regulator for osthole-triggered cancer
pathogenesis. Biomed Res Int. 2014:1752472014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jarząb A, Grabarska A, Skalicka-Woźniak K
and Stepulak A: Pharmacological features of osthole. Postepy Hig
Med Dosw (Online). 71:411–421. 2017. View Article : Google Scholar
|
|
12
|
D'Arcy MS: Cell death: A review of the
major forms of apoptosis, necrosis and autophagy. Cell Biol Int.
43:582–592. 2019. View Article : Google Scholar
|
|
13
|
Oakes S: Endoplasmic reticulum stress
signaling in cancer cells. Am J Pathol. 190:934–946. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guzel E, Arlier S, Guzeloglu-Kayisli O,
Tabak MS, Ekiz T, Semerci N, Larsen K, Schatz F, Lockwood CJ and
Kayisli UA: Endoplasmic reticulum stress and homeostasis in
reproductive physiology and pathology. Int J Mol Sci. 18:7922017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Almanza A, Carlesso A, Chintha C,
Creedican S, Doultsinos D, Leuzzi B, Luís A, McCarthy N,
Montibeller L, More S, et al: Endoplasmic reticulum stress
signalling-from basic mechanisms to clinical applications. FEBS J.
286:241–278. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Karagöz GE, Acosta-Alvear D and Walter P:
The unfolded protein response: Detecting and responding to
fluctuations in the protein-folding capacity of the endoplasmic
Reticulum. Cold Spring Harbor Perspect Biol. 11:a0338862019.
View Article : Google Scholar
|
|
17
|
Sano R and Reed JC: ER stress-induced cell
death mechanisms. Biochim Biophys Acta. 1833:3460–3470. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Verfaillie T, Salazar M, Velasco G and
Agostinis P: Linking ER Stress to autophagy: Potential implications
for cancer therapy. Int J Cell Biol. 2010:9305092010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Latorraca LB, Feitosa WB, Mariano C, Moura
MT, Fontes PK, Nogueira MFG and Paula-Lopes FF: Autophagy is a
pro-survival adaptive response to heat shock in bovine
cumulus-oocyte complexes. Sci Rep. 10:137112020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Di S, Fan C, Ma Z, Li M, Guo K, Han D, Li
X, Mu D and Yan X: PERK/eIF-2α/CHOP pathway dependent ROS
generation mediates butein-induced non-small-cell lung cancer
apoptosis and G2/M phase arrest. Int J Biol Sci. 15:1637–1653.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Park W, Park S, Song G and Lim W:
Inhibitory effects of osthole on human breast cancer cell
progression via induction of cell cycle arrest, mitochondrial
dysfunction, and ER stress. Nutrients. 11:27772019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Maiuri MC, Zalckvar E, Kimchi A and
Kroemer G: Self-eating and self-killing: Crosstalk between
autophagy and apoptosis. Nat Rev Mol Cell Biol. 8:741–752. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Song S, Tan J, Miao Y, Li M and Zhang Q:
Crosstalk of autophagy and apoptosis: Involvement of the dual role
of autophagy under ER stress. J Cell Physiol. 232:2977–2984. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pistritto G, Trisciuoglio D, Ceci C,
Garufi A and D'Orazi G: Apoptosis as anticancer mechanism: Function
and dysfunction of its modulators and targeted therapeutic
strategies. Aging. 8:603–619. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chipuk JE, Moldoveanu T, Llambi F, Parsons
MJ and Green DR: The BCL-2 family reunion. Mol Cell. 37:299–310.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shokoohinia Y, Jafari F, Mohammadi Z,
Bazvandi L, Hosseinzadeh L, Chow N, Bhattacharyya P, Farzaei MH,
Farooqi AA, Nabavi SM, et al: Potential anticancer properties of
osthol: A comprehensive mechanistic review. Nutrients. 10:362018.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Siddiqui WA, Ahad A and Ahsan H: The
mystery of BCL2 family: Bcl-2 proteins and apoptosis: An update.
Arch Toxicol. 89:289–317. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Devenport SN and Shah YM: Functions and
implications of autophagy in colon cancer. Cells. 8:13492019.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mathew R, Karantza-Wadsworth V and White
E: Role of autophagy in cancer. Nat Rev Cancer. 7:961–967. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mizushima N and Komatsu M: Autophagy:
Renovation of cells and tissues. Cell. 147:728–741. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang Y, Ren S, Liu Y, Gao K, Liu Z and
Zhang Z: Inhibition of starvation-triggered endoplasmic reticulum
stress, autophagy, and apoptosis in ARPE-19 cells by taurine
through modulating the expression of calpain-1 and calpain-2. Int J
Mol Sci. 18:21462017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lei C, Zhao B, Liu L, Zeng X, Yu Z and
Wang X: Expression and clinical significance of p62 protein in
colon cancer. Medicine (Baltimore). 99:e187912020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang H, Zhang Y, Zhu X, Chen C, Zhang C,
Xia Y, Zhao Y, Andrisani O and Kong L: DEAD box protein 5 inhibits
liver tumorigenesis by stimulating autophagy via interaction with
p62/SQSTM1. Hepatology. 69:1046–1063. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gong J, Wang XZ, Wang T, Chen JJ, Xie XY,
Hu H, Yu F, Liu HL, Jiang XY and Fan HD: Molecular signal networks
and regulating mechanisms of the unfolded protein response. J
Zhejiang Univ Sci B. 18:1–14. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sisinni L, Pietrafesa M, Lepore S,
Maddalena F, Condelli V, Esposito F and Landriscina M: Endoplasmic
reticulum stress and unfolded protein response in breast cancer:
The balance between apoptosis and autophagy and its role in drug
resistance. Int J Mol Sci. 20:8572019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Woo KJ, Lee TJ, Lee SH, Lee JM, Seo JH,
Jeong YJ, Park JW and Kwon TK: Elevated gadd153/chop expression
during resveratrol-induced apoptosis in human colon cancer cells.
Biochem Pharmacol. 73:68–76. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shen Z, Chen J and Lu H: Osthole induced
apoptosis in human normal liver cells by regulating cell
proliferation and endoplasmic reticulum stress. Environ Toxicol.
34:768–776. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mokarram P, Albokashy M, Zarghooni M,
Moosavi MA, Sepehri Z, Chen QM, Hudecki A, Sargazi A, Alizadeh J,
Moghadam AR, et al: New frontiers in the treatment of colorectal
cancer: Autophagy and the unfolded protein response as promising
targets. Autophagy. 13:781–819. 2017. View Article : Google Scholar : PubMed/NCBI
|