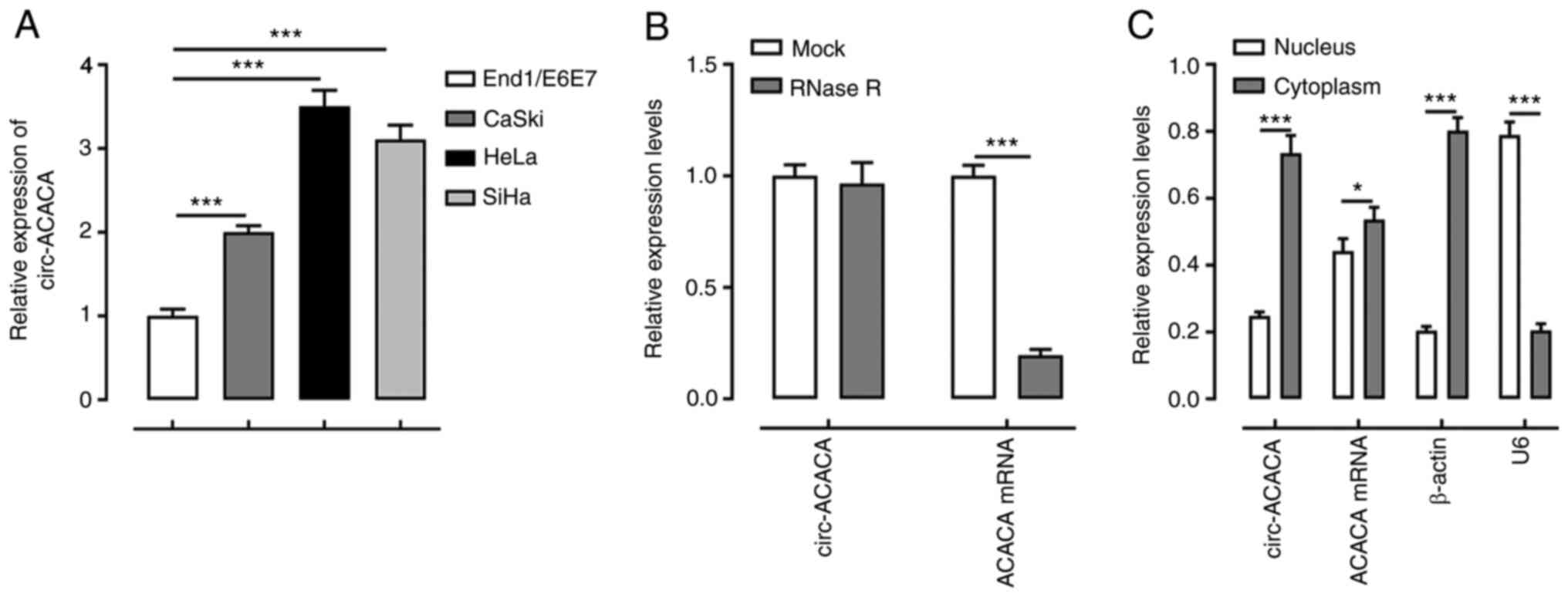
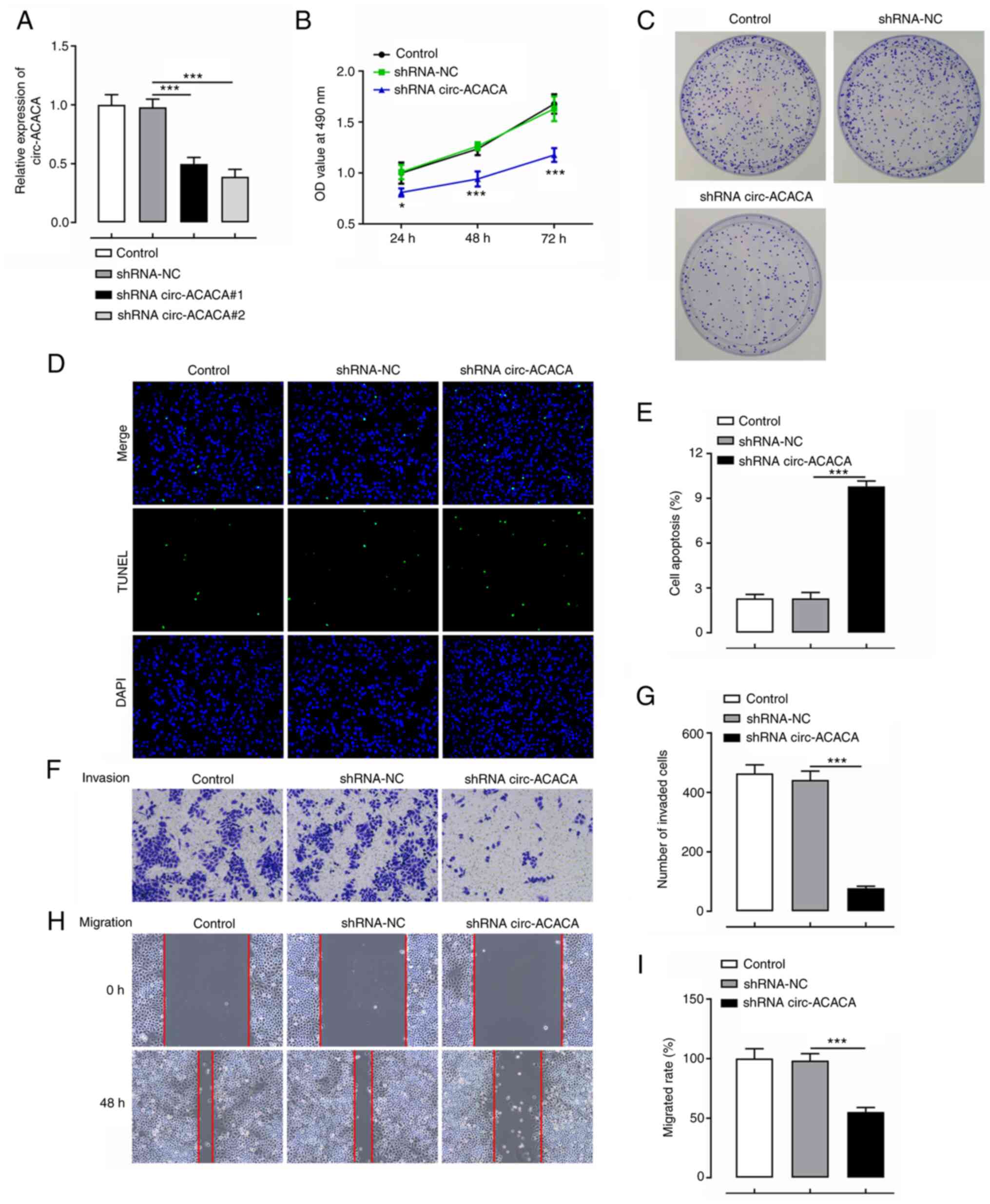
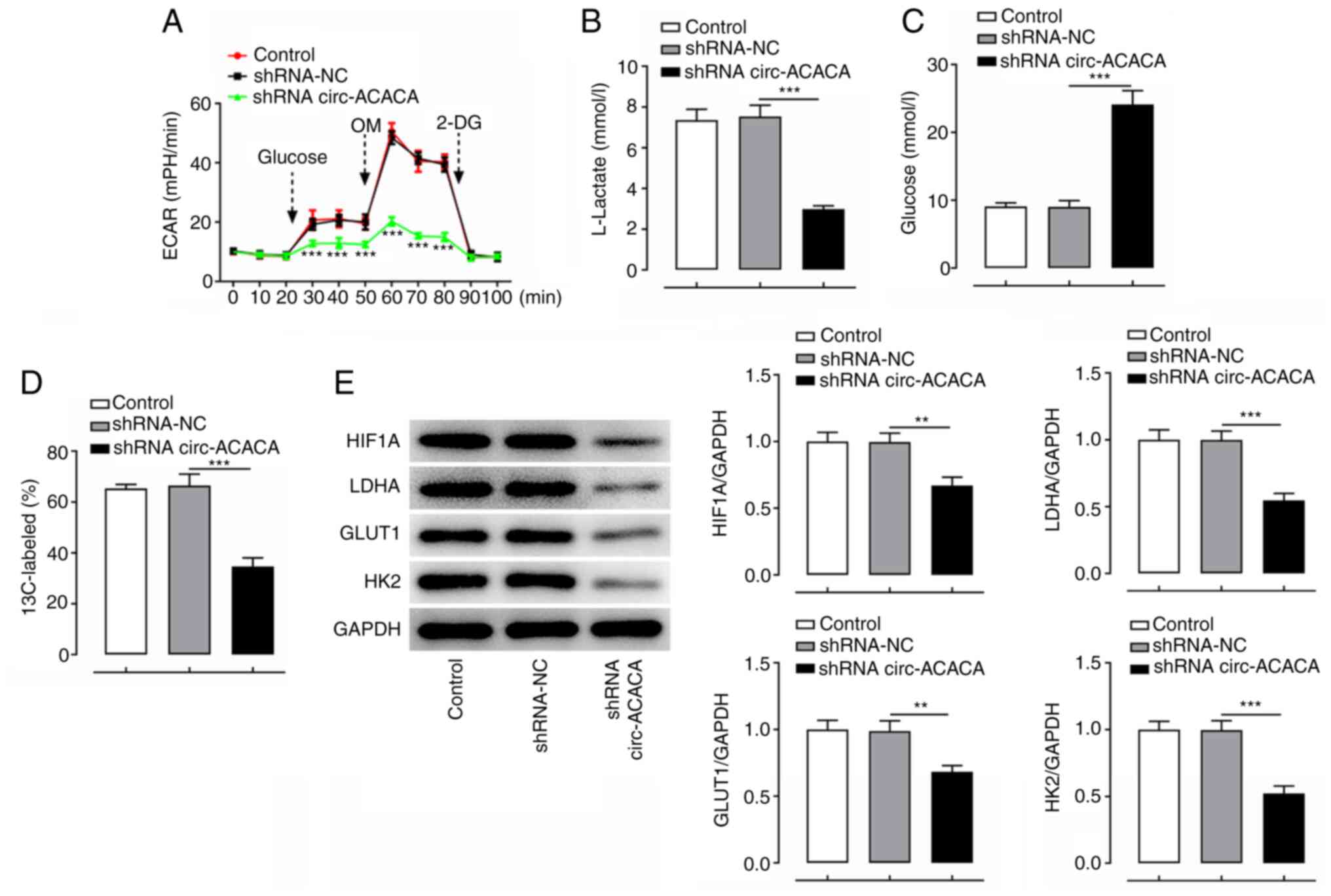
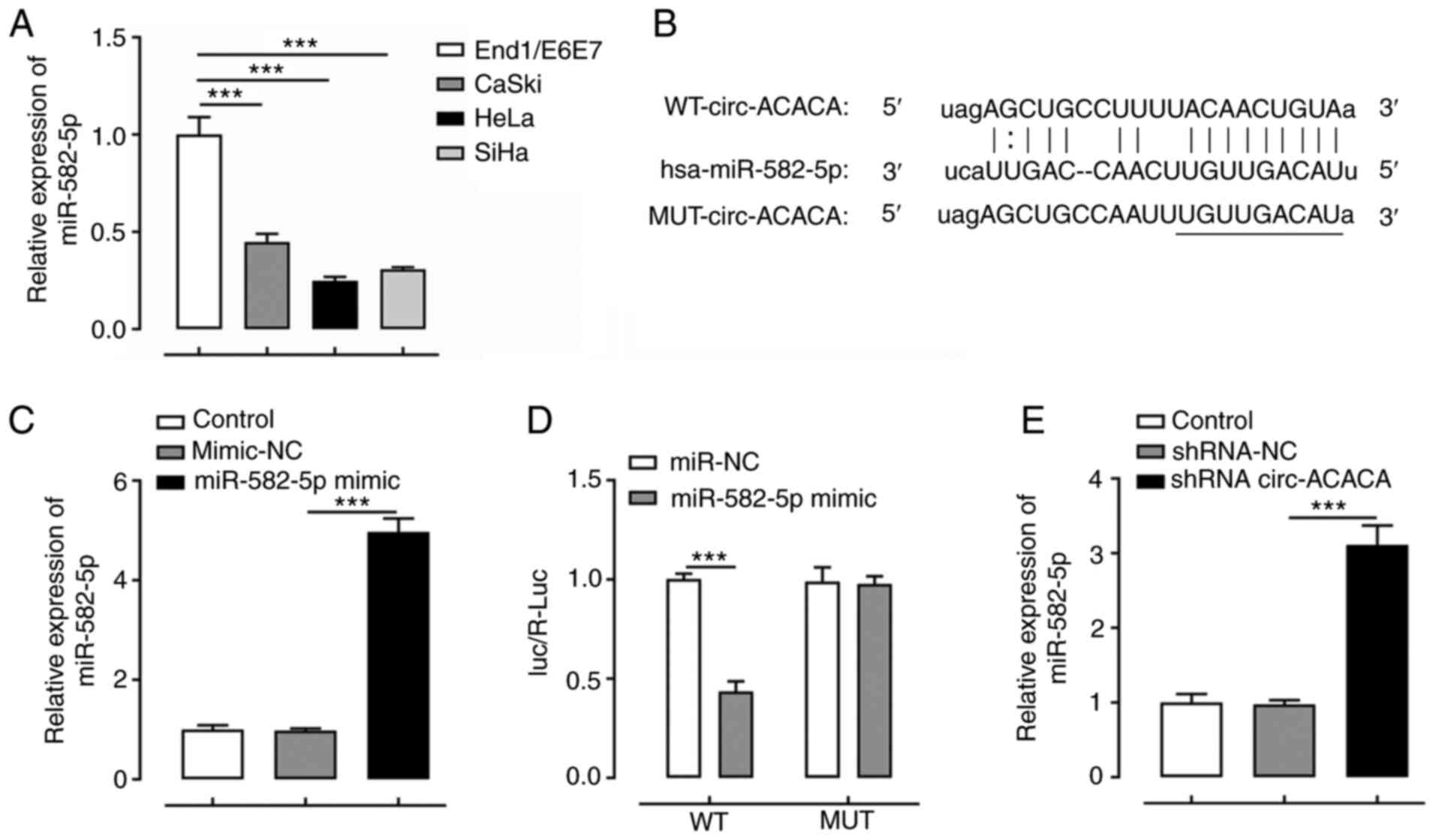
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fan Y, Sheng W, Meng Y, Cao Y and Li R:
LncRNA PTENP1 inhibits cervical cancer progression by suppressing
miR-106b. Artif Cells Nanomed Biotechnol. 48:393–407. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Fader AN: Surgery in cervical cancer. N
Engl J Med. 379:1955–1957. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fan Y, Meng Y, Yang S, Wang L, Zhi W,
Lazare C, Cao C and Wu P: Screening of cervical cancer with
self-collected cervical samples and next-generation sequencing. Dis
Markers. 2018:48265472018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wilusz JE and Sharp PA: Molecular biology.
A circuitous route to noncoding RNA. Science. 340:440–441. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jia YJ, Liu MJ and Wang SX: CircRNA
hsa_circRNA_0001776 inhibits proliferation and promotes apoptosis
in endometrial cancer via downregulating LRIG2 by sponging miR-182.
Cancer Cell Int. 20:4122020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang W, Liu T, Li TS and Zhao XD:
Hsa_circRNA_102002 facilitates metastasis of papillary thyroid
cancer through regulating miR-488-3p/HAS2 axis. Cancer Gene Ther.
28:279–293. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wu W, Xi W, Li H, Yang M and Yao X:
Circular RNA circ-ACACA regulates proliferation, migration and
glycolysis in non-small-cell lung carcinoma via miR-1183 and
PI3K/PKB pathway. Int J Mol Med. 45:1814–1824. 2020.PubMed/NCBI
|
|
10
|
Liu J, Liu S, Deng X, Rao J, Huang K, Xu G
and Wang X: MicroRNA-582-5p suppresses non-small cell lung cancer
cells growth and invasion via downregulating NOTCH1. PLoS One.
14:e02176522019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Huang S, Zou C, Tang Y, Wa Q, Peng X, Chen
X, Yang C, Ren D, Huang Y, Liao Z, et al: miR-582-3p and miR-582-5p
suppress prostate cancer metastasis to bone by repressing TGF-β
signaling. Mol Ther Nucleic Acids. 16:91–104. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wu J, Li W, Ning J, Yu W, Rao T and Cheng
F: Long noncoding RNA UCA1 targets miR-582-5p and contributes to
the progression and drug resistance of bladder cancer cells through
ATG7-mediated autophagy inhibition. Onco Targets Ther. 12:495–508.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li L and Ma L: Upregulation of miR-582-5p
regulates cell proliferation and apoptosis by targeting AKT3 in
human endometrial carcinoma. Saudi J Biol Sci. 25:965–970. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang Y, Huang W, Ran Y, Xiong Y, Zhong Z,
Fan X, Wang Z and Ye Q: miR-582-5p inhibits proliferation of
hepatocellular carcinoma by targeting CDK1 and AKT3. Tumour Biol.
36:8309–8316. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tanaka T, Kutomi G, Kajiwara T, Kukita K,
Kochin V, Kanaseki T, Tsukahara T, Hirohashi Y, Torigoe T, Okamoto
Y, et al: Cancer-associated oxidoreductase ERO1-α promotes immune
escape through up-regulation of PD-L1 in human breast cancer.
Oncotarget. 8:24706–24718. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang J, Yang J, Lin C, Liu W, Huo Y, Yang
M, Jiang SH, Sun Y and Hua R: Endoplasmic reticulum
stress-dependent expression of ERO1L promotes aerobic glycolysis in
pancreatic cancer. Theranostics. 10:8400–8414. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Han F, Xu Q, Zhao J, Xiong P and Liu J:
ERO1L promotes pancreatic cancer cell progression through
activating the Wnt/catenin pathway. J Cell Biochem. 119:8996–9005.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yan W, Wang X, Liu T, Chen L, Han L, Xu J,
Jin G, Harada K, Lin Z and Ren X: Expression of endoplasmic
reticulum oxidoreductase 1-α in cholangiocarcinoma tissues and its
effects on the proliferation and migration of cholangiocarcinoma
cells. Cancer Manag Res. 11:6727–6739. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zilli F, Marques Ramos P, Auf der Maur P,
Jehanno C, Sethi A, Coissieux MM, Eichlisberger T, Sauteur L,
Rouchon A, Bonapace L, et al: The NFIB-ERO1A axis promotes breast
cancer metastatic colonization of disseminated tumour cells. EMBO
Mol Med. 13:e131622021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang Y, Li T, Zhang L, Shangguan F, Shi
G, Wu X, Cui Y, Wang X, Wang X, Liu Y, et al: Targeting the
functional interplay between endoplasmic reticulum oxidoreductin-1α
and protein disulfide isomerase suppresses the progression of
cervical cancer. EBioMedicine. 41:408–419. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Seol SY, Kim C, Lim JY, Yoon SO, Hong SW,
Kim JW, Choi SH and Cho JY: Overexpression of Endoplasmic Reticulum
Oxidoreductin 1-α (ERO1L) is associated with poor prognosis of
gastric cancer. Cancer Res Treat. 48:1196–1209. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Takei N, Yoneda A, Sakai-Sawada K, Kosaka
M, Minomi K and Tamura Y: Hypoxia-inducible ERO1α promotes cancer
progression through modulation of integrin-β1 modification and
signalling in HCT116 colorectal cancer cells. Sci Rep. 7:93892017.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yamamoto T, Takano N, Ishiwata K, Ohmura
M, Nagahata Y, Matsuura T, Kamata A, Sakamoto K, Nakanishi T, Kubo
A, et al: Reduced methylation of PFKFB3 in cancer cells shunts
glucose towards the pentose phosphate pathway. Nat Commun.
5:34802014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Guarnerio J, Zhang Y, Cheloni G, Panella
R, Mae Katon J, Simpson M, Matsumoto A, Papa A, Loretelli C, Petri
A, et al: Intragenic antagonistic roles of protein and circRNA in
tumorigenesis. Cell Res. 29:628–640. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang G, Liu Y, Yang J, Wang H and Xing Z:
Inhibition of circ_0081234 reduces prostate cancer tumor growth and
metastasis via miR-1/MAP3K1 axis. J Gene Med. e33762021.PubMed/NCBI
|
|
27
|
Yang C, Shi J, Wang J, Hao D, An J and
Jiang J: Circ_0006988 promotes the proliferation, metastasis and
angiogenesis of non-small cell lung cancer cells by modulating
miR-491-5p/MAP3K3 axis. Cell Cycle. 20:1334–1346. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang J, Li S, Zhang G and Han H:
Sevoflurane inhibits malignant progression of colorectal cancer via
hsa_circ_0000231-mediated miR-622. J Biol Res (Thessalon).
28:142021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Xie J, Chen Q, Zhou P and Fan W: Circular
RNA hsa_circ_0000511 improves epithelial mesenchymal transition of
cervical cancer by regulating hsa-mir-296-5p/HMGA1. J Immunol Res.
2021:99645382021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ding L, Zhao Y, Dang S, Wang Y, Li X, Yu
X, Li Z, Wei J, Liu M and Li G: Circular RNA circ-DONSON
facilitates gastric cancer growth and invasion via NURF complex
dependent activation of transcription factor SOX4. Mol Cancer.
18:452019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li Y, Zheng F, Xiao X, Xie F, Tao D, Huang
C, Liu D, Wang M, Wang L, Zeng F and Jiang G: CircHIPK3 sponges
miR-558 to suppress heparanase expression in bladder cancer cells.
EMBO Rep. 18:1646–1659. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pelicano H, Martin DS, Xu RH and Huang P:
Glycolysis inhibition for anticancer treatment. Oncogene.
25:4633–4646. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hua Q, Mi BM, Xu F, Wen J, Zhao L, Liu J
and Huang G: Hypoxia-induced lncRNA-AC020978 promotes proliferation
and glycolytic metabolism of non-small cell lung cancer by
regulating PKM2/HIF-1α axis. Theranostics. 10:4762–4778. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sun SF, Li WW, Ma XM and Luan H: Long
noncoding RNA LINC00265 promotes glycolysis and lactate production
of colorectal cancer through regulating of miR-216b-5p/TRIM44 axis.
Digestion. 101:391–400. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shang RZ, Wang M, Dai B, Du J, Wang J, Liu
Z, Qu S, Yang X, Liu J, Xia C, et al: Long noncoding RNA SLC2A1-AS1
regulates aerobic glycolysis and progression in hepatocellular
carcinoma via inhibiting the STAT3/FOXM1/GLUT1 pathway. Mol Oncol.
14:1381–1396. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Xu J, Tan QQ and Lie T: USP22 promotes the
expression of GLUT1 and HK2 to facilitate growth and glycolysis in
cervical cancer cells. Eur J Gynaecol Oncol. 41:790–796. 2020.
View Article : Google Scholar
|
|
37
|
Yeung C, Gibson AE, Issaq SH, Oshima N,
Baumgart JT, Edessa LD, Rai G, Urban DJ, Johnson MS, Benavides GA,
et al: Targeting glycolysis through inhibition of lactate
dehydrogenase impairs tumor growth in preclinical models of ewing
sarcoma. Cancer Res. 79:5060–5073. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Walenta S, Wetterling M, Lehrke M,
Schwickert G, Sundfør K, Rofstad EK and Mueller-Klieser W: High
lactate levels predict likelihood of metastases, tumor recurrence,
and restricted patient survival in human cervical cancers. Cancer
Res. 60:916–921. 2000.PubMed/NCBI
|
|
39
|
Wang M, Wang W, Wang J and Zhang J:
MiR-182 promotes glucose metabolism by upregulating
hypoxia-inducible factor 1α in NSCLC cells. Biochem Biophys Res
Commun. 504:400–405. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kristensen LS, Hansen TB, Veno MT and
Kjems J: Circular RNAs in cancer: Opportunities and challenges in
the field. Oncogene. 37:555–565. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Rupaimoole R, Calin GA, Lopez-Berestein G
and Sood AK: miRNA deregulation in cancer cells and the tumor
microenvironment. Cancer Discov. 6:235–246. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Tian Y, Guan Y, Su Y, Luo W, Yang G and
Zhang Y: MiR-582-5p inhibits bladder cancer-genesis by suppressing
TTK Expression. Cancer Manag Res. 12:11933–11944. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Tang W, Ji M, He G, Yang L, Niu Z, Jian M,
Wei Y, Ren L and Xu J: Silencing CDR1as inhibits colorectal cancer
progression through regulating microRNA-7. Onco Targets Ther.
10:2045–2056. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Takei N, Yoneda A, Kosaka M, Sakai-Sawada
K and Tamura Y: ERO1α is a novel endogenous marker of hypoxia in
human cancer cell lines. BMC Cancer. 19:5102019. View Article : Google Scholar : PubMed/NCBI
|