The vascular endothelial growth factor (VEGF) family
consists of five mammalian factors, VEGFA, VEGFB, VEGFC, VEGFD and
placenta growth factor (PlGF). Genetic inactivation of VEGFA and
VEGFC in mice results in embryonic death due to defects in the
development of blood and lymphatic vessels, respectively. The VEGFs
bind to three different but structurally related tyrosine kinase
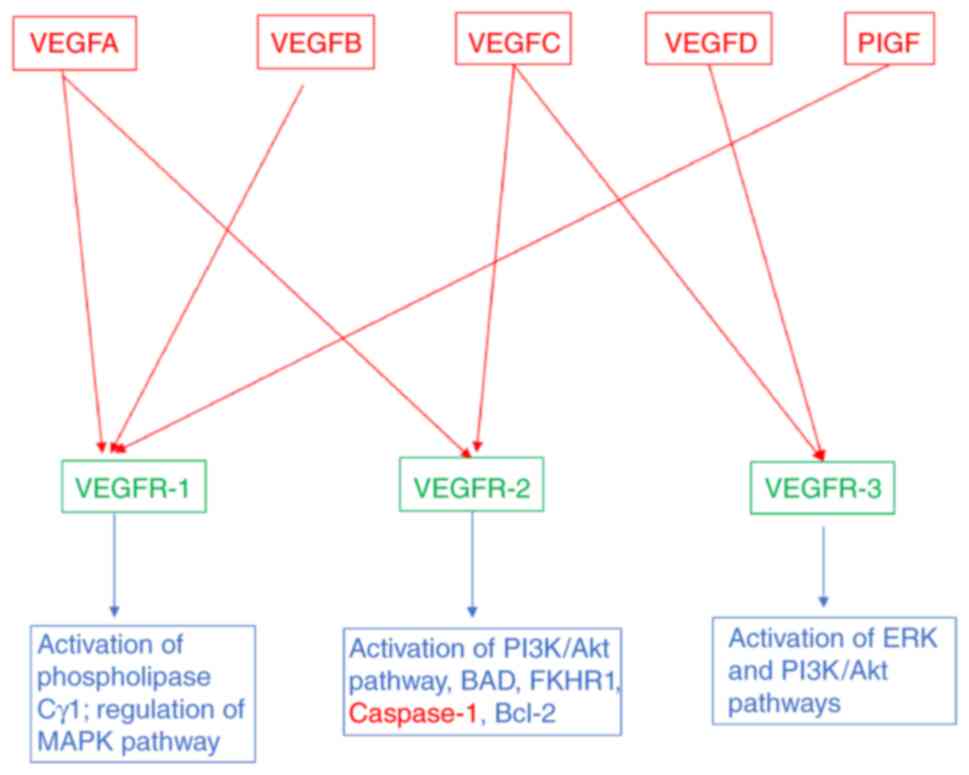
receptors, named VEGFR-1, VEGFR-2 and VEGFR-3 (Fig. 1). VEGFR-2 is preferentially
expressed on blood vascular endothelial cells, whereas VEGFR-3 is
primarily expressed on lymphatic endothelial cells. Moreover,
VEGFR-1 is expressed on a range of non-endothelial cells and is
essential for the regulation of leukocyte motility (1–3). All
VEGFRs contain seven immunoglobulin (Ig) homology domains, which
comprise the ligand-binding site, and an intracellular region
endowed with tyrosine kinase (TK) activity, which transduces the
signal. The downstream signaling includes activation of
phospholipase Cγ1, MAPK pathway via Ras/Raf1 activation and
PI3K/Akt pathway. Phospholipase Cγ1 regulates the concentration of
intracellular Ca+2 ions and formation of endothelial
nitric oxide synthase (Fig. 1).
The effect of all the cascades provides a balance of pro- and
anti-angiogenic signals that maintain the vasculature and/or result
in sprouting of new blood vessels, cell proliferation and cell
migration. The VEGF/VEGFR signaling pathway is upregulated in many
types of cancers, contributing to uncontrolled angiogenesis and
metastatic spreading.
VEGFA serves a critical role in vasculogenesis,
angiogenesis, tumors, inflammatory angiogenesis and
lymphangiogenesis. VEGFA was initially discovered as a vascular
permeability factor with a potency 50,000 times that of histamine
(1) and extravascular fluid
accumulation is prominent in tumor growth in body cavities, such as
the peritoneum. A striking positive correlation between VEGFA
expression levels, tumor progression and consequently reduced
patient survival, has also been demonstrated (2).
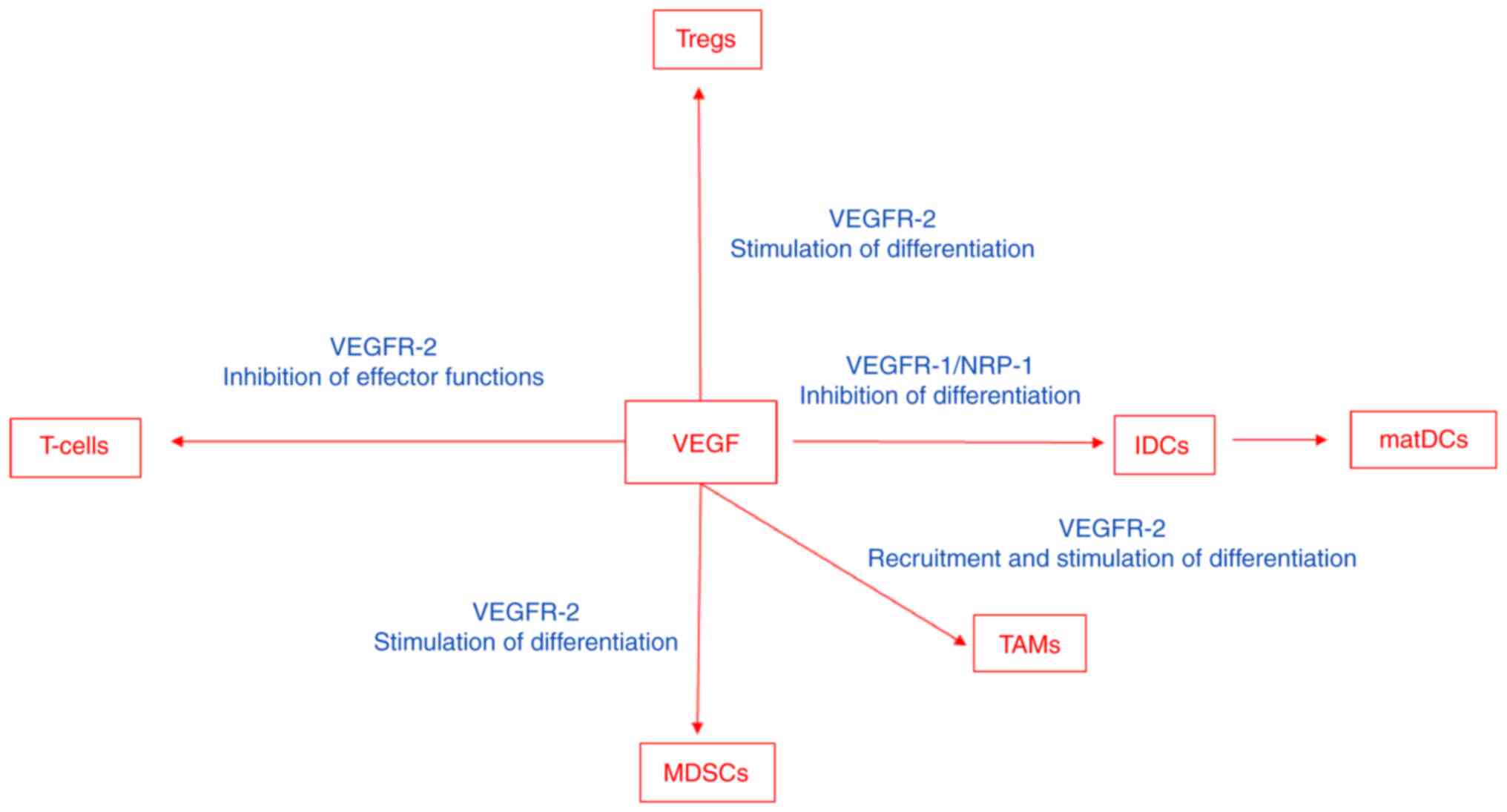
VEGF modulates the function of T-cells, suppressive
immune cells and the stroma in the tumor microenvironment, which
results in an immunosuppressive state (Fig. 2). VEGF impairs interactions between
leukocytes and endothelial cells. It achieves this via the
downregulation of the expression of adhesion molecules, including
vascular cell adhesion molecule-1 (VCAM-1) and intercellular cell
adhesion molecule-1 (ICAM-1), or via the inhibition of their
clustering, which impairs the ability of immune cells to adhere and
migrate across the endothelium into the tumor (3–7). The
anergic phenotype of tumor endothelial cells can be reversed via
antiangiogenic therapy, which upregulates the expression of
endothelial adhesion molecules in the tumor vasculature (8).
The number of tumors infiltrating lymphocytes has
been demonstrated to be markedly increased in animal tumor models
and in humans following VEGF inhibition (4,9,10).
VEGF signaling directly affects T-cell development, homing and
cytotoxic functions (7). Moreover,
VEGF negatively impacts T-cell migration from the lymph nodes into
the tumor bed via the stimulation of abnormal tumor vasculature
formation (11). VEGF/VEGFR-2
induces the production of pro-inflammatory molecules, including
interferon-γ and interleukin-2, and stimulates the migratory
response in memory CD4+ T-cells (12). Furthermore, VEGF induces regulatory
T-cells (T-regs) (13). VEGF also
impedes the differentiation of hematopoietic progenitor cells in
the thymus to CD8+ and CD4+ T-cells (14).
Dendritic cells are antigen-presenting cells that
serve a critical role in T-cell priming and activation. VEGF
inhibits dendritic maturation and antigen presentation (20). High levels of VEGF expression in
human cancers have been associated with defective dendritic cell
function and number (21–23). VEGFR-1 and neuropilin-1 are
involved in the VEGF-inhibition of dendritic cell maturation
(24,25). VEGF upregulates programmed
death-ligand 1 (PD-L1) in dendritic cells, which results in the
inhibition of T-cell expansion and function (26). Furthermore, it inhibits the
function of human mature dendritic cells to stimulate T-cells,
mediated by VEGFR-2 (27), via the
inhibition of NF-κB activation (28).
Myeloid-derived suppressor cells (MDSCs) are a mixed
cell population consisting mainly of neutrophils but also of
macrophages and dendritic cells with immunosuppressive and
tumor-promoting capacities (2,29).
VEGF induces the differentiation of myeloid cells into
immunosuppressive MDSCs (30) and
the VEGF concentration has been demonstrated to be associated with
the presence of MDSCs (31). In
tumors resistant to anti-VEGF treatment, the increased mobilization
and infiltration of MDSCs is distinguishable when compared with
treatment-sensitive tumors (32).
Tumor-associated macrophages (TAMs) differentiate
into anti-inflammatory M1 macrophages or pro-inflammatory and tumor
promoting M2 macrophages, which is dependent on the local cues
provided within the tumor microenvironment (33). M2 macrophages secrete
immunosuppressive cytokines, including IL-10, chemokine (C-C)
ligand (CCL)-7 and CCL-22 and angiogenic and tissue remodeling
factors, including VEGF, PlGF and matrix metalloproteinase-9
(33). VEGF recruits TAMs and
therefore contributes to the establishment of an immunosuppressive
microenvironment (34) and induces
the maturation of myeloid cells into the M2-like phenotype
(35). A reduced recruitment of
TAMs or the reprogramming of M2-like TAMs contributes towards
anti-cancer M1 phenotype reversed immunosuppression (36).
The increased recruitment of neutrophils during
anti-VEGF treatment promotes tumor progression and treatment
resistance (32).
Cancer-associated fibroblasts (CAFs) secrete several angiogenic
factors, including epidermal growth factor, hepatocyte growth
factor, insulin-like growth factor and fibroblast growth factor-2
(37). CAFs that are resistant to
anti-VEGF therapy promote tumor growth in anti-VEGF treatment
sensitive tumors (29).
VEGF may also contribute to the prevention of an
uncontrolled, detrimental immune response to the microbiota in the
lung. In this context, VEGF signaling in the alveolar
microenvironment is responsible for endothelial cell-mediated
tolerance to airborne pathogens and toxins via the interaction of
VEGF released by alveolar type I cells and secretory epithelial
cells with capillary endothelial cells (38). Moreover, VEGF is involved in
fibrotic lung disease (39).
Transfection of anti-VEGF gene therapy, in the form of the sFlt-1,
resulted in the attenuation of pulmonary fibrosis with a reduction
in lung collagen deposition and additional anti-inflammatory and
anti-angiogenic effects.
VEGF has served an important role in the clinic as a
therapeutic target in the anti-angiogenic approach to tumor
therapy. Bevacizumab binds VEGFA, which prevents VEGFA from
interacting with VEGFR-2 on vascular endothelial cells, endothelial
progenitor cells and megakaryocytes. Bevacizumab treatment in
patients with metastatic colorectal cancer induces an increase in
B- and T-cells (40). Furthermore,
the addition of bevacizumab to chemotherapy for patients with
non-small cell lung cancer results in improved dendritic cell
activation and T-cell cytotoxicity (41). Bevacizumab also reverses the
VEGF-induced inhibition of monocyte differentiation into dendritic
cells (42) and restores
peripheral blood dendritic cell numbers in patients with cancer
(43). In renal cell carcinoma,
bevacizumab reduces the number of MDSCs (44). Furthermore, VEGF induces T-reg
proliferation via VEGFR-2 activation. Bevacizumab also inhibits
T-reg accumulation in the peripheral blood of patients with
metastatic colorectal cancer (45). Moreover, ramucirumab (Cyramaza) is
a distinct monoclonal anti-VEGFR-2 antibody approved for the
treatment of non-small cell lung cancer, colorectal cancer,
hepatocellular carcinoma and gastric cancer (46).
Since VEGFRs possess a tyrosine kinase domain,
several small ATP mimetics that can inhibit the activity of
tyrosine kinase receptors involved in angiogenesis have been
developed. Sorafenib and sunitinib were the first multi-kinase
inhibitors approved for the treatment of metastatic renal cell and
hepatocellular carcinomas. Sunitinib, a multi-tyrosine kinase
inhibitor has been demonstrated to induce the upregulation of
ICAM-1 and VCAM-1 adhesion molecules on endothelial cells in
tumor-bearing mice (47). These
mice displayed increased Th1 responses via the reduction of
inhibitory molecule expression, including transforming growth
factor-β, IL-10, forkhead box protein-3, PD-1 and CTLA-4 (48). Sunitinib reduces the
immunosuppressive activity of MDSCs (49) and in human renal cell carcinoma
increases tumor infiltrating lymphocytes and reduces MDSCs
(50,51). Moreover, sunitinib in combination
with CD40 immuno-stimulating immunotherapy induces dendritic cell
activation, reduces MDSCs and enhances cytotoxic T-cell recruitment
(47).
PD-1, its ligand PD-L1 and CTLA-4 are negative
regulators of T-cell immune function. Following ligand binding,
PD-1 attenuates T-cell activation via recruiting SH2
domain-containing protein tyrosine phosphatase-2 and reducing
cytokine production and T-cell proliferation (52). PD-L1 is constitutively expressed
via T-cells, B-cells, dendritic cells, macrophages and
non-hematopoietic cells, including endothelial cells, epithelial
cells, hepatocytes and astrocytes (53). CTLA-4 is a cell surface receptor
expressed on activated T-cells. Following T-cell receptor
interactions with its cognate peptide/major histocompatibility
complex, and co-stimulation via CD28, CTLA-4 suppresses this
co-stimulation (54).
Numerous trials with anti-CTLA-4 and anti-PD-1/PD-L1
antibodies, which re-activate the anti-tumor processes of the human
immune system, have resulted in durable responses in patients with
cancer. In a previous study, VEGF expression decreased in patients
with metastatic melanoma responding to anti-CTLA-4 and anti-PD-1
therapy, but increased in non-responders, which indicated
therapeutic resistance (55).
Anti-angiogenic agents improve the effectiveness of
immunotherapy by transiently restoring the abnormal tumor
vasculature and increasing the infiltration of immune effector
cells into the tumor microenvironment (56). This induces the formation of high
endothelial venules that improve lymphocyte infiltration and
improve anti-tumor immunity (57).
A previous study demonstrated that patients with renal cell cancer
or non-small cell lung cancer with a high dendritic cell number
before treatment, exhibited an improved response to PD-L1
inhibition with atezolizumab (58).
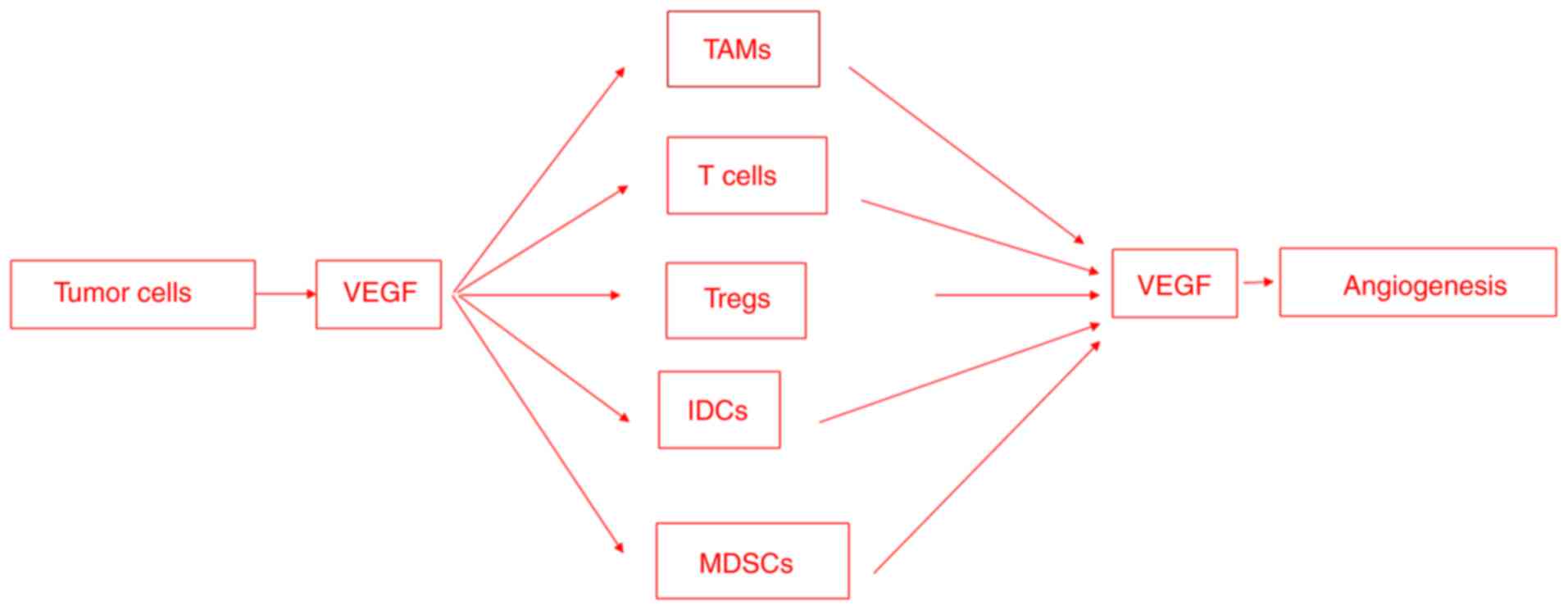
Angiogenesis and immunosuppression are closely
associated and occur simultaneously in response to different
stimuli (Fig. 3). The VEGF/VEGFR
signaling pathway is recognized as the master regulator of tumor
angiogenesis. VEGFRs may be targeted through monoclonal antibodies
to inhibit VEGF binding to the extracellular domain of the receptor
or, alternatively, using different small molecule tyrosine kinase
inhibitors blocking the ATP binding in the kinase domain and
phosphorylation of tyrosine residues, in clinical trials for the
treatment of renal cell carcinoma, hepatocellular carcinoma,
metastatic colorectal cancer and gastrointestinal stromal tumors.
Another strategy concerns the use of small-molecule inhibitors
targeting cell signaling pathways activated by VEGFRs, approved for
the treatment of metastatic melanoma and metastatic renal cell
carcinoma.
Tumor cells secrete soluble factors, such as VEGFA,
that recruit immunosuppressive cells, including TAMs, neutrophils,
MDSCs, dendritic cells, T-regs and natural killer cells. The
immunosuppressive cells, in turn, secrete VEGF, which promotes
endothelial cell proliferation and migration and/or induces the
release of MMPs. VEGFA exerts different immunosuppressive effects,
including the inhibition of dendritic cell maturation, the
induction of inhibitory molecule expression, such as PD-L1, on
dendritic cells, and the activation of T-regs. The present review
has demonstrated that VEGF can simultaneously promote angiogenesis
and mediate immunosuppression. These overlapping activities may
potentially explain the efficacy of anti-angiogenic and
immunotherapies in reversing the immunosuppressive effects of
VEGF.
Not applicable.
This research was supported by a grant from European Union
Seventh Framework Programme (FP7/2007-2013; grant no. 278570).
Not applicable.
DR wrote, edited and revised the manuscript. Data
authentication is not applicable. The author read and approved the
final manuscript.
Not applicable.
Not applicable.
The author declares that they have no competing
interests.
|
1
|
Senger DR, Galli SJ, Dvorak AM, Perruzzi
CA, Harvey VS and Dvorak HF: Tumor cells secrete a vascular
permeability factor that promotes accumulation of ascites fluid.
Science. 219:983–985. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ferrara N: Role of vascular endothelial
growth factor in physiologic and pathologic angiogenesis:
Therapeutic implications. Semin Oncol. 29:10–14. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bouzin C, Brouet A, De Vriese J, DeWever J
and Feron O: Effects of vascular endothelial growth factor on the
lymphocyte-endothelium interactions: Identification of Caveolin-1
and nitric oxide as control points of endothelial cell anergy. J
Immunol. 178:1505–1511. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dirkx AE, Oude Egbrink MG, Castermans K,
van der Schaft DW, Thijssen VL, Dings RP, Kwee L, Mayo KH, Wagstaff
J, Bouma-ter Steege JC and Griffioen AW: Anti-angiogenesis therapy
can overcome endothelial cell anergy and promote
leukocyte-endothelium interactions and infiltration in tumors.
FASEB J. 20:621–630. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Munn LL and Jain RK: Vascular regulation
of antitumor immunity. Science. 365:544–545. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tromp SC, oude Egbrink MG, Dings RP, van
Velzen S, Slaaf DW, Hillen HF, Tangelder GJ, Reneman RS and
Griffioen AW: Tumor angiogenesis factors reduce leukocyte adhesion
in vivo. Int Immunol. 12:671–676. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang J, Yan J and Liu B: Targeting
VEGF/VEGFR to modulate antitumor immunity. Front Immunol. 9:978.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Griffioen AW, Damen CA, Mayo KH,
Barendsz-Janson AF, Martinotti S, Blijham GH and Groenewegen G:
Angiogenesis inhibitors overcome tumor induced endothelial cell
anergy. Int J Cancer. 80:315–319. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shrimali RK, Yu Z, Theoret MR, Chinnasamy
D, Restifo NP and Rosenberg SA: Antiangiogenic agents can increase
lymphocyte infiltration into tumor and enhance the effectiveness of
adoptive immunotherapy of cancer. Cancer Res. 70:6171–6180. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wallin JJ, Bendell JC, Funke R, Sznol M,
Korski K, Jones S, Hernandez G, Mier J, He X, Hodi FS, et al:
Atezolizumab in combination with bevacizumab enhances migration of
antigen-specific T-cells in metastatic renal cell carcinoma. Nat
Commun. 7:126242016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hegde PS, Wallin JJ and Mancao C:
Predictive markers of anti-VEGF and emerging role of angiogenesis
inhibitors as immunotherapeutics. Semin Cancer Biol. 52:117–124.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Basu A, Hoerning A, Datta D, Edelbauer M,
Stack MP, Calzadilla K, Pal S and Briscoe DM: Cutting edge:
Vascular endothelial growth factor-mediated signaling in human
CD45RO+ CD4+ T cells promotes Akt and ERK activation and
costimulates IFN-gamma production. J Immunol. 184:545–549. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wada J, Suzuki H, Fuchino R, Yamasaki A,
Nagai S, Yanai K, Koga K, Nakamura M, Tanaka M, Morisaki T and
Katano M: The contribution of vascular endothelial growth factor to
the induction of regulatory T-cells in malignant effusions.
Anticancer Res. 29:881–888. 2009.PubMed/NCBI
|
|
14
|
Ohm JE, Gabrilovich DI, Sempowski GD,
Kisseleva E, Parman KS, Nadaf S and Carbone DP: VEGF inhibits
T-cell development and may contribute to tumor-induced immune
suppression. Blood. 101:4878–4886. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gavalas NG, Tsiatas M, Tsitsilonis O,
Politi E, Ioannou K, Ziogas AC, Rodolakis A, Vlahos G, Thomakos N,
Haidopoulos D, et al: VEGF directly suppresses activation of T
cells from ascites secondary to ovarian cancer via VEGF receptor
type 2. Br J Cancer. 107:1869–1875. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ziogas AC, Gavalas NG, Tsiatas M,
Tsitsilonis O, Politi E, Terpos E, Rodolakis A, Vlahos G, Thomakos
N, Haidopoulos D, et al: VEGF directly suppresses activation of T
cells from ovarian cancer patients and healthy individuals via VEGF
receptor type 2. Int J Cancer. 130:857–864. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim CG, Jang M, Kim Y, Leem G, Kim KH, Lee
H, Kim TS, Choi SJ, Kim HD, Han JW, et al: VEGF-A drives
TOX-dependent T cell exhaustion in anti-PD-1-resistant
microsatellite stable colorectal cancers. Science Immunol.
4:eaay05552019. View Article : Google Scholar
|
|
18
|
Voron T, Colussi O, Marcheteau E, Pernot
S, Nizard M, Pointet AL, Latreche S, Bergaya S, Benhamouda N,
Tanchot C, et al: VEGF-A modulates expression of inhibitory
checkpoints on CD8+ T cells in tumors. J Exp Med. 212:139–148.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Motz GT, Santoro SP, Wang LP, Garrabrant
T, Lastra RR, Hagemann IS, Lal P, Feldman MD, Benencia F and Coukos
G: Tumor endothelium FasL establishes a selective immune barrier
promoting tolerance in tumors. Nat Med. 20:607–615. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Apte RS, Chen DS and Ferrara N: VEGF in
signaling and disease: beyond discovery and development. Cell.
176:1248–1264. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen DS and Hurwitz H: Combinations of
Bevacizumab with cancer immunotherapy. Cancer J. 24:193–204. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lissoni P, Malugani F, Bonfanti A, Bucovec
R, Secondino S, Brivio F, Ferrari-Bravo A, Ferrante R, Vigoré L,
Rovelli F, et al: Abnormally enhanced blood concentrations of
vascular endothelial growth factor (VEGF) in metastatic cancer
patients and their relation to circulating dendritic cells, IL-12
and endothelin-1. J Biol Regul Homeost Agents. 15:140–144.
2001.PubMed/NCBI
|
|
23
|
Boissel N, Rousselot P, Raffoux E, Cayuela
JM, Maarek O, Charron D, Degos L, Dombret H, Toubert A and Rea D:
Defective blood dendritic cells in chronic myeloid leukemia
correlate with high plasmatic VEGF and are not normalized by
imatinib mesylate. Leukemia. 18:1656–1661. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Dikov MM, Ohm JE, Ray N, Tchekneva EE,
Burlison J, Moghanaki D, Nadaf S and Carbone DP: Differential roles
of vascular endothelial growth factor receptors 1 and 2 in
dendritic cell differentiation. J Immunol. 174:215–222. 2004.
View Article : Google Scholar
|
|
25
|
Oussa NA, Dahmani A, Gomis M, Richaud M,
Andreev E, Navab-Daneshmand AR, Taillefer J, Carli C, Boulet S,
Sabbagh L, et al: VEGF Requires the receptor NRP-1 to inhibit
lipopolysaccharide-dependent dendritic cell maturation. J Immunol.
197:3927–3935. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Curiel TJ, Wei S, Dong H, Alvarez X, Cheng
P, Mottram P, Krzysiek R, Knutson KL, Daniel B, Zimmermann MC, et
al: Blockade of B7-H1 improves myeloid dendritic cell-mediated
antitumor immunity. Nat Med. 9:562–567. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mimura K, Kono K, Takahashi A, Kawaguchi Y
and Fujii H: Vascular endothelial growth factor inhibits the
function of human mature dendritic cells mediated by VEGF
receptor-2. Cancer Immunol Immunother. 56:761–770. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Oyama T, Ran S, Ishida T, Nadaf S, Kerr L,
Carbone DP and Gabrilovich DI: Vascular endothelial growth factor
affects dendritic cell maturation through the inhibition of nuclear
factor-kappa B activation in hemopoietic progenitor cells. J
Immunol. 160:1224–1232. 1998.PubMed/NCBI
|
|
29
|
Crawford Y and Ferrara N: Tumor and
stromal pathways mediating refractoriness/resistance to
anti-angiogenic therapies. Trends Pharmacol Sci. 30:624–630. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Huang Y, Chen X, Dikov MM, Novitskiy SV,
Mosse CA, Yang L and Carbone DP: Distinct roles of VEGFR-1 and
VEGFR-2 in the aberrant hematopoiesis associated with elevated
levels of VEGF. Blood. 110:624–631. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Karakhanova S, Link J, Heinrich M,
Shevchenko I, Yang Y, Hassenpflug M, Bunge H, von Ahn K, Brecht R,
Mathes A, et al: Characterization of myeloid leukocytes and soluble
mediators in pancreatic cancer: Importance of myeloid-derived
suppressor cells. Oncoimmunology. 4:e9985192015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shojaei F, Wu X, Malik AK, Zhong C,
Baldwin ME, Schanz S, Fuh G, Gerber HP and Ferrara N: Tumor
refractoriness to anti-VEGF treatment is mediated by CD11b+Gr1+
myeloid cells. Nat Biotechnol. 25:911–920. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mantovani A and Locati M: Tumor-associated
macrophages as a paradigm of macrophage plasticity, diversity, and
polarization: Lessons and open questions. Arterioscler Thromb Vasc
Biol. 33:1478–1483. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Linde N, Lederle W, Depner S, van Rooijen
N, Gutschalk CM and Mueller MM: Vascular endothelial growth
factor-induced skin carcinogenesis depends on recruitment and
alternative activation of macrophages. J Pathol. 227:17–28. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Movahedi K, Laoui D, Gysemans C, Baeten M,
Stangé G, Van den Bossche J, Mack M, Pipeleers D, In't Veld P, De
Baetselier P and Van Ginderachter JA: Different tumor
microenvironments contain functionally distinct subsets of
macrophages derived from Ly6C(high) monocytes. Cancer Res.
70:5728–5739. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gabrusiewicz K, Liu D, Cortes-Santiago N,
Hossain MB, Conrad CA, Aldape KD, Fuller GN, Marini FC, Alonso MM,
Idoate MA, et al: Anti-vascular endothelial growth factor
therapy-induced glioma invasion is associated with accumulation of
Tie2-expressing monocytes. Oncotarget. 5:2208–2220. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bhowmick NA, Neilson EG and Moses HL:
Stromal fibroblasts in cancer initiation and progression. Nature.
432:332–337. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Raredon MSB, Adams TS, Suhail Y, Schupp
JC, Poli S, Neumark N, Leiby KL, Greaney AM, Yuan Y, Horien C, et
al: Single-cell connectomic analysis of adult mammalian lungs. Sci
Adv. 5:eaaw38512019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Barratt SL, Flower VA, Pauling JD and
Millar AB: VEGF (vascular endothelial growth factor) and fibrotic
lung disease. Int J Mol Med. 19:12692018.
|
|
40
|
Manzoni M, Rovati B, Ronzoni M, Loupakis
F, Mariucci S, Ricci V, Gattoni E, Salvatore L, Tinelli C, Villa E
and Danova M: Immunological effects of Bevacizumab-based treatment
in metastatic colorectal cancer. Oncology. 79:187–196. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Martino EC, Misso G, Pastina P, Costantini
S, Vanni F, Gandolfo C, Botta C, Capone F, Lombardi A, Pirtoli L,
et al: Immune-modulating effects of bevacizumab in metastatic
non-small-cell lung cancer patients. Cell Death Discov. 2:16025.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Alfaro C, Suarez N, Gonzalez A, Solano S,
Erro L, Dubrot J, Palazon A, Hervas-Stubbs S, Gurpide A,
Lopez-Picazo JM, et al: Influence of bevacizumab, sunitinib and
sorafenib as single agents or in combination on the inhibitory
effects of VEGF on human dendritic cell differentiation from
monocytes. Br J Cancer. 100:1111–1119. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Osada T, Chong G, Tansik R, Hong T,
Spector N, Kumar R, Hurwitz HI, Dev I, Nixon AB, Lyerly HK, et al:
The effect of anti-VEGF therapy on immature myeloid cell and
dendritic cells in cancer patients. Cancer Immunol Immunother.
57:1115–1124. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kusmartsev S, Eruslanov E, Kübler H, Tseng
T, Sakai Y, Su Z, Kaliberov S, Heiser A, Rosser C, Dahm P, et al:
Oxidative stress regulates expression of VEGFR1 in myeloid cells:
link to tumor-induced immune suppression in renal cell carcinoma. J
Immunol. 181:346–353. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Terme M, Pernot S, Marcheteau E, Sandoval
F, Benhamouda N, Colussi O, Dubreuil O, Carpentier AF, Tartour E
and Taieb J: VEGFA-VEGFR pathway blockade inhibits tumor-induced
regulatory T-cell proliferation in colorectal cancer. Cancer Res.
73:539–549. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Garon EB, Ciuleanu TE, Arrieta O, Prabhash
K, Syrigos KN, Goksel T, Park K, Gorbunova V, Kowalyszyn RD, Pikiel
J, et al: Ramucirumab plus docetaxel versus placebo plus docetaxel
for second-line treatment of stage IV non-small-cell lung cancer
after disease progression on platinum-based therapy (REVEL): A
multicentre, double-blind, randomised phase 3 trial. Lancet.
384:665–673. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
van Hooren L, Georganaki M, Huang H,
Mangsbo SM and Dimberg A: Sunitinib enhances the antitumor
responses of agonistic CD40-antibody by reducing MDSCs and
synergistically improving endothelial activation and T-cell
recruitment. Oncotarget. 7:50277–50289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ozao-Choy J, Ma G, Kao J, Wang GX, Meseck
M, Sung M, Schwartz M, Divino CM, Pan PY, Chen SH, et al: The novel
role of tyrosine kinase inhibitor in the reversal of immune
suppression and modulation of tumor microenvironment for
immune-based cancer therapies. Cancer Res. 69:2514–2522. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ko JS, Zea AH, Rini BI, Ireland JL, Elson
P, Cohen P, Golshayan A, Rayman PA, Wood L, Garcia J, et al:
Sunitinib mediates reversal of myeloid-derived suppressor cell
accumulation in renal cell carcinoma patients. Clin Cancer Res.
15:2148–2157. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Finke J, Ko J, Rini B, Rayman P, Ireland J
and Cohen P: MDSC as a mechanism of tumor escape from sunitinib
mediated anti-angiogenic therapy. Int Immunopharmacol. 11:856–861.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Guislain A, Gadiot J, Kaiser A, Jordanova
ES, Broeks A, Sanders J, van Boven H, de Gruijl TD, Haanen JB, Bex
A and Blank CU: Sunitinib pretreatment improves tumor-infiltrating
lymphocyte expansion by reduction in intratumoral content of
myeloid-derived suppressor cells in human renal cell carcinoma.
Cancer Immunol Immunother. 64:1241–1250. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Marasco M, Berteotti A, Weyershaeuser J,
Thorausch N, Sikorska J, Krausze J, Brandt HJ, Kirkpatrick J, Rios
P, Schamel WW, et al: Molecular mechanism of SHP2 activation by
PD-1 stimulation. Sci Adv. 6:eaay44582020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Sharpe AH, Wherry EJ, Ahmed R and Freeman
GJ: The function of programmed cell death 1 and its ligands in
regulating autoimmunity and infection. Nat Immunol. 8:239–245.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
54
|
Wei SC, Duffy CR and Allison JP:
Fundamental mechanisms of immune checkpoint blockade therapy.
Cancer Discov. 8:1069–1086. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chen PL, Roh W, Reuben A, Cooper ZA,
Spencer CN, Prieto PA, Miller JP, Bassett RL, Gopalakrishnan V,
Wani K, et al: Analysis of immune signatures in longitudinal tumor
samples yields insight into biomarkers of response and mechanisms
of resistance to immune checkpoint blockade. Cancer Discov.
6:827–837. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Huang Y, Yuan J, Righi E, Kamoun WS,
Ancukiewicz M, Nezivar J, Santosuosso M, Martin JD, Martin MR,
Vianello F, et al: Vascular normalizing doses of antiangiogenic
treatment reprogram the immunosuppressive tumor microenvironment
and enhance immunotherapy. Proc Natl Acad Sci USA. 109:17561–17566.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Allen E, Jabouille A, Rivera LB,
Lodewijckx I, Missiaen R, Steri V, Feyen K, Tawney J, Hanahan D,
Michael IP and Bergers G: Combined antiangiogenic and anti-PD-L1
therapy stimulates tumor immunity through HEV formation. Sci Transl
Med. 9:eaak96792017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Mayoux M, Roller A, Pulko V, Sammicheli S,
Chen S, Sum E, Jost C, Fransen MF, Buser RB, Kowanetz M, et al:
Dendritic cells dictate responses to PD-L1 blockade cancer
immunotherapy. Sci Transl Med. 12:5342020. View Article : Google Scholar : PubMed/NCBI
|