Introduction
The treatment of advanced or recurrent metastatic
solid tumors has been a major challenge in the medical community,
with limited treatment options and a poor prognosis. Furthermore,
the incidence of advanced solid tumors such as neuroendocrine
tumors (NETs) is still increasing globally (1). As the most notable means for the
treatment of unresectable tumors, molecularly targeted drugs such
as everolimus, sunitinib and capecitabine have been developed and
widely used in previous years (2–4).
Vascular endothelial growth factor (VEGF) is a key mediator of
tumor angiogenesis, and it is also considered an important
therapeutic target, receiving increasing attention from researchers
working on targeted molecular drugs (5). Surufatinib, also known as sulfatinib
in the past, is a potent, small-molecule tyrosine kinase inhibitor
(TKI), which selectively targets VEGF receptor (VEGFR)-1, −2 and
−3, fibroblast growth factor receptor-1 (FGFR-1) and
colony-stimulating factor-1 receptor (CSF-1R) (6).
Previous clinical studies revealed that surufatinib
had a notable effect on pancreatic and extra-pancreatic NETs
(7,8), and this drug also showed a high
objective response rate (ORR) for other solid tumors, such as
cholangiocarcinoma thyroid cancer and ovarian cancer (9,10).
However, high-quality evidence and verification studies by
randomized controlled trials are needed to clarify the efficacy and
safety of surufatinib. In the current study, a meta-analysis was
conducted to evaluate the safety and efficacy of surufatinib in
treating different types of advanced solid tumors. Overall, the
present study aimed to investigate whether surufatinib could exert
a practical effect on various solid tumors, including NETs, and
whether it could be considered a safe drug for treating patients
with solid tumors.
Materials and methods
Literature search
Literature screening was performed using four major
electronic databases, specifically PubMed (https://pubmed.ncbi.nlm.nih.gov/), EMBASE (https://www.embase.com/), Cochrane Library (https://www.cochranelibrary.com/) and ClinicalTrials.gov (https://clinicaltrials.gov/). The key words
‘surufatinib’ or ‘sulfatinib’ were used in the database search. All
screening results were divided into the categories ‘irrelevant’,
‘non-sofatinib’ and ‘possibly relevant’ based on the obtained study
abstracts. When multiple publications for the same clinical trial
were obtained, only the latest or most complete study was selected.
The databases were searched between the dates that they were
established and February 2022. To select the eligible studies, the
manuscripts in the ‘possibly relevant’ category were reviewed
according to the following inclusion criteria: i) Studies
describing surufatinib in advanced solid malignancies; and ii)
studies reporting tumor ORRs and/or toxicity. In addition, the
following exclusion criteria were applied: i) Case reports,
editorials, reviews, meta-analyses and review articles, as well as
animal and experimental studies; and ii) non-English articles.
Data extraction
The following data were extracted from every
selected article: Author(s), publication year, study design, number
of patients enrolled, age and sex of patients, Eastern Cooperative
Oncology Group-performance status (ECOG-ps) (11), as well as basic information,
including primary tumor location and tumor staging and grading
(12). Subsequently, stable disease
(SD), progressive disease (PD), partial response (PR), ORR, disease
control rate (DCR), duration of response and time to response were
extracted from these articles. In addition, the adverse effects,
including proteinuria, hypertriglyceridemia, diarrhea,
hypertension, abnormal high levels of alanine aminotransferase
(ALT), aspartate aminotransferase (AST) and blood bilirubin, were
added to the evaluation system.
Quality evaluation
Based on the particular content of the seven studies
selected, two quality assessment scales were used. Among the
aforementioned studies, five were observational studies using
Agency for Healthcare Research and Quality (AHRQ) scores (13), which included the following 11
items: i) A definition of the source of information (survey or
record review); ii) a listing of the inclusion and exclusion
criteria for exposed (case) and unexposed (controls) subjects or
reference to previous publications; iii) an indication of the time
period used to select patients; iv) an indication of whether all
patients were included in the study over a period of time if not
population-based; v) an indication of whether the evaluators of the
subjective components of every study were masked into other aspects
of the status of the patients; vi) a description of any assessments
undertaken for quality assurance purposes, such as testing or
re-testing of primary outcome measurements; vii) an explanation for
patient exclusion; viii) a description of how confounding variables
were assessed and/or controlled; ix) an explanation of how any
missing data were handled in the analysis, if applicable; x) a
summary of the patient response rates and completeness of data
collection; and xi) a clarification of the expected follow-up, if
any, and the percentage of patients for whom missing data or
follow-up was obtained. There were three answer forms for the AHRQ
scale: ‘yes’, ‘no’ or ‘unclear’. The other two studies were
randomized controlled trials (RCTs), and the Newcastle-Ottawa Scale
(NOS) was used (14). The NOS
scoring system included three aspects of evaluation: i) Selection;
ii) comparability; and iii) outcome. Selection was assessed by four
questions, comparability contained two options, while outcome was
also evaluated by three questions. A total of 9 points were
assigned.
Statistical analysis
The meta-analysis was performed using Microsoft
Office Excel (Microsoft Corporation) and StataCorp Stata 15.1
(StataCorp LP) software. Data were first transformed into
Freeman-Tukey double-arcsine, while the effect of treatment on the
incidence of adverse event ratios and their 95% confidence
intervals (95% CIs) were calculated using the DerSimonian and Laird
method (15) for meta-analyses with
random effects. To evaluate the heterogeneity among the included
studies, Higgins's I2 statistics and Q-tests were used.
Q-test results are expressed as P-values. An I2 value of
<25% indicated that there was no heterogeneity among studies.
Small, mild and strong heterogeneity were indicated by
I2 values of 25–50, 50–75 and >75%, respectively.
Overall, strong heterogeneity was assessed using heterogeneity
analysis. The results of the two RCTs were statistically analyzed
in the meta-analysis as aforementioned. P<0.05 indicated a
statistically significant difference. The current study was
registered on the International Platform of Registered Systematic
Review and Meta-analysis Protocols (INPLASY) platform (https://inplasy.com/) under the registration number,
INPLASY202260026.
Results
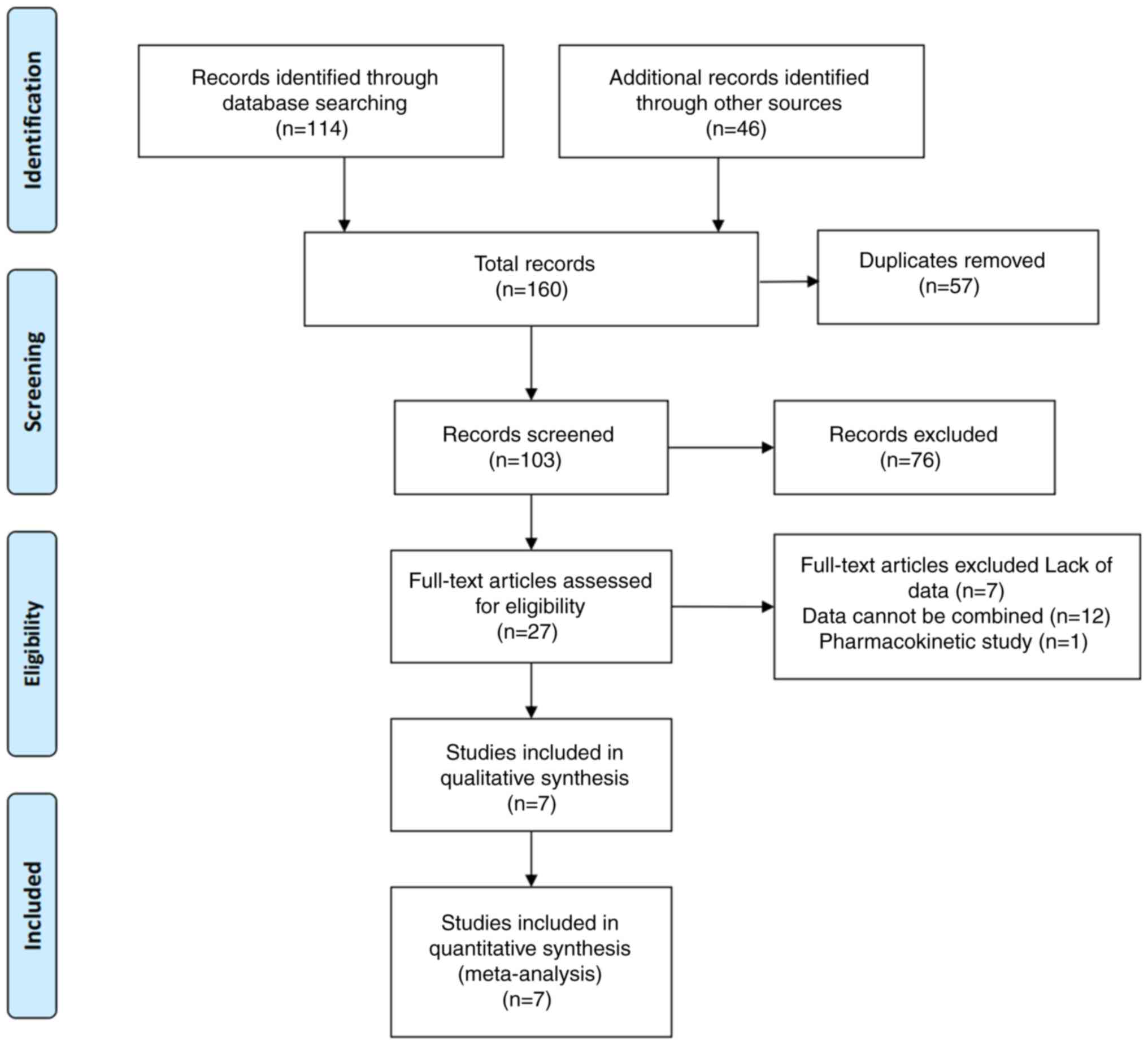
Search results
This systematic review was performed according to
the guidelines for the meta-analysis of observational
epidemiological studies (16). A
total of 160 articles were identified across the aforementioned
four major electronic databases. A total of 57 duplicate articles
were identified and excluded. Following literature screening by
title and abstract, a total of 76 irrelevant studies were also
excluded. After full text reading, an additional 20 studies were
excluded. Finally, seven clinical experimental studies that
included 638 participants (8–10,17–20)
were retained (Fig. 1). Among these
seven studies, five studies were single-arm trials, with a medium
or high score in the AHRQ scale (Table
I), while the other two were multicenter RCTs, with a score of
7–9 defining high quality studies on NOS (Table II).
 | Table I.Healthcare research and quality scale
score. |
Table I.
Healthcare research and quality scale
score.
| First author,
year | Definition of
source | Inclusion/exclusion
criteria | Time period | Included over
time | Masked
evaluators | Quality
assurance | Exclusion
explanation | Confounding
variable control | Missing data
processing | Summary | Clarification of
the follow-up | Total points | Quality | (Refs.) |
|---|
| Hamilton et
al, 2019 | Yes | No | Unclear | Unclear | Unclear | No | Yes | No | Yes | Yes | Yes | 5 | Medium | (19) |
| Chen et al,
2020 | Yes | Yes | No | Yes | Yes | No | Yes | Unclear | Yes | Yes | Yes | 8 | High | (10) |
| Xu et al,
2019 | Yes | Yes | No | Yes | Yes | Yes | No | No | Yes | Yes | Yes | 8 | High | (17) |
| Dasari et
al, 2020 | Yes | No | Unclear | Unclear | Unclear | No | Yes | No | Yes | Yes | No | 4 | Medium | (20) |
| Xu et al,
2021 | Yes | Yes | No | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | 9 | High | (9) |
 | Table II.Newcastle-Ottawa scale score. |
Table II.
Newcastle-Ottawa scale score.
|
|
|
|
|
| Comparability | Outcome |
|
|
|---|
|
| Selection |
|
|
|
|
|---|
|
|
| Analysis of the
most important factors | The second
important factor analysis | Determination of
exposure factors | Same approach | No response
rate | Score | (Refs.) |
|---|
| First author,
year | Case
identification | Representation of
cases | Choice of
comparison | Determination of
control |
|---|
| Xu et al,
20201st | 1 | 1 | - | 1 | 1 | 1 | 1 | 1 | - | 7 | (8) |
| Xu et al,
20202nd | 1 | 1 | - | 1 | 1 | 1 | 1 | 1 | - | 7 | (18) |
Treatment regimens
A total of 638 patients were included in the
meta-analysis; 510 were assigned to treatment arms, of which 35
patients participated in a dose escalation study (19). These patients were equally divided
into five groups and they were then treated with different
concentrations of surufatinib (50, 100, 200, 300 and 400 mg) once
daily and continuously for every 28-day cycle until disease
progression, intolerable toxicity or withdrawal of consent. An
additional 475 patients received 300 mg surufatinib once daily, in
28-day treatment cycles until disease progression, intolerable
toxicity or withdrawal of consent (8–10,17–20).
The 128 patients who participated in the RCT studies were randomly
assigned in either the placebo or the control arms, and the results
were not disclosed to the patients. Patients in the placebo group
received 300 mg placebo every day, in 4-week treatment cycles,
while patients in the control group received 300 mg of surufatinib
once daily in 4-week treatment cycles until disease progression,
intolerable toxicity, withdrawal of patient consent, poor
compliance, use of other antitumour medication, pregnancy, loss to
follow-up, or if the investigator deemed discontinuation to be in
the patient's best interest (8,18)
(Table III).
 | Table III.Characteristics of the study. |
Table III.
Characteristics of the study.
| First author,
year | Region | Type of study | Treatment
regimen | Patients, n | Sex, no. of
males/females | Mean age,
years | ECOG | Types of tumor | Pathological stage,
n | Funding | (Refs.) |
|---|
| Hamilton et
al, 2019 | USA | Open-label | Surufatinib,
50/100/200/300/400 mg qd; 28-day cycle | 35 | 23/34 | 62 | - | Any solid
tumor | - | Hutchison
MediPharma, Limited | (19) |
|
|
|
| Surufatinib, 300 mg
qd; 28-day cycle | 22 |
|
| - | Biliary tract
cancers; Pancreatic NETs; Extra-pancreatic NETs | - |
|
|
| Chen et al,
2020 | China | Open-label | Surufatinib, 300 mg
qd; 28-day cycle | 32 | 31/28 | 59 | 12/47 (0/1) | Differentiated
thyroid cancer | 1/31 (TNM stage
III/TNM stage IV) | Hutchison
MediPharma, Limited | (10) |
|
|
|
|
| 27 |
|
|
| Medullary thyroid
cancer | 27 (TNM stage
IV) |
|
|
| Xu et al,
2019 | China | Open-label | Surufatinib, 300 mg
qd; 28-day cycle | 42 | 25/17 | 46 | 32/10 (0/1) | Pancreatic
NETs | 7/35 (G1/G2) | Hutchison
MediPharma, Limited | (17) |
|
|
|
|
| 39 | 19/20 | 54 | 22/17 (0/1) | Extrapancreatic
NETs | 9/30 (G1/G2) |
|
|
| Dasari et
al, 2020 | USA | - | Surufatinib, 300
mg | 32 | - | - | - | Pancreatic
NETs | - | - | (20) |
|
|
|
|
|
|
|
|
| Extra-pancreatic
NETs |
|
|
|
| Xu et al,
20201st | China | RCT | Surufatinib, 300 mg
qd; 28-day cycle | 129 | 73/56 | 52 | 72/57 (0/1) | Extra-pancreatic
NETs | 21/108 (G1/G2) | Hutchison
MediPharma, Limited | (8) |
|
|
|
| Placebo, 300 mg qd;
28-day cycle | 69 | 35/34 | 54 | 46/23 (0/1) | Extra-pancreatic
NETs | 11/58 (G1/G2) |
|
|
| Xu et al,
20202nd | China | RCT | Surufatinib, 300 mg
qd; 28-day cycle | 113 | 53/60 | 51 | 73/40 (0/1) | Pancreatic
NETs | 14/99 (G1/G2) | Hutchison
MediPharma, Limited | (18) |
|
|
|
| Placebo, 300 mg qd;
28-day cycle | 59 | 31/28 | 48 | 43/16 (0/1) | Pancreatic
NETs | 9/50 (G1/G2) |
|
|
| Xu et al,
2021 | China | Open-label | Surufatinib of 300
mg qd; 28-day cycle | 29 | 22/17 | 56.2 | 10/29 (0/1) | Intra-hepatic
cholangio-carcinoma | 1/28 (TNM stage
III/TNM Stage IV) | Hutchison
MediPharma, Limited | (9) |
|
|
|
|
| 10 |
|
|
| Extra-hepatic
cholangio-carcinoma; Gallbladder cancer | 10 (TNM stage
IV) |
|
|
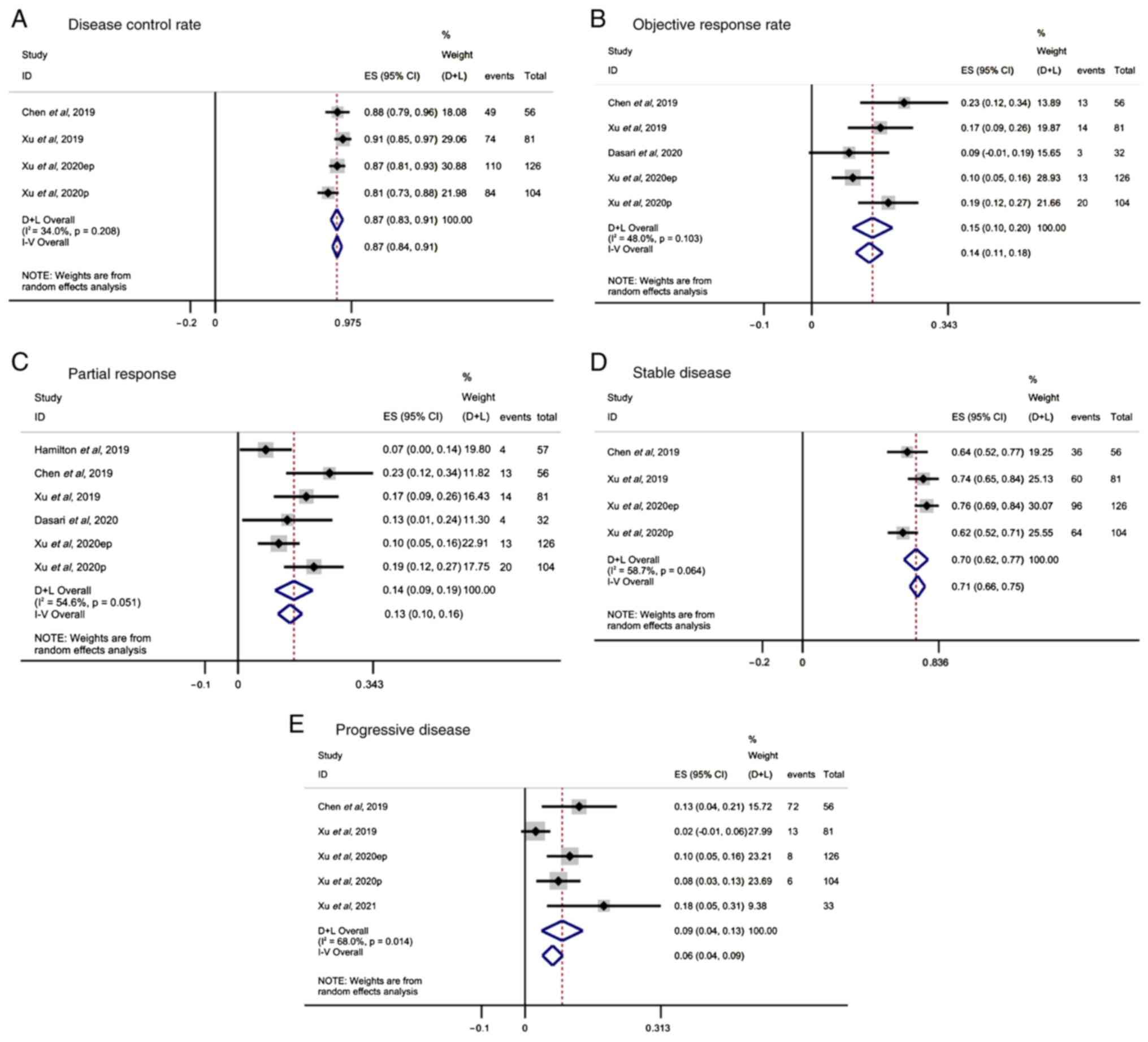
Tumor response
Tumor response was assessed in all studies using the
Response Evaluation Criteria in Solid Tumors (21). The DCR range reported in four
studies was 81–91%. The random effect analysis revealed that
surufatinib exerted a favorable DCR in patients with advanced solid
tumors (ES, 0.87; 95% CI, 0.84-0.91; I2=34%; P=0.208;
Fig. 2A). In addition, random
effect analysis of the pooled results from five studies also
indicated a beneficial ORR in patients with advanced solid tumors
(ES, 0.14; 95% CI, 0.11-0.18; I2=48%; P=0.103; Fig. 2B). PR in patients treated with
surufatinib was reported in seven studies. The pooled results
demonstrated that surufatinib significantly improved PR (ES, 0.13;
95% CI, 0.10-0.16; I2=54.6%; P=0.051; Fig. 2C). Additionally, SD was reported in
four studies. Overall, the analysis showed that SD was
significantly improved in patients with solid tumors treated with
surufatinib (ES, 0.71; 95% CI, 0.66-0.75; I2=58.7%;
P=0.064; Fig. 2D). Finally, the
pooled results regarding PD were reported in five studies and
suggested that surufatinib could also improve PD in the
aforementioned group of patients (ES, 0.06; 95% CI, 0.04-0.09;
I2=68%; P=0.014; Fig.
2E).
Safety assessment
In terms of drug safety, the analysis of the pooled
results from three studies suggested that treatment of patients
with advanced solid tumors with surufatinib notably reduced the
incidence of adverse events. The most common adverse effects in
patients treated with surufatinib were proteinuria (ES, 0.70; 95%
CI, 0.66-0.75; I2=69.3%; P=0.011; Fig. 3A), followed by hypertension (ES,
0.64; 95% CI, 0.59-0.68; I2=0%; P=0.588; Fig. 3B) and diarrhea (ES, 0.50; 95% CI,
0.45-0.54; I2=87.9%; P<0.001, Fig. 3C). Furthermore, increased AST (ES,
0.33; 95% CI, 0.28-0.38; I2=63.9%; P=0.040; Fig. 3D) and ALT (ES, 0.23; 95% CI,
0.18-0.27; I2=45.1%; P=0.141, Fig. 3E) levels were reported in four
studies. Additionally, three studies showed that surufatinib
enhanced the serum levels of thyrotropin (ES, 0.43; 95% CI,
0.38-0.48; I2=44.6%; P=0.164; Fig. 3F), while hypertriglyceridemia was
also reported in five studies (ES, 0.34; 95% CI, 0.30-0.39;
I2=35.6%; P=0.184; Fig.
3G). The I2 value for diarrhea was 87.9%, indicating
strong heterogeneity. Further sensitivity analysis resulted in a
reduced I2 value of 64.1% (ES, 0.43; 95% CI, 0.38-0.49;
I2=64.1%; P=0.039; Fig.
3H), supporting the robustness of the primary analysis. To
stabilize the changes in the ES ratios, double-arcsine
transformation was performed using the Freeman-Tukey transformation
formula (Table IV).
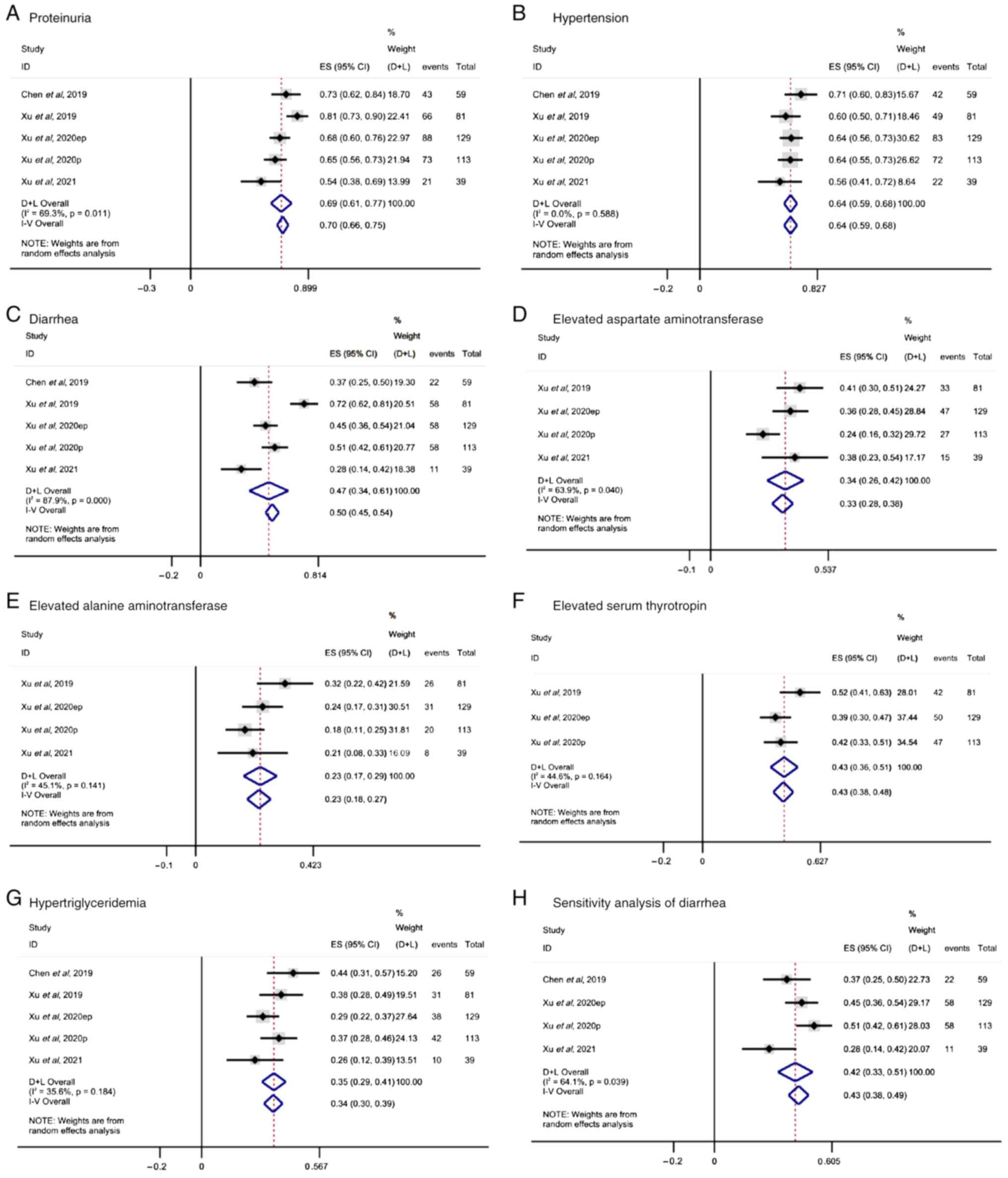 | Figure 3.Adverse events occurring in the
treatment of different solid tumors with surufatinib. (A)
Proteinuria, (B) hypertension, (C) diarrhea, (D) elevated aspartate
aminotransferase, (E) elevated alanine aminotransferase, (F)
elevated serum thyrotropin, (G) hypertriglyceridemia and (H)
sensitivity analysis of diarrhea. CI, confidence interval; ES,
effect size; D+L, Dersimonian-Laird; I–V, inverse variance. |
 | Table IV.Freeman-Tukey double arcsine
transformation. |
Table IV.
Freeman-Tukey double arcsine
transformation.
| Parameter | Rate | 95% CI | Post-conversion
rate | Transformed 95%
CI |
|---|
| DCR | 0.87 | 0.84-0.91 | 0.86 | 0.82-0.90 |
| ORR | 0.14 | 0.11-0.18 | 0.16 | 0.12-0.21 |
| PR | 0.13 | 0.10-0.16 | 0.15 | 0.11-0.20 |
| PD | 0.06 | 0.04-0.09 | 0.09 | 0.05-0.15 |
| Elevated ALT | 0.23 | 0.18-0.27 | 0.24 | 0.18-0.30 |
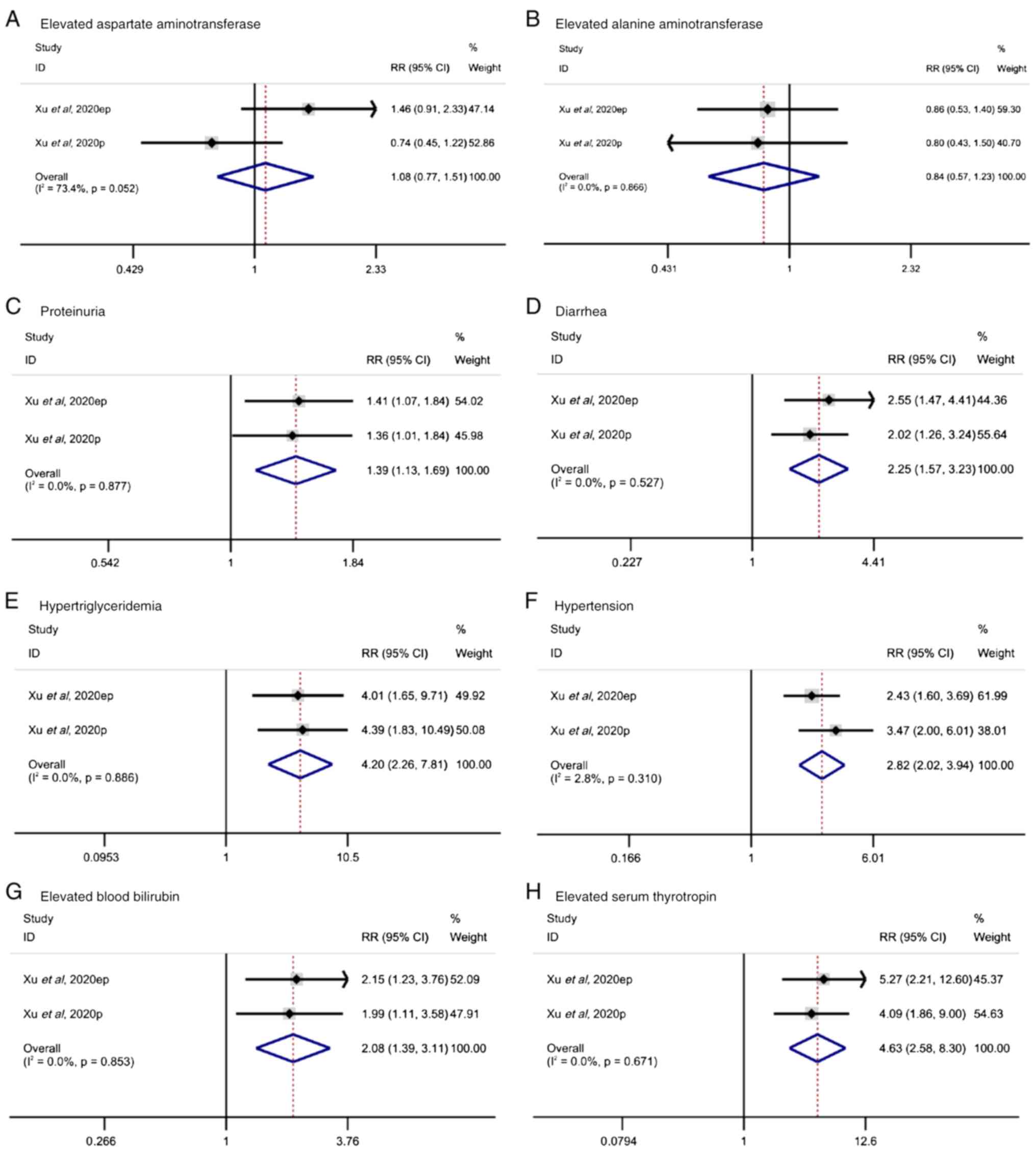
Relative risk analysis
To further verify the results of the current
meta-analysis, the results of two high-quality RCTs were analyzed.
These two RCTs compared the effect of surufatinib between patients
treated with surufatinib and those left untreated (placebo group).
The RR ratios of adverse events were also recorded. The levels of
AST were increased in the surufatinib group (RR, 1.08; 95% CI,
0.77-1.51), with mild heterogeneity (I2, 73.4%; P=0.052;
Fig. 4A). However, the difference
between treatment and placebo groups was not statistically
significant (P=0.658). Additionally, in both trials, increased ALT
levels were observed in the surufatinib group (RR, 0.84; 95% CI,
0.57-1.23), without heterogeneity (I2, 0%; P=0.886;
Fig. 4B). Again, statistical
significance was not reached (P=0.365). Among the other adverse
events, the pooled results for proteinuria (RR, 1.39; 95% CI,
1.13-1.69) showed no heterogeneity (I2, 0%; P=0.877;
Fig. 4C) with a statistically
significant difference (P=0.002). For diarrhea (RR, 2.25; 95% CI,
1.57-3.23), there was no heterogeneity (I2, 0%; P=0.527;
Fig. 4D), with a statistically
significant difference (P<0.001). For hypertriglyceridemia (RR,
4.2; 95% CI, 2.26-7.81), the pooled results revealed no
heterogeneity (I2, 0%; P=0.886; Fig. 4E), with a statistically significant
difference (P<0.001). For hypertension (RR, 2.82, 95% CI,
2.02-3.94), there was no heterogeneity (I2, 2.8%;
P=0.310; Fig. 4F), with a
statistically significant difference (P<0.001). For increased
blood bilirubin levels (RR, 2.08; 95% CI, 1.39-3.11), there was no
heterogeneity (I2, 0%; P=0.853; Fig. 4G), with a statistically significant
difference (P<0.001). Finally, for increased thyroid hormone
levels (RR, 4.63; 95% CI, 2.58-8.30), there was no heterogeneity
(I2, 0%; P=0.671; Fig.
4H), with a statistically significant difference
(P<0.001).
Publication bias
Since a small number of studies were included in the
meta-analysis, publication bias was not assessed. Therefore, bias
might have occurred in the analysis and the evaluation of the
research data.
Discussion
Different types of solid tumors, such as pancreatic
NETs, extra-pancreatic NETs and biliary tract cancer, are
characterized by different degrees of aggressiveness and require
different treatment approaches. Peptide receptor radionuclide
therapy with lutetium-177 dotatate (177 Lu-DOTATATE) has been
approved for advanced gastroenteropancreatic-NETs and the
antiangiogenic agent sunitinib is mainly used for pancreatic NETs
(22). In recent years, more and
more researchers have paid increasing attention to the most common
characteristics, such as abnormal angiogenesis and gene mutations,
of different types of solid tumors, such as tumors of the uterus,
pancreatic cancer and thyroid cancer (23,24),
while investigating more effective treatment approaches for several
of them (25–27).
It has been reported that VEGF and FGF can mediate
the formation of tumor blood vessels in solid tumors to provide
sufficient oxygen, thereby promoting tumor growth, cell migration
and invasion (28–30). It has been also suggested that
several VEGFR-targeted therapies are unsatisfactory, with numerous
patients exhibiting no or a limited respond to treatment, possibly
due to the induction of other pro-angiogenic pathways, including
those of FGFR and CSF-1R that promote drug resistance (7,28–32).
Surufatinib is a novel, orally administered,
small-molecule TKI of VEGFR-113, FGFR-1 and CSF-1R, which can
reduce tumor angiogenesis and immune system evasion, and also
enhance antitumor immunity (31) to
treat pancreatic and extra-pancreatic NETs (32). Several systemic therapies have been
approved for the treatment of NETs, including somatostatin analogue
therapy, the TKI sunitinib, mammalian target of rapamycin
inhibitors, peptide receptor radionuclide therapy (PRRT) and
cytotoxic chemotherapy (23,31–33).
The effect of aminokinase inhibitors can be counteracted by the
induction of other proangiogenic pathways, including the FGFR-1 and
CSF-1R signaling pathways (31).
Therefore, the application of surufatinib could provide a solution
to this issue to some extent. The first studies on surufatinib were
performed in China, while the scope of its application has
gradually expanded from NETs to several different types of advanced
solid tumors, thus attracting worldwide attention (8–10,17,18).
To the best of our knowledge, the present study was
the first meta-analysis to particularly focus on the efficacy and
safety of surufatinib in solid tumors. In the current
meta-analysis, seven studies involving 638 patients suffering from
different types of solid tumors, such as pancreatic NETs,
extra-pancreatic NETs, biliary tract cancer and thyroid cancer,
were included. The results of the meta-analysis revealed overall
DCR, ORR, PR, SD and PD rates of 87, 14, 1, 71 and 6%,
respectively. The results also showed that compared with
177Lu-DOTATATE PRRT, immunosuppressive agents, such as programmed
cell death protein-1 (PD-1)/PD-ligand-1 (PD-L1) inhibitors and
cytokine-induced killer cells, surufatinib resulted in higher DCR
and SD rates, while PD also remained at a lower level. This
supports the fact that surufatinib could exhibit a better efficacy
in the treatment of solid tumors (21,34,35).
The study by Xu et al (8)
showed that at the time of data cut-off, 77 (60%) patients in the
surufatinib group and 51 (74%) in the placebo group experienced
progression-free survival (PFS) events. The median follow-up time
for PFS was 13.8 months (95% CI, 11.1-16.7) and 16.6 months (95%
CI, 9.2-not calculable) in the surufatinib and placebo groups,
respectively. Additionally, the median investigator-assessed PFS
time was 9.2 months (95% CI, 7.4-11.1) in the surufatinib group and
3.8 months (95% CI, 3.7-5.7) in the placebo group (HR, 0.33; 95%
CI, 0.22-0.50; P<0.0001), which crossed the predefined P-value
threshold (two-sided, P=0.015) for the interim analysis (8). The aforementioned findings also
supported the beneficial effect of surufatinib on the prognosis of
patients with solid tumors.
In addition to analyzing the direct efficacy of
surufatinib in the treatment of solid tumors, the present study
also analyzed the adverse events that occurred during the treatment
of patients with surufatinib. More specifically, three studies
reported treatment-emergent adverse events with an overall
incidence of 44%, and four articles mentioned increased AST and ALT
levels with an incidence of 23 and 33%, respectively, while five
studies reported proteinuria (70%), diarrhea (50%),
hypertriglyceridemia (34%) and hypertension (64%). Increased
thyrotropin serum levels were reported in three articles (43%).
When compared with a placebo, there was no
statistically significant difference in increased AST and ALT
levels in patients treated with surufatinib, thus indicating that
surufatinib could not reduce the incidence of the aforementioned
adverse reactions. However, there was a statistically significant
difference in the incidence of proteinuria, diarrhea,
hypertriglyceridemia, hypertension, increased blood bilirubin and
thyrotropin serum levels between the placebo and surufatinib
groups, suggesting that treatment of patients with advanced solid
tumors with surufatinib could notably decrease the incidence of the
aforementioned adverse events. Conclusively, overall analysis
indicated that surufatinib could reduce the incidence of adverse
events, indicating a satisfactory safety profile.
On the other hand, the RR of adverse events, such as
diarrhea, hypertension and hyperbilirubinemia in patients treated
with surufatinib was decreased compared with those treated with
regorafenib (36). The incidence of
gastrointestinal-related adverse events during the treatment of
refractory thyroid cancer was lower in patients treated with
surufatinib compared with that in patients treated with sorafenib
(37). Additionally, surufatinib
was associated with a lower incidence of diarrhea compared with
sorafenib, sunitinib and pazopanib. Overall, the reduced RR of
gastrointestinal-related adverse events suggested that surufatinib
exhibited high safety in treating patients with solid tumors
(38).
The current study analyzed the results of seven
clinical studies with regard to the therapeutic effect of
surufatinib on solid tumors, such as pancreatic NETs,
extra-pancreatic NETs and cholangiocarcinoma. However, the present
study had some limitations. Surufatinib was approved in China in
December 2020 for the treatment of late-stage, well-differentiated,
extrapancreatic NETs (39,40). Surufatinib has been filed for
approval in China for the treatment of pancreatic NETs (41) and in the USA for the treatment of
pancreatic and extrapancreatic NETs (42). Since surufatinib has been only
recently used in clinical practice to treat advanced solid tumors
(5), the currently available
studies on surufatinib are limited. Further studies on different
types of solid tumors are urgently needed. In addition, this was a
single-ratio meta-analysis with high heterogeneity; even in the
categorical analyses performed, the existence of partial
heterogeneity could not be ruled out. However, the magnitude of the
observed treatment effect, the consistency of the diarrhea
incidence rates in the sensitivity analysis and the lower incidence
of adverse events in patients treated with surufatinib compared
with those treated with other agents, provided evidence of the
clinically meaningful benefit of surufatinib.
In conclusion, the findings of the current
meta-analysis suggested that surufatinib could be considered an
effective and safe drug in the treatment of solid tumors.
Additional clinical data and original studies of high quality are
required to further validate the results of the present study and
to provide more evidence to support the clinical applications of
surufatinib.
Acknowledgements
Not applicable.
Funding
This study was supported by funding from the National Natural
Science Foundation of China (grant no. 82170587), the Luzhou
Municipal People's Government-Southwest Medical University Science
and Technology Strategic Cooperation Project (grant no.
2021LZXNYD-Z01), the Sichuan Province International Science and
Technology Innovation Cooperation Project (grant no. 2023YFH0078)
and the Talent Development Project of the Affiliated Hospital of
Southwest Medical University.
Availability of data and materials
The datasets generated during and/or analyzed during
the current study are available from the corresponding author on
reasonable request.
Authors' contributions
TYC and YLC conceptualized the study and visualized
the data. YLC collected and analyzed data. TYC wrote and prepared
the original draft. WGF conceptualized and supervised the study.
YCD and PT collected and analyzed data, and edited the manuscript.
TXL and YFC analyzed data and edited the manuscript. LG performed
the statistical analysis, supervised the study and edited the
manuscript. All authors have read and approved the final version of
the manuscript. TYC and YLC confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Shah MH, Goldner WS, Benson AB, Bergsland
E, Blaszkowsky LS, Brock P, Chan J, Das S, Dickson PV, Fanta P, et
al: Neuroendocrine and adrenal tumors, version 2.2021, NCCN
clinical practice guidelines in oncology. J Natl Compr Canc Netw.
19:839–868. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yao JC, Pavel M, Lombard-Bohas C, Cutsem
EV, Voi M, Brandt U, He W, Chen D, Capdevila J, de Vries EGE, et
al: Everolimus for the treatment of advanced pancreatic
neuroendocrine tumors: Overall survival and circulating biomarkers
from the randomized, phase III RADIANT-3 study. J Clin Oncol.
34:3906–3913. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Raymond E, Dahan L, Raoul JL, Bang YJ,
Borbath I, Lombard-Bohas C, Valle J, Metrakos P, Smith D, Vinik A,
et al: Sunitinib malate for the treatment of pancreatic
neuroendocrine tumors. New Engl J Med. 364:501–513. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lu Y, Zhao Z, Wang J, Lv W, Lu L, Fu W and
Li W: Safety and efficacy of combining capecitabine and
temozolomide (CAPTEM) to treat advanced neuroendocrine neoplasms: A
meta-analysis. Medicine (Baltimore). 97:e127842018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hosaka K, Yang Y, Seki T, Du Q, Jing X, He
X, Wu J, Zhang Y, Morikawa H, Nakamura M, et al: Therapeutic
paradigm of dual targeting VEGF and PDGF for effectively treating
FGF-2 off-target tumors. Nat Commun. 11:37042020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Syed YY: Surufatinib: First approval.
Drugs. 81:723–732. 2021. View Article : Google Scholar
|
|
7
|
Xu JM, Wang Y, Chen YL, Jia R, Li J, Gong
JF, Li J, Qi C, Hua Y, Tan CR, et al: Sulfatinib, a novel kinase
inhibitor, in patients with advanced solid tumors: Results from a
phase I study. Oncotarget. 8:42076–42086. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xu J, Shen L, Zhou Z, Li J, Bai C, Chi Y,
Li Z, Xu N, Li E, Liu T, et al: Surufatinib in advanced
extrapancreatic neuroendocrine tumours (SANET-ep): A randomised,
double-blind, placebo-controlled, phase 3 study. Lancet Oncol.
21:1500–1512. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xu J, Bai Y, Sun H, Bai C, Jia R, Li Y,
Zhang W, Liu L, Huang C, Guan M, et al: A single-arm, multicenter,
open-label phase 2 trial of surufatinib in patients with
unresectable or metastatic biliary tract cancer. Cancer.
127:3975–3984. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen J, Ji Q, Bai C, Zheng X, Zhang Y, Shi
F, Li X, Tang P, Xu Z, Huang R, et al: Surufatinib in Chinese
patients with locally advanced or metastatic differentiated thyroid
cancer and medullary thyroid cancer: A multicenter, open-label,
phase II trial. Thyroid. 30:1245–1253. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern cooperative oncology group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
O'Sullivan B, Brierley J, Byrd D, Bosman
F, Kehoe S, Kossary C, Pineros M, Van Eycken E, Weir HK and
Gospodarowicz M: The TNM classification of malignant
tumours-towards common understanding and reasonable expectations.
Lancet Oncol. 18:849–851. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Carlson EA: Agency for healthcare research
and quality (AHRQ) web site. Orthop Nurs. 27:258–259. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Stang A: Critical evaluation of the
Newcastle-Ottawa scale for the assessment of the quality of
nonrandomized studies in meta-analyses. Eur J Epidemiol.
25:603–605. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
DerSimonian R and Laird N: Meta-analysis
in clinical trials. Control Clin Trials. 7:177–188. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: The PRISMA 2020 statement: An updated guideline for
reporting systematic reviews. BMJ. 372:n712021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xu J, Li J, Bai C, Xu N, Zhou Z, Li Z,
Zhou C, Jia R, Lu M, Cheng Y, et al: Surufatinib in advanced
well-differentiated neuroendocrine tumors: A multicenter,
single-arm, open-label, phase Ib/II trial. Clin Cancer Res.
25:3486–3494. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xu J, Shen L, Bai C, Wang W, Li J, Yu X,
Li Z, Li E, Yuan X, Chi Y, et al: Surufatinib in advanced
pancreatic neuroendocrine tumours (SANET-p): A randomised,
double-blind, placebo-controlled, phase 3 study. Lancet Oncol.
21:1489–1499. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hamilton E, Wang JS, Li D, Dasari NA,
Paulson S, Cohn AL, Sauter NP, Kania M, Kauh K and Falchook GS:
1393P-Safety and tolerability of surufatinib in western patients
with solid tumours. Ann Oncol. 30 (Suppl 5):v569–v570. 2019.
View Article : Google Scholar
|
|
20
|
Dasari A, Li D, Sung MW, Tucci C, Kauh JS,
Kania MK and Paulson AS: Efficacy and safety of surufatinib in
United States (US) patients (pts) with neuroendocrine tumors
(NETs). J Clin Oncol. 38 (15 Suppl):S46102020. View Article : Google Scholar
|
|
21
|
Ma Y, Zhang Z, Tang L, Xu YC, Xie ZM, Gu
XF and Wang HX: Cytokine-induced killer cells in the treatment of
patients with solid carcinomas: A systematic review and pooled
analysis. Cytotherapy. 14:483–493. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cives M and Strosberg JR:
Gastroenteropancreatic neuroendocrine tumors. CA Cancer J Clin.
68:471–487. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hyman DM, Smyth LM, Donoghue MTA, Westin
SN, Bedard PL, Dean EJ, Bando H, El-Khoueiry AB, Pérez-Fidalgo JA,
Mita A, et al: AKT inhibition in solid tumors with AKT1 mutations.
J Clin Oncol. 35:2251–2259. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ukidve A, Cu K, Kumbhojkar N, Lahann J and
Mitragotri S: Overcoming biological barriers to improve solid tumor
immunotherapy. Drug Deliv Transl Res. 11:2276–2301. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xiao WY, Wang Y, An HW, Hou D, Mamuti M,
Wang MD, Wang J, Xu W, Hu L and Wang H: Click Reaction-Assisted
Peptide Immune Checkpoint Blockade for Solid Tumor Treatment. ACS
Appl Mater Interfaces. 12:40042–40051. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Heery CR, O'Sullivan-Coyne G, Madan RA,
Cordes L, Rajan A, Rauckhorst M, Lamping E, Oyelakin I, Marté JL,
Lepone LM, et al: Avelumab for metastatic or locally advanced
previously treated solid tumours (JAVELIN Solid Tumor): A phase 1a,
multicohort, dose-escalation trial. Lancet Oncol. 18:587–598. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Speck T, Heidbuechel JPW, Veinalde R,
Jaeger D, von Kalle C, Ball CR, Ungerechts G and Engeland CE:
Targeted BiTE expression by an oncolytic vector augments
therapeutic efficacy against solid tumors. Clin Cancer Res.
24:2128–2137. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cébe-Suarez S, Zehnder-Fjällman A and
Ballmer-Hofer K: The role of VEGF receptors in angiogenesis;
complex partnerships. Cell Mol Life Sci. 63:601–615. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Daniele G, Corral J, Molife LR and de Bono
JS: FGF receptor inhibitors: Role in cancer therapy. Curr Oncol
Rep. 14:111–119. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Al-Ostoot FH, Sherapura A, V V, Basappa G,
H K V, B T P and Khanum SA: Targeting HIF-1α by newly synthesized
Indolephenoxyacetamide (IPA) analogs to induce
anti-angiogenesis-mediated solid tumor suppression. Pharmacol Rep.
73:1328–1343. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xu J: Current treatments and future
potential of surufatinib in neuroendocrine tumors (NETs). Ther Adv
Med Oncol. 13:175883592110426892021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lu X, Yan S, Koral KA and Chen Z:
Surufatinib for the treatment of advanced extrapancreatic
neuroendocrine tumors. Expert Rev Anticancer Ther. 21:917–926.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bodei L, Kwekkeboom DJ, Kidd M, Modlin IM
and Krenning EP: Radiolabeled somatostatin analogue therapy of
gastroenteropancreatic cancer. Semin Nucl Med. 46:225–238. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rao Q, Li M, Xu W, Pang K, Guo X, Wang D,
Liu J, Guo W and Zhang Z: Clinical benefits of PD-1/PD-L1
inhibitors in advanced hepatocellular carcinoma: A systematic
review and meta-analysis. Hepatol Int. 14:765–775. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang J, Song Q, Cai L, Xie Y and Chen Y:
The efficacy of 177Lu-DOTATATE peptide receptor
radionuclide therapy (PRRT) in patients with metastatic
neuroendocrine tumours: A systematic review and meta-analysis. J
Cancer Res Clin Oncol. 146:1533–1543. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yin X, Yin Y, Shen C, Chen H, Wang J, Cai
Z, Chen Z and Zhang B: Adverse events risk associated with
regorafenib in the treatment of advanced solid tumors:
Meta-analysis of randomized controlled trials. Onco Targets Ther.
11:6405–6414. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Feng G, Luo Y, Zhang Q, Zeng F, Xu J and
Zhu J: Sorafenib and radioiodine-refractory differentiated thyroid
cancer (RR-DTC): A systematic review and meta-analysis. Endocrine.
68:56–63. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Santoni M, Conti A, De Giorgi U, Iacovelli
R, Pantano F, Burattini L, Muzzonigro G, Berardi R, Santini D and
Cascinu S: Risk of gastrointestinal events with sorafenib,
sunitinib and pazopanib in patients with solid tumors: A systematic
review and meta-analysis of clinical trials. Int J Cancer.
135:763–773. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
HUTCHMED, . Chi-Med announces the NMPA
approval of surufatinib (Sulanda® in China) for
non-pancreatic neuroendocrine tumors. https://www.hutch-med.com/chi-med-announces-the-nmpa-approval-of-surufatinib-sulanda-in-china-for-epnet/December
30–2020
|
|
40
|
HUTCHMED, . Surufatinib (Sulanda): Chinese
prescribing information. https://www.hutch-med.com/sc/pipeline-and-products/our-products/#sulanda-scDecember
29–2020
|
|
41
|
HUTCHMED, . Chi-Med announces second NDA
acceptance in China for surufatinib in pancreatic neuroendocrine
tumors. https://www.hutch-med.com/chi-med-announces-second-nda-acceptance-in-china-for-surufatinib-in-pancreatic-net/September
17–2020
|
|
42
|
HUTCHMED, . Chi-Med initiates rolling
submission of NDA to U.S. FDA for surufatinib for the treatment of
advanced neuroendocrine tumors. https://www.hutch-med.com/chi-med-initiates-rolling-submission-of-nda-to-us-fda-for-surufatinib-for-the-treatment-of-advanced-net/December
28–2020
|