Introduction
Pancreatic cancer is the fourth leading cause of
cancer death and has one of the poorest prognoses, with a very low
5-year survival rate of about 10% in Japan and the United States
(1–3). Its incidence is increasing (1,3), and
most cases (−80%) are unresectable at diagnosis. FOLFIRINOX
(including fluorouracil, folinic acid, oxaliplatin, and irinotecan)
(4–6) and gemcitabine plus nab-paclitaxel
(7) have been reported as the
standard first-line therapies for advanced/recurrent pancreatic
cancer, but the prognosis remains poor with median overall survival
(OS) of 9–12 months in clinical trials (4,7).
Conversely, our real-world study, encompassing 846 cases of
metastatic pancreatic cancer initially treated between 2010 and
2020, revealed a median OS of just 6.8 months (8).
Even in cases with unfavorable prognoses, an
accurate prediction of the clinical outcomes is crucial. The
usefulness of inflammation-based and nutritional markers in
patients with cancer has been widely reported (9). Historically, various prognostic
markers and their correlation with outcomes in cancer patients have
been reported since the 1980s (10–12).
To date, numerous markers have been investigated for their utility
in diverse scenarios, and due to their convenience, they are widely
utilized in actual clinical practice. Nonetheless, data on their
direct comparisons are limited. It's essential to validate the most
suitable markers for each specific clinical setting.
The objective of this study is to conduct a direct
comparison of the various inflammation-based prognostic markers
reported to date, utilizing the aforementioned dataset (8), in order to identify the most accurate
markers for assessing prognosis in metastatic pancreatic
cancer.
Patients and methods
Study overview
The Tokushukai Real-world Data (TREAD) project is a
retrospective cohort study conducted at Tokushukai Medical Group
hospitals. Tokushukai Medical Group is a leading medical group in
Japan, encompassing 71 general hospitals nationwide. The study
utilizes a shared medical record system across these hospitals.
(e-Karte and Newtons2; Software Service Inc., Osaka, Japan) and
chemotherapy protocol system (srvApmDrop; Software Service Inc.,
Osaka, Japan), the details of which can be found in a separate
article (13). The project adhered
to the ethical guidelines for medical and biological research
involving human subjects in Japan (14) and followed the principles of the
Declaration of Helsinki. Approval for the study was obtained from
the Ethics Committee of the Tokushukai Group in April 2020
(approval no. TGE01427-024). Patients were informed about the
opt-out method, and the study was registered in the UMIN Clinical
Trial Registry under the number UMIN000050590.
Patients
We identified 846 patients with pathologically or
radiologically confirmed primary metastatic pancreatic cancer who
underwent first-line chemotherapy at Tokushukai Medical Group
hospitals between April 1, 2010, and March 31, 2020 (8). Briefly, the patients were treated with
gemcitabine, S-1, gemcitabine plus S-1, gemcitabine plus
nab-paclitaxel, or FOLFIRINOX as their first-line treatment.
Patients with pathological diagnoses of adenocarcinoma,
adenosquamous carcinoma, and carcinoma/malignant neoplasms were
included in the analysis. Patients with active double cancer,
inadequate treatment history, and missing fundamental patient data
were excluded from the study.
Data collection
As separately described, information on patients,
tumor-related factors, study period (A: 2010–2013, B: 2014–2016, C:
2017–2020), hospital volume (high- and low-volume hospitals),
hospital type (government-designated cancer hospital,
prefecture-designated cooperative cancer hospital, or
non-designated general hospital), and first-line chemotherapy
regimens was extracted from the medical record system, the
chemotherapy protocol system, and the National Cancer Registry Data
in Japan (15).
For supplemental analysis, blood laboratory data for
different parameters [white blood cells, neutrocytes, lymphocytes,
monocytes, hemoglobin, platelets, total bilirubin, aspartate
aminotransferase, alanine aminotransferase, lactate dehydrogenase
(LDH), γ-glutamyl transpeptidase, alkaline phosphatase, creatinine,
creatinine clearance, c-reactive protein (CRP), albumin, glucose,
hemoglobin A1c, carcinoembryonic antigen (CEA) and carbohydrate
antigen 19-9 (CA19-9)] collected within 14 days of first-line
treatment were extracted from the electronic medical record, and
levels of inflammation-based prognostic markers were calculated
(16–29). The markers used in this study are
listed in Table I. For the markers,
previously defined cutoff values were used, but for those that were
not defined, we referred to previous studies (24,25).
 | Table I.Systemic inflammation-based
prognostic scores and ratios. |
Table I.
Systemic inflammation-based
prognostic scores and ratios.
| Definition | Score or ratio |
|---|
| PNI |
|
| PNI;
Onodera et al (16) |
|
|
10 × albumin
(g/dl) + 0.005 × lymphocyte count (/dl) | ≥45 |
|
10 × albumin
(g/dl) + 0.005 × lymphocyte count (/dl) | <45 |
| GPS |
|
| GPS;
original (17) |
|
|
C-reactive protein
≤1.0 (mg/dl) and albumin ≥3.5 (g/dl) | 0 |
|
C-reactive protein
>1.0 (mg/dl) or albumin <3.5 (g/dl) | 1 |
|
C-reactive protein
>1.0 (mg/dl) and albumin <3.5 (g/dl) | 2 |
| J-mGPS
(20) |
|
|
C-reactive protein
≤0.5 (mg/dl) and albumin ≥3.5 (g/dl) | 0 |
|
C-reactive protein
>0.5 (mg/dl) or albumin <3.5 (g/dl) | 1 |
|
C-reactive protein
>0.5 (mg/dl) and albumin <3.5 (g/dl) | 2 |
| CAR (18) |
|
|
C-reactive protein
(mg/dl)/albumin (g/dl) | ≤0.22 |
|
C-reactive protein
(mg/dl)/albumin (g/dl) | >0.22 |
| NLR (19) |
|
|
Neutrophil count
(/µl)/lymphocyte count (/µl) | <3 |
|
Neutrophil count
(/µl)/lymphocyte count (/µl) | ≥3-<5 |
|
Neutrophil count
(/µl)/lymphocyte count (/µl) | ≥5 |
| PLR (20) |
|
|
Platelet count
(/µl)/lymphocyte count (/µl) | ≤150 |
|
Platelet count
(/µl)/lymphocyte count (/µl) | >150 |
| LMR (21) |
|
|
Lymphocyte count
(/µl)/monocyte count (/µl) | ≥2.40 |
|
Lymphocyte count
(/µl)/monocyte count (/µl) | <2.40 |
| dNLR (22) |
|
|
Neutrophil count
(/µl)/(leukocyte count (/µl)-neutrophil count (/µl)) | <3 |
|
Neutrophil count
(/µl)/(leukocyte count (/µl)-neutrophil count (/µl)) | ≥3-<5 |
|
Neutrophil count
(/µl)/(leukocyte count (/µl)-neutrophil count (/µl)) | ≥5 |
| NPS (23) |
|
|
Neutrophil count ≤7,500 (/µl)
and platelet count ≤400,000 (/µl) | 0 |
|
Neutrophil count >7,500
(/µl) or platelet count >400,000 (/µl) | 1 |
|
Neutrophil count >7,500
(/µl) and platelet count >400,000 (/µl) | 2 |
| NLS (24,25) |
|
|
Neutrophil count ≤7,500 (/µl)
and lymphocyte count ≥1,500 (/µl) | 0 |
|
Neutrophil count >7,500
(/µl) or lymphocyte count <1,500 (/µl) | 1 |
|
Neutrophil count >7,500
(/µl) and lymphocyte count <1,500 (/µl) | 2 |
| PLS (24,25) |
|
|
Platelet count ≤400,000 (/µl)
and lymphocyte count ≥1,500 (/µl) | 0 |
|
Platelet count >400,000
(/µl) or lymphocyte count <1,500 (/µl) | 1 |
|
Platelet count >400,000
(/µl) and lymphocyte count <1,500 (/µl) | 2 |
| LMS (24,25) |
|
|
Lymphocyte count ≥1,500 (/µl)
and monocyte count ≤800 (/µl) | 0 |
|
Lymphocyte count <1,500
(/µl) or monocyte count >800 (/µl) | 1 |
|
Lymphocyte count <1,500
(/µl) and monocyte count >800 (/µl) | 2 |
| PI (23) |
|
|
C-reactive protein ≤1.0
(mg/dl) and leukocyte count ≤11,000 (/µl) | 0 |
|
C-reactive protein >1.0
(mg/dl) or leukocyte count >11,000 (/µl) | 1 |
|
C-reactive protein >1.0
(mg/dl) and leukocyte count >11,000 (/µl) | 2 |
| SII (26) |
|
|
Neutrophil count (/µl) × 10 ×
platelet count (/µl)/lymphocyte count (/µl) | <300 |
|
Neutrophil count (/µl) × 10 ×
platelet count (/µl)/lymphocyte count (/µl) | ≥300-<600 |
|
Neutrophil count (/µl) × 10 ×
platelet count (/µl)/lymphocyte count (/µl) | ≥600-<1,000 |
|
Neutrophil count (/µl) × 10 ×
platelet count (/µl)/lymphocyte count (/µl) | ≥1,000 |
| SIRI (27) |
|
|
Neutrophil count (/µl) ×
monocyte count (/µl)/lymphocyte count (/µl) | <500 |
|
Neutrophil count (/µl) ×
monocyte count (/µl)/lymphocyte count (/µl) | ≥500-<1,000 |
|
Neutrophil count (/µl) ×
monocyte count (/µl)/lymphocyte count (/µl) |
≥1,000-<2,000 |
|
Neutrophil count (/µl) ×
monocyte count (/µl)/lymphocyte count (/µl) | ≥2,000 |
| LIPI (28) |
|
| dNLR ≤3
and lactate dehydrogenase ≤245 (U/l) | 0 |
| dNLR
>3 or lactate dehydrogenase >245 (U/l) | 1 |
| dNLR
>3 and lactate dehydrogenase >245 (U/l) | 2 |
| CALLY (29) |
|
| Albumin
(g/dl) × lymphocyte count (/µl)/C-reactive protein (mg/dl) | <5 |
| Albumin
(g/dl) × lymphocyte count (/µl)/C-reactive protein (mg/dl) | ≥5 |
Statistical analysis
Patients with complete data available were
evaluated. The primary endpoint evaluated in the current study was
OS, defined as the time from the start date of initial palliative
chemotherapy to the date of death or final survival
confirmation.
Basic statistics (absolute and relative frequencies
for categorical variables; quartiles, maximum values, minimum
values, means, or medians for continuous variables) were obtained
to summarize the distribution of variables related to patient
background factors, complications, other prognostic factors, and
primary and secondary endpoints. Survival analyses were performed
using OS as the primary endpoint. The censored cases included
patients who were alive at the end date of the study or had dropped
out of the study for any reason.
Kaplan-Meier curves (univariate analyses) were
obtained for each inflammation-based prognostic markers associated
with OS, and the log-rank test was utilized to compare survival
curves. We compared the predictive quality of markers against OS
using the Cox regression analysis concordance (rate) and the Akaike
Information Criterion (AIC). Concordance was defined as follows.
Assuming that (si, yi) is a pair of observed survival time (y) and
scores (s), a pair of observations (i, j) was considered
‘concordant’ if (yi > yj, si < sj) or (yi < yj, si
>sj), as these conditions are symmetrical. Conversely, it was
considered ‘discordant’ if the conditions (yi < yj, si < sj)
or (yi > yj, si > sj) applied. If c, d, and ts are
counts of pairs that are concordant, discordant, or tied when using
score s, then concordance C is defined as C=(c +
ts/2)/(c + d + ts) using the proportion of
concordant pairs. Although the above definition of ‘concordant’ and
‘discordant’ pairs appears to be reversed, survival is inversely
correlated with the height of the hazard, and this definition has
validity.
All analyses were performed using R, version 4.2.2
(R Foundation for Statistical Computing, Vienna, Austria). All
statistical assessments were conducted as two-sided, and
significance was determined with a threshold of P<0.05. All
statistical analyses were two-sided, and P<0.05 was considered
to indicate a statistically significant difference.
Results
Patients' characteristics
Among the 846 patients initially identified for this
study, a total of 487 individuals were selected in the analysis due
to the availability of complete data. Table II displays the characteristics of
both the entire patient cohort and the subset included in the
analysis. The two populations showed similar characteristics.
 | Table II.Medical and demographic
characteristics of patients. |
Table II.
Medical and demographic
characteristics of patients.
|
| No. of
patients |
|---|
|
|
|
|---|
| Characteristic | All cases
(n=846) | Analyzed cases
(n=487) |
|---|
| Age, years |
|
|
| Median
(quantile) | 70 (36, 64, 70, 76,
90) | 71 (37, 65, 71, 76,
90) |
| ≥75, n
(%) | 266 (31.4) | 166 (34.1) |
| Sex, n (%) |
|
|
|
Male | 503 (59.5) | 279 (57.3) |
|
Female | 343 (40.5) | 208 (42.7) |
| PS, n (%) |
|
|
| 0 | 232 (27.4) | 124 (25.5) |
| 1 | 290 (34.3) | 188 (38.6) |
| 2 | 53 (6.3) | 36 (7.4) |
|
N/A | 271 (32.0) | 139 (28.5) |
| BMI,
kg/m2 |
|
|
| Median
(quantile) | 19.7 (11.2, 17.4,
19.7, 21.9, 35.4) | 19.7 (11.2, 17.4,
19.7, 21.9, 35.4) |
| Smoking
status, n (%) |
|
|
| Current
or former (BI >0) | 217 (25.7) | 125 (25.7) |
| Never
smoked (BI=0) | 562 (66.4) | 333 (68.4) |
|
N/A | 67 (7.9) | 29 (5.9) |
| Pathology, n
(%) |
|
|
|
Yes | 745 (88.1) | 435 (89.3) |
|
Adenocarcinoma | 418 (49.4) | 243 (49.9) |
|
Adenosquamous
carcinoma | 7 (0.8) | 4 (0.8) |
|
Carcinoma/malignant
neoplasm | 320 (37.8) | 188 (38.6) |
| No
(Radiological diagnosis only) | 101 (11.9) | 52 (10.7) |
| Primary disease
site, n (%) |
|
|
|
Pancreas head | 359 (42.5) | 199 (40.9) |
|
Pancreas body | 232 (27.4) | 140 (28.7) |
|
Pancreas tail | 220 (26.0) | 129 (26.5) |
| Not
evaluable | 35 (4.1) | 19 (3.9) |
| Previous
procedures, n (%) |
|
|
|
Surgery | 123 (14.5) | 47 (9.7) |
|
Endoscopic procedure | 44 (5.2) | 19 (3.9) |
|
Radiotherapy | 47 (5.6) | 25 (5.1) |
| Study period, n
(%) |
|
|
| Period
A (2010–2013) | 268 (31.7) | 135 (27.7) |
| Period
B (2014–2016) | 251 (29.6) | 159 (32.7) |
| Period
C (2017–2020) | 327 (38.7) | 193 (39.6) |
| Hospital scale, n
(%) |
|
|
| High
volume (n ≥50) | 509 (60.2) | 303 (62.2) |
| Low
volume (n <50) | 337 (39.8) | 184 (37.8) |
| Hospital type, n
(%) |
|
|
|
Government-designated cancer
hospital | 218 (25.7) | 137 (28.1) |
|
Prefectural designated cancer
hospital | 316 (37.4) | 181 (37.2) |
| General
hospital | 312 (36.9) | 169 (34.7) |
| First-line systemic
therapy, n (%) |
|
|
|
Gemcitabine monotherapy | 302 (35.7) | 167 (34.3) |
| S-1
monotherapy | 197 (23.3) | 102 (20.9) |
|
Gemcitabine plus S-1 | 66 (7.8) | 38 (7.8) |
|
Gemcitabine plus
nab-paclitaxel | 229 (27.1) | 146 (30.0) |
|
FOLFIRINOX | 52 (6.1) | 34 (7.0) |
Comparison of the ratios and
scores
Table III
summarizes the OS statistics by 17 inflammation-based prognostic
markers. All markers had statistically significant prognostic
value. In addition, when comparing ratios computed as continuous
variables with scores determined using categorical variables
employing specific cutoff values, it was evident that similar
hazard ratio (HR)s were observed. For instance, when we analyzed
the HRs and their corresponding 95% confidential intervals for NLR
(<3 compared to ≥3-<5 and ≥5) and NLS (0 compared to 1 and
2), the outcomes were as follows: 1.76 (1.35–2.29) and 2.67
(2.07–3.44) for NLR, and 1.60 (1.26–2.03) and 2.46 (1.75–3.47) for
NLS, respectively.
 | Table III.Overall survival summary statistics
for each score. |
Table III.
Overall survival summary statistics
for each score.
| Score/ratio | n (%) | Events | Median overall
survival (95% CI) | Hazard ratio (95%
CI) | P-value |
|---|
| PNI |
|
|
|
|
|
|
≥45 | 200 (41.1) | 139 | 7.4 (6.4–9.1) | Reference | - |
|
<45 | 287 (58.1) | 230 | 3.9 (3.0–5.1) | 1.82
(1.47–2.27) | <0.0001 |
| GPS |
|
|
|
|
|
| 0 | 184 (37.8) | 129 | 7.4 (6.6–9.1) | Reference | - |
| 1 | 157 (32.2) | 118 | 5.7 (4.1–7.9) | 1.33
(1.03–1.71) | 0.0286 |
| 2 | 146 (30.0) | 122 | 2.7 (1.8–4.6) | 2.40
(1.86–3.11) | <0.0001 |
| mGPS |
|
|
|
|
|
| 0 | 137 (28.1) | 91 | 8.2 (7.2–9.9) | Reference | - |
| 1 | 185 (38.0) | 143 | 6.0 (4.8–8.3) | 1.39
(1.07–1.81) | 0.0150 |
| 2 | 165 (33.9) | 135 | 2.9 (1.9–4.6) | 2.63
(2.00–3.45) | <0.0001 |
| CAR |
|
|
|
|
|
|
≤0.22 | 216 (46.4) | 147 | 8.3 (7.2–10.5) | Reference | - |
|
>0.22 | 271 (55.6) | 222 | 4.5 (3.2–5.5) | 2.06
(1.65–2.57) | <0.0001 |
| NLR |
|
|
|
|
|
|
<3 | 177 (24.0) | 119 | 8.3 (7.2–10.7) | Reference | - |
|
≥3-<5 | 152 (31.2) | 115 | 5.9 (4.8–7.4) | 1.76
(1.35–2.29) | <0.0001 |
| ≥5 | 158 (32.4) | 135 | 3.3 (2.8–4.6) | 2.67
(2.07–3.44) | <0.0001 |
| PLR |
|
|
|
|
|
|
≤150 | 203 (41.7) | 151 | 7.1 (5.6–8.7) | Reference | - |
|
>150 | 284 (58.3) | 218 | 4.8 (4.0–6.4) | 1.35
(1.09–1.67) | 0.0056 |
| LMR |
|
|
|
|
|
|
≥2.40 | 329 (67.6) | 237 | 7.1 (6.0–8.6) | Reference | - |
|
<2.40 | 158 (32.4) | 132 | 3.9 (2.8–5.1) | 1.83
(1.47–2.28) | <0.0001 |
| dNLR |
|
|
|
|
|
|
<3 | 326 (66.9) | 233 | 7.2 (6.0–8.7) | Reference | - |
|
≥3-<5 | 114 (23.4) | 94 | 3.9 (3.0–5.2) | 2.02
(1.58–2.58) | <0.0001 |
| ≥5 | 47 (9.7) | 42 | 2.2 (1.0–4.8) | 2.05
(1.46–2.88) | <0.0001 |
| NLS |
|
|
|
|
|
| 0 | 157 (32.2) | 112 | 8.3 (7.2–10.6) | Reference | - |
| 1 | 273 (56.1) | 204 | 5.0 (4.0–6.8) | 1.60
(1.26–2.03) | 0.00011 |
| 2 | 57 (11.7) | 53 | 3.4 (2.8–4.8) | 2.46
(1.75–3.47) | <0.0001 |
| PLS |
|
|
|
|
|
| 0 | 172 (35.3) | 125 | 7.4 (6.4–9.1) | Reference | - |
| 1 | 307 (63.0) | 238 | 4.8 (3.9–6.0) | 1.48
(1.18–1.84) | 0.00066 |
| 2 | 8 (1.6) | 6 | 4.7 (0.8-N/A) | 1.54
(0.64–3.69) | 0.33526 |
| LMS |
|
|
|
|
|
| 0 | 174 (35.7) | 125 | 7.9 (6.6–9.5) | Reference | - |
| 1 | 293 (60.2) | 224 | 4.8 (4.0–6.1) | 1.52
(1.21–1.90) | <0.0001 |
| 2 | 20 (4.1) | 20 | 1.6 (1.0–4.6) | 4.34
(2.64–7.15) | <0.0001 |
| NPS |
|
|
|
|
|
| 0 | 386 (79.3) | 286 | 6.5 (5.3–8.1) | Reference | - |
| 1 | 88 (18.1) | 72 | 4.1 (2.9–5.3) | 1.56
(1.18–2.05) | 0.00155 |
| 2 | 13 (2.7) | 11 | 3.7 (1.9-N/A) | 3.32
(1.78–6.18) | <0.0001 |
| PI |
|
|
|
|
|
| 0 | 235 (48.3) | 166 | 7.9 (6.9–9.6) | Reference | - |
| 1 | 195 (40.0) | 152 | 5.1 (4.1–6.6) | 1.58
(1.26–1.99) | <0.0001 |
| 2 | 57 (11.7) | 51 | 1.9 (1.6–3.9) | 3.35
(2.39–4.72) | <0.0001 |
| SII |
|
|
|
|
|
|
<300 | 40 (8.2) | 30 | 8.7 (7.2–13.0) | Reference | - |
|
≥300-<600 | 131 (26.9) | 94 | 8.0 (6.7–10.9) | 1.10
(0.72–1.66) | 0.6690 |
|
≥600-<1,000 | 121 (24.8) | 87 | 6.0 (4.8–8.7) | 1.76
(1.15–2.70) | 0.0089 |
|
≥1,000 | 195 (40.0) | 158 | 3.9 (3.0–4.9) | 2.50
(1.68–3.72) | <0.0001 |
| SIRI |
|
|
|
|
|
|
<500 | 39 (8.0) | 27 | 9.6 (8.6–15.5) | Reference | - |
|
≥500-<1,000 | 120 (24.6) | 84 | 7.2 (5.3–10.1) | 1.60
(1.03–2.50) | 0.0370 |
|
≥1,000-<2,000 | 140 (28.7) | 99 | 7.1 (5.1–8.7) | 1.96
(1.26–3.03) | 0.00266 |
|
≥2,000 | 188 (38.6) | 159 | 3.7 (2.9–4.9) | 3.37
(2.20–5.17) | <0.0001 |
| LIPI |
|
|
|
|
|
| 0 | 267 (54.8) | 184 | 7.9 (7.1–9.9) | Reference | - |
| 1 | 153 (31.4) | 126 | 4.2 (3.6–6.1) | 1.86
(1.47–2.34) | <0.0001 |
| 2 | 67 (13.8) | 59 | 2.2 (1.5–4.3) | 3.33
(2.45–4.53) | <0.0001 |
| CALLY |
|
|
|
|
|
|
<5 | 431 (88.5) | 332 | 5.1 (4.2–6.6) | Reference | - |
| ≥5 | 56 (11.5) | 37 | 10.3
(8.2–13.9) | 0.50
(0.35–0.70) | <0.0001 |
Kaplan-Meier curves
The Kaplan-Meier curves for OS by each
inflammation-based prognostic marker are shown in Fig. 1. Each marker alone showed a
significant correlation with prognosis. Additionally, for the
majority of markers, each increment in the numerical value was
linked to a gradual deterioration in prognosis.
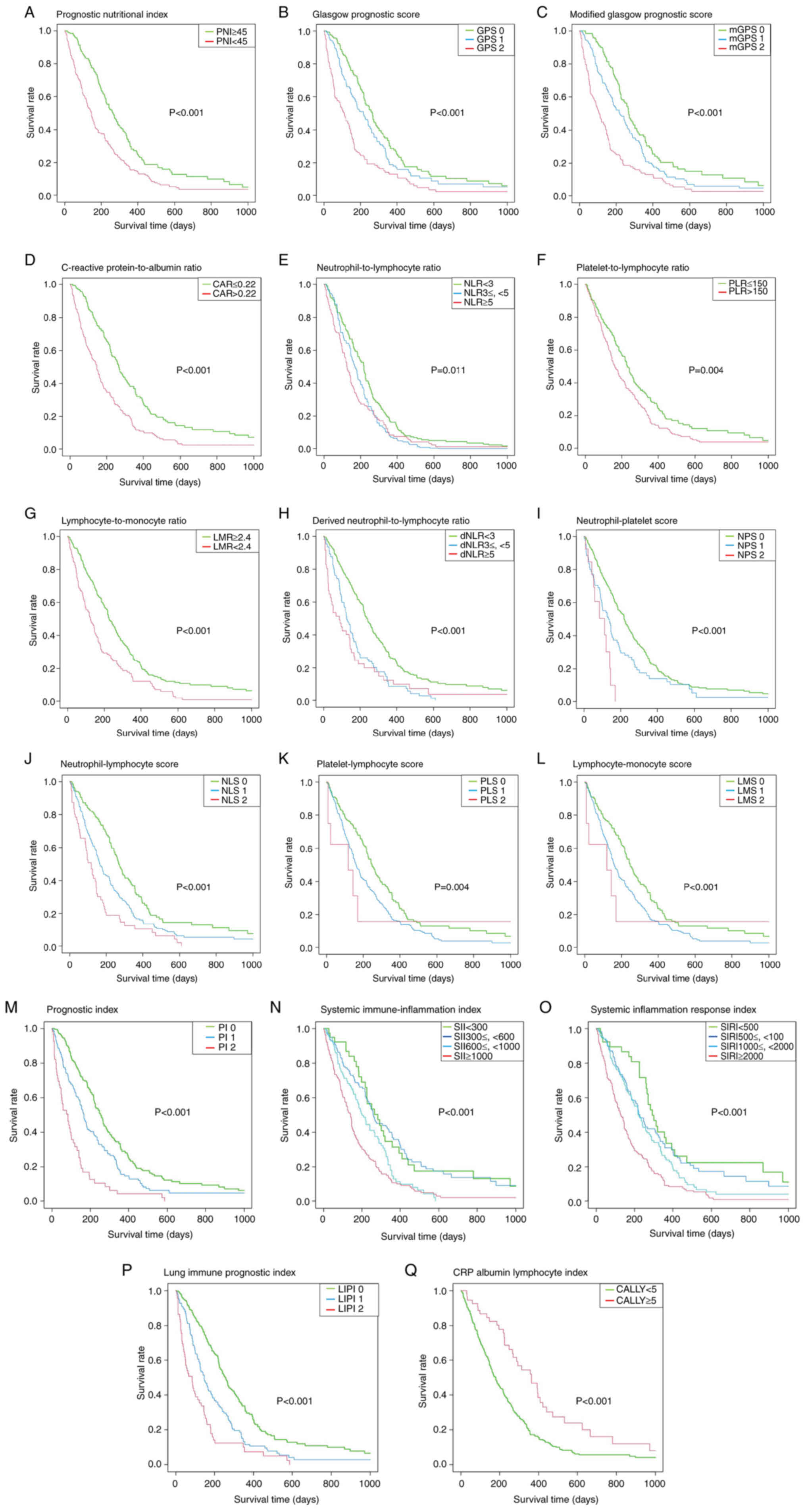 | Figure 1.Overall survival based on the
different scores/ratios. A log-rank test was used to calculate
significance. (A) Prognostic nutritional index, (B) Glasgow
prognostic score, (C) modified Glasgow prognostic score, (D)
C-reactive protein-to-albumin ratio, (E) neutrophil-to-lymphocyte
ratio, (F) platelet-to-lymphocyte ratio, (G) lymphocyte-to-monocyte
ratio, (H) derived neutrophil-to-lymphocyte ratio, (I)
neutrophil-platelet score, (J) neutrophil-lymphocyte score, (K)
platelet-lymphocyte score, (L) lymphocyte-monocyte score, (M)
prognostic index, (N) systemic immune-inflammation index, (O)
systemic inflammation response index, (P) lung immune prognostic
index and (Q) CRP albumin lymphocyte index. CRP, C-reactive
protein. |
Comparison of the inflammation-based
prognostic markers
The concordance rates and AICs calculated for the 17
inflammation-based prognostic markers are shown in Table IV. The concordance values ranged
from 0.616 to 0.679, and the AIC values ranged from 3784 to 3836.
Among them, the mGPS correlated best with OS, followed by GPS and
lung immune prognostic index (LIPI).
 | Table IV.Prognostic scores/ratios, and their
concordance rates and AIC for overall survival. |
Table IV.
Prognostic scores/ratios, and their
concordance rates and AIC for overall survival.
| Score | Concordance | AIC |
|---|
| mGPS | 0.679 | 3,796 |
| GPS | 0.672 | 3,802 |
| LIPI | 0.669 | 3,784 |
| PI | 0.666 | 3,799 |
| CAR | 0.666 | 3,801 |
| NLR | 0.665 | 3,788 |
| SIRI | 0.657 | 3,797 |
| SII | 0.653 | 3,801 |
| dNLR | 0.650 | 3,808 |
| PNI | 0.650 | 3,814 |
| LMR | 0.645 | 3,817 |
| NLS | 0.642 | 3,816 |
| LMS | 0.632 | 3,814 |
| NPS | 0.628 | 3,827 |
| CALLY | 0.626 | 3,825 |
| PLS | 0.618 | 3,834 |
| PLR | 0.616 | 3,836 |
Discussion
In this study, we compared inflammation-based
prognostic markers helpful in predicting prognosis in patients
undergoing chemotherapy for metastatic pancreatic cancer using
real-world data from the Tokushukai medical database. While
numerous prognostic and predictive markers have been reported, to
the best of our knowledge, this study represents the most
comprehensive comparison of inflammation-based prognostic markers
to date. All 17 markers we evaluated demonstrated significant
prognostic value, irrespective of whether they were ratio-based or
scored systems. Among them, the mGPS, GPS, and LIPI emerged as the
most accurate in predicting prognosis following first-line
treatment of metastatic pancreatic cancer.
GPS is probably the most widely used prognostic
score, with numerous reports supporting its usefulness. It was
defined and reported by Forrest et al (17) in 2003 as CRP and albumin levels in
patients with unresectable non-small cell lung cancer. According to
multivariate analyses, the combined score of CRP and albumin was
identified as an independent prognostic factor, and a validation
study was subsequently reported in 2004 (30). The validity of the GPS has been
reported in the Glasgow Inflammation Outcome Study (18,19),
and the GPS is now widely used as an inflammation-based prognostic
marker (11). In addition, several
modifications of the GPS with adjusted cutoff values have been
reported with improved accuracy. In studies of patients with
colorectal cancer, low albumin levels did not correlate with poor
prognosis because few patients have low albumin levels without
elevated CRP levels. Thus, low albumin level alone looked less
associated with poor prognosis (31), and a modified GPS that partially
excludes the albumin level has been suggested (11). In a study examining the correlation
between the mGPS and prognosis in Japanese patients with colorectal
cancer, the best cutoff value of 0.5 mg/dl was reported for CRP
based on its receiver operating characteristic curve (32).
LIPI, another recently reported score, is based on a
combination of dNLR and LDH scores. The prognostic correlation of
LIPI was reported by Mezquita et al in 2018 in patients
treated with immune checkpoint inhibitors for advanced non-small
cell lung cancer (28). LIPI has
been developed as a prognostic score because dNLR and LDH level
were independent prognostic factors in two large retrospective
studies of patients with metastatic melanoma treated with
ipilimumab (33) or pembrolizumab
(34). The combination of these two
factors has been reported to be of prognostic value in patients
treated with immune checkpoint inhibitors for lung cancer (35,36),
urothelial carcinoma (37), or
solid tumors in general (38).
Although the LIPI score was developed for patients treated with
immune checkpoint inhibitors, the present study shows it is also a
useful prognostic indicator in metastatic pancreatic cancer. The
literature examining the usefulness of LIPI is still limited, and
further studies are needed to determine whether it is reproducible
in other cancers.
This study has a few limitations. First, due to the
retrospective design, a number of patients had deficiencies in
blood tests such as CRP, albumin, and LDH, which are not essential
for chemotherapy induction. Accordingly, fewer patients qualified
for the complete analysis. Second, new inflammation-based
prognostic markers are introduced each year, and not all of them
are included in this study. Third, this study included only
Japanese subjects. Hence, its external validity may be limited in
non-Asian populations. However, a nationwide study conducted in the
Netherlands on metastatic pancreatic cancer identified CA19-9,
albumin, CRP, LDH, C-reactive protein-to-albumin ratio, and
GPS/mGPS as easily measurable prognostic biomarkers (39). Additionally, a systematic review
from 2013 confirmed the prognostic potential of GPS/mGPS and NLR
(40). Hence, it is safe to assume
that at least GPS/mGPS are applicable prognostic biomarkers for
both Asian and non-Asian patients with metastatic pancreatic
cancer.
Lastly, additional parameters such as hemoglobin,
CEA, and CA19-9 may also hold prognostic significance. Despite
these limitations, the robustness of this study lies in the
incorporation of a sizable cohort and the simultaneous assessment
of real-world data for numerous inflammation-based prognostic
markers. Our future research will evaluate each laboratory
parameter and develop a novel prognostic score.
In conclusion, our real-world data analysis
demonstrated that 17 inflammation-based markers that previously
reported held significant prognostic value for patients with
metastatic pancreatic cancer. Among these markers, the mGPS
exhibited the highest accuracy.
Acknowledgements
The authors would like to thank Dr Shinnichi
Higashiue (chair of the Medical Corporation Tokushukai, Tokyo,
Japan and General Incorporated Association Tokushukai, Tokyo,
Japan) and Dr Hisaaki Afuso (chief advisor of the Medical
Corporation Tokushukai, Tokyo, Japan) for their support in
conducting clinical research within the Tokushukai Group, and Mr.
Katsuhiko Ozaki (President of Tokushukai Information System, Inc.,
Osaka, Japan) for their assistance in using the medical
database.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
RS, YI and MO made substantial contributions to the
study design and conception. RS, YF, MS and MH were responsible for
data acquisition. RS and YI interpreted the data and drafted the
manuscript. KU, TM, KO, NS and HM provided advice on research
design and aided in the critical interpretation of this research
for critical content. RS and YI confirm the authenticity of all the
raw data. NS and HM comprehensively reviewed and approved the final
version of this manuscript. All authors have read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
The project adhered to the ethical guidelines for
medical and biological research involving human subjects in Japan
and followed the principles of The Declaration of Helsinki.
Approval for the study was obtained from the Ethics Committee of
the Tokushukai Group in April 2020 (approval no. TGE01427-024), and
the study was registered in the UMIN Clinical Trial Registry under
the number UMIN000050590. Patients were provided with information
using opt-out methods and no patient declared not to
participate.
Patient consent for publication
Patient consent for publication was obtained through
opt-out methods.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
AIC
|
akaike information criterion
|
|
BMI
|
body mass index
|
|
CALLY
|
CRP albumin lymphocyte index
|
|
CAR
|
CRP-to-albumin ratio
|
|
CIs
|
confidence intervals
|
|
dNLR
|
derived neutrophil-to-lymphocyte
ratio
|
|
FOLFIRINOX
|
fluorouracil, folic acid, oxaliplatin
and irinotecan
|
|
GPS
|
Glasgow prognostic score
|
|
HRs
|
hazard ratios
|
|
LIPI
|
lung immune prognostic index
|
|
LMR
|
lymphocyte-to-monocyte ratio
|
|
LMS
|
lymphocyte-monocyte score
|
|
mGPS
|
modified Glasgow prognostic score
|
|
NLR
|
neutrophil-to-lymphocyte ratio
|
|
NLS
|
neutrophil-lymphocyte score
|
|
NPS
|
neutrophil-platelet score
|
|
OS
|
overall survival
|
|
PI
|
prognostic index
|
|
PLR
|
platelet-to-lymphocyte ratio
|
|
PLS
|
platelet-lymphocyte score
|
|
PNI
|
prognostic nutritional index
|
|
S-1
|
tegafur/gimeracil/oteracil
|
|
SII
|
systemic immune-inflammation index
|
|
SIRI
|
systemic inflammation response
index
|
|
TREAD
|
tokushukai real-world data
|
|
UMIN
|
university hospital medical
information network
|
References
|
1
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer statistics, 2021. CA Cancer J Clin. 71:7–33. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
SEER 5-year relative survival rate,
2012–2018, . Cancer Statistics Explorer Network. National Cancer
Institute; United States: https://seer.cancer.gov/statistics-network/explorer/application.htmlApril
1–2023
|
|
3
|
Cancer Information Service, National
Cancer Center, Japan and National Cancer Registry (In Japanese)
(Ministry of Health, Labour and Welfare), . https://ganjoho.jp/reg_stat/statistics/data/dl/en.htmlApril
1–2023
|
|
4
|
Conroy T, Desseigne F, Ychou M, Bouché O,
Guimbaud R, Bécouarn Y, Adenis A, Raoul JL, Gourgou-Bourgade S, de
la Fouchardière C, et al: FOLFIRINOX versus gemcitabine for
metastatic pancreatic cancer. N Engl J Med. 364:1817–1825. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Okusaka T, Ikeda M, Fukutomi A, Ioka T,
Furuse J, Ohkawa S, Isayama H and Boku N: Phase II study of
FOLFIRINOX for chemotherapy-naïve Japanese patients with metastatic
pancreatic cancer. Cancer Sci. 105:1321–1326. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ozaka M, Ishii H, Sato T, Ueno M, Ikeda M,
Uesugi K, Sata N, Miyashita K, Mizuno N, Tsuji K, et al: A phase II
study of modified FOLFIRINOX for chemotherapy-naïve patients with
metastatic pancreatic cancer. Cancer Chemother Pharmacol.
81:1017–1023. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Von Hoff DD, Ervin T, Arena FP, Chiorean
EG, Infante J, Moore M, Seay T, Tjulandin SA, Ma WW, Saleh MN, et
al: Increased survival in pancreatic cancer with nab-paclitaxel
plus gemcitabine. N Engl J Med. 369:1691–1703. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shimoyama R, Imamura Y, Uryu K, Mase T,
Fujimura Y, Hayashi M, Ohtaki M, Ohtani K, Shinozaki N and Minami
H: Real-world treatment outcomes among patients with metastatic
pancreatic cancer in Japan: The Tokushukai real-world data project.
Mol Clin Oncol. 19:982023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
McMillan DC: An inflammation-based
prognostic score and its role in the nutrition-based management of
patients with cancer. Proc Nutr Soc. 67:257–262. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Buzby GP, Mullen JL, Matthews DC, Hobbs C
and Rosato EF: Prognostic nutritional index in gastrointestinal
surgery. Am J Surg. 139:160–167. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
McMillan DC: Systemic inflammation,
nutritional status and survival in patients with cancer. Curr Opin
Clin Nutr Metab Care. 12:223–226. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dolan RD, McSorley ST, Horgan PG, Laird B
and McMillan DC: The role of the systemic inflammatory response in
predicting outcomes in patients with advanced inoperable cancer:
Systematic review and meta-analysis. Crit Rev Oncol Hematol.
116:134–146. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shimoyama R, Imamura Y, Uryu K, Mase T,
Fujimura Y, Hayashi M, Ohtaki M, Ohtani K, Shinozaki N and Minami
H: Real-world outcomes of systemic therapy in Japanese patients
with cancer (Tokushukai REAl-world data project: TREAD): Study
protocol for a nationwide cohort study. Healthcare (Basel).
10:21462022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Eba J and Nakamura K: Overview of the
ethical guidelines for medical and biological research involving
human subjects in Japan. Jpn J Clin Oncol. 52:539–544. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
National Cancer Registry (Ministry of
Health, Labour and Welfare), tabulated by Cancer Information
Service, National Cancer Center, Japan, . https://ganjoho.jp/reg_stat/statistics/data/dl/en.htmlApril
1–2023
|
|
16
|
Onodera T, Goseki N and Kosaki G:
Prognostic nutritional index in gastrointestinal surgery of
malnourished cancer patients. Nihon Geka Gakkai Zasshi.
85:1001–1005. 1984.(In Japanese). PubMed/NCBI
|
|
17
|
Forrest LM, McMillan DC, McArdle CS,
Angerson WJ and Dunlop DJ: Evaluation of cumulative prognostic
scores based on the systemic inflammatory response in patients with
inoperable non-small-cell lung cancer. Br J Cancer. 89:1028–1030.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fairclough E, Cairns E, Hamilton J and
Kelly C: Evaluation of a modified early warning system for acute
medical admissions and comparison with C-reactive protein/albumin
ratio as a predictor of patient outcome. Clin Med (Lond). 9:30–33.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nakahara K, Monden Y, Ohno K, Fujii Y,
Hashimoto J, Kitagawa Y and Kawashima Y: Importance of biologic
status to the postoperative prognosis of patients with stage III
nonsmall cell lung cancer. J Surg Oncol. 36:155–160. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Smith RA, Bosonnet L, Raraty M, Sutton R,
Neoptolemos JP, Campbell F and Ghaneh P: Preoperative
platelet-lymphocyte ratio is an independent significant prognostic
marker in resected pancreatic ductal adenocarcinoma. Am J Surg.
197:466–472. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wilcox RA, Ristow K, Habermann TM, Inwards
DJ, Micallef IN, Johnston PB, Colgan JP, Nowakowski GS, Ansell SM,
Witzig TE, et al: The absolute monocyte and lymphocyte prognostic
score predicts survival and identifies high-risk patients in
diffuse large-B-cell lymphoma. Leukemia. 25:1502–1509. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Proctor MJ, McMillan DC, Morrison DS,
Fletcher CD, Horgan PG and Clarke SJ: A derived neutrophil to
lymphocyte ratio predicts survival in patients with cancer. Br J
Cancer. 107:695–699. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Watt DG, Proctor MJ, Park JH, Horgan PG
and McMillan DC: The neutrophil-platelet score (NPS) predicts
survival in primary operable colorectal cancer and a variety of
common cancers. PLoS One. 10:e01421592015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Dolan RD, McSorley ST, Park JH, Watt DG,
Roxburgh CS, Horgan PG and McMillan DC: The prognostic value of
systemic inflammation in patients undergoing surgery for colon
cancer: comparison of composite ratios and cumulative scores. Br J
Cancer. 119:40–51. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dolan RD, Alwahid M, McSorley ST, Park JH,
Stevenson RP, Roxburgh CS, Horgan PG and McMillan DC: A comparison
of the prognostic value of composite ratios and cumulative scores
in patients with operable rectal cancer. Sci Rep. 10:179652020.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kasymjanova G, MacDonald N, Agulnik JS,
Cohen V, Pepe C, Kreisman H, Sharma R and Small D: The predictive
value of pre-treatment inflammatory markers in advanced
non-small-cell lung cancer. Curr Oncol. 17:52–58. 2010.PubMed/NCBI
|
|
27
|
Qi Q, Zhuang L, Shen Y, Geng Y, Yu S, Chen
H, Liu L, Meng Z, Wang P and Chen Z: A novel systemic inflammation
response index (SIRI) for predicting the survival of patients with
pancreatic cancer after chemotherapy. Cancer. 122:2158–2167. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mezquita L, Auclin E, Ferrara R, Charrier
M, Remon J, Planchard D, Ponce S, Ares LP, Leroy L,
Audigier-Valette C, et al: Association of the lung immune
prognostic index with immune checkpoint inhibitor outcomes in
patients with advanced non-small cell lung cancer. JAMA Oncol.
4:351–357. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Müller L, Hahn F, Mähringer-Kunz A, Stoehr
F, Gairing SJ, Michel M, Foerster F, Weinmann A, Galle PR, Mittler
J, et al: Immunonutritive scoring for patients with hepatocellular
carcinoma undergoing transarterial chemoembolization: Evaluation of
the CALLY index. Cancers (Basel). 13:50182021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Forrest LM, McMillan DC, McArdle CS,
Angerson WJ and Dunlop DJ: Comparison of an inflammation-based
prognostic score (GPS) with performance status (ECOG) in patients
receiving platinum-based chemotherapy for inoperable non-small-cell
lung cancer. Br J Cancer. 90:1704–1706. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
McMillan DC, Crozier JEM, Canna K,
Angerson WJ and McArdle CS: Evaluation of an inflammation-based
prognostic score (GPS) in patients undergoing resection for colon
and rectal cancer. Int J Colorectal Dis. 22:881–886. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Toiyama Y, Miki C, Inoue Y, Tanaka K,
Mohri Y and Kusunoki M: Evaluation of an inflammation-based
prognostic score for the identification of patients requiring
postoperative adjuvant chemotherapy for stage II colorectal cancer.
Exp Ther Med. 2:95–101. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ferrucci PF, Ascierto PA, Pigozzo J, Del
Vecchio M, Maio M, Antonini Cappellini GC, Guidoboni M, Queirolo P,
Savoia P, Mandalà M, et al: Baseline neutrophils and derived
neutrophil-to-lymphocyte ratio: Prognostic relevance in metastatic
melanoma patients receiving ipilimumab. Ann Oncol. 27:732–738.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Weide B, Martens A, Hassel JC, Berking C,
Postow MA, Bisschop K, Simeone E, Mangana J, Schilling B, Di
Giacomo AM, et al: Baseline biomarkers for outcome of melanoma
patients treated with pembrolizumab. Clin Cancer Res. 22:5487–5496.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Huang L, Han H, Zhou L, Chen X, Xu Q, Xie
J, Zhan P, Chen S, Lv T and Song Y: Evaluation of the lung immune
prognostic index in non-small cell lung cancer patients treated
with systemic therapy: A retrospective study and meta-analysis.
Front Oncol. 11:6702302021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang Q, Gong X, Sun L, Miao L and Zhou Y:
The predictive value of pretreatment lactate dehydrogenase and
derived neutrophil-to-lymphocyte ratio in advanced non-small cell
lung cancer patients treated with PD-1/PD-L1 inhibitors: A
meta-analysis. Front Oncol. 12:7914962022. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Banna GL, Di Quattro R, Malatino L,
Fornarini G, Addeo A, Maruzzo M, Urzia V, Rundo F, Lipari H, De
Giorgi U and Basso U: Neutrophil-to-lymphocyte ratio and lactate
dehydrogenase as biomarkers for urothelial cancer treated with
immunotherapy. Clin Transl Oncol. 22:2130–2135. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu H, Yang XL, Yang XY, Dong ZR, Chen ZQ,
Hong JG and Li T: The prediction potential of the pretreatment lung
immune prognostic index for the therapeutic outcomes of immune
checkpoint inhibitors in patients with solid cancer: A systematic
review and meta-analysis. Front Oncol. 11:6910022021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Strijker M, van Veldhuisen E, van der
Geest LG, Busch OR, Bijlsma MF, Mohammad NH, Homs MY, van Hooft JE,
Verheij J, de Vos-Geelen J, et al: Readily available biomarkers
predict poor survival in metastatic pancreatic cancer. Biomarkers.
26:325–334. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ahmad J, Grimes N, Farid S and
Morris-Stiff G: Inflammatory response related scoring systems in
assessing the prognosis of patients with pancreatic ductal
adenocarcinoma: A systematic review. Hepatobiliary Pancreat Dis
Int. 13:474–481. 2014. View Article : Google Scholar : PubMed/NCBI
|















