Introduction
Breast cancer (BC) is the most prevalent type of
cancer and the second highest cause of cancer mortality in women
worldwide with an estimated 2.3 million new cases and >685,000
deaths reported in 2020 (1).
Invasive lobular carcinoma (ILC) is the second most common
histologic subtype of breast cancer worldwide, accounting for
10–15% of all breast cancer cases (2,3). Over
the past two decades, the incidence of ILC has increased, likely
due to improvements in diagnostic techniques in breast imaging
(4). ILC is a distinct entity with
unique clinical and molecular features, including a diffuse,
multifocal, and poorly circumscribed growth pattern, and is
associated with difficult and delayed early diagnosis, inherent
resistance to conventional therapies and a risk of late recurrence,
which all present significant challenges in management (5). As with other breast cancers, the
treatment of early lobular BC involves conservative surgery or
‘lumpectomy’ followed by adjuvant radiation. However, due to the
multifocality and diffuse growth pattern, the rate of positive
margins and thus second revision surgeries is higher in invasive
lobular carcinomas than in ductal carcinomas (6).
The purpose of radiation therapy in the adjuvant
setting is to sterilize subclinical localized residual disease that
persists after surgical resection but is undetectable by clinical
or radiographic assays. Whole breast irradiation decreases the risk
of ipsilateral breast cancer recurrence by ~50% (7–10) in
both invasive ductal carcinoma and ILC. Moreover, partial breast
irradiation of ILC is associated with higher ipsilateral breast
cancer recurrence rates and is relatively contraindicated in ILC
(11). Nevertheless, the majority
of women with ILC treated with breast conserving surgery do not
develop in-breast local recurrence, which suggests that the use of
whole breast irradiation is overtreating these women. There is a
clear clinical need for more effective biomarkers to predict the
benefits of radiation for patients with cancer, including women
with breast cancer.
The measurement of circulating tumor DNA (ctDNA) has
emerged as a biomarker with wide-ranging applications in cancer
management (12). ctDNA levels have
both prognostic and predictive value in many cancers including
breast cancer. Moreover, dynamic changes in ctDNA levels, including
ctDNA clearance have been reported to be associated with response
to various systemic therapies, such as PI3K inhibitors. The power
of ctDNA detection derives from its role in the signaling of
clinically occult metastatic disease or minimal residual disease
(MRD). There are many approaches to measure ctDNA in the plasma,
with the most sensitive approaches involving the sequencing of
tumors followed by the development of tumor-specific assays
personalized to the patient's tumor. These ‘tumor-bespoke’ assays
use either sequencing or PCR reactions to identify the individual
mutations in a patient's tumor (13,14).
The Signatera™ assay uses tumor whole-exome sequencing
to identify 16 patient-specific somatic single-nucleotide variants
and short indels to generate multiplex PCR primer pairs. These
pairs are used to amplify universal libraries, which are then
sequenced on a NGS sequencing platform (HiSeq 2500 system;
Illumina, Inc.) (15). The present
study reports a case of invasive lobular carcinoma, in which ctDNA
clearance suggested response to local breast radiation therapy.
Case report
In January 2021, a 46-year-old pre-menopausal
female, with a family history of breast cancer, presented with a
palpable 1 cm (length) mass in her left breast with no palpable
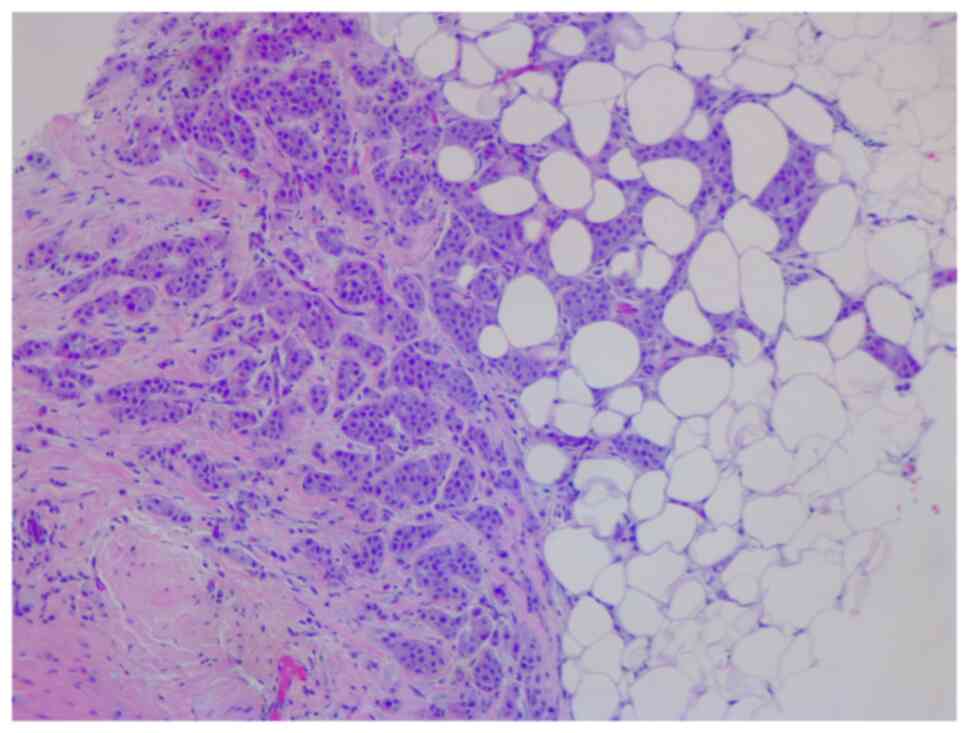
lymphadenopathy. The breast biopsy showed an invasive lobular
carcinoma breast cancer (Fig. 1),
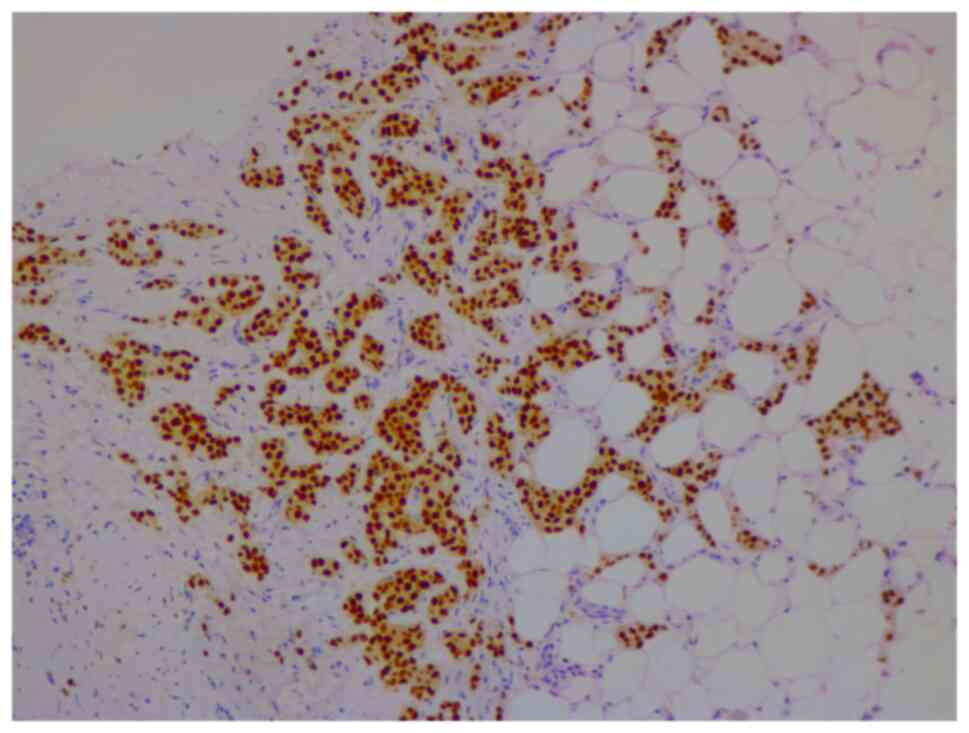
Nottingham grade 2/3, estrogen (98%; Fig. 2) and progesterone (45%) receptor
positive (Fig. 3), Her2 score 2+
based on the immunohistochemistry (according to ASCO-CAP clinical
laboratory guidelines) (16),
fluorescence in-situ hybridisation not amplified. Pre-operative
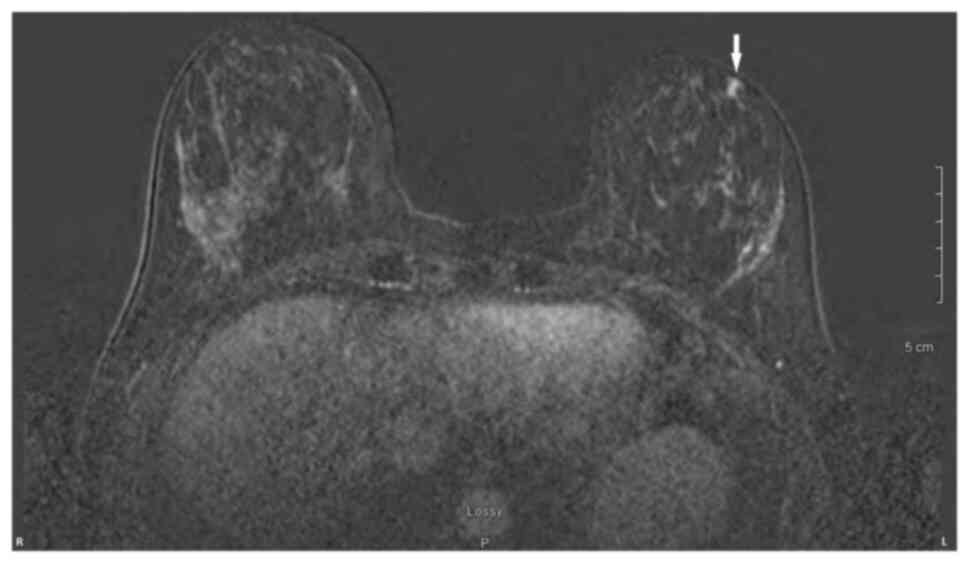
breast MRI did not reveal any other suspicious focus of enhancement
in the left breast but did reveal a 4×3×4 mm non-mass enhancement
in the right breast, classified as a BI-RADS 4B lesion (17). A breast Magnetic resonance imaging
(MRI) guided biopsy was done, which showed a lobular carcinoma in
situ, Nottingham grade 2 (Fig. 4).
In April 2021, the patient underwent 2 partial mastectomies, one on
each breast, localized with radioactive seeds and sentinel lymph
node biopsy in the left axilla. Pathological examination of the
resection sample showed a 1 cm (length) grade 2 invasive lobular
carcinoma, without lymphovascular invasion, with lobular carcinoma
in-situ, negative margins and 0/4 sentinel lymph nodes on the left
side (pT1pN0; AJCC-Breast Cancer Staging; 8th edition) (18). The right breast sample showed only
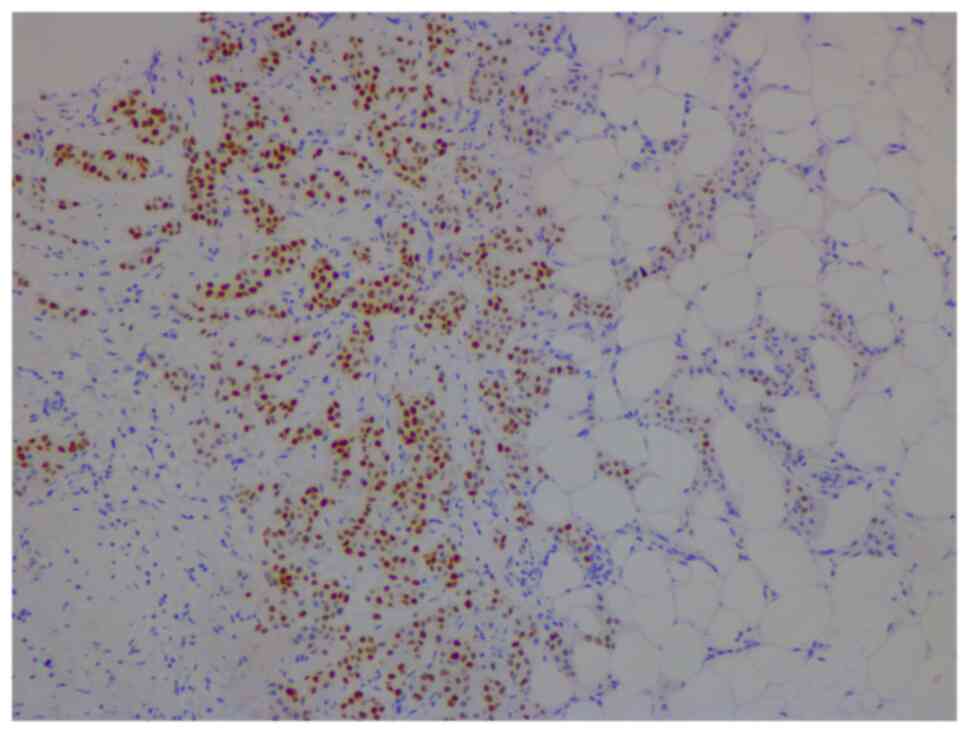
lobular carcinoma in-situ with negative margins. The Ki67 staining
of the invasive lobular carcinoma was 7–8% (Fig. 5), but the Oncotype DX®
(Exact Sciences, Inc.) Breast Recurrence Score was 24. A germline
test was performed on the blood samples of patients, which included
DNA extraction, PCR amplification, sequencing, bioinformatics
analysis and variant interpretation. The test revealed no
clinically relevant pathogenic sequence variants in the BRCA1,
BRCA2 and PALB2 genes. After tumor board discussion, based on the
Oncotype Dx result, the hormonal status and age of the patient,
adjuvant chemotherapy was offered but the patient refused.
Concurrently, a personalized, tumor-informed multiplex-PCR
(m-PCR)-NGS assay (Signatera™) was performed. The first
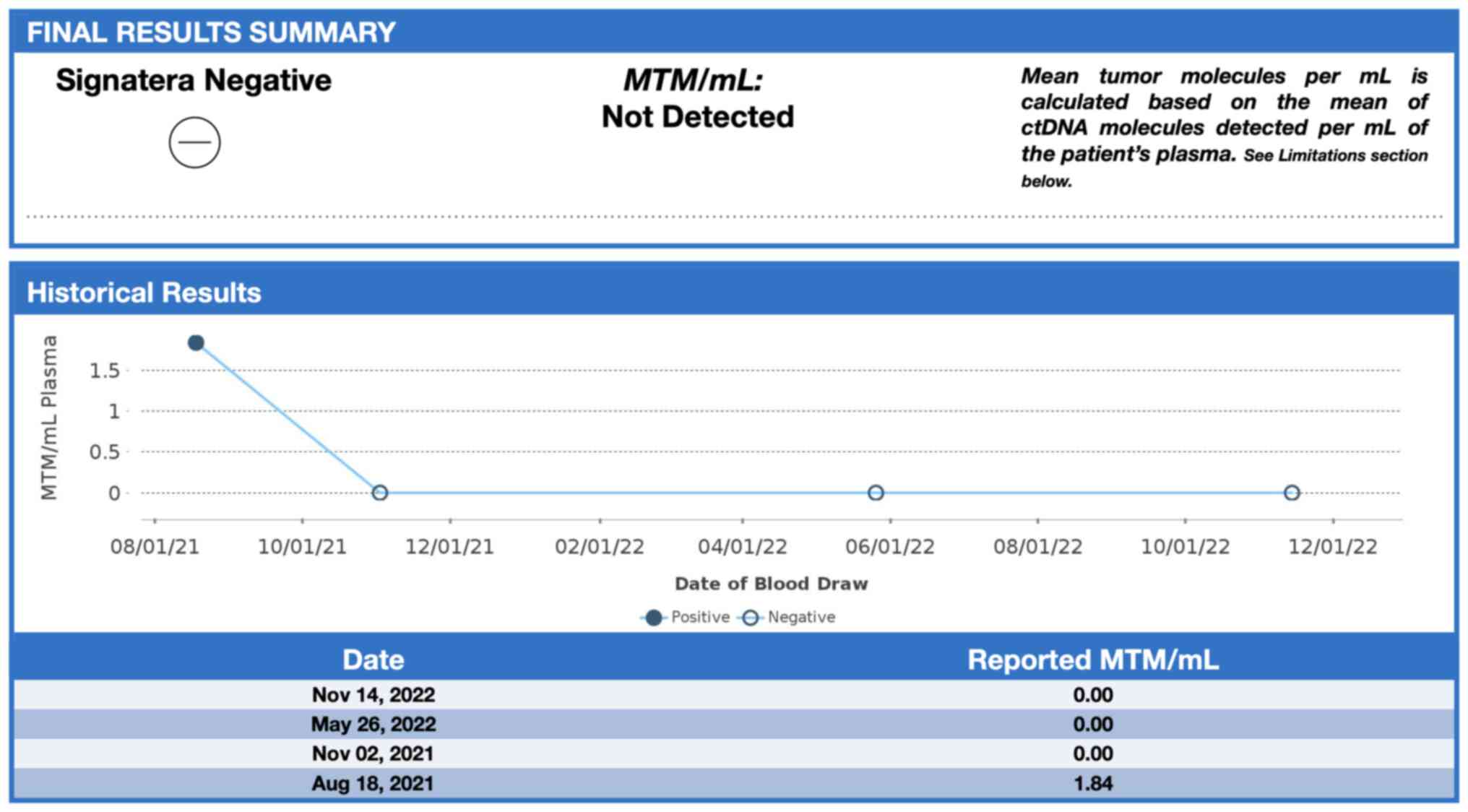
sample was collected in August 2021 and the result was reported as
a positive test (1.85 MTM/ml) (Fig.
6). Adjuvant therapy was again offered but the patient refused
both chemotherapy and endocrine therapy (Tamoxifen) +/- ovarian
suppression. Over 28 days in September and October 2021, the
patient received adjuvant radiation therapy: A total of 42.4 Gy in
16 fractions was delivered to the whole left breast, followed by a
boost to the surgical bed of 10 Gy in 4 fractions. After radiation
therapy, 3 ctDNA analyses were performed in November 2021 and, May
and November 2022 and all were reported as undetectable (0.00
MTM/ml) (Fig. 6). Tumor markers
(CA15-3 and CA-125) were assessed every 6 months and were always
negative. Computed tomography of the head was performed in November
2021 due to new symptoms (postural dizziness) and was negative for
metastatic disease. The patient was followed up by the breast
surgeon every 6 months, with her last visit was in November 2022
during which the physical exam was negative for local recurrence.
The patient's last breast imaging was performed in March 2023 and
both ultrasound and mammogram showed BI-RADS2 with benign findings.
At the time of this report, the patient remains with no signs of
local or distant recurrence of disease and excellent performance
status and.
Clinical testing
In order to predict an early relapse after adjuvant
treatment, the patient consented to periodic ctDNA monitoring in
her blood. She was first tested 3 months after surgery and 1, 7 and
13 months after radiation therapy using the Signatera™ test, in
addition to a physical exam every six months and a mammogram once a
year.
Discussion
Breast cancer is a heterogeneous disease and ctDNA
can accurately reflect this heterogeneity, supporting the
detection, monitoring and understanding of the evolution of the
disease (19). Previous studies
have reported the value of ctDNA in detecting MRD, monitoring
treatment response or resistance and predicting early relapse
(20,21). Nevertheless, studies investigating
the clinical utility of serial ctDNA monitoring for treatment
guidance are few (22,23). It is important to note that the
sensitivity and specificity of these assays varies depending on the
method used to measure ctDNA in plasma. Many ctDNA assays use
pre-defined recurrently mutated gene panels, which, for the purpose
of tracking MRD have the limitation of missing mutations or
alterations not included in or tested by the panel. Tumour
‘bespoke’ assays use the primary tumour to screen and select
specific mutation variants belonging to the tumour. These highly
sensitive and specific personalized assays are best used to detect
and track MRD (24). These tumor
bespoke assays provide accurate prognostic information related to
distant disease recurrence, as evidence of micrometastatic disease.
However, they appear to be less accurate in predicting local
response to therapy (25). This is
apparent in studies of ctDNA in the context of neoadjuvant
treatment for breast cancer. For instance, in the large I-SPY study
reported that early clearance of ctDNA was associated with
pathological complete response (pCR) in the breast only in patients
with triple negative breast cancer (26). Cavallone et al (27) reported that early clearance of ctDNA
had a relatively low predictive value of 50% for pCR. These same
assays are highly predictive of distant relapse free survival. In
previous studies, all recurrences reported in patients with
positive tumor bespoke assays were distant recurrences (21,28,29).
In the present study, the detection and
quantification of breast cancer-derived ctDNA using a personalized
tumor bespoke assay is reported. In this patient, with a small
invasive lobular carcinoma in a background of lobular carcinoma in
situ (LCIS), the commercial tumor bespoke Signatera™ assay, was
positive for the detection of ctDNA 17 weeks after surgery but
prior to local radiotherapy treatment. The ctDNA was cleared after
radiation therapy to the breast, which suggested that radiation
therapy eliminated residual breast cancer cells, a possibility
which is supported by multiple randomized clinical trials (30,31).
Pre-operative breast imaging including MRI was unable to detect
suspicious breast disease beyond the target cancer. To the best of
our knowledge, there have been no previous reports of the clearance
of ctDNA due to local radiation therapy for early breast cancer.
Several follow up ctDNA tests did not reveal any further ctDNA up
to 22 months of follow up. As the patient had LCIS in her breast,
it is possible that there were multiple other foci of lobular
cancer in her diseased breast, invisible to imaging. These foci may
have accounted for the presence of ctDNA in her blood after
surgery. Radiotherapy appears to be effective in eliminating this
disease, based on the lack of local recurrence during almost 2
years of follow up.
As the present study is limited to a single patient,
this alone cannot establish it as a disease-monitoring tool.
However, this case demonstrated that bespoke tumor assays have the
capacity to detect local subclinical breast cancer. These results
provide a rationale for larger prospective studies to establish if
ctDNA analysis could be considered an effective decision-making
tool in the management of radiation therapy decisions in
early-stage breast cancer. In a future study, ctDNA levels should
evaluated before and after treatment, including locoregional
treatments, if necessary. During the adjuvant period, ctDNA should
be compared with traditional blood markers such as CA15.3, CEA and
CA125 in order to detect recurrences.
Acknowledgements
Not applicable.
Funding
Funding: Not applicable.
Availability of data and materials
The data generated in the present study are not
publicly available due to proprietary information but may be
requested from the corresponding author. Sequencing data are not
available as the Signatera™ test is a commercially available
clinical test, with these data not being provided by the
company.
Authors' contributions
Conceptualization was performed by MPC, PF and MB.
Data acquisition and curation was performed by MPC, PF and MB. PF
was involved in discussions about the treatments of the patient.
Data analysis and interpretation were performed by MPC and MB. The
manuscript was written by MPC and MB. All authors read and approved
the final manuscript. MB and MP confirm the authenticity of all the
raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for publication of the details of her medical case.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ctDNA
|
circulating tumor DNA
|
|
BC
|
breast cancer
|
|
ILC
|
invasive lobular carcinoma
|
|
MRD
|
minimal residual disease
|
|
MRI
|
magnetic resonance imaging
|
|
pCR
|
pathological complete response
|
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mouabbi JA, Hassan A, Lim B, Hortobagyi
GN, Tripathy D and Layman RM: Invasive lobular carcinoma: An
understudied emergent subtype of breast cancer. Breast Cancer Res
Treat. 193:253–264. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Thomas M, Kelly ED, Abraham J and Kruse M:
Invasive lobular breast cancer: A review of pathogenesis,
diagnosis, management, and future directions of early stage
disease. Semin Oncol. 46:121–132. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wilson N, Ironside A, Diana A and
Oikonomidou O: Lobular breast cancer: A review. Front Oncol.
10:5913992021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pramod N, Nigam A, Basree M, Mawalkar R,
Mehra S, Shinde N, Tozbikian G, Williams N, Majumder S and
Ramaswamy B: Comprehensive review of molecular mechanisms and
clinical features of invasive lobular cancer. Oncologist.
26:e943–e953. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Biglia N, Maggiorotto F, Liberale V,
Bounous VE, Sgro LG, Pecchio S, D'Alonzo M and Ponzone R:
Clinical-pathologic features, long term-outcome and surgical
treatment in a large series of patients with invasive lobular
carcinoma (ILC) and invasive ductal carcinoma (IDC). Eur J Surg
Oncol. 39:455–460. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Choi YJ, Shin YD and Song YJ: Comparison
of ipsilateral breast tumor recurrence after breast-conserving
surgery between ductal carcinoma in situ and invasive breast
cancer. World J Surg Oncol. 14:1262016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Giannakeas V, Sopik V and Narod SA:
Association of radiotherapy with survival in women treated for
ductal carcinoma in situ with lumpectomy or mastectomy. JAMA Netw
Open. 1:e1811002018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Boyages J and Baker L: Evolution of
radiotherapy techniques in breast conservation treatment. Gland
Surg. 7:5762018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tsai CJ, Huang HY, Chen FM, Yang YH, Chen
LC and Hsieh KP: Investigating the effectiveness of adjuvant
therapy for patients with hormone receptor-positive ductal
carcinoma in situ. PLoS One. 17:e02629342022. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mills MN, Russo NW, Fahey M, Nanda RH,
Raiker S, Jastrzebski J, Stout LL, Wilson JP, Altoos TA, Allen KG,
et al: Increased risk for ipsilateral breast tumor recurrence in
invasive lobular carcinoma after accelerated partial breast
irradiation brachytherapy. Oncologist. 26:e1931–e1938. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sanz-Garcia E, Zhao E, Bratman SV and Siu
LL: Monitoring and adapting cancer treatment using circulating
tumor DNA kinetics: Current research, opportunities, and
challenges. Sci Adv. 8:eabi86182022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zavarykina TM, Lomskova PK, Pronina IV,
Khokhlova SV, Stenina MB and Sukhikh GT: Circulating tumor DNA is a
variant of liquid biopsy with predictive and prognostic clinical
value in breast cancer patients. Int J Mol Sci. 24:170732023.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Arisi MF, Dotan E and Fernandez SV:
Circulating tumor DNA in precision oncology and its applications in
colorectal cancer. Int J Mol Sci. 23:44412022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Reinert T, Henriksen TV, Christensen E,
Sharma S, Salari R, Sethi H, Knudsen M, Nordentoft I, Wu HT, Tin
AS, et al: Analysis of plasma cell-free DNA by ultradeep sequencing
in patients with stages I to III colorectal cancer. JAMA Oncol.
5:1124–1131. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wolff AC, Hammond MEH, Hicks DG, Dowsett
M, McShane LM, Allison KH, Allred DC, Bertlett JMS, Bilous M,
Fitzgibbons P, et al: Recommendations for human epidermal growth
factor receptor 2 testing in breast cancer: American Society of
Clinical Oncology/College of American Pathologists clinical
practice guideline update. J Clin Oncol. 31:3997–4013. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
D'Orsi C, Bassett L and Feig S: Breast
imaging reporting and data system (BI-RADS). Breast Imaging Atlas.
4th edition. American College of Radiology; Reston, VA, USA: pp.
29–47. 2018
|
|
18
|
Amin MD, Gress DM, Meyer Vega LR, Edge SB,
Greene FL, Byrd DR, Brookland RK, Washington MK and Compton CC;
American Joint Committee on Cancer (AJCC), : AJCC cancer staging
manual. 8th edition. Springer; New York, NY, USA: pp. 10322017
|
|
19
|
Olsson E, Winter C, George A, Chen Y,
Howlin J, Tang MH, Dahlgren M, Schulz R, Grabau D and van Westen D:
Serial monitoring of circulating tumor DNA in patients with primary
breast cancer for detection of occult metastatic disease. EMBO Mol
Med. 7:1034–1047. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Vlataki K, Antonouli S, Kalyvioti C,
Lampri E, Kamina S, Mauri D, Harissis HV and Magklara A:
Circulating tumor DNA in the management of early-stage breast
cancer. Cells. 12:15732023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Magbanua MJM, Swigart LB, Wu HT, Hirst GL,
Yau C, Wolf DM, Tin A, Salari R, Shchegrova S, Pawar H, et al:
Circulating tumor DNA in neoadjuvant-treated breast cancer reflects
response and survival. Ann Oncol. 32:229–239. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fiste O, Liontos M, Koutsoukos K, Terpos
E, Dimopoulos MA and Zagouri F: Circulating tumor DNA-based
predictive biomarkers in breast cancer clinical trials: A narrative
review. Ann Transl Med. 8:16032020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Adejolu M, Huo L, Rohren E, Santiago L and
Yang WT: False-positive lesions mimicking breast cancer on FDG PET
and PET/CT. AJR Am J Roentgenol. 198:W304–W314. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Garcia-Murillas I, Schiavon G, Weigelt B,
Ng C, Hrebien S, Cutts RJ, Cheang M, Osin P, Nerurkar A, Kozarewa
I, et al: Mutation tracking in circulating tumor DNA predicts
relapse in early breast cancer. Sci Transl Med. 7:302ra1332015.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lipsyc-Sharf M, de Bruin EC, Santos K,
McEwen R, Stetson D, Patel A, Kirkner GJ, Hughes ME, Tolaney SM,
Partridge AH, et al: Circulating tumor DNA and late recurrence in
high-risk hormone receptor-positive, human epidermal growth factor
receptor 2-negative breast cancer. J Clin Oncol. 40:2408–2419.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Magbanua MJM, van't Veer L, Clark AS,
Chien AJ, Boughey JC, Han HS, Wallace A, Beckwith H, Liu MC, Yau C,
et al: Outcomes and clinicopathologic characteristics associated
with disseminated tumor cells in bone marrow after neoadjuvant
chemotherapy in high-risk early stage breast cancer: The I-SPY
SURMOUNT study. Breast Cancer Res Treat. 198:383–390. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cavallone L, Aguilar-Mahecha A, Lafleur J,
Brousse S, Aldamry M, Roseshter T, Lan C, Alirezaie N, Bareke E,
Majewski J, et al: Prognostic and predictive value of circulating
tumor DNA during neoadjuvant chemotherapy for triple negative
breast cancer. Sci Rep. 10:147042020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Coombes RC, Page K, Salari R, Hastings RK,
Armstrong A, Ahmed S, Ali S, Cleator S, Kenny L, Stebbing J, et al:
Personalized detection of circulating tumor DNA antedates breast
cancer metastatic recurrence. Clin Cancer Res. 25:4255–4263. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cailleux F, Agostinetto E, Lambertini M,
Rothé F, Wu HT, Balcioglu M, Kalashnikova E, Vincent D, Viglietti
G, Gombos A, et al: Circulating tumor DNA after neoadjuvant
chemotherapy in breast cancer is associated with disease relapse.
JCO Precis Oncol. 6:e22001482022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lin PH, Wang MY, Lo C, Tsai LW, Yen TC,
Huang TY, Huang WC, Yang K, Chen CK, Fan SC, et al: Circulating
tumor DNA as a predictive marker of recurrence for patients with
stage II–III breast cancer treated with neoadjuvant therapy. Front
Oncol. 11:7367692021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lee TH, Kim H, Kim YJ, Park WY, Park W,
Cho WK and Kim N: Implication of pre- and post-radiotherapy ctDNA
dynamics in patients with residual triple-negative breast cancer at
surgery after neoadjuvant chemotherapy: Findings from a prospective
observational study. Cancer Res Treat. Nov 10–2023.(Epub ahead of
print).
|