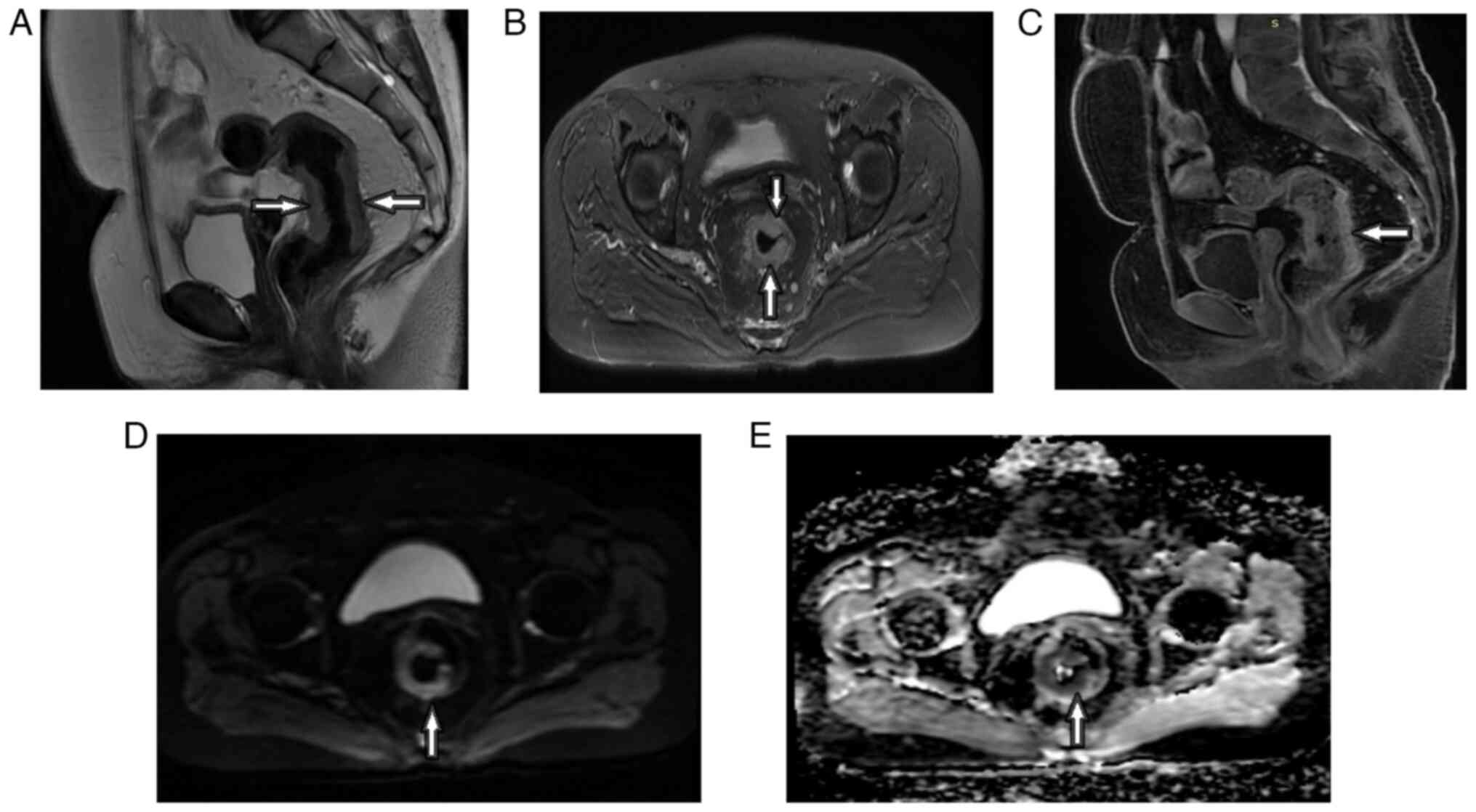
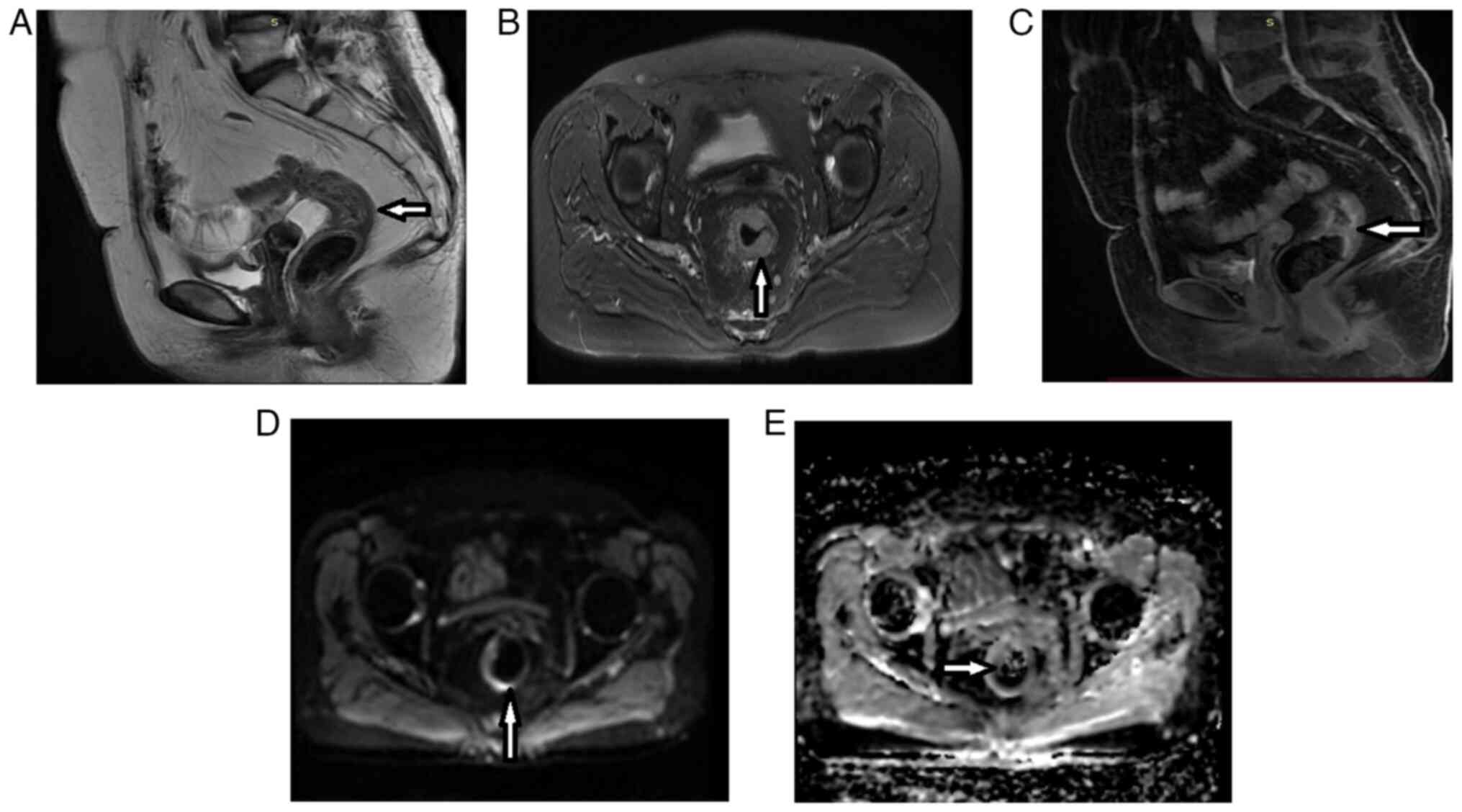
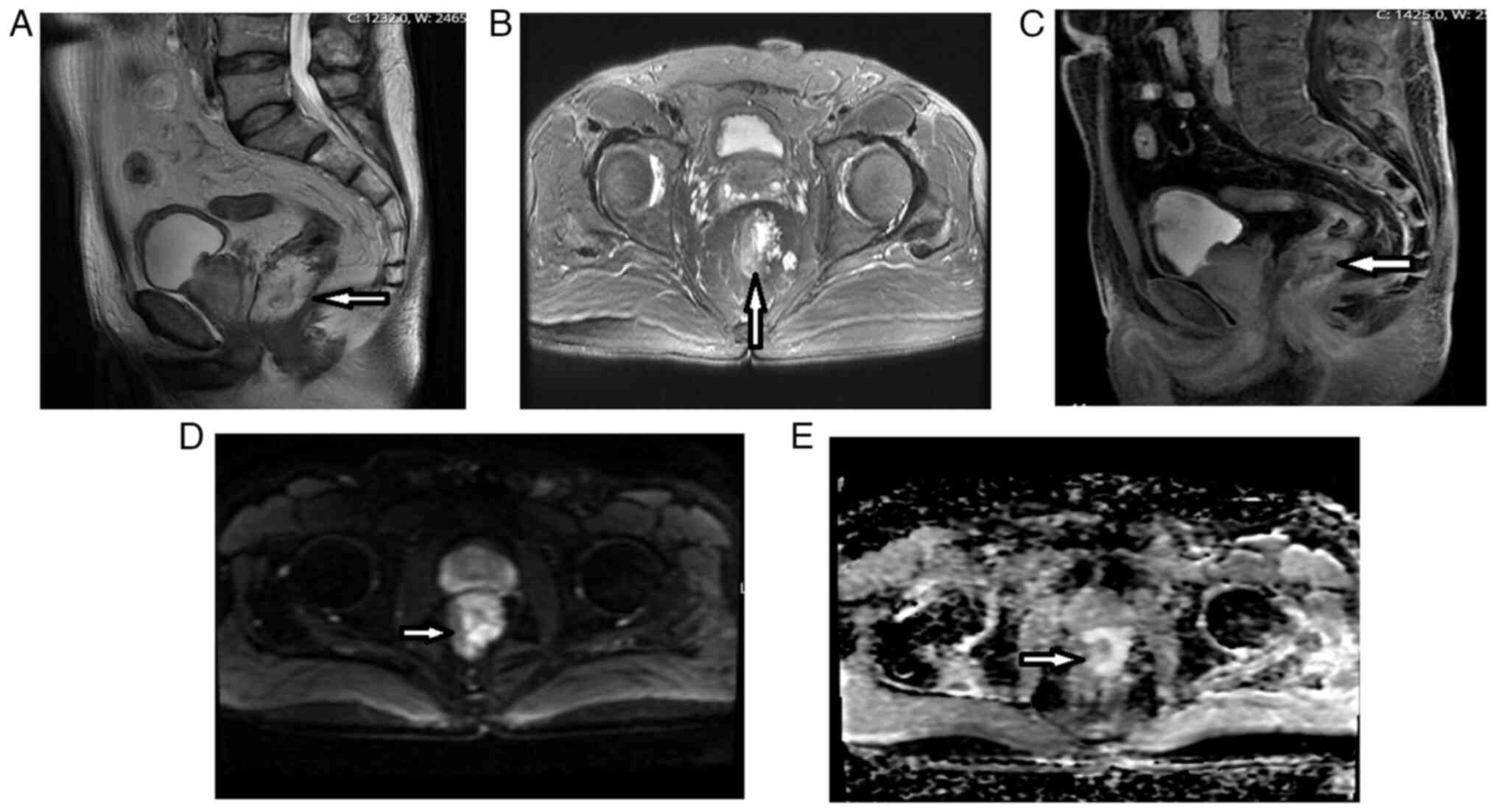
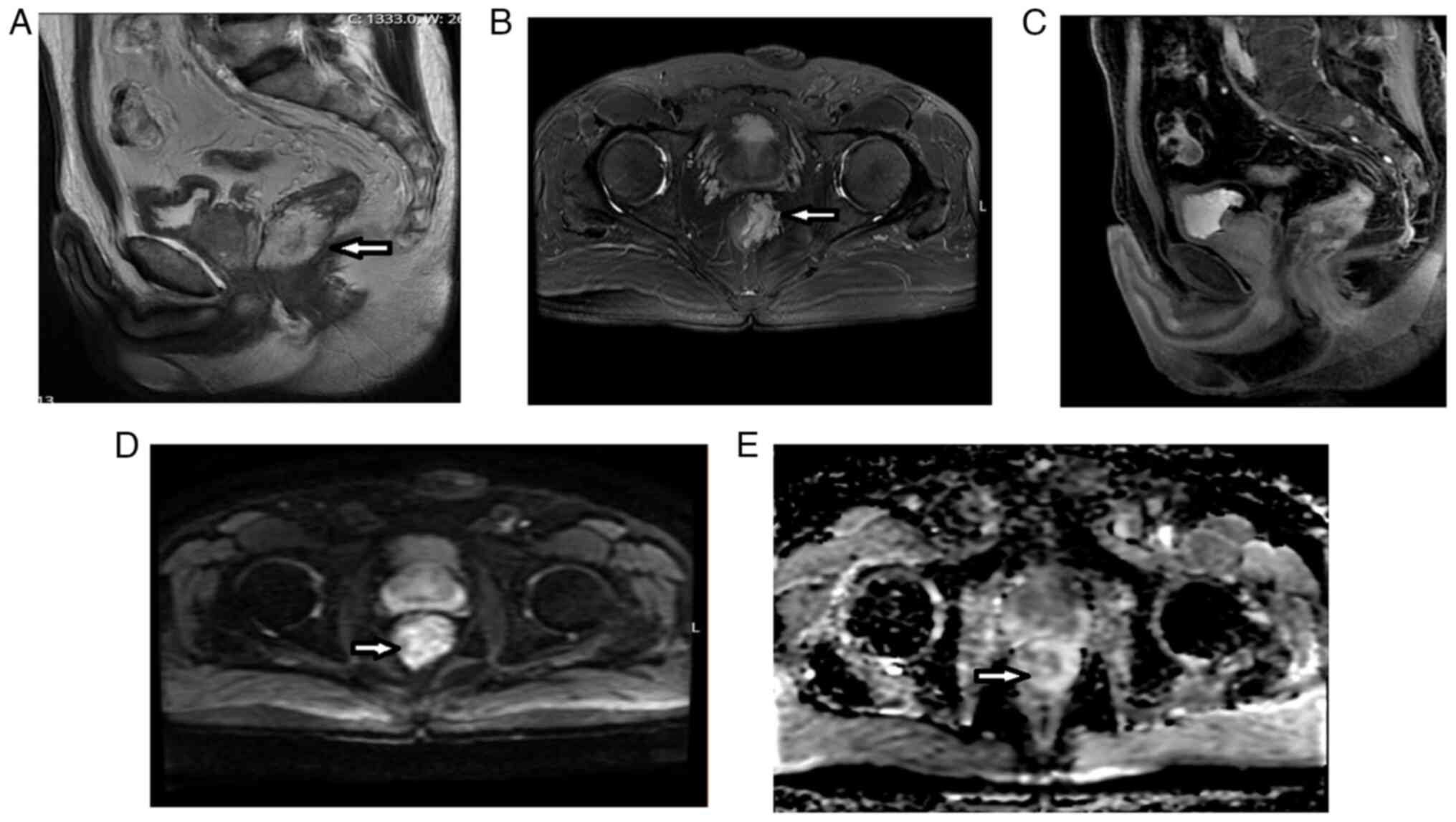
Introduction
Colorectal cancer is the second most common cause of
cancer-related deaths in women and the third most common in men
worldwide. Rectal cancer has an incidence rate of 40 per 100,000
patients and accounts for more than one-third of colorectal cancer
cases. Due to anatomical limitations, rectal cancer is more
challenging to resect, has a higher risk of local recurrence, and
generally has a worse prognosis (1). Colorectal cancer also has a higher
risk of metastasizing to the lungs compared to colon cancer. The
5-year mortality rate for patients diagnosed at Stage IV is
approximately 90% (2). The
treatment approach for rectal cancer is total mesorectal excision.
In individuals with locally advanced rectal cancer (LARC), the use
of neoadjuvant chemoradiotherapy (CRT) has resulted in significant
improvements in local disease control and fewer side effects
compared with adjuvant therapy administered after surgery (3,4).
High-resolution magnetic resonance imaging (MRI) is
the preferred method for diagnosing rectal cancer; it plays a
crucial role in preoperative diagnosis and staging, in the
identification of cases that may benefit from preoperative
neoadjuvant CRT and in post-CRT restaging to assess treatment
response. This imaging technique is vital for guiding surgical
approaches and oncological treatment alternatives (5–8).
Patients with LARC receive personalized CRT
treatments. In recent years, there has been an increasing demand
for more detailed and reliable radiological response assessments of
such Patients with LARC receive personalized CRT treatments. In
recent years, there has been an increasing demand for more detailed
and reliable radiological response assessments of such patients.
Post-CRT MRI plays a critical role in the decision-making process
for managing rectal cancer by providing crucial information on
prognostic factors, such as tumor grade, circumferential resection
margin (CRM) status, mesorectal fascia (MRF) involvement, the
presence of extramural vascular invasion (EMVI) and the mucin
content of the tumor (9–11).
MRI is the most sensitive noninvasive imaging method
for staging rectal cancer patients. High-resolution T2-weighted
(T2-W) images are regarded as the most sensitive sequences for
evaluation. However, existing MRI techniques face certain
limitations in assessing post-CRT treatment responses, particularly
in regions affected by post-radiation fibrosis. Consequently, it is
crucial to incorporate functional MRI sequences, such as
diffusion-weighted imaging (DWI) sequences, in order to enhance the
distinction between the tumor and fibrosis are increasingly being
adopted in clinical MRI protocols to reliably determine the tumor
response and achieve a balance between oncological safety and
patient quality of life (5,11–14).
Dynamic contrast-enhanced magnetic resonance imaging
(DCE-MRI), the magnetization transfer ratio, and textural analysis
(i.e., radiomics) are being investigated to overcome some
limitations in the restaging of rectal cancer post-CRT. However,
none of these techniques are currently used in routine clinical
practice (6).
To reduce costs, shorten imaging and assessment
times, and avoid unnecessary sequences that might negatively impact
patient comfort, abbreviated MRI protocols have emerged as
alternatives to standard protocols (13). According to the ESGAR guidelines,
the MRI protocol for staging rectal cancer should include T2-W
sequences in three planes and DWI, which can provide adequate
disease assessment in approximately 10–15 min, thus mimicking an
abbreviated protocol (9). However,
non-contrast and contrast-enhanced fat-suppressed (FS) T1-W
sequences are not routinely recommended during DCE-MRI or after the
administration of contrast agents (5,8,12).
Combining morphological and functional assessments using T2-W
sequences with DWI and DCE-MRI sequences could be beneficial for
the restaging of rectal cancer. Because T1 and postcontrast
T1-weighted sequences are not recommended because they do not
provide optimum information about the anatomy (5,12,15–18).
Currently, the accuracy of MRI is lower in post-CRT
staging than in pre-CRT assessment. Furthermore, there is no
consensus on a standard protocol or the utility of DCE-MRI and DWI
for post-CRT staging (12). DCE-MRI
is part of the routine imaging protocol for assessing tumor
response after neoadjuvant CRT, as it provides a reliable basis for
evaluating tumor response after an initial contrast-enhanced study.
It is recommended that post-CRT tumor restaging be performed using
the same protocol and scanner as the initial staging phase
(5,8,12,19,20).
Although AB-MRI protocols are considered promising for
significantly shortening imaging time while maintaining diagnostic
accuracy in breast and prostate cancer, and other cancer types, few
studies have evaluated their effectiveness in the post-CRT
restaging of rectal cancer (5,6,11,20–24).
The aim of the present study was to compare the
effectiveness of two post-CRT abbreviated rectal MRI protocols in
evaluating tumor response to neoadjuvant CRT in patients with LARC.
This comparison was also evaluated against pretreatment DCE-MRI
findings.
Materials and methods
Study population
The images used in the present study were created
during assessments performed Recep Tayyip Erdogan University
Faculty of Medicine, Training and Research Hospital Radiology
Department between December 2015 and December 2021. Our exclusion
criteria include low image quality, claustrophobia, refusal to
participate in the study, patients who cannot be pathologically
confirmed, contraindications to contrast agents, and cases where
MRI could not be performed before or after neoadjuvant
chemoradiotherapy. After applying the exclusion criteria, a total
of 62 patients were included in the study. In order to evaluate the
treatment response after the exclusion criteria, 62 patients
diagnosed with rectal cancer who underwent neoadjuvant CRT and
standard contrast-enhanced rectal MRI examination before (for
staging purposes) and after CRT (for evaluating the treatment
response) were included in the study. The patients had a median age
of 58 years (range: 37–85 years), and 54.8% were male. The AB-MRI
protocols, which were obtained via retrospective screening and
re-analysis of standard contrast-enhanced MRI (T2-W, postcontrast
T1-W, and DWI) data registered in picture archiving and
communication systems (PACS), included the AB1 (T2-W and DWI) and
AB2 (postcontrast FS T1-W and DWI) protocols. Two radiologists,
blinded to the results, reached a consensus after jointly reviewing
images obtained using standard MRI sequences at baseline and
protocol AB-MRI sequences obtained after treatment.
Written informed consent was obtained from each
subject following a detailed explanation of the objectives and
protocol of the study, which was conducted in accordance with the
ethical principles in the ‘Declaration of Helsinki’ and approved by
the Recep Tayyip Erdogan University Faculty of Medicine
Non-interventional Clinical Research Ethics Committee (Date of
Approval: 07/06/2022; Reference/Protocol No.: 2022/134). Informed
consent was obtained from the participants whose images were used
in the publication.
Assessments
Demographic data, tumor histopathology, metastatic
lymph node status, and residual tumor size were recorded for each
patient. The pretreatment MRI and post-CRT (with two AB-MRI
protocols) data included measurements of lesion length, lesion
width, lymph node size, tumor stage, CRM, EMVI, mucoid accumulation
in the lumen (MAIN), extramural extension, mucinous response, MRF
involvement, and MRI-based tumor regression grade (mrTRG). TNM
classification was used for staging rectal cancer (7). Additionally, the reliability and
concordance of the AB1 and AB2 protocols in assessing tumor
response were analyzed.
mrTRG was classified based on four grades: Grade 1
(significant fibrosis but no residual tumor), grade 2 (significant
fibrosis and minimal residual tumor), grade 3 (fibrosis and tumor)
and grade 4 (significant residual tumor and minimal fibrosis). EMVI
was defined as positive when minimal or <25% fibrotic change was
detected on the treated tumor component within the extramural
venous structure. Mucinous response was categorized as poor (i.e.,
the tumor remained non-mucinous or mucinous after treatment) or
good (i.e., there was a non-mucinous to mucinous conversion after
treatment or the presence of tumor cell necrosis with mucinous
structures persisting between them).
MRI protocol
Prior to medical treatment and following neoadjuvant
CRT, MRI was conducted using a 3 Tesla MRI system (GE Healthcare
Discovery MR750, Waukesha, WI) and a pelvic phased-array surface
coil (eight-channel body coil). The contrast agent gadobutrol
(Gadovist; Bayer AG) was injected and the remaining contrast agent
in the catheter used for MRI was flushed with 20 ml of saline to
flush the remaining contrast agent from the catheter after the
contrast injection at a flow rate of 2 ml/sec at 0.1 mmol/kg body
weight. Post-contrast T1-WI was obtained approximately 2 min after
the injection of the contrast age.
The MRI protocol for staging rectal cancer and
evaluating treatment response included the following sequences:
axial FS T1-W imaging, axial non-fat-saturated T2-W imaging, axial
fat-saturated T2-W imaging, postcontrast fat-saturated T1-W
imaging, coronal fat-saturated T2-W imaging, sagittal
high-resolution DWI, and axial DWI. For the purpose of response
assessment, abbreviated protocols created from the full MRI
protocol were AB1, consisting of T2-WI and DWI, and AB2, consisting
of post-contrast axial FS T1-WI and axial DWI.
Statistical analysis
Statistical analysis was performed using IBM SPSS
Statistics for Windows, version 26.0 (IBM Corp.). The conformity of
the variables to the normal distribution was examined visually
(histogram and probability graphs) and using the Kolmogorov-Smirnov
method. The Friedman test was used to compare the difference
between continuous variables, with Bonferroni's correction for
pairwise results. The McNemar test was used to assess the
statistical difference for related categorical variables.
Reliability analysis was performed using the intraclass correlation
test with consideration of intraclass correlation coefficient (ICC)
values <0.5, between 0.5 and 0.75, between 0.75 and 0.9, and
>0.9, to indicate poor, moderate, good and excellent
reliability, respectively. Agreement between the AB1 and AB2
protocols was analyzed using Cohen's κ coefficient values and
interpreted as poor (<0.20), fair (0.20-0.40), moderate
(0.41-0.60), good (0.61-0.80) and very good (0.81-1.00). Absolute
agreement was calculated as the percentage of the same results for
the variables. Data are expressed as the median, confidence
interval (CI) and n (%) where appropriate. P<0.05 was considered
to indicate a statistically significant difference.
Results
Patients
The median patient age was 58 years (range, 37–85
years), and 54.8% of the patients were male. The histological
subtype was adenocarcinoma in 90.3% of the patients, and surgical
metastatic lymphanedopathy (LAP) was noted in 43.5% of the patients
(Table I).
 | Table I.Demographic characteristics. |
Table I.
Demographic characteristics.
| Characteristic | Value |
|---|
| Age,
yearsa | 58 (37–85) |
| Sex, n (%) |
|
|
Female | 28 (45.2) |
|
Male | 34 (54.8) |
| Histological
subtype, n (%) |
|
|
Adenocarcinoma | 56 (90.3) |
|
Mucinous cancer | 6 (9.7) |
| Surgical metastatic
LAP, n (%) |
|
| No | 35 (56.5) |
|
Yes | 27 (43.5) |
| Residual
tumora mm | 29.5 (0–124) |
| ADC,
mm2/seca |
|
|
Pre-treatment | 0.91
(0.50-1.58) |
|
Post-treatment | 1.21
(0.13-1.8) |
Tumor response to neoadjuvant CRT
As shown in Figs. 1
and 2, and Table II, the post-CRT AB1 and AB2
protocols revealed increases in the percentages of T0-T2 stage
tumors (from 0% for pretreatment to 22.5% for AB1 and 24.2% for
AB2) and decreases in the percentages of T4a (from 38.7 to 17.7%
for AB1 and 19.4% for AB2) and T4b (from 11.3 to 0.0% for both AB1
and AB2) stage tumors. There were also decreases in the rate of
lymph node positivity (from 90.3 to 50.0% for AB1 and 51.6% for
AB2), CRM involvement (from 72.6 to 38.7% for both protocols), MRF
involvement (from 58.1 to 38.7% for AB1 and 42.0% for AB2), EMVI
(from 72.6 to 48.4% for AB1 and 38.7% for AB2), MAIN (from 40.3 to
17.7% for AB1 and 14.5% for AB2) and extramural extension (from
61.3 to 43.5% for AB1 and 41.9% for AB2).
 | Table II.Tumor response to neoadjuvant
chemoradiotherapy. |
Table II.
Tumor response to neoadjuvant
chemoradiotherapy.
|
|
| Post-treatment
AB-MRI protocol |
|---|
|
|
|
|
|---|
| Parameter | Pretreatment | AB1 (T2-WI and
DWI) | AB2 (post-contrast
FS T1-WI and DWI) |
|---|
| Tumor stage, n
(%) |
|
|
|
| 0 | 0 (0.0) | 1 (1.6) | 0 (0.0) |
| 1 | 0 (0.0) | 2 (3.2) | 3 (4.8) |
| 2 | 0 (0.0) | 11 (17.7) | 12 (19.4) |
| 3a | 1 (1.6) | 3 (4.8) | 2 (3.2) |
| 3b | 10 (16.1) | 21 (33.9) | 20 (32.3) |
| 3c | 14 (22.6) | 4 (6.5) | 6 (9.7) |
| 3d | 6 (9.7) | 9 (14.5) | 7 (11.3) |
| 4a | 24 (38.7) | 11 (17.7) | 12 (19.4) |
| 4b | 7 (11.3) | 0 (0.0) | 0 (0.0) |
| Lymph node
positivity, n (%) |
|
|
|
| No | 6 (9.7) | 31 (50.0) | 30 (48.4) |
|
Yes | 56 (90.3) | 31 (50.0) | 32 (51.6) |
| CRM involvement, n
(%) |
|
|
|
| No | 17 (27.4) | 38 (61.3) | 38 (61.3) |
|
Yes | 45 (72.6) | 24 (38.7) | 24 (38.7) |
| MRF involvement, n
(%) |
|
|
|
| No | 26 (41.9) | 38 (61.3) | 36 (58.1) |
|
Yes | 36 (58.1) | 24 (38.7) | 26 (41.9) |
| EMVI, n (%) |
|
|
|
| No | 17 (27.4) | 32 (51.6) | 38 (61.3) |
|
Yes | 45 (72.6) | 30 (48.4) | 24 (38.7) |
| MAIN, n (%) |
|
|
|
| No | 37 (59.7) | 51 (82.3) | 53 (85.5) |
|
Yes | 25 (40.3) | 11 (17.7) | 9 (14.5) |
| Extramural
extension, n (%) |
|
|
|
| No | 24 (38.7) | 35 (56.5) | 36 (58.1) |
|
Yes | 38 (61.3) | 27 (43.5) | 26 (41.9) |
| Mucinous response,
n (%) |
|
|
|
|
Poor | 30 (48.4) | 53 (85.5) | 18 (29.0) |
|
Good | 32 (51.6) | 9 (14.5) | 44 (71.0) |
| mrTRG, n (%) |
|
|
|
| Grade 1
(significant fibrosis, no residue tumor) |
| 5 (8.1) | 5 (8.1) |
| Grade 2
(significant fibrosis, minimal residue tumor) |
| 13 (21.0) | 15 (24.2) |
| Grade 3
(fibrosis and tumor) |
| 27 (43.5) | 29 (46.8) |
| Grade 4
(significant residual tumor, minimal fibrosis) |
| 17 (27.4) | 13 (21.0) |
As shown in Table
II, the rate of poor mucinous response was 48.4% for the
pretreatment MRI and 85.5 and 29.0% for the post-CRT AB1 and AB2
protocols, respectively. Post-CRT mrTRG was at grade 3 in nearly
half of the imaging assessed by the AB1 (43.5%) and AB2 (46.8%)
protocols.
Lesion and lymph node
characteristics
As shown in Table
III, the post-CRT AB1 and AB2 protocols revealed significant
decreases in median lesion length from 63 mm (range, 28–132 mm) for
pretreatment MRI to 37.5 mm (range, 0–128 mm) for AB1 and 37.5 mm
(range, 0–120 mm) for AB2 (both P<0,001). There were also
significant decreases in lesion width from 19 mm (range, 12–56 mm)
to 14.0 mm (range, 0–23 mm) and 12 mm (range, 0–24 mm),
respectively (both P<0.05), and significant decreases in lymph
node size from 9 mm (range, 0–19 mm) to 6 mm (0–14 mm) and 6 mm
(range, 0–13 mm), respectively (both P<0.05).
 | Table III.Lesion and lymph node
characteristics. |
Table III.
Lesion and lymph node
characteristics.
|
|
| Post-treatment
AB-MRI protocol |
|
|---|
|
|
|
|
|
|---|
| Characteristic | Pretreatment | AB1 (T2-WI and
DWI) | AB2 (post-contrast
FS T1-WI and DWI) | P-value |
|---|
| Lesion length along
long axis mm (n=62) | 63.0
(28.0-132.0) | 37.5
(0–128.0)a | 37.5
(0–120.0)a |
<0.001b |
| Lesion width mm
(n=62) | 19.0
(12.0-56.0) | 14.0
(0–23.0)a | 12.0
(0–24.0)a |
<0.001b |
| Lymph node size mm
(n=53) | 9.0 (0–19.0) | 6.0
(0–14.0)a | 6.0
(0–13.0)a |
<0.001b |
Reliability and agreement analysis for
the AB1 and AB2 protocols
As shown in Table
IV, the post-CRT AB1 and AB2 protocols had excellent
reliability in identifying lesion length (ICC, 0.988; 95% CI,
0.980-0.993; P<0.001), lesion width (ICC, 0.972; 95% CI,
0.953-0.983; P<0.001) and lymph node size (ICC, 0.979; 95% CI,
0.963-0.988; P<0.001). Cohen's κ coefficient indicated very good
agreement between the AB1 and AB2 protocols in terms of identifying
lymph node positivity (κ, 0.903; absolute agreement, 95.2%), CRM
involvement (κ, 0.864; absolute agreement, 93.5%), EMVI (κ, 0.805;
absolute agreement, 90.3%), MAIN (κ, 0.881; absolute agreement,
96.8%) and MRF involvement (κ, 0.838; absolute agreement, 91.9%).
There was good agreement between the AB1 and AB2 protocols in
identifying extramural extension (κ, 0.769; absolute agreement,
88.7%). However, no agreement was observed between the protocols in
terms of identifying mucinous response (κ, 0.084; absolute
agreement, 41.9%).
 | Table IV.Reliability and agreement analysis
for abbreviated magnetic resonance imaging protocols. |
Table IV.
Reliability and agreement analysis
for abbreviated magnetic resonance imaging protocols.
|
| AB1 (T2-WI and DWI)
and AB2 (post-contrast FS T1-WI and DWI) |
|---|
|
|
|
|---|
| Parameters | ICC (CI) |
|
P-valuea |
|---|
| Lesion length
mm | 0.988
(0.980-0.993) |
| <0.001 |
| Lesion width,
mm | 0.972
(0.953-0.983) |
| <0.001 |
| Lymph node size
mm | 0.979
(0.963-0.988) |
| <0.001 |
|
|
Parameters | Cohen's κ
value | Absolute
agreement, % |
P-valueb |
|
| Lymph node
positivity | 0.903 | 95.2 | <0.001 |
| CRM
involvement | 0.864 | 93.5 | <0.001 |
| EMVI | 0.805 | 90.3 | <0.001 |
| MAINa | 0.881 | 96.8 | <0.001 |
| Extramural
extension | 0.769 | 88.7 | <0.001 |
| MRF
involvement | 0.838 | 91.9 | <0.001 |
| Mucinous
response | 0.084 | 41.9 | 0.190 |
Cross-analysis of the pretreatment and
post-CRT protocols
As shown in Figs. 3,
and 4, and Table V, the post-CRT AB1 and AB2 protocols
revealed similar improvements in lymph node positivity (disappeared
after CRT in 44.6 and 42.8% of patients, respectively), CRM
involvement (disappeared in 48.6 and 46.0% of patients,
respectively), and MRF involvement (disappeared in 41.7 and 38.9%
of patients, respectively). The AB1 and AB2 protocols yielded lower
rates of improvement for EMVI (disappeared in 33.3% vs. 46.7% of
patients, respectively), MAIN (disappeared in 60.0% vs. 68.0% of
patients, respectively), and, especially, mucinous response.
 | Table V.Cross-analysis of pretreatment and
post-treatment protocols. |
Table V.
Cross-analysis of pretreatment and
post-treatment protocols.
|
| Post-treatment
AB-MRI protocol |
|---|
|
|
|
|---|
|
| AB1 (T2-WI and
DWI) | AB2 (post-contrast
FS T1-WI and DWI) |
|---|
|
|
|
|
|---|
| Pretreatment
MRI | No | Yes | No | Yes |
|---|
| Lymph node
positivity (n=62) |
|
|
|
|
| No
(n=6) | 6 (100.0) | 0 (0.0) | 6 (100.0) | 0 (0.0) |
| Yes
(n=56) | 25 (44.6) | 31 (55.4) | 24 (42.9) | 32 (57.1) |
|
P-valuea | <0.001 |
| <0.001 |
|
| CRM involvement
(n=62) |
|
|
|
|
| No
(n=25) | 20 (80.0) | 5 (20.0) | 21 (84.0) | 4 (16.0) |
| Yes
(n=37) | 18 (48.6) | 19 (51.4) | 17 (45.9) | 20 (54.1) |
|
P-valuea | 0.011 |
| 0.007 |
|
| EMVI (n=62) |
|
|
|
|
| No
(n=17) | 17 (100.0) | 0 (0.0) | 17 (100.0) | 0 (0.0) |
| Yes
(n=45) | 15 (33.3) | 30 (66.7) | 21 (46.7) | 24 (53.3) |
|
P-valuea | <0.001 |
| <0.001 |
|
| MAIN (n=62) |
|
|
|
|
| No
(n=37) | 36 (97.3) | 1 (2.7) | 36 (97.3) | 1 (2.7) |
| Yes
(n=25) | 15 (60.0) | 10 (40.0) | 17 (68.0) | 8 (32.0) |
|
P-valuea | 0.001 |
| <0.001 |
|
| Extramural
extension (n=62) |
|
|
|
|
| No
(n=24) | 23 (95.8) | 1 (4.2) | 22 (91.7) | 2 (8.3) |
| Yes
(n=38) | 12 (31.6) | 26 (68.4) | 14 (36.8) | 24 (63.2) |
|
P-valuea | 0.003 |
| 0.004 |
|
| MRF involvement
(n=62) |
|
|
|
|
| No
(n=26) | 23 (88.5) | 3 (11.5) | 22 (84.6) | 4 (15.4) |
| Yes
(n=36) | 15 (41.7) | 21 (58.3) | 14 (38.9) | 22 (61.1) |
|
P-valuea | 0.008 |
| 0.031 |
|
Cross-analysis of the pretreatment and
post-CRT protocols
As shown in Table
VI, the post-CRT AB1 and AB2 protocols revealed had lower
recovery rates for mucoid response (lost in 10.3 and 48.3% of
patients, respectively).
 | Table VI.Cross-analysis of pretreatment and
post-treatment protocols. |
Table VI.
Cross-analysis of pretreatment and
post-treatment protocols.
|
| Post-treatment
AB-MRI protocol |
|---|
|
|
|
|---|
|
| AB1 (T2-WI and
DWI) | AB2 (post-contrast
FS T1-WI and DWI) |
|---|
|
|
|
|
|---|
| Mucinous response
(n=62) | Poor | Good | Poor | Good |
|---|
| Good (n=33) | 27 (81.8) | 6 (18.2) | 4 (12.1) | 29 (87.9) |
| Poor (n=29) | 26 (89.7) | 3 (10.3) | 15 (51.7) | 14 (48.3) |
|
P-valuea | 0.353 |
| <0.001 |
|
Cross-analysis of the AB1 and AB2
protocols
As shown in Table
VII, with regard to patients with lymph node positivity, MRF
involvement, CRM involvement, mucinous response, extramural
extension, MAIN and EMVI in the AB1 protocol, the AB2 protocol
confirmed the findings in 96.8, 95.8, 91.7, 88.9, 85.2, 81.8 and
80.0% of the patients, respectively.
 | Table VII.Cross analysis of AB1 and AB2
protocols. |
Table VII.
Cross analysis of AB1 and AB2
protocols.
|
| AB2 (post-contrast
FS T1-WI and DWI) |
|---|
|
|
|
|---|
| AB1 (T2-WI and
DWI) | No | Yes |
|---|
| Lymph node
positivity (n=62) |
|
|
| No | 29 (93.5) | 2 (6.5) |
|
Yes | 1 (3.2) | 30 (96.8) |
|
P-value | 1.0 |
|
| CRM involvement
(n=62) |
|
|
| No | 36 (94.7) | 2 (5.3) |
|
Yes | 2 (8.3) | 22 (91.7) |
|
P-value | >0.999 |
|
| EMVI (n=62) |
|
|
| No | 32 (100) | 0 (0) |
|
Yes | 6 (20.0) | 24 (80.0) |
|
P-value | 0.031 |
|
| MAIN (n=62) |
|
|
| No | 51 (100) | 0 (0) |
|
Yes | 2 (18.2) | 9 (81.8) |
|
P-value | 0.500 |
|
| Extramural
extension (n=62) |
|
|
| No | 32 (91.4) | 3 (8.6) |
|
Yes | 4 (14.8) | 23 (85.2) |
|
P-value | >0.999 |
|
| MRF involvement
(n=62) |
|
|
| No | 35 (92.1) | 3 (7.9) |
|
Yes | 1 (4.2) | 23 (95.8) |
|
P-value | 0.625 |
|
| Mucinous response
(n=62) |
|
|
|
Good | 1 (11.1) | 8 (88.9) |
|
Poor | 18 (34.6) | 35 (63.4) |
|
P-value | 0,111 |
|
Discussion
In the present study, some of the patients who
received neoadjuvant CRT experienced improvements in clinical and
pathological outcomes, such as reductions in tumor, lesion and
lymph node sizes, and decreases in lymph node positivity, CRM and
MRF involvement, EMVI, MAIN. These findings suggest that
neoadjuvant CRT supports sphincter-sparing surgery and even
nonsurgical treatment approaches in some cases, alongside tumor
shrinkage (in over 50% of cases) and pathological complete response
(pCR) (in 15–38% of cases) (7,8).
Therefore, the use of an abbreviated rectal MRI protocol via T2-W
and DWI or postcontrast FS T1-W and DWI series facilitates the
assessment of tumor response after neoadjuvant CRT in rectal cancer
patients. It also allows for a reduction in examination time
without compromising imaging quality during post-CRT follow-up.
Diffusion-weighted imaging (DWI) is considered an
optional MRI sequence that is sensitive to the movement of water
molecules in vivo and is used for tumor and lymph node
detection in primary staging (2,6). The
movement caused by diffusion is influenced by the characteristics
of tissues and cells, the integrity of cell membranes, and the
viscosity of fluids. This type of imaging can be particularly
useful in the restaging of tumors after CRT (6,8,24–27).
This is because patients who respond to treatment often develop
fibrosis or necrosis accompanied by a decrease in cellular density,
(which leads to an increase in the apparent diffusion coefficient
(ADC) value (5,28,29).
ADC is a quantitative parameter used to assess the magnitude of
water diffusion movement within tissues and provides information
related to tissue cellularity. ADC values have been proven to be
associated with tumor cellularity and grade. ADC parameters have
been validated as potential predictors for early therapeutic
response in various types of cancer, including head and neck
cancer, Hodgkin lymphoma, lung cancer, breast cancer, and prostate
cancer (2).
DCE-MRI studies of rectal cancer have been
recognized as significant for pre- and post-CRT staging (15). While contrast-enhanced T1-W imaging
does not contribute to an increase in diagnostic accuracy in the
local staging of rectal cancer, it can be particularly useful
during post-CRT staging. This type of imaging helps radiologists
better identify local recurrence and assess common iliac and lower
para-aortic nodes and incidental pelvic findings. Additionally, it
has proven valuable in cases of mucinous neoplasms where T2 signal
intensities might closely resemble those of fat (5,6,15,30).
Indeed, a morphological and functional evaluation
combining the T2-W sequence with DWI and DCE-MRI sequences has been
shown to be useful in assessing post-CRT tumor response in rectal
cancer patients (5,12,15–18).
Likewise, besides the excellent reliability and very good agreement
between the AB1 and AB2 protocols in identifying several tumor
response criteria in the present study, the AB2 protocol yielded
significantly higher percentages of disappearances of EMVI and MAIN
and poor mucinous response after CRT compared to the AB1 protocol.
These findings seem to support the suggested value of including DWI
and T1-W sequences in addition to T2-W sequences in the MRI-based
restaging of rectal cancer patients (5,6).
The mrTRG scores provide important information in
follow-up evaluations after CRT. The theoretical success of pCR is
higher in patients with mrTRG 1 and 2, and these individuals show
improved disease-free survival and overall survival compared with
those with higher-grade mrTRG (9,11,31,32).
In the present study, the post-CRT AB1 and AB2 protocols yielded
similar results regarding the prevalence of mrTRG 1 (8.1% for each)
and 2 (21.0% for AB1 and 24.2% for AB2). This suggests that at
least one-third of our advanced-stage rectal cancer patients could
become candidates for organ-preserving treatments after neoadjuvant
CRT. However, post-CRT MRI is not reliable for making
determinations about more radical surgical dissection or
organ-preserving strategies due to its inadequacy in distinguishing
between residual viable tumors and CRT-related changes (such as
edema, necrosis, and, especially, fibrotic changes). Therefore,
T2-W imaging carries a risk of tumor overstaging because
CRT-induced fibrotic changes (low signal intensity on T2-W images)
at the interface between the tumor and mesorectal fat as well as
CRT-induced submucosal edema (thickened and intermediate to high
signal intensity on T2-W images) on the rectal wall adjacent to the
tumor can be misinterpreted as residual tumors (6,11,20,29).
Although fibrotic changes manifest as reduced signal
intensity on T2-W images, the dark signal intensity observed on MRI
does not solely indicate fibrotic change; approximately 50% of
viable residual tumor may remain in the fibrotic tissue (12,31–34).
Hence, determining tumor relationships with the surrounding
structures on the sole basis of T2 signal intensity changes is
likely to jeopardize oncologic safety. Meanwhile, supplementary
functional MRI sequences, such as DWI, have yielded encouraging
outcomes in the MRI-based restaging of tumors by distinguishing
between residual tumor (manifesting as high signal intensity on
DWI) and fibrosis (manifesting as low signal intensity on high
b-value DWI) (6,11,14,33,34).
Notably, our findings revealed considerable tumor downstaging due
to the post-CRT AB1 and AB2 protocols, including increases in T0-T2
stage tumors (to 22.5 and 24.2%, respectively) and decreases in T4a
(from 38.7 to 17.7 and 19.4%, respectively) and T4b (from 11.3 to
0.0%) stage tumors. These results seem notable given that the AB1
and AB2 protocols both included DWI and that DWI-based restaging
has been shown to have improved sensitivity and offers a solution
for overcoming the risk of tumor overstaging in T2-W imaging
(20,29).
In addition to the frequent intermixing of fibrosis
(i.e., decreased T2-W signal intensity) with viable tumor tissues,
the development of a mucinous response (i.e., increased T2-W signal
intensity) after CRT in rectal tumors challenges the prediction of
residual tumor viability in T2-W MRI protocols (11,14,35).
Following CRT, non-mucinous tumors have the potential to transform
into mucinous tumors, presenting with a mucinous (or colloid
degeneration) response. This response signals a positive reaction
to treatment and a more favorable prognosis. In contrast, mucinous
tumors can exhibit an acellular mucin response (i.e., a
pathological response specific to mucinous tumors) without
affecting recurrence-free survival, or they may not respond to CRT,
resulting in an elevated risk of local recurrence and a poorer
outcome (6,20,36).
In the present study, a conversion from a poor mucinous response to
a good mucinous response was observed in 3 out of 29 patients
(10.3%) based on pretreatment MRI and 14 out of 29 patients (48.3%)
based on the post-CRT AB1 and AB2 protocols, indicating a
significant discrepancy between the AB1 and AB2 protocols in
detecting changes in mucinous status. The presence of intermingled
tumor cells with fibrosis and mucin is likely the reason behind the
low level of agreement between mrTRG and pathological tumor
regression grade (pTRG) (11,37).
The very good agreement in determining CRM
involvement between the AB1 and AB2 protocols is important given
that CRM is one of the key components in determining the rate of
local tumor recurrence after CRT; Positive CRM involvement has been
observed in 25–26% of patients with CRM in previous studies
(9,11,38).
In addition, the protocols had very high concordance in terms of
MRF involvement in the current study, which seems important as
applying MRF involvement in addition to CRM (implementation of
distance from MRF) is likely to overcome certain limitations of
post-CRT MRI in detecting positive CRM [i.e., low positive
predictive value (PPV)], making more aggressive and additional
treatment unnecessary (11,34,39–41).
The potential utility of DWI in predicting tumor
clearance at the MRF after CRT has been documented. The positive
predictive value (PPV) of the AB 1 protocol for MRF involvement was
higher (82–91%) compared to T2W imaging alone (30–45%) (42). In this context, the presence of CRM
and MRF involvement in the post-CRT AB1 (38.7% for each) and AB2
(38.7% for CRM and 42.0% for MRF) protocols is noteworthy. This
finding is significant due to the potential introduction of
additional treatment options, such as extramesorectal excision or
multivisceral resection, boost radiotherapy at the site of MRF
invasion, and upfront chemotherapy. This is also true for patients
who are CRM and MRF positive after CRT (11).
The post-CRT AB1 and AB2 protocols revealed
comparable enhancements in lymph node positivity, which disappeared
after CRT in 44.6 and 42.8% of the patients, respectively.
Additionally, there were significant reductions in lymph node size,
which decreased from a median of 9 to 6 mm for each protocol. These
results suggest that a substantial number of irradiated lymph nodes
might vanish following CRT. Reliable predictors of negative node
status after surgery include a reduction in lymph node size of at
least 70% and a nodal size of less than 2.5 mm in the short axis
(6,43,44).
Persistent EMVI on post-CRT MRI has been shown to
increase the risk of metastatic disease and to serve as an
independent negative prognostic marker for disease-free survival;
regression or persistence of EMVI after neoadjuvant therapy has
been associated with improved survival (45,46).
In the present study, the AB1 and AB2 protocols showed the
disappearance of EMVI in 33.3 and 46.7% of patients who had EMVI on
pretreatment MRI, respectively. Furthermore, regarding patients who
had EMVI or MAIN according to the AB1 protocol, the AB2 protocol
revealed the absence of EMVI and MAIN in 20% of those cases,
indicating a potential discordance between the AB1 and AB2
protocols in the detection of changes in EMVI and MAIN.
Post-CRT MRI is regarded as the primary option for
assessing treatment response. It not only functions as a surgical
roadmap but also helps identify complete responders and facilitate
organ-preserving treatments. However, current MRI techniques have
certain limitations in evaluating post-CRT treatment response,
jeopardizing the reliability and safety of tumor response
evaluation. Furthermore, the use of post-CRT MRI-based data to
inform treatment strategies remains controversial (11,14).
Given the risk of radiological interpretation resulting in either a
faulty decision for organ-preserving treatment or unnecessary
radical surgery, improved knowledge of specific morphology and
diffusion signal patterns and the complementary use of functional
MRI sequences (such as DWI) in addition to morphological (T2-W) MRI
are important for increasing post-CRT MRI's ability to identify
complete responders and ensuring the absence of extramural tumor
infiltration and residual metastatic lymph nodes (11,14).
In this context, the present study's findings on the similar
effectiveness of abbreviated post-CRT MRI protocols and the use of
DWI, especially in determining CRM and MRF involvement, are
significant.
The present study has some limitations. The main
limitation is the lack of data on tumor response on post-CRT AB
protocols in relation to postoperative pathological findings.
Additionally, the impact of tumor histological findings and subtype
on treatment response with each MRI protocol, as well as the
assessment of residual tumor size based on the largest dimension
rather than the three-dimensional volume measurement, are
limitations that otherwise would extend the knowledge obtained in
the current study.
In conclusion, the use of an abbreviated rectal MRI
protocol has been shown to be a reliable method for evaluating
tumor response in patients with rectal cancer following neoadjuvant
CRT. Both AB protocols demonstrated excellent reliability and good
to very good agreement in identifying improved outcomes in post-CRT
clinicopathological results, including reductions in lesion length,
lesion width, lymph node size, lymph node positivity, CRM
involvement, EMVI, MAIN and MRF involvement. However, there were
variations in the diagnostic performance of the T2-WI and T1-WI
sequences in the detection of changes in EMVI, MAIN and,
especially, mucinous response. Larger-scale prospective studies in
patients with LARC are needed to evaluate the comparative
effectiveness of AB-MRI protocols and to assess how specialized
sequences could overcome MRI limitations in accurately assessing
treatment response.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
FT and YM confirm authenticity of all the raw data.
FT conceived the study and reviewed the manuscript. YM conceived
the study and analyzed data. NOM analyzed data. SR designed the
experiments and wrote the manuscript. MGG interpreted data. ET
performed experiments, interpreted data and wrote and reviewed the
manuscript. All authors have read and approved the manuscript.
Ethics approval and consent to
participate
The study was evaluated and approved by Erdogan
University Faculty of Medicine Non-interventional Clinical Research
Ethics Committee (approval no. 2022/134). The principles outlined
in the Declaration of Helsinki have been followed.
Patient consent for publication
Informed consent was obtained from the participants
whose images were used in the publication. Consent was obtained
before MRI and for use of images for study purposes.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Maheshwari E, Bajaj G, Jambhekar K, Pandey
T and Ram R: Magnetic Resonance Imaging of Rectal Cancer. J
Gastrointestinal Abdominal Radiol ISGAR. 2:18–32. 2019. View Article : Google Scholar
|
|
2
|
Zhao M, Zhao L, Yang H and Duan Y:
Apparent diffusion coefficient for the prediction of tumor response
to neoadjuvant chemo-radiotherapy in locally advanced rectal
cancer. Radiat Oncol. 16:172021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Heald RJ and Ryall RD: Recurrence and
survival after total mesorectal excision for rectal cancer. Lancet.
1:1479–1482. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Adjuvant therapy for patients with colon
and rectum cancer. Consens Statement. 8:1–25. 1990.
|
|
5
|
Sauer R, Becker H, Hohenberger W, Rödel C,
Wittekind C, Fietkau R, Martus P, Tschmelitsch J, Hager E, Hesss
CF, et al: Preoperative versus postoperative chemoradiotherapy for
rectal cancer. N Engl J Med. 351:1731–1740. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Haak HE, Maas M, Trebeschi S and Beets-Tan
RGH: Modern MR imaging technology in rectal cancer;there is more
than meets the eye. Front Oncol. 10:5375322020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Horvat N, Carlos Tavares Rocha C, Clemente
Oliveira B, Petkovska I and Gollub MJ: MRI of rectal cancer: Tumor
staging, imaging techniques, and management. Radiographics.
39:367–387. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Glimelius B, Tiret E, Cervantes A and
Arnold D; ESMO Guidelines Working Group, : Rectal cancer: ESMO
Clinical Practice Guidelines for diagnosis, treatment and
follow-up. Ann Oncol. 24 (Suppl 6):vi81–vi88. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Beets-Tan RGH, Lambregts DMJ, Maas M,
Bipat S, Barbaro B, Curvo-Semedo L, Fenlon HM, Gollub MJ,
Gourtsoyianni S, Halligan S, et al: Magnetic resonance imaging for
clinical management of rectal cancer: Updated recommendations from
the 2016 European Society of Gastrointestinal and Abdominal
Radiology (ESGAR) consensus meeting. Eur Radiol. 28:1465–1475.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Patel UB, Taylor F, Blomqvist L, George C,
Evans H, Tekkis P, Quirke P, Sebag-Montefiore D, Moran B, HAeld R,
et al: Magnetic resonance imaging-detected tumor response for
locally advanced rectal cancer predicts survival outcomes: MERCURY
experience. J Clin Oncol. 29:3753–3760. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Taylor FG, Swift RI, Blomqvist L and Brown
G: A systematic approach to the interpretation of preoperative
staging MRI for rectal cancer. AJR Am J Roentgenol. 191:1827–1835.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Seo N, Kim H, Cho MS and Lim JS: Response
assessment with MRI after Chemoradiotherapy in rectal cancer:
Current evidences. Korean J Radiol. 20:1003–1018. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Granata V, Bicchierai G, Fusco R, Cozzi D,
Grazzini G, Danti G, De Muzio F, Maggialetti N, Smorchkova O,
D'Elia M, et al: Diagnostic protocols in oncology: Workup and
treatment planning. Part 2: Abbreviated MR protocol. Eur Rev Med
Pharmacol Sci. 25:6499–6528. 2021.PubMed/NCBI
|
|
14
|
Fornell-Perez R, Vivas-Escalona V,
Aranda-Sanchez J, Gonzalez-Dominguez MC, Rubio-Garcia J,
Aleman-Flores P, Lozano-Rodriguez A, Porcel-de-Peralta G and
Loro-Ferrer JF: Primary and post-chemoradiotherapy MRI detection of
extramural venous invasion in rectal cancer: The role of
diffusion-weighted imaging. Radiol Med. 125:522–530. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lambregts DMJ, Boellaard TN and Beets-Tan
RGH: Response evaluation after neoadjuvant treatment for rectal
cancer using modern MR imaging: A pictorial review. Insights
Imaging. 10:152019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Dijkhoff RAP, Beets-Tan RGH, Lambregts
DMJ, Beets GL and Maas M: Value of DCE-MRI for staging and response
evaluation in rectal cancer: A systematic review. Eur J Radiol.
95:155–168. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Petrillo A, Fusco R, Granata V, Filice S,
Sansone M, Rega D, Delrio P, Bianco F, Romano GM, Tatangelo F, et
al: Assessing response to neo-adjuvant therapy in locally advanced
rectal cancer using Intra-voxel Incoherent Motion modelling by DWI
data and Standardized Index of Shape from DCE-MRI. Ther Adv Med
Oncol. 10:17588359188098752018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fusco R, Sansone M, Granata V, Grimm R,
Pace U, Delrio P, Tatangelo F, Botti G, Avallone A, Pecori B and
Petrillo A: Diffusion and perfusion MR parameters to assess
preoperative short-course radiotherapy response in locally advanced
rectal cancer: A comparative explorative study among Standardized
Index of Shape by DCEMRI, intravoxel incoherent motion- and
diffusion kurtosis imaging-derived parameters. Abdom Radiol (NY).
44:3683–3700. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fusco R, Granata V, Rega D, Russo C, Pace
U, Pecori B, Tatangelo F, Botti G, Izzo F, Cascella M, et al:
Morphological and functional features prognostic factor of magnetic
resonance imaging in locally advanced rectal cancer. Acta Radiol.
60:815–825. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Granata V, Grassi R, Fusco R, Izzo F,
Brunese L, Delrio P, Avallone A, Pecori B and Petrillo A: Current
status on response to treatment in locally advanced rectal cancer:
What the radiologist should know. Eur Rev Med Pharmacol Sci.
24:12050–12062. 2020.PubMed/NCBI
|
|
21
|
Patel UB, Blomqvist LK, Taylor F, George
G, Guthrie A, Bees N and Brown G: MRI After treatment of locally
advanced rectal cancer: How to report tumor response-the MERCURY
Experience. AJR Am J Roentgenol. 199:W486–W495. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kuhl CK: Abbreviated magnetic resonance
imaging (MRI) for breast cancer screening: Rationale, concept, and
transfer to clinical practice. Annu Rev Med. 70:501–519. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gao Y and Heller SL: Abbreviated and
ultrafast breast MRI in clinical practice. Radiographics.
40:1507–1527. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hötker AM, Vargas HA and Donati OF:
Abbreviated MR protocols in prostate MRI. Life (Basel).
12:5522022.PubMed/NCBI
|
|
25
|
Lee SL, Shin YR and Kim K: The added value
of pelvic surveillance by MRI during postoperative follow-up of
rectal cancer, with a focus on abbreviated MRI. Eur Radiol.
30:3113–3124. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Maas M, Nelemans PJ, Valentini V, Das P,
Rödel C, Kuo LJ, Calvo FA, Garcia-Aguilar J, Glynne-Jones R,
Haustermans K, et al: Long-term outcome in patients with a
pathological complete response after chemoradiation for rectal
cancer: A pooled analysis of individual patient data. Lancet Oncol.
11:835–844. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Garcia-Aguilar J, Marcet J, Coutsoftides
T, Cataldo P, Fichera A, Smith LE, Oomen S, Hunt SR, Herzig D,
Dietz D, et al: Impact of neoadjuvant chemotherapy following
chemoradiation on tumor response, adverse events, and surgical
complications in patients with advanced rectal cancer treated with
TME. J Clin Oncol. 29 (15 suppl):S35142011. View Article : Google Scholar
|
|
28
|
Jhaveri KS and Hosseini-Nik H: MRI of
rectal cancer: An overview and update on recent advances. AJR Am J
Roentgenol. 205:W42–W55. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jung SH, Heo SH, Kim JW, Jeong YY, Shin
SS, Soung MG, Kim HR and Kang HK: Predicting response to
neoadjuvant chemoradiation therapy in locally advanced rectal
cancer: Diffusion-weighted 3 Tesla MR imaging. J Magn Reson
Imaging. 35:110–116. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
van der Paardt MP, Zagers MB, Beets-Tan
RG, Stoker J and Bipat S: Patients who undergo preoperative
chemoradiotherapy for locally advanced rectal cancer restaged by
using diagnostic MR imaging: A systematic review and meta-analysis.
Radiology. 269:101–112. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Vliegen RF, Beets GL, von Meyenfeldt MF,
Kessels AG, Lemaire EE, van Engelshoven JM and Beets-Tan RG: Rectal
Cancer: MRI in local staging-is gadolinium-based contrast material
helpful? Radiology. 234:179–188. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jang JK, Lee JL, Park SH, Park HJ, Park
IJ, Kim JH, Choi SH, Kim J, Yu CS and Kim JC: Magnetic resonance
tumour regression grade and pathological correlates in patients
with rectal cancer. Br J Surg. 105:1671–1679. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Battersby NJ, Dattani M, Rao S, Cunningham
D, Tait D, Adams R, Moran BJ, Khakoo S, Tekkis P, Rasheed S, et al:
A rectal cancer feasibility study with an embedded phase III trial
design assessing magnetic resonance tumour regression grade (mrTRG)
as a novel biomarker to stratify management by good and poor
response to chemoradiotherapy (TRIGGER): Study protocol for a
randomised controlled trial. Trials. 18:3942017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lambregts DM, Rao SX, Sassen S, Martens
MH, Heijnen LA, Buijsen J, Sosef M, Beets GL, Vliegen RA and
Beets-Tan RGH: MRI and Diffusion-weighted MRI volumetry for
identification of complete tumor responders after preoperative
chemoradiotherapy in patients with rectal cancer: A
Bi-institutional validation study. Ann Surg. 262:1034–1039. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Vliegen RF, Beets GL, Lammering G, Dresen
RC, Rutten HJ, Kessels AG, Oei TK, de Bruine PA, van Engelshoven JM
and Beets-Tan RGH: Mesorectal fascia invasion after neoadjuvant
chemotherapy and radiation therapy for locally advanced rectal
cancer: Accuracy of MR imaging for prediction. Radiology.
246:454–462. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Park SH, Lim JS, Lee J, Kim HY, Koom WS,
Hur H, Park MS, Kim MJ and Kim H: Rectal mucinous adenocarcinoma:
MR imaging assessment of response to concurrent chemotherapy and
radiation Therapy-A hypothesis-generating study. Radiology.
285:124–133. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shia J, McManus M, Guillem JG, Leibold T,
Zhou Q, Tang LH, Riedel ER, Weiser MR, Paty PB, Temple LK, et al:
Significance of acellular mucin pools in rectal carcinoma after
neoadjuvant chemoradiotherapy. Am J Surg Pathol. 35:127–134. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kaur H, Choi H, You YN, Rauch GM, Jensen
CT, Hou P, Chang GJ, Skibber JM and Ernst RD: MRI for preoperative
evaluation of primary rectal cancer: Practical considerations.
Radiographics. 32:389–409. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sauer R, Liersch T, Merkel S, Fietkau R,
Hohenberger W, Hess C, Becker H, Raab HR, Villanueva MT, Witzigmann
H, et al: Preoperative versus postoperative chemoradiotherapy for
locally advanced rectal cancer: Results of the German
CAO/ARO/AIO-94 randomized phase III trial after a median followup
of 11 years. J Clin Oncol. 30:1926–1933. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
MERCURY Study Group, : Diagnostic accuracy
of preoperative magnetic resonance imaging in predicting curative
resection of rectal cancer: Prospective observational study. BMJ.
333:7792006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kulkarni T, Gollins S, Maw A, Hobson P,
Byrne R and Widdowson D: Magnetic resonance imaging in rectal
cancer downstaged using neoadjuvant chemoradiation: Accuracy of
prediction of tumour stage and circumferential resection margin
status. Colorectal Dis. 10:479–489. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kim KH, Park MJ, Lim JS, Kim NK, Min BS,
Ahn JB, Kim TI, Kim HG and Koom WS: Circumferential resection
margin positivity after preoperative chemoradiotherapy based on
magnetic resonance imaging for locally advanced rectal cancer:
Implication of boost radiotherapy to the involved mesorectal
fascia. Jpn J Clin Oncol. 46:316–322. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Park MJ, Kim SH, Lee SJ, Jang KM and Rhim
H: Locally advanced rectal cancer: Added value of
diffusion-weighted MR imaging for predicting tumor clearance of the
mesorectal fascia after neoadjuvant chemotherapy and radiation
therapy. Radiology. 260:771–780. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
van Heeswijk MM, Lambregts DM, Palm WM,
Henddriks BM, Mass M, Beets GL and Beets-Tan RG: DWI for assessment
of rectal cancer nodes after chemoradiotherapy: Is the Absence of
Nodes at DWI proof of a negative nodal status? AJR Am J Roentgenol.
208:W79–W84. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chand M, Evans J, Swift RI, Tekkis PP,
West NP, Stamp G, Heald RJ and Brown G: The prognostic significance
of postchemoradiotherapy High-resolution MRI and histopathology
detected extramural venous invasion in rectal cancer. Ann Surg.
261:473–479. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Tan JJ, Carten RV, Babiker A, Abulafi M,
Lord AC and Brown G: Prognostic Importance of MRI-Detected
extramural venous invasion in rectal cancer: A literature review
and systematic meta-analysis. Int J Radiat Oncol Biol Phys.
111:385–394. 2021. View Article : Google Scholar : PubMed/NCBI
|