Introduction
Uterine corpus endometrial carcinoma (UCEC) is a
prevalent form of cancer in women, and its incidence is rapidly
increasing (estimated annual percentage change, 0.69% per year)
while its mortality is decreasing (estimated annual percentage
change, 0.85% per year) worldwide in recent years (1). UCEC can be broadly classified into two
primary subtypes: Type I, which is hormone-driven and generally
associated with a favorable prognosis; and type II, which is
hormone-independent and typically characterized by a poorer
prognosis (2). In China, the
majority of newly diagnosed UCEC cases (80–90%) fall under type I
classification (3). A notable
proportion of UCEC type I cases are detected at an early stage,
leading to favorable outcomes with standard surgical intervention
and subsequent chemotherapy; the 5-year relative survival rate for
such patients is ~80% (4,5). However, certain patients experiencing
recurrent UCEC may not achieve satisfactory therapeutic responses
from existing treatment modalities. The unfavorable outcome of
these individuals is primarily attributed to the malignant growth
and distant metastasis (6,7). Therefore, comprehending the underlying
mechanisms governing malignant behaviors in UCEC is of
importance.
The interaction between cells serves a significant
role in influencing the progression of tumors, and several studies
have demonstrated its direct impact on tumor metastasis (8,9).
Communication among cells within the microenvironment can be
facilitated through extracellular exosomes (10). Extracellular membranous vesicles
known as exosomes, ranging from 30 to 150 nm in size, consist of
proteins, lipids, DNAs and various forms of RNAs (11). Following fusion with the cytoplasmic
membrane, exosomes are released into the extracellular space by
most cell types (12).
Subsequently, recipient cells internalize these exosomes for
modulation purposes. Previous studies suggest that the
intercellular transfer of microRNA (miRNA) via exosomes serves a
pivotal role in modulating the expression levels of multiple target
genes within recipient cells, thereby endowing exosomes with the
ability to regulate tumor cell proliferation, migration and
invasion (13,14). Recently, numerous exosomal miRNAs,
such as exosomal miR-27a-5p (15)
and miR-499a-5p (16), have been
reported to mediate the development of UCEC. miR-32-5p has been
implicated as an oncogenic factor in pancreatic (17), colorectal (18) and gynecological cancer, such as
ovarian cancer (19). However, its
role in UCEC progression remains elusive. Furthermore, it was
recently demonstrated that serum exosomal miR-32-5p holds potential
as a diagnostic biomarker for coronary artery disease (20). Therefore, the impact of exosomal
miR-32-5p on the progression of UCEC and its underlying mechanism
has gained notable interest in current research.
The present study aimed to investigate the
expression of miR-32-5p in UCEC, and perform preliminary
investigations into the role and mechanism of exosomal miR-32-5p
during the progression of UCEC.
Materials and methods
Data acquisition
miRNA expression data in UCEC were downloaded from
The Cancer Genome Atlas (TCGA) database (https://portal.gdc.cancer.gov/). A cohort comprising
578 samples were analyzed, with 33 categorized as normal and 545 as
UCEC tumor samples. Additionally, 49 DE-miRNAs previously
identified by Zhou et al (21) from the comparison of 56 plasma
samples from patients with endometrial cancer and healthy controls
were analyzed in the present study.
Screening of differentially expressed
(DE) miRNAs
The Limma package (version 3.52.0; Bioconductor) was
employed for the analysis of DE-miRNAs between UCEC and normal
samples. miRNAs with a false discovery rate ≤0.05,
|log2fold change|≥1.2 and P-value <0.01 were
considered to be statistically significant. The 49 DE-miRNAs
identified from analysis by Zhou et al (21) were included in the present study
analysis.
Patient samples
From October 2023 to April 2024, a total of 9
patients (age, 44–72 years; mean age, 57.56±8.93 years) diagnosed
with UCEC via pathological examination were enrolled in Renmin
Hospital of Wuhan University (Wuhan, China). Inclusion criteria:
Patients had been admitted to the hospital for the first time and
did not receive any malignant tumor treatments, such as
chemotherapy or radiotherapy before surgery; postoperative tissues
were diagnosed by pathologists as UCEC with parallel International
Federation of Gynecology and Obstetrics (FIGO) staging; patients
were aware of and agreed to the present study process; and patient
clinical data and follow-up data were complete and available.
Exclusion criteria: Patients with other malignant tumors; patients
with immune system diseases or infectious diseases; and patients
with severe impairment of the heart, liver, kidney and other organ
functions. According to the FIGO staging system (22), the tumor stages of the included
patients were as follows: Stage I, 1; stage III, 1; and stage IV, 7
cases. A total of 9 sex and age-matched healthy individuals were
recruited as normal controls (age, 41–70 years; mean age,
55.42±7.28 years). Peripheral blood samples (10 ml) from both
patients with UCEC and healthy individuals were collected, followed
by centrifugation at 3,000 × g for 10 min at 4°C within 4 h to
obtain plasma supernatant. The collected plasma samples were
further subjected to centrifugation at 16,000 × g for 10 min at 4°C
and subsequently stored at −80°C until further use. The present
study adhered to the principles outlined in the Declaration of
Helsinki, and ethical approval was obtained from Renmin Hospital of
Wuhan University Ethics Committee (approval no. WDRY2023-K161). All
participants signed a written informed consent form.
Cell culture
Procell Life Science & Technology, Ltd. provided
two UCEC cell lines (HEC-1-A and Ishikawa). HEC-1-A and Ishikawa
cells were grown in RPMI-1640 medium (Gibco; Thermo Fisher
Scientific, Inc.) with 10% FBS (Gibco; Thermo Fisher Scientific,
Inc.) and 1% penicillin/streptomycin (Invitrogen; Thermo Fisher
Scientific, Inc.), with an environment with 5% CO2 and a
temperature of 37°C. Cells in the logarithmic growth phase were
used for the subsequent experiments.
Cell transfection
miR-32-5p-inhibitor, miR-32-5p inhibitor-negative
control (NC), overexpression (OE)-Forkhead Box N2 (FOXN2), OE-NC,
miR-32-5p-mimic and miR-32-5p-mimic-NC were synthesized by Ribo
Biotech, Ltd. The vector used for the overexpression transfections
was pcDNA™3.1(+) (Invitrogen; Thermo Fisher
Scientific, Inc.). Lipofectamine® 3000 (Invitrogen;
Thermo Fisher Scientific, Inc.), HEC-1-A cells were transfected
with miR-32-5p-inhibitor or miR-32-5p inhibitor-NC, while Ishikawa
cells were transfected with miR-32-5p-mimic or miR-32-5p-mimic-NC.
The concentration of nucleic acid used was 50 nM; the transfection
process lasted for 48 h at 37°C, after which the cells were
collected further functional experiments. The sequences of the
miRNA mimic and inhibitors as well as those of the negative
controls are listed in Table I.
 | Table I.Sequences of the miRNA mimic and
inhibitor. |
Table I.
Sequences of the miRNA mimic and
inhibitor.
| Gene | Sequences
(5′-3′) |
|---|
|
miR-32-5p-mimic | S:
UAUUGCACAUUACUAAGUUGCA |
|
| AS:
CAACUUAGUAAUGUGCAAUAUU |
|
miR-32-5p-mimic-NC | S:
UUCUCCGAACGUGUCACGU |
|
| AS:
GUGACACGUUCGGAGAAUU |
|
miR-32-5p-inhibitor |
UGCAACUUAGUAAUGUGCAAUA |
|
miR-32-5p-inhibitor-NC |
ACGUGACACGUUCGGAGAA |
Exosome isolation and
characterization
Exosomes from UCEC plasma samples (UCEC-Exo) and
normal controls (Normal-Exo) were isolated using an
ExoQuick™ Exosome Precipitation Kit (System Biosciences,
LLC) according to the manufacturer's instructions. Briefly, plasma
samples (500 µl) were combined with exosome precipitation reagent
(100 µl) and incubated at room temperature for 10 min. The mixture
was then subjected to centrifugation at a speed of 10,000 × g for a
duration of 10 min at 4°C. The resulting pellet containing the
exosomes was subsequently resuspended in PBS for subsequent
analysis.
To isolate exosomes from the UCEC cell lines
(HEC-1-A-Exo and Ishikawa-Exo), HEC-1-A or Ishikawa cells were
cultured in RPMI-1640 medium supplemented with 10% FBS, at a
temperature of 37°C at 5% CO2. Following a culture
period of 72 h, the supernatant containing exosomes and cellular
debris was separated using centrifugation (10,000 × g for 30 min at
4°C). Exosomes from the collected supernatant were extracted using
the GM™ Exosome Isolation Reagent kit (Guangzhou
Geneseed Biotech. Co., Ltd.). The resulting mixture was incubated
at a temperature of 4°C for 30 min before centrifugation at 2,000 ×
g for 30 min. Finally, the isolated exosomes were PBS-resuspended
for further analysis.
Exosome structure was analyzed using transmission
electron microscopy (TEM; Hitachi, Ltd.) as previous described
(23). Nanoparticle tracking
analysis (NTA) was conducted using the NanoSight NS300 (Malvern
Panalytical, Ltd.) to analyze the size distribution of the isolated
exosomes. Western blotting was utilized to identify the presence of
exosomal indicators, including CD9, CD81, tumor susceptibility gene
101 (TSG101) and calnexin.
Co-culture system construction
The HEC-1-A donor cells (transfected with
miR-32-5p-inhibitor-NC or miR-32-5p inhibitor) or Ishikawa donor
cells (transfected with miR-32-5p-mimic-NC or miR-32-5p mimic) were
seeded on a Transwell polyester permeable support. Simultaneously,
the receptor HEC-1-A or Ishikawa cells (both untransfected) were
cultured in the lower chamber of Transwell culture plate. After a
24 h incubation, the receptor cells were collected for subsequent
experiments.
Quantitative reverse transcription-PCR
(qRT-PCR)
TRIzol (Invitrogen; Thermo Fisher Scientific, Inc.)
was used for RNA isolation from UCEC cell lines according to the
manufacturer's instructions. The Multi-type Sample DNA/RNA
Extraction-Purification Kit (Sansure Biotech, Inc.) was used to
isolate RNA from exosomes according to the manufacturer's
instructions. The RNA concentration was measured using a Nanodrop
spectrophotometer (Thermo Fisher Scientific, Inc.). The optical
density 260/280 nm ratios of all RNA samples were ≥1.8. cDNA was
obtained using the ReverTra Ace qPCR RT Kit (Toyobo Co., Ltd.)
according to the manufacturer's instructions and subject to PCR
analysis using a SYBR High-Sensitivity qPCR Supermix Kit
(Novoprotein Scientific, Inc.) with the Real-time PCR System
(Applied Biosystems; Thermo Fisher Scientific, Inc.). The following
thermocycling conditions were used for the PCR: Initial
denaturation at 95°C for 10 min; 40 cycles of 95°C for 15 sec and
60°C for 30 sec. The 2−ΔΔCq values (24) reflected the RNA expression levels
using U6 (for miR-32-5p) (25,26)
and GAPDH (for AKT, PI3K, Bcl-2 and FOXN2) as controls. qPCR
primers used are shown in Table
II.
 | Table II.PCR primer list. |
Table II.
PCR primer list.
| Gene | Sequences
(5′-3′) | Accession no. |
|---|
| miR-32-5p | F:
ACACTCCAGCTGGGTATTGCACATTACTAA | NC_000009.12 |
|
| R:
TGGTGTCGTGGAGTCG |
|
| miR-32-5p stem-loop
primera |
CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGTGCAACTT |
|
| U6 | F:
CTCGCTTCGGCAGCACA | NC_000015.10 |
|
| R:
AACGCTTCACGAATTGTGCGT |
|
| AKT | F:
AGAAGCAGGAGGAGGAGGAG | NM_005163.2 |
|
| R:
TCTCCTTCACCAGGATCACC |
|
| PI3K | F:
CCACGACCATCATCAGGTGAA | NM_006218.4 |
|
| R:
CCTCACGGAGGCATTCTAAAGT |
|
| Bcl-2 | F:
GAGGATTGTGGCCTTCTTTG | NM_000633.3 |
|
| R:
ACAGTTCCACAAAGGCATCC |
|
| FOXN2 | F:
CCAGGTCTAGCGTGTCTTCC | NM_001375442.1 |
|
| R:
AGCCACTGTCTCCAAGAGGA |
|
| GAPDH | F:
ACAACTTTGGTATCGTGGAAGG | NM_002046.7 |
|
| R:
GCCATCACGCCACAGTTTC |
|
Cell proliferation assays
The viability of UCEC cells was measured via CCK-8
assay. HEC-1-A or Ishikawa cells (3×103 cells/ml) were
grown in 96-well plates for 0, 24, 48 and 72 h before adding the
CCK-8 solution (15 µl; Dojindo Laboratories, Inc.). Following a 2 h
incubation, UCEC cell viability was calculated via microplate
reader (Bio-Rad Laboratories, Inc.), with the absorbance at a
wavelength of 450 nm.
Colony forming experiments were carried out to
further estimate the proliferative capacities. HEC-1-A or Ishikawa
cells (~1,000 cells) were seeded into each well of 6-well plates
and cultured for 2 weeks. The culture medium was refreshed
biweekly. Following that, the cells were rinsed with PBS, treated
with 100 % methanol for fixation at 25°C for 10 min and
subsequently dyed with 0.1% crystal violet (Sigma-Adrich; Merck
KGaA) for a duration of 20 min at 25°C. Cells were then washed
three times, and cell colonies were counted using a light
microscope (Olympus Corporation).
The 5-ethynyl-2′-deoxyuridine (EdU) proliferation
assay Kit (Abcam) was used according to the manufacturer's
instructions. HEC-1-A or Ishikawa cells were initially incubated
with 50 µM EdU at 37°C for 2 h. Fixation was performed using 4%
formaldehyde at 25°C for 15 min and permeabilized with 0.5% Triton
X-100 at 25°C for 20 min. Following this, the cells were incubated
with an Apollo reaction cocktail (1X; Abcam) at room temperature
for ~30 min. To visualize DNA, cells were stained with DAPI at 25°C
for 30 min. A fluorescence microscope (Carl Zeiss AG) was utilized
to observe the EdU-positive cells.
Transwell migration assay
The upper chamber of the Transwell insert was
pre-coated with 50 µl of Matrigel at 37°C for 30 min, which had
been diluted 5-fold in serum-free RPMI-1640. HEC-1-A or Ishikawa
cells (5×104 cells) were suspended in a RPMI-1640 medium
without serum and then placed into the upper chamber.
Simultaneously, the lower chamber was supplemented with RPMI-1640
medium containing 10% FBS. Following overnight incubation at 37°C,
the cells in the lower chamber were treated with 0.1% crystal
violet at 37°C for 15 min. Images of the stained cells were
captured using a light microscope (magnification, ×400).
Flow cytometry analysis
The apoptotic UCEC cells were assessed by utilizing
the Annexin V-FITC apoptosis detection kit (Thermo Fisher
Scientific, Inc.) following the guidelines provided by the
manufacturer. The HEC-1-A or Ishikawa cells (2×105) were
suspended in 500 µl of binding buffer and then treated with Annexin
V-EGFP and PI (5 µl each) at a temperature of 4°C for a duration of
15 min in the absence of light. Thereafter, the apoptosis of the
cells was evaluated using a FACScan flow cytometer (Becton,
Dickinson and Company) and analyzed using the BD CellQuest software
(version 3.3; Becton, Dickinson and Company).
Target prediction
The mRNA targets of miR-32-5p were predicted using
the StarBase software (version 2.0; http://starbase.sysu.edu.cn/), and 2,143 targets were
predicted. FOXN2 was selected for the subsequent experiments due to
its key role in endometrial cancer (27) and unknown regulatory relationship
with miR-32-5p.
Dual luciferase reporter (DLR)
assay
The 3′-untranslated region (UTR) of FOXN2 containing
the putative binding sites of miR-32-5p was cloned into the
luciferase reporter plasmid pGL3 vector (Promega Corporation) to
construct the FOXN2 wild-type (WT)/mutant type (MUT). Ishikawa
cells (5×103) were seeded onto a 24-well plate and
co-transfected with one of the aforementioned plasmids (80 ng),
along with either miR-32-5p mimic or mimic-NC (10 nM), using
Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.). After 48 h at 37°C, the relative luciferase
activity was determined using a Dual-Luciferase Reporter Assay
System Kit (Promega Corporation). The activity of firefly
luciferase was normalized to that of Renilla luciferase.
Western blotting
RIPA lysis buffer (Beyotime Institute of
Biotechnology) containing protease inhibitors was utilized for
protein extraction from cells, and protein concentrations were
determined using the BCA Protein Assay Kit (Beyotime Institute of
Biotechnology). In each lane, ~30 µg of proteins was separated
using 10% SDS-PAGE and transferred onto a PVDF membrane. Blocking
of the membrane was carried out at room temperature using 5% bovine
serum albumin (Beyotime Institute of Biotechnology) for 2 h at
25°C. Afterward, the membrane was placed in a 4°C environment and
incubated overnight with primary antibodies against AKT (1:1,500;
cat. no. #9272; Cell Signaling Technology), p-AKT (1:1,500; cat.
no. #4060; Cell Signaling Technology), PI3K (1:1,500; cat. no.
#4249; Cell Signaling Technology), Bcl-2 (1:1,500; cat. no. #3498;
Cell Signaling Technology), FOXN2 (1:1,500; cat. no. ab236385;
Abcam), GAPDH (1:1,500; cat. no. #2118; Cell Signaling Technology),
Calnexin (1:1,500; cat. no. #2679; Cell Signaling Technology), CD9
(1:1,500; cat. no. #13174; Cell Signaling Technology), CD81
(1:1,500; cat. no. ab79559; Abcam) and TSG101 (1:1,500; cat. no.
ab125011; Abcam). Subsequently, the membranes were washed three
times using tris-buffered saline with 0.05% Tween 20. Then, the
HRP-conjugated secondary antibody (1:3,000; cat. no. #7074; Cell
Signaling Technology) was added at room temperature for 1 h. The
reference gene used was GAPDH. The immunoreactive protein bands
were observed using an ECL Basic Kit (ABclonal Biotech Co., Ltd.)
using a Gel-Pro analyzer (version 4.0; Media Cybernetics,
Inc.).
Statistical analysis
Data analysis was conducted utilizing SPSS software
(version 20.0; IBM Corp.). The findings were presented as the mean
value ± standard deviation. Student's t-test (unpaired) or one-way
ANOVA with Tukey's post hoc test was used for comparisons.
Multivariate Cox regression analysis was performed on DE-miRNAs
utilizing the Survival package in R (Posit Software, PBC). The
median was selected as the cut-off value, and Kaplan-Meier survival
curves were generated using the Survminer package (version 0.5.0;
DataNovia) and analyzed using log-rank test. P<0.05 was
considered to indicate a statistically significant difference.
Results
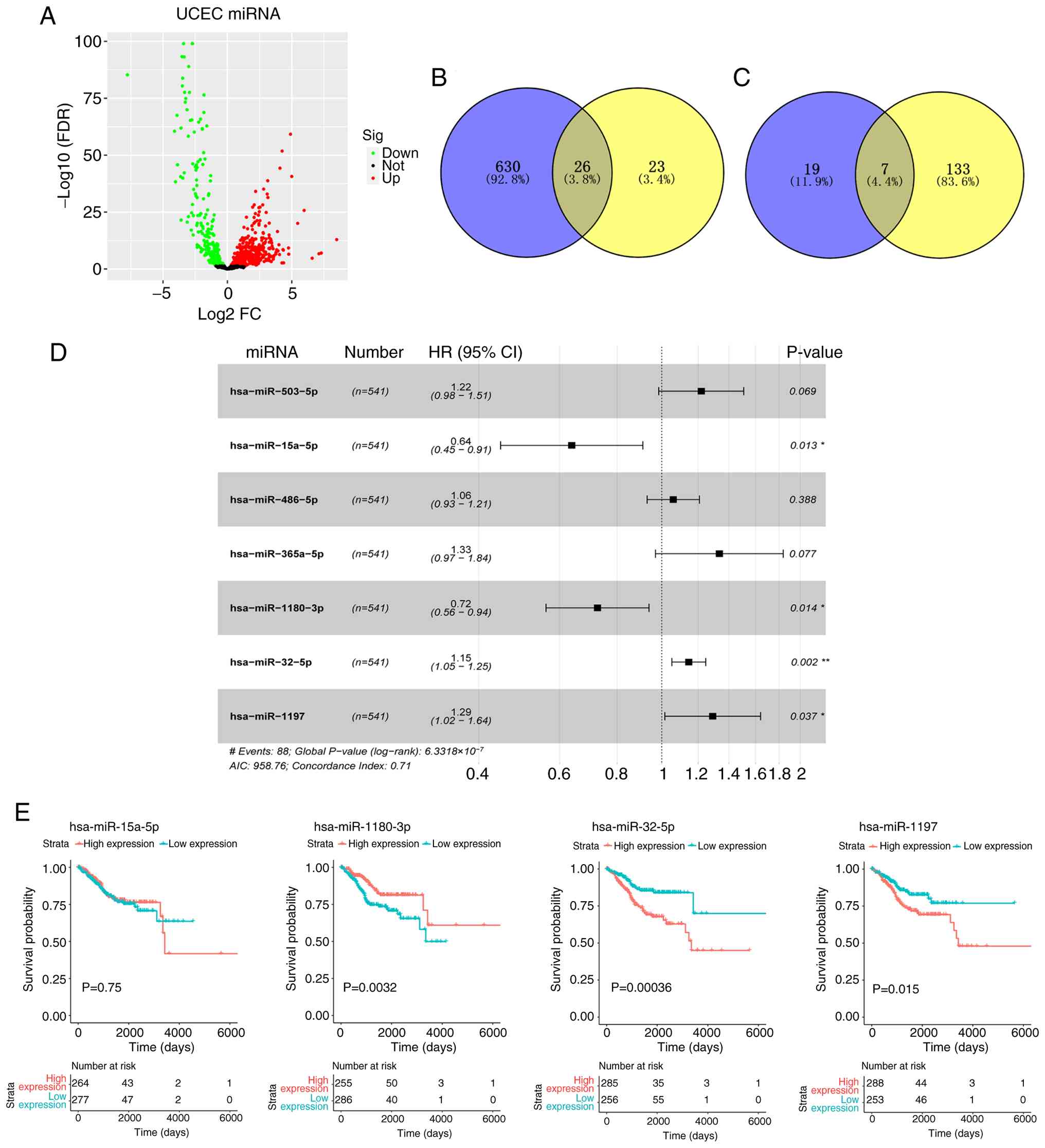
miRNA screening
The present study screened miRNA expression data
from patients with UCEC of TCGA database and DE-miRNA data from
Zhou et al (21). Compared
with that in normal samples, there were 656 miRNAs differentially
expressed in UCEC tissues from TCGA database (Fig. 1A). Among them, 402 miRNAs were
differentially upregulated, while 254 miRNAs were differentially
downregulated in UCEC. The heatmap of top 50 DE-miRNAs is shown in
Fig. S1. Through intersecting
these 656 DE-miRNAs with DE-miRNA data (comprising 49 DE-miRNAs)
from Zhou et al (21), a
total of 26 DE-miRNAs were obtained (Fig. 1B). Univariate Cox risk regression
analysis (P<0.05) was conducted on the TCGA data, and 140 miRNAs
were identified as potential prognostic miRNAs in patients with
UCEC. These 140 miRNAs were then intersected with 26 DE-miRNAs from
the aforementioned analysis, from which 7 overlapping DE-miRNAs
were identified (Fig. 1C).
Multivariate Cox regression analysis was used to demonstrate that
miR-15a-5p (P=0.013), miR-1180-3p (P=0.014), miR-32-5p (P=0.002)
and miR-1197 (P=0.037) were independent prognostic factors of UCEC
(Fig. 1D). Survival probability
analysis was conducted to investigate the effects of the expression
of these four miRNAs on survival outcomes, from which miR-32-5p
demonstrated the most significant effects on survival probability
and was therefore selected for further investigation (P=0.00036;
Fig. 1E).
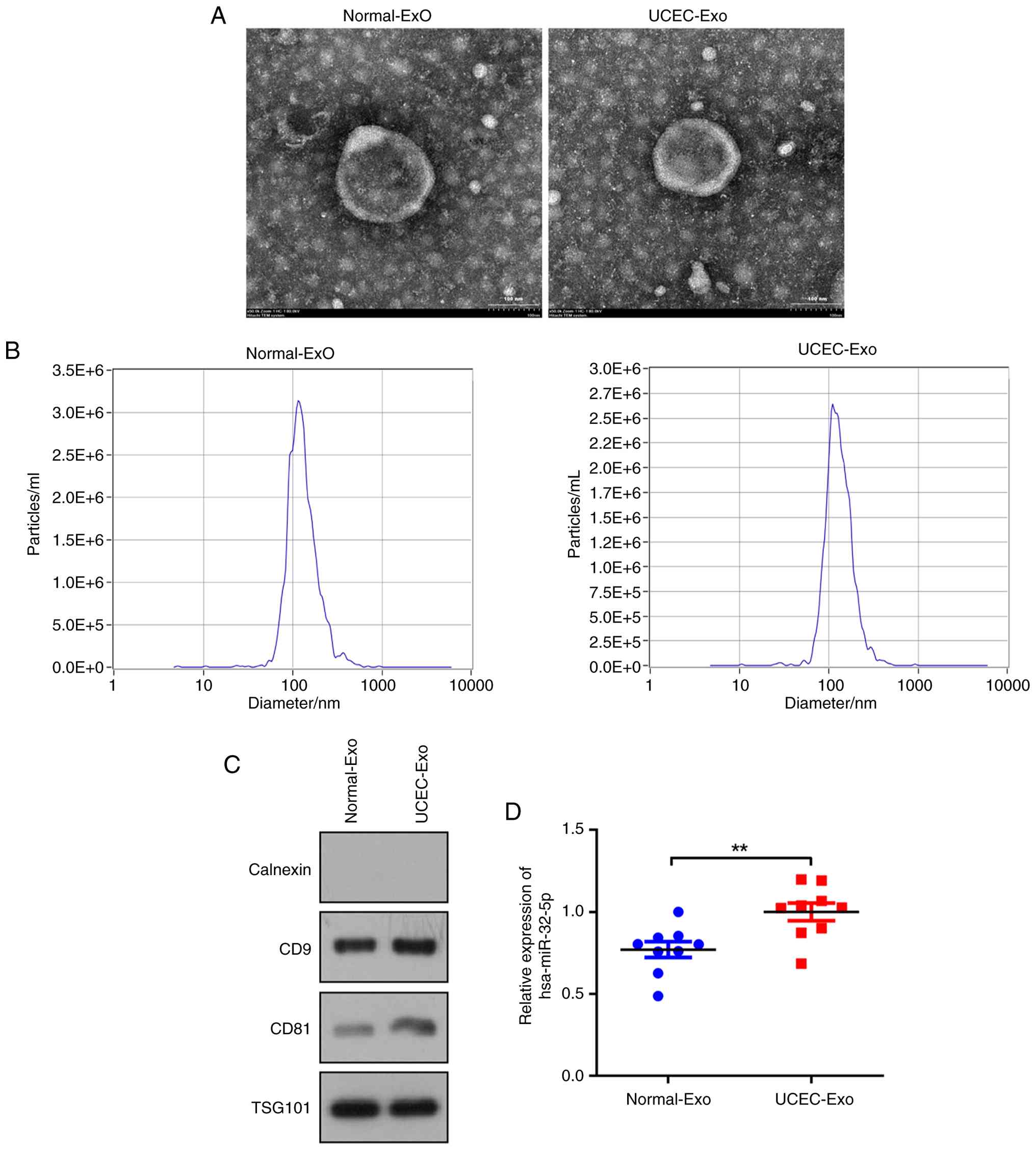
miR-32-5p is overexpressed in
UCEC-Exo
To confirm the successful isolation of exosomes from
plasma samples from patients with UCEC and healthy controls, TEM
and NTA were employed to characterize the isolated exosomes. The
isolated vesicles exhibited a double-layered membrane structure and
a size distribution of ~100 nm in diameter, which were in
accordance with the expected characteristics of exosomes (Fig. 2A and B) (28). Western blotting demonstrated that
the vesicles expressed the exosome-positive markers CD9, CD81 and
TSG101, while the exosome-negative marker, Calnexin, was absent in
the isolated vesicles (Fig. 2C),
which demonstrated successful isolation of exosomes. The expression
of miR-32-5p in normal-Exo and UCEC-Exo was then determined;
miR-32-5p expression was upregulated in UCEC-Exo compared with that
of normal-Exo (P<0.05; Fig.
2D).
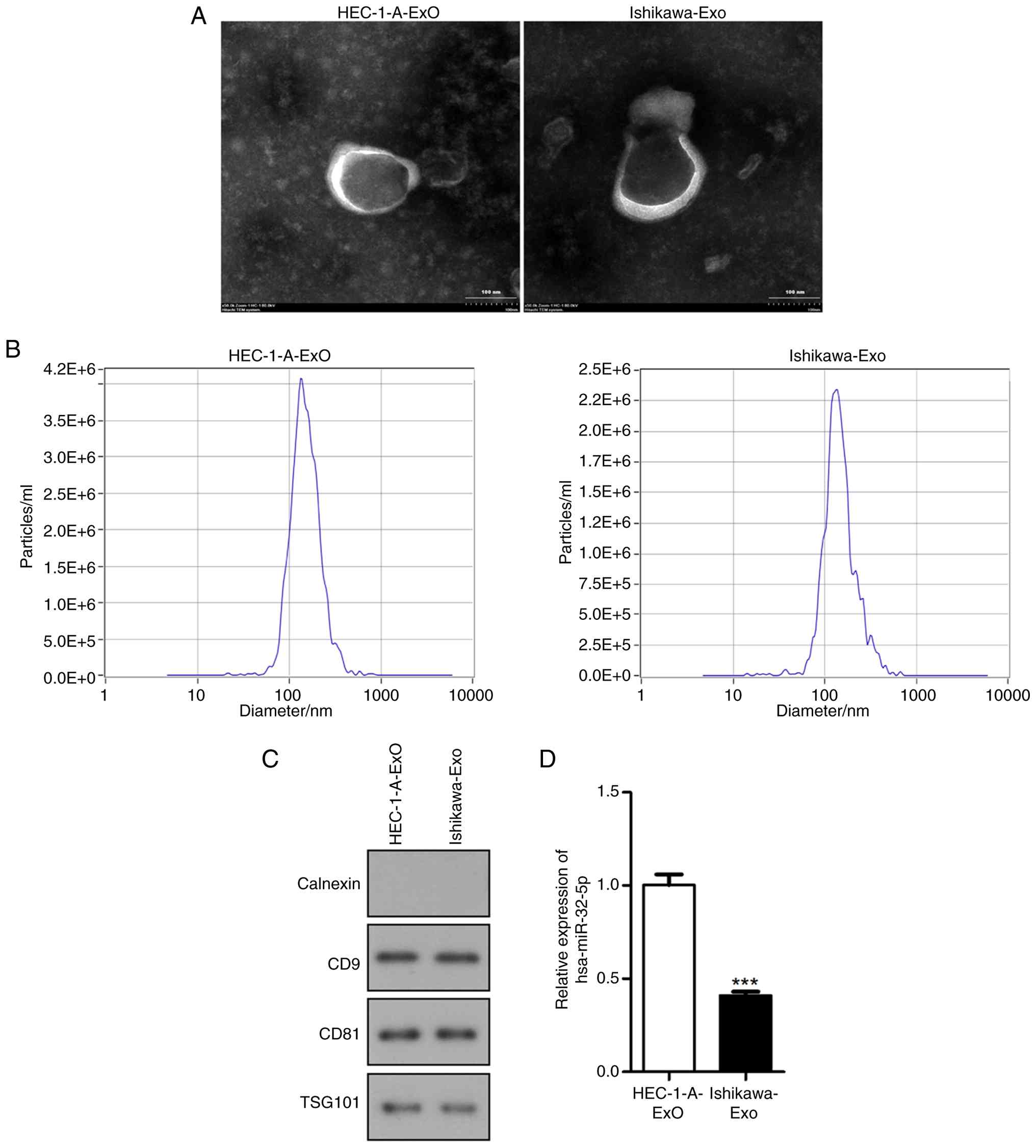
Expression of miR-32-5p in exosomes
isolated from UCEC cell lines
Subsequently, exosomes were isolated from two UCEC
cell lines (HEC-1-A and Ishikawa). The characteristics of vesicles
from UCEC cell lines were similar to exosomes isolated from plasma
samples (Fig. 3A and B). Western
blotting demonstrated presence of exosome-positive markers CD9,
CD81 and TSG101 in the exosomes-enriched fractions, and absence of
Calnexin (Fig. 3C). Additionally,
compared with that of HEC-1-A-Exo, miR-32-5p expression was
downregulated in Ishikawa-Exo (P<0.001; Fig. 3D).
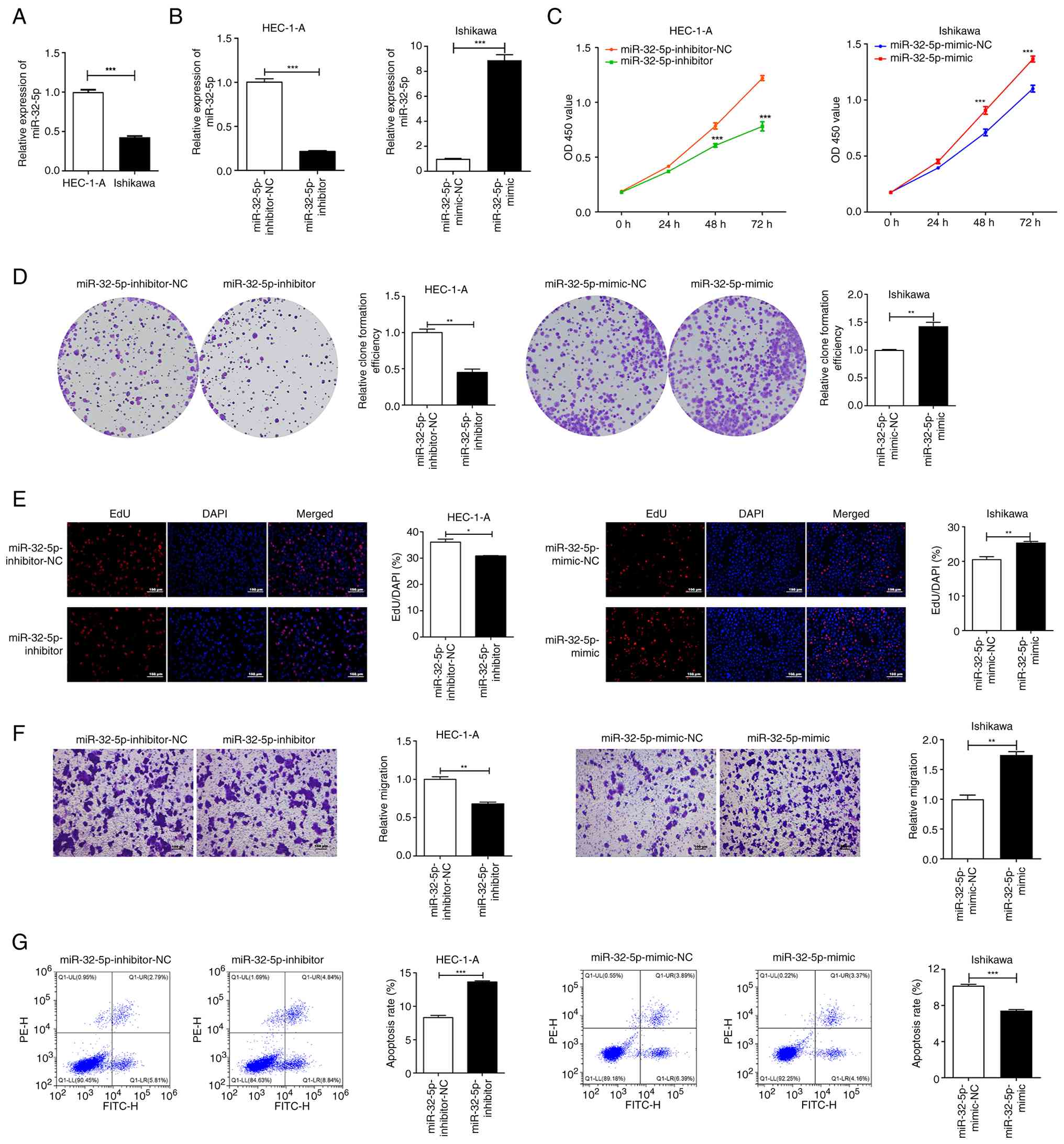
miR-32-5p affects the proliferative
capacities, migratory abilities and apoptotic rate of UCEC
cells
miR-32-5p expression levels were significantly
reduced in Ishikawa cells compared with that of HEC-1-A cells
(P<0.001; Fig. 4A). As miR-32-5p
demonstrated high expression levels in HEC-1-A cells, they were
selected for transfection with the miR-32-5p inhibitor to achieve
interference in miR-32-5p expression, whereas the Ishikawa cells
were chosen for transfection with the miR-32-5p mimic to achieve
overexpression, due to the low expression levels of miR-32-5p in
Ishikawa cells. qRT-PCR results that miR-32-5p expression in
HEC-1-A cells was significantly decreased following transfection
with the miR-32-5p inhibitor (P<0.001), while miR-32-5p was
overexpressed in Ishikawa cells upon transfection with the
miR-32-5p mimic (Fig. 4B;
P<0.001). miR-32-5p inhibition significantly suppressed the
viability of HEC-1-A cells, as evidenced by a CCK-8 assay (Fig. 4C; P<0.001). Conversely,
overexpression of miR-32-5p increased the cell viability of
Ishikawa cells (Fig. 4C;
P<0.001). Colony formation and EdU proliferation assays further
confirmed the impacts of miR-32-5p expression status on cell
proliferation. Transfection with miR-32-5p inhibitor led to a
significant decrease in colony formation efficiency and the
percentages of EdU-positive cells in HEC-1-A cells (Fig. 4D and E; P<0.05), while
transfection with miR-32-5p mimic significantly increased colony
formation efficiency and EdU-positive cell percentages in Ishikawa
cells (P<0.01). The migratory abilities of UCEC cell were
assessed using a Transwell assay, demonstrating that downregulation
of miR-32-5p significantly suppressed HEC-1-A cell migration
(Fig. 4F; P<0.01), whereas
miR-32-5p overexpression significantly increased the migratory
capacities of Ishikawa cells (P<0.01). Furthermore, flow
cytometry analysis demonstrated increased apoptosis in HEC-1-A
cells upon treatment with miR-32-5p inhibitor (Fig. 4G; P<0.001), while Ishikawa cells
exhibited a significantly reduced apoptotic rate following
treatment with miR-32-5p mimic (P<0.001).
miR-32-5p negatively regulates FOXN2
and activates PI3K/AKT/Bcl-2 pathway
The Starbase software predicted a potential binding
site between miR-32-5p and FOXN2 (Fig.
5A). The DLR assay was conducted in Ishikawa cells, which
demonstrated a significant decrease in relative luciferase activity
in the FOXN2-WT + miR-32-5p mimic group when compared with that of
the FOXN2-WT + mimic-NC group (Fig.
5B; P<0.001), while no statistical changes were observed
between the FOXN2-MUT + miR-32-5p mimic and FOXN2-MUT + mimic-NC
groups. These findings suggested that FOXN2 was a direct target of
miR-32-5p. The PI3K/AKT pathway is a well-established mechanism
implicated in the promotion of cancer development (29,30),
including in UCEC (31), but
whether it is regulated by miR-32-5p in UCEC remains unknown.
Therefore, we further explored the effects of miR-32-5p on PI3K/AKT
pathway in UCEC cells. Inhibition of miR-32-5p resulted in a
significant inhibition of AKT and PI3K mRNA expression (Fig. 5C; P<0.001). Additionally,
significantly decreased Bcl-2 mRNA expression levels (P<0.05)
and increased FOXN2 mRNA expression levels (P<0.01) were
observed upon miR-32-5p inhibition. The opposite effects were
observed in the results of mRNA expression of AKT, PI3K, Bcl-2 and
FOXN2 in Ishikawa cells following transfection of miR-32-5p mimic
(Fig. 5D; P<0.05). Western
blotting was used to assess protein levels in HEC-1-A and Ishikawa
cells. The expression levels of AKT protein were unaltered by both
miR-32-5p overexpression and inhibition (Fig. 5E and F). When transfected with
miR-32-5p inhibitor in HEC-1-A cells, the protein expression levels
of p-AKT, PI3K and Bcl-2 were significantly decreased (P<0.05),
but FOXN2 protein expression levels were increased (P<0.001).
The ratio of p-AKT/AKT was significantly decreased in HEC-1-A cells
transfected with miR-32-5p-inhibitor (P<0.001). However,
overexpression with miR-32-5p mimic showed the opposite effects
(P<0.05). These findings suggested that miR-32-5p negatively
regulated FOXN2 and activated PI3K/AKT/Bcl-2 pathway. Rescue
experiments were performed in Ishikawa cells to further verify the
effects of miR-32-5p-mediated FOXN2 on PI3K/AKT/Bcl-2 pathway. mRNA
expression of FOXN2 was significantly increased followed
transfection of OE-FOXN2 (Fig. 6A;
P<0.0001), which demonstrated the successful transfection.
Overexpression of FOXN2 significantly reversed the effects of
miR-32-5p-mimic transfection that promoted the activation of
PI3K/AKT/Bcl-2 pathway (Fig. 6B and
C; P<0.05), and the inhibitory effect on FOXN2 expression
levels (Fig. 6B and C;
P<0.001).
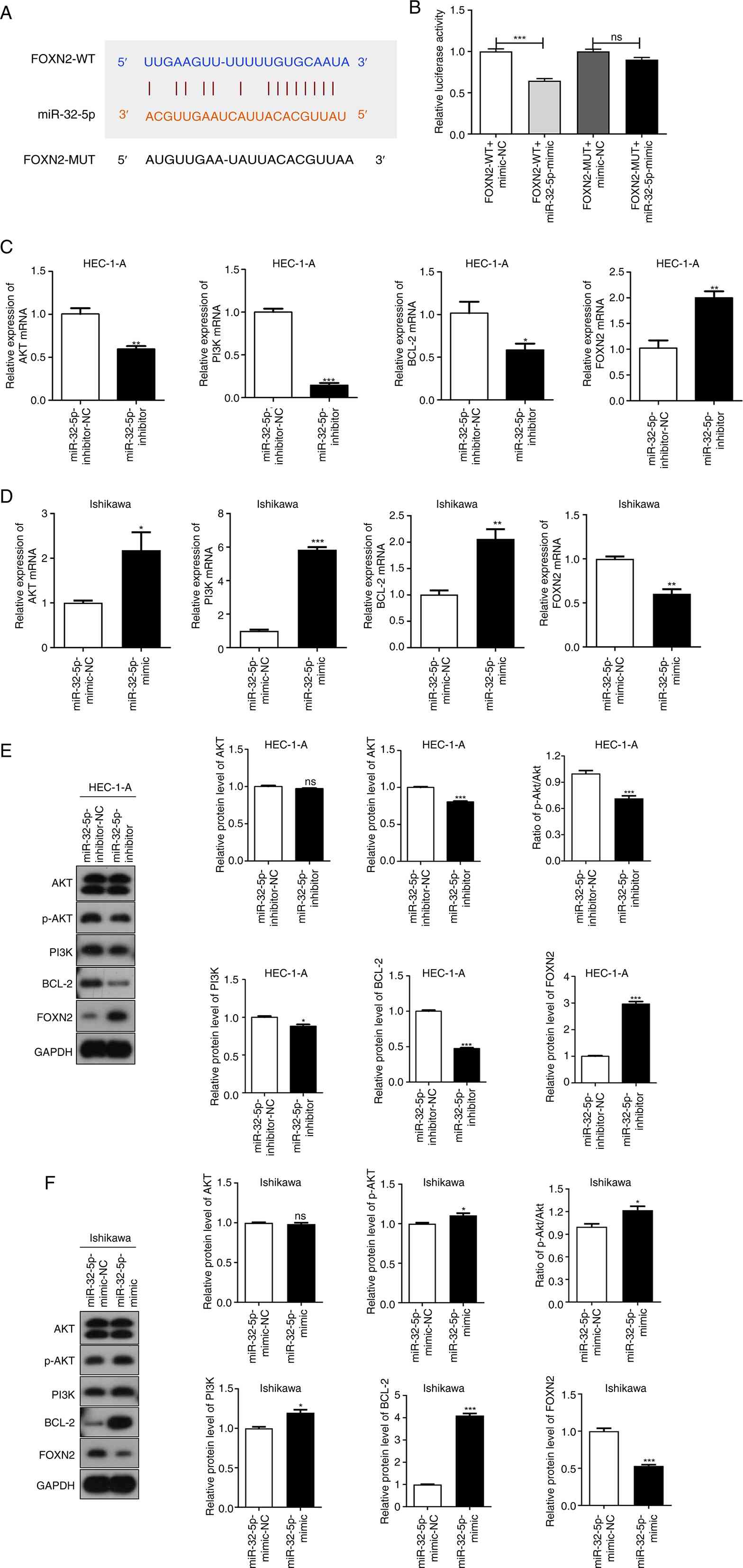 | Figure 5.miR-32-5p negatively regulates FOXN2
and activates PI3K/AKT pathway. (A) The Starbase software shows the
potential binding site for miR-32-5p and FOXN2. (B) A dual
luciferase reporter assay was conducted to assess the relationship
between miR-32-5p and FOXN2. ***P<0.001 vs. FOXN2-WT
+ mimic-NC. (C) The mRNA expression of AKT, PI3K, Bcl-2 and FOXN2
in HEC-1-A cells transfected with miR-32-5p inhibitor or
inhibitor-NC was detected using qRT-PCR. *P<0.05, **P<0.01,
***P<0.001 vs. miR-32-5p inhibitor-NC. (D) The mRNA expression
of AKT, PI3K, Bcl-2 and FOXN2 in Ishikawa cells transfected with
miR-32-5p mimic or mimic-NC was detected by qRT-PCR. *P<0.05,
**P<0.01 and ***P<0.001 vs. miR-32-5p mimic-NC. (E) The
protein levels of AKT, PI3K, p-PI3K, Bcl-2, FOXN2 and the ratio of
p-AKT/AKT in HEC-1-A cells transfected with miR-32-5p inhibitor or
inhibitor-NC were measured using western blotting. *P<0.05 and
***P<0.001 vs. miR-32-5p inhibitor-NC. (F) The protein levels of
AKT, PI3K, p-PI3K, Bcl-2, FOXN2 and the ratio of p-AKT/AKT in
Ishikawa cells transfected with miR-32-5p mimic or mimic-NC were
measured using western blotting. *P<0.05 and ***P<0.001 vs.
miR-32-5p inhibitor-NC. UCEC, uterine corpus endometrial carcinoma;
miRNA, microRNA; NC, negative control; NS, not significant; WT,
wild-type; MUT, mutant; qRT-PCR, quantitative reverse
transcription-PCR; p-, phosphorylated; FOXN2, Forkhead Box N2. |
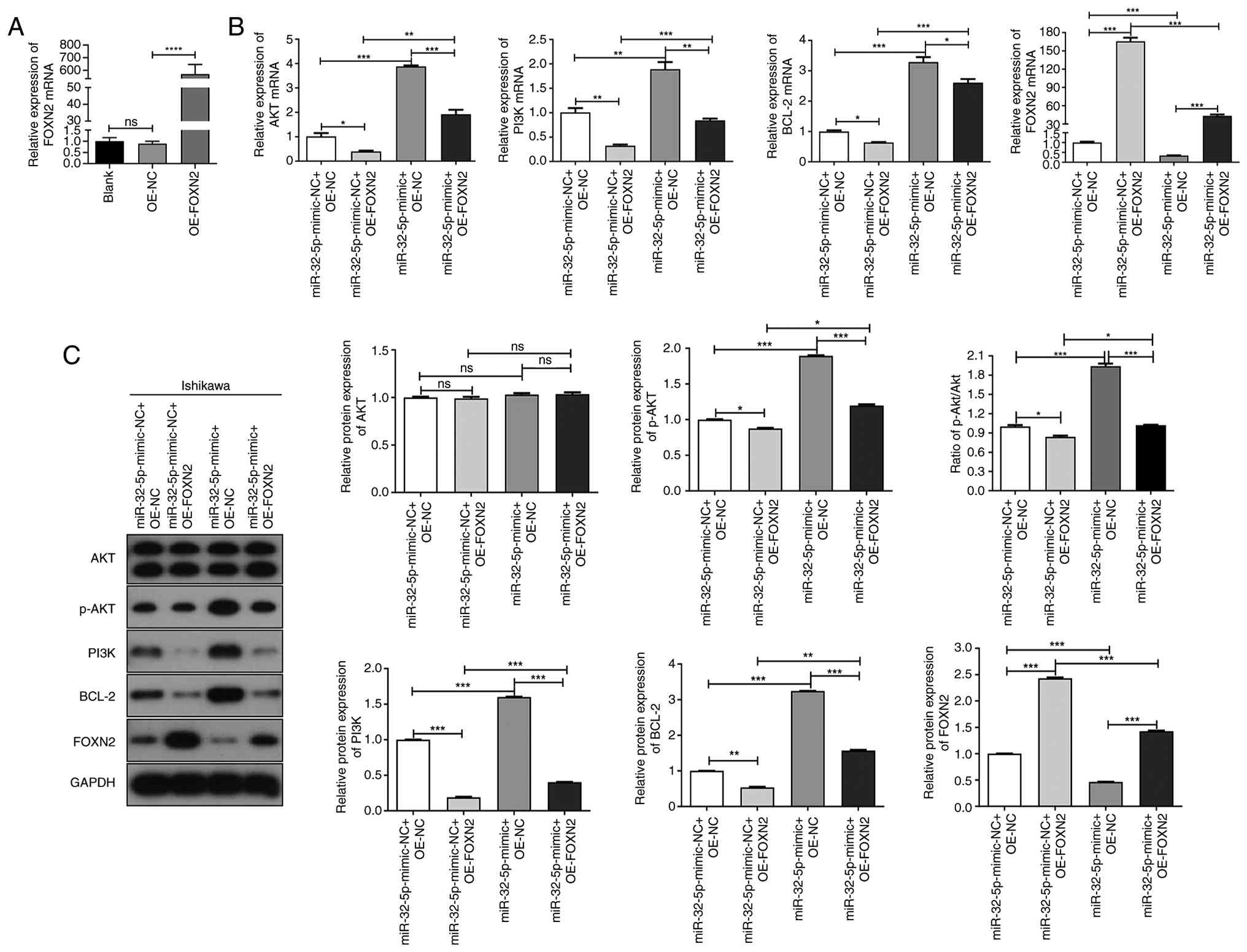 | Figure 6.Effects of miR-32-5p-mediated FOXN2
on PI3K/AKT/Bcl-2 pathway. (A) Following transfection of
OE-FOXN2/OE-NC in Ishikawa cells, the mRNA expression of FOXN2 was
determined via qRT-PCR. Following transfection of miR-32-5p-mimic
or OE-FOXN2 in Ishikawa cells, (B) the mRNA expression and (C)
protein levels of FOXN2/PI3K/AKT/Bcl-2 and the ratio of p-AKT/AKT
were determined through qRT-PCR and western blotting, respectively.
*P<0.05, **P<0.01, ***P<0.001, ****P<0.0001. UCEC,
uterine corpus endometrial carcinoma; miRNA, microRNA; NC, negative
control; NS, not significant; WT, wild-type; MUT, mutant; qRT-PCR,
quantitative reverse transcription-PCR; p-, phosphorylated; FOXN2,
Forkhead Box N2; OE, overexpression. |
Exo-miR-32-5p regulates the
proliferation, migration and apoptosis of UCEC cells through
regulating FOXN2 expression and PI3K/AKT/Bcl-2 pathway
The impacts of Exo-miR-32-5p on the proliferation,
migration and apoptosis of UCEC cells were examined utilizing a
co-culture model. Firstly, Exo-miR-32-5p expression in HEC-1-A
cells transfected with miR-32-5p-inhibitor or in Ishikawa cells
transfected with miR-32-5p-mimic was determined using qRT-PCR.
miR-32-5p expression was significantly downregulated in
Exo-miR-32-5p-inhibitor group when compared with the
Exo-miR-32-5p-inhibitor-NC group (Fig.
7A; P<0.001), while miR-32-5p expression levels were
significantly increased in the Exo-miR-32-5p-mimic group when
compared with the Exo-miR-32-5p-mimic-NC group (Fig. 7A; P<0.001). Compared with the
Exo-miR-32-5p-inhibitor-NC group, the proliferation and migration
of HEC-1-A cells were significantly decreased in the
Exo-miR-32-5p-inhibitor group (Fig.
7B-E; P<0.01), while the rate of apoptosis was increased
(Fig. 7F; P<0.001). Conversely,
compared with the Exo-miR-32-5p-mimic-NC group, a significant
increase of proliferative and migratory capacities (Fig. 7B-E; P<0.01) and significant
decrease of the apoptosis rate in Ishikawa cells of the
Exo-miR-32-5p-mimic group was demonstrated (Fig. 7F; P<0.001). Additionally, FOXN2
levels were increased, Bcl-2 levels were decreased and the PI3K/AKT
pathway was inactivated in HEC-1-A cells (Fig. 7G and H; P<0.01), while these
effects were all reversed in Ishikawa cells (Fig. 7G and H; P<0.01).
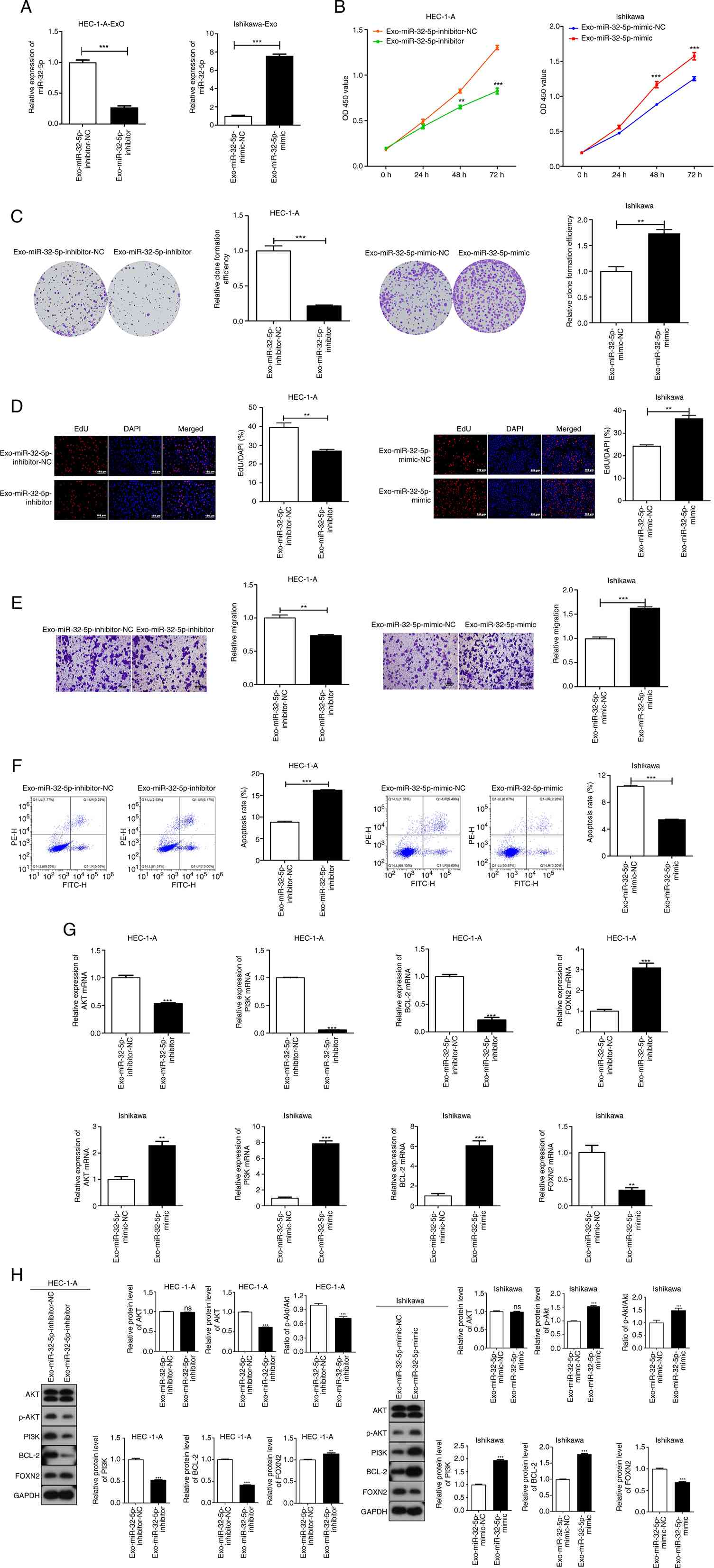 | Figure 7.Exo-miR-32-5p regulates the
proliferation, migration and apoptosis of UCEC cells through
regulating FOXN2 expression and PI3K/AKT/Bcl-2 pathway. The impact
of Exo-miR-32-5p on the proliferation, migration and apoptosis of
UCEC cells were examined utilizing a co-culture model. (A)
Exo-miR-32-5p expression in HEC-1-A cells transfected with
miR-32-5p-inhibitor or in Ishikawa cells transfected with
miR-32-5p-mimic was determined through qRT-PCR. (B) The viability
of UCEC cell lines were measured using the CCK-8 assay. (C)
Relative colony formation efficiency of UCEC cell lines was
evaluated using the colony formation assay. (D) EdU-positive cells
were measured through EdU proliferation assay. (Scale bar, 100 µm).
(E) The migratory potential of UCEC cell lines were determined via
the Transwell migration assay. (F) Flow cytometry was used to
detect apoptosis. (G) The mRNA expression of AKT, PI3K, Bcl-2 and
FOXN2 was detected by qRT-PCR. (H) The protein levels of AKT, PI3K,
p-PI3K, Bcl-2, FOXN2 and the ratio of p-AKT/AKT were measured by
western blotting. **P<0.01, ***P<0.001 vs. Exo-miR-32-5p
inhibitor-NC or Exo-miR-32-5p mimic-NC. UCEC, uterine corpus
endometrial carcinoma; miRNA, microRNA; NC, negative control; NS,
not significant; WT, wild-type; MUT, mutant; qRT-PCR, quantitative
reverse transcription-PCR; p-, phosphorylated; FOXN2, Forkhead Box
N2; EdU, 5-ethynyl-2′-deoxyuridine. |
Discussion
UCEC is a highly prevalent malignant tumor affecting
the female reproductive system, particularly within obstetrics and
gynecological tumors in Western countries (32). The incidence of UCEC has been
progressively increasing in China, posing a significant threat to
women's health (33). Diagnosis of
UCEC typically relies on clinical symptoms, medical history and
pathological findings. Patients are required to undergo uterine
apoxesis for pathology examination in order to accurately diagnose
UCEC, as there is a lack of effective biomarkers (34). Exosomal miRNAs have gained
significant attention in the field of precision medicine due to
their non-invasive nature, accessibility and stability (35).
Previous research has demonstrated that exosomal
miRNAs hold potential as effective biomarkers for cancer screening,
diagnosis and monitoring purposes (36,37).
In the present study, among the four candidate miRNAs, miR-32-5p
was identified as the most significantly associated with the
survival probability (P=0.00036) of patients with UCEC. Meanwhile,
the role and mechanism of exosomal miR-32-5p in UCEC progression
in vitro have been preliminarily elucidated, indicating that
exosomal-miR-32-5p can regulate the proliferation, migration and
apoptosis of UCEC cells through FOXN2/PI3K/AKT pathway.
miRNAs, a subclass of non-coding RNAs ranging from
19 to 25 nucleotides in length, serve key roles in extensive
modulation by specifically targeting the 3′-UTRs of mRNAs (38). miR-32-5p, belonging to miR-32
family, is located on chromosome 9 (39). Numerous studies have provided
evidence for the functional significance of miR-32-5p in various
cancer types, highlighting its capacity to modulate tumor
development, growth patterns, metastatic potential and invasive
characteristics. For example, overexpression of the long non-coding
RNA GAS5 suppressed the proliferative and metastatic capacities of
pancreatic cancer cells, while upregulation of miR-32-5p reversed
this inhibitory effect (17).
Notably high expression levels of miR-32-5p were observed in
colorectal cancer tissues and strongly correlated with poor
prognosis; however, miR-32-5p downregulation markedly impeded the
metastasis of colorectal cancer cells (18). Additionally, upregulation of
miR-32-5p expression has also been documented in gynecological
malignancies such as ovarian cancer, and its overexpression
significantly accelerated cancer cell growth and metastasis
(19). In the present study, a
significant increase level of miR-32-5p was identified in UCEC
tissues in contrast to that of normal tissues, implying that
miR-32-5p may be an onco-miRNA affecting the development of UCEC.
miR-32-5p expression was then inhibited or overexpressed to further
explore the dysregulation of miR-32-5p on the malignant behaviors
of UCEC cells. High levels of miR-32-5p expression were found to
significantly increase the proliferative and migratory capacities,
while concurrently suppressing the UCEC apoptosis rate. Meanwhile,
inhibition of miR-32-5p yielded the opposite results. These
findings further substantiate the present hypothesis that miR-32-5p
can induce the progression of UCEC by enhancing cancer cell
proliferation and migration, while inhibiting apoptosis.
Additionally, miR-32-5p has been reported to target
the downstream mRNAs, exerting regulation effects in various human
solid tumors, such as miR-32-5p-SIK1 in ovarian cancer (19), miR-32-5p-HOXB8 in cervical cancer
(40) and miR-32-5p-TOB1 in breast
cancer (41). Considering the
regulatory function of miR-32-5p in UCEC cells, the existence of
potential downstream genes modulated by miR-32-5p during UCEC
progression could be considered. In the present study, a putative
binding site for miR-32-5p and FOXN2 was identified using the
Starbase software prediction, and this interaction was
experimentally validated using DLR assay. The negatively regulatory
effect of miR-32-5p on FOXN2 expression was further demonstrated.
Over the past decade, the PI3K/AKT pathway has garnered attention
as it serves a pivotal role in orchestrating an array of cellular
functions, encompassing transcription, metabolism, growth and
apoptosis (42). Recently, the
oncogene role of the PI3K/AKT pathway in UCEC has been elucidated,
underscoring its potential as a promising therapeutic target
(43,44). A PI3K/AKT inhibitor, PF-04691502,
has proven to be beneficial for patients with recurrent UCEC from a
clinical perspective (29). It was
thus considered that a potential interaction may exist between
miR-32-5p and the PI3K/AKT pathway in UCEC cells. The upregulation
of miR-32-5p was found to effectively activate the PI3K/AKT
pathway. Conversely, downregulation of miR-32-5p exhibited a
suppressive effect on the activity of the PI3K/AKT pathway.
Additionally, AKT exerts a negative regulatory effect on the
function or expression of Bcl-2 homology domain 3-only proteins,
which are recognized for their role in promoting cell death by
deactivating the anti-cell death members of the Bcl-2 family
(30). Therefore, inhibiting the
PI3K/AKT signaling pathway has been demonstrated to promote
apoptosis in UCEC cells (30). The
present study further demonstrated that the upregulation of
miR-32-5p could effectively increase the expression levels of
Bcl-2, whereas its downregulation exerted inhibitory effects on
Bcl-2 expression. Collectively, the present findings suggested that
miR-32-5p may exert its regulatory effects on UCEC by targeting the
FOXN2/PI3K/AKT/Bcl-2 pathway, thereby promoting proliferation and
migration while inhibiting apoptosis.
The increasing attention towards miRNAs in exosomes
stems from their pivotal role in recruiting and reprogramming
essential components of the tumor microenvironment (45,46).
miRNAs in exosomes are also considered vital for intercellular
communication through cell-to-cell contact (47). Numerous reports have demonstrated
that the transfer of miRNA through exosomes alters the
microenvironment of tumors, ultimately contributing to the
tumorigenesis of UCEC. For example, Yao et al (48) identified four hub exosomal miRNAs
including miR-320d, miR-193a-5p, miR-99b-3p and miR-17-3p based on
bioinformatic analysis, and demonstrated their potential as
prognostic biomarkers or therapeutic targets in UCEC. Jing et
al (16) reported that exosomal
miR-499a-5p interacts with VAV3 to suppress the growth of UCEC. Li
et al (49) found an
exosomal miR-148b from cancer-associated fibroblasts serve as a
tumor suppressor in UCEC through the targeting of DNA
methyltransferase 1. In the present study, considering the effects
of miR-32-5p on the malignant behaviors of UCEC cells, it was
further considered that exosomal miR-32-5p might induce comparable
impacts on the growth, metastasis and apoptosis of UCEC cells. A
co-culture system was established in the present study;
Exo-miR-32-5p-inhibitor or Exo-miR-32-5p-inhibitor-NC was
internalized by HEC-1-A cells, while Exo-miR-32-5p-mimic or
Exo-miR-32-5p-mimic-NC was internalized by Ishikawa cells.
Following internalization, Exo-miR-32-5p-inhibitor significantly
suppressed the proliferative and migratory capacities, and induced
apoptosis of HEC-1-A cells, while the opposite results were
observed in Ishikawa cells with the Exo-miR-32-5p-mimic. These
findings demonstrated the regulatory role of exosomal miR-32-5p in
the malignant behaviors of UCEC cells. Furthermore, in HEC-1-A
cells, the Exo-miR-32-5p-inhibitor inhibited PI3K/AKT/Bcl-2 pathway
and elevated FOXN2 expression levels, whereas Exo-miR-32-5p-mimic
exerted the opposite regulatory effects in Ishikawa cells. These
results demonstrated that the transfer of exosomal miR-32-5p serves
a pivotal role in modulating the expression levels of multiple
target genes within recipient cells, which was in line with
findings of previous studies (13,14).
Therefore, it was demonstrated that exosomal-miR-32-5p regulates
the proliferation, migration and apoptosis of UCEC through
FOXN2/PI3K/AKT/Bcl-2 pathway.
There were some limitations to the present study.
First, polydispersity index (PDI) serves as an important indicator
that characterizes the uniformity of particle size distribution in
samples. It is commonly used to evaluate the particle size
distribution of exosomes (50). In
this study, the absence of PDI may not accurately evaluate the
purity of exosomes, thereby affecting the accuracy of experimental
results. Furthermore, exosomes may exhibit agglomeration phenomenon
in the samples, and PDI is an important indicator for determining
agglomeration (50). If this
indicator is ignored, it may not be possible to detect and handle
the agglomeration issues in a timely manner, which could affect the
separation and purification efficiency of exosomes (50). Therefore, when investigating
exosomes in future research, PDI will be incorporated as a
significant parameter to facilitate a more comprehensive
identification of the extracted exosomes. Second, the sample size
of patients was relatively small. Larger sample sizes should be
considered in future studies. Third, cell line studies possess
limitations in predicting clinical outcomes; the present study only
conducted preliminary research on the regulatory mechanisms of
exosomal-miR-32-5p in UCEC at the cellular level, and lacked
validation of biological integrity, experimentation in an in
vivo microenvironment and feasibility of clinical
transformation, therefore further investigation in animal models is
warranted.
In summary, the present study demonstrates a novel
insight into the role of exosomal miR-32-5p in promoting the
proliferation and migration, while inhibiting the apoptosis rate of
UCEC cells through regulation of the FOXN2/PI3K/AKT/Bcl-2 pathway.
The findings presented may provide information and insights for
potential clinical therapeutic strategies for patients with
UCEC.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present work was supported by Mechanism Study of Recombinant
Human Interleukin-11 Combined with Dendritic Cell Vaccine Loading
of Tumor Stem Cell Antigen in the Treatment of Head and Neck
Squamous Cell Cancer (grant. no. KYC0000000807) and Mechanism study
of circular RNA hsa_circ 0000523 as a competitive endogenous RNA
upregulating METTL3 to promote invasion and metastasis of colon
cancer (grant. no. 2024HX0021).
Availability of data and materials
The data analyzed during the current study are
available from the corresponding author on reasonable request.
Authors' contributions
XinC made substantial contributions to the
conception and design of the study. XinC, HL, XiaC, YC and GG made
substantial contributions to the acquisition, analysis and
interpretation of the data. XinC and HL drafted the manuscript. All
authors critically revised the manuscript for intellectual content.
XiaC, YC and GG confirmed the authenticity of all the raw data. All
authors have read and approved the final manuscript.
Ethics approval and consent to
participate
The present study adheres to the principles outlined
in the Declaration of Helsinki, and ethical approvals have been
obtained from Renmin Hospital of Wuhan University Ethics Committee
(approval no. WDRY2023-K161; Wuhan, China). All participants
provided informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
miRNA
|
microRNA
|
|
mRNA
|
messenger RNA
|
|
UCEC
|
uterine corpus endometrial
carcinoma
|
|
TCGA
|
The Cancer Genome Atlas
|
|
DE
|
differentially expressed
|
|
EdU
|
5-ethynyl-2′-deoxyuridine
|
|
WT
|
wild-type
|
|
MUT
|
mutant
|
References
|
1
|
Crosbie EJ, Kitson SJ, McAlpine JN,
Mukhopadhyay A, Powell ME and Singh N: Endometrial cancer. Lancet.
399:1412–1428. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hafizz AMHA, Zin RRM, Aziz NHA, Kampan NC
and Shafiee MN: Beyond lipid-lowering: Role of statins in
endometrial cancer. Mol Biol Rep. 47:8199–8207. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li Y, Huang C, Kavlashvili T, Fronk A,
Zhang Y, Wei Y, Dai D, Devor EJ, Meng X, Thiel KW, et al: Loss of
progesterone receptor through epigenetic regulation is associated
with poor prognosis in solid tumors. Am J Cancer Res. 10:1827–1843.
2020.PubMed/NCBI
|
|
4
|
Francis SR, Ager BJ, Do OA, Huang YJ,
Soisson AP, Dodson MK, Werner TL, Sause WT, Grant JD and Gaffney
DK: Recurrent early stage endometrial cancer: Patterns of
recurrence and results of salvage therapy. Gynecol Oncol.
154:38–44. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bregar AJ, Melamed A, Diver E, Clemmer JT,
Uppal S, Schorge JO, Rice LW, Del Carmen MG and Rauh-Hain JA:
Minimally invasive staging surgery in women with early-stage
endometrial cancer: Analysis of the national cancer data base. Ann
Surg Oncol. 24:1677–1687. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Connor EV and Rose PG: Management
strategies for recurrent endometrial cancer. Expert Rev Anticancer
Ther. 18:873–885. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pölcher M, Rottmann M, Brugger S, Mahner
S, Dannecker C, Kiechle M, Brambs C, Grab D, Anthuber C, von Koch
F, et al: Lymph node dissection in endometrial cancer and clinical
outcome: A population-based study in 5546 patients. Gynecol Oncol.
154:65–71. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wessler S, Aberger F and Hartmann TN: The
sound of tumor cell-microenvironment communication-composed by the
cancer cluster salzburg research network. Cell Commun Signal.
15:202017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lauko A, Mu Z, Gutmann DH, Naik UP and
Lathia JD: Junctional adhesion molecules in cancer: A paradigm for
the diverse functions of cell-cell interactions in tumor
progression. Cancer Res. 80:4878–4885. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Paskeh MDA, Entezari M, Mirzaei S,
Zabolian A, Saleki H, Naghdi MJ, Sabet S, Khoshbakht MA, Hashemi M,
Hushmandi K, et al: Emerging role of exosomes in cancer progression
and tumor microenvironment remodeling. J Hematol Oncol. 15:832022.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wei H, Chen Q, Lin L, Sha C, Li T, Liu Y,
Yin X, Xu Y, Chen L, Gao W, et al: Regulation of exosome production
and cargo sorting. Int J Biol Sci. 17:163–177. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cully M: Exosome-based candidates move
into the clinic. Nat Rev Drug Discov. 20:6–7. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Au Yeung CL, Co NN, Tsuruga T, Yeung TL,
Kwan SY, Leung CS, Li Y, Lu ES, Kwan K, Wong KK, et al: Exosomal
transfer of stroma-derived miR21 confers paclitaxel resistance in
ovarian cancer cells through targeting APAF1. Nat Commun.
7:111502016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Baroni S, Romero-Cordoba S, Plantamura I,
Dugo M, D'Ippolito E, Cataldo A, Cosentino G, Angeloni V, Rossini
A, Daidone MG and Iorio MV: Exosome-mediated delivery of miR-9
induces cancer-associated fibroblast-like properties in human
breast fibroblasts. Cell Death Dis. 7:e23122016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Che X, Jian F, Chen C, Liu C, Liu G and
Feng W: PCOS serum-derived exosomal miR-27a-5p stimulates
endometrial cancer cells migration and invasion. J Mol Endocrinol.
64:1–12. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jing L, Hua X, Yuanna D, Rukun Z and
Junjun M: Exosomal miR-499a-5p inhibits endometrial cancer growth
and metastasis via targeting VAV3. Cancer Manag Res.
12:13541–13552. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gao ZQ, Wang JF, Chen DH, Ma XS, Wu Y,
Tang Z and Dang XW: Long non-coding RNA GAS5 suppresses pancreatic
cancer metastasis through modulating miR-32-5p/PTEN axis. Cell
Biosci. 7:662017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liang H, Tang Y, Zhang H and Zhang C:
MiR-32-5p regulates radiosensitization, migration and invasion of
colorectal cancer cells by targeting TOB1 gene. Onco Targets Ther.
12:9651–9661. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jin Y and Wang H: Circ_0078607 inhibits
the progression of ovarian cancer via regulating the miR-32-5p/SIK1
network. J Ovarian Res. 15:32022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang P, Liang T, Chen Y, Wang X, Wu T,
Xie Z, Luo J, Yu Y and Yu H: Circulating exosomal miRNAs as Novel
biomarkers for stable coronary artery disease. Biomed Res Int.
2020:35939622020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhou L, Wang W, Wang F, Yang S, Hu J, Lu
B, Pan Z, Ma Y, Zheng M, Zhou L, et al: Plasma-derived exosomal
miR-15a-5p as a promising diagnostic biomarker for early detection
of endometrial carcinoma. Mol Cancer. 20:572021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Berek JS, Matias-Guiu X, Creutzberg C,
Fotopoulou C, Gaffney D, Kehoe S, Lindemann K, Mutch D and Concin
N; Endometrial Cancer Staging Subcommittee, FIGO Women's Cancer
Committee, : FIGO staging of endometrial cancer: 2023. Int J
Gynaecol Obstet. 162:383–394. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Pan Y, Wang X, Li Y, Yan PY and Zhang H:
Human umbilical cord blood mesenchymal stem cells-derived exosomal
microRNA-503-3p inhibits progression of human endometrial cancer
cells through downregulating MEST. Cancer Gene Ther. 29:1130–1139.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhu Z, Chen Z, Wang M, Zhang M, Chen Y,
Yang X, Zhou C, Liu Y, Hong L and Zhang L: Detection of plasma
exosomal miRNA-205 as a biomarker for early diagnosis and an
adjuvant indicator of ovarian cancer staging. J Ovarian Res.
15:272022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu W, Yang D, Chen L, Liu Q, Wang W, Yang
Z, Shang A, Quan W and Li D: Plasma exosomal miRNA-139-3p is a
novel biomarker of colorectal cancer. J Cancer. 11:4899–4906. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Taghehchian N, Lotfi M, Zangouei AS,
Akhlaghipour I and Moghbeli M: MicroRNAs as the critical regulators
of Forkhead box protein family during gynecological and breast
tumor progression and metastasis. Eur J Med Res. 28:3302023.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xie SM, Zhang Q and Jiang L: Current
knowledge on exosome biogenesis, cargo-sorting mechanism and
therapeutic implications. Membranes (Basel). 12:4982022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jiang N, Dai Q, Su X, Fu J, Feng X and
Peng J: Role of PI3K/AKT pathway in cancer: The framework of
malignant behavior. Mol Biol Rep. 47:4587–4629. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
He Y, Sun MM, Zhang GG, Yang J, Chen KS,
Xu WW and Li B: Targeting PI3K/Akt signal transduction for cancer
therapy. Signal Transduct Target Ther. 6:4252021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pavlidou A and Vlahos NF: Molecular
alterations of PI3K/Akt/mTOR pathway: A therapeutic target in
endometrial cancer. ScientificWorldJournal. 2014:7097362014.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Buhtoiarova TN, Brenner CA and Singh M:
Endometrial carcinoma: Role of current and emerging biomarkers in
resolving persistent clinical dilemmas. Am J Clin Pathol. 145:8–21.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hu ZY, Tang LD, Zhou Q, Xiao L and Cao Y:
Aberrant promoter hypermethylation of p16 gene in endometrial
carcinoma. Tumour Biol. 36:1487–1491. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Muinelo-Romay L, Casas-Arozamena C and
Abal M: Liquid biopsy in endometrial cancer: New opportunities for
personalized oncology. Int J Mol Sci. 19:23112018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Falcone G, Felsani A and D'Agnano I:
Signaling by exosomal microRNAs in cancer. J Exp Clin Cancer Res.
34:322015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bjørnetrø T, Redalen KR, Meltzer S,
Thusyanthan NS, Samiappan R, Jegerschöld C, Handeland KR and Ree
AH: An experimental strategy unveiling exosomal microRNAs 486-5p,
181a-5p and 30d-5p from hypoxic tumour cells as circulating
indicators of high-risk rectal cancer. J Extracell Vesicles.
8:15672192019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zheng M, Hou L, Ma Y, Zhou L, Wang F,
Cheng B, Wang W, Lu B, Liu P, Lu W and Lu Y: Exosomal let-7d-3p and
miR-30d-5p as diagnostic biomarkers for non-invasive screening of
cervical cancer and its precursors. Mol Cancer. 18:762019.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Panda AC: Circular RNAs Act as miRNA
sponges. Adv Exp Med Biol. 1087:67–79. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zeng ZL, Zhu Q, Zhao Z, Zu X and Liu J:
Magic and mystery of microRNA-32. J Cell Mol Med. 25:8588–8601.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liu YJ, Zhou HG, Chen LH, Qu DC, Wang CJ,
Xia ZY and Zheng JH: MiR-32-5p regulates the proliferation and
metastasis of cervical cancer cells by targeting HOXB8. Eur Rev Med
Pharmacol Sci. 23:87–95. 2019.PubMed/NCBI
|
|
41
|
Wang R, Huang Z, Qian C, Wang M, Zheng Y,
Jiang R and Yu C: LncRNA WEE2-AS1 promotes proliferation and
inhibits apoptosis in triple negative breast cancer cells via
regulating miR-32-5p/TOB1 axis. Biochem Biophys Res Commun.
526:1005–1012. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lawrence MS, Stojanov P, Mermel CH,
Robinson JT, Garraway LA, Golub TR, Meyerson M, Gabriel SB, Lander
ES and Getz G: Discovery and saturation analysis of cancer genes
across 21 tumour types. Nature. 505:495–501. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wei ST, Zhang J, Zhao R, Shi R, An L, Yu
Z, Zhang Q, Zhang J, Yao Y, Li H and Wang H: Histone lactylation
promotes malignant progression by facilitating USP39 expression to
target PI3K/AKT/HIF-1α signal pathway in endometrial carcinoma.
Cell Death Discov. 10:1212024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ma LJ, Huang WN, Liang XL, Bai G, Wang X,
Jiang H, Xin Y, Hu L, Chen X and Liu C: Inhibition of squalene
epoxidase linking with PI3K/AKT signaling pathway suppresses
endometrial cancer. Cancer Sci. 114:3595–3607. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Li C, Zhou T, Chen J, Li R, Chen H, Luo S,
Chen D, Cai C and Li W: The role of exosomal miRNAs in cancer. J
Transl Med. 20:62022. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Nail HM, Chiu CC, Leung CH, Ahmed MMM and
Wang HMD: Exosomal miRNA-mediated intercellular communications and
immunomodulatory effects in tumor microenvironments. J Biomed Sci.
30:692023. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Fasken MB, Morton DJ, Kuiper EG, Jones SK,
Leung SW and Corbett AH: The RNA exosome and human disease. Methods
Mol Biol. 2062:3–33. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yao Y, Shi L and Zhu X: Four
differentially expressed exosomal miRNAs as prognostic biomarkers
and therapy targets in endometrial cancer: Bioinformatic analysis.
Medicine (Baltimore). 102:e349982023. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Li BL, Lu W, Qu JJ, Ye L, Du GQ and Wan
XP: Loss of exosomal miR-148b from cancer-associated fibroblasts
promotes endometrial cancer cell invasion and cancer metastasis. J
Cell Physiol. 234:2943–2953. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wang Y, Guo MM, Lin DM, Liang D, Zhao L,
Zhao R and Wang Y: Docetaxel-loaded exosomes for targeting
non-small cell lung cancer: preparation and evaluation in vitro and
in vivo. Drug Deliv. 28:1510–1523. 2021. View Article : Google Scholar : PubMed/NCBI
|