Introduction
There is a notable variation in survival rates among
individuals diagnosed with penile squamous cell cancer (PSCC). For
patients diagnosed with localized PSCC (N0 stage), the 5-year OS
rate is ~90% (1). Once the tumor
metastasizes to the lymph nodes, the prognosis becomes markedly
worse. In a previous study, patients with unilateral inguinal lymph
node (ILN) involvement and ≤2 lymph nodes (N1 stage) experienced a
5-year OS rate of 80%, those with bilateral or pelvic lymph node
involvement (N2 or N3 stage) experienced an OS rate of 10–20% and
those patients with extranodal lymph node involvement had a
survival rate of <10% (2).
Therefore, early diagnosis and treatment are critical factors in
determining patient survival (2).
Lymph node metastasis is a recognized determinant of prognosis in
PSCC, and early radical ILN dissection (rILND) has been reported to
confer a survival advantage. For patients with positive lymph
nodes, early rILND can achieve a 5-year overall survival (OS) rate
of 71–80% compared with those waiting for observation (3–5).
However, complications following rILND, such as lymphatic fistula,
necessitating prolonged periods with drainage tubes, and
occasionally leading to lower limb edema, flap infection/necrosis
at the incision site and hernia formation, could significantly
impact the patient's quality of life in the long term. Appropriate
boundaries for dissection are therefore crucial to ensure
tumor-free resection, achieve a complete cure and reduce surgical
complications.
In rILND for PSCC, the standards for the extent of
resection are not uniform across various guidelines; for example,
the scope of dissection in Mei Hui Urology (6) surgical area is larger than that in the
NCCN guidelines (7). Daseler et
al (8) introduced the concept
of rILND and its approximate boundaries in 1948; however, a
detailed summary on the number and anatomical distribution of ILNs
remains lacking, especially the locations of pathologically
positive nodes (8). This has led to
current and ongoing controversy over different surgical approaches
for rILND in recent decades (9–14).
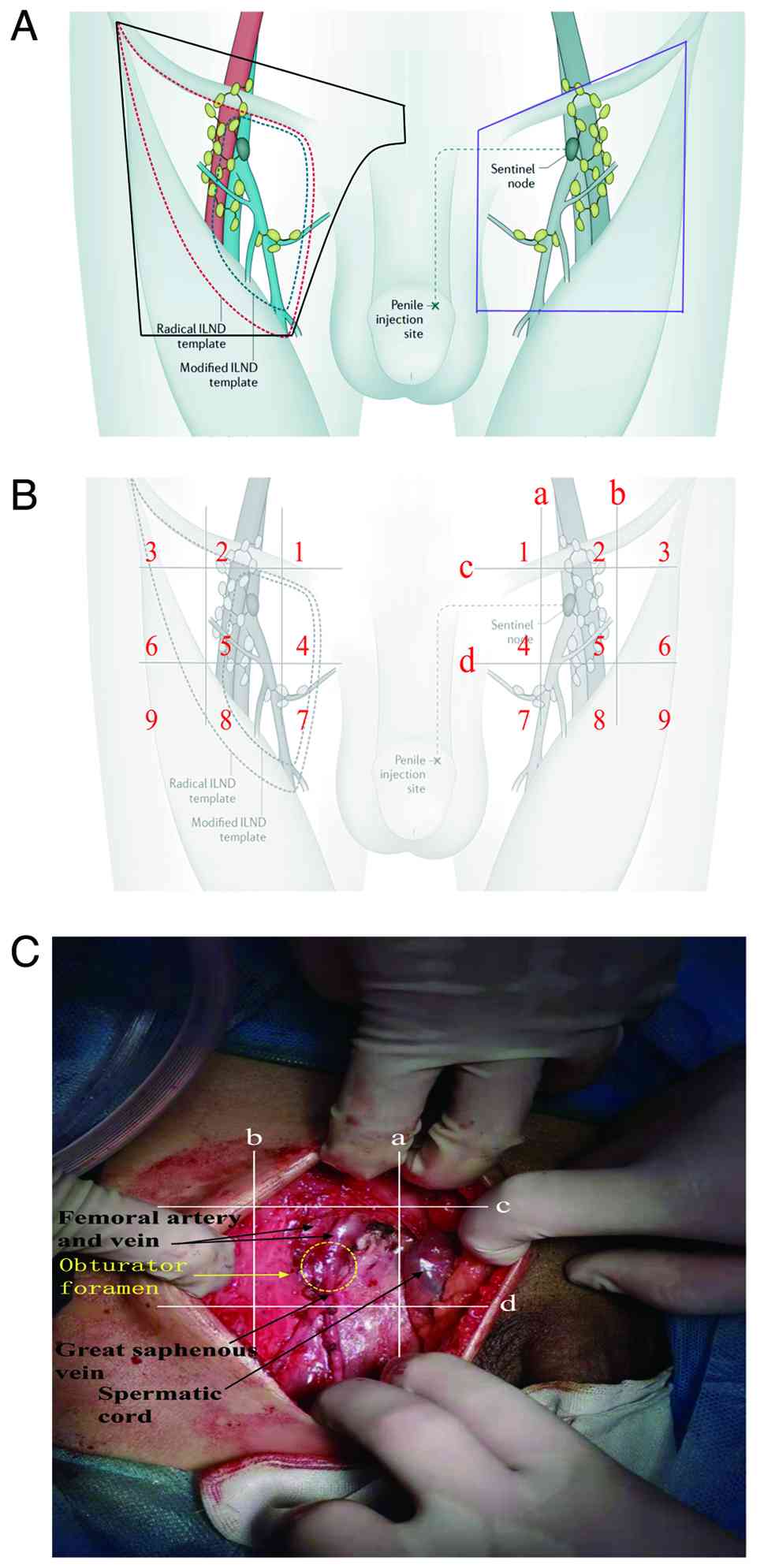
When discussing the boundaries of rILND, three main
scopes are primarily considered: i) The traditional rILND scope
(8) (Fig. 1A; purple outlined area); ii) the
standard rILND scope (15)
(Fig. 1A; red outlined area); iii)
the modified rILND scope (9,12,16)
(the external boundary narrowed from the sartorius muscle to the
femoral artery, resulting in a smaller dissection range; Fig. 1A; blue outlined area). The
controversy surrounding the boundaries of rILND is mainly
attributed to the lack of detailed anatomical partitioning data for
ILNs and corresponding pathological results.
In the clinical process at Yunnan Cancer Hospital
(Kunming, China), it had been observed that, occasionally, there
was recurrence and metastasis of the lymph nodes in the area above
the pubic bone after bilateral rILND, which is currently excluded
in several rILND scopes. Furthermore, it was suggested that the
number of all resected lymph nodes, the number of pathologically
positive lymph nodes and the lymph node metastasis density after
rILND in different regions could guide the surgical strategy,
reduce unnecessary dissection ranges, prevent the omission of
micro-metastases and reduce the recurrence rate in affected
patients.
The present study aimed to optimize the scope of
lymphadenectomy, specifically to include the suprapubic lymph
nodes. Building upon Daseler's five-region framework, a nine-region
model (regions 1–9) is proposed, centered around the saphenous
opening and the bifurcation of the great saphenous vein as the key
anatomical landmarks. This model involves expanding the
superomedial dissection boundary and independently designating the
suprapubic area as region 1. By calculating the lymph node
metastasis density within each predefined region, the present study
sought to achieve a more precise evaluation of metastasis density
and patterns, ultimately aiming to reduce unnecessary surgical
interventions and improve patient survival rates.
Materials and methods
Study population and ethics
statement
The present prospective study enrolled patients
diagnosed with PSCC at Yunnan Cancer Hospital between November 2021
and December 2024. All patients provided informed written consent
for participation, and the study protocol was approved by the
Medical Ethics Committee of Yunnan Cancer Hospital (approval no.
KYLX2022051) before data collection. The study was conducted in
accordance with the Declaration of Helsinki (2013 revision) and was
registered with the Chinese Clinical Trial Registry under the
clinical trial number ChiCTR2200064529 (registration date,
2022-10-11). All patients underwent simultaneous or elective
(within 3 months) bilateral rILND.
The dissection scope was expanded based on the
traditional rILND range (6), with
the upper boundary extending up to 2 cm above the inguinal ligament
and the inner upper boundary extending to the pubic symphysis,
adding an area in front of the pubic bone (Fig. 1A; black outlined area).
The traditional rILND (Mei Hui Urology) (6) scope includes the following: A line
from the external ring to the anterior superior iliac spine; the
lateral boundary represented by a line extending 20 cm downward
from the anterior superior iliac spine; and the medial boundary,
represented by a line extending 15 cm downward from the pubic
tubercle (Fig. 1A; purple
area).
Inclusion and exclusion criteria
The inclusion criteria were as follows: i) Diagnosed
with PSCC and undergoing rILND concurrently/electively; ii) age ≥18
years; and iii) willingness to participate in the research protocol
and undergo follow-up.
The exclusion criteria were as follows: i) Previous
rILND or biopsy; ii) prior systemic treatment (chemotherapy,
radiotherapy, immunotherapy, etc.); iii) pathology with other
differentiation types; iv) patients with incomplete medical records
or unable to obtain required clinical information; and v) patients
unwilling to provide clinical information.
In summary, the present study included patients who
underwent partial or total penectomy with rILND. All patients had
complete imaging information, clinical pathological data and
biochemical parameters.
rILND boundaries and the nine-section
anatomical division of ILNs
In the present study, the boundaries of rILND are
presented in Fig. 1A (black
outlined area). The surgical templates proposed by the NCCN
(7) and EAU (17) guidelines define the standard scope
(superior: inguinal ligament; inferior: saphenofemoral junction;
lateral: sartorius muscle; medial: adductor longus muscle) and the
modified scope (narrowed laterally to the femoral artery and
inferiorly to the femoral canal, with preservation of the great
saphenous vein). By contrast, the rILND boundaries in the present
study adopted traditional radical margins (8) but further extended this dissection
range by expanding the superior boundary to 2 cm above the inguinal
ligament and the medial limit to the pubic symphysis, thereby
incorporating the prepubic anatomical area. Superficial ILNs were
completely removed and zonally diagnosed according to anatomical
landmarks (Fig. 1B and C). The
inguinal region was divided using the nine-region method as
follows: Taking the saphenofemoral junction and the bifurcation of
the great saphenous vein as the center, 4 lines (a, b, c and d)
were drawn 1 cm above, below, medial, and lateral to this center,
respectively, forming 9 regions. Lymph nodes from each region were
collected for pathological examination. Line ‘a’ was located 1 cm
medial to the saphenofemoral junction, line ‘b’ was 1 cm lateral to
the saphenofemoral junction, line ‘c’ was at the level of the
midpoint of the inguinal ligament and line ‘d’ was located at the
inferior border of the femoral triangle below the saphenofemoral
junction (Fig. 1B and C). Region 1
is an area not included in the dissection ranges recommended by
current guidelines, and during surgery, lymph nodes in regions 1
through 9 were each subjected to separate pathological
examinations. The anterior and posterior sides, as well as the
superior and inferior directions, were marked with sutures or
Hem-o-locks. The actual division according to the nine-section
method is shown in Fig. 1C. The
rILND was performed by experienced surgeons with the credentials of
Associate Chief Physician or higher. The ILNs harvested after
dissection were categorized by trained Resident Physicians to
ensure consistency.
Pathological examination
Primary tumors and the 9 regions of ILNs were fixed
in 10% neutral buffered formalin at room temperature for 24 h, cut
into 4-µm sections for standard hematoxylin and eosin staining, and
subsequently evaluated under a light microscope at ×20 and ×100
magnification by experienced genitourinary subspecialty
pathologists. All isolated lymph nodes were analyzed using
5-mm-thick longitudinal whole-tissue sections to ascertain the
status of lymph node metastasis. Clinical pathological
characteristics were documented according to the 8th Edition of the
Tumor-Node-Metastasis Staging System for penile cancer (18).
Tx staging was assigned to patients who were
referred to Yunnan Cancer Hospital specifically for rILND following
initial excision of the primary tumor at an external hospital,
where the definitive pathological specimen was retained, thus
precluding an accurate T-stage assessment. The T1 category
represents a broad classification. This is because the original
pathology reports for the specimens obtained after primary tumor
resection lacked the detailed descriptions necessary to
consistently discriminate among the Tis, Ta and T1 substages.
Consequently, the broader T1 category was used in the present study
to encompass all non-invasive and early invasive diseases that were
not classified as T2 or higher.
Statistical analysis
Data were collected and analyzed using SPSS version
26.0 (IBM Corp.) and R version 4.2.1 (R Core Team, R Foundation for
Statistical Computing). The number of lymph nodes is expressed as
the mean ± standard deviation, and differences between groups were
compared using unpaired Student's t-test. P<0.05 was considered
to indicate a statistically significant difference. Fisher's exact
test was used to compare the proportion of positive lymph nodes
between the right and left inguinal regions. The Kaplan-Meier
method was used to estimate survival curves, and differences
between groups were compared with the log-rank test. Multivariate
Cox proportional hazards regression models were used to identify
independent prognostic factors. The results are presented as hazard
ratios (HRs) with corresponding 95% confidence intervals (CIs).
Results
Baseline characteristics of 26
patients with PSCC undergoing rILND
A total of 26 patients with PSCC underwent extended
bilateral rILND. Detailed baseline data are summarized in Table I. A total of 8 patients with PSCC
(30.8%) were found to have regional lymph node metastasis (pN+).
Overall, 26 left-sided and 26 right-sided ILNs were analyzed.
 | Table I.Baseline characteristics of 26
patients with PSCCa. |
Table I.
Baseline characteristics of 26
patients with PSCCa.
| Variables | Value |
|---|
| Median age (IQR),
years | 54 (41.8–59.5) |
| pT stage, n (%) |
|
| Tx | 15 (57.7) |
| ≤T1 | 6 (23.1) |
| T2 | 2 (7.7) |
| T3 | 3 (11.5) |
| AJCC 8th edition pN,
n (%) |
|
| pN0 | 18 (69.2) |
| pN1 | 3 (11.5) |
| pN2 | 5 (19.2) |
| Lymph
node dissection method, n (%) |
|
|
Unilateral | 0 (0.0) |
|
Bilateral | 26 (100.0) |
| pM, n (%) |
|
| pM0 | 26 (100.0) |
|
pM1 | 0 (0.0) |
| Grade, n (%) |
|
| G1 | 9 (34.6) |
| G2 | 13 (50.0) |
| G3 | 4 (15.4) |
| Adjuvant therapy, n
(%) | 9 (34.6) |
Distribution of resected and positive
ILNs in PSCC
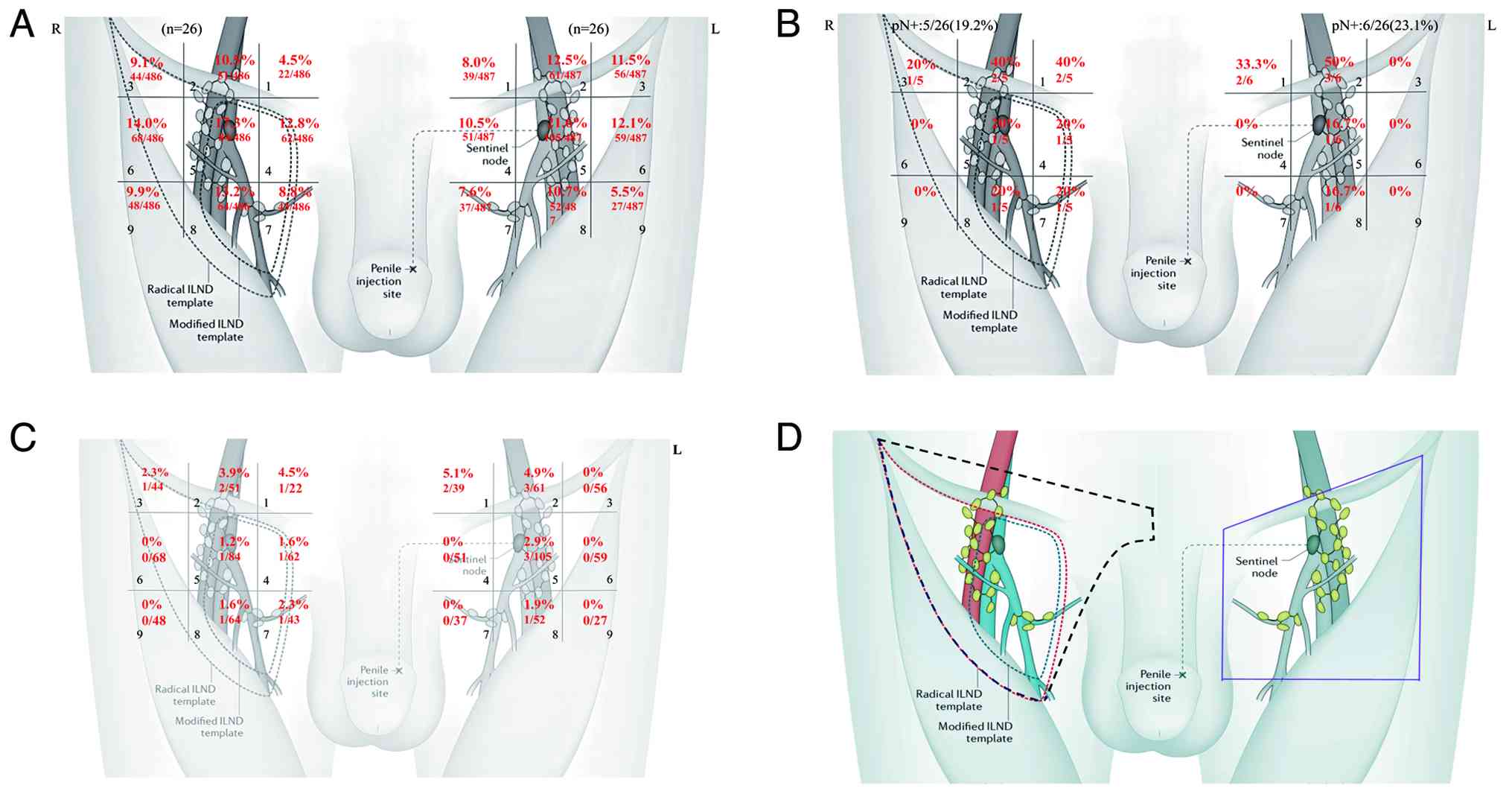
In this study, a mean of 18.81±7.22 left ILNs and
18.69±7.35 right ILNs were resected (Fig. 2A; Table
II). The number of ILNs in each region was relatively uniform.
Comparing the upper and lower regions unilaterally, regions 4, 5
and 6 exhibited a slightly higher number of ILNs. Unilaterally
comparing the left and right regions unilaterally, regions 2, 5,
and 8 had a slightly higher number of ILNs. However, there was no
significant difference between the bilateral groins (Fig. 2A; Table
II).
 | Table II.Mean number of lymph nodes dissected
in each region. |
Table II.
Mean number of lymph nodes dissected
in each region.
| Region | Right ILNs
(n=26) | Left ILNs
(n=26) | P-value |
|---|
| Total | 18.69±7.35 | 18.81±7.22 | 0.985 |
| Region 1 | 0.85±1.52 | 1.50±1.68 | 0.147 |
| Region 2 | 1.96±2.46 | 2.35±2.06 | 0.543 |
| Region 3 | 1.69±2.11 | 2.15±2.11 | 0.434 |
| Region 4 | 2.38±2.10 | 1.96±1.71 | 0.429 |
| Region 5 | 3.23±2.25 | 4.04±3.01 | 0.279 |
| Region 6 | 2.62±2.56 | 2.27±2.20 | 0.604 |
| Region 7 | 1.65±1.85 | 1.42±2.16 | 0.681 |
| Region 8 | 2.46±2.27 | 2.00±2.64 | 0.502 |
| Region 9 | 1.85±1.76 | 1.04±1.68 | 0.097 |
In these cases, 19.2% (5/26) of the right ILNs and
23.1% (6/26) of the left ILNs (Fig.
2B) had metastatic lymph nodes. Notably, region 2 was the most
frequently metastatic area, accounting for >40% of patients with
pN+, followed by regions 1, 5, and 8. Lymph node metastasis in
regions 3, 4, 6, 7 and 9 was less common, accounting for 0–20% of
patients with pN+. The number of positive ILNs is presented in
Table III, with no statistically
significant differences shown between the two groups. Region 1 had
the highest lymph node metastasis density, followed by regions 2, 5
and 8, with lower densities in regions 3, 4, and 7, and no lymph
node metastasis in regions 6 and 9. The metastasis density of ILNs
is presented in Fig. 2C. The
results indicated that positive ILNs were distributed in most areas
within the extended radical boundaries (excluding regions 6 and 9),
and were mainly concentrated in the upper medial regions (regions 1
and 2). This suggests that traditional rILND may not achieve
adequate tumor-free resection, and that there is an over-resection
in the lower lateral areas. Notably, the current guidelines for
rILND do not include region 1. Therefore, the results of this study
suggested that the boundaries of the dissection range during rILND
should be appropriately adjusted based on the traditional
boundaries. The upper medial boundary should be extended medially
upwards to include the area in front of the pubic bone, to reduce
the recurrence rate, while the lower lateral boundary can be
appropriately reduced to minimize unnecessary damage and surgical
complications. The lymph node dissection range recommended by the
present study is shown in Fig.
2D.
 | Table III.Number of positive lymph nodes. |
Table III.
Number of positive lymph nodes.
| Region | Positive right ILNs
(n=5) | Positive left ILNs
(n=6) | P-value |
|---|
| Case | 5/26 (19.2%) | 6/26 (23.1%) | 0.711 |
| Total | 0.35±0.85 | 0.35±0.75 | 1.000 |
| Region 1 | 0.08±0.27 | 0.08±0.27 | 1.000 |
| Region 2 | 0.08±0.27 | 0.12±0.33 | 0.646 |
| Region 3 | 0.04 ±0.20 | 0.00±0.00 | 0.327 |
| Region 4 | 0.04 ±0.20 | 0.00±0.00 | 0.327 |
| Region 5 | 0.04±0.20 | 0.04±0.20 | 1.000 |
| Region 6 | 0.00±0.00 | 0.00±0.00 | - |
| Region 7 | 0.04±0.20 | 0.00±0.00 | 0.327 |
| Region 8 | 0.04±0.20 | 0.04±0.20 | 1.000 |
| Region 9 | 0.00±0.00 | 0.00±0.00 | - |
First station metastasis distribution
of single positive ILN or region and lymphatic drainage
patterns
The sentinel lymph node is the first lymph node on
the lymphatic drainage pathway of the primary tumor, acting as a
barrier to prevent the spread of tumor cells through the lymphatic
system (17,19,20).
During the process of cancer dissemination, cancer cells typically
first metastasize to this lymph node, making the sentinel lymph
node the first stop for cancer cells to spread from the primary
tumor area to other locations (17,19,20).
The metastatic pathway of PSCC usually first flows through the
superficial lymphatics of the penis to the superficial ILNs, then
transfers downward to the deep ILNs, where it finally reaches the
iliac lymph nodes (21).
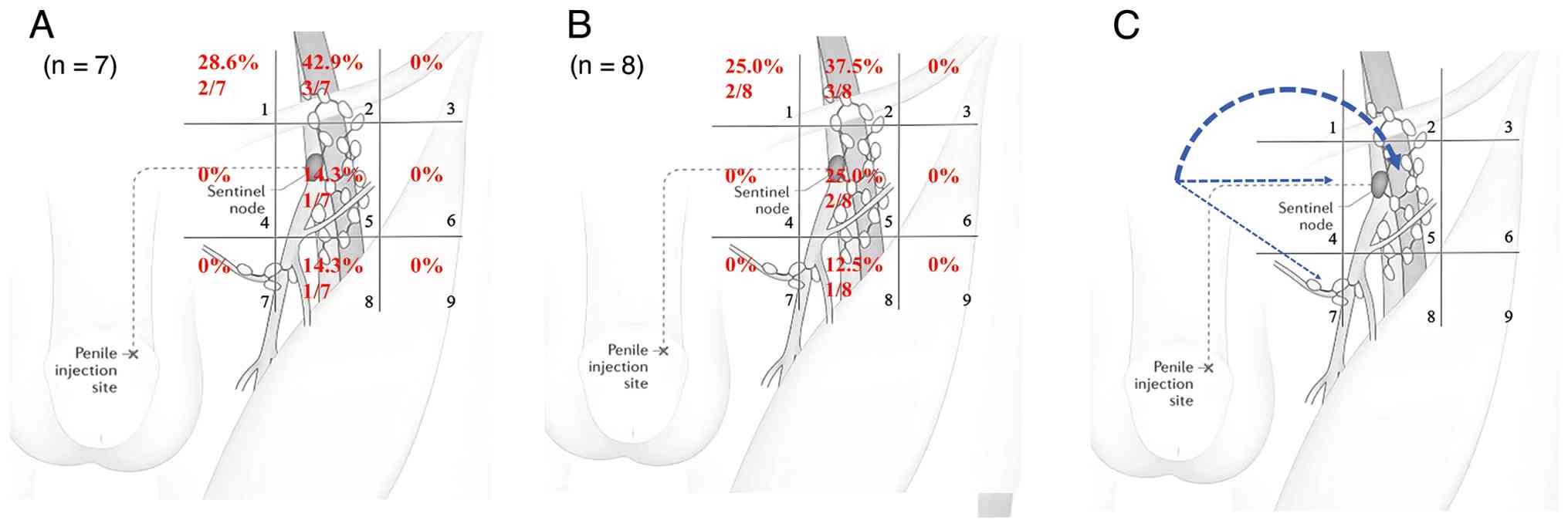
In the present study, single metastatic lymph nodes
in the unilateral inguinal region were analyzed to obtain direct
pathological evidence regarding the distribution of the first
positive sentinel lymph nodes in the superficial ILNs of PSCC
(Fig. 3A). In this study cohort, 7
cases that fulfilled the criteria for a single positive lymph node
in the inguinal region were analyzed separately (Fig. 3A). The distribution of
single-positive lymph nodes included regions 1, 2, 5 and 8. Region
2 was the most frequent site for positive lymph nodes, accounting
for 42.9%, followed by regions 1, 5 and 8, which accounted for
28.6, 14.3 and 14.3%, respectively (Fig. 3A). Additionally, the location of the
first-station metastatic region in each inguinal region was
investigated, which was defined as the only metastatic region in
each inguinal region (Fig. 3B). It
was found that the first-station metastatic regions occurred in
regions 1, 2, 5 and 8. Region 2 was the most common, accounting for
37.5%, followed by regions 1, 5 and 8, which accounted for 25.0,
25.0 and 12.5%, respectively (Fig.
3B). Based on the current data, it was possible to infer the
potential lymphatic metastatic pathway involved according to
postoperative pathological findings, and further studies may be
conducted using a dynamic sentinel node biopsy approach (Fig. 3C).
Region 1 had a high proportion of positive sentinel
lymph nodes, and the traditional rILND had limitations in the
medial upper boundary of the dissection range, which may omit
metastatic lymph nodes. This finding is of importance for the
treatment strategy of PSCC, as it suggests that it may be necessary
to reevaluate and adjust the scope of rLND to ensure more effective
treatment outcomes.
Region 1 lymph node positivity is a
risk factor for a poor prognosis
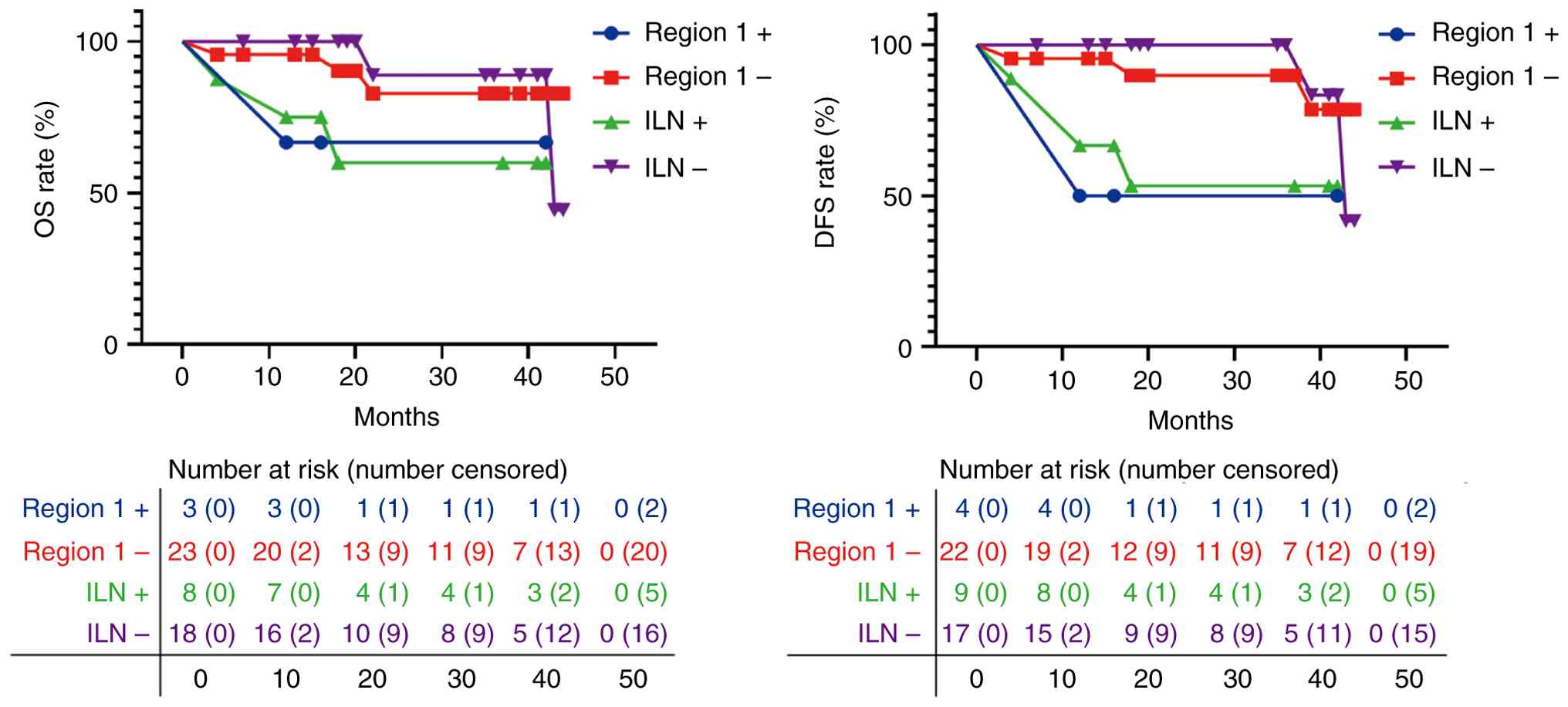
With a median follow-up time of 30 months
(interquartile range, 16.0–63.5 months) for the 26 enrolled
patients, 5 experienced recurrence and 4 died. Kaplan-Meier
survival analysis revealed that ILN positivity was associated with
inferior OS (HR, 8.9; 95% CI, 1.0–76.3; log-rank P=0.0207) and
inferior DFS (HR, 8.7; 95% CI, 1.3–57.6; log-rank P=0.0082)
compared with ILN negativity. Region 1 positivity was preliminarily
associated with inferior DFS (HR, 11.0; 95% CI, 0.8–152.3; log-rank
P=0.0464), but not associated with OS (HR, 6.4; 95% CI, 0.2–188.0;
log-rank P=0.2342) compared with region 1 negativity, as shown in
Fig. 4 and Table IV.
 | Table IV.K-M and Cox regression model for OS
and DFS. |
Table IV.
K-M and Cox regression model for OS
and DFS.
|
| OS | DFS |
|---|
|
|
|
|
|---|
|
| K-M analysis | Cox | K-M analysis | Cox |
|---|
|
|
|
|
|
|
|---|
| Variables | HR | 95% CI | P-value | P-value | HR | 95% CI | P-value | P-value |
|---|
| Age, years |
|
|
|
|
|
|
|
|
|
<52 | Reference | 0.113–5.828 | 0.847 | 0.482 | Reference | 0.103–5.621 | 0.928 | 0.968 |
|
≥52 | 0.811 |
|
|
| 0.761 |
|
|
|
| Smoking status |
|
|
|
|
|
|
|
|
| No | Reference |
| 0.002 | 0.268 | Reference |
| 0.002 | 0.218 |
|
Yes | 44.045 | 4.726–410.513 |
|
| 30.902 | 3.538–269.931 |
|
|
| N stage |
|
|
|
|
|
|
|
|
| 0 | Reference |
|
| 0.011 | Reference |
|
| 0.093 |
| 1 | 10.421 | 0.259–418.398 | 0.085 | 0.003 | 15.245 | 0.305–761.467 | 0.172 | 0.030 |
| 2 | 21.503 | 1.211–381.795 | 0.014 | 0.099 | 18.021 | 1.072–302.871 | 0.052 | 0.285 |
| Histological
type |
|
|
|
|
|
|
|
|
|
Well-differentiated | Reference |
|
| 0.321 | Reference |
|
| 0.191 |
|
Moderately differentiated | 4.752 | 0.405–55.753 | 0.220 | 0.151 | 5.561 | 0.516–59.907 | 0.165 | 0.074 |
| Poorly
differentiated | 25.790 | 0.369–1802.026 | 0.134 | 0.216 | 25.79 | 0.369–1802.026 | 0.134 | 0.139 |
| Total number of
left ILNs |
|
|
|
|
|
|
|
|
|
<18 | Reference | 0.123–6.377 | 0.885 | 0.529 | Reference | 0.144–7.600 | 0.823 | 0.899 |
|
≥18 | 0.885 |
|
|
| 1.046 |
|
|
|
| Total number of
right ILNs |
|
|
|
|
|
|
|
|
|
<18 | Reference | 0.555–30.080 | 0.156 | 0.463 | Reference | 0.634–37.659 | 0.090 | 0.162 |
|
≥18 | 4.084 |
|
|
| 4.885 |
|
|
|
| Total number of
ILNs |
|
|
|
|
|
|
|
|
|
<37 | Reference | 0.212–11.938 | 0.691 | 0.868 | Reference | 0.242–15.375 | 0.429 | 0.443 |
|
≥37 | 1.589 |
|
|
| 1.930 |
|
|
|
| Region
1-positive |
|
|
|
|
|
|
|
|
| No | Reference | 0.218–188.016 | 0.234 | 0.097 | Reference | 0.792–152.282 | 0.046 | 0.285 |
|
Yes | 6.405 |
|
|
| 10.984 |
|
|
|
| ILN-positive |
|
|
|
|
|
|
|
|
| No | Reference | 1.036–76.260 | 0.021 | 0.003 | Reference | 1.329–57.558 | 0.008 | 0.030 |
|
Yes | 8.889 |
|
|
| 8.747 |
|
|
|
Discussion
ILN metastasis is a notable prognostic factor in
patients with PSCC. According to the NCCN (16) and EAU guidelines (17,22),
patients with intermediate to high-risk PSCC should undergo
concurrent rILND, which has a significant curative value for the
treatment of early- to intermediate-stage disease. However, there
is currently no international consensus regarding the extent of
rILND, and incomplete dissection may lead to tumor recurrence and
affect treatment outcomes.
Since the concept of rILND was introduced by Daseler
et al (8) in 1948, the
optimal boundaries of rILND have been controversial due to a lack
of high-quality pathological data from patients with PSCC (9–14,23).
The root of this controversy is in the scarcity of clinical
evidence regarding the distribution and metastatic patterns of ILNs
in PSCC (14). In 2024, a
retrospective summary was conducted for clinical pathological data
from 414 patients with PSCC who underwent concurrent rILND, and for
the first time, the distribution of ILNs in PSCC was mapped,
particularly the distribution patterns of pathologically positive
lymph nodes (14). It was concluded
that the upper part of the lymph nodes had the highest positivity
rate for metastatic disease, which is similar to the conclusion of
the present study. It was also found that the upper inner part of
the saphenofemoral junction had the highest positivity rate (51.7%)
(14), a location similar to region
1 in this study, thus indirectly demonstrating the importance of
region 1.
At Yunnan Cancer Hospital, occasional lymph node
recurrence and metastasis have been observed in the area anterior
to the pubic bone, which is currently excluded from the existing
rILND ranges. Incomplete rILND may lead to cancer recurrence in the
inguinal region, which may become an extremely serious situation.
Even when patients undergo salvage rILND, nearly one-half of the
patients die after ~16.4 months, and these patients also experience
more complications (24). To
explore more effective dissection boundaries and improve the
treatment outcomes of PSCC, Yunnan Cancer Hospital has adopted a
method of extending rILND boundaries, including the area anterior
to the pubic bone, and pioneered the nine-section method to clarify
the number of lymph nodes, positivity rate and lymph node density
in each region, aiming to further clarify the standard of radical
dissection range of ILNs to ensure the best tumor control effect
for patients.
Within the expanded radical dissection boundaries,
ILNs appeared anywhere within the observed boundaries (in all 9
regions), with a cross-shaped distribution of ILNs, high density in
the central areas and lower density in the peripheral areas. A
greater number of ILNs were located in the central regions of 2, 4,
5, 6 and 8; however, up to 4.5–8.0% of ILNs were found in the
superior medial area of region 1. Compared with the five and
six-region division methods, the further anatomical division
refined the superficial ILNs (regions 1–9), appropriately extending
the upper boundary of the dissection range on the traditional
boundaries, and adding the ILNs in the anterior region of the pubic
symphysis (region 1).
Additionally, the results confirmed to some extent
the safety and feasibility of the reduced rILND proposed by
Catalona (15) and the common
femoral triangle-limited laparoscopic rILND in the laparoscopic
era, which narrowed the lateral boundary of dissection. More
importantly, it was confirmed that metastatic ILNs could be found
in region 1, which is not included in the traditional rILND range.
This suggested the necessity of including the anterior region of
the pubic symphysis (region 1) in the rILND range.
Penile cancer has a relatively low incidence rate,
with only 87 patients with penile cancer treated at Yunnan Cancer
Hospital between November 2021 and December 2024. Among these, 48
patients fulfilled the inclusion criteria and, after applying the
exclusion criteria, 26 remained. The small sample size may have
resulted in statistical bias. In small cohorts, the Cox model often
yields excessively wide confidence intervals (95% CI, 0.8–52.3),
resulting in insufficient statistical power to confirm the
significance of the hazard ratio. Consequently, the prognostic
value of region 1 positivity is interpreted as a preliminary
clinical trend that warrants further validation in larger,
multicenter cohorts. Therefore, future studies may require
multi-center collaboration to obtain more reliable and
generalizable findings. Compared with the traditional dissection
range, the inclusion of region 1 in the surgical field may increase
the potential risks of retropubic lymphatic leakage, infection,
flap necrosis and hernia formation. Since all enrolled patients to
date have developed ≥1 of these complications, further studies with
an expanded sample size and using the standardized complication
grading systems are necessary to confirm these findings. Future
investigations will also integrate surgical optimizations from
multiple centers to determine the most effective surgical approach
(such as single-port laparoscopic techniques) (25) and the optimal dissection range.
In summary, the present study used the nine-section
method to detail the distribution of ILNs in PSCC, firstly
including the pubic symphysis area in the rILND range for PSCC, and
provided direct pathological evidence of the distribution of ILNs
in PSCC. The results of the study emphasized the necessity of
including the area anterior to the pubic symphysis in the
dissection range and adjusting the traditional dissection
boundaries to reduce recurrence rates and surgical complications.
These findings provide clinical guidance for optimizing surgical
strategies for treating PSCC.
In conclusion, a metastatic density map of ILN
metastasises in penile cancer was established using a the
nine-section method. Region 1 contained positive lymph nodes and
was recommended for surgical inclusion. Regions 1, 2, 5 and 8 may
be the first-station regions of ILN metastasis. Region 1 positivity
was preliminarily associated with inferior DFS.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Scientific Research Fund
of Yunnan Provincial Department of Education (grant no.
2026J0278).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
CH was responsible for study design,
conceptualization, methodology, access to and verification of the
underlying data, formal analysis, validation, visualization,
writing of the original draft, and reviewing and editing of the
manuscript. LD performed database establishment, statistical
analysis and visualisation, and writing, reviewing and editing of
the manuscript. HW was responsible for conceptualization. YB was
responsible for the literature search and methodology and performed
the surgical resection. HY, YB, HJ, WW, JL performed sample
collection, management and labeling. RL performed the surgical
resection and project supervision. HS was responsible for
conceptualization, reviewing and editing of the manuscript, project
supervision, administration and funding acquisition. All authors
have read and approved the final manuscript. CH and HS confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Yunnan Cancer Hospital (Kunming, China; approval no. KYLX2022051)
and was registered with the Chinese Clinical Trial Registry, with
the clinical trial number ChiCTR2200064529 (registration date,
2022-10-11). All patients have provided consent for
participation.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Djajadiningrat RS, Graafland NM, van
Werkhoven E, Meinhardt W, Bex A, van der Poel HG, van Boven HH,
Valdés Olmos RA and Horenblas S: Contemporary management of
regional nodes in penile cancer-improvement of survival. J Urol.
191:68–73. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pagliaro LC and Crook J: Multimodality
therapy in penile cancer: When and which treatments? World J Urol.
27:221–225. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Woldu SL, Ci B, Hutchinson RC, Krabbe LM,
Singla N, Passoni NM, Clinton TN, Raj GV, Miller DS and Sagalowsky
AI: Usage and survival implications of surgical staging of inguinal
lymph nodes in intermediate- to high-risk, clinical localized
penile cancer: A propensity-score matched analysis. Urol Oncol.
36:159.e7–159.e17. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Brouwer OR, Albersen M, Parnham A, Protzel
C, Pettaway CA, Ayres B, Antunes-Lopes T, Barreto L, Campi R, Crook
J, et al: European association of Urology-American Society of
clinical oncology collaborative guideline on penile cancer: 2023
update. Eur Urol. 83:548–560. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chipollini J, Necchi A and Spiess PE:
Outcomes for patients with node-positive penile cancer: Impact of
perioperative systemic therapies and the importance of surgical
intervention. Eur Urol. 74:241–242. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mei H, Gu FL, Guo YL, et al: Urology
Operative Surgery[M]. 3rd ed. Beijing, People's Medical Publishing
House; China: pp. 646–648. 2008
|
|
7
|
Campbell RA, Slopnick EA, Ferry EK, Zhu H,
Kim SP and Abouassaly R: Disparity between pre-existing management
of penile cancer and NCCN guidelines. Urol Oncol.
35:531.e9–531.e14. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Daseler EH, Anson BJ and Reimann AF:
Radical excision of the inguinal and iliac lymph glands; a study
based upon 450 anatomical dissections and upon supportive clinical
observations. Surg Gynecol Obstet. 87:679–694. 1948.PubMed/NCBI
|
|
9
|
Shao Y, Hu X, Ren S, Liao D, Yang Z, Liu
Y, Lia T, Wu K, Xiong S, Yang W, et al: Comparison of different
surgical methods and strategies for inguinal lymph node dissection
in patients with penile cancer. Sci Rep. 12:25602022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cindolo L, Spiess PE, Bada M, Chipollini
JJ, Nyirády P, Chiodini P, Varga J, Ditonno P, Battaglia M, De
Nunzio C, et al: Adherence to EAU guidelines on penile cancer
translates into better outcomes: A multicenter international study.
World J Urol. 37:1649–1657. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang S, Du P, Tang X, An C, Zhang N and
Yang Y: Comparison of efficiency of video endoscopy and open
inguinal lymph node dissection. Anticancer Res. 37:4623–4628.
2017.PubMed/NCBI
|
|
12
|
Singh A, Jaipuria J, Goel A, Shah S,
Bhardwaj R, Baidya S, Jain J, Jain C and Rawal S: Comparing
outcomes of robotic and open inguinal lymph node dissection in
patients with carcinoma of the penis. J Urol. 199:1518–1525. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fankhauser CD, Ayres Be, Issa A, Albersen
M, Watkin N, Muneer A, Sangar V and Parnham A: Practice patterns
among penile cancer surgeons performing dynamic sentinel lymph node
biopsy and radical inguinal lymph node dissection in men with
penile cancer: A eUROGEN survey. Eur Urol Open Sci. 24:39–42. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tan X, Cai T, Wang Y, Wu Z, Zhou Q, Guo S,
Li J, Yuan G, Liu Z, Li Z, et al: Regional lymph node mapping in
patients with penile cancer undergoing radical inguinal lymph node
dissection-a retrospective cohort study. Int J Surg. 110:2865–2873.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Catalona WJ: Modified inguinal
lymphadenectomy for carcinoma of the penis with preservation of
saphenous veins: Technique and preliminary results. J Urol.
140:306–310. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hu J, Li H, Cui Y, Liu P, Zhou X, Liu L,
Chen H, Chen J and Zu X: Comparison of clinical feasibility and
oncological outcomes between video endoscopic and open inguinal
lymphadenectomy for penile cancer: A systematic review and
meta-analysis. Medicine (Baltimore). 98:e158622019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Vreeburg M, Donswijk ML, Albersen M,
Parnham A, Ayres B, Protzel C, Pettaway C, Spiess PE and Brouwer
OR: New EAU/ASCO guideline recommendations on sentinel node biopsy
for penile cancer and remaining challenges from a nuclear medicine
perspective. Eur J Nucl Med Mol Imaging. 51:2861–2868. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cubilla AL, Velazquez EF, Amin MB, Epstein
J, Berney DM and Corbishley CM; Members of the ISUP Penile Tumor
Panel, : The World Health Organisation 2016 classification of
penile carcinomas: A review and update from the International
Society of Urological Pathology expert-driven recommendations.
Histopathology. 72:893–904. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Naumann CM, Colberg C, Jüptner M, Marx M,
Zhao Y, Jiang P, Hamann MF, Jünemann KP, Zuhayra M and Lützen U:
Evaluation of the diagnostic value of preoperative sentinel lymph
node (SLN) imaging in penile carcinoma patients without palpable
inguinal lymph nodes via single photon emission computed
tomography/computed tomography (SPECT/CT) as compared to planar
scintigraphy. Urol Oncol. 36:92.e17–92.e24. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pizzocaro G: Editorial comment on:
Anatomical mapping of lymphatic drainage in penile carcinoma with
SPECT-CT: Implications for the extent of inguinal lymph node
dissection. Eur Urol. 54:8922008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Johnston MJ and Nigam R: Recent advances
in the management of penile cancer. F1000 Res 8, F1000 Faculty Rev.
–558. 2019.
|
|
22
|
Brouwer OR, Rumble RB, Ayres B, Martínez
DF, Oliveira P, Spiess PE, Johnstone PAS, Crook J, Pettaway CA and
Tagawa ST; Members of the EAU-ASCO Penile Cancer Update Panel, :
Penile cancer: EAU-ASCO Collaborative guidelines update Q and A.
JCO Oncol Pract. 20:33–37. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Marshall K, Nair SM, Willmore KE,
Beveridge TS and Power NE: Anatomical characterization of the
inguinal lymph nodes using microcomputed tomography to inform
radical inguinal lymph node dissections in penile cancer. J Surg
Oncol. 122:1785–1790. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chakiryan NH, Dahmen A, Bandini M,
Pederzoli F, Marandino L, Albersen M, Roussel E, Zhu Y, Ye DW,
Ornellas AA, et al: Patterns of recurrence following inguinal lymph
node dissection for penile cancer: Optimizing surveillance
strategies. J Urol. 206:960–969. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Perdonà S, Izzo A, Tufano A, Passaro F,
Quarto G, Aveta A, Contieri R, Pandolfo SD, Autorino R and Spena G:
Advancing surgical management of penile cancer: Single port
bilateral inguinal lymph node dissection. International Braz J
Urol. 51:e202406632025. View Article : Google Scholar : PubMed/NCBI
|