Gastric cancer (GC) represents a prominent public
health concern globally, characterized by limited effective
treatment options, poor prognosis and high mortality rates
(1). Anti-GC treatments, which are
primarily based on chemotherapy, radiation therapy and targeted
therapy, face a major challenge in the form of acquired resistance
(2). GC cells acquire resistance
through a range of mechanisms and associated signaling, all
involving both intrinsic and extrinsic factors (3). According to the Lauren classification,
GC is categorized into two major histopathological subtypes with
distinct biological behaviors: Intestinal-type and diffuse-type
(4). Intestinal-type GC, accounting
for 50–60% of cases, arises from intestinal metaplasia (a
premalignant lesion), exhibits glandular differentiation and is
associated with environmental risk factors [such as Helicobacter
pylori (H. pylori) infection, high-salt diet and
smoking] (5). It typically presents
as an exophytic or ulcerative mass in the distal stomach,
progresses slowly and has a relatively favorable prognosis
(5). By contrast, diffuse-type GC
(30–40% of cases) lacks glandular structure, is characterized by
signet ring cells with intracellular mucin accumulation and is
linked to genetic predispositions [such as cadherin-1 (CDH1)
mutations] (6). It invades
diffusely through the gastric wall (linitis plastica), has
early metastatic potential and confers a worse outcome. A third
subtype, mixed-type GC (10–15% of cases), combines features of both
intestinal and diffuse types and exhibits intermediate clinical
behavior (6).
GC progression follows a well-defined natural
history, from premalignant lesions (gastric atrophy, intestinal
metaplasia and dysplasia) to early stage localized tumors, and
ultimately to advanced disease with lymph node (LN) and distant
metastasis. Multiple factors modulate this process, including: i)
Microenvironment (ME) factors: Beyond hypoxia, H. pylori
infection (the strongest risk factor) induces chronic inflammation,
oxidative stress and epithelial dysregulation, which synergize with
hypoxia-inducible factors (HIFs) to promote carcinogenesis; ii)
genetic/epigenetic alterations: Mutations in TP53, CDH1 and
epidermal growth factor receptor 2 (ERBB2) and microsatellite
instability (MSI) contribute to subtype-specific progression, with
HIFs interacting with these pathways to enhance malignancy; and
iii) lifestyle and host factors: Obesity, alcohol consumption and
immune dysregulation further exacerbate GC development by altering
tumor ME (TME) hypoxia and HIF activation.
Pathological staging of GC adheres to the American
Joint Committee on Cancer/Union for International Cancer
Control/TNM system (8th edition) (7), which classifies tumors based on
primary tumor invasion depth (T), lymph node involvement (N) and
distant metastasis (M) (7).
Pathological staging (pTNM) is the gold standard for prognosis,
with pT1 (tumor invading mucosa/submucosa) and pT2 (invading
muscularis propria) defining early-stage GC (I–II), and pT3 (serosa
invasion) and pT4 (adjacent organ invasion) indicating locally
advanced disease (III) (8). LN
involvement is stratified as pN0 (no LN metastasis), pN1 (1–2
positive LNs), pN2 (3–6 positive LNs) and pN3 (≥7 positive LNs),
with pN3 indicating high metastatic potential (8). Clinical staging (cTNM), based on
preoperative imaging and endoscopy, guides treatment selection but
is less accurate than pTNM, particularly for LN assessment
(9). Clinically, early-stage GC
(cI–II) is curable with surgery, while advanced-stage GC (cIII–IV)
requires multimodal therapy (3).
Notably, HIF expression is associated with TNM stage: HIF-1α
upregulation is detected in 40–70% of GC cases, with higher levels
in pT3-pT4 tumors, pN2-pN3 LN status and M1 disease, making it an
independent prognostic marker (10).
Anti-GC treatments, primarily based on chemotherapy,
radiation therapy, targeted therapy and immunotherapy, face a major
challenge in acquired resistance. GC cells acquire resistance
through intrinsic (genetic mutations and metabolic reprogramming)
and extrinsic (TME hypoxia and immunosuppression) mechanisms, all
involving HIF-mediated signaling (11). For most patients with end-stage
cancer, the primary cause of treatment failure is resistance to
cancer therapy, and primary or acquired drug resistance
particularly acts as a major impediment in clinical oncology
(11). Therefore, studying drug
resistance mechanisms is of equal importance to drug development,
and both pharmacological factors, including insufficient drug
concentration at the tumor site, and cellular factors, such as
aberrant activation of signal transducer and activator of
transcription 3 (STAT3) signaling, enhanced tumor cell stemness,
epithelial-mesenchymal transition (EMT) and overexpression of
ATP-binding cassette (ABC) transporters, can contribute to clinical
resistance in gastric cancer (12).
The mechanisms governing drug resistance in tumors are precise yet
complex and multifactorial, and they can be grouped into three
categories: i) Inadequacies in pharmacokinetic properties; ii)
intrinsic factors of tumor cells; and iii) external conditions of
tumor cells in the TME (13).
Accumulating evidence has demonstrated that the TME
drives cancer progression through multiple mechanisms, with a
prominent role in mediating therapeutic resistance (14). On the one hand, the TME impairs drug
penetration, confers surviving cancer cells with proliferative and
antiapoptotic properties to facilitate therapeutic resistance, and
induces common morphological alterations of the disease (15). On the other hand, it is enriched
with soluble factors secreted by both tumor and stromal cells,
which in turn contribute to aberrant cell proliferation,
pathological angiogenesis, tumor metastasis and the development of
drug resistance (16). As the rapid
and uncontrolled proliferation of tumor cells restricts oxygen
availability, leading to insufficient blood supply or hypoxia, this
condition has become a typical ME feature in nearly all solid
tumors (16). Hypoxia elicits
intratumoral oxygen gradients, and subsequently contributes to the
plasticity and heterogeneity of tumors while enhancing the
emergence of more aggressive, metastatic phenotypic traits
(11). Moreover, the TME undergoes
remodeling under hypoxic conditions, which in turn affects the
stemness, chemoresistance, epithelial-mesenchymal transition (EMT)
and angiogenesis of GC cells and tissues (Fig. 1) (17–19).
In this process, the increased expression of HIFs is a pivotal
hallmark. The HIF family consists of HIF-1, HIF-2 and HIF-3, and
serves a central role in cellular mechanisms triggered in response
to hypoxia (20,21).
HIF-1, the most important component of the HIF
family, is predominantly made up of two subunits, namely HIF-1α and
HIF-1β (22). Under normal oxygen
concentrations, HIF-1α undergoes degradation and fails to maintain
stable expression, whereas under hypoxic conditions, it
translocates into the nucleus and dimerizes with HIF-1β to promote
the transcription of downstream genes (Fig. 2) (23). The specific mechanism is as follows:
Under normoxic conditions, HIF-1α undergoes prolyl hydroxylation
catalyzed by prolyl hydroxylase (PHD). This modified HIF-1α is
subsequently recognized and bound by the von Hippel-Lindau tumor
suppressor protein and upon this binding, is ubiquitinated and
ultimately degraded (24–26). Under hypoxic conditions, PHD
inactivation blocks oxygen-dependent prolyl hydroxylation, which
inhibits HIF-1α degradation; the accumulated HIF-1α subsequently
enters the nucleus, combines with HIF-1β to form a dimer, and this
dimer regulates the expression of hypoxia-related genes with the
participation of transcriptional co-activators such as histone
acetyltransferase p300, finally facilitating cellular adaptation to
hypoxia (27–31).
HIF-2 is composed of HIF-2α and HIF-2β, with HIF-2α
serving as the primary functional subunit that is abundant in
tissues including vascular endothelial cells and fetal lung
fibroblasts, and upon activation, HIF-2α binds to aryl hydrocarbon
receptor nuclear translocator (ARNT) to form a heterodimer
(32,33). Hypoxia-inducible factor 3 (HIF-3) is
a relatively understudied member of the HIF gene family, and it is
composed of HIF-3α and HIF-3β subunits. The HIF-3α gene undergoes
complex transcriptional regulation, generating multiple
alternatively spliced HIF-3α variants through the use of different
promoters and transcription initiation sites (34). These variants exhibit differential
expression patterns, which are tightly regulated not only by
hypoxia but also by multiple non-hypoxic factors (34). Notably, these non-hypoxic regulatory
factors include pro-inflammatory cytokines (which regulate HIF-3α
via NF-κB-dependent epigenetic modifications), insulin-mediated
PI3K/protein kinase B (AKT) signaling (which modulates HIF-3α
stability through phosphorylation) and the von Hippel-Lindau (VHL)
E3 ubiquitin ligase complex (which targets specific HIF-3α variants
for ubiquitination and degradation in an oxygen-dependent manner)
(34). Full-length HIF-3α protein
functions as an oxygen-regulated transcriptional activator
(35).
In 2003, HIF-1α was first identified as exhibiting
stable expression in GC tissues and was involved in the initiation
and progression of GC (36). Since
this initial discovery, numerous subsequent investigations have
confirmed that HIF exerts a regulatory effect on the initiation and
progression of GC and the development of HIF-targeted agents could
represent a promising therapeutic strategy for advanced GC
(37–40). Therefore, the present review focuses
on the role of HIF in GC, specifically addressing its regulatory
effects on GC cell proliferation, metastasis, apoptosis, drug
resistance, angiogenesis, stemness and metabolism, and also covers
several HIF-targeted therapeutic agents for GC.
Tumor cells induce the formation of new blood
vessels as an adaptive response to low oxygen and nutrient levels,
a process termed de novo angiogenesis (11). The newly generated blood vessels
exhibit leakiness due to their discontinuous endothelium and
impaired lymphatic drainage, ultimately leading to vascular
hyperpermeability and increased permeation (41). Hence, hypoxia induces vascular
leakage and dysregulated lymphatic drainage in the tumor,
ultimately resulting in elevated interstitial fluid pressure
(42). The TME denotes the local
biological environment where solid tumors reside, which comprises
cancer cells and adjacent stromal cells, specifically normal host
cells (such as fibroblasts, various immune cells and
blood/lymphatic cells) recruited by cancer cells and embedded in
densely packed extracellular matrix (43–45).
The secondary development of adverse metabolic and physical MEs
leads to an imbalance between the positive and negative regulators
that govern the activation and dysregulation of angiogenesis,
desmoplasia and inflammation (44,46–48).
Most neoplasms harbor hypoxic regions, and the formation of an
abnormal vasculature alongside a hypoxic ME promotes aberrant
angiogenesis, desmoplasia and inflammation, all of which contribute
to tumor progression and therapeutic resistance (49,50). A
previous review highlighted that cancers evade host regulatory
mechanisms and disrupt systemic homeostasis by secreting a spectrum
of neurohormonal mediators (including cytokines, neurotransmitters
and pituitary hormones) and immune factors, as validated in both
human and animal models (51). It
is hypothesized that these tumor-derived molecules enable
bidirectional crosstalk with central neuroendocrine axes (such as
hypothalamic-pituitary-adrenal and thyroid axes) and peripheral
autonomic/sensory nerves, allowing tumors to hijack host
homeostatic regulation to support their progression (51). This paradigm suggests that malignant
cells actively manipulate central neuroendocrine and immune
systems, reshaping systemic balance at the expense of the host, to
facilitate tumor expansion (51).
HIF-1α orchestrates hypoxia-induced signaling that
regulates multiple steps of the metabolic substrate transport
cascade, mainly glucose and lactate transport, to support cellular
adaptation to hypoxic microenvironments (52). Research findings have demonstrated
that cancer-associated fibroblasts (CAFs) and myeloid cells
facilitate tumor metastasis (53).
Under hypoxic conditions, activated HIF-1α enhances the activity of
Snail and Twist, two transcription factors that downregulate
E-cadherin expression and drive EMT (54–56).
Of note, although EMT-associated signaling is not required for the
metastatic process, it can enhance multiple malignant traits of
tumor cells, including invasion, senescence, cancer stem cell-like
phenotype and chemoresistance (57). HIF-1α can additionally modulate the
expression of enzymes responsible for collagen fiber polymerization
and alignment regulation, as well as integrin activity, to promote
cancer migration (52). Moreover,
hypoxia induces leakiness and compression of blood and lymphatic
vessels, a process mediated by HIF-regulated factors including
angiopoietin-2, vascular endothelial growth factor (VEGF) and
angiopoietin-like 4, thereby facilitating the transmigration of
metastatic cancer cells through the vessel wall (58).
The hypoxic ME supports glycolysis and subsequent
lactic acid generation mediated by glycolytic key enzymes and
lactate dehydrogenase A; this excess lactic acid accumulation leads
to an acidic extracellular pH (59–61).
Moreover, HIF can facilitate the reverse conversion of carbon
dioxide and water, generated via the activation of carbonic
anhydrase IX or XII into bicarbonate ions
(HCO3−). HCO3− then
diffuses across the cell membrane, leading to
HCO3− accumulation in the TME and a
subsequent reduction in extracellular pH (62). Numerous studies have demonstrated
that reduced intracellular pH of endosomes and lysosomes in tumor
cells can facilitate tumor metastasis via protease activation
(42,63). Indeed, changes in extracellular pH
induce drug resistance via suppressing cellular and humoral immune
functions, as acidic pH is prevalent at sites of inflammation and
other immunologically active regions (64–67). A
reduction in pH mainly inhibits the chemotaxis, respiratory
activity and bactericidal ability of polymorphonuclear leukocytes
(68–70). Under acidic pH conditions, impaired
cytotoxicity and proliferation of lymphocytes have been reported,
and similarly, cytotoxic T lymphocytes exhibit decreased lysis of
various tumor cell lines in this acidic extracellular environment,
while neutralization of T cell effector function and tumor acidity
can improve the response to immunotherapy (63,71–73).
Furthermore, studies focusing on macrophages and eosinophils have
indicated that acidic conditions induce the activation of
complement proteins and the alternative complement pathway, which
is accompanied by increased binding of antibodies to leukocytes
under lower pH (63,72,74).
Notably, reactive oxygen species (ROS) levels are
demonstrated to be elevated in cancer cells under hypoxic
conditions (75). Decreased oxygen
utilization impairs electron transfer through the mitochondrial
electron transport chain (ETC) complexes, which in turn promotes
electron leakage from the ETC and ultimately results in the
overproduction of ROS (76).
Moreover, excessive ROS production disrupts genomic stability and
impairs the function of DNA repair pathways (77). ROS are further capable of inducing
cell survival or apoptosis through a mechanism known as oxidative
stress, thereby contributing to enhanced cytotoxicity and apoptosis
(78). Notably, at high
concentrations (10–30 µM), ROS can induce damage to cellular
biomolecules including proteins, DNA and RNA, and trigger mutations
that either drive carcinogenesis in normal cells or confer
multidrug resistance (MDR) in cancer cells (79). However, most cancer cells still
survive under internal oxidative stress, hence avoiding apoptosis
and developing resistance to chemotherapy (80). Elevated ROS exposure can drive
cancer cell resistance via activating redox-sensitive transcription
factors, including nuclear factor κB nuclear factor
(erythroid-derived 2)-like factor 2, c-Jun and HIF-1α (80). Subsequently, the activation of these
genes enhances the activation of the antioxidant system and
promotes the expression of cell survival proteins (80). In addition, ROS promote the
transition from apoptosis to autophagy in methotrexate-resistant
choriocarcinoma Jeg-3 cells, thereby supporting the survival of
these cells against methotrexate (81). ROS can also stimulate the
differentiation of cancer stem cells, thus promoting EMT and
inducing metabolic reprogramming involved in the resistance of
cancer cells (79).
Hypoxic stress induces immunosuppression through
regulating angiogenesis, as well as promoting immune evasion and
tumor resistance (82–84). Notably, macrophages serve as a key
component of the immune infiltrate in solid tumors via their
differentiation into tumor-associated macrophages (TAMs), which are
preferentially localized in hypoxic regions of tumors (85). Furthermore, cytokines derived from
tumors can induce the conversion of TAMs into polarized type 2 (M2)
macrophages, which exhibit enhanced immunosuppressive activity and
thereby contribute to tumor progression (86–88).
Additionally, myeloid-derived suppressor cells (MDSCs) directly
contribute to immune tolerance, and in hypoxic zones, HIF-1
directly modulates the differentiation and function of MDSCs with
these tumor-derived MDSCs exhibiting greater immunosuppressive
activity than splenic MDSCs (89).
Previous studies have shown the upregulation of the
expression of programmed death-ligand 1 (PD-L1) under hypoxia
(89–91). HIF-1 serves as a key regulator of
both PD-L1 mRNA and protein expression, specifically by directly
binding to a hypoxia response element (HRE) within the proximal
promoter of PD-L1 (89). The
originally elevated immunosuppressive function of tumor derived
MDSCs under hypoxia was found to be abrogated following PD-L1
blockade. Consistent with PD-L1 blockade, the hypoxia-driven
upregulation of IL-6 and IL-10 in MDSCs was notably attenuated
(92). Currently, immunotherapeutic
strategies that elicit antitumor immunity exhibit limited efficacy
due to the diverse mechanisms by which tumors evade
immunosurveillance (92). Antibody
blockade of the T-cell immune checkpoint receptors programmed cell
death protein-1 (PD-1) and cytotoxic T-lymphocyte-associated
protein 4 exhibit poor efficacy in certain tumors due to sparse or
absent T cells in the TM, and hypoxia-driven modulation of T-cell
exclusion and apoptosis helps sustain this state (93–95).
While T cells can enter hypoxic tumors, hypoxia-driven
acidification of the extracellular environment impairs their
ability to proliferate or exert cytotoxic effector functions
(72,96). Taken together, tumor hypoxia
predicts poor outcomes across numerous types of cancer, and hypoxia
plays a key role in establishing and maintaining tumor immune
privilege or resistance to immunotherapy (97).
HIF-1 is a basic helix-loop-helix (bHLH)
transcription factor that mediates homeostatic adaptations to
hypoxic conditions (98). HIF-1
functions as a heterodimer composed of HIF-1α (encoded by the
HIF-1A gene) in complex with HIF-1β (encoded by the ARNT gene)
(99). Previous studies reported
the assignment of the HIF-1A gene to mouse chromosome 12 and the
HIF-1A gene to human chromosome 14, respectively, with HIF-1A
mapped to human chromosome 14q21-q24 via somatic cell hybrid
analysis and fluorescence in situ hybridization and HIF-1A
localized through interspecific backcross analysis to a region of
>30 centimorgans on mouse chromosome 12 that exhibits
conservation of synteny with the segment of human chromosome 14
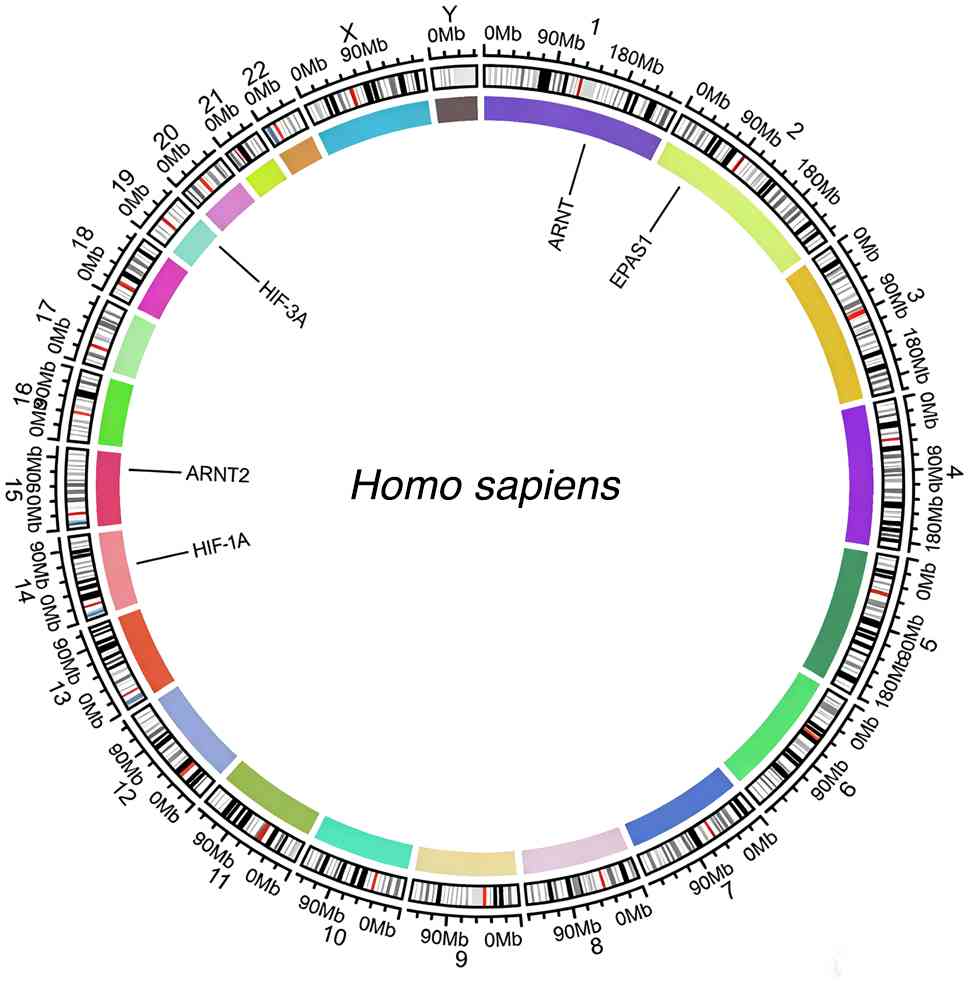
spanning from PAX9 at 14q12-q13 to IGHC at 14q32.33 (Fig. 3) (98,100).
Furthermore, ARNT, also known as HIF-1β, is chromosomally localized
to human chromosome 1q21 (Fig. 3)
and structurally categorized as a bHLH-Per-Arnt-Sim (PAS) family
transcription factor, featuring conserved functional domains
including the bHLH domain (for dimerization and DNA binding), PAS
A/B domains (for protein-protein interactions, especially with
HIF-1α) and a C-terminal transactivation domain (for mediating
transcriptional activation of downstream target genes) (101).
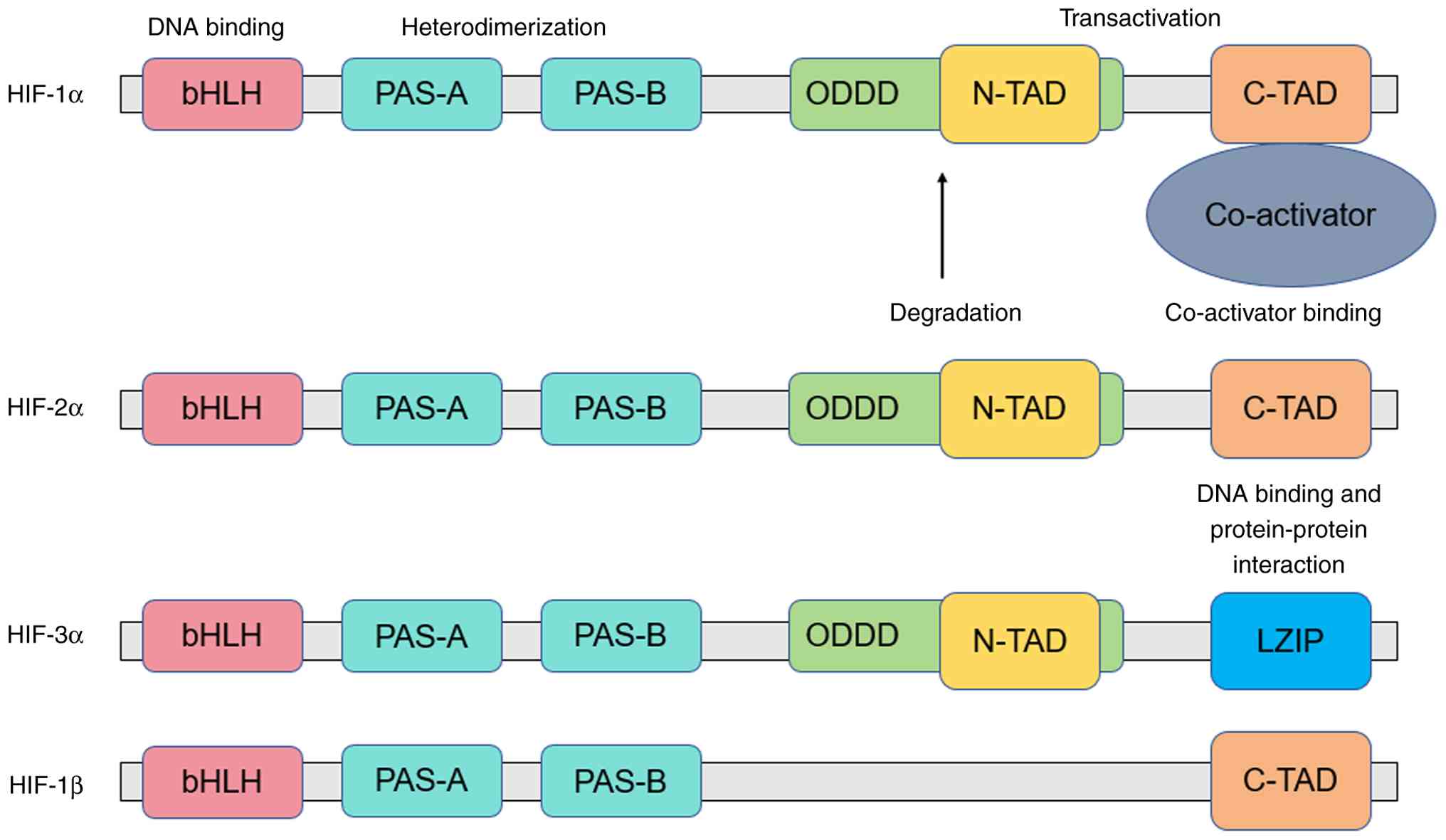
Both HIF-1α and HIF-1β possess a bHLH domain, which
is critical for their binding to the HRE within the promoter region
of target genes. The N-terminal region of HIF-1α additionally
contains PAS domains, which enable its heterodimerization with
HIF-1β (Fig. 4) (102,103). Two transactivation domains (TADs)
are located in the C-terminal segment of HIF-1α: An N-terminal TAD
(N-TAD) and a C-terminal TAD (C-TAD), and these TADs are
responsible for modulating the interaction between HIFs and
co-activators (104). Notably,
HIF-1α harbors an oxygen-dependent degradation domain (ODDD) that
overlaps with the N-TAD-a structural characteristic that
differentiates HIF-1α from HIF-1β (Fig.
4). The ODDD functions as a recognition motif for the VHL tumor
suppressor protein and is involved in regulating the stability of
HIF-1α in an oxygen-dependent manner (104). When two proline residues within
the ODDD are hydroxylated by oxygen-sensitive (PHD)
domain-containing proteins, HIF-1α undergoes rapid proteasomal
degradation (105). In contrast to
HIF-1α, which has a short half-life, HIF-1β maintains a stable
protein expression level because it lacks the ODDD and N-TAD
(Fig. 4).
Numerous previous studies have demonstrated that
HIF-1 exerts important regulatory effects on proliferation,
metastasis, metabolism, apoptosis, angiogenesis, cancer cell
stemness, drug resistance and other aspects of GC cells (106–112). HIF-1α can promote GC cell
proliferation by binding to the promoter of hypoxia-induced
proliferation-associated long non-coding RNA (lncRNA; HYPAL) to
promote its transcription, which in turn activates the
Wnt/β-catenin signaling pathway via the HYPAL/microRNA
(miR)-431-5p/CDK14 axis and induces GC cell proliferation (106). An additional study further
revealed that HIF-1α can bind to the promoter region of miR-17-5p
to activate the transcription of both pre-miR-17-5p and mature
miR-17-5p; this miR-17-5p then binds to the untranslated region of
programmed cell death 4 (PDCD4), a suppressor gene in GC that
primarily functions to inhibit cell proliferation, ultimately
leading to the degradation of PDCD4 mRNA (113). Furthermore, whilst the
upregulation of HIF-1α can promote the proliferation of GC cells,
tumor suppressor gene Linc-pint is capable of inhibiting GC cell
proliferation by downregulating HIF-1α expression (114). Additionally, 3-deazaneplanocin A,
a histone methyltransferase inhibitor that likely acts by
regulating histone methylation, can inhibit HIF-1A expression and
thus suppress the proliferation of BGC-823 GC cells; observations
from a study indirectly confirm the critical role of HIF-1A in GC
cell proliferation (115). An
additional study demonstrated that lncRNA ZEB2-AS1 is upregulated
in GC and regulates cell proliferation via the miR-143-5p/HIF-1α
axis (116).
Metastasis is the primary cause of mortality in most
human malignant tumors, including hepatocellular carcinoma, GC and
colorectal cancer (117–119). HIF-1α can promote the metastasis
of GC by facilitating the EMT of GC cells (107). Furthermore, HIF-1α can directly
bind to the promoter of liver X receptor-α (LXRα) to promote its
transcription; elevated LXRα levels subsequently activate the EMT
of GC cells, thereby enhancing the metastatic capacity of GC
(120). Another line of research
has shown that HIF-1α can induce GC cells to secrete exosomes
enriched with miR-301a-3p, thereby promoting GC cell metastasis via
the miR-301a-3p/PHD3/HIF-1α positive feedback loop (121). Functional studies have shown that
collagen triple helix repeat containing 1 (CTHRC1) increases C-X-C
chemokine receptor type 4 (CXCR4) expression by upregulating
HIF-1α, ultimately promoting the migration and invasion of GC
cells. Inhibiting HIF-1α expression can reduce CXCR4 expression and
suppress the migration and invasion of GC cells, which demonstrates
that the HIF-1α/CXCR4 signaling pathway mediates the promoting
effect of CTHRC1 on the migration and invasion of GC cells
(122). Moreover, hypoxia promotes
migration and invasion of GC cells by activating HIF-1α and
inhibiting N-myc downstream-regulated gene 2 (NDRG2) associated
signaling pathways, which are widely recognized to exert
tumor-suppressive roles via constraining cell motility,
invasiveness and metastatic potential in gastric cancer and
multiple malignancies (123).
Apoptosis refers to autonomous and orderly
gene-controlled cell death that maintains internal environmental
stability and is not a phenomenon of autologous injury under
pathological conditions, but an active death process aimed at
adapting to the living environment (124–126). HIF-1 can upregulate the expression
of the adhesion molecule MGr1-Ag/37LRP by activating ERK, thereby
inhibiting apoptosis in GC cells (108). Research has further shown that HIF
can inhibit the apoptosis of GC cells by upregulating integrin
alpha-5 (127). However, the role
of HIF in GC cell apoptosis remains controversial, as some studies
have demonstrated that HIF can promote GC cell apoptosis (128,129). To summarize, the role of HIF in GC
cell apoptosis remains controversial to date, necessitating further
investigations to clarify its function in GC.
Drug resistance represents a major barrier to
achieving effective cancer treatment, notably compromising the
therapeutic efficacy of both conventional chemotherapeutics (such
as platinum-based drugs) and targeted agents (such as EGFR
inhibitors) (130,131). Clinically, the emergence of drug
resistance often leads to treatment failure, frequent disease
recurrence and substantially reduced overall survival rates in
patients with various cancers, including lung, breast and
gastrointestinal malignancies, underscoring its critical impact on
clinical outcomes (132–134). The molecular mechanisms driving
cancer drug resistance are complex and multifactorial, involving
processes such as upregulation of drug efflux transporters (such as
P-glycoprotein), acquired mutations in drug target genes (such as
KRAS or BRAF), dysregulation of cell survival signaling pathways
(such as PI3K/Akt/mTOR) and the adaptive changes in the hypoxic or
immunosuppressive TME (82,135–139). Despite decades of research
advancing the understanding of these resistance mechanisms,
translating this basic science knowledge into clinically applicable
strategies to overcome or prevent resistance remains a central
challenge in oncology. Therefore, addressing drug resistance,
whether through developing novel agents that target
resistance-related molecules, designing rational combination
therapies to bypass resistance or implementing personalized
treatment strategies based on molecular profiling has become a key
priority to improve the durability and effectiveness of cancer
treatment. HIF-1 can promote drug resistance in GC cells by
regulating the expression of pyruvate kinase muscle 1 (PKM1), a
gene associated with chemotherapy resistance in GC (109). Knocking down HIF-1α via small
interfering RNA can attenuate the drug resistance of GC cells and
enhance the cytotoxic effect of 5-fluorouracil (5-FU) on these
cells (140). A study has
demonstrated that HIF-1α directly binds to the promoter of miR-27a
to upregulate its expression, with miR-27a subsequently promoting
drug resistance in GC cells by inhibiting the expression of
multidrug resistance protein 1 (MDR1)/P-gp, LRP and Bcl-2 (141). The role of HIF-1 in GC drug
resistance is relatively well-established; HIF-1 enhances drug
resistance in GC, a finding corroborated by numerous studies
(142–144).
Anti-angiogenesis has long been recognized as a
pivotal strategy in the design of antitumor drugs, primarily
targeting the abnormal neovascularization that tumors depend on to
obtain nutrients, oxygen and a pathway for metastatic spread
(145,146). By interfering with the activation
of pro-angiogenic signaling pathways (such as the VEGF/VEGFR
pathway) or the maturation of tumor-associated blood vessels,
anti-angiogenic therapies effectively disrupt the ‘vascular niche’
of tumors, thereby suppressing their growth and limiting their
ability to invade surrounding tissues (147). This approach has been clinically
validated: Numerous anti-angiogenic agents (such as bevacizumab, a
VEGF-specific monoclonal antibody) have been approved for the
treatment of solid tumors such as colorectal and non-small cell
lung cancer, further confirming its status as a core antitumor
strategy (148–150). Studies have shown that HIF-1α can
promote angiogenesis in GC, a process potentiated by natriuretic
peptide receptor A, the primary receptor for atrial natriuretic
peptide, which plays a significant role in driving GC development
and progression (151–154). Additionally, another study has
indicated that HIF-1α can facilitate angiogenesis in gastric cancer
by upregulating the expression of VEGF-A (148). HIF-1α drives gastric angiogenesis
via the β-catenin/VEGF signaling pathway, thereby promoting GC
progression (110,153,155,156).
Cancer stem cells (CSCs) represent a small
subpopulation within tumors that generate heterogeneous tumor cells
while possessing self-renewal capacity and are regarded as critical
factors driving tumor progression (157–159). One study demonstrated that HIF-1α
can drive the progression of GC by promoting the stemness of GC
cells (111). Other research has
confirmed that HIF-1α further facilitates peritoneal dissemination
of GC by enhancing the stemness of its cells (160). Currently, research on the role of
HIF in gastric CSCs remains limited, and no drugs targeting CSCs
have achieved successful clinical application; thus, this role
warrants further investigation.
Metabolism is intricately linked to tumor
initiation, progression and therapeutic response, with tumor cells
exhibiting hallmark metabolic reprogramming that distinguishes
their glucose, lipid and protein metabolism from that of normal
cells. Notably, tumor cells typically rely on aerobic glycolysis
(the Warburg effect) even under oxygen-sufficient conditions, this
preference for glycolysis enables rapid production of ATP and
biosynthetic precursors (such as pyruvate and lactate) to support
unchecked proliferation, whereas normal cells primarily use
oxidative phosphorylation for efficient energy generation (161). Beyond glucose metabolism, tumor
cells also upregulate de novo lipid synthesis (rather than
utilizing exogenous lipids) to build cell membranes for expanding
cell populations, and they exhibit dysregulated protein metabolism,
including increased uptake of amino acids (such as glutamine) and
enhanced protein synthesis, to meet the demands of maintaining
stemness and resisting nutrient stress (162,163). Collectively, these metabolic
alterations are not merely adaptive changes but active drivers of
tumor biology, highlighting the importance of targeting
tumor-specific metabolic pathways as a potential therapeutic
strategy. Studies have demonstrated that HIF-1α promotes GC
progression by enhancing glycolysis in GC cells (112,164,165). HIF-1α also regulates glycolysis in
GC cells via the FOXO4/Lactate dehydrogenase A (LDHA) axis and
thereby influences the progression of GC cells (164). Another line of research has shown
that HIF-1α can promote glycolysis in GC cells via the circular RNA
(circ-)MAT2B/miR-515-5p axis and thereby facilitate the occurrence
and development of GC (165).
Notably, extracellular matrix protein-1 facilitates GC cell
metastasis and glucose metabolism by inducing the integrin β4/focal
adhesion kinase/SOX2/HIF-1α signaling pathway (166). Moreover, miR-186 can inhibit
aerobic glycolysis in GC via HIF-1α regulation (167). While 5-FU is known to exert an
antitumor effect by inhibiting nucleic acid metabolism, HIF-1,
which is closely associated with GC metabolism, can promote GC
progression by enhancing glycolysis under hypoxic conditions,
making it a promising target for GC drug development (168,169).
HIF-2 is a member of the basic bHLH-PAS
transcription factor family that mediates cellular adaptive
responses to hypoxia, with distinct functional specificity compared
with HIF-1 (99,170,171). Functionally, HIF-2 acts as a
heterodimer, typically composed of the oxygen-sensitive HIF-2α
subunit [encoded by the endothelial PAS domain-containing protein 1
(EPAS1) gene] and a constitutively expressed β subunit, which can
be either an ARNT (HIF-1β) or ARNT2 (HIF-2β) depending on tissue
context (170,172,173). The EPAS1 gene, which encodes
HIF-2α, is localized to the human chromosome 2p21 region, while the
ARNT2 gene, which encodes HIF-2β, is localized to the human
chromosome 15q23-q25 region (Fig.
3). Structurally, both HIF-2α and its β partners contain a
conserved bHLH domain at the N-terminus, which is essential for
sequence-specific binding to the HRE (core sequence 5′-TACGTG-3′)
in target gene promoters (174–177).
HIF-2α has been linked to the malignant behavior of
several types of cancers. In cervical cancer, it was identified as
a hub gene among hypoxia and ferroptosis related genes (178). Furthermore, in a previous study,
HIF-2α was markedly upregulated in cervical cancer tissues and
could increase the proliferation, invasion and migration of
cervical cancer cells while reducing apoptosis (179). In esophageal squamous cell
carcinoma (ESCC), molecular deregulation of EPAS1 was observed,
with 7.5% of patients harboring mutations in this gene; these
alterations were associated with tumor location and T stages
(180). Suppression of EPAS1 in
ESCC cells led to reduced proliferation, wound healing, migration
and invasion (180). HIF-2β
(ARNT2) also plays a role in the development of cancer. In oral
squamous cell carcinoma (OSCC), ARNT2 was found to be
downregulated, and overexpression of ARNT2 led to decreased
cellular proliferation, suggesting its potential as a therapeutic
target against OSCC progression (181). In GC, the expression of ARNT2 was
lower in cancer tissues compared with adjacent non-tumor tissues
and overexpression of ARNT2 inhibited cell proliferation (182).
HIF-2α plays a crucial role in hypoxia-induced
pathways, which are relevant to the development and progression of
GC. In hypoxic conditions, HIF-2α is stabilized and activates the
transcription of numerous genes involved in cell survival,
angiogenesis and metabolism. In a study on non-small cell lung
cancer, hypoxic-stabilized HIF-2α proteins transactivated DNA
methyltransferase 1-the major maintenance DNA methyltransferase
responsible for preserving genomic methylation patterns following
DNA replication by preferentially targeting hemi-methylated DNA and
catalyzing methyl group transfer to cytosine nucleotides-which in
turn caused promoter hypermethylation and transcription inhibition
of HIF-2α itself, creating a negative-feedback regulation loop
(183). This mechanism may also be
present in GC cells, where hypoxia is a common ME condition.
Hypoxia can drive the upregulation of HIF-2α, leading to increased
expression of genes such as VEGF, which promotes angiogenesis
(178). Angiogenesis is essential
for tumor growth as it provides the necessary nutrients and oxygen
to the growing tumor cells (123).
In addition, HIF-2α can regulate genes involved in cell metabolism,
enabling cancer cells to adapt to the hypoxic environment (179). For example, it may upregulate
genes involved in glycolysis, allowing cancer cells to generate
energy even in low-oxygen conditions (179). Currently, research on HIF-2 in GC
remains insufficiently clear, and a number of its mechanisms have
not yet been fully elucidated.
HIF-3 is a heterodimer, which consists of HIF-α and
oxygen-insensitive β subunits and is a member of the ARNT family
(101). HIF-3α is located at
chromosome 19q13.13–13.2 (Fig. 3).
HIF-3α shares a conserved bHLH-PAS domain architecture with HIF-1α
and HIF-2α but exhibits unique structural modifications (101,170). The N-terminal bHLH domain mediates
DNA binding to HREs with the consensus sequence ‘TACGTG’ (170). Adjacent to the bHLH domain are two
PAS domains (PAS-A and PAS-B), which facilitate heterodimerization
with the β-subunit (ARNT) and ligand binding (170,171). Notably, the PAS-B domain of HIF-3α
contains a 510 Å3 hydrophobic cavity, a structural
feature absent in HIF-1α and HIF-2α, enabling it to selectively
bind endogenous lipids such as oleoylethanolamide and
monoacylglycerols (99,170,171). This lipid-binding capability
suggests HIF-3α acts as a metabolic sensor integrating hypoxia and
lipid signaling (170,171). Unlike HIF-1α and HIF-2α, HIF-3α
lacks the C-TAD (170). Instead,
its C-terminus contains a unique leucine zipper domain and a LxxLL
motif, which mediates protein-protein interactions with
co-regulators such as HSP90 (170,171). The absence of C-TAD reduces the
intrinsic transcriptional activity of HIF-3α, but it retains
partial activity through N-TAD, which shares 58 and 52% sequence
identity with HIF-1α and HIF-2α, respectively (171).
HIF-3 is a critical regulator in cancer progression,
primarily through its α-subunit (HIF-3α) and its splice variants,
such as inhibitory PAS domain protein (IPAS) (184). Unlike HIF-1α and HIF-2α, HIF-3α
acts as a context-dependent modulator rather than a canonical
transcription factor. For instance, IPAS competitively inhibits
HIF-1α binding to HREs, thereby suppressing downstream genes
involved in angiogenesis (such as VEGF) and glycolysis (such as
GLUT1), which are pivotal for tumor growth under hypoxic conditions
(184,185).
Currently, relevant studies on the role of HIF-3 in
GC are scarce and the specific mechanisms remain unclear. While the
pathogenic roles of HIF-1α and HIF-2α in GC cancer such as
promoting angiogenesis, glycolysis and EMT have been extensively
characterized, HIF-3 has long been overlooked due to its structural
complexity (such as multiple splice variants with potentially
divergent functions) and the lack of specific tools to dissect its
isoform-specific effects. Notably, existing investigations on HIF-3
in GC are often limited to in vitro cell line experiments or
small-scale tissue samples, with inconsistent findings regarding
its pro-tumor or antitumor properties-certain studies suggest
inhibitory effects via antagonizing HIF-1α, while others imply
pro-metastatic roles through undefined signaling axes (182,184,185). Furthermore, the crosstalk between
HIF-3 and key oncogenic pathways in GC (such as TGF-β/Smad,
Wnt/β-catenin or PI3K/Akt) has rarely been systematically explored,
and its clinical relevance such as associations with patient
prognosis, tumor stage or response to anti-hypoxic therapies
remains largely unvalidated. This knowledge gap not only hinders a
comprehensive understanding of the hypoxic regulatory network in GC
but also limits the development of targeted strategies that harness
HIF-3 for therapeutic intervention.
Small-molecule drugs (SMDs) are typically defined as
organic compounds with a molecular weight generally not exceeding
1,000 Da and most of them possess physicochemical properties such
as good cell membrane permeability and structural modifiability,
which lay the foundation for their subsequent biological activity
(186). Due to their favorable
pharmacokinetic characteristics (such as easy absorption,
distribution and metabolic adjustability) and relatively low
production costs, SMDs have been widely applied in the clinical
treatment of various diseases, including tumors, cardiovascular
disorders and infectious diseases, and have long occupied a core
position in therapeutic regimens (187). The theoretical system supporting
SMD research and development which covers key fields such as
medicinal chemistry, structure-activity relationship analysis and
high-throughput screening has become increasingly mature, providing
solid technical support for the efficient discovery and
optimization of novel small-molecule therapeutic agents (188–190).
Numerous SMDs capable of suppressing the
progression of GC through HIF targeting have been identified
(Table I). A previous study
demonstrated that apigenin, a flavonoid component present in
traditional medicines, fruits and vegetables, inhibits
HIF-1α-induced autophagy-related cell death (191). Low-dose tipifarnib-a
non-peptidomimetic quinolinone farnesyltransferase inhibitor (FTI)
that exerts antineoplastic effects by inhibiting protein
farnesylation, suppressing mTOR signaling, and reducing ROS
levels-inhibits tumors by suppressing the expression of HIF-1α, and
dextran sulphate-a sulfated polysaccharide (anionic derivative of
dextran) with anticoagulant, antiviral, and protein-sequestering
properties-can inhibit EMT in GC cells by inhibiting the expression
of HIF (192,193). A study constructed R8-modified
vinorelbine-plus-schisandrin B liposomes, validated via in
vitro experiments on BGC-823 cells and in vivo
experiments on nude mice bearing BGC-823 cell xenografts, were
shown to synergistically induce GC apoptosis, enhance tumor cell
targeting and inhibit GC metastasis by downregulating HIF-1α,
thereby exerting antitumor effects and providing a safe and
effective therapeutic strategy for GC (194). Moreover, numerous small-molecule
therapeutic agents exert an inhibitory effect on GC progression by
targeting HIF (195–201). Despite the identification of
numerous small-molecule agents that inhibit GC progression by
targeting HIF, none have yet translated to clinical application,
highlighting the need for additional basic and clinical
research.
GC development and progression are driven by the
interplay of environmental, genetic, microenvironmental and host
factors, all of which interact with HIF-mediated hypoxia
responses.
High-salt intake (≥5 g/day) induces gastric mucosal
damage and hypoxia, upregulating HIF-1α (205). Red meat and processed food
consumption increases N-nitrosamine exposure, which synergizes with
HIFs to promote DNA damage (206).
Conversely, fruits, vegetables (rich in flavonoids) and green tea
(contains epigallocatechin gallate) inhibit HIF-1α and reduce GC
risk (207).
Cigarette smoke contains polycyclic aromatic
hydrocarbons that stabilize HIF-1α and induce EMT (208). Heavy alcohol consumption (≥3
drinks/day) causes gastric mucosal hypoxia and acetaldehyde
accumulation, enhancing HIF-2α expression (209).
i) Driver mutations: TP53 mutations (40–50% of GC)
enhance HIF-1α stability by inhibiting PHD activity; ii) CDH1
mutations (5–10% of diffuse-type GC) disrupt E-cadherin, promoting
hypoxia and HIF-1α-dependent EMT (210); and iii) ERBB2 amplification
(10–15% of GC) activates PI3K/Akt signaling to upregulate HIF-1α
and VEGF (210).
DNA methylation of CDKN2A and DNA mismatch repair
protein Mlh1 (MSI-high GC) correlates with HIF-1α upregulation
(211). Histone acetylation
(p300/CBP) enhances HIF-1α transcriptional activity (212). lncRNAs (such as H19 and MALAT1)
and miRNAs (such as miR-143 and miR-515-5p) regulate HIF expression
in a subtype-specific manner (165,213).
CAFs secrete TGF-β and IL-6 to stabilize HIF-1α,
promoting desmoplasia and hypoxia (214). Diffuse-type GC has a higher CAF
density than intestinal-type GC, contributing to its TME rigidity
and hypoxia (214). These
alterations collectively foster a highly immunosuppressive,
pro-tumorigenic microenvironment that accelerates diffuse-type GC
progression, invasion and therapeutic resistance.
TAMs and MDSCs enhance HIF-1α expression via
cytokine secretion (IL-1β and TNF-α) (215). CD8+ T cell exhaustion, mediated by
HIF-1α/PD-L1 signaling, correlates with poor prognosis in advanced
GC (215). This forms a positive
immunosuppressive feedback loop: TAMs and MDSCs secrete IL-1β and
TNF-α to upregulate HIF-1α, which in turn transcriptionally
elevates PD-L1 expression in tumor and stromal cells; elevated
PD-L1 binds to its receptor on CD8+ cytotoxic T cells, triggering
functional exhaustion, impairing tumor-specific cytotoxicity and
enabling immune evasion, thereby facilitating unrestricted GC
proliferation, invasion and metastasis, and ultimately resulting in
dismal clinical outcomes.
The natural history of GC varies by subtype:
Intestinal-type GC progresses from premalignant lesions to invasive
cancer over 10–20 years, while diffuse-type GC develops rapidly
(3–5 years) with early metastasis (216). Pathological staging strongly
influences prognosis. The 5-year survival rates are 90–95% for
stage 1, 60–70% for stage II, 30–40% for stage III and <10% for
stage IV (216). HIF expression
correlates with staging. HIF-1α positivity increases from 20–30% in
stage I to 70–80% in stage IV, while HIF-2α is more frequently
expressed in stage III–IV intestinal-type GC (217). LN metastasis (pN2-pN3) is
associated with higher HIF-1α levels (99), as hypoxic tumors have increased
lymphatic invasion potential.
White light endoscopy: The standard screening tool
for GC, but limited in detecting early lesions (sensitivity
60–70%).
i) Narrow-band imaging. Improves visualization of
mucosal microvessels and glands, increasing early GC detection
sensitivity to 85–90% (218); ii)
confocal laser endomicroscopy. Enables in vivo histological
evaluation, distinguishing benign from malignant lesions with
90–95% accuracy (219); and iii)
artificial intelligence (AI)-assisted endoscopy. AI algorithms
analyze endoscopic images to identify early GC and premalignant
lesions, with sensitivity and specificity comparable to expert
endoscopists (220). AI-based
systems also correlate mucosal hypoxia (via vessel density) with
HIF expression, providing prognostic information (221).
Liquid biopsies: Circulating tumor DNA (ctDNA)
detection of driver mutations (TP53 and ERBB2) and HIF pathway
genes (HIF-1A and EPAS1) enable non-invasive staging and treatment
monitoring (222). ctDNA levels
correlate with TNM stage, with detectable ctDNA in 20% of stage I,
50% of stage II, 70% of stage III and 90% of stage IV GC (223).
i) Positron emission tomography-computed
tomography: F-FDG uptake reflects HIF-mediated glycolysis, aiding
in staging (sensitivity 80–85% for LN metastasis) and treatment
response assessment (1,3); ii) magnetic resonance imaging (MRI):
Diffusion-weighted imaging and dynamic contrast-enhanced MRI
evaluate tumor hypoxia and angiogenesis, correlating with HIF-1α
expression (1,3); and iii) contrast-enhanced ultrasound.
Assesses tumor vascularity, with hypovascular lesions (diffuse-type
GC) showing higher HIF-1α expression (1,3).
i) Immunohistochemistry (IHC): HIF-1α, HIF-2α and
PD-L1 IHC staining aids in subtype classification and prognosis
prediction. HIF-1α IHC positivity (≥10% tumor cells) is associated
with diffuse-type GC and poor prognosis (224); and ii) next-generation sequencing
(NGS): Tumor tissue NGS identifies MSI status, tumor mutational
burden and HIF pathway alterations (such as HIF-1A amplification,
EPAS1 mutations), guiding targeted and immunotherapy selection
(224). MSI-high GC (10–15% of
cases) exhibits lower HIF-1α expression and better response to PD-1
inhibitors (225).
The present review systematically synthesizes the
regulatory roles, molecular mechanisms and therapeutic potential of
HIFs in GC, a malignancy with high global morbidity and mortality
and suboptimal outcomes for advanced-stage patients. First,
hypoxia, an iconic hallmark of the TME in GC, drives TME remodeling
by inducing acidification ROS accumulation and immunosuppression
(such as M2 polarization of TAMs, MDSC recruitment and PD-L1
upregulation) and aberrant angiogenesis, all of which converge to
promote GC progression and therapeutic resistance (11,14).
Central to this hypoxic response is the HIF family, whose three
subtypes (HIF-1, HIF-2, HIF-3) exhibit distinct structural features
and functional heterogeneity in GC. HIF-1, the most
well-characterized subtype, exerts a pleiotropic oncogenic role in
GC: Its α-subunit (HIF-1α) regulates GC cell proliferation via the
HYPAL/miR-431-5p/CDK14 or HIF-1α/miR-17-5p/PDCD4 axis, drives EMT
through direct transactivation of LXRα or exosomal miR-301a-3p
secretion, enhances chemoresistance by modulating PKM1 or
MDR1/P-gp, promotes angiogenesis via VEGF-A or β-catenin/VEGF
signaling and supports aerobic glycolysis (Warburg effect) through
the FOXO4/LDHA or circ-MAT2B/miR-515-5p axis (106,113,120,121). By contrast, HIF-2 (primarily
HIF-2α encoded by EPAS1) remains less well studied in GC; existing
evidence links it to vascular endothelial function and GC cell
metabolism, but its precise role in proliferation, metastasis or
resistance, especially across GC subtypes (intestinal vs. diffuse),
remains elusive (226–228). HIF-3, the least explored subtype,
exhibits context-dependent activity; its splice variant IPAS
competitively inhibits HIF-1α-mediated transactivation of pro-tumor
genes, yet some studies suggest pro-metastatic effects via
uncharacterized signaling axes, highlighting unresolved
controversies in the functional role of HIF-3 (35,184,185,229–233). Finally, a panel of small-molecule
agents (such as apigenin, tipifarnib and schisandrin B) have been
summarized that target HIFs to suppress GC progression, although
all remain confined to preclinical models without clinical
translation.
The findings presented in the present review
reinforce the centrality of HIFs as integrators of hypoxic TME
signals and GC malignant phenotypes, aligning with the broader
consensus that hypoxia-driven HIF activation is a non-negotiable
step in solid tumor progression (10,11).
Induction of melanogenesis robustly upregulates HIF-1α expression
and its downstream target genes related to angiogenesis and
glycolysis, along with a subset of HIF-1-independent genes, in
melanoma cells and tissues, highlighting the critical role of
HIF-1α in mediating the metabolic regulatory effects of specific
cellular processes (234). A
critical insight is the bidirectional crosstalk between HIFs and
the TME: HIFs not only respond to hypoxia but also actively reshape
the TME. For example, HIF-1α-induced lactate production (via LDHA)
acidifies the extracellular ME, which in turn inhibits T-cell
cytotoxicity and enhances MDSC immunosuppressive activity;
concurrently, ROS generated by mitochondrial dysfunction under
hypoxia stabilizes HIF-1α, creating a positive feedback loop that
amplifies pro-tumor signaling (235–237). This crosstalk explains why
targeting HIFs may simultaneously disrupt multiple hallmarks of GC
(such as angiogenesis, immune evasion and metabolic reprogramming)
rather than acting on a single pathway.
Another notable observation is the subtype-specific
functional diversity of HIFs. HIF-1α is widely regarded as a
universal oncogenic driver in GC, consistent with its
overexpression in most GC tissues and association with poor
prognosis (34). By contrast,
HIF-2α exhibits tissue-specific expression (such as vascular
endothelial cells) and may play a more context-dependent role. For
instance, it promotes angiogenesis in GC via VEGF but has also been
linked to differentiation in certain cell types (34). HIF-3α, meanwhile, represents a
‘double-edged sword’; its full-length isoform may act as a
transcriptional activator, while splice variants such as IPAS
antagonize HIF-1α (34). This
heterogeneity underscores the need for subtype-specific targeting
strategies, as pan-HIF inhibitors may disrupt physiological hypoxia
responses (such as wound healing) and cause off-target toxicity
which is a major barrier to clinical translation. From a
therapeutic perspective, the preclinical efficacy of HIF-targeted
small molecules (Table I) validates
HIFs as viable targets for GC. For example, low-dose tipifarnib
inhibits HIF-1α without affecting Ras signaling, addressing the
toxicity concerns of earlier FTIs; R8-modified
vinorelbine-schisandrin B liposomes enhance tumor targeting while
downregulating HIF-1α, overcoming the poor bioavailability of
numerous small molecules (192,194). However, a critical translational
gap persists as no HIF-targeted agent has entered clinical trials
for GC, despite the Food and Drug Administration approval of
Welireg (a HIF-2α inhibitor) for von Hippel-Lindau syndrome. This
gap likely stems from two challenges: i) The lack of predictive
biomarkers to identify patients most likely to benefit (such as
HIF-1α-high vs. HIF-2α-high GC); and ii) the potential for adaptive
resistance, as HIF inhibition may activate compensatory pathways
(such as PI3K/Akt/mTOR). Future studies should prioritize
combinatorial strategies, for example, HIF inhibitors combined with
immune checkpoint blockers given that HIF-1α regulates PD-L1
expression; or HIF inhibitors plus anti-angiogenic agents (such as
bevacizumab), to synergistically disrupt the vascular niche.
While the present review discusses the current
state of HIF research in GC, it is important to acknowledge several
limitations that reflect broader gaps in the field. First, the
evidence base for HIF-2α and HIF-3α in GC remains sparse and
heterogeneous. Most studies on HIF-2α focus on other cancers (such
as cervical cancer and ESCC), and GC-specific data are limited to
in vitro experiments with a narrow panel of cell lines (such
as BGC-823or SGC-7901). Similarly, HIF-3α studies in GC are
confined to small-scale tissue samples or cell models, with
conflicting reports on its pro- vs. antitumor roles. This paucity
of data limits our ability to draw definitive conclusions about
these subtypes, emphasizing the need for more GC-specific research.
Second, the review focuses primarily on HIF-mediated mechanisms in
generic GC, overlooking the profound heterogeneity of the disease.
GC is classified into distinct histological (intestinal, diffuse
and mixed) and molecular subtypes, and HIF expression and function
may vary across subtypes. For example, diffuse-type GC is
characterized by EMT and high metastatic potential, but it remains
unclear whether HIF-1α serves a more prominent role in this subtype
than in intestinal-type GC. This lack of subtype-specific analysis
limits the clinical relevance of the present review, as
personalized therapy requires understanding HIF biology in distinct
GC subsets. Third, the discussion of SMDs is restricted to
preclinical studies, with no consideration of pharmacokinetic and
pharmacodynamic challenges or clinical safety. Numerous HIF
inhibitors exhibit poor solubility, short half-lives or off-target
effects (such as inhibiting PHDs involved in collagen synthesis),
which may explain their failure to advance to clinical trials.
Additionally, the review does not address the potential for
HIF-independent hypoxia responses (such as AMPK activation or
autophagy), which may contribute to resistance to HIF-targeted
therapy and warrants further exploration. Finally, the review
relies heavily on in vitro and xenograft models, which
poorly recapitulate the human GC TME. Xenograft models use
immunocompromised mice, precluding analysis of HIF-mediated immune
interactions (such as T-cell exhaustion or MDSC recruitment)
(14); 3D cell culture models,
while more physiologically relevant, are underutilized in HIF-GC
research. These model limitations may lead to overestimation of HIF
inhibitor efficacy, highlighting the need for more translational
models (such as patient-derived xenografts, organoids) in future
studies.
In summary, the present review highlights that
HIFs, particularly HIF-1α, play a central role in GC progression by
integrating hypoxic TME signals to regulate proliferation,
metastasis, metabolism and immune evasion. While HIF-2α and HIF-3α
remain less well understood, their subtype-specific functions
suggest they may represent untapped targets. Despite notable
preclinical progress in developing HIF-targeted small molecules,
translational challenges (such as lack of biomarkers and adaptive
resistance) and gaps in our understanding of HIF biology (such as
subtype heterogeneity and pathway crosstalk) persist. Addressing
these limitations through future research will be critical to
realizing the potential of HIF-targeted therapy as a novel strategy
for improving outcomes in patients with GC.
Funding: No funding was received.
Not applicable.
JX contributed to writing the original draft and
performing the literature search. GD contributed to writing, review
and editing, literature search and visualization. YW contributed to
manuscript writing, reviewing and editing and literature search. CL
contributed to writing, reviewing, editing, conceptualization,
supervision and project administration. All authors read and
approved the final version of the manuscript. Data authentication
is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Sundar R, Nakayama I, Markar SR, Shitara
K, van Laarhoven HWM, Janjigian YY and Smyth EC: Gastric cancer.
Lancet. 405:2087–2102. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tanprasert P, Limpakan Yamada S,
Chattipakorn SC, Chattipakorn N and Shinlapawittayatorn K:
Targeting mitochondria as a therapeutic anti-gastric cancer
approach. Apoptosis. 27:163–183. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Luo D, Liu Y, Lu Z and Huang L: Targeted
therapy and immunotherapy for gastric cancer: Rational strategies,
novel advancements, challenges, and future perspectives. Mol Med.
31:522025. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li Y, Xue XW, Luo YF, Wu HW, Chen J and
Zhou WX: Clinicopathologic features of gastric adenocarcinoma based
on the revised Lauren's classification. Zhonghua Bing Li Xue Za
Zhi. 47:486–491. 2018.(In Chinese). PubMed/NCBI
|
|
5
|
Shi W, Wang Y, Xu C, Li Y, Ge S, Bai B,
Zhang K, Wang Y, Zheng N, Wang J, et al: Multilevel proteomic
analyses reveal molecular diversity between diffuse-type and
intestinal-type gastric cancer. Nat Commun. 14:8352023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu LW, Jang SJ, Shapiro C, Fazlollahi L,
Wang TC, Ryeom SW and Moy RH: Diffuse gastric cancer: A
comprehensive review of molecular features and emerging
therapeutics. Target Oncol. 19:845–865. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tanaka S, Komatsu S, Ohta A, Furuke H,
Kumano T, Imura K, Shimomura K, Ikeda J, Taniguchi F and Shioaki Y:
Validation of the 8th edition of the UICC TNM classification for
stage III gastric cancer. Gan To Kagaku Ryoho. 46:502–504.
2019.PubMed/NCBI
|
|
8
|
Liu Z, Liu H, Zhang Y, Yang Y and Gao H:
Prognostic enhancement in gastric cancer through the integration of
inflammatory indices into the pTNM-inflammation staging system
(pTNM-I). J Inflamm Res. 18:11869–11882. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jeong O, Jung MR, Kang JH and Ryu SY:
Prognostic performance of preoperative staging: Assessed by using
multidetector computed tomography-between the new clinical
classification and the pathological classification in the eighth
American joint committee on cancer classification for gastric
carcinoma. Ann Surg Oncol. 27:545–551. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sumiyoshi Y, Kakeji Y, Egashira A,
Mizokami K, Orita H and Maehara Y: Overexpression of
hypoxia-inducible factor 1alpha and p53 is a marker for an
unfavorable prognosis in gastric cancer. Clin Cancer Res.
12:5112–5117. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jing X, Yang F, Shao C, Wei K, Xie M, Shen
H and Shu Y: Role of hypoxia in cancer therapy by regulating the
tumor microenvironment. Mol Cancer. 18:1572019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gatti L and Zunino F: Overview of tumor
cell chemoresistance mechanisms. Methods Mol Med. 111:127–148.
2005.PubMed/NCBI
|
|
13
|
Gottesman MM: Mechanisms of cancer drug
resistance. Annu Rev Med. 53:615–627. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sun Y: Tumor microenvironment and cancer
therapy resistance. Cancer Lett. 380:205–215. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen F, Zhuang X, Lin L, Yu P, Wang Y, Shi
Y, Hu G and Sun Y: New horizons in tumor microenvironment biology:
Challenges and opportunities. BMC Med. 13:452015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wu T and Dai Y: Tumor microenvironment and
therapeutic response. Cancer Lett. 387:61–68. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Saxena K, Jolly MK and Balamurugan K:
Hypoxia, partial EMT and collective migration: Emerging culprits in
metastasis. Transl Oncol. 13:1008452020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu X, Huang Z, Chen Q, Chen K, Liu W, Liu
G, Chu X, Li D, Ma Y, Tian X and Yang Y: Hypoxia-induced epigenetic
regulation of miR-485-3p promotes stemness and chemoresistance in
pancreatic ductal adenocarcinoma via SLC7A11-mediated ferroptosis.
Cell Death Discov. 10:2622024. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lee PWT, Koseki LR, Haitani T, Harada H
and Kobayashi M: Hypoxia-inducible factor-dependent and independent
mechanisms underlying chemoresistance of hypoxic cancer cells.
Cancers (Basel). 16:17292024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Huang Y, Lin D and Taniguchi CM: Hypoxia
inducible factor (HIF) in the tumor microenvironment: Friend or
foe? Sci China Life Sci. 60:1114–1124. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
McKeown SR: Defining normoxia, physoxia
and hypoxia in tumours-implications for treatment response. Br J
Radiol. 87:201306762014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yang C, Zhong ZF, Wang SP, Vong CT, Yu B
and Wang YT: HIF-1: Structure, biology and natural modulators. Chin
J Nat Med. 19:521–527. 2021.PubMed/NCBI
|
|
23
|
Knutson AK, Williams AL, Boisvert WA and
Shohet RV: HIF in the heart: Development, metabolism, ischemia, and
atherosclerosis. J Clin Invest. 131:e1375572021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yu F, White SB, Zhao Q and Lee FS:
HIF-1alpha binding to VHL is regulated by stimulus-sensitive
proline hydroxylation. Proc Natl Acad Sci USA. 98:9630–9635. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Maxwell PH, Wiesener MS, Chang GW,
Clifford SC, Vaux EC, Cockman ME, Wykoff CC, Pugh CW, Maher ER and
Ratcliffe PJ: The tumour suppressor protein VHL targets
hypoxia-inducible factors for oxygen-dependent proteolysis. Nature.
399:271–275. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Epstein AC, Gleadle JM, McNeill LA,
Hewitson KS, O'Rourke J, Mole DR, Mukherji M, Metzen E, Wilson MI,
Dhanda A, et al: C. elegans EGL-9 and mammalian homologs define a
family of dioxygenases that regulate HIF by prolyl hydroxylation.
Cell. 107:43–54. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xue X, Kang JB, Yang X, Li N, Chang L, Ji
J, Meng XK, Zhang HQ, Zhong Y, Yu SP, et al: An efficient strategy
for digging protein-protein interactions for rational drug design-A
case study with HIF-1α/VHL. Eur J Med Chem. 227:1138712022.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
He W, Batty-Stuart S, Lee JE and Ohh M:
HIF-1α hydroxyprolines modulate oxygen-dependent protein stability
via single VHL interface with comparable effect on ubiquitination
rate. J Mol Biol. 433:1672442021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang Y, Zou P, He L, Shao J, Tang Y and Li
J: CBL aggravates Ang II-induced cardiac hypertrophy via the
VHL/HIF-1α pathway. Exp Cell Res. 405:1127302021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chakraborty AA: Coalescing lessons from
oxygen sensing, tumor metabolism, and epigenetics to target VHL
loss in kidney cancer. Semin Cancer Biol. 67:34–42. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Cargill K, Hemker SL, Clugston A, Murali
A, Mukherjee E, Liu J, Bushnell D, Bodnar AJ, Saifudeen Z, Ho J, et
al: Von Hippel-Lindau acts as a metabolic switch controlling
nephron progenitor differentiation. J Am Soc Nephrol. 30:1192–1205.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Erbel PJA, Card PB, Karakuzu O, Bruick RK
and Gardner KH: Structural basis for PAS domain heterodimerization
in the basic helix-loop-helix-PAS transcription factor
hypoxia-inducible factor. Proc Natl Acad Sci USA. 100:15504–15509.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Diao X, Shang Q, Guo M, Huang Y, Zhang M,
Chen X, Liang Y, Sun X, Zhou F, Zhuang J, et al: Structural basis
for the ligand-dependent activation of heterodimeric AHR-ARNT
complex. Nat Commun. 16:12822025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li M, Li G, Yang X, Yin W, Lv G and Wang
S: HIF in gastric cancer: Regulation and therapeutic target.
Molecules. 27:48932022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Duan C: Hypoxia-inducible factor 3
biology: Complexities and emerging themes. Am J Physiol Cell
Physiol. 310:C260–C269. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Park JH, Kim TY, Jong HS, Kim TY, Chun YS,
Park JW, Lee CT, Jung HC, Kim NK and Bang YJ: Gastric epithelial
reactive oxygen species prevent normoxic degradation of
hypoxia-inducible factor-1alpha in gastric cancer cells. Clin
Cancer Res. 9:433–440. 2003.PubMed/NCBI
|
|
37
|
Li H, Jia Y and Wang Y: Targeting HIF-1α
signaling pathway for gastric cancer treatment. Pharmazie. 74:3–7.
2019.PubMed/NCBI
|
|
38
|
She X, Geng L, Zhao Q, Guo H, Rong G, Luo
Y, Li X, Xu L, Ran F and Liu S: Targeting hypoxia-induced
HIF-1α/JMJD3/Notch axis in gastric cancer therapy. J Bioenerg
Biomembr. 57:107–117. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li D, Wang T, Lai J, Zeng D, Chen W, Zhang
X, Zhu X, Zhang G and Hu Z: Silencing TRPM2 enhanced erastin- and
RSL3-induced ferroptosis in gastric cancer cells through
destabilizing HIF-1α and Nrf2 proteins. Cytotechnology. 74:559–577.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lin Z, Song J, Gao Y, Huang S, Dou R,
Zhong P, Huang G, Han L, Zheng J, Zhang X, et al: Hypoxia-induced
HIF-1α/lncRNA-PMAN inhibits ferroptosis by promoting the
cytoplasmic translocation of ELAVL1 in peritoneal dissemination
from gastric cancer. Redox Biol. 52:1023122022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Maeda H, Wu J, Sawa T, Matsumura Y and
Hori K: Tumor vascular permeability and the EPR effect in
macromolecular therapeutics: A review. J Control Release.
65:271–284. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Nelson DA, Tan TT, Rabson AB, Anderson D,
Degenhardt K and White E: Hypoxia and defective apoptosis drive
genomic instability and tumorigenesis. Genes Dev. 18:2095–2107.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Frye M, Taddei A, Dierkes C,
Martinez-Corral I, Fielden M, Ortsäter H, Kazenwadel J, Calado DP,
Ostergaard P, Salminen M, et al: Matrix stiffness controls
lymphatic vessel formation through regulation of a GATA2-dependent
transcriptional program. Nat Commun. 9:15112018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Fan W, Adebowale K, Váncza L, Li Y, Rabbi
MF, Kunimoto K, Chen D, Mozes G, Chiu DK, Li Y, et al: Matrix
viscoelasticity promotes liver cancer progression in the
pre-cirrhotic liver. Nature. 626:635–642. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Baghy K, Ladányi A, Reszegi A and
Kovalszky I: Insights into the tumor microenvironment-components,
functions and therapeutics. Int J Mol Sci. 24:175362023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ahluwalia A, Jones MK, Deng X, Sandor Z,
Szabo S and Tarnawski AS: An imbalance between VEGF and endostatin
underlies impaired angiogenesis in gastric mucosa of aging rats. Am
J Physiol Gastrointest Liver Physiol. 305:G325–G332. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zhao ZC, Zheng SS, Wan YL, Jia CK and Xie
HY: The molecular mechanism underlying angiogenesis in
hepatocellular carcinoma: the imbalance activation of signaling
pathways. Hepatobiliary Pancreat Dis Int. 2:529–536.
2003.PubMed/NCBI
|
|
48
|
Cho C, Mukherjee R, Peck AR, Sun Y,
McBrearty N, Katlinski KV, Gui J, Govindaraju PK, Puré E, Rui H and
Fuchs SY: Cancer-associated fibroblasts downregulate type I
interferon receptor to stimulate intratumoral stromagenesis.
Oncogene. 39:6129–6137. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Jain RK: Antiangiogenesis strategies
revisited: From starving tumors to alleviating hypoxia. Cancer
Cell. 26:605–622. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Whatcott CJ, Han H and Von Hoff DD:
Orchestrating the tumor microenvironment to improve survival for
patients with pancreatic cancer: Normalization, not destruction.
Cancer J. 21:299–306. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Slominski RM, Raman C, Chen JY and
Slominski AT: How cancer hijacks the body's homeostasis through the
neuroendocrine system. Trends Neurosci. 46:263–275. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Semenza GL: Oxygen sensing,
hypoxia-inducible factors, and disease pathophysiology. Annu Rev
Pathol. 9:47–71. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Gaggioli C, Hooper S, Hidalgo-Carcedo C,
Grosse R, Marshall JF, Harrington K and Sahai E: Fibroblast-led
collective invasion of carcinoma cells with differing roles for
RhoGTPases in leading and following cells. Nat Cell Biol.
9:1392–1400. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Yang MH, Wu MZ, Chiou SH, Chen PM, Chang
SY, Liu CJ, Teng SC and Wu KJ: Direct regulation of TWIST by
HIF-1alpha promotes metastasis. Nat Cell Biol. 10:295–305. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wang HY, Zhang XP and Wang W: Regulation
of epithelial-to-mesenchymal transition in hypoxia by the HIF-1α
network. FEBS Lett. 596:338–349. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Imai T, Horiuchi A, Wang C, Oka K, Ohira
S, Nikaido T and Konishi I: Hypoxia attenuates the expression of
E-cadherin via up-regulation of SNAIL in ovarian carcinoma cells.
Am J Pathol. 163:1437–1447. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Pastorek J and Pastorekova S:
Hypoxia-induced carbonic anhydrase IX as a target for cancer
therapy: From biology to clinical use. Semin Cancer Biol. 31:52–64.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Peng X, He Z, Yuan D, Liu Z and Rong P:
Lactic acid: The culprit behind the immunosuppressive
microenvironment in hepatocellular carcinoma. Biochim Biophys Acta
Rev Cancer. 1879:1891642024. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Cui XG, Han ZT, He SH, Wu XD, Chen TR,
Shao CH, Chen DL, Su N, Chen YM, Wang T, et al: HIF1/2α mediates
hypoxia-induced LDHA expression in human pancreatic cancer cells.
Oncotarget. 8:24840–24852. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lu H, Li X, Luo Z, Liu J and Fan Z:
Cetuximab reverses the Warburg effect by inhibiting HIF-1-regulated
LDH-A. Mol Cancer Ther. 12:2187–2199. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Harris AL: Hypoxia-a key regulatory factor
in tumour growth. Nat Rev Cancer. 2:38–47. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Pilon-Thomas S, Kodumudi KN, El-Kenawi AE,
Russell S, Weber AM, Luddy K, Damaghi M, Wojtkowiak JW, Mulé JJ,
Ibrahim-Hashim A and Gillies RJ: Neutralization of tumor acidity
improves antitumor responses to immunotherapy. Cancer Res.
76:1381–1390. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Huber V, Camisaschi C, Berzi A, Ferro S,
Lugini L, Triulzi T, Tuccitto A, Tagliabue E, Castelli C and
Rivoltini L: Cancer acidity: An ultimate frontier of tumor immune
escape and a novel target of immunomodulation. Semin Cancer Biol.
43. pp. 74–89. 2017, Zhu J, Shen K, Su Q, Yao G and Chen X:
Metabolic reprogramming and immunosenescence in colorectal cancer:
Mechanisms and therapeutic implications. Front Cell Dev Biol. 13.
pp. 16624642025, View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Wojtkowiak JW, Verduzco D, Schramm KJ and
Gillies RJ: Drug resistance and cellular adaptation to tumor acidic
pH microenvironment. Mol Pharm. 8:2032–2038. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Hosonuma M and Yoshimura K: Association
between pH regulation of the tumor microenvironment and
immunological state. Front Oncol. 13:11755632023. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Dobos GJ, Böhler J, Zhou XJ, Andre M,
Norgauer J, Kownatzki E, Schollmeyer PJ and Vaziri ND: Persistent
inhibition of neutrophil function by glucose based dialysis
solutions. ASAIO J. 40:M435–M439. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Rotstein OD, Nasmith PE and Grinstein S:
pH-dependent impairment of the neutrophil respiratory burst by the
Bacteroides byproduct succinate. Clin Invest Med. 11:259–265.
1988.PubMed/NCBI
|
|
69
|
Vasconcelles MJ, Weitzman SA, Lee SN,
Prachard S and Gordon LI: Inhibition of human polymorphonuclear
leukocyte respiratory burst activity and aggregation by
6-ketocholestanol. Free Radic Res Commun. 8:185–193. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Sgarbi G, Gorini G, Liuzzi F, Solaini G
and Baracca A: Hypoxia and IF1 expression promote ROS decrease in
cancer cells. Cells. 7:642018. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Bosticardo M, Ariotti S, Losana G,
Bernabei P, Forni G and Novelli F: Biased activation of human T
lymphocytes due to low extracellular pH is antagonized by B7/CD28
costimulation. Eur J Immunol. 31:2829–2838. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Müller B, Fischer B and Kreutz W: An
acidic microenvironment impairs the generation of non-major
histocompatibility complex-restricted killer cells. Immunology.
99:375–384. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Foreman KE and Bjornson AB: The
alternative complement pathway promotes IgM antibody-dependent and
-independent adherence of Bacteroides to polymorphonuclear
leukocytes through CR3 and CR1. J Leukoc Biol. 55:603–611. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhu X and Zuo L: Characterization of
oxygen radical formation mechanism at early cardiac ischemia. Cell
Death Dis. 4:e7872013. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Guzy RD, Hoyos B, Robin E, Chen H, Liu L,
Mansfield KD, Simon MC, Hammerling U and Schumacker PT:
Mitochondrial complex III is required for hypoxia-induced ROS
production and cellular oxygen sensing. Cell Metab. 1:401–408.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Nita M and Grzybowski A: The role of the
reactive oxygen species and oxidative stress in the pathomechanism
of the age-related ocular diseases and other pathologies of the
anterior and posterior eye segments in adults. Oxid Med Cell
Longev. 2016:31647342016. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Bridge G, Rashid S and Martin SA: DNA
mismatch repair and oxidative DNA damage: Implications for cancer
biology and treatment. Cancers (Basel). 6:1597–1614. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Syu JP, Chi JT and Kung HN: Nrf2 is the
key to chemotherapy resistance in MCF7 breast cancer cells under
hypoxia. Oncotarget. 7:14659–14672. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Shen Y, Yang J, Zhao J, Xiao C, Xu C and
Xiang Y: The switch from ER stress-induced apoptosis to autophagy
via ROS-mediated JNK/p62 signals: A survival mechanism in
methotrexate-resistant choriocarcinoma cells. Exp Cell Res.
334:207–218. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Corzo CA, Condamine T, Lu L, Cotter MJ,
Youn JI, Cheng P, Cho HI, Celis E, Quiceno DG, Padhya T, et al:
HIF-1α regulates function and differentiation of myeloid-derived
suppressor cells in the tumor microenvironment. J Exp Med.
207:2439–2453. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Schito L: Bridging angiogenesis and immune
evasion in the hypoxic tumor microenvironment. Am J Physiol Regul
Integr Comp Physiol. 315:R1072–R1084. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Abou Khouzam R, Brodaczewska K, Filipiak
A, Zeinelabdin NA, Buart S, Szczylik C, Kieda C and Chouaib S:
Tumor hypoxia regulates immune escape/invasion: influence on
angiogenesis and potential impact of hypoxic biomarkers on cancer
therapies. Front Immunol. 11:6131142021. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Obeagu EI: Oxygen deprivation in breast
cancer: Mechanisms, pathways, and implications. Ann Med Surg
(Lond). 87:3635–3659. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Mantovani A, Sozzani S, Locati M, Allavena
P and Sica A: Macrophage polarization: tumor-associated macrophages
as a paradigm for polarized M2 mononuclear phagocytes. Trends
Immunol. 23:549–555. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Li M, Xu H, Qi Y, Pan Z, Li B, Gao Z, Zhao
R, Xue H and Li G: Tumor-derived exosomes deliver the tumor
suppressor miR-3591-3p to induce M2 macrophage polarization and
promote glioma progression. Oncogene. 41:4618–4632. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Gao B, Wang L, Wen T, Xie X, Rui X and
Chen Q: Colon cancer-derived exosomal LncRNA-XIST promotes M2-like
macrophage polarization by regulating PDGFRA. Int J Mol Sci.
25:114332024. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Nowak M and Klink M: The Role of
tumor-associated macrophages in the progression and chemoresistance
of ovarian cancer. Cells. 9:12992020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Noman MZ, Desantis G, Janji B, Hasmim M,
Karray S, Dessen P, Bronte V and Chouaib S: PD-L1 is a novel direct
target of HIF-1α, and its blockade under hypoxia enhanced
MDSC-mediated T cell activation. J Exp Med. 211:781–790. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Barsoum IB, Smallwood CA, Siemens DR and
Graham CH: A mechanism of hypoxia-mediated escape from adaptive
immunity in cancer cells. Cancer Res. 74:665–674. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Noman MZ and Chouaib S: Targeting hypoxia
at the forefront of anticancer immune responses. Oncoimmunology.
3:e9544632015. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Saggar JK, Yu M, Tan Q and Tannock IF: The
tumor microenvironment and strategies to improve drug distribution.
Front Oncol. 3:1542013. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Winograd R, Byrne KT, Evans RA, Odorizzi
PM, Meyer AR, Bajor DL, Clendenin C, Stanger BZ, Furth EE, Wherry
EJ and Vonderheide RH: Induction of T-cell immunity overcomes
complete resistance to PD-1 and CTLA-4 blockade and improves
survival in pancreatic carcinoma. Cancer Immunol Res. 3:399–411.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Sun J, Zhang Y, Yang M, Zhang Y, Xie Q, Li
Z, Dong Z, Yang Y, Deng B, Feng A, et al: Hypoxia induces T-cell
apoptosis by inhibiting chemokine C receptor 7 expression: the role
of adenosine receptor A(2). Cell Mol Immunol. 7:77–82. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Alicea Pauneto CDM, Riesenberg BP, Gandy
EJ, Kennedy AS, Clutton GT, Hem JW, Hurst KE, Hunt EG, Green JM,
Miller BC, et al: Intra-tumoral hypoxia promotes CD8+ T
cell dysfunction via chronic activation of integrated stress
response transcription factor ATF4. Immunity. 58:2489–2504.e8.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Fischbeck AJ, Ruehland S, Ettinger A,
Paetzold K, Masouris I, Noessner E and Mendler AN: Tumor lactic
acidosis: protecting tumor by inhibiting cytotoxic activity through
motility arrest and bioenergetic silencing. Front Oncol.
10:5894342020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Li H, Rokavec M, Jiang L, Horst D and
Hermeking H: Antagonistic effects of p53 and HIF1A on microRNA-34a
regulation of PPP1R11 and STAT3 and hypoxia-induced epithelial to
mesenchymal transition in colorectal cancer cells.
Gastroenterology. 153:505–520. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Semenza GL, Rue EA, Iyer NV, Pang MG and
Kearns WG: Assignment of the hypoxia-inducible factor 1alpha gene
to a region of conserved synteny on mouse chromosome 12 and human
chromosome 14q. Genomics. 34:437–439. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Wang GL, Jiang BH, Rue EA and Semenza GL:
Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS
heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci
USA. 92:5510–5514. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Iyer NV, Leung SW and Semenza GL: The
human hypoxia-inducible factor 1alpha gene: HIF1A structure and
evolutionary conservation. Genomics. 52:159–165. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Mandl M and Depping R: Hypoxia-inducible
aryl hydrocarbon receptor nuclear translocator (ARNT) (HIF-1β): Is
it a rare exception? Mol Med. 20:215–220. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Jiang BH, Rue E, Wang GL, Roe R and
Semenza GL: Dimerization, DNA binding, and transactivation
properties of hypoxia-inducible factor 1. J Biol Chem.
271:17771–17778. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Jiang H, Guo R and Powell-Coffman JA: The
Caenorhabditis elegans hif-1 gene encodes a bHLH-PAS protein that
is required for adaptation to hypoxia. Proc Natl Acad Sci USA.
98:7916–7921. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Dames SA, Martinez-Yamout M, De Guzman RN,
Dyson HJ and Wright PE: Structural basis for Hif-1 alpha/CBP
recognition in the cellular hypoxic response. Proc Natl Acad Sci
USA. 99:5271–5276. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Bruick RK and McKnight SL: A conserved
family of prolyl-4-hydroxylases that modify HIF. Science.
294:1337–1340. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Piao HY, Liu Y, Kang Y, Wang Y, Meng XY,
Yang D and Zhang J: Hypoxia associated lncRNA HYPAL promotes
proliferation of gastric cancer as ceRNA by sponging miR-431-5p to
upregulate CDK14. Gastric Cancer. 25:44–63. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Guan D and Li C, Li Y, Li Y, Wang G, Gao F
and Li C: The DpdtbA induced EMT inhibition in gastric cancer cell
lines was through ferritinophagy-mediated activation of p53 and
PHD2/hif-1α pathway. J Inorg Biochem. 218:1114132021. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Liu L, Zhang H, Sun L, Gao Y, Jin H, Liang
S, Wang Y, Dong M, Shi Y, Li Z and Fan D: ERK/MAPK activation
involves hypoxia-induced MGr1-Ag/37LRP expression and contributes
to apoptosis resistance in gastric cancer. Int J Cancer.
127:820–829. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Okazaki M, Fushida S, Tsukada T, Kinoshita
J, Oyama K, Miyashita T, Ninomiya I, Harada S and Ohta T: The
effect of HIF-1α and PKM1 expression on acquisition of
chemoresistance. Cancer Manag Res. 10:1865–1874. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Tang E, Wang Y, Liu T and Yan B: Gastrin
promotes angiogenesis by activating HIF-1α/β-catenin/VEGF signaling
in gastric cancer. Gene. 704:42–48. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Luo Z, Luo Y and Xiao K: A-kinase
interacting protein 1 promotes cell invasion and stemness via
activating HIF-1α and β-catenin signaling pathways in gastric
cancer under hypoxia condition. Front Oncol. 11:7985572022.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wu T, Yang Y, Zhang B, Zhang ZS, Zhou S,
Jia GZ, Liu SQ, He XL, He JX and Wang N: EDDM3A drives gastric
cancer progression by promoting HIF-1α-dependent aerobic
glycolysis. Oncogenesis. 11:32022. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Zhao J, Xiao A, Liu C, Ye C, Yin K, Lu M,
Jiao R, Chen X, Zhang C and Liu M: The HIF-1A/miR-17-5p/PDCD4 axis
contributes to the tumor growth and metastasis of gastric cancer.
Signal Transduct Target Ther. 5:462020. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Hong L, Wang J, Wang H, Wei S, Zhang F,
Han J, Liu Y, Ma M, Liu C, Xu Y and Liu W: Linc-pint overexpression
inhibits the growth of gastric tumors by downregulating HIF-1α. Mol
Med Rep. 20:2875–2881. 2019.PubMed/NCBI
|
|
114
|
Huang R, Jin X, Gao Y, Yuan H, Wang F and
Cao X: DZNep inhibits Hif-1α and Wnt signalling molecules to
attenuate the proliferation and invasion of BGC-823 gastric cancer
cells. Oncol Lett. 18:4308–4316. 2019.PubMed/NCBI
|
|
115
|
Wu F, Gao H, Liu K, Gao B, Ren H, Li Z and
Liu F: The lncRNA ZEB2-AS1 is upregulated in gastric cancer and
affects cell proliferation and invasion via miR-143-5p/HIF-1α axis.
Onco Targets Ther. 12:657–667. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Chaffer CL and Weinberg RA: A perspective
on cancer cell metastasis. Science. 331:1559–1564. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Alvikas J, Lo W, Tohme S and Geller DA:
Outcomes and patient selection in laparoscopic vs open liver
resection for HCC and colorectal cancer liver metastasis. Cancers
(Basel). 15:11792023. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Li GZ, Doherty GM and Wang J: Surgical
management of gastric cancer: A review. JAMA Surg. 157:446–454.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Guo R and Yang B: Hypoxia-induced LXRα
contributes to the migration and invasion of gastric cancer cells.
Folia Biol (Praha). 67:91–101. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Xia X, Wang S, Ni B, Xing S, Cao H, Zhang
Z, Yu F, Zhao E and Zhao G: Hypoxic gastric cancer-derived exosomes
promote progression and metastasis via MiR-301a-3p/PHD3/HIF-1α
positive feedback loop. Oncogene. 39:6231–6244. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Ding X, Huang R, Zhong Y, Cui N, Wang Y,
Weng J, Chen L and Zang M: CTHRC1 promotes gastric cancer
metastasis via HIF-1α/CXCR4 signaling pathway. Biomed Pharmacother.
123:1097422020. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Ou XW, Wang RX, Kang MF and Shi JQ:
Hypoxia promotes migration and invasion of gastric cancer cells by
activating HIF-1α and inhibiting NDRG2 associated signaling
pathway. Eur Rev Med Pharmacol Sci. 22:8237–8247. 2018.PubMed/NCBI
|
|
123
|
Fadeel B and Orrenius S: Apoptosis: A
basic biological phenomenon with wide-ranging implications in human
disease. J Intern Med. 258:479–517. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Pereira WO and Amarante-Mendes GP:
Apoptosis: A programme of cell death or cell disposal? Scand J
Immunol. 73:401–407. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Silvestris F, Ribatti D, Nico B,
Silvestris N, Romito A and Dammacco F: Apoptosis or programmed cell
death: Regulatory and pathophysiological mechanisms. Ann Ital Med
Int. 10:7–13. 1995.(In Italian). PubMed/NCBI
|
|
126
|
Rohwer N, Welzel M, Daskalow K, Pfander D,
Wiedenmann B, Detjen K and Cramer T: Hypoxia-inducible factor
1alpha mediates anoikis resistance via suppression of alpha5
integrin. Cancer Res. 68:10113–10120. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Tanaka T, Kitajima Y, Miyake S, Yanagihara
K, Hara H, Nishijima-Matsunobu A, Baba K, Shida M, Wakiyama K,
Nakamura J and Noshiro H: The apoptotic effect of HIF-1α inhibition
combined with glucose plus insulin treatment on gastric cancer
under hypoxic conditions. PLoS One. 10:e01372572015. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Hao YX, Zhong H, Yu PW, Zhang C, Zeng DZ,
Shi Y and Tang B: Effects of HIF-1alpha on human gastric cancer
cell apoptosis at different CO(2) pressures. Clin Exp Med.
9:139–147. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Russo M, Crisafulli G, Sogari A, Reilly
NM, Arena S, Lamba S, Bartolini A, Amodio V, Magrì A, Novara L, et
al: Adaptive mutability of colorectal cancers in response to
targeted therapies. Science. 366:1473–1480. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Geller LT, Barzily-Rokni M, Danino T,
Jonas OH, Shental N, Nejman D, Gavert N, Zwang Y, Cooper ZA, Shee
K, et al: Potential role of intratumor bacteria in mediating tumor
resistance to the chemotherapeutic drug gemcitabine. Science.
357:1156–1160. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Sequist LV, Soria JC, Goldman JW, Wakelee
HA, Gadgeel SM, Varga A, Papadimitrakopoulou V, Solomon BJ, Oxnard
GR, Dziadziuszko R, et al: Rociletinib in EGFR-mutated
non-small-cell lung cancer. N Engl J Med. 372:1700–1709. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Haynes J and Manogaran P: Mechanisms and
strategies to overcome drug resistance in colorectal cancer. Int J
Mol Sci. 26:19882025. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Adil M, Kanwal S, Rasheed S, Iqbal M and
Abbas G: Cancer chemoresistance; recent challenges and future
considerations. Cancer Treat Res. 185:237–253. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Tian Y, Lei Y, Wang Y, Lai J, Wang J and
Xia F: Mechanism of multidrug resistance to chemotherapy mediated
by P-glycoprotein (Review). Int J Oncol. 63:1192023. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Yu F, Zheng S, Yu C, Gao S, Shen Z, Nar R,
Liu Z, Huang S, Wu L, Gu T and Qian Z: KRAS mutants confer platinum
resistance by regulating ALKBH5 posttranslational modifications in
lung cancer. J Clin Invest. 135:e1851492025. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Sameen S, Barbuti R, Milazzo P, Cerone A,
Del Re M and Danesi R: Mathematical modeling of drug resistance due
to KRAS mutation in colorectal cancer. J Theor Biol. 389:263–273.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Dulgar O, Kutuk T and Eroglu Z: Mechanisms
of resistance to BRAF-targeted melanoma therapies. Am J Clin
Dermatol. 22:1–10. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Alkan FK and Korkaya H: Therapeutic
utility of immunosuppressive TREM2+ macrophages: An important step
forward in potentiating the immune checkpoint inhibitors. Signal
Transduct Target Ther. 5:2642020. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Chen Y, Sun L, Guo D, Wu Z and Chen W:
Co-delivery of hypoxia inducible factor-1α small interfering RNA
and 5-fluorouracil to overcome drug resistance in gastric cancer
SGC-7901 cells. J Gene Med. 19:e29982017. View Article : Google Scholar
|
|
140
|
Zhao Q, Li Y, Tan BB, Fan LQ, Yang PG and
Tian Y: HIF-1α induces multidrug resistance in gastric cancer cells
by inducing MiR-27a. PLoS One. 10:e01327462015. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Xuan Y, Hur H, Ham IH, Yun J, Lee JY, Shim
W, Kim YB, Lee G, Han SU and Cho YK: Dichloroacetate attenuates
hypoxia-induced resistance to 5-fluorouracil in gastric cancer
through the regulation of glucose metabolism. Exp Cell Res.
321:219–230. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Sun XP, Dong X, Lin L, Jiang X, Wei Z,
Zhai B, Sun B, Zhang Q, Wang X, Jiang H, et al: Up-regulation of
survivin by AKT and hypoxia-inducible factor 1α contributes to
cisplatin resistance in gastric cancer. FEBS J. 281:115–128. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Rohwer N, Dame C, Haugstetter A,
Wiedenmann B, Detjen K, Schmitt CA and Cramer T: Hypoxia-inducible
factor 1alpha determines gastric cancer chemosensitivity via
modulation of p53 and NF-kappaB. PLoS One. 5:e120382010. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Ferrara N: Vascular endothelial growth
factor: Basic science and clinical progress. Endocr Rev.
25:581–611. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Crunkhorn S: Anticancer drugs:
Tumour-specific angiogenesis inhibition. Nat Rev Drug Discov.
15:3102016. View Article : Google Scholar
|
|
146
|
Butler JM, Kobayashi H and Rafii S:
Instructive role of the vascular niche in promoting tumour growth
and tissue repair by angiocrine factors. Nat Rev Cancer.
10:138–146. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Hicklin DJ and Ellis LM: Role of the
vascular endothelial growth factor pathway in tumor growth and
angiogenesis. J Clin Oncol. 23:1011–1027. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Venook AP, Niedzwiecki D, Lenz HJ,
Innocenti F, Fruth B, Meyerhardt JA, Schrag D, Greene C, O'Neil BH,
Atkins JN, et al: Effect of first-line chemotherapy combined with
cetuximab or bevacizumab on overall survival in patients with KRAS
wild-type advanced or metastatic colorectal cancer: A randomized
clinical trial. JAMA. 317:2392–2401. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Lieu CH and Messersmith WA: Cetuximab or
bevacizumab with first-line chemotherapy in advanced KRAS wild-type
colorectal cancer: No difference, but not the same. JAMA.
317:2376–2378. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Li Z, Wang JW, Wang WZ, Zhi XF, Zhang Q,
Li BW, Wang LJ, Xie KL, Tao JQ, Tang J, et al: Natriuretic peptide
receptor A inhibition suppresses gastric cancer development through
reactive oxygen species-mediated G2/M cell cycle arrest and cell
death. Free Radic Biol Med. 99:593–607. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Li Z, Fan H, Cao J, Sun G, Wang S, Lv J,
Xuan Z, Xia Y, Wang L, Zhang D, et al: Natriuretic peptide receptor
a promotes gastric malignancy through angiogenesis process. Cell
Death Dis. 12:9682021. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Stoeltzing O, McCarty MF, Wey JS, Fan F,
Liu W, Belcheva A, Bucana CD, Semenza GL and Ellis LM: Role of
hypoxia-inducible factor 1alpha in gastric cancer cell growth,
angiogenesis, and vessel maturation. J Natl Cancer Inst.
96:946–956. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Ma J, Zhang L, Ru GQ, Zhao ZS and Xu WJ:
Upregulation of hypoxia inducible factor 1alpha mRNA is associated
with elevated vascular endothelial growth factor expression and
excessive angiogenesis and predicts a poor prognosis in gastric
carcinoma. World J Gastroenterol. 13:1680–1686. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Tsai CY, Wang CS, Tsai MM, Chi HC, Cheng
WL, Tseng YH, Chen CY, Lin CD, Wu JI, Wang LH and Lin KH:
Interleukin-32 increases human gastric cancer cell invasion
associated with tumor progression and metastasis. Clin Cancer Res.
20:2276–2288. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Zhang J, Wu Y, Lin YH, Guo S, Ning PF,
Zheng ZC, Wang Y and Zhao Y: Prognostic value of hypoxia-inducible
factor-1 alpha and prolyl 4-hydroxylase beta polypeptide
overexpression in gastric cancer. World J Gastroenterol.
24:2381–2391. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Stoica G: Cancer stem cells: Current
status and future directions. Vet J. 205:124–125. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Pastushenko I, Mauri F, Song Y, de Cock F,
Meeusen B, Swedlund B, Impens F, Van Haver D, Opitz M, Thery M, et
al: Fat1 deletion promotes hybrid EMT state, tumour stemness and
metastasis. Nature. 589:448–455. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Miao Y, Yang H, Levorse J, Yuan S, Polak
L, Sribour M, Singh B, Rosenblum MD and Fuchs E: Adaptive immune
resistance emerges from tumor-initiating stem cells. Cell.
177:1172–1186.e14. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Miao ZF, Wang ZN, Zhao TT, Xu YY, Gao J,
Miao F and Xu HM: Peritoneal milky spots serve as a hypoxic niche
and favor gastric cancer stem/progenitor cell peritoneal
dissemination through hypoxia-inducible factor 1α. Stem Cells.
32:3062–3074. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
García-Caballero M, Sokol L, Cuypers A and
Carmeliet P: Metabolic reprogramming in tumor endothelial cells.
Int J Mol Sci. 23:110522022. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Menendez JA and Lupu R: Fatty acid
synthase and the lipogenic phenotype in cancer pathogenesis. Nat
Rev Cancer. 7:763–777. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Vander Heiden MG, Cantley LC and Thompson
CB: Understanding the Warburg effect: The metabolic requirements of
cell proliferation. Science. 324:1029–1033. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Wang XH, Jiang ZH, Yang HM, Zhang Y and Xu
LH: Hypoxia-induced FOXO4/LDHA axis modulates gastric cancer cell
glycolysis and progression. Clin Transl Med. 11:e2792021.
View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Liu J, Liu H, Zeng Q, Xu P, Liu M and Yang
N: Circular RNA circ-MAT2B facilitates glycolysis and growth of
gastric cancer through regulating the miR-515-5p/HIF-1α axis.
Cancer Cell Int. 20:1712020. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Gan L, Meng J, Xu M, Liu M, Qi Y, Tan C,
Wang Y, Zhang P, Weng W, Sheng W, et al: Extracellular matrix
protein 1 promotes cell metastasis and glucose metabolism by
inducing integrin β4/FAK/SOX2/HIF-1α signaling pathway in gastric
cancer. Oncogene. 37:744–755. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Liu L, Wang Y, Bai R, Yang K and Tian Z:
MiR-186 inhibited aerobic glycolysis in gastric cancer via HIF-1α
regulation. Oncogenesis. 5:e2242016. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Neoptolemos JP, Moore MJ, Cox TF, Valle
JW, Palmer DH, McDonald AC, Carter R, Tebbutt NC, Dervenis C, Smith
D, et al: Effect of adjuvant chemotherapy with fluorouracil plus
folinic acid or gemcitabine vs observation on survival in patients
with resected periampullary adenocarcinoma: The ESPAC-3
periampullary cancer randomized trial. JAMA. 308:147–156. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Cadman E, Heimer R and Davis L: Enhanced
5-fluorouracil nucleotide formation after methotrexate
administration: Explanation for drug synergism. Science.
205:1135–1137. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Loboda A, Jozkowicz A and Dulak J: HIF-1
versus HIF-2-is one more important than the other? Vascul
Pharmacol. 56:245–251. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Fiorini G and Schofield CJ: Biochemistry
of the hypoxia-inducible factor hydroxylases. Curr Opin Chem Biol.
79:1024282024. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Tian H, McKnight SL and Russell DW:
Endothelial PAS domain protein 1 (EPAS1), a transcription factor
selectively expressed in endothelial cells. Genes De. 11:72–82.
1997. View Article : Google Scholar
|
|
172
|
Freeburg PB and Abrahamson DR: Divergent
expression patterns for hypoxia-inducible factor-1beta and aryl
hydrocarbon receptor nuclear transporter-2 in developing kidney. J
Am Soc Nephrol. 15:2569–2578. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Nagao K and Oka K: HIF-2 directly
activates CD82 gene expression in endothelial cells. Biochem
Biophys Res Commun. 407:260–265. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Wang V, Davis DA, Veeranna RP, Haque M and
Yarchoan R: Characterization of the activation of protein tyrosine
phosphatase, receptor-type, Z polypeptide 1 (PTPRZ1) by hypoxia
inducible factor-2 alpha. PLoS One. 5:e96412010. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Kristan A, Debeljak N and Kunej T:
Integration and visualization of regulatory elements and variations
of the EPAS1 gene in human. Genes (Basel). 12:17932021. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Kling L, Schreiber A, Eckardt KU and
Kettritz R: Hypoxia-inducible factors not only regulate but also
are myeloid-cell treatment targets. J Leukoc Biol. 110:61–75. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Lu X, Zhang W, Zhang J, Ren D, Zhao P and
Ying Y: EPAS1, a hypoxia- and ferroptosis-related gene, promotes
malignant behaviour of cervical cancer by ceRNA and super-enhancer.
J Cell Mol Med. 28:e183612024. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Jiang L, Xia Y, Zhong T, Zhang H, Jin Q,
Li F and Shi S: HIF2A overexpression reduces cisplatin sensitivity
in cervical cancer by inducing excessive autophagy. Transl Cancer
Res. 9:75–84. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Islam F, Gopalan V, Law S, Lam AK and
Pillai S: Molecular deregulation of EPAS1 in the pathogenesis of
esophageal squamous cell carcinoma. Front Oncol. 10:15342020.
View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Kimura Y, Kasamatsu A, Nakashima D,
Yamatoji M, Minakawa Y, Koike K, Fushimi K, Higo M, Endo-Sakamoto
Y, Shiiba M, et al: ARNT2 regulates tumoral growth in oral squamous
cell carcinoma. J Cancer. 7:702–710. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Jia Y, Hao S, Jin G, Li H, Ma X, Zheng Y,
Xiao D and Wang Y: Overexpression of ARNT2 is associated with
decreased cell proliferation and better prognosis in gastric
cancer. Mol Cell Biochem. 450:97–103. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Xu XH, Bao Y, Wang X, Yan F, Guo S, Ma Y,
Xu D, Jin L, Xu J and Wang J: Hypoxic-stabilized EPAS1 proteins
transactivate DNMT1 and cause promoter hypermethylation and
transcription inhibition of EPAS1 in non-small cell lung cancer.
FASEB J. fj2017007152018.(Epub ahead of print). PubMed/NCBI
|
|
183
|
Makino Y, Kanopka A, Wilson WJ, Tanaka H
and Poellinger L: Inhibitory PAS domain protein (IPAS) is a
hypoxia-inducible splicing variant of the hypoxia-inducible
factor-3alpha locus. J Biol Chem. 277:32405–32408. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Jang MS, Park JE, Lee JA, Park SG, Myung
PK, Lee DH, Park BC and Cho S: Binding and regulation of
hypoxia-inducible factor-1 by the inhibitory PAS proteins. Biochem
Biophys Res Commun. 337:209–215. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Su Y, Liu J, Tan S, Liu W, Wang R and Chen
C: PLGA sustained-release microspheres loaded with an insoluble
small-molecule drug: Microfluidic-based preparation, optimization,
characterization, and evaluation in vitro and in vivo. Drug Deliv.
29:1437–1446. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Lao Y, Skiba MA, Chun SW, Narayan ARH and
Smith JL: Structural basis for control of methylation extent in
polyketide synthase metal-dependent C-methyltransferases. ACS Chem
Biol. 17:2088–2098. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Huang Y, Zhu Y, Cai D, Guo Q, Wang J, Lei
L, Li X and Shi S: Penetrating-peptide-mediated non-invasive
Axitinib delivery for anti-neovascularisation. J Control Release.
347:449–459. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Ovens AJ, Gee YS, Ling NXY, Yu D, Hardee
JP, Chung JD, Ngoei KRW, Waters NJ, Hoffman NJ, Scott JW, et al:
Structure-function analysis of the AMPK activator SC4 and
identification of a potent pan AMPK activator. Biochem J.
479:1181–1204. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Ahamad S, Mathew S, Khan WA and Mohanan K:
Development of small-molecule PCSK9 inhibitors for the treatment of
hypercholesterolemia. Drug Discov Today. 27:1332–1349. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Kim TW and Lee HG: Apigenin Induces
autophagy and cell death by targeting EZH2 under hypoxia conditions
in gastric cancer cells. Int J Mol Sci. 22:134552021. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Egawa N, Tanaka T, Matsufuji S, Yamada K,
Ito K, Kitagawa H, Okuyama K, Kitajima Y and Noshiro H: Antitumor
effects of low-dose tipifarnib on the mTOR signaling pathway and
reactive oxygen species production in HIF-1α-expressing gastric
cancer cells. FEBS Open Bio. 11:1465–1475. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Huang YN, Xu YY, Ma Q, Li MQ, Guo JX, Wang
X, Jin X, Shang J and Jiao LX: Dextran sulfate effects EMT of human
gastric cancer cells by reducing HIF-1α/TGF-β. J Cancer.
12:3367–3377. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Li XY, Shi LX, Yao XM, Jing M, Li QQ, Wang
YL and Li QS: Functional vinorelbine plus schisandrin B liposomes
destroying tumor metastasis in treatment of gastric cancer. Drug
Dev Ind Pharm. 47:100–112. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Wang J, Wu K, Bai F, Zhai H, Xie H, Du Y,
Liang J, Han S, Chen Y, Lin T and Fan D: Celecoxib could reverse
the hypoxia-induced Angiopoietin-2 upregulation in gastric cancer.
Cancer Lett. 242:20–27. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Wang K, Liu R, Li J, Mao J, Lei Y, Wu J,
Zeng J, Zhang T, Wu H, Chen L, et al: Quercetin induces protective
autophagy in gastric cancer cells: Involvement of Akt-mTOR- and
hypoxia-induced factor 1α-mediated signaling. Autophagy. 7:966–978.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Kim DH, Sung B, Kang YJ, Hwang SY, Kim MJ,
Yoon JH, Im E and Kim ND: Sulforaphane inhibits hypoxia-induced
HIF-1α and VEGF expression and migration of human colon cancer
cells. Int J Oncol. 47:2226–2232. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Xu Y, Jin X, Huang Y, Dong J, Wang H, Wang
X and Cao X: Inhibition of peritoneal metastasis of human gastric
cancer cells by dextran sulphate through the reduction in HIF-1α
and ITGβ1 expression. Oncol Rep. 35:2624–2634. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Dong J, Zheng N, Wang X, Tang C, Yan P,
Zhou HB and Huang J: A novel HDAC6 inhibitor exerts an anti-cancer
effect by triggering cell cycle arrest and apoptosis in gastric
cancer. Eur J Pharmacol. 828:67–79. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Deng X, Li Z, Xiong R, Liu J, Liu R, Peng
J, Chen Y, Lei X, Cao X, Zheng X, et al: FS-7 inhibits MGC-803
cells growth in vitro and in vivo via down-regulating glycolysis.
Biomed Pharmacother. 109:1659–1669. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Wang SJ, Zhao JK, Ren S, Sun WW, Zhang WJ
and Zhang JN: Wogonin affects proliferation and the energy
metabolism of SGC-7901 and A549 cells. Exp Ther Med. 17:911–918.
2019.PubMed/NCBI
|
|
201
|
Idowu S, Bertrand PP and Walduck AK:
Gastric organoids: Advancing the study of H. pylori
pathogenesis and inflammation. Helicobacter. 27:e128912022.
View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Lee DY, Jung DE, Yu SS, Lee YS, Choi BK
and Lee YC: Regulation of SIRT3 signal related metabolic
reprogramming in gastric cancer by Helicobacter pylori
oncoprotein CagA. Oncotarget. 8:78365–78378. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Xu HM, Han Y, Liu ZC, Yin ZY, Wang MY, Yu
C, Ma JL, Sun D, Liu WD, Zhang Y, et al: Helicobacter pylori
treatment and gastric cancer risk among individuals with high
genetic risk for gastric cancer. JAMA Netw Open. 7:e24137082024.
View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Li N: Hypoxia inducible factor-1α-mediated
gene activation in the regulation of renal medullary function and
salt sensitivity of blood pressure. Am J Cardiovasc Dis. 2:208–215.
2012.PubMed/NCBI
|
|
205
|
Di Y, Ding L, Gao L and Huang H:
Association of meat consumption with the risk of gastrointestinal
cancers: A systematic review and meta-analysis. BMC Cancer.
23:7822023. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Wu X, Qin G, Zhang R, Liu Y, Chen L, Su J,
Kong C, Fang Z and Wu J: Effect of vegetable consumption on risk of
gastric cancer: A systematic review and multi-level meta-analysis
of prospective studies. Nutr Res Rev. 38:192–201. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Eurlings IM, Reynaert NL, van den Beucken
T, Gosker HR, de Theije CC, Verhamme FM, Bracke KR, Wouters EF and
Dentener MA: Cigarette smoke extract induces a phenotypic shift in
epithelial cells; involvement of HIF1α in mesenchymal transition.
PLoS One. 9:e1077572014. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Seitz HK: A narrative review on alcohol
and alimentary tract cancer with special emphasis on acetaldehyde
and oxidative stress. Z Gastroenterol. 63:960–974. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Benusiglio PR, Malka D, Rouleau E, De Pauw
A, Buecher B, Noguès C, Fourme E, Colas C, Coulet F, Warcoin M, et
al: CDH1 germline mutations and the hereditary diffuse gastric and
lobular breast cancer syndrome: A multicentre study. J Med Genet.
50:486–489. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Ning M, Hoshii T, Nakagawa T, Usui G,
Izumi S, Hayashi K, Matsumoto M, Rahmutulla B, Fukuyo M, Abe H, et
al: Non-catalytic role of SETD1A promotes gastric cancer cell
proliferation through the E2F4-TAF6 axis in the cell cycle. Cell
Death Dis. 16:6392025. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Arany Z, Huang LE, Eckner R, Bhattacharya
S, Jiang C, Goldberg MA, Bunn HF and Livingston DM: An essential
role for p300/CBP in the cellular response to hypoxia. Proc Natl
Acad Sci USA. 93:12969–12973. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
He M, Zhan M, Chen W, Xu S, Long M, Shen
H, Shi Y, Liu Q, Mohan M and Wang J: MiR-143-5p deficiency triggers
EMT and metastasis by targeting HIF-1α in gallbladder cancer. Cell
Physiol Biochem. 42:2078–2092. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
213
|
LaGory EL and Giaccia AJ: The
ever-expanding role of HIF in tumour and stromal biology. Nat Cell
Biol. 18:356–365. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Corcoran SE and O'Neill LA: HIF1α and
metabolic reprogramming in inflammation. J Clin Invest.
126:3699–3707. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Catalano V, Labianca R, Beretta GD, Gatta
G, de Braud F and Van Cutsem E: Gastric cancer. Crit Rev Oncol
Hematol. 54:209–241. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
216
|
Nam S and Lee Y: HIF1A protein expression
is correlated with clinical features in gastric cancer: An updated
systematic review and meta-analysis. Sci Rep. 14:137362024.
View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Zhu HM and Wang SY: Efficacy of magnifying
endoscopy combined with narrowband imaging in detection of early
gastric cancer: A systematic evaluation and meta-analysis. Am J
Transl Res. 16:4268–4278. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Canakis A, Deliwala SS, Kadiyala J, Bomman
S, Canakis J and Bilal M: The diagnostic performance of probe-based
confocal laser endomicroscopy in the detection of gastric cancer: A
systematic review and meta-analysis. Ann Gastroenterol. 35:496–502.
2022.PubMed/NCBI
|
|
219
|
He X, Wu L, Dong Z, Gong D, Jiang X, Zhang
H, Ai Y, Tong Q, Lv P, Lu B, et al: Real-time use of artificial
intelligence for diagnosing early gastric cancer by magnifying
image-enhanced endoscopy: A multicenter diagnostic study (with
videos). Gastrointest Endosc. 95:671–67.e4. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Mizokami K, Kakeji Y, Oda S, Irie K,
Yonemura T, Konishi F and Maehara Y: Clinicopathologic significance
of hypoxia-inducible factor 1alpha overexpression in gastric
carcinomas. J Surg Oncol. 94:149–154. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Li J, Li Z, Ding Y, Xu Y, Zhu X, Cao N,
Huang C, Qin M, Liu F and Zhao A: TP53 mutation and MET
amplification in circulating tumor DNA analysis predict disease
progression in patients with advanced gastric cancer. PeerJ.
9:e111462021. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Liu Z, Shi Z, Jiang W, Shen Z, Chen W,
Shen K, Sun Y, Tang Z and Wang X: Circulating tumor DNA analysis
for prediction of prognosis and molecular insights in patients with
resectable gastric cancer: Results from a prospective study.
MedComm (2020). 6:e700652025. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Wu YL, Hu LN, Zheng CD, Sun RC, Zhang SX,
Yan Q and Li YX: Expression of hypoxia-inducible factor 1α in
gastric cancer and its clinical signficance. Zhonghua Yi Xue Za
Zhi. 96:1418–1423. 2016.(In Chinese). PubMed/NCBI
|
|
224
|
Lee D, Choi J, Oh HJ, Ham IH, Lee SH,
Nomura S, Han SU and Hur H: Molecular and immune profiling of
syngeneic mouse models predict response to immune checkpoint
inhibitors in gastric cancer. Cancer Res Treat. 55:167–178. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Griffiths EA, Pritchard SA, McGrath SM,
Valentine HR, Price PM, Welch IM and West CM: Hypoxia-associated
markers in gastric carcinogenesis and HIF-2alpha in gastric and
gastro-oesophageal cancer prognosis. Br J Cancer. 98:965–973. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Tong WW, Tong GH, Chen XX, Zheng HC and
Wang YZ: HIF2α is associated with poor prognosis and affects the
expression levels of survivin and cyclin D1 in gastric carcinoma.
Int J Oncol. 46:233–242. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Zhao J, Bai Z, Feng F, Song E, Du F, Zhao
J, Shen G, Ji F, Li G, Ma X, et al: Cross-talk between
EPAS-1/HIF-2α and PXR signaling pathway regulates multi-drug
resistance of stomach cancer cell. Int J Biochem Cell Biol.
72:73–88. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Torii S, Goto Y, Ishizawa T, Hoshi H,
Goryo K, Yasumoto K, Fukumura H and Sogawa K: Pro-apoptotic
activity of inhibitory PAS domain protein (IPAS), a negative
regulator of HIF-1, through binding to pro-survival Bcl-2 family
proteins. Cell Death Differ. 18:1711–1725. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Zhou X, Guo X, Chen M, Xie C and Jiang J:
HIF-3α promotes metastatic phenotypes in pancreatic cancer by
transcriptional regulation of the RhoC-ROCK1 signaling pathway. Mol
Cancer Res. 16:124–134. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Pasanen A, Heikkilä M, Rautavuoma K,
Hirsilä M, Kivirikko KI and Myllyharju J: Hypoxia-inducible factor
(HIF)-3alpha is subject to extensive alternative splicing in human
tissues and cancer cells and is regulated by HIF-1 but not HIF-2.
Int J Biochem Cell Biol. 42:1189–1200. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Heikkilä M, Pasanen A, Kivirikko KI and
Myllyharju J: Roles of the human hypoxia-inducible factor (HIF)-3α
variants in the hypoxia response. Cell Mol Life Sci. 68:3885–3901.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
López-Mejía A, Briseño-Díaz P and
Robles-Flores M: The role of hypoxia-inducible factor-3α in human
disease. Biochim Biophys Acta Mol Cell Res. 1872:1200072025.
View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Slominski A, Kim TK, Brożyna AA,
Janjetovic Z, Brooks DL, Schwab LP, Skobowiat C, Jóźwicki W and
Seagroves TN: The role of melanogenesis in regulation of melanoma
behavior: Melanogenesis leads to stimulation of HIF-1α expression
and HIF-dependent attendant pathways. Arch Biochem Biophys.
563:79–93. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Basheeruddin M and Qausain S:
Hypoxia-inducible factor 1-alpha (HIF-1α) and cancer: Mechanisms of
tumor hypoxia and therapeutic targeting. Cureus.
16:e707002024.PubMed/NCBI
|
|
235
|
Liu Y, Suhail Y, Novin A, Afzal J and Pant
A: Kshitiz: Lactate in breast cancer cells is associated with
evasion of hypoxia-induced cell cycle arrest and adverse patient
outcome. Hum Cell. 37:768–781. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Hashemzadeh S, Shahmorad S, Rafii-Tabar H
and Omidi Y: Computational modeling to determine key regulators of
hypoxia effects on the lactate production in the glycolysis
pathway. Sci Rep. 10:91632020. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Shay JE and Celeste Simon M:
Hypoxia-inducible factors: Crosstalk between inflammation and
metabolism. Semin Cell Dev Biol. 23:389–394. 2012. View Article : Google Scholar : PubMed/NCBI
|















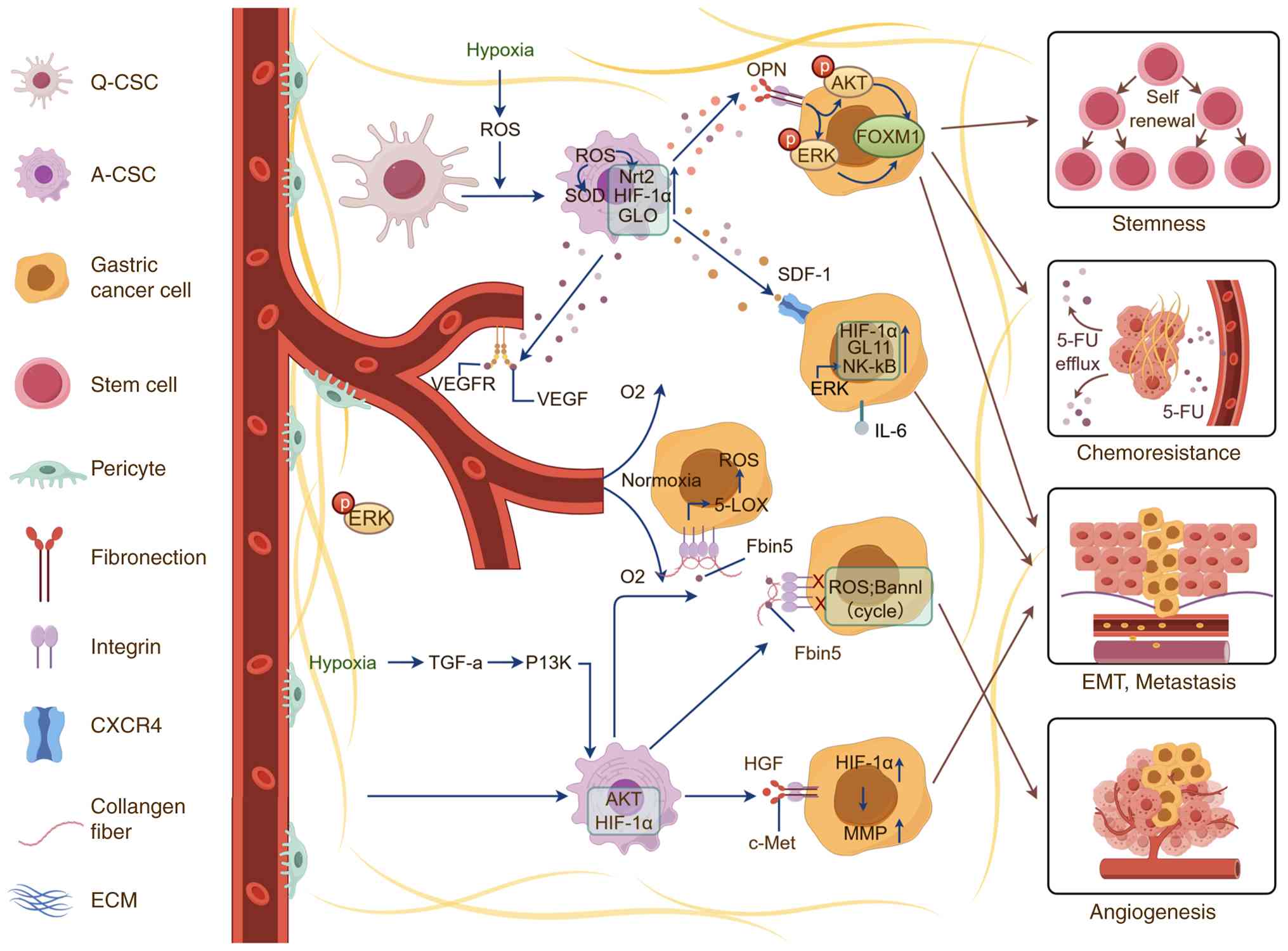
![Hypoxia activates downstream
signaling pathways. Normoxic degradation of HIF-α: In normoxia
(normal oxygen tension), HIF-α is hydroxylated by PHDs and FIH,
utilizing O2 and 2-KG as cofactors. This modification triggers
binding to the VHL complex, leading to Ub and proteasomal
degradation. Metabolites such as succinate, lactate, pyruvate,
fumarate and the oncometabolite 2-HG, along with ROS, can inhibit
PHD activity, while sirtuins (SIRT1/SIRT6) modulate HIF-α stability
via deacetylation. Hypoxic stabilization and activation of HIF-α:
In hypoxia [low oxygen tension, (aO2)], reduced O₂ availability
inhibits PHD/FIH function, stabilizing HIF-α. Stabilized HIF-α
translocates to the nucleus, heterodimerizes with aryl hydrocarbon
receptor nuclear translocator (ARNT/HIF-1β) and recruits
co-activators p300/CBP to bind HREs in target gene promoters. This
drives transcription of genes involved in proliferation, cell
survival, metabolic reprogramming, angiogenesis (via VEGF, PDGF,
SDF-1, FGF, ANGPT2), ECM remodeling, metastasis, stem cell
maintenance, migration/invasion, apoptosis, autophagy,
lymphangiogenesis, EMT and immune evasion. Upstream signaling and
microenvironment interactions: Extracellular signals, including
GFRs, TCRs, IL-6R and TLRs, activate pathways such as
PI3K/AKT/mTOR, Ras/ERK, JAK/STAT3 and NF-κB to modulate HIF-α
translation and transcription. Hypoxia also drives angiogenesis by
activating endothelial cells and VSMCs, reprograms CAFs for
metabolic support and suppresses immune cell function, collectively
promoting gastric cancer progression. VEGF, vascular endothelial
growth factor; PDGF, platelet-derived growth factor; SDF-1, stromal
cell-derived factor-1; FGF, fibroblast growth factor; ANGPT2,
angiopoietin-2; 2-HG, 2-hydroxyglutarate; PHDs, prolyl hydroxylase;
ARNT, aryl hydrocarbon receptor nuclear translocator; p300/CBP, E1A
binding protein p300/CREB binding protein; SIRT1, sirtuin 1; TCR, T
cell receptor; GFR, growth factor receptor; IL-6R, interleukin-6
receptor; TLR, toll-like receptor; ENOs, enolases; AMPK,
AMP-activated protein kinase; PI3K, phosphatidylinositol 3-kinase;
AKT, protein kinase B; mTOR, mechanistic target of rapamycin;
REDD1, regulated in development and DNA damage responses 1; Ras,
Rat sarcoma viral oncogene homolog; ERK, extracellular
signal-regulated kinase; p70S6K, p70 ribosomal S6 kinase; 4E-BP1,
eukaryotic translation initiation factor 4E-binding protein 1;
eIF4E, eukaryotic translation initiation factor 4E; MNK,
MAPK-interacting kinase; Jak, Janus kinase; IKK, IκB kinase; STAT3,
signal transducer and activator of transcription 3; NF-κB, nuclear
factor-κB; HRE, hypoxia response element; EMT,
epithelial-mesenchymal transition; FIH, factor inhibiting HIF;
2-KG, α-ketoglutarate; VHL, von Hippel-Lindau; Ub, ubiquitination;
ROS, reactive oxygen species; VSMCs, vascular smooth muscle cells;
CAFs, cancer-associated fibroblasts.](/article_images/ol/31/6/ol-31-06-15578-g01.jpg)