Introduction
Cervical cancer (CC) is the third most common
malignant tumor among women worldwide (1). It has been reported that ~600,000
novel cases are diagnosed annually, leading to ~340,000 mortalities
(2). Human papillomavirus
(HPV)-based screening and prophylactic vaccination have become
critical interventions. Despite continuous advancements in
screening technologies, 5–11% of CC cases are unassociated with HPV
infection (3). These cases,
characterized by distinct histopathological, molecular and clinical
features, are referred to as HPV-independent CCs, with major
subtypes including gastric-type adenocarcinoma, clear cell,
mesonephric and endometrioid carcinoma (4). Current treatment strategies for CC
vary by disease stage, with surgical intervention being the primary
approach for early-stage disease, while advanced-stage disease
relies on chemotherapy, radiotherapy and immunotherapy (5). However, challenges such as
surgery-related complications, damage to normal tissues caused by
chemotherapy, radiotherapy and immunotherapy, as well as the
pervasive issue of drug resistance, remain notable obstacles in the
clinical management of CC (6,7).
Therefore, the exploration of novel therapeutic strategies has
emerged as a critical direction in current research.
Mesenchymal stem cells (MSCs) are a type of adult
stem cells with self-renewal and multi-directional differentiation
potential (8). Over the past 3
decades, their unique biological properties and broad therapeutic
potential have garnered extensive attention in various diseases
(9). For instance, MSCs have
demonstrated potential therapeutic value in fields such as
tuberculosis (10), osteoarthritis
(11), and oral (12), neurodegenerative (13), kidney (14) and rheumatic diseases (15). More importantly, MSCs possess innate
tumor-homing capabilities and potent immunomodulatory functions,
positioning them as notable research subjects in cancer therapy
(16). As key mediators of MSC
function, their derived exosomes can serve a central role in
regulating the tumor microenvironment (TME) (17,18)
and influencing resistance to cancer therapies, including
chemotherapy, radiotherapy, targeted therapy and immunotherapy
(19,20), by mediating intercellular
communication (21).
Selecting CC as a model to investigate the role of
MSCs and their derived exosomes is supported by unique scientific
rationale. Firstly, persistent infection with high-risk HPV,
particularly HPV-16 and HPV-18, is the primary driver of cervical
carcinogenesis (22,23). This viral oncogenic mechanism
fundamentally shapes the TME of CC. HPV oncoproteins, such as E6
and E7, while disrupting cell cycle regulation, also compromise
host immune surveillance, thereby establishing a chronic
inflammatory and immunosuppressive microenvironment conducive to
tumorigenesis (24). The potent
immunomodulatory properties of MSCs offer distinct advantages in
this context for CC therapy (25).
Secondly, the TME of CC is characterized by complex interactions
between infected epithelial cells and various stromal components,
including cancer-associated fibroblasts and infiltrating immune
cells (26,27). MSCs and their derived exosomes are
known to be recruited to tumor sites and can differentiate into
cancer-associated fibroblasts, promoting tissue remodeling and
tumor progression (16). In CC,
persistent viral stimulation further complicates these
interactions, potentially inducing plasticity in the functions of
MSCs and their derived exosomes, leading to dual pro-tumorigenic or
antitumorigenic effects (28,29).
This dualistic and paradoxical role inherent to MSCs
and their derived exosomes underscores the complexity that must be
addressed prior to their clinical translation. Therefore, the
present review aims to systematically elucidate the specific
mechanisms of action of MSCs and their derived exosomes in cervical
carcinogenesis and progression, delve into the underlying reasons
for their dual effects and summarize recent advances in therapeutic
research based on these findings.
MSCs and MSC exosomes (MSC-Exos)
MSCs
The origin of MSCs can be traced back to the
mid-to-late 20th century (30).
After decades of exploration, researchers have now developed a
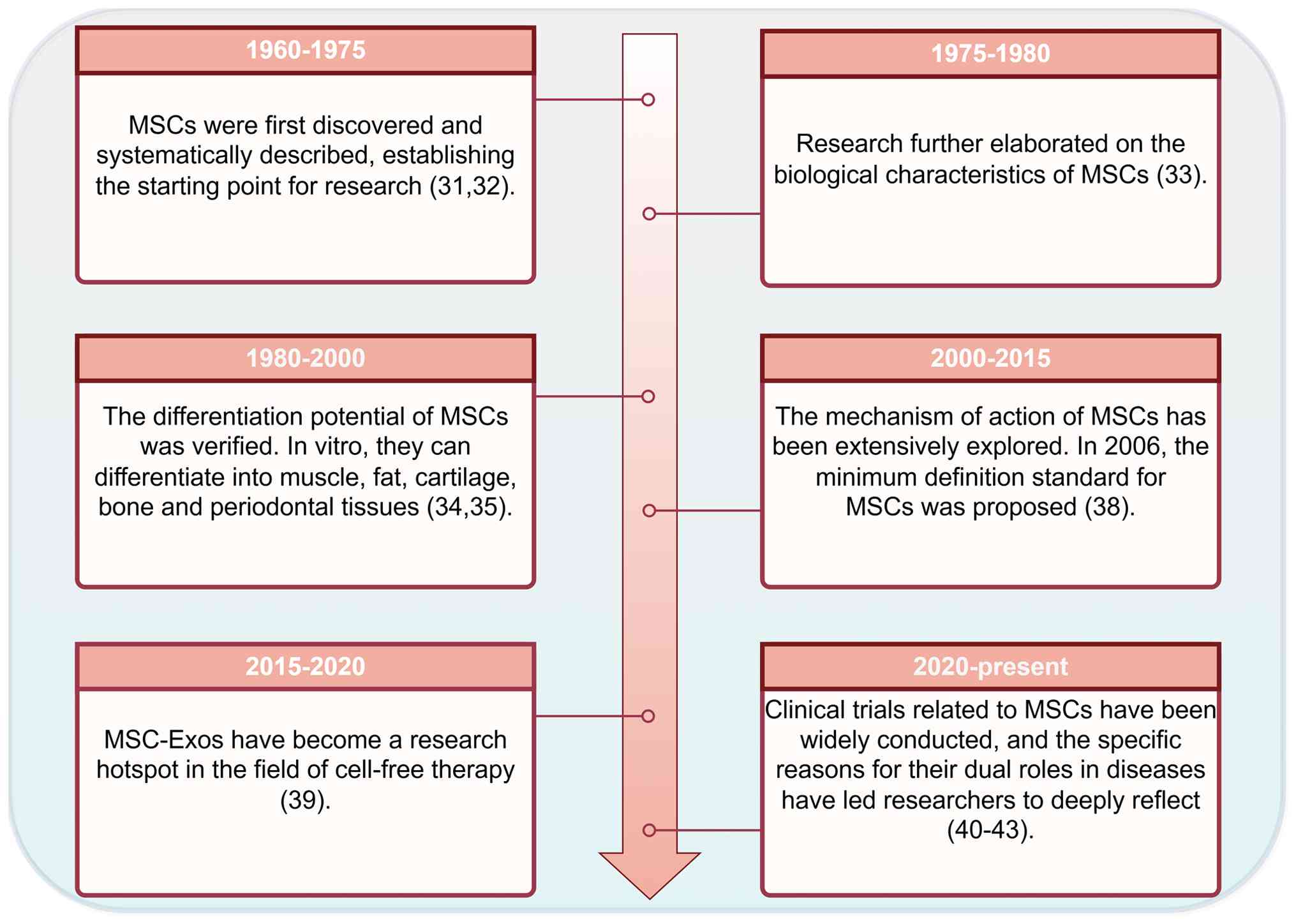
relatively systematic understanding of these cells (Fig. 1). As early as 1960-1975, researchers
first identified a type of non-hematopoietic stem cell in the bone
marrow with clonogenic capacity and adherence-dependent growth
(31). Friedenstein et al
(32) were the first to
systematically isolate and characterize these cells, which is
considered the starting point of research on MSCs. This discovery
demonstrated for the first time that within the complex
hematopoietic microenvironment of the bone marrow, there exists a
unique group of non-hematopoietic precursor cells, laying the
theoretical foundation for subsequent studies on MSCs.
From 1975 to 1980, research further elaborated on
the biological characteristics of MSCs, such as the cloning
formation characteristics and radiation resistance (33). Further studies gradually revealed
the multi-lineage differentiation potential of these cells. In
1988, Grigoriadis et al (34) found that a subset of mesenchymal
progenitor cells could differentiate into four distinct cell types,
namely, muscle, adipose tissue, cartilage and bone, in vitro
under the induction of glucocorticoids. This discovery suggested
that MSCs possessed multi-directional differentiation capabilities,
thus opening up the possibility of application in regenerative
medicine. In 1995, research further confirmed the presence of MSCs
in periodontal tissues (35). This
finding indicated that the source of MSCs expanded from bone marrow
to other tissues, providing an important basis for the subsequent
isolation of MSCs from various tissues, such as fat and the
umbilical cord, and also revealed the extensive distribution of
MSCs in the body.
From 2000 to 2015, the mechanisms of MSCs were
extensively explored and researchers gradually recognized that MSCs
possessed multiple functions in disease contexts, including
immunomodulation, inflammation suppression, promotion of repair
(for example, repairing bones, cartilage, tendons and cardiac
muscle) and specific homing to tumor sites (36,37).
In 2006, the International Society for Cellular Therapy proposed
the minimal defining criteria for MSCs: i) Adherence to plastic;
ii) specific expression of CD105, CD73 and CD90; iii) lack of
expression of surface markers CD45, CD34, CD14, CD11b, CD79α, CD19
and human leukocyte antigen-DR isotype; and iv) multi-lineage
differentiation potential. This criteria established a foundation
in standardizing research on MSCs (38). The proposal of this standardized
definition marks a notable milestone in the development of this
field. It enables the results of different researchers to be
similar and establishes standards for the basic research of
MSCs.
Between 2015 and 2020, extracellular vesicles (EVs)
secreted by MSCs, particularly exosomes, were identified as key
carriers mediating their effects and gradually became a research
hotspot in regenerative medicine as an alternative to cell therapy
(39). From 2020 to the present,
clinical trials associated with MSCs have been widely conducted.
Certain studies have demonstrated that MSCs exhibit notable
therapeutic efficacy (40,41), while others have reported potential
negative effects; for instance, there are complex interactions
between MSCs and tumor cells, which can create a microenvironment
conducive to tumor cell proliferation, angiogenesis, migration,
invasion and metastasis, thereby promoting tumor progression
(42). Furthermore, in another
study, it was found that MSCs co-cultured with colorectal cancer
cells exhibited enhanced invasiveness and proliferation ability due
to changes in their tumor protein p53/transforming growth factor β1
(p53/TGF-β1) levels (43). The core
contribution of this stage is that it has pushed the research on
MSCs to enter a novel phase from ‘verification of effectiveness’ to
‘precision of treatment’ (44).
These results have prompted researchers to reflect, suggesting that
this bidirectional nature of effects may be associated with factors
such as cell source, route of administration and dosage (45,46).
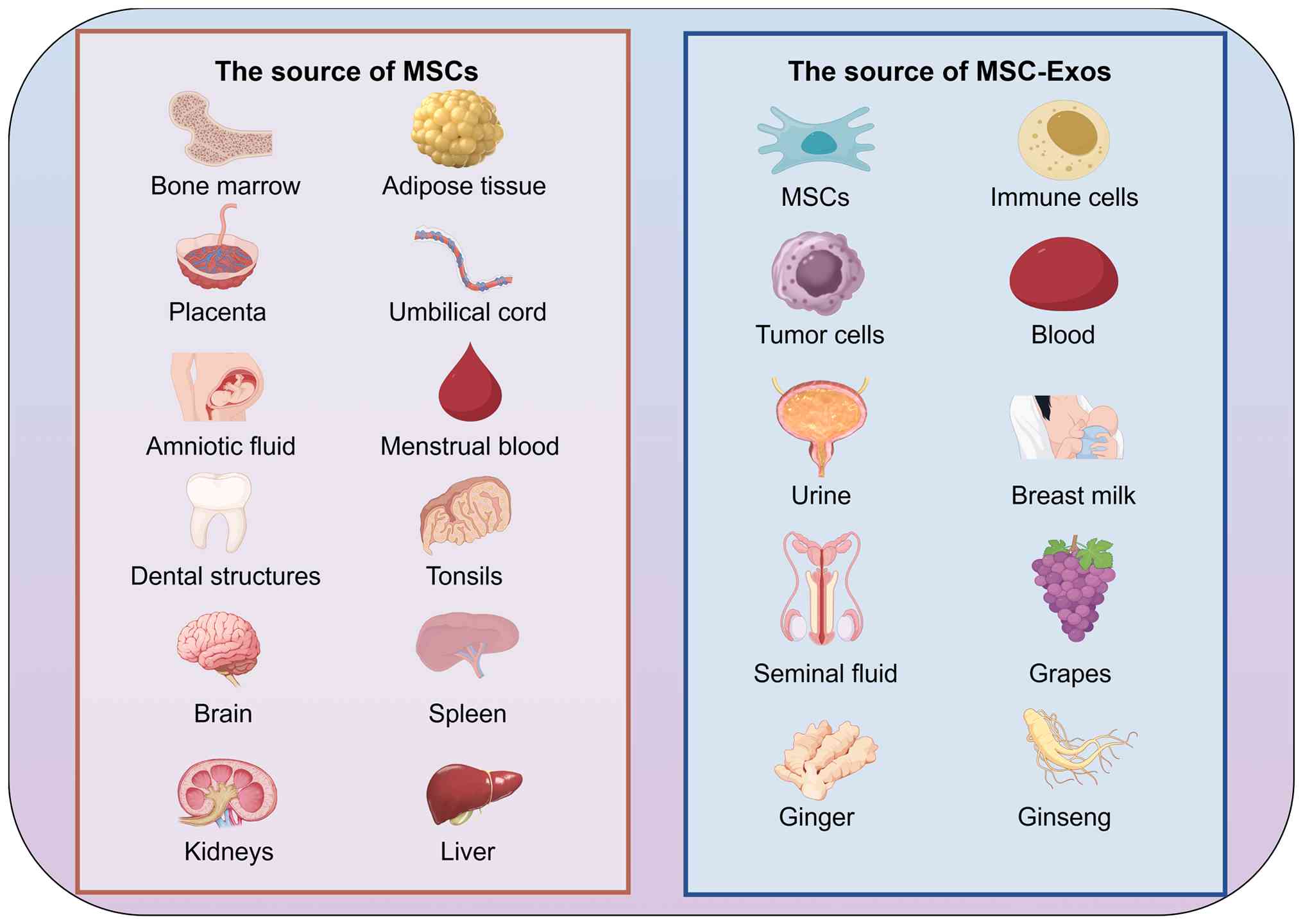
Of note, MSCs can be isolated from various biological tissues. In
addition to common sources such as bone marrow, adipose tissue,
umbilical cord, amniotic fluid, placenta and menstrual blood
(47–51), several studies have also reported
that MSCs can be successfully extracted and isolated from teeth
(52), tonsils (53), as well as visceral tissues including
the brain, spleen, kidney and liver (54–56).
Exosomes
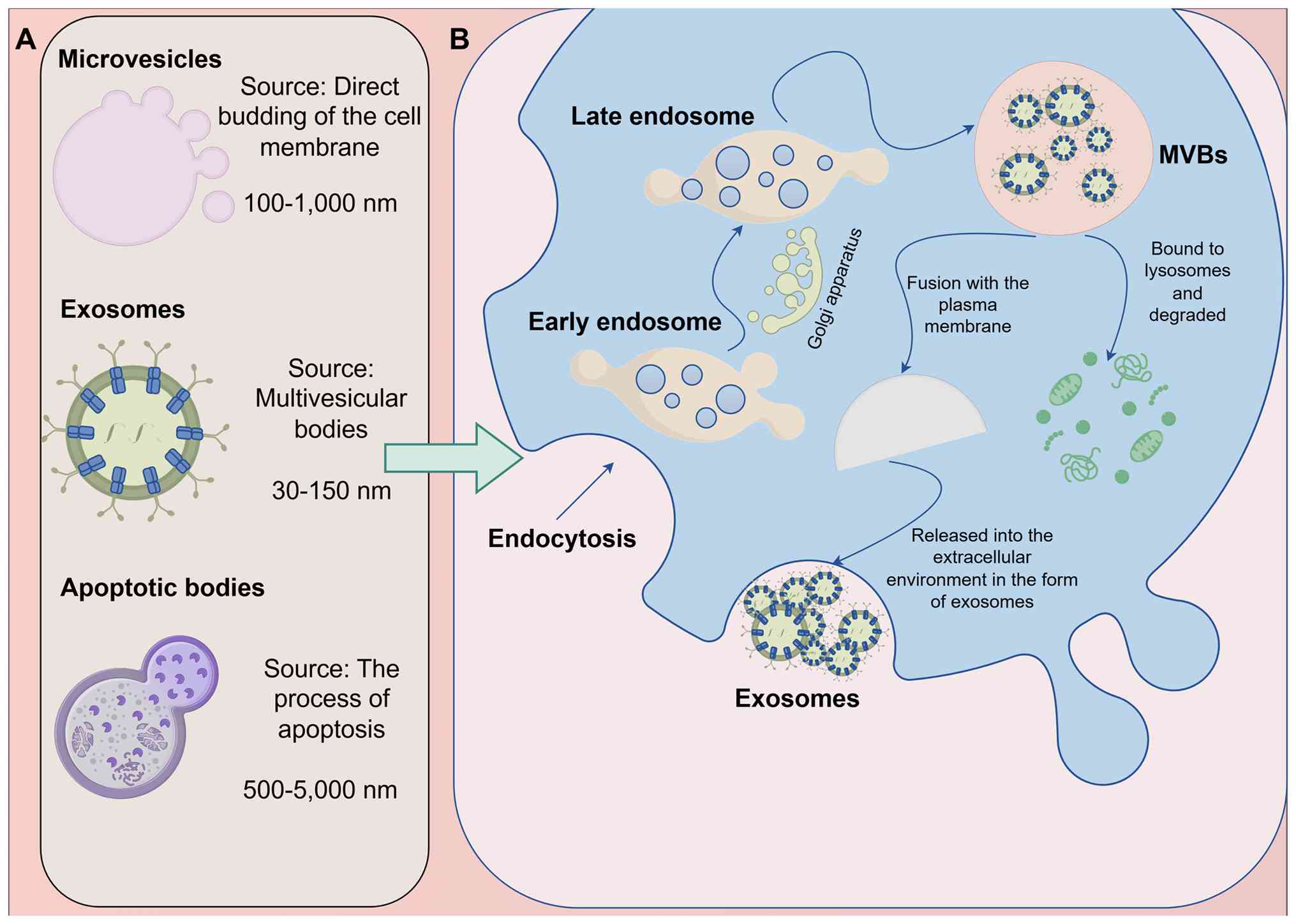
EVs are heterogeneous collections of particles
enclosed by a lipid bilayer membrane. EVs are typically classified
into three main types based on their biogenesis pathways and size
(57). Among them, exosomes are
small nanovesicles (diameter, 30–150 nm) derived from the endosomal
system (58), which distinguishes
them from the larger microvesicles (diameter, 100–1,000 nm) and the
distinctively originating apoptotic bodies (diameter, 500–5,000 nm)
(59,60) (Fig.
2A). The biogenesis of exosomes is a complex process (Fig. 2B). It begins with endocytosis at the
cell membrane surface, forming early endosomes. Early endosomes
further mature, receive transport vesicles from organelles such as
the Golgi apparatus and undergo processes such as acidification to
transform into late endosomes. Within late endosomes, the membrane
invaginates inwardly a second time, forming multiple intraluminal
vesicles (ILVs) through budding (61–63).
Late endosomes containing numerous ILVs are termed multivesicular
bodies (MVBs). MVBs undergo either of two pathways: Most MVBs fuse
with lysosomes, leading to the degradation of the ILVs, while a
small portion of MVBs fuse with the plasma membrane, releasing the
ILVs into the extracellular environment. These released ILVs are
termed exosomes (61,64,65).
Similar to MSCs, the sources of exosomes are diverse (Fig. 3). Besides being present in MSCs and
various immune cells (66–68), exosomes are also widely found in
tumor cells (69,70). Furthermore, exosomes exist in
various bodily fluids, including blood, urine, breast milk and
semen (71,72). Exosomes have also been detected in
certain foods and plants (73–77).
Tumor-promoting mechanism of MSCs and their
derived exosomes in CC
In the initiation and progression of CC, the role of
MSCs appear to be more extensively documented compared with that of
their derived exosomes. MSCs primarily exert pro-tumorigenic
effects through multiple mechanisms, including promoting tumor cell
proliferation, inducing tumor angiogenesis and suppressing the
immune response of the body, working in synergy (78–80)
(Table I).
 | Table I.Mechanisms by which MSCs promote the
occurrence and development of CC. |
Table I.
Mechanisms by which MSCs promote the
occurrence and development of CC.
| Source | Research type | Mechanism of
action | (Refs.) |
|---|
| Bone marrow | In vitro
cell model | At low doses, MSCs
can promote the proliferation of CC cells | (46) |
| CC | In vitro
cell model | Increases the
expression level of CD73 in tumor cells | (78) |
| Adipose tissue | In vitro
cell models and in vivo animal models | Activates the NF-κB
signaling pathway and promotes angiogenesis in CC | (79) |
| CC | In vitro
cell model | Promotes the
secretion of TGF-β1 and the expression level of PD-L1, thereby
inhibiting the antitumor activity of CD8+ T
lymphocytes | (80) |
| CC | In vitro
cell models and in vivo animal models | Expression level of
transcription factor NANOG can promote the proliferation of CC SiHa
cells | (81) |
| Umbilical cord | In vitro
cell models and in vivo animal models | Secretion of VEGF
to enhance in vivo angiogenesis of CC HeLa cells | (83) |
| CC | In vitro
cell model | Enhances the
ability of M2 type macrophages to polarize | (84,85) |
| CC | In vitro
cell model | High expression
levels of CD39 and CD73, promoting the occurrence of immune
escape | (86) |
Promoting the proliferation of tumor
cells
The abnormal proliferation of tumor cells is a
central aspect of tumor progression. A study based on in
vitro cell models found that MSCs derived from CC cells
(CC-MSCs) upregulated the expression level of CD73 in tumor cells,
thereby accelerating the progression of CC (78). Another study using both in
vitro and in vivo models further revealed that the
expression level of the transcription factor NANOG in CC
cell-derived MSCs can promote the proliferation of CC SiHa cells,
thereby driving CC growth (81).
Notably, further research has observed that the effect of bone
marrow-derived MSCs (B-MSCs) on the proliferation of CC cells
exhibits a dose-dependent pattern: Low doses can promote
proliferation, whereas high doses inhibit proliferation; this
phenomenon may be associated with the modulation of the
mitogen-activated protein kinase/phosphatidylinositol 3-kinase
(PI3K) signaling pathways (46).
This finding suggested that the dosage of MSCs used may be one of
the key factors influencing whether MSCs exert pro-tumor or
antitumor effects.
Promoting tumor angiogenesis
Due to the high demand for oxygen and nutrients by
tumor cells, tumor angiogenesis is key to sustaining the early
stages of tumor development and its progression (82). Previously, a study based on in
vitro and in vivo models reported that adipose
tissue-derived MSCs (AD-MSCs) may promote CC angiogenesis by
activating the nuclear factor κ-light-chain-enhancer of activated B
cells signaling pathway, thereby accelerating tumor growth and
metastasis (79). Similarly,
umbilical cord-derived MSCs (UC-MSCs) may enhance in vivo
angiogenesis in CC HeLa cells by secreting vascular endothelial
growth factor (VEGF), thereby promoting the progression of CC
(83).
Suppressing the immune response
MSCs can promote cancer progression by modulating
the immune response within the TME. For instance, CC-MSCs exhibit a
stronger ability to induce M2 macrophage polarization compared with
MSCs derived from normal cervical tissue, thereby fostering an
immunosuppressive microenvironment (84,85).
Furthermore, it has been reported that CC-MSCs can promote the
secretion of TGF-β1 and the expression level of programmed
death-ligand 1 through an adenosine-dependent pathway, subsequently
inhibiting the antitumor activity of CD8+ T lymphocytes
and contributing to cancer promotion (80). Another study also revealed that,
compared with MSCs from normal cervical cells, MSCs derived from CC
cells highly express CD39 and CD73 on their cell membrane. These
molecules serve a notable role in extracellular adenosine
generation and immune evasion (86). However, the aforementioned
mechanisms are primarily based on in vitro experiments and
their in vivo effects require further validation.
Antitumor mechanisms of MSCs and their
derived exosomes in CC
Consistent with their tumor-promoting effects, MSCs
and their derived exosomes can, under specific conditions, exert
antitumor effects by inhibiting tumor cell proliferation, inducing
apoptosis, regulating immune responses and suppressing tumor
angiogenesis (Table II).
 | Table II.MSCs in inhibiting the occurrence and
development of CC. |
Table II.
MSCs in inhibiting the occurrence and
development of CC.
| First author,
year | Source | Product
administered | Research type | Mechanism of
action | (Refs.) |
|---|
| Abas et al,
2022 | Umbilical cord | MSC-Exos | In vitro
cell model | Induces apoptosis
in CC HeLa cells and inhibits the expression levels of proteins
associated with epithelial-mesenchymal transition and as an
effective delivery carrier for paclitaxel, it enhances its
antitumor efficacy | (87) |
| Li et al,
2024 | Umbilical cord | MSC-Exos | In vitro
cell model | miR-370-3p inhibits
the proliferation and migration of CC cells by targeting
DHCR24 | (88) |
| Li et al,
2023 | Umbilical cord | MSC-Exos | In vitro
cell model | Activates the Notch
signaling pathway, promotes the squamous differentiation of CC
CaSki cells, thereby inhibiting tumor proliferation and
metastasis | (89) |
| Kenarkoohi et
al, 2020 | Adipose tissue | MSCs | In vitro
cell models and in vivo animal models | Carries the HSV-TK
lentiviral vector to induce apoptosis in CC cells | (90) |
| Meng et al,
2021 | Bone marrow | MSC-Exos | In vitro
cell models and in vivo animal models | miR-144-3p inhibits
the proliferation and invasion of CC cells and promotes their
apoptosis by targeting CEP55 | (91) |
| Liu et al,
2019 | Menstrual
blood | MSCs | In vitro
cell models and in vivo animal models | Inhibition of CC
HeLa cell proliferation through the TGF-β1-mediated JNK/p21
signaling pathway | (92) |
| Yi et al,
2024 | Umbilical cord | MSC-Exos | In vitro
cell model | Reduces the
expression levels of pro-inflammatory factors while increasing the
expression levels of anti-inflammatory factors | (96) |
| Zhou et al,
2023 | Amniotic fluid | MSCs | In vitro
cell model | Inhibits tumor
angiogenesis | (97) |
| Yang et al,
2022 | Bone marrow | MSC-Exos | In vitro
cell models and in vivo animal models | Delivers miR-331-3p
to regulate the methylation level of LIMS2 in CC cells | (99) |
Inhibiting the proliferation of tumor
cells and inducing tumor cell apoptosis
MSCs and their derived exosomes can inhibit the
growth of CC through multiple signaling pathways. For example,
in vitro cell models have reported that UC-MSC-Exos can
induce apoptosis in CC HeLa cells and suppress the expression
levels of epithelial-mesenchymal transition (EMT)-related proteins,
thereby exerting antitumor effects (87). Additionally, microRNA (miR)-370-3p
carried by such exosomes can inhibit the proliferation and
migration of CC cells by targeting 24-dehydrocholesterol reductase
in in vitro cell models (88). It has also been reported that
UC-MSC-Exos can promote squamous differentiation of CC CaSki cells
by activating the Notch signaling pathway, thereby inhibiting their
growth and metastasis (89). In
in vivo animal models, AD-MSCs carrying the herpes simplex
virus thymidine kinase lentiviral vector can induce cancer cell
apoptosis, thereby delaying CC progression (90). Furthermore, miR-144-3p in B-MSC-Exos
can inhibit the proliferation and invasion of CC cells and promote
their apoptosis by targeting centrosomal protein 55 (91). Menstrual blood-derived MSCs can
inhibit the proliferation of CC HeLa cells through the
TGF-β1-mediated c-Jun N-terminal kinase/p21 signaling pathway,
thereby exerting antitumor effects (92).
Regulating immune responses and
inflammatory microenvironment
Due to the immunosuppressive properties of MSCs,
MSCs are often perceived more as tumor-promoting rather than
antitumor agents (93–95). However, research has also suggested
that MSCs may exert antitumor effects by modulating inflammatory
responses. For example, a previous study by Yi et al
(96) demonstrated that UC-MSC-Exos
can markedly reduce the expression levels of pro-inflammatory
factors such as tumor necrosis factor-α (TNF-α), IL-1β and IL-6,
while increasing the expression of the anti-inflammatory factor
IL-10, thereby alleviating cervical inflammation and potentially
reducing the risk of carcinogenesis by inhibiting the EMT process.
This finding indicated that the anti-inflammatory regulatory
functions of MSCs and their derived exosomes may be associated with
their antitumor effects.
Inhibiting tumor angiogenesis
In contrast to the pro-angiogenic effects of
AD-MSC-Exos (79) and UC-MSC-Exos
(83), a previous study based on an
in vitro cell model indicated that amniotic fluid-derived
MSC-Exos can inhibit the proliferation of CC HeLa cells by
suppressing tumor angiogenesis, thereby exerting antitumor effects
(97). This opposing role in tumor
vascular regulation exhibited by MSCs and their exosomes derived
from different tissues suggests that the cellular source may be a
key factor in determining their functional direction.
Delivering active substances
MSC-Exos inherently carry various bioactive
substances, such as lipids, proteins and nucleic acids, serving as
crucial mediators in intercellular communication (98). Furthermore, following engineering
modifications, MSC-Exos can also function as drug delivery systems
to enhance therapeutic precision. For example, UC-MSC-Exos can
serve as effective delivery vehicles for paclitaxel, augmenting its
antitumor efficacy (79).
Furthermore, B-MSC-Exos can regulate the methylation level of LIM
zinc finger domain 2 in CC cells by delivering miR-331-3p, thereby
inhibiting tumor progression (99).
In summary, MSCs and their exosomes exhibit a
distinct dual role in CC (Fig. 4):
MSCs and their exosomes can both promote tumor cell proliferation,
angiogenesis and immune escape, as well as inhibit tumor cell
proliferation, induce apoptosis and modulate the immune
microenvironment. This bidirectional effect is likely closely
associated with their tissue origin, dosage, administration route
and the specific signaling molecules carried by the exosomes. These
aspects will be discussed in detail in the following sections.
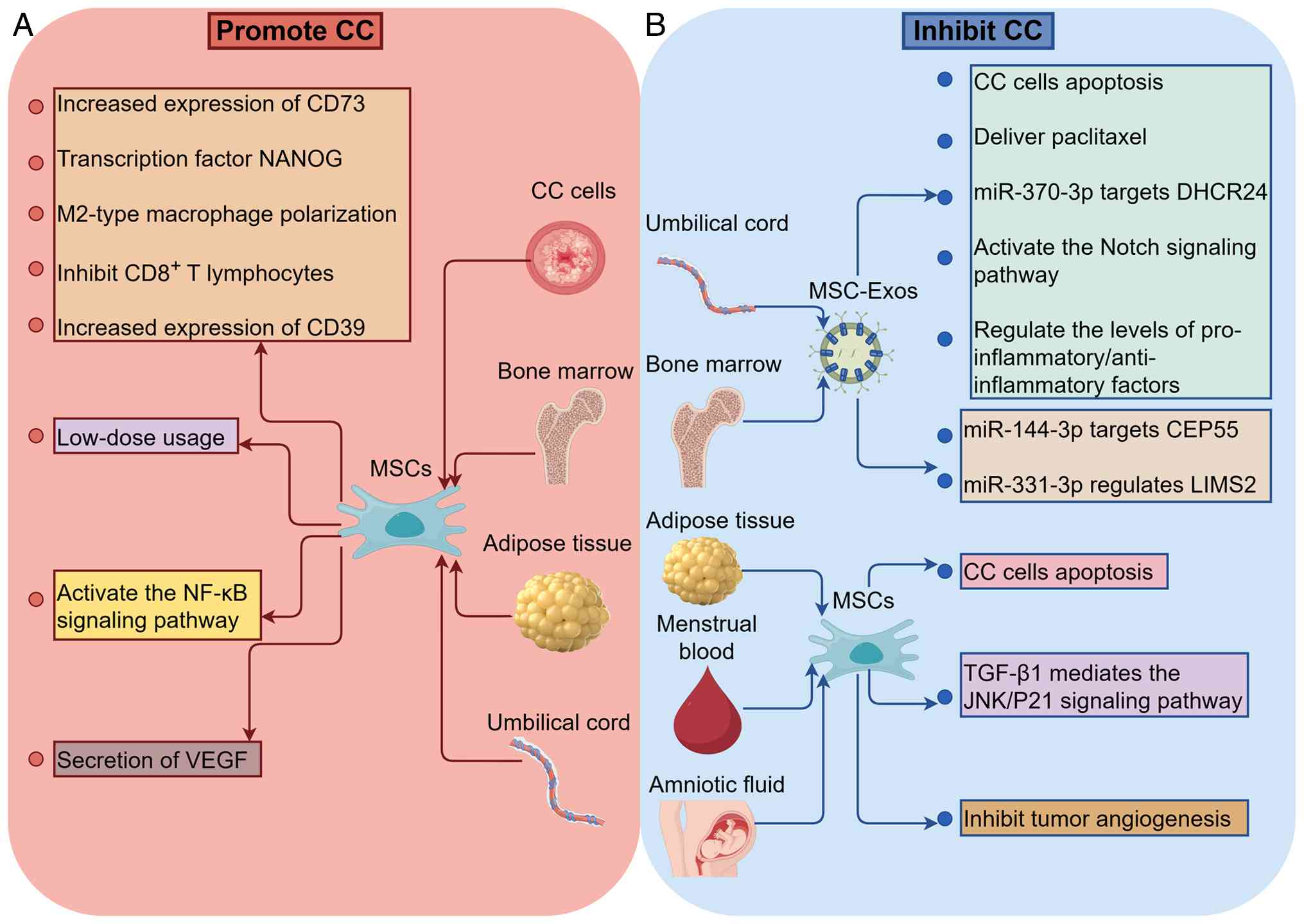 | Figure 4.Bidirectional regulatory mechanism of
MSCs and their derived exosomes in CC. (A) Mechanism by which MSCs
and their derived exosomes promote the development of CC. (B)
Mechanism by which MSCs and their derived exosomes inhibit the
development of CC. The figure was generated using Figdraw
(www.figdraw.com). CC, cervical cancer; NF-κB,
nuclear factor κ-light-chain-enhancer of activated B cells; VEGF,
vascular endothelial growth factor; MSCs, mesenchymal stem cells;
MSC-Exos, exosomes derived from mesenchymal stem cells; DHCR24,
24-dehydrocholesterol reductase; CEP55, centrosomal protein 55;
LIMS2, LIM and senescent cell antigen-like domains 2; TGF-β1,
transforming growth factor β1; JNK/p21, c-Jun N-terminal
kinase/p21. |
Analysis of the bidirectional role of MSCs
and their derived exosomes in CC
During the initiation and progression of CC, MSCs
and their derived exosomes serve a complex and dual-functional
role. Although, MSCs and their derived exosomes can promote tumor
progression through various mechanisms (78–80);
by contrast, under specific conditions, MSCs and their derived
exosomes exhibit antitumor effects (87,96,97,99).
This functional paradox suggests that their dual role may be
regulated by multiple factors, potentially including tissue origin,
dosage and administration methods (46,100,101).
Influence of organizational
origin
The differences in tissue origin may be an important
factor influencing the direction of the role of MSCs and their
derived exosomes in CC. A previous study by Song et al
(102) demonstrated that MSCs from
different sources exhibited notable differences in their
chemotactic ability towards CC cells. This research conducted a
series of in vitro experiments to compare the chemotactic
effects of MSCs from adipose tissue, umbilical cord, amniotic
membrane and chorion on CC cells. The results demonstrated that the
MSCs from chorion had the strongest chemotactic ability. Underlying
this phenomenon may involve the epigenetic signatures carried by
MSCs from different tissue origins. A previous study demonstrated
that MSCs retain the epigenetic characteristics of their tissue of
origin during development, thereby influencing their gene
expression profiles and secretome (103). For instance, B-MSCs may be more
inclined to express factors associated with osteogenesis, whereas
AD-MSCs are enriched in molecules associated with lipid metabolism
and angiogenesis (104). This
finding suggested that although MSCs from different tissue sources
are similar in morphology, immunophenotype and basic activity,
their biological functions may differ, which was consistent with
the conclusion of Kern et al (100).
Influence of dosage
The different dosages used may also result in
opposite effects of the same source of MSCs on CC cells. Long et
al (46) conducted co-culture
experiments of B-MSCs with CC cells HeLa in vitro. The
results indicated that a low proportion of B-MSCs could promote the
proliferation of CC cells, while a high proportion inhibited their
proliferation. This dose-dependent effect may be associated with a
threshold effect in signaling pathways. Specifically, at low doses,
growth factors secreted by MSCs (such as VEGF and TGF-β) might
primarily activate pro-survival signaling pathways (for example,
PI3K/protein kinase B) in tumor cells, thereby promoting
proliferation. However, when the MSCs dose increases to a certain
level, the concentration of their secreted factors also rises,
potentially exceeding a threshold and instead activating
pro-apoptotic or anti-proliferative signaling pathways (involving
molecules such as interferon-γ or TNF-α). Nevertheless, this
hypothesis requires further investigation for confirmation. The
aforementioned phenomenon suggests that when applying MSCs in CC
therapy, the selection of dosage is critically important and may
directly impact therapeutic outcomes.
Differences in functions between MSCs
and their exosomes
Although exosomes are derived from MSCs and share
high functional similarity with MSCs, they may exert different or
even opposing effects during the development of CC. For instance,
UC-MSCs can promote tumor progression by inducing tumor
angiogenesis (83), whereas their
derived exosomes demonstrate antitumor effects (87). This functional discrepancy is
closely associated with the distinct mechanisms of action between
the two. Specifically, MSC-Exos can modulate their secretion and
uptake behavior through various means and interventions, thereby
exhibiting functional characteristics that differ from those of
their parent cells (105). This
understanding suggests that in future research, the relationship
between the two should be regarded as both interconnected and
independent, warranting separate investigation rather than being
simplistically treated as a single entity.
Influence of the administration
route
MSCs and their derived exosomes can be administered
through various routes and the method of administration may
influence their ultimate effects. A previous study has reported
that in mouse models, local injection of B-MSCs exacerbate tumor
growth, whereas no notable difference was observed between the
treatment and control groups after intravenous injection (101). Although this study was based on an
ascites cancer cell model, the results suggested that the route of
administration may affect the function of MSCs. Based on this
phenomenon, the present review hypothesizes that local injection
may result in a high concentration of MSCs aggregating at the tumor
site, yet the sphere of action may be limited and MSCs might be
induced to exhibit pro-tumorigenic effects due to pro-tumoral
signals within the local microenvironment. Furthermore, different
routes of administration lead to distinct patterns of contact
between MSCs and the immune system of the host, potentially
triggering different rates of immune recognition and clearance
(106). This could, in turn,
affect their persistence and functional duration in vivo,
which may also contribute to their differential effects. Therefore,
using CC models in future research is necessary to further
elucidate the differential impacts of various administration routes
in CC.
Challenges and prospects
Although MSCs and their derived exosomes demonstrate
potential for application in CC treatment, their translation to
clinical practice faces a series of notable challenges. Currently,
the large-scale and standardized production of exosomes remains a
considerable challenge (107).
While various isolation and extraction methods exist, such as
ultracentrifugation (108),
ultrafiltration (109),
size-exclusion chromatography (110), precipitation-based separation
(111), immunoaffinity capture
(112) and microchip-based
techniques (113), each of these
approaches has its respective advantages and drawbacks (114). None of the currently available
methods can simultaneously meet the requirements for high purity
and efficiency, operational simplicity and low cost. Therefore,
achieving efficient, accurate and economical isolation of
high-purity exosomes remains a critical issue that urgently needs
to be addressed (115,116). Regarding in vivo
application, the pharmacokinetic profiles, targeted delivery
efficiency and tissue distribution patterns of MSCs and their
derived exosomes are still not well understood, which markedly
compromises the precision and controllability of their therapeutic
use. Furthermore, the long-term safety and potential
tumor-promoting risks require thorough evaluation through
systematic and rigorous preclinical and clinical studies.
To address the aforementioned challenges, future
research could focus on the following directions: First, leveraging
multi-omics technologies to gain deeper insights into the molecular
composition and mechanisms of action of MSCs and their derived
exosomes, thereby providing a theoretical foundation for their
precise application. Second, developing efficient and standardized
isolation and extraction methods to enable the large-scale,
standardized production of MSC-Exos. Additionally, in vivo
experimental studies should be strengthened to further elucidate
their pharmacokinetic behavior, targeted delivery characteristics
and tissue distribution patterns, providing a basis for optimizing
administration strategies. Notably, although MSCs and their derived
exosomes from natural sources hold potential in cancer therapy,
their targeting specificity remains limited. The emergence of
engineering modification techniques offers a novel strategy to
address this issue. MSCs and their derived exosomes can be modified
to express specific tumor-suppressive miRNAs or cytokines by
engineering, thereby enhancing their antitumor activity (117). Concurrently, the feasibility and
efficacy of MSCs and their derived exosomes, whether employed as
monotherapy or as part of combination treatment strategies for CC,
should be systematically evaluated through well-designed
preclinical and clinical trials. This is essential to facilitate
their translation into clinical application. Beyond therapeutic
applications, MSC-Exos also demonstrate marked potential in the
diagnosis and prognosis of CC. As novel tools for liquid biopsy,
the molecular information carried by exosomes can reflect tumor
initiation, progression and response to therapy (118).
Conclusion
MSCs and their derived exosomes serve a complex and
dual-regulatory role in the initiation, progression and metastasis
of CC. Their functions are closely associated with multiple factors
such as tissue origin, dosage and route of administration. A
systematic and comprehensive understanding of this bidirectional
regulatory mechanism not only contributes to further insight into
their biological behavior but also provides a theoretical
foundation in developing novel diagnostic and therapeutic
strategies based on MSCs and their exosomes. Although numerous
challenges remain in their current clinical application for CC,
with the aid of advanced technologies such as engineering
modifications, MSCs and their derived exosomes are expected to
become important tools integrating both diagnostic and therapeutic
functions, thereby opening new avenues for the diagnosis and
treatment of CC in the future.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
XL and DW contributed to the conception and overall
design of the present review. XL and DW drafted the manuscript and
prepared the figures and tables. DW reviewed and revised the
manuscript. Both authors read and approved the final version of the
manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global Cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI
|
|
2
|
Ferrara P, Dallagiacoma G, Alberti F,
Gentile L, Bertuccio P and Odone A: Prevention, diagnosis and
treatment of cervical cancer: A systematic review of the impact of
COVID-19 on patient care. Prev Med. 164:1072642022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Giannella L, Grelloni C, Natalini L,
Sartini G, Bordini M, Delli Carpini G, Di Giuseppe J, Dugo E, Piva
F and Ciavattini A: Molecular features of HPV–Independent cervical
cancers. Pathogens. 14:6682025. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hurjui RM, Hurjui IA, Buțureanu TA,
Popovici D, Avădănei ER and Balan RA: HPV-independent cervical
cancer-a new challenge of modern oncology. Int J Mol Sci.
26:100512025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cohen PA, Jhingran A, Oaknin A and Denny
L: Cervical cancer. Lancet. 393:169–182. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dilruba S and Kalayda GV: Platinum-based
drugs: Past, present and future. Cancer Chemother Pharmacol.
77:1103–1124. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Duska LR, Podwika SE and Randall LM: Top
advances of the year: Cervical cancer. Cancer. 130:2571–2576. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhidu S, Ying T, Rui J and Chao Z:
Translational potential of mesenchymal stem cells in regenerative
therapies for human diseases: Challenges and opportunities. Stem
Cell Res Ther. 15:2662024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pittenger MF, Discher DE, Péault BM,
Phinney DG, Hare JM and Caplan AI: Mesenchymal stem cell
perspective: Cell biology to clinical progress. NPJ Regen Med.
4:222019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang X, Xie Q, Ye Z, Li Y, Che Z, Huang M
and Zeng J: Mesenchymal stem cells and tuberculosis: Clinical
challenges and opportunities. Front Immunol. 12:6952782021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yu H, Huang Y and Yang L: Research
progress in the use of mesenchymal stem cells and their derived
exosomes in the treatment of osteoarthritis. Ageing Res Rev.
80:1016842022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Smojver I, Katalinić I, Bjelica R, Gabrić
D, Matišić V, Molnar V and Primorac D: Mesenchymal stem cells based
treatment in dental medicine: A narrative review. Int J Mol Sci.
23:16622022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mattei V and Delle Monache S: Mesenchymal
stem cells and their role in neurodegenerative diseases. Cells.
13:7792024. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Huang Y and Yang L: Mesenchymal stem cells
and extracellular vesicles in therapy against kidney diseases. Stem
Cell Res Ther. 12:2192021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang Y, Ma D, Wu Z, Yang B, Li R, Zhao X,
Yang H and Zhang L: Clinical application of mesenchymal stem cells
in rheumatic diseases. Stem Cell Res Ther. 12:5672021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
TomyTomcy A and Sindhu ER: Mesenchymal
stem cells-an excellent therapeutic agent for cancer. Asia Pac J
Clin Oncol. 20:7–15. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Vakhshiteh F, Atyabi F and Ostad SN:
Mesenchymal stem cell exosomes: A two-edged sword in cancer
therapy. Int J Nanomedicine. 14:2847–2859. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang Y, Wang H, Tan J, Cao Z, Wang Q, Wang
H, Yue S, Li W and Wang D: Therapeutic effect of mesenchymal stem
cells and their derived exosomes in diseases. Mol Biomed. 6:342025.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Weng Z, Zhang B, Wu C, Yu F, Han B, Li B
and Li L: Therapeutic roles of mesenchymal stem cell-derived
extracellular vesicles in cancer. J Hematol Oncol. 14:1362021.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lyu T, Wang Y, Li D, Yang H, Qin B, Zhang
W, Li Z, Cheng C, Zhang B, Guo R and Song Y: Exosomes from BM-MSCs
promote acute myeloid leukemia cell proliferation, invasion and
chemoresistance via upregulation of S100A4. Exp Hematol Oncol.
10:242021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xunian Z and Kalluri R: Biology and
therapeutic potential of mesenchymal stem cell-derived exosomes.
Cancer Sci. 111:3100–3110. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ahmed HG, Bensumaidea SH, Alshammari FD,
Alenazi FSH, ALmutlaq BA, Alturkstani MZ and Aladani IA: Prevalence
of Human Papillomavirus subtypes 16 and 18 among Yemeni Patients
with Cervical Cancer. Asian Pac J Cancer Prev. 18:1543–1548.
2017.PubMed/NCBI
|
|
23
|
Brandt HM, Footman A, Adsul P, Ramanadhan
S and Kepka D: Implementing interventions to start HPV vaccination
at age 9: Using the evidence we have. Hum Vaccin Immunother.
19:21802502023. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Moody CA and Laimins LA: Human
papillomavirus oncoproteins: Pathways to transformation. Nat Rev
Cancer. 10:550–560. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang Y, Fang J, Liu B, Shao C and Shi Y:
Reciprocal regulation of mesenchymal stem cells and immune
responses. Cell Stem Cell. 29:1515–1530. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lin Z, Zhou Y, Liu Z, Nie W, Cao H, Li S,
Li X, Zhu L, Lin G, Ding Y, et al: Deciphering the tumor immune
microenvironment: single-cell and spatial transcriptomic insights
into cervical cancer fibroblasts. J Exp Clin Cancer Res.
44:1942025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ou Z, Lin S, Qiu J, Ding W, Ren P, Chen D,
Wang J, Tong Y, Wu D, Chen A, et al: Single-Nucleus RNA sequencing
and spatial transcriptomics reveal the immunological
microenvironment of cervical squamous cell carcinoma. Adv Sci
(Weinh). 9:e22030402022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Taheri M, Tehrani HA, Dehghani S,
Rajabzadeh A, Alibolandi M, Zamani N, Arefian E and Ramezani M:
Signaling crosstalk between mesenchymal stem cells and tumor cells:
Implications for tumor suppression or progression. Cytokine Growth
Factor Rev. 76:30–47. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ramírez Idarraga JA and Restrepo Múnera
LM: Mesenchymal stem cells: Their role in the tumor
microenvironment. Tissue Eng Part B Rev. 29:681–691. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bianco P, Robey PG and Simmons PJ:
Mesenchymal stem cells: Revisiting history, concepts, and assays.
Cell Stem Cell. 2:313–319. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Maloney MA and Patt HM: On the origin of
hematopoietic stem cells after local marrow extirpation. Proc Soc
Exp Biol Med. 149:94–97. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Friedenstein AJ, Chailakhjan RK and
Lalykina KS: The development of fibroblast colonies in monolayer
cultures of guinea-pig bone marrow and spleen cells. Cell Tissue
Kinet. 3:393–403. 1970.PubMed/NCBI
|
|
33
|
Friedenstein AJ, Gorskaja JF and Kulagina
NN: Fibroblast precursors in normal and irradiated mouse
hematopoietic organs. Exp Hematol. 4:267–274. 1976.PubMed/NCBI
|
|
34
|
Grigoriadis AE, Heersche JN and Aubin JE:
Differentiation of muscle, fat, cartilage, and bone from progenitor
cells present in a bone-derived clonal cell population: Effect of
dexamethasone. J Cell Biol. 106:2139–2151. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lang H, Schüler N, Arnhold S, Nolden R and
Mertens T: Formation of differentiated tissues in vivo by
periodontal cell populations cultured in vitro. J Dent Res.
74:1219–1225. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Patel SA, Sherman L, Munoz J and Rameshwar
P: Immunological properties of mesenchymal stem cells and clinical
implications. Arch Immunol Ther Exp (Warsz). 56:1–8. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Porada CD and Almeida-Porada G:
Mesenchymal stem cells as therapeutics and vehicles for gene and
drug delivery. Adv Drug Deliv Rev. 62:1156–1166. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Dominici M, Le Blanc K, Mueller I,
Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A,
Prockop Dj and Horwitz E: Minimal criteria for defining multipotent
mesenchymal stromal cells. The International society for cellular
therapy position statement. Cytotherapy. 8:315–317. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Keshtkar S, Azarpira N and Ghahremani MH:
Mesenchymal stem cell-derived extracellular vesicles: Novel
frontiers in regenerative medicine. Stem Cell Res Ther. 9:632018.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li X, Liu H, Han C, Luo J, Guan X, Wang L,
Li Y, Wang J, Piao H, Zou W and Liu J: A Human brain model
mimicking umbilical cord mesenchymal stem cells for the treatment
of hypoxic-ischemic brain injury. Int J Mol Sci. 24:142082023.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
da Silva KN, Marim FM, Rocha GV,
Costa-Ferro ZSM, França LSA, Nonaka CKV, Paredes BD, Rossi EA,
Loiola EC, Adanho CSA, et al: Functional heterogeneity of
mesenchymal stem cells and their therapeutic potential in the
K18-hACE2 mouse model of SARS-CoV-2 infection. Stem Cell Res Ther.
16:152025. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Antoon R, Overdevest N, Saleh AH and
Keating A: Mesenchymal stromal cells as cancer promoters. Oncogene.
43:3545–3555. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Oh IR, Raymundo B, Kim M and Kim CW:
Mesenchymal stem cells co-cultured with colorectal cancer cells
showed increased invasive and proliferative abilities due to its
altered p53/TGF-β1 levels. Biosci Biotechnol Biochem. 84:256–267.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lu W and Allickson J: Mesenchymal stromal
cell therapy: Progress to date and future outlook. Mol Ther.
33:2679–2688. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kresse JC, Gregersen E, Atay JCL, Eijken M
and Nørregaard R: Does the route matter? A preclinical review of
mesenchymal stromal cell delivery to the kidney. APMIS.
131:687–697. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Long X, Matsumoto R, Yang P and Uemura T:
Effect of human mesenchymal stem cells on the growth of HepG2 and
Hela cells. Cell Struct Funct. 38:109–121. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Park S and Jung SC: New sources,
differentiation, and therapeutic uses of mesenchymal stem cells.
Int J Mol Sci. 22:52882021. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Patel AN, Park E, Kuzman M, Benetti F,
Silva FJ and Allickson JG: Multipotent menstrual blood stromal stem
cells: Isolation, characterization, and differentiation. Cell
Transplant. 17:303–311. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Igura K, Zhang X, Takahashi K, Mitsuru A,
Yamaguchi S and Takashi TA: Isolation and characterization of
mesenchymal progenitor cells from chorionic villi of human
placenta. Cytotherapy. 6:543–553. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Reyhani S, Abbaspanah B and Mousavi SH:
Umbilical cord-derived mesenchymal stem cells in neurodegenerative
disorders: From literature to clinical practice. Regen Med.
15:1561–1578. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Joerger-Messerli MS, Marx C, Oppliger B,
Mueller M, Surbek DV and Schoeberlein A: Mesenchymal stem cells
from Wharton's Jelly and amniotic fluid. Best Pract Res Clin Obstet
Gynaecol. 31:30–44. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Entezami S and Sam MR: The role of
mesenchymal stem cells-derived from oral and teeth in regenerative
and reconstructive medicine. Tissue Cell. 93:1027662025. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Jung N, Park S, Choi Y, Park JW, Hong YB,
Park HH, Yu Y, Kwak G, Kim HS, Ryu KH, et al: Tonsil-Derived
mesenchymal stem cells differentiate into a Schwann cell phenotype
and promote peripheral nerve regeneration. Int J Mol Sci.
17:18672016. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Pombero A, Garcia-Lopez R and Martinez S:
Brain mesenchymal stem cells: Physiology and pathological
implications. Dev Growth Differ. 58:469–480. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wang Y, Yu X, Chen E and Li L:
Liver-derived human mesenchymal stem cells: A novel therapeutic
source for liver diseases. Stem Cell Res Ther. 7:712016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
da Silva Meirelles L, Chagastelles PC and
Nardi NB: Mesenchymal stem cells reside in virtually all post-natal
organs and tissues. J Cell Sci. 119((Pt 11)): 2204–2213. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Kao CY and Papoutsakis ET: Extracellular
vesicles: Exosomes, microparticles, their parts, and their targets
to enable their biomanufacturing and clinical applications. Curr
Opin Biotechnol. 60:89–98. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Kowal J, Tkach M and Théry C: Biogenesis
and secretion of exosomes. Curr Opin Cell Biol. 29:116–125. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Zhang Y, Liu Y, Liu H and Tang WH:
Exosomes: Biogenesis, biologic function and clinical potential.
Cell Biosci. 9:192019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Farooqi AA, Desai NN, Qureshi MZ,
Librelotto DRN, Gasparri ML, Bishayee A, Nabavi SM, Curti V and
Daglia M: Exosome biogenesis, bioactivities and functions as new
delivery systems of natural compounds. Biotechnol Adv. 36:328–334.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Colombo M, Raposo G and Théry C:
Biogenesis, secretion, and intercellular interactions of exosomes
and other extracellular vesicles. Annu Rev Cell Dev Biol.
30:255–289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Simons M and Raposo G: Exosomes-vesicular
carriers for intercellular communication. Curr Opin Cell Biol.
21:575–581. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Stoorvogel W, Strous GJ, Geuze HJ,
Oorschot V and Schwartz AL: Late endosomes derive from early
endosomes by maturation. Cell. 65:417–427. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Hanson PI and Cashikar A: Multivesicular
body morphogenesis. Annu Rev Cell Dev Biol. 28:337–362. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Sahu R, Kaushik S, Clement CC, Cannizzo
ES, Scharf B, Follenzi A, Potolicchio I, Nieves E, Cuervo AM and
Santambrogio L: Microautophagy of cytosolic proteins by late
endosomes. Dev Cell. 20:131–139. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Zhang S, Duan Z, Liu F, Wu Q, Sun X and Ma
H: The impact of exosomes derived from distinct sources on
rheumatoid arthritis. Front Immunol. 14:12407472023. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Xu F, Zhang Q, Liu Y, Tang R, Li H, Yang H
and Lin L: The role of exosomes derived from various sources in
facilitating the healing of chronic refractory wounds. Pharmacol
Res. 216:1077532025. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Si C, Gao J and Ma X: Natural killer
cell-derived exosome-based cancer therapy: From biological roles to
clinical significance and implications. Mol Cancer. 23:1342024.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Dai X, Ye Y and He F: Emerging innovations
on exosome-based onco-therapeutics. Front Immunol. 13:8652452022.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Liang B, Peng P, Chen S, Li L, Zhang M,
Cao D, Yang J, Li H, Gui T, Li X and Shen K: Characterization and
proteomic analysis of ovarian cancer-derived exosomes. J
Proteomics. 80:171–182. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Panfoli I, Granata S, Candiano G, Verlato
A, Lombardi G, Bruschi M and Zaza G: Analysis of urinary exosomes
applications for rare kidney disorders. Expert Rev Proteomics.
17:735–749. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Di SJ, Cui XW, Liu TJ and Shi YY:
Therapeutic potential of human breast milk-derived exosomes in
necrotizing enterocolitis. Mol Med. 30:2432024. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Cao M, Diao N, Cai X, Chen X, Xiao Y, Guo
C, Chen D and Zhang X: Plant exosome nanovesicles (PENs): green
delivery platforms. Mater Horiz. 10:3879–3894. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Yi Q, Xu Z, Thakur A, Zhang K, Liang Q,
Liu Y and Yan Y: Current understanding of plant-derived
exosome-like nanoparticles in regulating the inflammatory response
and immune system microenvironment. Pharmacol Res. 190:1067332023.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Li Q, Zhang Y, Shi B, Lin C, Feng Q, Zhou
H, Hao M, Ding Y, Ma C, Mu J and Wang D: Galangin exosomes induce
cell apoptosis through miR-10b/P53 axis in gastric cancer. Sci Rep.
15:128762025. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Kim NH, Kim J, Lee JY, Bae HA and Kim CY:
Application of milk exosomes for musculoskeletal health: Talking
points in recent outcomes. Nutrients. 15:46452023. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Karabay AZ, Barar J, Hekmatshoar Y and
Rahbar Saadat Y: Multifaceted therapeutic potential of
plant-derived exosomes: Immunomodulation, anticancer, anti-aging,
anti-melanogenesis, detoxification, and drug delivery.
Biomolecules. 15:3942025. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Ávila-Ibarra LR, Mora-García ML,
García-Rocha R, Hernández-Montes J, Weiss-Steider B, Montesinos JJ,
Lizano Soberon M, García-López P, López CAD, Torres-Pineda DB, et
al: Mesenchymal stromal cells derived from normal cervix and
cervical cancer tumors increase CD73 expression in cervical cancer
cells through TGF-β1 production. Stem Cells Dev. 28:477–488. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Castro-Oropeza R, Vazquez-Santillan K,
Díaz-Gastelum C, Melendez-Zajgla J, Zampedri C, Ferat-Osorio E,
Rodríguez-González A, Arriaga-Pizano L and Maldonado V:
Adipose-derived mesenchymal stem cells promote the malignant
phenotype of cervical cancer. Sci Rep. 10:142052020. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Marín-Aquino LA, Mora-García ML,
Moreno-Lafont MC, García-Rocha R, Montesinos-Montesinos JJ,
López-Santiago R, Sánchez-Torres LE, Torres-Pineda DB,
Weiss-Steider B, Hernández-Montes J, et al: Adenosine increases
PD-L1 expression in mesenchymal stromal cells derived from cervical
cancer through its interaction with A2AR/A2BR and the production of
TGF-β1. Cell Biochem Funct. 42:e40102024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Gu TT, Liu SY and Zheng PS: Cytoplasmic
NANOG-positive stromal cells promote human cervical cancer
progression. Am J Pathol. 181:652–661. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Burlacu A, Grigorescu G, Rosca AM, Preda
MB and Simionescu M: Factors secreted by mesenchymal stem cells and
endothelial progenitor cells have complementary effects on
angiogenesis in vitro. Stem Cells Dev. 22:643–653. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Lin L, Sun W and Wang L: Effects of
mesenchymal stem cells on angiogenesis of cervical cancer HeLa
cancer cell line HeLa in vivo. Zhonghua Yi Xue Za Zhi.
95:1175–1178. 2015.(In Chinese). PubMed/NCBI
|
|
84
|
Cortés-Morales VA, Chávez-Sánchez L,
Rocha-Zavaleta L, Espíndola-Garibay S, Monroy-García A,
Castro-Manrreza ME, Fajardo-Orduña GR, Apresa-García T,
Gutiérrez-de la Barrera M, Mayani H and Montesinos JJ: Mesenchymal
stem/stromal cells derived from cervical cancer promote M2
macrophage polarization. Cells. 12:10472023. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Bautista-Sebastián E, Cortés-Morales VA,
Fajardo-Orduña GR, Monroy-García A, Castro-Manrreza ME,
Saucedo-Campos AD, Barrera MG, Mayani H and Montesinos JJ:
Interaction between mesenchymal stromal cells and tumor cells
present in cervical cancer influences macrophage polarization.
Cancers (Basel). 17:30992025. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
de Lourdes Mora-García M, García-Rocha R,
Morales-Ramírez O, Montesinos JJ, Weiss-Steider B, Hernández-Montes
J, Ávila-Ibarra LR, Don-López CA, Velasco-Velázquez MA,
Gutiérrez-Serrano V and Monroy-García A: Mesenchymal stromal cells
derived from cervical cancer produce high amounts of adenosine to
suppress cytotoxic T lymphocyte functions. J Transl Med.
14:3022016. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Abas BI, Demirbolat GM and Cevik O:
Wharton jelly-derived mesenchymal stem cell exosomes induce
apoptosis and suppress EMT signaling in cervical cancer cells as an
effective drug carrier system of paclitaxel. PLoS One.
17:e02746072022. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Li W, Zhang C, Gao T, Sun Y, Yang H, Liu
L, Shi M, Ding L, Zhang C, Deng DYB and Li T: Human umbilical cord
mesenchymal stem cells small extracellular vesicles-derived
miR-370-3p inhibits cervical precancerous lesions by targeting
DHCR24. Stem Cells Transl Med. 14:szae0872025. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Li W, Zhang X, Gao T, Liu L, Zhang C, Yang
H, Xie J, Pan W, Deng DYB, Zhang C and Li T: Jagged1 contained in
MSC-derived small extracellular vesicles promotes squamous
differentiation of cervical cancer by activating NOTCH pathway. J
Cancer Res Clin Oncol. 149:18093–18102. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Kenarkoohi A, Bamdad T, Soleimani M,
Soleimanjahi H, Fallah A and Falahi S: HSV-TK expressing
mesenchymal stem cells exert inhibitory effect on cervical cancer
model. Int J Mol Cell Med. 9:146–154. 2020.PubMed/NCBI
|
|
91
|
Meng Q, Zhang B, Zhang Y, Wang S and Zhu
X: Human bone marrow mesenchymal stem cell-derived extracellular
vesicles impede the progression of cervical cancer via the
miR-144-3p/CEP55 pathway. J Cell Mol Med. 25:1867–1883. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Liu QY, Ruan F, Li JY, Wei L, Hu P, Chen
HW and Liu QW: Human menstrual blood-derived stem cells inhibit the
proliferation of HeLa cells via TGF-β1-Mediated JNK/P21 signaling
pathways. Stem Cells Int. 2019:92802982019. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Ridge SM, Sullivan FJ and Glynn SA:
Mesenchymal stem cells: Key players in cancer progression. Mol
Cancer. 16:312017. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Melzer C, Yang Y and Hass R: Interaction
of MSC with tumor cells. Cell Commun Signal. 14:202016. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Norozi F, Ahmadzadeh A, Shahrabi S,
Vosoughi T and Saki N: Mesenchymal stem cells as a double-edged
sword in suppression or progression of solid tumor cells. Tumour
Biol. 37:11679–11689. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Yi C, Wang H, Din MAU, Xia Y, Akanyibah FA
and Mao F: Human umbilical cord mesenchymal stem cells
derived-exosomes alleviate LPS-induced cervical inflammation and
epithelial-mesenchymal transition. Am J Transl Res. 16:6903–6913.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zhou J, Liang T, Wang D, Li L, Cheng Y,
Guo Q and Zhang G: IFNα-expressing amniotic fluid-derived
mesenchymal stem cells migrate to and suppress HeLa cell-derived
tumors in a mouse model. Stem Cells Int. 2018:12413232018.
View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Veziroglu EM and Mias GI: Characterizing
extracellular vesicles and their diverse RNA contents. Front Genet.
11:7002020. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Yang S, Wang L, Gu L, Wang Z, Wang Y, Wang
J and Zhang Y: Mesenchymal stem cell-derived extracellular vesicles
alleviate cervical cancer by delivering microRNA-331-3p to reduce
LIM zinc finger domain containing 2 methylation in tumor cells. Hum
Mol Genet. 31:3829–3845. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Kern S, Eichler H, Stoeve J, Klüter H and
Bieback K: Comparative analysis of mesenchymal stem cells from bone
marrow, umbilical cord blood, or adipose tissue. Stem Cells.
24:1294–1301. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Mostafa A, Mohamed Abdelsalam S, S Sabbah
W and Mekawey D: Mesenchymal stem cells treatment aggravates tumor
growth regardless its route of administration: An in vivo study.
Asian Pac J Cancer Prev. 23:3309–3315. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Song Y, Li R, Ye M, Pan C, Zheng L, Wang
ZW and Zhu X: Differences in chemotaxis of human mesenchymal stem
cells and cervical cancer cells. Apoptosis. 27:840–851. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Godini R, Karami K and Fallahi H: Genome
imprinting in stem cells: A mini-review. Gene Expr Patterns.
34:1190632019. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Costela-Ruiz VJ, Melguizo-Rodríguez L,
Bellotti C, Illescas-Montes R, Stanco D, Arciola CR and Lucarelli
E: different sources of mesenchymal stem cells for tissue
regeneration: A guide to identifying the most favorable one in
orthopedics and dentistry applications. Int J Mol Sci. 23:63562022.
View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Liu Z, Liu Y, Li Y, Xu S and Wang Y, Zhu
Y, Jiang C, Wang K, Zhang Y and Wang Y: ECM stiffness affects cargo
sorting into MSC-EVs to regulate their secretion and uptake
behaviors. J Nanobiotechnology. 22:1242024. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Galipeau J and Sensébé L: Mesenchymal
stromal cells: Clinical challenges and therapeutic opportunities.
Cell Stem Cell. 22:824–833. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
van Niel G, Carter DRF, Clayton A, Lambert
DW, Raposo G and Vader P: Challenges and directions in studying
cell-cell communication by extracellular vesicles. Nat Rev Mol Cell
Biol. 23:369–382. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Johnstone RM, Bianchini A and Teng K:
Reticulocyte maturation and exosome release: Transferrin receptor
containing exosomes shows multiple plasma membrane functions.
Blood. 74:1844–1851. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Yu LL, Zhu J, Liu JX, Jiang F, Ni WK, Qu
LS, Ni RZ, Lu CH and Xiao MB: A comparison of traditional and novel
methods for the separation of exosomes from human samples. Biomed
Res Int. 2018:36345632018. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Lathe GH and Ruthven CR: The separation of
substances on the basis of their molecular weights, using columns
of starch and water. Biochem J. 60:xxxiv1955.PubMed/NCBI
|
|
111
|
Cheng H, Fang H, Xu RD, Fu MQ, Chen L,
Song XY, Qian JY, Zou YZ, Ma JY and Ge JB: Development of a rinsing
separation method for exosome isolation and comparison to
conventional methods. Eur Rev Med Pharmacol Sci. 23:5074–5083.
2019.PubMed/NCBI
|
|
112
|
Yoo CE, Kim G, Kim M, Park D, Kang HJ, Lee
M and Huh N: A direct extraction method for microRNAs from exosomes
captured by immunoaffinity beads. Anal Biochem. 431:96–98. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Wunsch BH, Smith JT, Gifford SM, Wang C,
Brink M, Bruce RL, Austin RH, Stolovitzky G and Astier Y: Nanoscale
lateral displacement arrays for the separation of exosomes and
colloids down to 20 nm. Nat Nanotechnol. 11:936–940. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Li P, Kaslan M, Lee SH, Yao J and Gao Z:
Progress in. exosome isolation techniques Theranostics. 7:789–804.
2017.PubMed/NCBI
|
|
115
|
Royo F, Théry C, Falcón-Pérez JM,
Nieuwland R and Witwer KW: Methods for separation and
characterization of extracellular vesicles: Results of a worldwide
survey performed by the ISEV rigor and standardization
subcommittee. Cells. 9:19552020. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Ramirez MI, Amorim MG, Gadelha C, Milic I,
Welsh JA, Freitas VM, Nawaz M, Akbar N, Couch Y, Makin L, et al:
Technical challenges of working with extracellular vesicles.
Nanoscale. 10:881–906. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Ye M, Liu T, Miao L, Ji H, Xu Z, Wang H,
Zhang J and Zhu X: Cisplatin-encapsulated TRAIL-engineered exosomes
from human chorion-derived MSCs for targeted cervical cancer
therapy. Stem Cell Res Ther. 15:3962024. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Alen BO, Estévez-Pérez LS, Otero Alén M,
Domínguez Hormaetxe S, Simón L and Concha Á: Expression of
epithelial and mesenchymal markers in plasmatic extracellular
vesicles as a diagnostic tool for neoplastic processes. Int J Mol
Sci. 24:35782023. View Article : Google Scholar : PubMed/NCBI
|