Introduction
Peripheral T-cell lymphoma, not otherwise specified
(PTCL-NOS), is the most common histological subtype of PTCL
(1); its clinical, immunophenotypic
and genetic features are highly heterogeneous. The global incidence
of PTCL-NOS is 0.5–1.7 cases per 100,000 person-years (2,3). The
disease shows marked geographic variation; in China, PTCL-NOS
accounts for 23–27% of all non-Hodgkin lymphoma cases, a figure
notably higher than the 10% reported in Western countries (4). In the absence of distinctive
immunophenotypic and molecular signatures, diagnosis is based on
exclusion of other diseases (5).
The clinical course is characteristically aggressive, with most
patients presenting with advanced stage (III/IV) disease (5). Extranodal involvement is seen in ~50%
of cases, most frequently in the bone marrow, skin and
gastrointestinal tract (6). Typical
laboratory abnormalities include anaemia, thrombocytopenia,
elevated lactate dehydrogenase, increased
β2-microglobulin, eosinophilia and
hypergammaglobulinemia (7).
Histologically, diffuse effacement of lymph-node architecture is
the most common pattern; residual B cells are often seen at the
periphery of the infiltrate, and some cases show selective
interfollicular or paracortical involvement. Tumour cells are not
morphologically distinctive, consisting of medium- to large-sized
pleomorphic lymphocytes with irregular nuclei, admixed with
reactive elements, including eosinophils, neutrophils, histiocytes
and plasma cells. Immunohistochemistry (IHC) may reveal loss of
pan-T-cell antigens, most commonly CD5 and CD7. T-follicular helper
(TFH)-associated markers [BCL6, CD10, inducible T-cell
costimulator/CD278, serum amyloid P component, C-X-C motif
chemokine ligand 13 and C-C chemokine receptor type 5 (CCR5)] can
aid in identifying TFH-derived lymphoma; however, isolated
positivity for a single TFH marker does not exclude a diagnosis of
PTCL-NOS (8). PTCL-NOS is
associated with poor outcomes, and no standard-of-care therapy
exists. Current management still borrows from B-cell lymphoma
protocols, with the cyclophosphamide, vincristine, doxorubicin and
prednisone (CHOP) regimen remaining the default first-line regimen.
Although the initial objective response rate is modest, early
relapse is common. For patients treated with CHOP regimen, the
objective response rate (ORR) is 56%, and nearly one-half of the
patients experienced relapse or progression within 6 months after
primary therapy, with a 5-year progression-free survival (PFS) rate
of only 29% (6,9,10). In
recent years, increasing attention in lymphoma research has shifted
toward molecular prognostic biomarkers and pathogenic mechanisms
(11,12). In parallel, a wave of novel targeted
agents has emerged for PTCL, including chidamide, romidepsin and
brentuximab vedotin (BV), all of which have conferred measurable
clinical benefit (8).
Nevertheless, the long-term outlook for
relapsed/refractory PTCL-NOS remains poor. As the disease is rare,
its molecular pathogenesis and mechanisms of drug resistance are
still inadequately defined, and research progress has been slow;
consequently, these patients face a therapeutic impasse. The
present study was therefore designed to delineate the clinical
characteristics of PTCL-NOS, identify potential prognostic
determinants, and provide data to facilitate an earlier diagnosis
and improve long-term survival.
Patients and methods
Patient selection
A retrospective analysis of 30 consecutive,
previously untreated patients with newly diagnosed PTCL-NOS who
attended the Department of Haematology, The Second Hospital of
Hebei Medical University (Shijiazhuang, China) between September
2013 and September 2023 was conducted. The cohort consisted of 22
men and 8 women (male-to-female ratio, 2.75:1), with a median age
of 59 years (age range, 17–70 years). The inclusion criteria for
patient enrollment were as follows: i) Patients were diagnosed by
histopathology and immunohistochemistry, in accordance with the
2017 World Health Organisation classification of haematolymphoid
neoplasms (13); and ii) complete
clinical and follow-up data were available. The exclusion criteria
for patient enrollment were as follows: i) Patients with a second
malignancy or a previous history of malignant tumor; and ii)
patients with severe dysfunction of the heart, lung, liver, kidney
or other vital organs. The study protocol was approved by the
Clinical Research Ethics Committee of The Second Hospital of Hebei
Medical University (approval no. 2024-R760), and written informed
consent was obtained from each patient. All procedures were
performed in accordance with the Institutional Review Board of The
Second Hospital of Hebei Medical University and The Declaration of
Helsinki. Clinical data were extracted from the electronic
medical-record system and included demographics (age and sex),
sites of involvement, Ann Arbor stage (14), Eastern Cooperative Oncology Group
(ECOG) performance status (PS) (15), presence or absence of B symptoms,
laboratory parameters (haemoglobin, platelet count, lactate
dehydrogenase, albumin, β2-microglobulin and Ki-67
proliferation index), imaging studies [positron emission
tomography/computed tomography (CT), abdominal ultrasonography,
superficial lymph-node ultrasonography, and chest and abdominal
CT], histopathological and IHC findings, treatment regimens,
response and long-term outcomes. All pathological diagnoses were
independently reviewed and confirmed by two haematopathologists at
The Second Hospital of Hebei Medical University. Any discrepancies
were resolved via consensus consultation or by a third senior
haematopathologist.
Treatment regimens
In this retrospective cohort, the treatment plans
for the 30 patients with PTCL-NOS were formulated by a
multidisciplinary team after comprehensive evaluation of each
patient's PS, disease burden, Ann Arbor stage, International
Prognostic Index (IPI)/PTCL-NOS Prognostic Index (PIT) score,
molecular prognostic indicators and organ reserve function, in
accordance with the current National Comprehensive Cancer Network
Clinical Practice Guidelines and the Chinese Guidelines for the
Diagnosis and Treatment of Adult Peripheral T-cell Lymphoma, and
were adjusted according to real-world drug accessibility and
patient preferences (16–19).
For first-line chemotherapy regimens, CHOP or
CHOP-like protocols were primarily used. For younger patients with
a good PS (ECOG PS ≤1) who displayed high-risk clinical features
(advanced stage disease, high IPI/PIT score, bulky disease, central
nervous system involvement and leukemic phase), a Ki-67
proliferation index ≥70% and a highly aggressive clinical course,
the cyclophosphamide, doxorubicin, vincristine, prednisone and
etoposide (CHOPE) regimen was preferred. Patients with low- or
intermediate-risk disease without the aforementioned high-risk
factors, elderly patients or those with a slightly poorer PS (ECOG
PS ≥2) were recommended to receive CHOP. For patients with
concomitant cardiovascular disease, anthracycline-containing agents
were avoided where possible, and the gemcitabine, dexamethasone and
cisplatin (GDP) regimen or the gemcitabine and oxaliplatin (GemOx)
regimen was preferred. For patients with hepatic or renal
insufficiency, a reduced-intensity CHOP regimen, GDP/GemOx
regimens, bendamustine monotherapy or bendamustine-based
combination protocols were used based on the patient's specific
conditions.
For second-line chemotherapy regimens, the
dexamethasone, ifosfamide, cisplatin and etoposide (DICE) regimen,
the gemcitabine, vinorelbine and liposomal mitoxantrone (GVM)
regimen, the GDP regimen, single-agent liposomal mitoxantrone, or
the ifosfamide, carboplatin and etoposide (ICE) regimen were
primarily used.
Regarding targeted agents, as chidamide has been
approved for use in China, all newly diagnosed high-risk and
relapsed/refractory patients in the present study were recommended
to receive continuous chidamide until disease progression was
reached or intolerable toxicity to chidamide occurred. For
CD30-positive patients, BV in combination with chemotherapy or used
as monotherapy was recommended; however, overall utilization of
targeted drugs was constrained by multiple real-world factors.
Haematopoietic stem-cell transplantation (HSCT) was
performed in high-risk young patients who achieved complete
response (CR) after first-line induction, and sequential autologous
HSCT as consolidation was considered. For relapsed/refractory
patients who responded to salvage therapy, evaluation for
allogeneic HSCT (allo-HSCT) was performed; due to the profound
influence of real-world circumstances, the proportion of patients
undergoing transplantation in this cohort remained low.
Response assessment and adverse-event
evaluation
Response was graded according to the Lugano criteria
for malignant lymphoma (2014) (20)
and classified as CR, partial response (PR), stable disease (SD) or
progressive disease. Treatment-related adverse events were assessed
and recorded with the Common Terminology Criteria for Adverse
Events version 5.0 (21).
Follow-up
Follow-up was conducted via telephone, and review of
outpatient and inpatient medical records, and concluded in
September 2023. Overall survival (OS) was defined as the time from
diagnosis to death or loss to follow-up for any reason, and PFS was
defined as the time from diagnosis to disease progression,
recurrence, death or last follow-up.
Statistical analysis
Statistical analysis of the case data was performed
using SPSS version 29.0 (IBM Corp.). Continuous data are presented
as the median (range), and categorical data are presented as n (%).
Univariate survival analysis was performed using Kaplan-Meier
analysis, and survival curves were plotted. Intergroup differences
were compared using a log-rank test. Variables with a P-value
<0.05 in the univariate analysis were included in the
multivariate Cox regression model analysis. Hazard ratio (HR) and
its 95% confidence interval (CI) values were calculated. The
χ2 test and Fisher's exact test were used for group
comparisons in Tables V and
VI. Graphs were generated using
GraphPad Prism version 9.5 (Dotmatics). P<0.05 was considered to
indicate a statistically significant difference.
 | Table V.Comparison of baseline clinical
characteristics between the CHOP (n=13) and CHOPE (n=13) regimen
groups. |
Table V.
Comparison of baseline clinical
characteristics between the CHOP (n=13) and CHOPE (n=13) regimen
groups.
| Clinical
characteristic | CHOP regimen group,
n (%) | CHOPE regimen
group, n (%) |
P-valuea |
|---|
| Male sex | 9 (69.2) | 10 (76.9) | >0.999 |
| Age >60
years | 3 (23.1) | 4 (30.8) | >0.999 |
| Ann Arbor stage
III/IV | 12 (92.3) | 11 (84.6) | >0.999 |
| B symptoms | 3 (23.1) | 6 (46.2) | 0.236 |
| Extranodal
involvement sites >1 | 3 (23.1) | 7 (53.8) | 0.226 |
| ECOG score ≥1 | 4 (30.8) | 2 (15.4) | 0.649 |
| Serous cavity
effusion present | 5 (38.5) | 3 (23.1) | 0.673 |
| Hepatomegaly | 2 (15.4) | 3 (23.1) | 0.645 |
| Splenomegaly | 4 (30.8) | 7 (53.8) | 0.428 |
| Bone marrow
involvement | 3 (23.1) | 6 (46.2) | 0.411 |
| HGB <110
g/l | 4 (30.8) | 8 (61.5) | 0.238 |
| PLTs
<150×109/l | 1 (7.7) | 2 (15.4) | >0.999 |
| Β2-MG >4.0
mg/l | 3 (23.1) | 6 (46.2) | 0.411 |
| ALB <35 g/l | 3 (23.1) | 7 (53.8) | 0.226 |
| LDH ≥245 U/l | 9 (69.2) | 8 (61.5) | >0.999 |
| Ki-67 index
≥70% | 6 (46.2) | 11 (84.6) | 0.094 |
 | Table VI.Comparison of baseline
characteristics between the chidamide treatment group (n=18) and
the non-chidamide treatment group (n=12). |
Table VI.
Comparison of baseline
characteristics between the chidamide treatment group (n=18) and
the non-chidamide treatment group (n=12).
| Clinical
characteristic | Chidamide treatment
group, n (%) | Non-chidamide
treatment group, n (%) |
P-valuea |
|---|
| Male sex | 12 (66.7) | 10 (83.3) | 0.419 |
| Age >60
years | 6 (33.3) | 3 (25.0) | 0.704 |
| Ann Arbor stage
III/IV | 14 (77.8) | 11 (91.7) | 0.622 |
| B symptoms | 8 (44.4) | 5 (41.7) | >0.999 |
| Extranodal
involvement sites >1 | 9 (50.0) | 3 (25.0) | 0.264 |
| ECOG score ≥1 | 8 (44.4) | 2 (16.7) | 0.235 |
| Serous cavity
effusion present | 9 (50.0) | 4 (33.3) | 0.465 |
| Hepatomegaly | 6 (33.3) | 2 (16.7) | 0.419 |
| Splenomegaly | 10 (55.6) | 4 (33.3) | 0.284 |
| Bone marrow
involvement | 7 (38.9) | 4 (33.3) | >0.999 |
| HGB <110
g/l | 10 (55.6) | 6 (50.0) | >0.999 |
| PLTs
<150×109/l | 3 (16.7) | 2 (16.7) | >0.999 |
| Β2-MG >4.0
mg/l | 7 (38.9) | 4 (33.3) | >0.999 |
| ALB <35 g/l | 5 (27.8) | 7 (58.3) | 0.130 |
| LDH ≥24 5U/l | 11 (61.1) | 9 (75.0) | 0.694 |
| Ki-67 index
≥70% | 11 (61.1) | 8 (66.7) | >0.999 |
Results
Clinical characteristics
The present study included 30 patients with
PTCL-NOS. The most common presenting symptom was painless
lymphadenopathy, although other symptoms included abdominal pain,
nasal congestion, lower-limb oedema and limb weakness. A total of
13 patients presented with B symptoms, primarily fever. Pleural
effusion was observed at initial diagnosis in 13 patients, with
pleural, abdominal and pelvic effusions most commonly observed
(data not shown). The majority of patients (26 out of 30) were
diagnosed with advanced-stage disease (stage III/IV). Lesions
originated from superficial or deep lymph nodes in 26 patients; in
the remaining 4 cases, they were primary cases in extranodal sites
and presented solely with extranodal involvement (2 in the
gastrointestinal tract, 1 in the skin and 1 in skeletal muscle). At
diagnosis, 23 patients had both nodal and extranodal involvement,
of whom 12 had ≥2 extranodal sites affected (data not shown).
Additional findings included splenomegaly in 14 patients,
hepatomegaly in 8, bone marrow involvement in 11, and extranodal
involvement at other sites, including the lungs and pleura (4
cases), skin (3 cases), thyroid gland (2 cases), kidney (1 case),
and testis (1 case). A total of 3 patients had a history of immune
abnormalities, including 2 with autoimmune diseases and 1 with
acquired immunodeficiency due to human immunodeficiency virus
infection. Additionally, 2 patients were diagnosed with
hemophagocytic syndrome, and 1 patient was diagnosed with
myelofibrosis (data not shonw). All laboratory tests were based on
baseline levels at the first visit. Among the 30 patients, 16 had
anaemia, 5 had thrombocytopenia, 20 had elevated lactate
dehydrogenase levels, 19 had normal β2-microglobulin
levels and 12 had hypoalbuminemia (Table I).
 | Table I.Clinical characteristics of 30
patients with peripheral T-cell lymphoma, not otherwise
specified. |
Table I.
Clinical characteristics of 30
patients with peripheral T-cell lymphoma, not otherwise
specified.
| Clinical
feature | Number of
cases | Percentage |
|---|
| Sex |
|
|
|
Male | 22 | 73.3 |
|
Female | 8 | 26.7 |
| Age, years |
|
|
|
≤60 | 21 | 70.0 |
|
>60 | 9 | 30.0 |
| Ann Arbor
stage |
|
|
|
I/II | 4 | 13.3 |
|
III/IV | 26 | 86.7 |
| B symptoms |
|
|
|
Present | 13 | 43.3 |
|
Absent | 17 | 56.7 |
| Presence of >1
extranodal involvement sites |
|
|
|
Yes | 12 | 40.0 |
| No | 18 | 60.0 |
| ECOG score |
|
|
|
<1 | 20 | 66.7 |
| ≥1 | 10 | 33.3 |
| Serous cavity
effusion |
|
|
|
Present | 13 | 43.3 |
|
Absent | 17 | 56.7 |
| Hepatomegaly |
|
|
|
Present | 8 | 26.7 |
|
Absent | 22 | 73.3 |
| Splenomegaly |
|
|
|
Present | 14 | 46.7 |
|
Absent | 16 | 53.3 |
| Bone marrow
involvement |
|
|
|
Present | 11 | 36.7 |
|
Absent | 19 | 63.3 |
| HGB, g/l |
|
|
|
<110 | 16 | 53.3 |
|
≥110 | 14 | 46.7 |
| PLTs |
|
|
|
<150×109/l | 5 | 16.7 |
|
≥150×109/l | 25 | 83.3 |
| Β2-MG, mg/l |
|
|
|
≤4.0 | 19 | 63.3 |
|
>4.0 | 11 | 36.7 |
| ALB, g/l |
|
|
|
<35 | 12 | 40.0 |
|
≥35 | 18 | 60.0 |
| LDH, U/l |
|
|
|
<245 | 10 | 33.3 |
|
≥245 | 20 | 66.7 |
| Ki-67 index, % |
|
|
|
<70 | 11 | 36.7 |
|
≥70 | 19 | 63.3 |
Immunophenotype
The immunophenotypic features of all 30 patients
were examined (initial pathology reports and stained sections
obtained at The Second Hospital of Hebei Medical University were
collated and re-reviewed; the panel of markers tested was
determined by diagnostic requirements, so the number of evaluable
cases varied between antigens). Antigens were assessed as follows:
Pan-T-cell surface antigen CD2-positive in 100% (27/27),
CD3-positive in 83.3% (20/24), CD5-positive in 56.7% (17/30) and
CD7-positive in 40.0% (12/30) of cases; helper-T-cell subset
CD4-positive in 73.3% (22/30) of cases; cytotoxic-T-cell subset
CD8-positive in 46.7% (14/30) of cases; cytotoxic marker
T-cell-restricted intracellular antigen-1 (TIA-1)-positive in 60.0%
(15/25) of cases; TFH-associated marker BCL-6-positive in 23.1%
(6/26) of cases; and CD30-positive in 53.8% (14/26) of cases
(Table II).
 | Table II.Immunophenotypic characteristics of
30 patients with peripheral T-cell lymphoma, not otherwise
specified. |
Table II.
Immunophenotypic characteristics of
30 patients with peripheral T-cell lymphoma, not otherwise
specified.
| Immune
phenotype | Cases, n/total
na | Percentage |
|---|
| CD2 | 27/27 | 100.0 |
| CD3 | 20/24 | 83.3 |
| CD4 | 22/30 | 73.3 |
| CD5 | 17/30 | 56.7 |
| CD7 | 12/30 | 40.0 |
| CD8 | 14/30 | 46.7 |
| CD30 | 14/26 | 53.8 |
| TIA-1 | 15/25 | 60.0 |
| BCL-6 | 6/26 | 23.1 |
Evaluation of treatment efficacy
In the present study, 28 patients received
chemotherapy alone, whereas 2 patients received sequential
radiotherapy after chemotherapy. Anthracycline-based regimens were
the mainstay of treatment: 13 patients received CHOP, 13 received
CHOPE and 4 received other chemotherapeutic regimens. Chidamide, an
oral histone deacetylase inhibitor, was administered in combination
to 18 patients: 8 received chidamide-based induction followed by
maintenance and 10 received chidamide solely as maintenance after
completing eight cycles of chemotherapy. After four cycles, the ORR
among the 30 patients was 56.7% (17/30): 11 achieved a CR (36.7%)
and 6 a PR (20.0%). A total of 6 patients were assessed as
exhibiting SD (6/30; 20%). In the first-line chemotherapy arms, the
CHOP-treated group of 13 patients achieved an ORR of 46.1% (6/13)
with 5 patients attaining a CR (38.4%) and 1 patient a PR (7.7%),
whereas the CHOPE-treated group of 13 patients achieved an ORR of
61.5% (8/13) with 5 patients attaining a CR (38.4%) and 3 patients
achieving a PR (23.1%). In the chidamide-plus-chemotherapy subgroup
of 8 patients, the ORR was 75.0% (6/8), 5 patients achieved a CR
(62.5%) and 1 achieved a PR (12.5%). At the final efficacy
evaluation, 9 of the aforementioned 11 patients who achieved a CR
in the entire cohort maintained their CR (data not shown).
During the observation period, there were 21
relapsed/refractory patients, among whom 17 received at least one
full cycle of salvage therapy, whereas 4 did not receive any
systemic salvage treatment due to rapid disease progression.
Salvage treatment was predominantly based on conventional
combination chemotherapy, including the DICE regimen in 5 patients,
the GVM regimen in 3 patients, the GDP regimen in 3 patients, the
ICE regimen in 2 patients, the bendamustine plus liposomal
mitoxantrone regimen in 1 patient, and liposomal mitoxantrone
monotherapy in 3 frail patients who could not tolerate intensive
chemotherapy. Within the cohort of 17 patients, 5 received
chemotherapy (GDP/ICE/GVM regimens) combined with chidamide and 1
CD30-positive patient received BV in combination. After 2 cycles, a
response evaluation was performed in the 17 patients who received
salvage therapy, yielding an ORR of 23.5% (4/17), with 2 patients
achieving a CR (11.8%) and 2 achieving a PR (11.8%) (data not
shown).
A total of 5 patients underwent HSCT: 3 received
autologous stem-cell transplantation after achieving CR, and all 3
experienced a sustained CR at the time of last follow-up. Another 2
patients received allo-HSCT after achieving a PR following salvage
therapy for relapsed disease, and both experienced a CR after
transplantation; 1 patient remained alive with a continued CR and 1
succumbed due to severe bloodstream infection post-transplant
(Table III).
 | Table III.Treatment information and follow-up
outcomes of patients post-transplant. |
Table III.
Treatment information and follow-up
outcomes of patients post-transplant.
| Case no. | Age, years | Sex | Ann Arbor
stage | First-line
therapy | Short-term
therapeutic effect | Second-line
therapy | Pre-transplant
condition | Transplant
type | Donor | Conditioning
regimen | Best response after
transplantation | Outcome of
follow-up |
|---|
| 1 | 35 | Male | IV | CHOP regimen
×2 | SD | Chidamide + ICE
regimen ×2 | CR2 | AHCT | / | BEAM regimen | CR | Survival
status |
| 2 | 31 | Female | IV | Chidamide + CHOPE
regimen ×6 | CR | / | CR1 | AHCT | / | BEAM regimen | CR | Survival
status |
| 3 | 54 | Male | IV | CHOP regimen
×6 | CR | Chidamide + GDP
regimen ×4 | CR2 | AHCT | / | CBV regimen | CR | Survival
status |
| 4 | 44 | Male | IV | Chidamide + CHOP
regimen ×4 | SD | Chidamide + GDP
regimen ×6 | PR | Allo-HSCT | Younger
brother | Bu/Cy regimen | CR | Mortality due to
transplant complications |
| 5 | 19 | Male | IV | DA-EPOCH regimen
×2 | SD | BV + ICE regimen
×2 | PR | Allo-HSCT | Father | Bu/Cy regimen | CR | Survival
status |
Survival and prognostic analysis
Follow-up ended in September 2023; among the 30
patients, 15 were alive and 15 had died, 14 of whom succumbed to
disease progression or relapse and 1 who had succumbed to severe
infection after transplantation. The median follow-up duration was
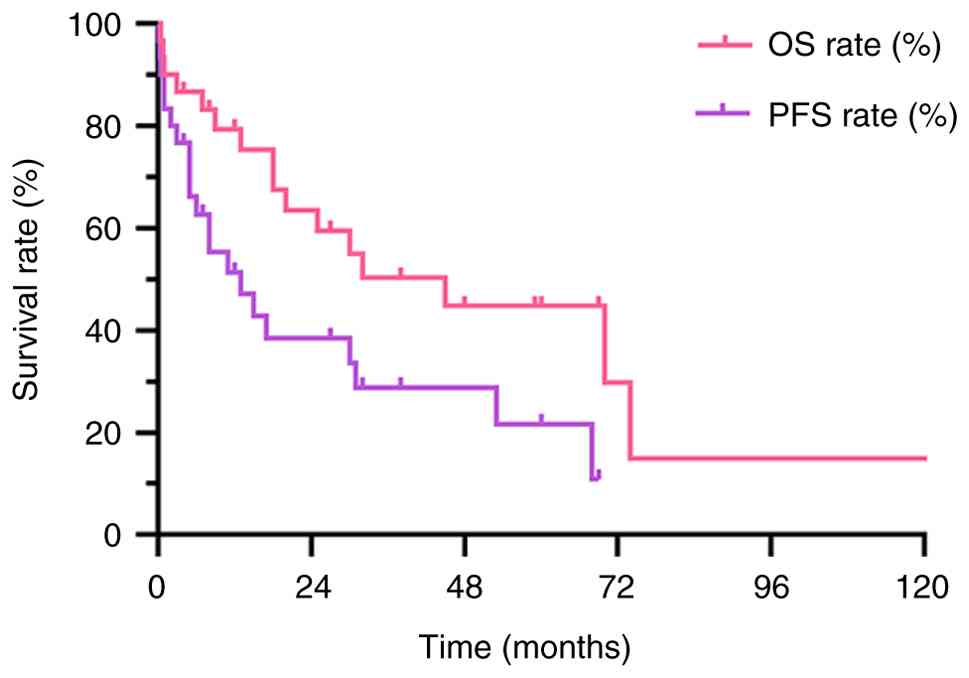
59.0 months. For the entire cohort, the 3-year OS and PFS rates
were 50.4 and 28.9%, respectively, while the 5-year OS and PFS
rates were 44.8 and 21.7% (Fig.
1).
Among the 21 relapsed/refractory patients, the
3-year OS and PFS rates calculated from the date of documented
relapse/refractoriness were 21.8 and 17.9%, respectively (Fig. 2). Based on whether salvage therapy
was administered, these 21 patients were divided into two groups;
given the extremely small sample size of the untreated group (n=4),
no statistical analysis was performed and the data are presented
for descriptive comparison only; baseline characteristics were
compared only descriptively (Table
IV). All 4 patients who did not receive salvage therapy were
male, had serous effusion, haemoglobin <110 g/l and Ki-67 ≥70%,
and were all stage III/IV; the two groups were similar with respect
to PS, number of extranodal sites and bone marrow involvement.
Survival analysis showed that the salvage-treatment group had
3-year OS and PFS rates of 26.9 and 15.7%, respectively, whereas
every patient in the non-salvage therapy group experienced rapid
progression and died; their median PFS time was only 0.22 months
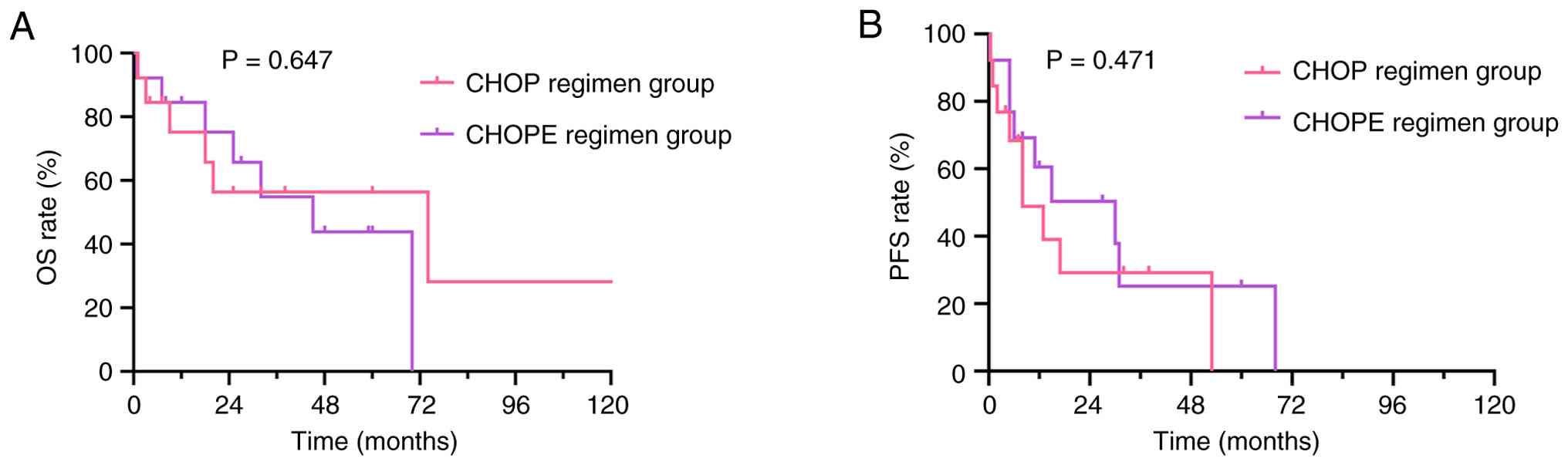
and median OS time was 0.50 months (Fig. 3). Survival analyses were performed
for patients who received CHOP vs. CHOPE (baseline characteristics
had no significant differences; Table
V). The CHOP cohort had a 3-year OS rate of 56.4% and a 3-year
PFS rate of 29.3%, whereas the CHOPE cohort had a 3-year OS rate of
54.8% and a 3-year PFS rate of 25.2%. Neither OS (HR, 0.786; 95%
CI, 0.268–2.334; P=0.647) nor PFS (HR, 1.384; 95% CI, 0.549–3.531;
P=0.471) differed significantly between the two groups (Fig. 4).
 | Table IV.Descriptive comparison of baseline
clinical characteristics between patients in the salvage therapy
group (n=17) and non-salvage therapy group (n=4). |
Table IV.
Descriptive comparison of baseline
clinical characteristics between patients in the salvage therapy
group (n=17) and non-salvage therapy group (n=4).
| Clinical
characteristic | Salvage therapy
group, n (%) | Non-salvage therapy
group, n (%) |
|---|
| Male sex | 11 (64.7) | 4 (100.0) |
| Age >60
years | 11 (64.7) | 3 (75.0) |
| Ann Arbor stage
III/IV | 14 (82.4) | 4 (100.0) |
| B symptoms | 10 (58.8) | 3 (75.0) |
| Extranodal
involvement sites >1 | 10 (58.8) | 2 (50.0) |
| ECOG score ≥1 | 10 (58.8) | 2 (50.0) |
| Serous cavity
effusion present | 11 (64.7) | 4 (100.0) |
| Hepatomegaly | 4 (23.5) | 1 (20.0) |
| Splenomegaly | 8 (47.1) | 1 (20.0) |
| Bone marrow
involvement | 10 (58.8) | 2 (50.0) |
| HGB <110
g/l | 9 (52.9) | 4 (100.0) |
| PLTs
<150×109/l | 15 (88.2) | 2 (50.0) |
| Β2-MG >4.0
mg/l | 6 (35.3) | 3 (75.0) |
| ALB <35 g/l | 8 (47.1) | 3 (75.0) |
| LDH ≥245 U/l | 12 (70.6) | 3 (75.0) |
| Ki-67 index
≥70% | 10 (58.8) | 4 (100.0) |
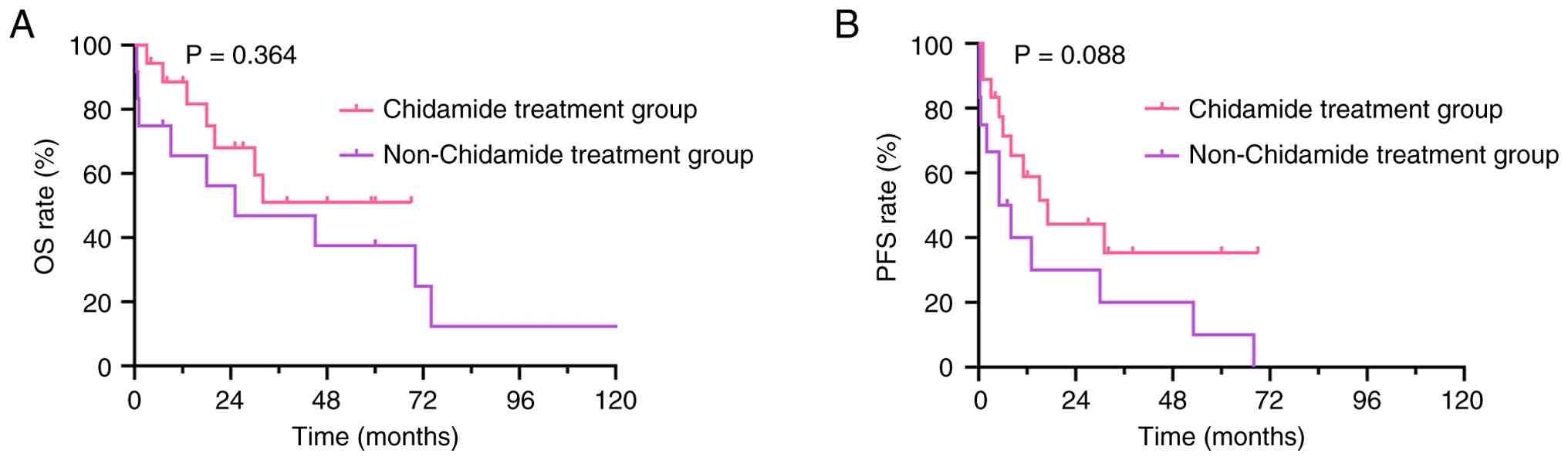
Survival analyses were performed for patients who
received chidamide vs. those who did not (baseline characteristics
had no significant differences; Table
VI). The chidamide group had a 3-year OS rate of 51.1% and a
3-year PFS rate of 35.4%, whereas the non-chidamide group had a
3-year OS rate of 46.9% and a 3-year PFS rate of 20%. Neither OS
(HR, 0.786; 95% CI, 0.268–2.334; P=0.364) nor PFS (HR, 1.384; 95%
CI, 0.549–3.531; P=0.088) differed significantly between the two
groups, although a trend toward improved PFS was observed (Fig. 5).
Prognostic-factor analysis
Univariate analysis
Univariate analysis of the clinical characteristics
of the 30 patients (sex, age, disease stage, B symptoms, number of
extranodal sites, ECOG PS, serous effusion, hepatosplenomegaly,
bone marrow involvement and laboratory parameters), showed that an
ECOG PS >1, serous effusion, bone marrow involvement, a platelet
count <150×109/l and decreased ALB were significantly
associated with worse OS (P<0.05), whereas ECOG PS, bone marrow
involvement, a platelet count <150×109/l,
β2-microglobulin levels >4.0 mg/l, and albumin levels <35 g/l
were significantly associated with worse PFS (P<0.05) (Table VII).
 | Table VII.Influence of different factors on OS
and PFS. |
Table VII.
Influence of different factors on OS
and PFS.
|
| OS | PFS |
|---|
|
|
|
|
|---|
| Clinical
characteristic | Median time,
months | P-value | HR (95% CI) | Median time,
months | P-value | HR (95% CI) |
|---|
| Sex |
|
|
|
|
|
|
|
Male | 30 | 0.850 | 1.119
(0.349–3.586) | 8 | 0.067 | 2.824
(0.932–8.559) |
|
Female | 45 |
|
| 31 |
|
|
| Age, years |
|
|
|
|
|
|
|
≤60 | 32 | 0.814 | 1.130
(0.408–3.130) | 17 | 0.947 | 0.970
(0.388–2.423) |
|
>60 | 45 |
|
| 13 |
|
|
| Ann Arbor
stage |
| 0.644 | 0.739
(0.205–2.660) |
|
|
|
|
I/II | 45 |
|
| 11 | 0.700 | 1.278
(0.367–4.455) |
|
III/IV | 30 |
|
| 13 |
|
|
| B symptoms |
|
|
|
|
|
|
|
Present | 25 | 0.312 | 1.665
(0.620–4.469) | 5 | 0.057 | 2.412
(0.973–5.979) |
|
Absent | 70 |
|
| 30 |
|
|
| Presence of >1
extranodal involvement sites |
|
|
|
|
|
|
|
Yes | 74 | 0.533 | 0.713
(0.247–2.064) | 31 | 0.401 | 0.673
(0.267–1.695) |
| No | 30 |
|
| 8 |
|
|
| ECOG score |
|
|
|
|
|
|
| ≤1 | 7 | 0.001a | 6.327
(2.090–19.152) | 1 | 0.021a | 2.922
(1.178–7.249) |
|
>1 | 70 |
|
| 15 |
|
|
| Serous cavity
effusion |
|
|
|
|
|
|
|
Present | 3 | 0.025a | 3.507
(1.167–10.536) | 5 | 0.261 | 1.642
(0.691–3.901) |
|
Absent | 30 |
|
| 15 |
|
|
| Hepatomegaly |
|
|
|
|
|
|
|
Present | - | 0.831 | 0.870
(0.242–3.124) | 1 | 0.729 | 1.199
(0.429–3.351) |
|
Absent | 32 |
|
| 5 |
|
|
| Splenomegaly |
|
|
|
|
|
|
|
Present | 30 | 0.637 | 1.269
(0.471–3.418) | 11 | 0.441 | 1.426
(0.578–3.518) |
|
Absent | 70 |
|
| 17 |
|
|
| Bone marrow
involvement |
|
|
|
|
|
|
|
Present | 9 | 0.003a | 4.705
(1.679–13.183) | 2 |
<0.001a | 6.391
(2.514–16.248) |
|
Absent | 70 |
|
| 30 |
|
|
| HGB, g/l |
|
|
|
|
|
|
|
<110 | 20 | 0.276 | 1.759
(0.636–4.862) | 5 | 0.139 | 1.955
(0.805–4.750) |
|
≥110 | 70 |
|
| 31 |
|
|
| PLTs |
|
|
|
|
|
|
|
<150×109/l | 3 | 0.002a | 11.474
(2.484–52.997) | 1 | 0.014a | 4.438
(1.353–14.559) |
|
≥150×109/l | 70 |
|
| 15 |
|
|
| Β2-MG, mg/l |
|
|
|
|
|
|
|
≤4.0 | 70 | 0.087 | 2.524
(0.873–7.296) | 30 | 0.031a | 2.276
(1.098–6.765) |
|
>4.0 | 13 |
|
| 3 |
|
|
| ALB, g/l |
|
|
|
|
|
|
|
<35 | 18 | 0.035a | 2.969
(1.077–8.183) | 6 | 0.016a | 3.152
(1.243–7.995) |
|
≥35 | 70 |
|
| 53 |
|
|
| LDH, U/l |
|
|
|
|
|
|
|
<245 | 30 | 0.176 | 2.226
(0.698–7.102) | 8 | 0.108 | 2.264
(0.806–6.361) |
|
≥245 | 70 |
|
| 30 |
|
|
| Ki-67 index, % |
|
|
|
|
|
|
|
<70 | 45 | 0.776 | 0.857
(0.296–2.478) | 13 | 0.365 | 1.529
(0.599–3.905) |
|
≥70 | 75 |
|
| 17 |
|
|
Multivariate analysis
Factors significant in the univariate analysis were
entered into a Cox regression model. ECOG PS >1, bone marrow
involvement and platelet count <150×109/l were
independent predictors of OS (Table
VIII), whereas bone marrow involvement and albumin levels
<35 g/l were independent predictors of PFS (Table IX).
 | Table VIII.Multivariate analysis of OS with Cox
proportional hazards regression. |
Table VIII.
Multivariate analysis of OS with Cox
proportional hazards regression.
|
| OS |
|---|
|
|
|
|---|
| Clinical
characteristic | HR | 95% CI | P-value |
|---|
| ECOG score
>1 | 4.223 | 1.089–16.383 | 0.037a |
| Serous cavity
effusion present | 2.705 | 0.730–10.029 | 0.137 |
| Bone marrow
involvement | 3.354 | 1.016–11.065 | 0.047a |
| PLTs
<150×109/l | 7.353 | 1.096–49.330 | 0.040a |
| ALB <35 g/l | 2.648 | 0.787–8.915 | 0.116 |
 | Table IX.Multivariate analysis of PFS with Cox
proportional hazards regression. |
Table IX.
Multivariate analysis of PFS with Cox
proportional hazards regression.
|
| PFS |
|---|
|
|
|
|---|
| Clinical
characteristic | HR | 95% CI | P-value |
|---|
| ECOG score
>1 | 1.154 | 0.340–3.916 | 0.819 |
| Bone marrow
involvement | 6.140 | 1.982–19.018 | 0.002a |
| PLTs
<150×109/l | 3.887 | 0.897–16.841 | 0.070 |
| Β2-MG >4.0
mg/l | 2.090 | 0.669–6.531 | 0.205 |
| ALB <35 g/l | 3.595 | 1.159–11.154 | 0.027a |
Adverse events
All 30 patients experienced treatment-related
adverse events. Grade 3–4 toxicities were predominantly
haematological in nature (leukopenia, anaemia and
thrombocytopenia). Each episode was managed with supportive care
[subcutaneous granulocyte-colony stimulating factor (G-CSF) or
recombinant human thrombopoietin, or blood product transfusion],
and blood counts recovered to acceptable levels before the next
cycle. Non-haematological toxicities were primarily grade 1–2.
Every patient developed some degree of gastrointestinal symptoms
(nausea, vomiting or loss of appetite); symptomatic management
(oral or parenteral vitamin B6 or 5-HT3 receptor
antagonists) led to improvement. A total of 10 patients had
pneumonia, 2 of which were grade 3–4 and delayed the next
chemotherapy cycle. Neurotoxic events consisted primarily of mild
peripheral neuropathy; follow-up of 6 patients showed gradual
resolution after drug withdrawal. A total of 3 patients developed
grade 1–2 hepatic dysfunction; only 1 episode occurred during
active treatment, which resolved after suspension of chemotherapy
and addition of glycyrrhizin preparations. None of the patients
discontinued therapy due to severe adverse events (Table X).
 | Table X.Adverse events during treatment of 30
patients with peripheral T-cell lymphoma, not otherwise
specified. |
Table X.
Adverse events during treatment of 30
patients with peripheral T-cell lymphoma, not otherwise
specified.
| Adverse event | Number of
cases | Percentage | Cumulative
percentage |
|---|
| Hematological
toxicity |
|
|
|
|
Leukopenia |
|
| 80.0 |
|
Grade 1–2 | 15 | 50.0 |
|
|
Grade 3–4 | 9 | 30.0 |
|
|
Anemia |
|
| 56.7 |
|
Grade 1–2 | 12 | 40.0 |
|
|
Grade 3–4 | 5 | 16.7 |
|
|
Thrombocytopenia |
|
| 86.7 |
|
Grade 1–2 | 12 | 40.0 |
|
|
Grade 3–4 | 6 | 20.0 |
|
| Non-haematological
toxicity |
|
|
|
|
Gastrointestinal
reactions |
|
| 100.0 |
|
Grade 1–2 | 28 | 93.3 |
|
|
Grade 3–4 | 2 | 6.7 |
|
|
Peripheral neuropathy |
|
| 20.0 |
|
Grade 1–2 | 6 | 20.0 |
|
|
Grade 3–4 | 0 | 0.0 |
|
|
Pneumonia |
|
| 36.7 |
|
Grade 1–2 | 9 | 30.0 |
|
|
Grade 3–4 | 2 | 6.7 |
|
|
Abnormal liver function |
|
| 10.0 |
|
Grade 1–2 | 3 | 10.0 |
|
|
Grade 3–4 | 0 | 0.0 |
|
Discussion
PTCL-NOS is the most common subtype of PTCL,
accounting for 21–27% of all PTCL cases (22). Although PTCL-NOS remains defined as
a heterogeneous collection of tumours that cannot be assigned to
any other recognized PTCL category, recent advances in PTCL
genomics have led to the reclassification of cases exhibiting a TFH
phenotype or Epstein-Barr virus (EBV) positivity as distinct
entities, thereby narrowing the diagnostic scope of PTCL-NOS
(23).
In the present study, the clinical characteristics
of 30 patients with PTCL-NOS were analysed; the median age at
diagnosis was 59 years, and the disease was more common among men
(73.3%). The majority of patients presented with painless
peripheral lymphadenopathy, and approximately one-half (43%) had B
symptoms (fever, night sweats or weight loss). Occasional
co-occurrence of hemophagocytic syndrome and myelofibrosis was also
observed, which is consistent with a previous report (6). PTCL-NOS typically arises in the lymph
nodes, but the present cohort also included rare primary extranodal
cases involving the gastrointestinal tract, skin and muscle.
Previous research has shown that the primary site influences
outcome: Tumours originating in the kidney or gastrointestinal
tract are associated with poorer survival than nodal disease,
whereas primary bone, skin or soft-tissue involvement confers a
better prognosis (24).
Immunophenotypically, all patients in the present
cohort exhibited partial or complete loss/downregulation of
pan-T-cell antigens, most frequently CD7 and CD5. Overall, 60% of
cases were positive for the cytotoxic marker TIA-1, a figure
considerably higher than the 32% reported in Western countries
(North America and Europe) (6) and
further evidence that cytotoxic PTCL-NOS (cPTCL-NOS) is more common
in Asian populations (25).
cPTCL-NOS is defined by expression of ≥1 cytotoxic marker (TIA-1,
granzyme B or perforin) in ≥50% of tumour cells (25). These patients often have an
immunocompromised background, such as a history of malignancy or
autoimmune disease, and the pathogenesis is linked to mutations in
epigenetic regulators, including tet methylcytosine dioxygenase 2
and DNA methyltransferase 3α. Studies have shown that cPTCL-NOS
typically expresses the T-helper 1-associated transcription factors
TBX21 and C-X-C chemokine receptor type 3 (CXCR3), and belongs to
the PTCL-TBX21 subgroup, which is associated with a poor prognosis
(26). PTCL-TBX21 is driven by the
transcription factor TBX21 and characterized by aberrant activation
of the STAT3 pathway; another subgroup identified by gene
expression profiling is PTCL-GATA3, which is driven by the
transcription factor GATA3. Compared with PTCL-TBX21, PTCL-GATA3
exhibits a more aberrant genomic expression profile and is
associated with PI3K pathway activation, with a worse overall
prognosis (27). An IHC algorithm
using TBX21/CXCR3 and GATA3/CCR4 as markers has been developed to
distinguish PTCL-TBX21 and PTCL-GATA3 in clinical practice
(28). Due to the lack of fresh,
formalin-fixed, paraffin-embedded pathological samples, it was not
possible to perform supplementary IHC staining for the
aforementioned markers and gene-expression profiling in the present
study, thereby limiting the ability to validate the prognostic
value of molecular subtypes and specific gene mutations in
cPTCL-NOS in the real-world setting. Notably, previous cohort
studies of cPTCL-NOS primarily included EBV-positive cases
(28,29); these have now been reclassified as
distinct entities. Large-scale investigations are urgently needed
to determine whether EBV-negative cPTCL-NOS exhibits clinical
features and survival outcomes that differ from those of
non-cPTCL-NOS (30). In the present
study, the CD30 positivity rate was 53.8%, which is higher than
that reported in an earlier study (22), likely reflecting differences in the
CD30 positivity cutoff and the limited sample size. This finding
indicates that BV, an antibody-drug conjugate targeting CD30, could
be a therapeutic option for these patients.
Currently, the clinical prognostic indices most
frequently used for PTCL-NOS are the IPI, the PIT and the modified
PIT; all three models stratify patients according to age, ECOG PS,
LDH level, number of extranodal sites and Ki-67 expression
(6,19,31,32).
The present study confirmed that an ECOG PS >1, bone marrow
involvement and platelet count <150×109/l are independent
predictors of OS, which is consistent with previous reports
(6,31,32).
In addition to bone marrow involvement, albumin levels <35 g/l
were an independent predictor of PFS. Low serum albumin levels have
been linked to poor outcomes in a variety of malignancies,
including gastric, small-cell lung and breast cancer (33–35),
and its prognostic value across PTCL subtypes has been confirmed by
multiple studies (36–39). Albumin reflects not only systemic
nutritional status but also the inflammatory burden and tumour
load. Pro-inflammatory cytokines released within the tumour
microenvironment, such as IL-6 and tumour necrosis factor-α,
suppress hepatic albumin synthesis, whereas increased capillary
leakage accelerates the development of hypoalbuminemia. Raj et
al (40) analysed 149 patients
with large B-cell lymphoma and demonstrated that C-reactive protein
and IL-6 levels were inversely associated with serum albumin level,
confirming that albumin behaves as a negative acute-phase protein
that mirrors the high inflammatory state of lymphoma. Further
mechanistic studies have shown that IL-6 is a key component of a
positive inflammatory feedback loop, activating the JAK1-STAT3
pathway, and thereby promoting proliferation and migration of PTCL
cells (41,42). Federico et al (36) incorporated the prognostic value of
serum albumin into a T-cell scoring system that combines serum
albumin level, ECOG PS, disease stage and absolute neutrophil
count, and validated its ability to predict both OS and PFS.
Although this model, derived from a large prospective dataset,
achieved greater predictive accuracy than earlier indices, it
remains limited to clinical variables and offers limited guidance
for treatment selection. In the present study, albumin was measured
only once prior to treatment initiation, precluding any dynamic
assessment of its relationship with therapeutic response. Owing to
the retrospective nature of the data, inflammatory markers such as
C-reactive protein and IL-6 were excluded from the model;
therefore, the proposed link between hypoalbuminemia and systemic
inflammation could not be formally validated. Large prospective
cohorts are still needed to clarify how pretreatment albumin levels
are associated with nutritional status and inflammatory burden in
real-world PTCL-NOS, with the ultimate goal of developing robust
predictive algorithms that can reliably identify high-risk
patients.
In the present cohort, anthracycline-based
chemotherapy was the primary strategy, yielding an ORR of 56.7% and
a 5-year OS of 44.8%, figures that mirror historical data (6). CHOP-like regimens perform poorly in
PTCL-NOS. Although first-line response rates are acceptable, early
relapse often occurs, and long-term outlook remains poor. The value
of anthracyclines is itself debated; several studies have found no
notable survival difference between anthracycline-containing and
anthracycline-free schedules (6,43). In
the present study, the outcomes between patients receiving CHOP and
those receiving CHOPE were compared, and no statistically
significant difference in either OS or PFS was observed. A previous
report has suggested that CHOPE may improve PFS rate in patients
<60 years old, possibly as elderly or frail individuals are less
tolerant of the myelosuppressive effects of etoposide (44); therefore, age-stratified analyses
are required to clarify the true benefit of the CHOPE regimen.
Chidamide, a selective histone deacetylase inhibitor
developed in China, is now widely used to manage PTCL and shows
favourable activity (45). In the
present study, patients who received chidamide exhibited higher
3-year OS and PFS rates compared with those who did not receive the
treatment, with a notable trend toward improved PFS. However, this
difference did not reach statistical significance, likely due to
the limited sample size. The small sample size also precluded
stratified analysis of outcomes based on different chidamide
treatment strategies, such as up-front combination therapy vs.
maintenance therapy. Additionally, clinicians tend to initiate
chidamide treatment earlier in patients deemed high-risk.
Furthermore, financial constraints prevent some patients from
completing the planned number of treatment cycles. These factors
may bias the efficacy estimates toward the null, potentially
obscuring the true therapeutic benefits of chidamide. The small
cohort and variable follow-up duration further reduced the
statistical power in the present study. Consequently, the negative
result observed did not refute the therapeutic value of chidamide,
rather, it simply documents the heterogeneity of its real-world
use. Large prospective studies are required to define the precise
role of chidamide as first-line and maintenance therapy, and to
determine whether molecular subgroups derive differential benefit.
BV, an anti-CD30 antibody-drug conjugate, has demonstrated
therapeutic potential in CD30-positive lymphoma (46,47).
In the present study, 1 patient who relapsed (repeat lymph-node
biopsy confirming CD30 positivity) received BV combined with
chemotherapy as salvage therapy, and achieved PR, before proceeding
to allo-HSCT. This single-case experience suggests that BV is
active in PTCL-NOS. Beyond improving drug accessibility (such as
reducing economic burden), an evidence-based CD30 cutoff must be
established, and prospective real-world studies on the efficacy and
safety of BV in China are urgently needed to inform clinical
decision-making.
A total of 3 patients in the present study underwent
autologous stem-cell transplantation after achieving CR, and all
remain alive at the latest follow-up. The precise value of upfront
autologous stem-cell transplantation in PTCL-NOS is still
contested. A large retrospective study showed improved OS when
patients aged <65 years with stage II–IV disease received
autologous stem-cell transplantation as consolidation (48), whereas a prospective trial found no
survival benefit for sequential autologous stem-cell
transplantation in first-remission PTCL-NOS (49). Although existing data are
conflicting, and large-scale studies specific to the PTCL-NOS
subtype are lacking, current guidelines and clinical practice
continue to favour sequential autologous stem-cell transplantation
for chemotherapy-sensitive patients, on the assumption that it may
achieve long-term disease control.
Patients with relapsed or refractory PTCL-NOS have
poor outcomes, effective agents are limited, and they remain in a
challenging therapeutic situation. In the present cohort, the
3-year OS was only 21.8%, and 4 of the 21 patients died from rapid
progression, within a median OS time of 0.50 months, without ever
receiving salvage therapy. Although the sample is too small to
identify statistically robust high-risk features, all four had high
stage disease, serous effusion, anaemia and a Ki-67 index ≥70%,
suggesting that an inherently aggressive biology, rather than poor
PS, drives the fulminant course. Larger studies integrated with
molecular genetics are required to determine whether an
ultra-high-risk molecular subset exists. Salvage regimens in the
present study were highly heterogeneous; conventional chemotherapy
(GDP and ICE) conferred only a modest survival benefit, which is
consistent with previous reports (50–52).
Notably, the mere opportunity to receive salvage therapy emerged as
a survival ‘watershed’, implying that published response rates may
be systemically overestimated. Additionally, the use of targeted
agents (chidamide and BV) in Chinese patients with
relapsed/refractory PTCL-NOS was piloted, providing preliminary
experience for individualized, targeted combination strategies.
Transplant-related mortality is undeniable, yet allo-HSCT remains
the only curative option for relapsed/refractory PTCL-NOS. In the
present study, only 2 patients were bridged to allo-HSCT; most
younger individuals never achieved a response adequate for
transplant, and the need to locate a suitable donor and secure
costly procedures before further progression created additional,
real-world barriers. More effective pre-transplant salvage regimens
are urgently required, while frail patients who are ineligible for
transplants need lower-intensity alternatives that can meaningfully
prolong survival. As the genetic understanding of PTCL pathogenesis
improves, novel targeted agents, such as the JAK1 inhibitor
golidocitinib, the PI3K inhibitor linperlisib and the programmed
cell death protein 1 inhibitor pembrolizumab, are emerging,
offering patients with relapsed/refractory PTCL-NOS additional
therapeutic options. Thus, patients with PTCL-NOS may achieve
sustained remission or even disease-free status with effective
targeted therapies.
In the present study, treatment-related toxicities
were generally manageable; the most common grade 3–4 adverse events
were myelosuppression-neutropenia (30%), anaemia (16.7%) and
thrombocytopenia (20%). These rates are largely consistent with
those of a previous report (53).
Pneumonia was the leading cause of treatment delay, and the two
grade 3–4 pulmonary infections were both neutropenia-related.
Clearly, maintaining chemotherapy intensity to achieve remission
while avoiding severe myelosuppression-related complications
remains a formidable clinical challenge. Prophylactic G-CSF can
reduce the incidence of serious infections and maintain cycle
schedules, but cumulative toxicity, dynamic blood count trends and
individual infection histories must all be considered. Therefore, a
multidisciplinary consultation should be sought promptly when
necessary. Overall, the regimens used in the present study were
considered safe, and no treatment-related deaths occurred.
The present study has several important limitations
that restrict the generalizability of the findings. First, the
overall sample size and the sizes of individual subgroups are
small, limiting statistical power and increasing the risk of type
II errors; comparisons of survival between groups must therefore be
interpreted with caution. Second, the study is a single-centre
retrospective analysis and is subject to inherent selection and
recall biases; although baseline clinical characteristics were
compared across all subgroups, potential confounders such as
socioeconomic factors, patient treatment preferences and individual
physician choices may still have influenced survival outcomes.
Third, the absence of an external validation cohort means that the
results are exploratory only; they provide clinical evidence and
generate preliminary hypotheses for this rare disease, but a large,
multicentre, prospective study is required for confirmation.
In summary, PTCL-NOS predominantly affects
middle-aged and elderly men, follows an aggressive clinical course
and is associated with poor overall outcomes. Conventional
CHOP-like regimens provide sub-optimal disease control; relapse is
common and robust prognostic tools are lacking. Multicentre
prospective studies with larger patient cohorts are urgently
required, together with comprehensive mapping of the PTCL-NOS
molecular landscape, to elucidate the biological basis of its
clinical heterogeneity and to develop effective targeted
combination strategies that improve long-term survival for patients
with this disorder.
Acknowledgements
The abstract was presented at the 66th Annual
Meeting of the American Society of Hematology, December 7–10, 2024,
in San Diego, CA, and published as abstract no. 204589 in Blood 144
(Suppl 1): 6404, 2024.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
MM and YZ contributed equally to this work. MM and
YZ were involved in the conception and design of the study, data
analysis and interpretation, drafting of the manuscript and
preparation of the figures. LX and QW participated in data
collection, data collation and data curation. SQ contributed to the
conception and design of the study, supervised the research,
critically revised the manuscript for important intellectual
content, and gave final approval of the version to be published. MM
and YZ confirm the authenticity of all the raw data. All authors
have read and approved the final manuscript, and agree to be
accountable for all aspects of the work.
Ethics approval and consent to
participate
All procedures involved in this study were approved
by the Institutional Review Board and Ethics Committee of The
Second Hospital of Hebei Medical University (Shijiazhuang, China;
approval no. 2024R760). Written informed consent was obtained from
each patient.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Swerdlow SH, Campo E, Pileri SA, Harris
NL, Stein H, Siebert R, Advani R, Ghielmini M, Salles GA, Zelenetz
AD and Jaffe ES: The 2016 revision of the World Health Organization
classification of lymphoid neoplasms. Blood. 127:2375–2390. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Adams SV, Newcomb PA and Shustov AR:
Racial patterns of peripheral T-cell lymphoma incidence and
survival in the United States. J Clin Oncol. 34:963–971. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sant M, Allemani C, Tereanu C, De Angelis
R, Capocaccia R, Visser O, Marcos-Gragera R, Maynadié M, Simonetti
A, Lutz JM, et al: Incidence of hematologic malignancies in Europe
by morphologic subtype: Results of the HAEMACARE project. Blood.
116:3724–3734. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shi Y: Current status and progress of
lymphoma management in China. Int J Hematol. 107:405–412. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
d'Amore F, Gaulard P, Trümper L, Corradini
P, Kim WS, Specht L, Bjerregaard Pedersen M and Ladetto M; ESMO
Guidelines Committee, : Peripheral T-cell lymphomas: ESMO clinical
practice guidelines for diagnosis, treatment and follow-up. Ann
Oncol. 26 (Suppl 5):v108–v115. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Weisenburger DD, Savage KJ, Harris NL,
Gascoyne RD, Jaffe ES, MacLennan KA, Rüdiger T, Pileri S, Nakamura
S, Nathwani B, et al: Peripheral T-cell lymphoma, not otherwise
specified: A report of 340 cases from the international peripheral
T-cell lymphoma project. Blood. 117:3402–3408. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Oluwasanjo A, Kartan S, Johnson W,
Alpdogan O, Gru A, Mishra A, Haverkos BM, Gong J and Porcu P:
Peripheral T-cell lymphoma, not otherwise specified (PTCL-NOS).
Cancer Treat Res. 176:83–98. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pileri SA, Tabanelli V, Fiori S, Calleri
A, Melle F, Motta G, Lorenzini D, Tarella C and Derenzini E:
Peripheral T-cell lymphoma, not otherwise specified: Clinical
manifestations, diagnosis, and future treatment. Cancers (Basel).
13:45352021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sorial M, Aniagboso K, Buttermore E, Lei
M, Kendall E, Nasto K, Koh MJ, Han JX, Boussi L, Malpica L, et al:
Early time-to-relapse as a predictor of survival in mature
T-cell/NK-cell lymphomas: Results from the PETAL consortium. Blood.
146 (Suppl 1):S8892025. View Article : Google Scholar
|
|
10
|
Broccoli A and Zinzani PL: Peripheral
T-cell lymphoma, not otherwise specified. Blood. 129:1103–1112.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lee C and An Y: Deciphering the genetic
complexity of classical hodgkin lymphoma: Insights and effective
strategies. Curr Genomics. 25:334–342. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lyu G and Li D: PLEKHG7 expression: A
biomarker for prognosis and targeted therapy in diffuse large
B-cell lymphoma. Protein Pept Lett. 32:657–666. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Swerdlow SH, Campo E, Harris NL, Jaffe ES,
Pileri SA, Stein H, Thiele J and Vardiman JW: WHO classification of
tumours of haematopoietic and lymphoid tissues. International
agency for research on cancer Lyon; France: 2017
|
|
14
|
Carbone PP, Kaplan HS, Musshoff K,
Smithers DW and Tubiana M: Report of the committee on Hodgkin's
disease staging classification. Cancer Res. 31:1860–1861.
1971.PubMed/NCBI
|
|
15
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the eastern cooperative oncology group. Am J Clin
Oncol. 5:649–656. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
International Non-Hodgkin's Lymphoma
Prognostic Factors Project, . A predictive model for aggressive
non-Hodgkin's lymphoma. N Engl J Med. 329:987–994. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Horwitz SM, Ansell S, Ai WZ, Barnes J,
Barta SK, Brammer J, Clemens MW, Dogan A, Foss F, Ghione P, et al:
NCCN Clinical Practice Guidelines in Oncology. T-Cell Lymphomas,
Version 2.2022. J Natl Compr Canc Netw. 20:285–308. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang QY, Feng JF, Wang HQ, Huang HQ,
Zhang HL, Li XC, Gao YY, Song YP, Li ZM, et al: China Anti-Cancer
Association (CACA) guidelines for holistic integrative management
of lymphoma (version 2022). Holist Integ Oncol. 2:412023.
View Article : Google Scholar
|
|
19
|
Gallamini A, Stelitano C, Calvi R, Bellei
M, Mattei D, Vitolo U, Morabito F, Martelli M, Brusamolino E,
Iannitto E, et al: Peripheral T-cell lymphoma unspecified (PTCL-U):
A new prognostic model from a retrospective multicentric clinical
study. Blood. 103:2474–2479. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cheson BD, Fisher RI, Barrington SF,
Cavalli F, Schwartz LH, Zucca E, Lister TA; Alliance, Australasian
Leukaemia; Lymphoma Group and Eastern Cooperative Oncology Group, ;
et al: Recommendations for initial evaluation, staging, and
response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano
classification. J Clin Oncol. 32:3059–3068. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Common Terminology Criteria for Adverse
Events (CTCAE) Version 5.0. U.S. Department of Health and Human
Services; Washington, DC: 2017
|
|
22
|
Chen NC, Chang H, Kuo MC, Lin TL, Shih LY,
Chuang WY and Kao HW: Predictive model for treatment outcomes of
peripheral T-cell lymphoma, not otherwise specified, in Taiwanese
patients. J Formos Med Assoc. 123:188–197. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Khoury JD, Solary E, Abla O, Akkari Y,
Alaggio R, Apperley JF, Bejar R, Berti E, Busque L, Chan JKC, et
al: The 5th edition of the World Health Organization classification
of haematolymphoid tumours: Myeloid and histiocytic/dendritic
neoplasms. Leukemia. 36:1703–1719. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Davis O, Truong D, Day S, Pandey M,
Ibrahimi S, Khawandanah M, Holter-Chakrabarty J, Asch A and
Al-Juhaishi T: Impact of primary organ site of involvement by
peripheral T-cell lymphoma not otherwise specified on survival.
Cancer Med. 12:21770–21778. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nicolae A, Bouilly J, Lara D, Fataccioli
V, Lemonnier F, Drieux F, Parrens M, Robe C, Poullot E, Bisig B, et
al: Nodal cytotoxic peripheral T-cell lymphoma occurs frequently in
the clinical setting of immunodysregulation and is associated with
recurrent epigenetic alterations. Mod Pathol. 35:1126–1136. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Herek TA, Bouska A, Lone W, Sharma S,
Amador C, Heavican TB, Li Y, Wei Q, Jochum D, Greiner TC, et al:
DNMT3A mutations define a unique biological and prognostic subgroup
associated with cytotoxic T cells in PTCL-NOS. Blood.
140:1278–1290. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Iqbal J, Wright G, Wang C, Rosenwald A,
Gascoyne RD, Weisenburger DD, Greiner TC, Smith L, Guo S, Wilcox
RA, et al: Gene expression signatures delineate biological and
prognostic subgroups in peripheral T-cell lymphoma. Blood.
123:2915–2923. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Amador C, Greiner TC, Heavican TB, Smith
LM, Galvis KT, Lone W, Bouska A, D'Amore F, Pedersen MB, Pileri S,
et al: Reproducing the molecular subclassification of peripheral
T-cell lymphoma-NOS by immunohistochemistry. Blood. 134:2159–2170.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Asano N, Suzuki R, Kagami Y, Ishida F,
Kitamura K, Fukutani H, Morishima Y, Takeuchi K and Nakamura S:
Clinicopathologic and prognostic significance of cytotoxic molecule
expression in nodal peripheral T-cell lymphoma, unspecified. Am J
Surg Pathol. 29:1284–1293. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kato S, Takahashi E, Asano N, Tanaka T,
Megahed N, Kinoshita T and Nakamura S: Nodal cytotoxic molecule
(CM)-positive Epstein-Barr virus (EBV)-associated peripheral T cell
lymphoma (PTCL): A clinicopathological study of 26 cases.
Histopathology. 61:186–199. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Went P, Agostinelli C, Gallamini A,
Piccaluga PP, Ascani S, Sabattini E, Bacci F, Falini B, Motta T,
Paulli M, et al: Marker expression in peripheral T-cell lymphoma: A
proposed clinical-pathologic prognostic score. J Clin Oncol.
24:2472–2479. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gutiérrez-García G, García-Herrera A,
Cardesa T, Martínez A, Villamor N, Ghita G, Martínez-Trillos A,
Colomo L, Setoain X, Rodríguez S, et al: Comparison of four
prognostic scores in peripheral T-cell lymphoma. Ann Oncol.
22:397–404. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang H, Shi J, Xie H, Liu X, Ruan G, Lin
S, Ge Y, Liu C, Chen Y, Zheng X, et al: Superiority of
CRP-albumin-lymphocyte index as a prognostic biomarker for patients
with gastric cancer. Nutrition. 116:1121912023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu X, Meng QH, Ye Y, Hildebrandt MAT, Gu
J and Wu X: Prognostic significance of pretreatment serum levels of
albumin, LDH and total bilirubin in patients with non-metastatic
breast cancer. Carcinogenesis. 36:243–248. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Guo Y, Wei L, Patel SH, Lopez G, Grogan M,
Li M, Haddad T, Johns A, Ganesan LP, Yang Y, et al: Serum albumin:
Early prognostic marker of benefit for immune checkpoint inhibitor
monotherapy but not chemoimmunotherapy. Clin Lung Cancer.
23:345–355. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Federico M, Bellei M, Marcheselli L,
Schwartz M, Manni M, Tarantino V, Pileri S, Ko YH, Cabrera ME,
Horwitz S, et al: Peripheral T cell lymphoma, not otherwise
specified (PTCL-NOS). A new prognostic model developed by the
international T cell project network. Br J Haematol. 181:760–769.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kaito S, Kanemasa Y, Sasaki Y, Okuya T,
Yamaguchi T, Funasaka C, Shimoyama T, Omuro Y, Hishima T and Maeda
Y: A new prognostic score comprising lactate dehydrogenase, albumin
and neutrophil to lymphocyte ratio to predict sensitivity to
first-line chemotherapy in patients with peripheral T-cell
lymphomas. Int J Hematol. 107:451–459. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen H, Tao Y, Zhou Y, Liu P, Yang J, He
X, Zhou S, Qin Y, Song Y, Gui L, et al: The clinical features,
treatment, and prognostic factors for peripheral T-cell lymphomas:
A single-institution analysis of 240 Chinese patients. Asia Pac J
Clin Oncol. 19:e202–e214. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shen HR, Tang J, Li WY, Liang JH, Li Y, Wu
JZ, Wang L, Li JY, Gao R, Yin H and Xu W: 25-Hydroxy vitamin D
deficiency is an inferior predictor of peripheral T-cell lymphomas.
Ann Hematol. 103:565–574. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Raj SS, Fei T, Fried S, Ip A, Fein JA,
Leslie LA, Alarcon Tomas A, Leithner D, Peled JU, Corona M, et al:
An inflammatory biomarker signature of response to CAR-T cell
therapy in non-Hodgkin lymphoma. Nat Med. 31:1183–1194. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhu F, Wang KB and Rui L: STAT3 activation
and oncogenesis in lymphoma. Cancers (Basel). 12:192019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Jaeger A, Gambheer SMM, Sun X, Chernyakov
D, Skorobohatko O, Mack T, Kissel S, Pfeifer D, Zeiser R, Fisch P,
et al: Activated granulocytes and inflammatory cytokine signaling
drive T-cell lymphoma progression and disease symptoms. Blood.
141:2824–2840. 2023.PubMed/NCBI
|
|
43
|
Vose J, Armitage J and Weisenburger D;
International T-Cell Lymphoma Project, : International peripheral
T-cell and natural killer/T-cell lymphoma study: Pathology findings
and clinical outcomes. J Clin Oncol. 26:4124–4130. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ellin F, Landström J, Jerkeman M and
Relander T: Real-world data on prognostic factors and treatment in
peripheral T-cell lymphomas: A study from the Swedish lymphoma
registry. Blood. 124:1570–1577. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wang J, Su N, Fang Y, Ma S, Zhang Y, Cai
J, Zou Q, Tian X, Xia Y, Liu P, et al: Comparison of chemotherapy
combined with chidamide versus chemotherapy in the frontline
treatment for peripheral T-cell lymphoma. Front Immunol.
13:8351032022. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Horwitz S, O'Connor OA, Pro B, Illidge T,
Fanale M, Advani R, Bartlett NL, Christensen JH, Morschhauser F,
Domingo-Domenech E, et al: Brentuximab vedotin with chemotherapy
for CD30-positive peripheral T-cell lymphoma (ECHELON-2): A global,
double-blind, randomised, phase 3 trial. Lancet. 393:229–240. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sureda A, García-Sanz R, Domingo-Domenech
E, Capote FJ, Gutiérrez A, Rodriguez A, Aguiar D, Giraldo P,
Infante MS, López-Jiménez J, et al: Retreatment with brentuximab
vedotin in patients with relapsed/refractory CD30+ malignancies: A
retrospective medical chart review study in Spain-The BELIEVE
study. Cancers (Basel). 17:11372025. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Brink M, Meeuwes FO, van der Poel MWM,
Kersten MJ, Wondergem M, Mutsaers PGNJ, Böhmer LH, Woei-A-Jin FSH,
Visser O, Oostvogels R, et al: Impact of etoposide and ASCT on
survival among patients aged <65 years with stage II to IV PTCL:
A population-based cohort study. Blood. 140:1009–1019. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Park SI, Horwitz SM, Foss FM, Pinter-Brown
LC, Carson KR, Rosen ST, Pro B, Hsi ED, Federico M, Gisselbrecht C,
et al: The role of autologous stem cell transplantation in patients
with nodal peripheral T-cell lymphomas in first complete remission:
Report from COMPLETE, a prospective, multicenter cohort study.
Cancer. 125:1507–1517. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Meeuwes FO, Serroukh YIM, van der Poel
MWM, Plattel WJ and Nijland M: Relapsed and refractory peripheral
T-cell lymphoma; treatment, challenges and future perspectives.
Leuk Lymphoma. 66:2341–2356. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Qi F, Dong M, He X, Li Y, Wang W, Liu P,
Yang J, Gui L, Zhang C, Yang S, et al: Gemcitabine, dexamethasone,
and cisplatin (GDP) as salvage chemotherapy for patients with
relapsed or refractory peripheral T cell lymphoma-not otherwise
specified. Ann Hematol. 96:245–251. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mikesch JH, Kuhlmann M, Demant A, Krug U,
Thoennissen GB, Schmidt E, Kessler T, Schliemann C, Pohlen M, Mohr
M, et al: DexaBEAM versus ICE salvage regimen prior to autologous
transplantation for relapsed or refractory aggressive peripheral T
cell lymphoma: A retrospective evaluation of parallel patient
cohorts of one center. Ann Hematol. 92:1041–1048. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Bachy E, Camus V, Thieblemont C, Sibon D,
Casasnovas RO, Ysebaert L, Damaj G, Guidez S, Pica GM, et al:
Romidepsin Plus CHOP Versus CHOP in Patients With Previously
Untreated Peripheral T-Cell Lymphoma: Results of the Ro-CHOP Phase
III Study (Conducted by LYSA). J Clin Oncol. 40:242–251. 2022.
View Article : Google Scholar : PubMed/NCBI
|