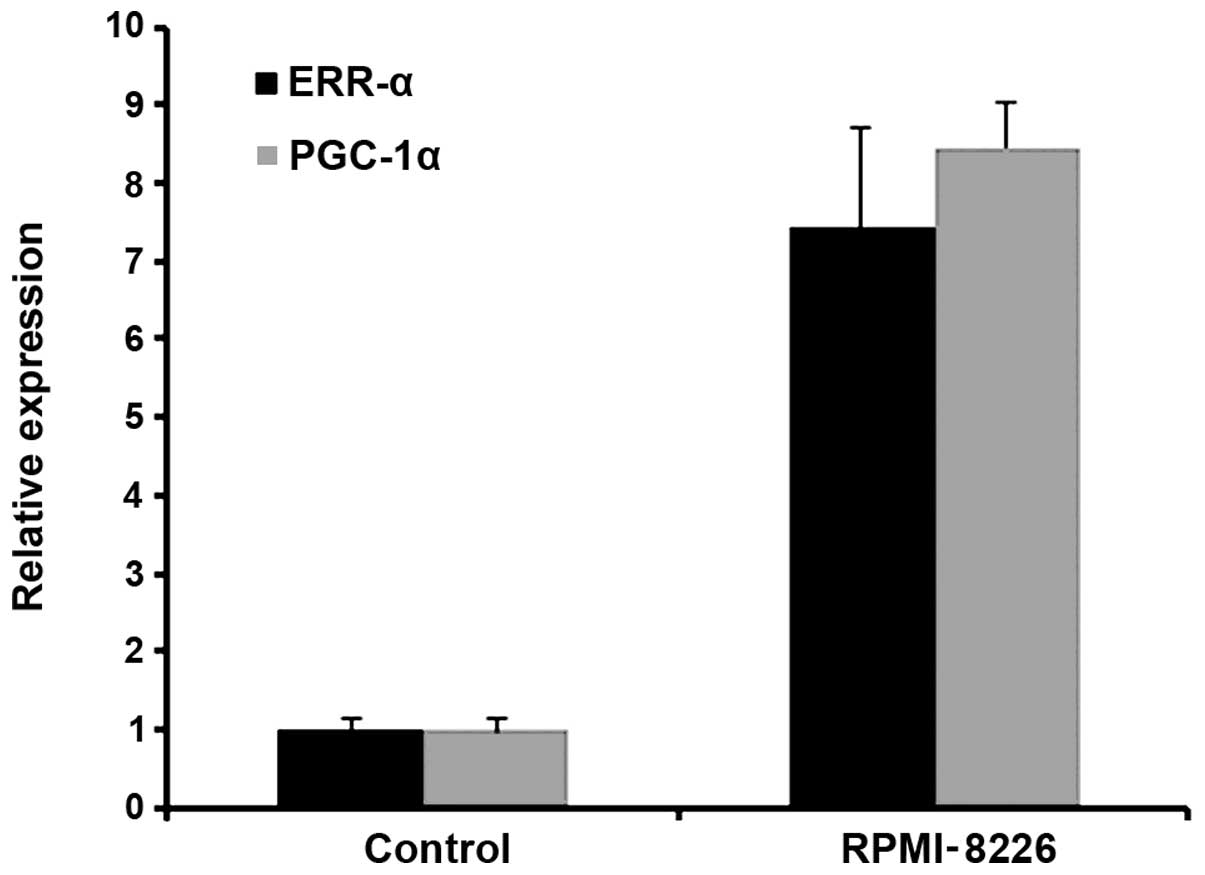
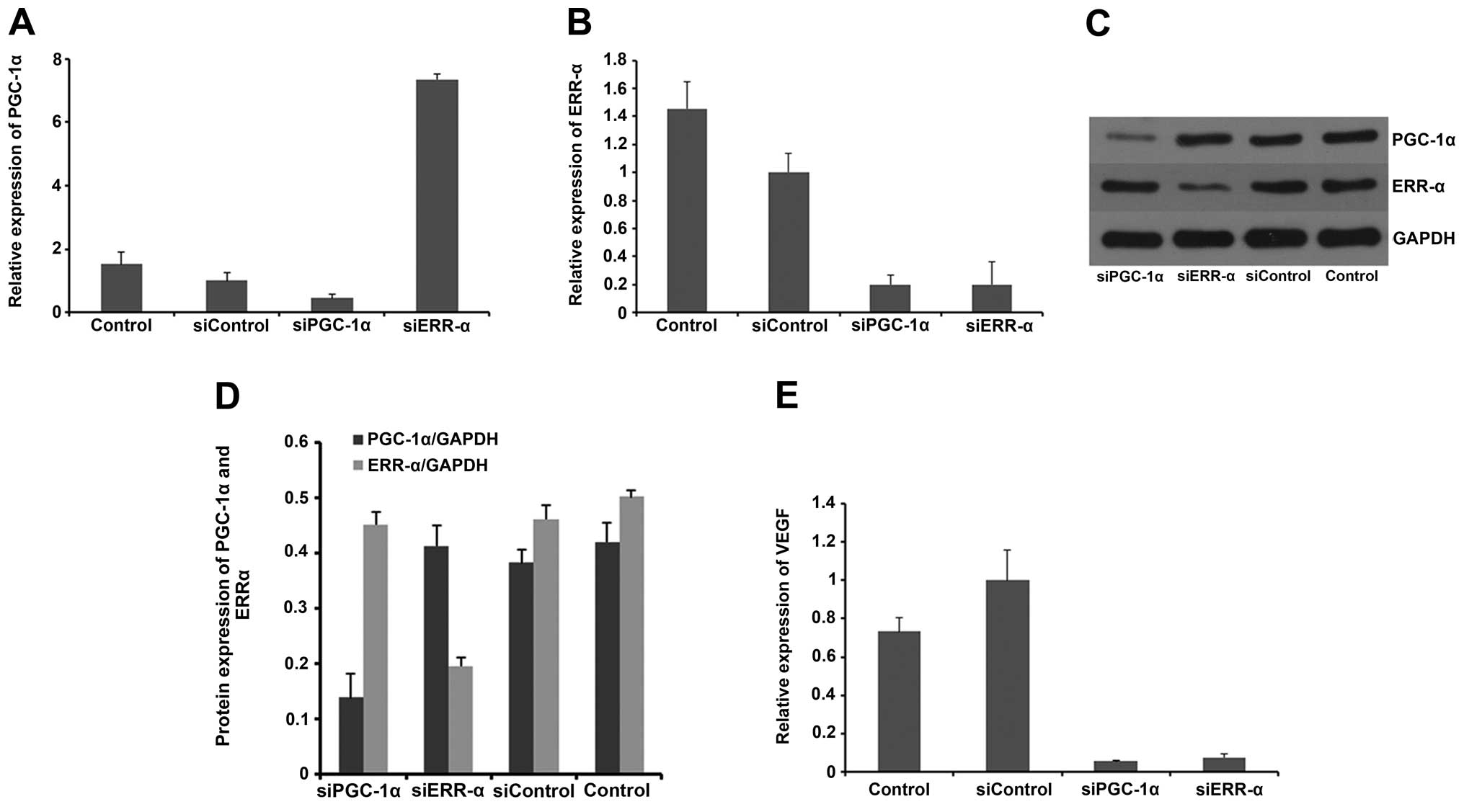
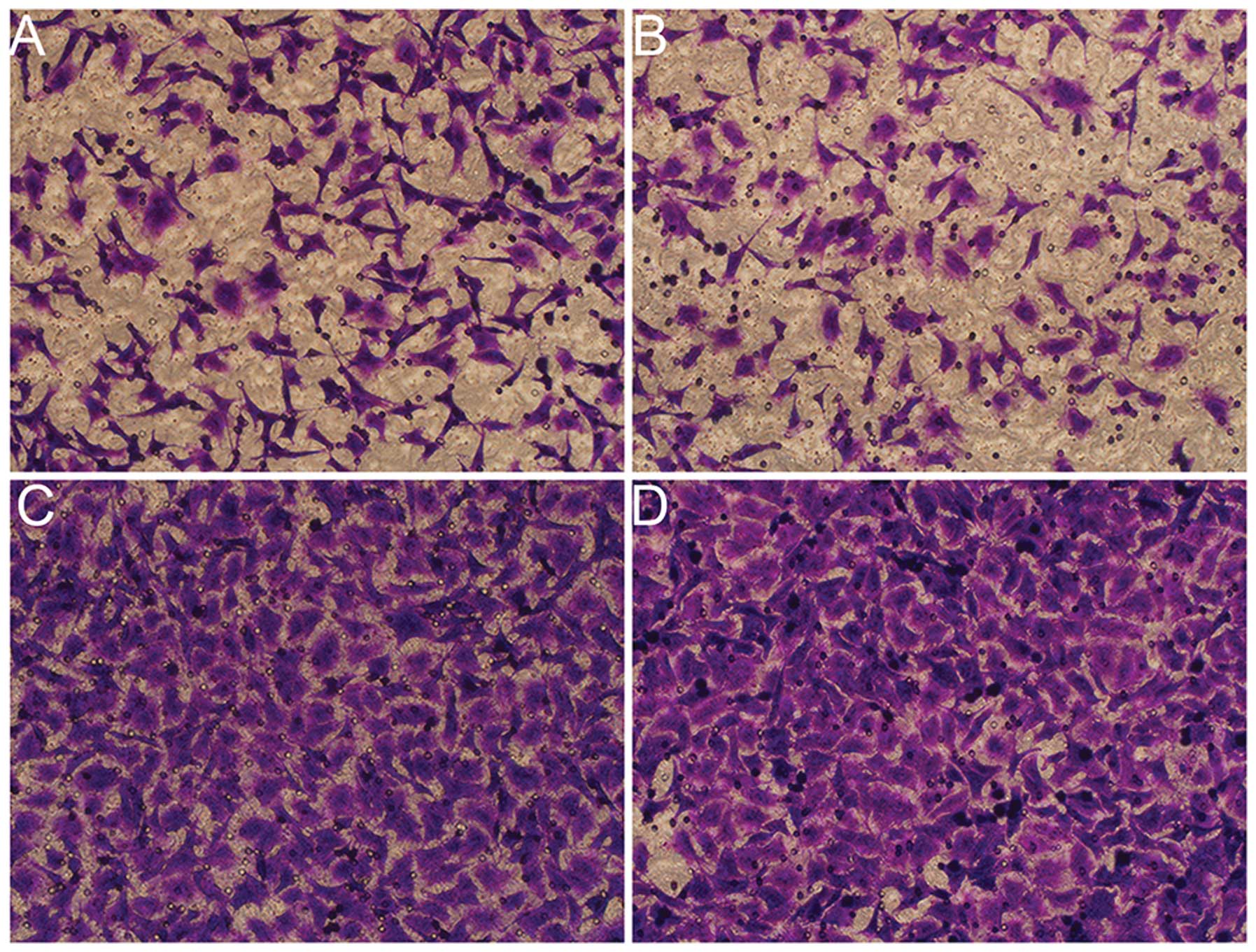
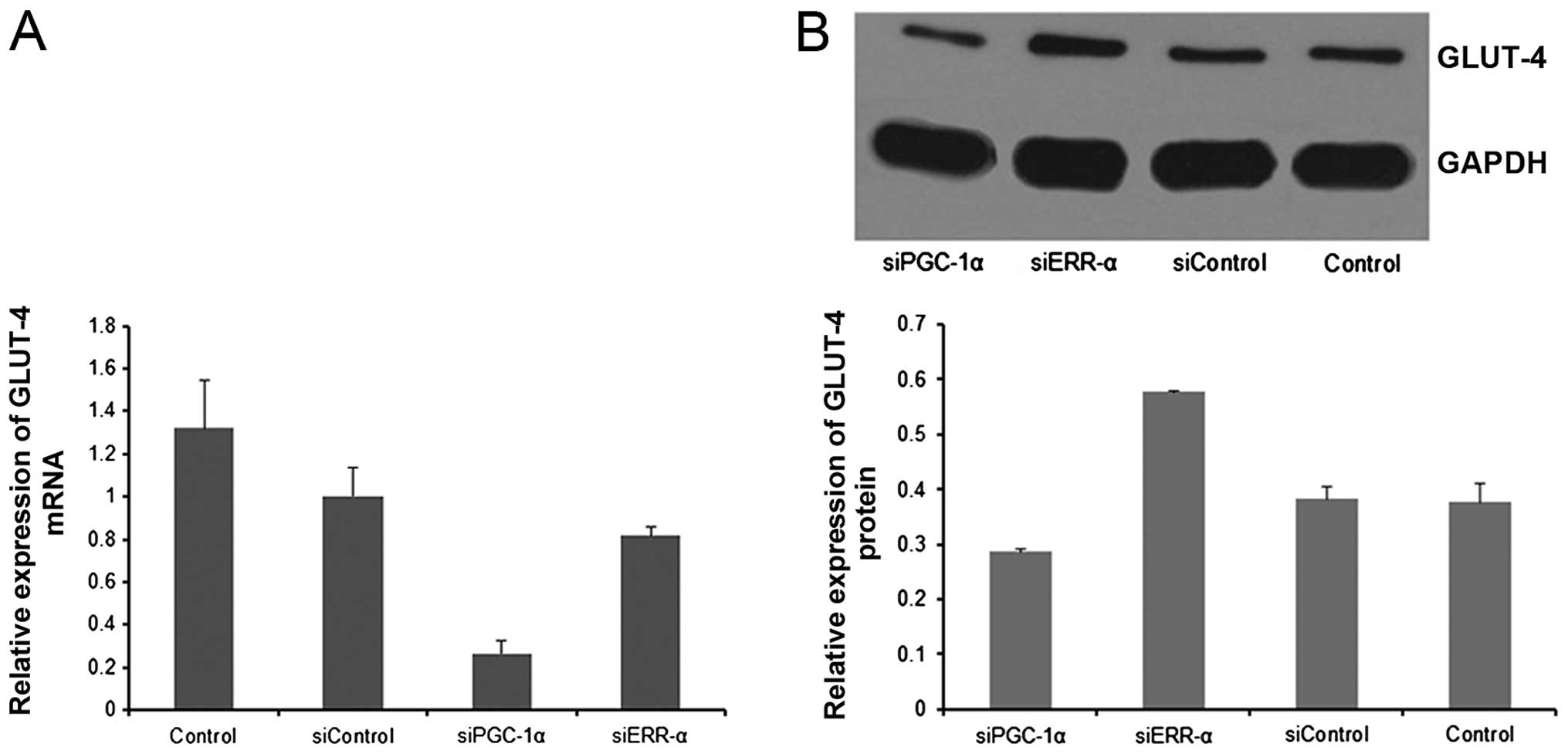
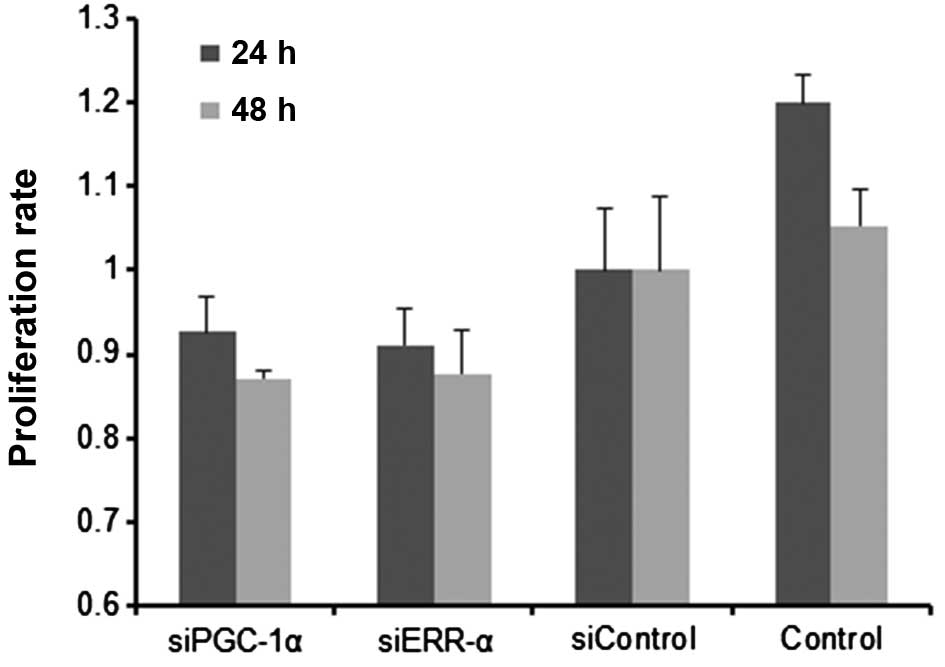
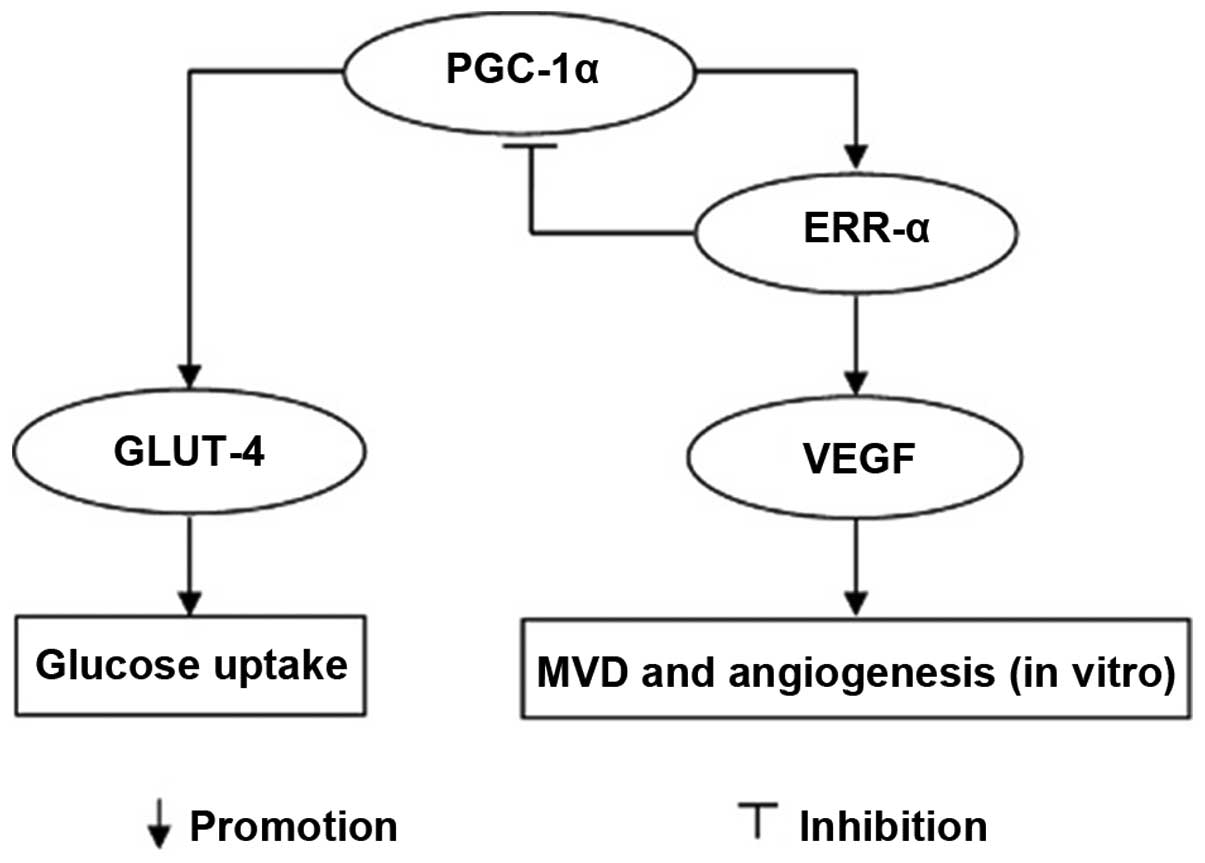
|
1
|
Munshi NC: Plasma cell disorders: an
historical perspective. Hematology Am Soc Hematol Educ Program.
297:2008.PubMed/NCBI
|
|
2
|
Ria R, Reale A, De Luisi A, Ferrucci A,
Moschetta M and Vacca A: Bone marrow angiogenesis and progression
in multiple myeloma. Am J Blood Res. 1:76–89. 2011.PubMed/NCBI
|
|
3
|
McBrayer SK, Cheng JC, Singhal S, Krett
NL, Rosen ST and Shanmugam M: Multiple myeloma exhibits novel
dependence on GLUT4, GLUT8, and GLUT11: implications for glucose
transporter-directed therapy. Blood. 119:4686–4697. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stocks T, Rapp K, Bjorge T, et al: Blood
glucose and risk of incident and fatal cancer in the metabolic
syndrome and cancer project (me-can): analysis of six prospective
cohorts. PLoS Med. 6:e10002012009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vacca A, Ribatti D, Roccaro AM, Frigeri A
and Dammacco F: Bone marrow angiogenesis in patients with active
multiple myeloma. Semin Oncol. 28:543–550. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Giatromanolaki A, Bai M, Margaritis D, et
al: Hypoxia and activated VEGF/receptor pathway in multiple
myeloma. Anticancer Res. 30:2831–2836. 2010.PubMed/NCBI
|
|
7
|
Sun CY, Hu Y, Huang J, et al:
Brain-derived neurotrophic factor induces proliferation, migration,
and VEGF secretion in human multiple myeloma cells via activation
of MEK-ERK and PI3K/AKT signaling. Tumour Biol. 31:121–128. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Puigserver P, Wu Z, Park CW, Graves R,
Wright M and Spiegelman BM: A cold-inducible coactivator of nuclear
receptors linked to adaptive thermogenesis. Cell. 92:829–839. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Handschin C and Spiegelman BM: Peroxisome
proliferator-activated receptor γ coactivator 1 coactivators,
energy homeostasis, and metabolism. Endocr Rev. 27:728–735.
2006.
|
|
10
|
Puigserver P and Spiegelman BM: Peroxisome
proliferator-activated receptor-γ coactivator 1 α (PGC-1 α):
transcriptional coactivator and metabolic regulator. Endocr Rev.
24:78–90. 2003.
|
|
11
|
Finck BN and Kelly DP: PGC-1 coactivators:
inducible regulators of energy metabolism in health and disease. J
Clin Invest. 116:615–622. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Arany Z, Foo SY, Ma Y, et al:
HIF-independent regulation of VEGF and angiogenesis by the
transcriptional coactivator PGC-1α. Nature. 451:1008–1012.
2008.PubMed/NCBI
|
|
13
|
Lu J, Zhang K, Chen S and Wen W: Grape
seed extract inhibits VEGF expression via reducing HIF-1α protein
expression. Carcinogenesis. 30:636–644. 2009.PubMed/NCBI
|
|
14
|
Otjacques E, Binsfeld M, Noel A, Beguin Y,
Cataldo D and Caers J: Biological aspects of angiogenesis in
multiple myeloma. Int J Hematol. 94:505–518. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nikhil C and Kenneth C: Advances in
Biology and Therapy of Multiple Myeloma. 1. Basic Science.
Springer; New York, NY: 2012
|
|
16
|
Adekola K, Rosen ST and Shanmugam M:
Glucose transporters in cancer metabolism. Curr Opin Oncol.
24:650–654. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ak I and Gulbas Z: F-18 FDG uptake of bone
marrow on PET/CT scan: its correlation with CD38/CD138 expressing
myeloma cells in bone marrow of patients with multiple myeloma. Ann
Hematol. 90:81–87. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bartel TB, Haessler J, Brown TL, et al:
F18-fluorodeoxyglucose positron emission tomography in the context
of other imaging techniques and prognostic factors in multiple
myeloma. Blood. 114:2068–2076. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kaira K, Endo M, Abe M, et al: Biologic
correlation of 2-[18F]-fluoro-2-deoxy-D-glucose uptake
on positron emission tomography in thymic epithelial tumors. J Clin
Oncol. 28:3746–3753. 2010.
|
|
20
|
Podar K and Anderson KC: Emerging
therapies targeting tumor vasculature in multiple myeloma and other
hematologic and solid malignancies. Curr Cancer Drug Targets.
11:1005–1024. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Klimcakova E, Chenard V, McGuirk S, et al:
PGC-1α promotes the growth of ErbB2/Neu-induced mammary tumors by
regulating nutrient supply. Cancer Res. 72:1538–1546. 2012.
|
|
22
|
Altenberg B and Greulich KO: Genes of
glycolysis are ubiquitously overexpressed in 24 cancer classes.
Genomics. 84:1014–1020. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Durie BG, Waxman AD, D’Agnolo A and
Williams CM: Whole-body 18F-FDG PET identifies high-risk
myeloma. J Nucl Med. 43:1457–1463. 2002.PubMed/NCBI
|
|
24
|
Zhang K, Lu J, Mori T, et al: Baicalin
increases VEGF expression and angiogenesis by activating the
ERRα/PGC-1α pathway. Cardiovasc Res. 89:426–435. 2011.PubMed/NCBI
|
|
25
|
Bredella MA, Steinbach L, Caputo G, Segall
G and Hawkins R: Value of FDG PET in the assessment of patients
with multiple myeloma. AJR Am J Roentgenol. 184:1199–1204. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gendron MC, Schrantz N, Metivier D, et al:
Oxidation of pyridine nucleotides during Fas- and ceramide-induced
apoptosis in Jurkat cells: correlation with changes in
mitochondria, glutathione depletion, intracellular acidification
and caspase 3 activation. Biochem J. 353:357–367. 2001. View Article : Google Scholar
|
|
27
|
Mathupala SP, Ko YH and Pedersen PL:
Hexokinase II: cancer’s double-edged sword acting as both
facilitator and gatekeeper of malignancy when bound to
mitochondria. Oncogene. 25:4777–4786. 2006.
|
|
28
|
Gottlob K, Majewski N, Kennedy S, Kandel
E, Robey RB and Hay N: Inhibition of early apoptotic events by
Akt/PKB is dependent on the first committed step of glycolysis and
mitochondrial hexokinase. Genes Dev. 15:1406–1418. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
DeBerardinis RJ, Mancuso A, Daikhin E, et
al: Beyond aerobic glycolysis: transformed cells can engage in
glutamine metabolism that exceeds the requirement for protein and
nucleotide synthesis. Proc Natl Acad Sci USA. 104:19345–19350.
2007. View Article : Google Scholar
|
|
30
|
Zhao Y, Altman BJ, Coloff JL, et al:
Glycogen synthase kinase 3α and 3β mediate a glucose-sensitive
antiapoptotic signaling pathway to stabilize Mcl-1. Mol Cell Biol.
27:4328–4339. 2007.
|
|
31
|
Danial NN, Gramm CF, Scorrano L, et al:
BAD and glucokinase reside in a mitochondrial complex that
integrates glycolysis and apoptosis. Nature. 424:952–956. 2003.
View Article : Google Scholar
|
|
32
|
Rathmell JC, Fox CJ, Plas DR, Hammerman
PS, Cinalli RM and Thompson CB: Akt-directed glucose metabolism can
prevent Bax conformation change and promote growth
factor-independent survival. Mol Cell Biol. 23:7315–7328. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gatenby RA and Gillies RJ: Why do cancers
have high aerobic glycolysis? Nat Rev Cancer. 4:891–899. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Plas DR and Thompson CB: Cell metabolism
in the regulation of programmed cell death. Trends Endocrinol
Metab. 13:75–78. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chang CY and McDonnell DP: Molecular
pathways: the metabolic regulator estrogen-related receptor α as a
therapeutic target in cancer. Clin Cancer Res. 18:6089–6095.
2012.
|