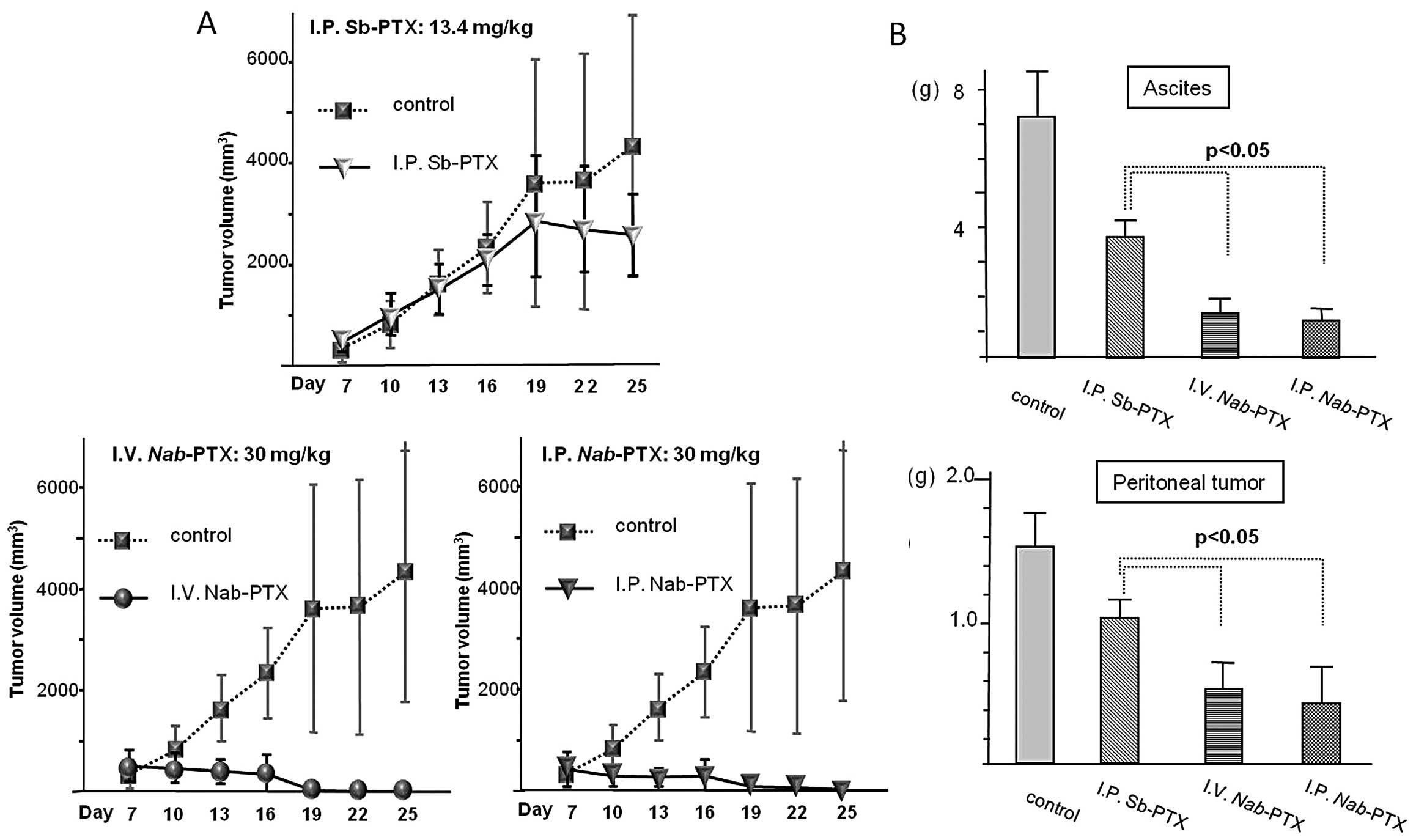
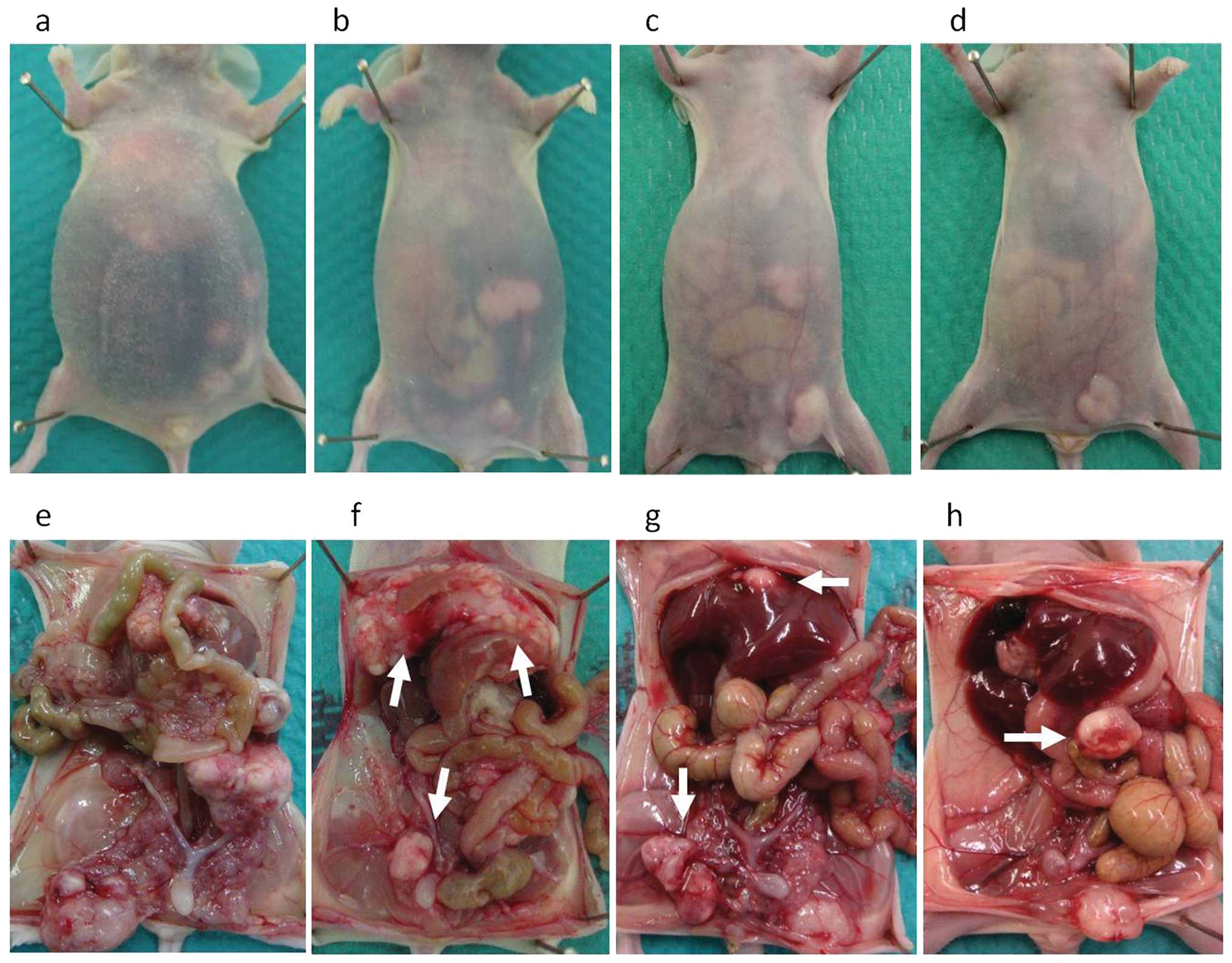
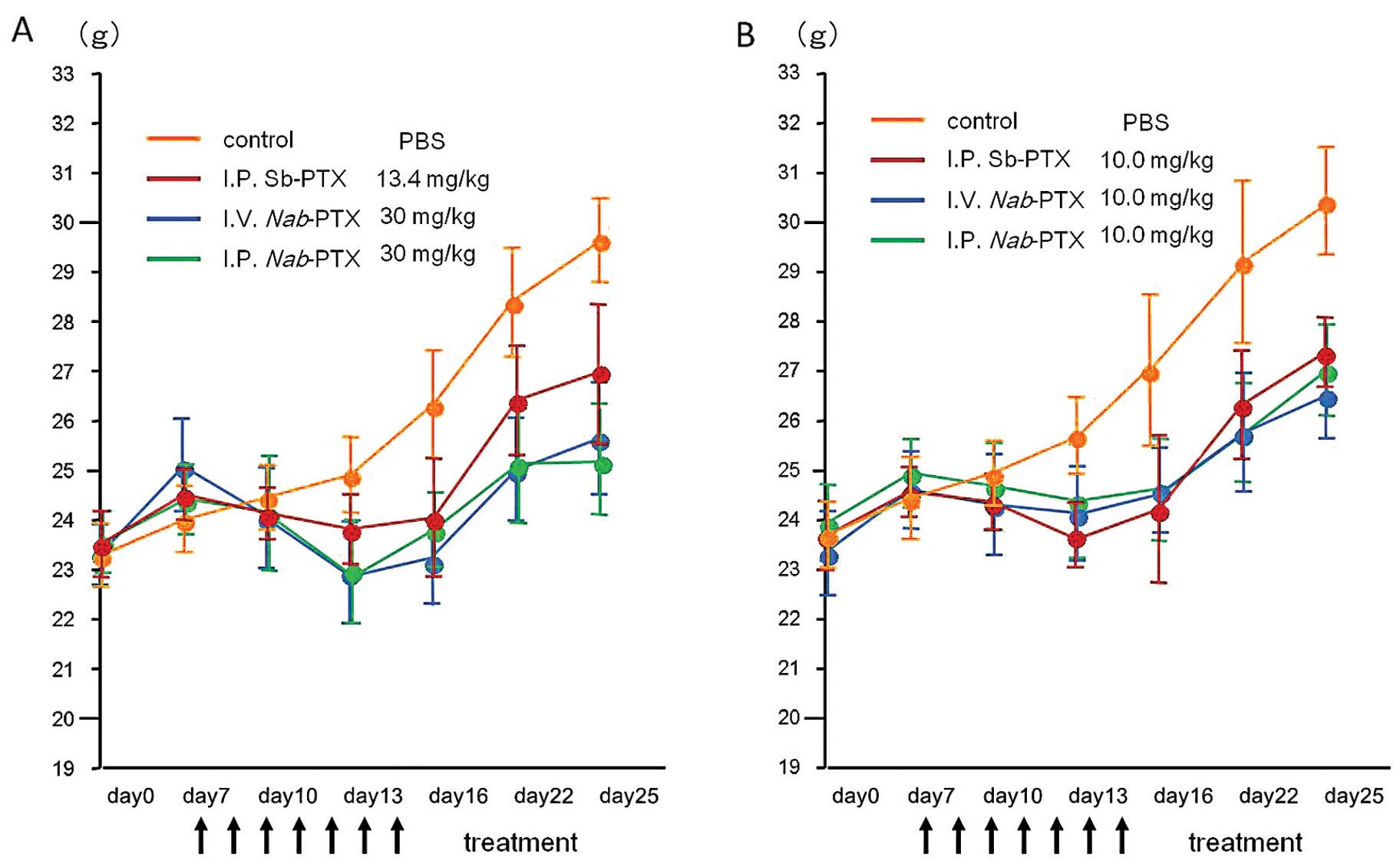
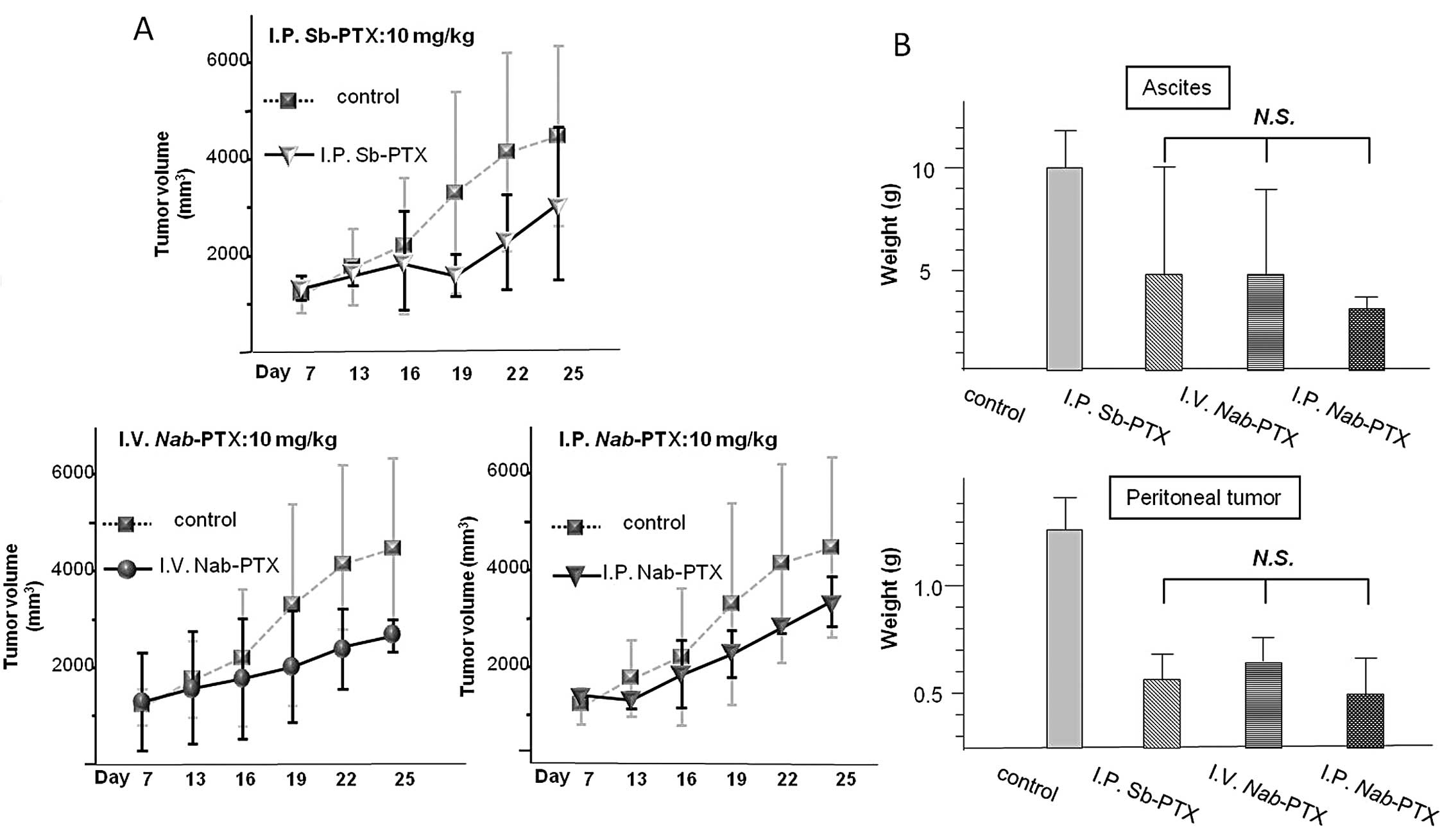
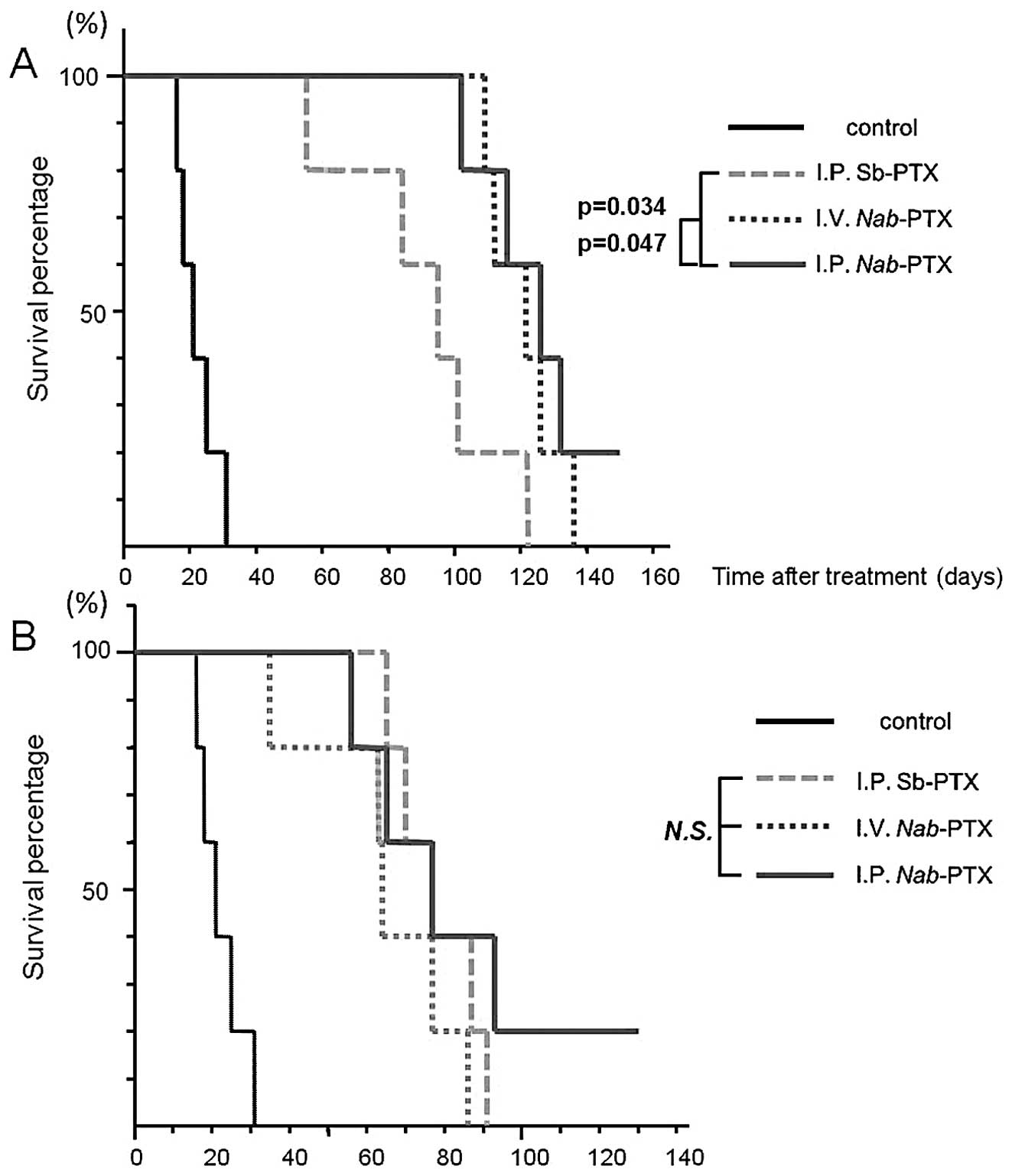
|
1
|
Siewert JR, Bottcher K, Roder JD, Busch R,
Hermanek P and Meyer HJ: Prognostic relevance of systematic lymph
node dissection in gastric carcinoma. German Gastric Carcinoma
Study Group. Br J Surg. 80:1015–1018. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Allum WH, Powell DJ, McConkey CC and
Fielding JW: Gastric cancer: a 25-year review. Br J Surg.
76:535–540. 1989.PubMed/NCBI
|
|
3
|
Rowinsky EK, Cazenave LA and Donehower RC:
Taxol: a novel investigational antimicrotubule agent. J Natl Cancer
Inst. 82:1247–1259. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Carney DN: Chemotherapy in the management
of patients with inoperable non-small cell lung cancer. Semin
Oncol. 23:71–75. 1996.PubMed/NCBI
|
|
5
|
Crown J and O’Leary M: The taxanes: an
update. Lancet. 355:1176–1178. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gelmon K: The taxoids: paclitaxel and
docetaxel. Lancet. 344:1267–1272. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ohtsu A, Boku N, Tamura F, et al: An early
phase II study of a 3-hour infusion of paclitaxel for advanced
gastric cancer. Am J Clin Oncol. 21:416–419. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yamada Y, Shirao K, Ohtsu A, et al: Phase
II trial of paclitaxel by three-hour infusion for advanced gastric
cancer with short premedication for prophylaxis against
paclitaxel-associated hypersensitivity reactions. Ann Oncol.
12:1133–1137. 2001. View Article : Google Scholar
|
|
9
|
Hironaka S, Zenda S, Boku N, et al: Weekly
paclitaxel as second-line chemotherapy for advanced or recurrent
gastric cancer. Gastric Cancer. 9:14–18. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Francis P, Rowinsky E, Schneider J, Hakes
T, Hoskins W and Markman M: Phase I feasibility and pharmacologic
study of weekly intraperitoneal paclitaxel: a Gynecologic Oncology
Group pilot study. J Clin Oncol. 13:2961–2967. 1995.PubMed/NCBI
|
|
11
|
Markman M, Brady MF, Spirtos NM, Hanjani P
and Rubin SC: Phase II trial of intraperitoneal paclitaxel in
carcinoma of the ovary, tube, and peritoneum: a Gynecologic
Oncology Group Study. J Clin Oncol. 16:2620–2624. 1998.PubMed/NCBI
|
|
12
|
Armstrong DK, Bundy B, Wenzel L, Huang HQ,
Baergen R, Lele S, Copeland LJ, Walker JL and Burger RA;
Gynecologic Oncology Group. Intraperitoneal cisplatin and
paclitaxel in ovarian cancer. N Engl J Med. 354:34–43. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ishigami H, Kitayama J, Kaisaki S, et al:
Phase II study of weekly intravenous and intraperitoneal paclitaxel
combined with S-1 for advanced gastric cancer with peritoneal
metastasis. Ann Oncol. 21:67–70. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fushida S, Kinoshita J, Kaji M, Hirono Y,
Goda F, Yagi Y, Oyama K, Sudo Y, Watanabe Y and Fujimura T; Society
for the Study of Peritoneal Carcinomatosis in Gastric Cancer. Phase
I/II study of intraperitoneal docetaxel plus S-1 for the gastric
cancer patients with peritoneal carcinomatosis. Cancer Chemother
Pharmacol. 71:1265–1272. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fujiwara Y, Takiguchi S, Nakajima K, et
al: Intraperitoneal docetaxel combined with S-1 for advanced
gastric cancer with peritoneal dissemination. J Surg Oncol.
105:38–42. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Imano M, Yasuda A, Itoh T, et al: Phase II
study of single intraperitoneal chemotherapy followed by systemic
chemotherapy for gastric cancer with peritoneal metastasis. J
Gastrointest Surg. 16:2190–2196. 2012. View Article : Google Scholar
|
|
17
|
Weiss RB, Donehower RC, Wiernik PH, et al:
Hypersensitivity reactions from Taxol. J Clin Oncol. 8:1263–1268.
1990.PubMed/NCBI
|
|
18
|
Kloover JS, den Bakker MA, Gelderblom H
and van Meerbeeck JP: Fatal outcome of a hypersensitivity reaction
to paclitaxel: a critical review of premedication regimens. Br J
Cancer. 90:304–305. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
ten Tije AJ, Verweij J, Loos WJ and
Sparreboom A: Pharmacological effects of formulation vehicles:
implications for cancer chemotherapy. Clin Pharmacokinet.
42:665–685. 2003.PubMed/NCBI
|
|
20
|
Sparreboom A, van Zuylen L, Brouwer E, et
al: Cremophor EL- mediated alteration of paclitaxel distribution in
human blood: clinical pharmacokinetic implications. Cancer Res.
59:1454–1457. 1999.
|
|
21
|
Gradishar WJ, Tjulandin S, Davidson N, et
al: Phase III trial of nanoparticle albumin-bound paclitaxel
compared with polyethylated castor oil-based paclitaxel in women
with breast cancer. J Clin Oncol. 23:7794–7803. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Simionescu M, Gafencu A and Antohe F:
Transcytosis of plasma macromolecules in endothelial cells: a cell
biological survey. Microsc Res Tech. 57:269–288. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
John TA, Vogel SM, Tiruppathi C, Malik AB
and Minshall RD: Quantitative analysis of albumin uptake and
transport in the rat microvessel endothelial monolayer. Am J
Physiol Lung Cell Mol Physiol. 284:L187–L196. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Desai N, Trieu V, Damascelli B and
Soon-Shiong P: SPARC expression correlates with tumor response to
albumin-bound paclitaxel in head and neck cancer patients. Transl
Oncol. 2:59–64. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Von Hoff DD, Ramanathan RK, Borad MJ, et
al: Gemcitabine plus nab-paclitaxel is an active regimen in
patients with advanced pancreatic cancer: a phase I/II trial. J
Clin Oncol. 29:4548–4554. 2011.
|
|
26
|
Socinski MA, Bondarenko I, Karaseva NA, et
al: Weekly nab-paclitaxel in combination with carboplatin versus
solvent-based paclitaxel plus carboplatin as first-line therapy in
patients with advanced non-small-cell lung cancer: final results of
a phase III trial. J Clin Oncol. 30:2055–2062. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Takiuchi H, Sasaki Y, Nishina T, et al:
ABI-007 in the treatment of unresectable or recurrent gastric
cancer refractory to fluoropyrimidine-containing regimen: Updated
data from the multicenter phase II study. J Clin Oncol (Meeting
Abstracts). 30(Suppl 4): 902012.
|
|
28
|
Yashiro M, Chung YS, Nishimura S, Inoue T
and Sowa M: Peritoneal metastatic model for human scirrhous gastric
carcinoma in nude mice. Clin Exp Metastasis. 14:43–54. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Desai N, Trieu V, Yao Z, et al: Increased
antitumor activity, intratumor paclitaxel concentrations, and
endothelial cell transport of cremophor-free, albumin-bound
paclitaxel, ABI-007, compared with cremophor-based paclitaxel. Clin
Cancer Res. 12:1317–1324. 2006. View Article : Google Scholar
|
|
30
|
Brouwer E, Verweij J, De Bruijn P, et al:
Measurement of fraction unbound paclitaxel in human plasma. Drug
Metab Dispos. 28:1141–1145. 2000.PubMed/NCBI
|
|
31
|
Kumar GN, Walle UK, Bhalla KN and Walle T:
Binding of taxol to human plasma, albumin and alpha 1-acid
glycoprotein. Res Commun Chem Pathol Pharmacol. 80:337–344.
1993.PubMed/NCBI
|
|
32
|
Gardner ER, Dahut WL, Scripture CD, et al:
Randomized crossover pharmacokinetic study of solvent-based
paclitaxel and nab-paclitaxel. Clin Cancer Res.
14:4200–4205. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang CS, Lin KH, Chen SL, Chan YF and
Hsueh S: Overexpression of SPARC gene in human gastric carcinoma
and its clinic-pathologic significance. Br J Cancer. 91:1924–1930.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhao ZS, Wang YY, Chu YQ, Ye ZY and Tao
HQ: SPARC is associated with gastric cancer progression and poor
survival of patients. Clin Cancer Res. 16:260–268. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Said N and Motamed K: Absence of
host-secreted protein acidic and rich in cysteine (SPARC) augments
peritoneal ovarian carcinomatosis. Am J Pathol. 167:1739–1752.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kamijo Y, Ito C, Sai Y and Miyamoto K:
Surfactants influence the distribution of taxanes in peritoneal
dissemination tumor-bearing rats. Cancer Lett. 287:182–186. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Matsumura Y and Maeda H: A new concept for
macromolecular therapeutics in cancer chemotherapy: mechanism of
tumoritropic accumulation of proteins and the antitumor agent
smancs. Cancer Res. 46:6387–6392. 1986.
|
|
38
|
Vicent MJ and Duncan R: Polymer
conjugates: nanosized medicines for treating cancer (Review).
Trends Biotechnol. 24:39–47. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Duncan R: Polymer conjugates as anticancer
nanomedicines (Review). Nat Rev Cancer. 6:688–701. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Emoto S, Yamaguchi H, Kishikawa J,
Yamashita H, Ishigami H and Kitayama J: Antitumor effect and
pharmacokinetics of intraperitoneal NK105, a nanomicellar
paclitaxel formulation for peritoneal dissemination. Cancer Sci.
103:1304–1310. 2012. View Article : Google Scholar : PubMed/NCBI
|