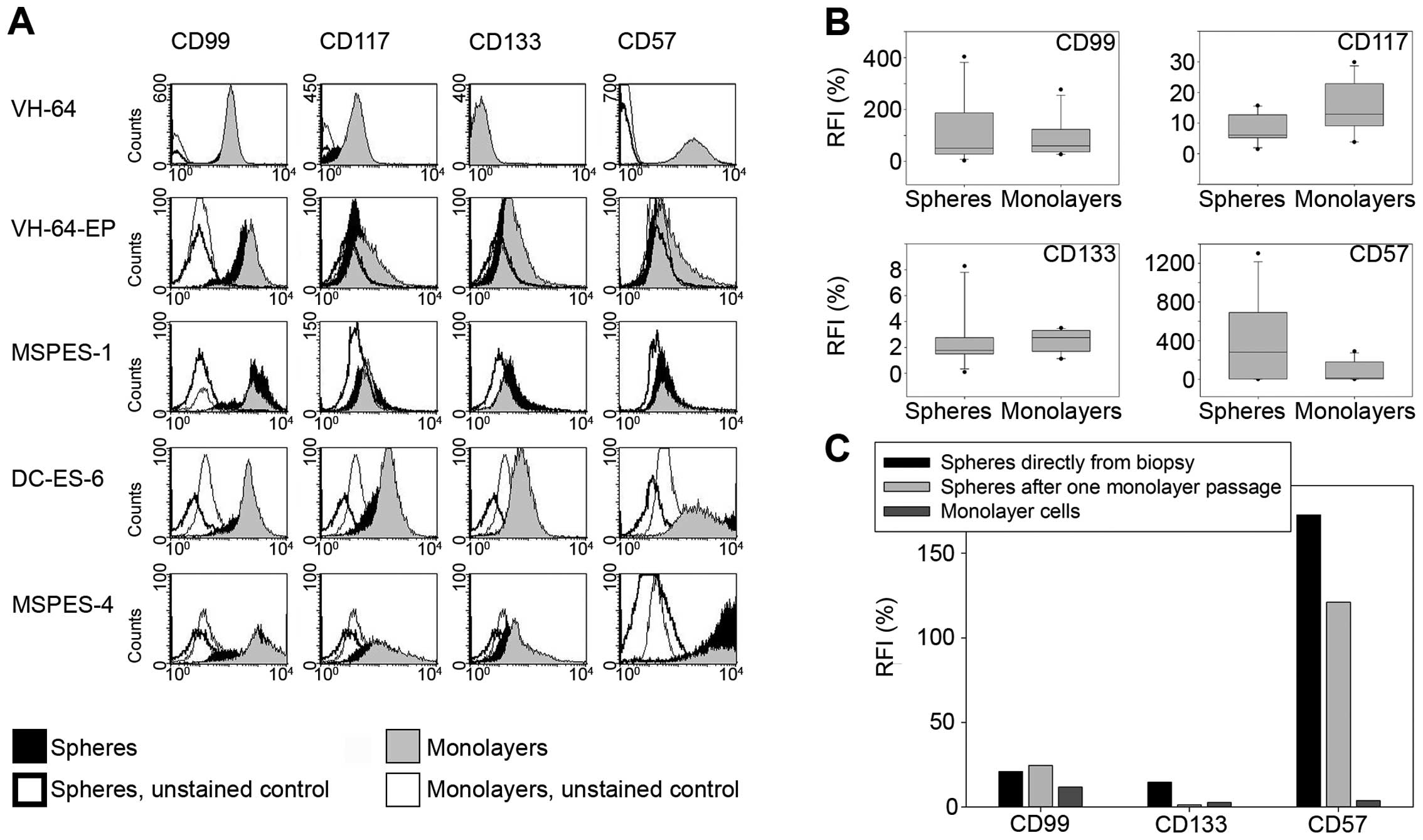
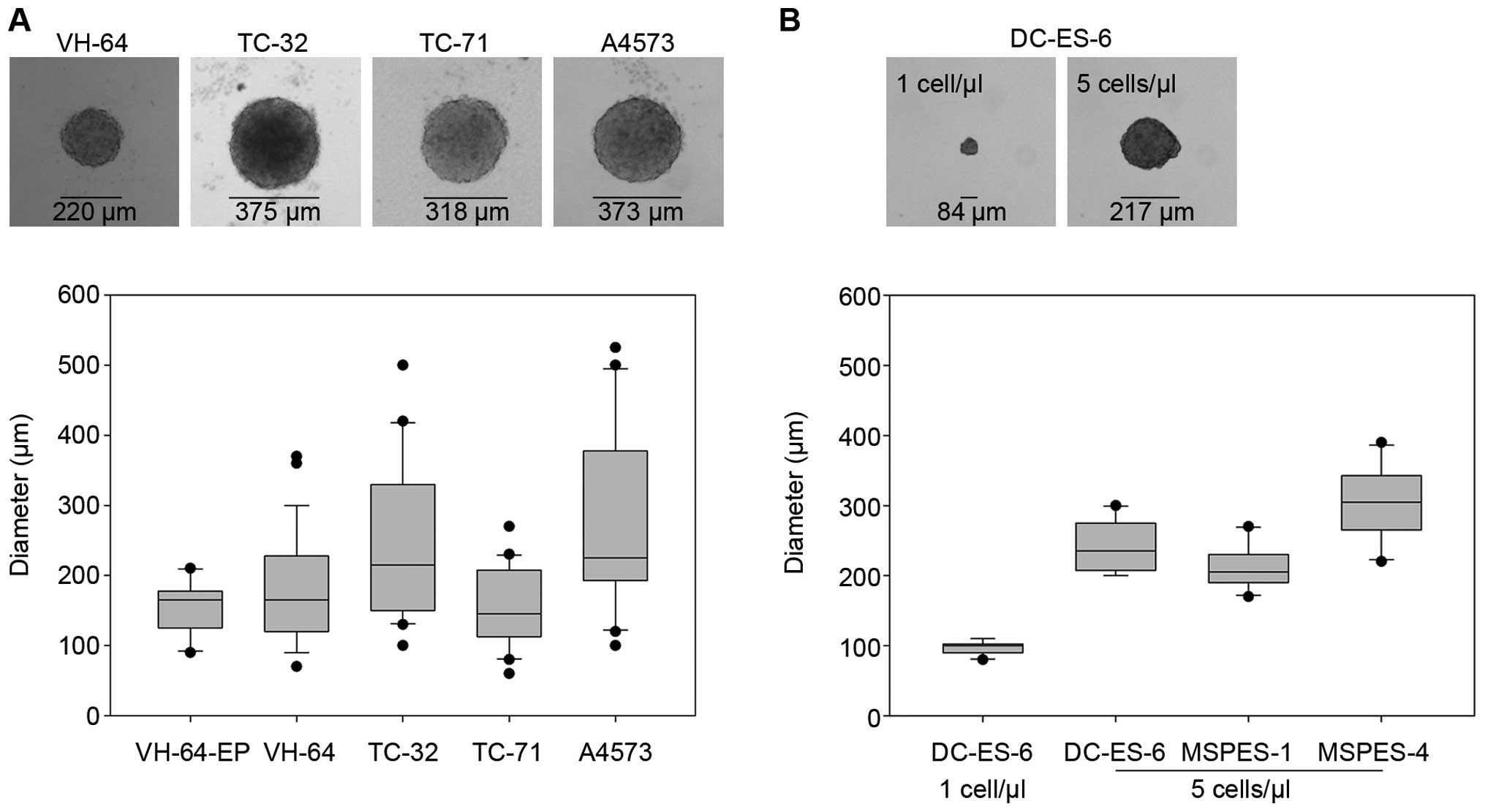
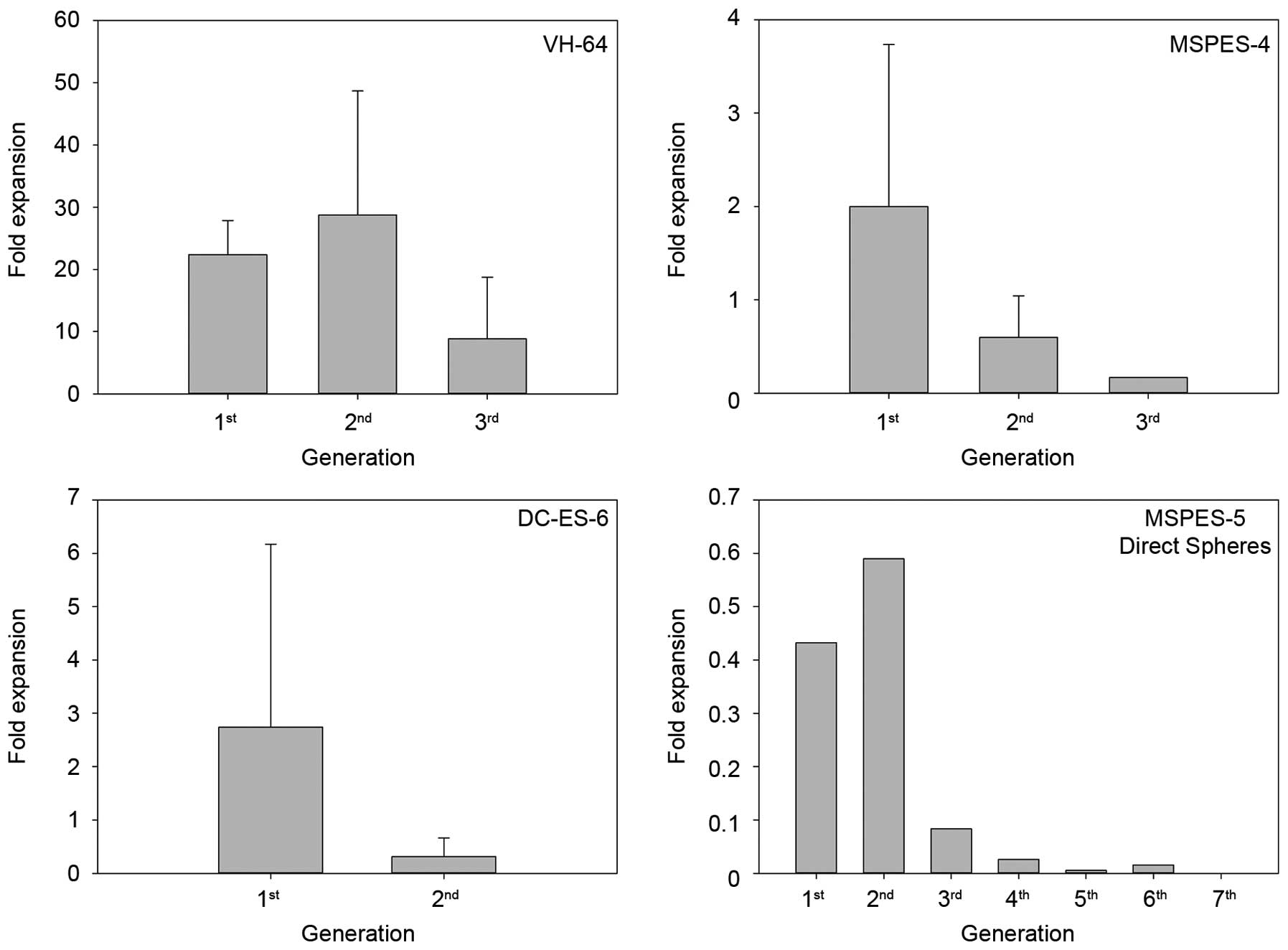
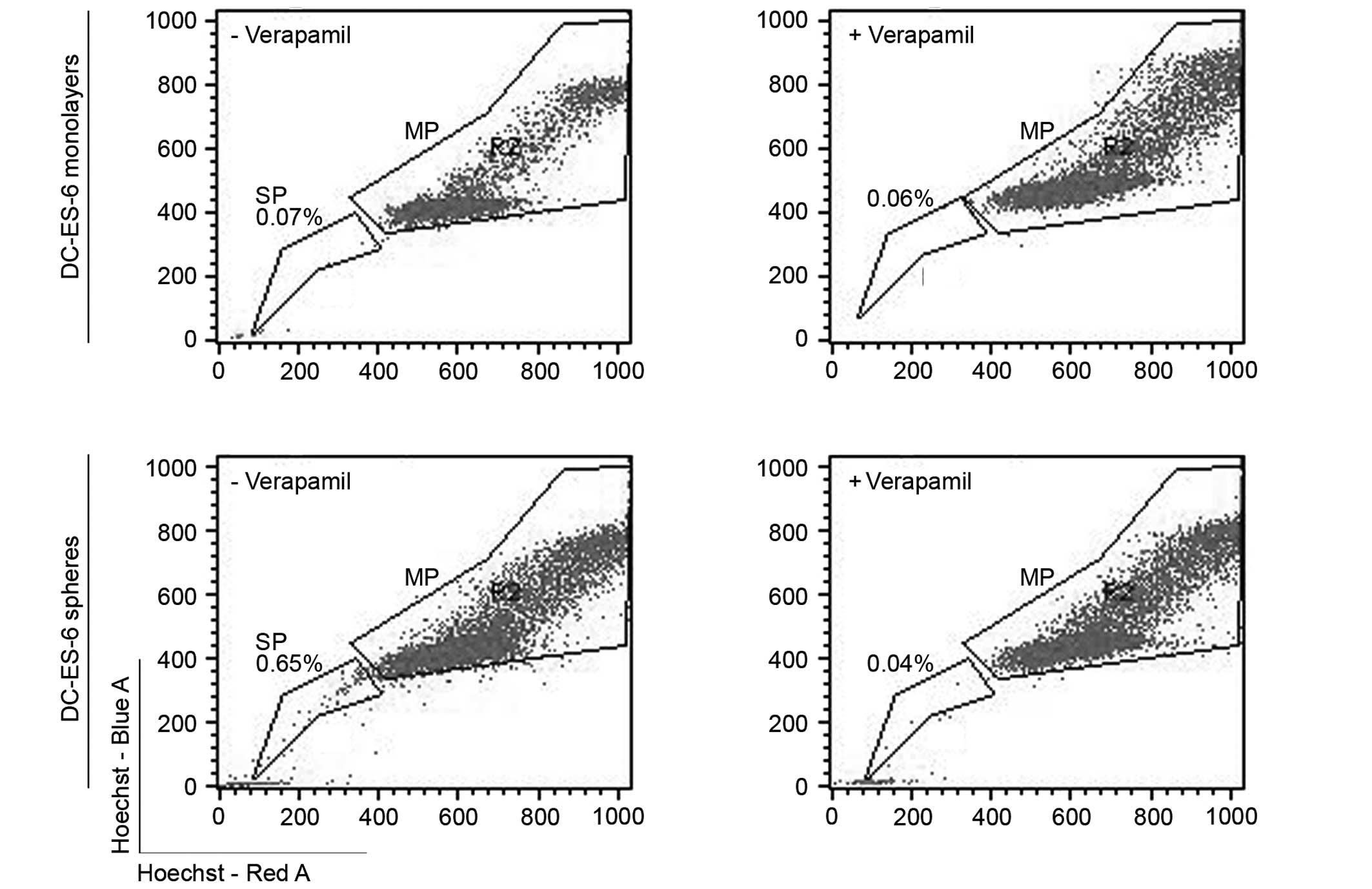
|
1
|
Paulussen M, Craft AW, Lewis I, et al:
Results of the EICESS-92 study: two randomized trials of Ewing’s
sarcoma treatment - cyclophosphamide compared with ifosfamide in
standard-risk patients and assessment of benefit of etoposide added
to standard treatment in high-risk patients. J Clin Oncol.
26:4385–4393. 2008.PubMed/NCBI
|
|
2
|
Paulussen M, Ahrens S, Burdach S, et al:
Primary metastatic (stage IV) Ewing tumor: survival analysis of 171
patients from the EICESS studies. European Intergroup Cooperative
Ewing Sarcoma Studies. Ann Oncol. 9:275–281. 1998. View Article : Google Scholar
|
|
3
|
Stahl M, Ranft A, Paulussen M, et al: Risk
of recurrence and survival after relapse in patients with Ewing
sarcoma. Pediatr Blood Cancer. 57:549–553. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cho D, Shook DR, Shimasaki N, Chang YH,
Fujisaki H and Campana D: Cytotoxicity of activated natural killer
cells against pediatric solid tumors. Clin Cancer Res.
16:3901–3909. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mackall CL, Rhee EH, Read EJ, et al: A
pilot study of consolidative immunotherapy in patients with
high-risk pediatric sarcomas. Clin Cancer Res. 14:4850–4858. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Thiel U, Pirson S, Müller-Spahn C, et al:
Specific recognition and inhibition of Ewing tumour growth by
antigen-specific allo-restricted cytotoxic T cells. Br J Cancer.
104:948–956. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Erkizan HV, Kong Y, Merchant M, et al: A
small molecule blocking oncogenic protein EWS-FLI1 interaction with
RNA helicase A inhibits growth of Ewing’s sarcoma. Nat Med.
15:750–756. 2009.PubMed/NCBI
|
|
8
|
Braun S, Pantel K, Müller P, et al:
Cytokeratin-positive cells in the bone marrow and survival of
patients with stage I, II, or III breast cancer. N Engl J Med.
342:525–533. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Woelfle U, Breit E, Zafrakas K, et al:
Bi-specific immunomagnetic enrichment of micrometastatic tumour
cell clusters from bone marrow of cancer patients. J Immunol
Methods. 300:136–145. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Freedman VH and Shin SI: Cellular
tumorigenicity in nude mice: correlation with cell growth in
semi-solid medium. Cell. 3:355–359. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shin SI, Freedman VH, Risser R and Pollack
R: Tumorigenicity of virus-transformed cells in nude mice is
correlated specifically with anchorage independent growth in vitro.
Proc Natl Acad Sci USA. 72:4435–4439. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lee J, Kotliarova S, Kotliarov Y, et al:
Tumor stem cells derived from glioblastomas cultured in bFGF and
EGF more closely mirror the phenotype and genotype of primary
tumors than do serum-cultured cell lines. Cancer Cell. 9:391–403.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lawlor ER, Scheel C, Irving J and Sorensen
PH: Anchorage-independent multi-cellular spheroids as an in vitro
model of growth signaling in Ewing tumors. Oncogene. 21:307–318.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wahl J, Bogatyreva L, Boukamp P, et al:
Ewing’s sarcoma cells with CD57-associated increase of
tumorigenicity and with neural crest-like differentiation capacity.
Int J Cancer. 127:1295–1307. 2010.
|
|
15
|
Ottaviano L, Schaefer KL, Gajewski M, et
al: Molecular characterization of commonly used cell lines for bone
tumor research: a trans-European EuroBoNet effort. Genes
Chromosomes Cancer. 49:40–51. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Landuzzi L, De Giovanni C, Nicoletti G, et
al: The metastatic ability of Ewing’s sarcoma cells is modulated by
stem cell factor and by its receptor c-kit. Am J Pathol.
157:2123–2131. 2000.
|
|
17
|
Suvà ML, Riggi N, Stehle JC, et al:
Identification of cancer stem cells in Ewing’s sarcoma. Cancer Res.
69:1776–1781. 2009.
|
|
18
|
Al Hajj M, Wicha MS, Benito-Hernandez A,
Morrison SJ and Clarke MF: Prospective identification of
tumorigenic breast cancer cells. Proc Natl Acad Sci USA.
100:3983–3988. 2003.
|
|
19
|
Singh SK, Hawkins C, Clarke ID, et al:
Identification of human brain tumour initiating cells. Nature.
432:396–401. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Goodell MA, Rosenzweig M, Kim H, et al:
Dye efflux studies suggest that hematopoietic stem cells expressing
low or undetectable levels of CD34 antigen exist in multiple
species. Nat Med. 3:1337–1345. 1997. View Article : Google Scholar
|
|
21
|
Coulon A, Flahaut M, Mühlethaler-Mottet A,
et al: Functional sphere profiling reveals the complexity of
neuroblastoma tumor-initiating cell model. Neoplasia. 13:991–1004.
2011.PubMed/NCBI
|
|
22
|
Hansford L, Smith K, Datti A, et al: Tumor
initiating cells from neuroblastoma, a neural crest-derived tumor.
Int J Dev Neurosci. 24:4892006. View Article : Google Scholar
|
|
23
|
Mahller YY, Williams JP, Baird WH, et al:
Neuroblastoma cell lines contain pluripotent tumor initiating cells
that are susceptible to a targeted oncolytic virus. PLoS One.
4:e42352009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rainusso N, Brawley VS, Ghazi A, et al:
Immunotherapy targeting HER2 with genetically modified T cells
eliminates tumor-initiating cells in osteosarcoma. Cancer Gene
Ther. 19:212–217. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Walter D, Satheesha S, Albrecht P, et al:
CD133 positive embryonal rhabdomyosarcoma stem-like cell population
is enriched in rhabdospheres. PLoS One. 6:e195062011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang S, Balch C, Chan MW, et al:
Identification and characterization of ovarian cancer-initiating
cells from primary human tumors. Cancer Res. 68:4311–4320. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Brown CE, Starr R, Martinez C, et al:
Recognition and killing of brain tumor stem-like initiating cells
by CD8+ cytolytic T cells. Cancer Res. 69:8886–8893.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gibbs CP, Kukekov VG, Reith JD, et al:
Stem-like cells in bone sarcomas: implications for tumorigenesis.
Neoplasia. 7:967–976. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhong Y, Guan K, Guo S, et al: Spheres
derived from the human SK-RC-42 renal cell carcinoma cell line are
enriched in cancer stem cells. Cancer Lett. 299:150–160. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Charafe-Jauffret E, Ginestier C, Iovino F,
et al: Breast cancer cell lines contain functional cancer stem
cells with metastatic capacity and a distinct molecular signature.
Cancer Res. 69:1302–1313. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
O’Brien CA, Pollett A, Gallinger S and
Dick JE: A human colon cancer cell capable of initiating tumour
growth in immunodeficient mice. Nature. 445:106–110.
2007.PubMed/NCBI
|
|
32
|
Yin S, Li J, Hu C, et al: CD133 positive
hepatocellular carcinoma cells possess high capacity for
tumorigenicity. Int J Cancer. 120:1444–1450. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jiang X, Gwye Y, Russell D, et al: CD133
expression in chemo-resistant Ewing sarcoma cells. BMC Cancer.
10:1162010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hirschmann-Jax C, Foster AE, Wulf GG, et
al: A distinct ‘side population’ of cells with high drug efflux
capacity in human tumor cells. Proc Natl Acad Sci USA.
101:14228–14233. 2004.
|
|
35
|
Murase M, Kano M, Tsukahara T, et al: Side
population cells have the characteristics of cancer stem-like
cells/cancer-initiating cells in bone sarcomas. Br J Cancer.
101:1425–1432. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Broadley KW, Hunn MK, Farrand KJ, et al:
Side population is not necessary or sufficient for a cancer stem
cell phenotype in glioblastoma multiforme. Stem Cells. 29:452–461.
2011. View
Article : Google Scholar : PubMed/NCBI
|
|
37
|
Barrett LE, Granot Z, Coker C, et al:
Self-renewal does not predict tumor growth potential in mouse
models of high-grade glioma. Cancer Cell. 21:11–24. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Feder-Mengus C, Ghosh S, Weber WP, et al:
Multiple mechanisms underlie defective recognition of melanoma
cells cultured in three-dimensional architectures by
antigen-specific cytotoxic T lymphocytes. Br J Cancer.
96:1072–1082. 2007. View Article : Google Scholar
|
|
39
|
Fischer K, Hoffmann P, Voelkl S, et al:
Inhibitory effect of tumor cell-derived lactic acid on human T
cells. Blood. 109:3812–3819. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hirschhaeuser F, Leidig T, Rodday B,
Lindemann C and Mueller-Klieser W: Test system for trifunctional
antibodies in 3D MCTS culture. J Biomol Screen. 14:980–990. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Jääskeläinen J, Kalliomäki P, Paetau A and
Timonen T: Effect of LAK cells against three-dimensional tumor
tissue. In vitro study using multi-cellular human glioma spheroids
as targets. J Immunol. 142:1036–1045. 1989.PubMed/NCBI
|
|
42
|
Lakshmikanth T, Burke S, Ali TH, et al:
NCRs and DNAM-1 mediate NK cell recognition and lysis of human and
mouse melanoma cell lines in vitro and in vivo. J Clin Invest.
119:1251–1263. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Parkhurst MR, Riley JP, Dudley ME and
Rosenberg SA: Adoptive transfer of autologous natural killer cells
leads to high levels of circulating natural killer cells but does
not mediate tumor regression. Clin Cancer Res. 17:6287–6297. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Geller MA, Cooley S, Judson PL, et al: A
phase II study of allogeneic natural killer cell therapy to treat
patients with recurrent ovarian and breast cancer. Cytotherapy.
13:98–107. 2011. View Article : Google Scholar : PubMed/NCBI
|