Introduction
Non-small cell lung cancer (NSCLC) is a common form
of lung adenocarcinoma, with a significant threat to public health
and accounts for 80% of the total lung cancer cases all over the
world (1). Although improvements in
diagnosis and treatment have made significant strides, the 5-year
survival rate still remains poor (2,3). Thus,
it is crucial for research to expand our understanding of non-small
cell lung cancer, to investigate the molecular mechanism on the
NSCLC progression and identify novel diagnosis and therapeutic
targets. Proliferation and epithelial-mesenchymal transition (EMT)
in cancer are two critical malignant characteristics (4–6). Cell
proliferation is considered as a complicated progress, which is
regulated by a series of genes, among them, CCND1, was a classic
oncogenic protein that could promote cell proliferation and the
beginning of S phase in many cancers (7).
Epithelial-mesenchymal transition is a complicated
progress, during which, epithelial cells lose their epithelial
features and gain mesenchymal characteristics, and become invasive.
As shown by many groups, loss of epithelial marker E-cadherin and
gain of mesenchymal markers N-cadherin are usually considered as
the molecular markers of EMT (8–10).
Recent studies have shown that many transcription factors are
involved in the progress of EMT, such as Snail (11,12).
However, the regulation of Snail in NSCLC EMT and metastasis
remains poorly understood.
There are over 40 members in the forkhead
transcription factor family, which are characterized by a forkhead
winged helix-turn-helix DNA binding domain, and regulate a diverse
range of tumor progression, such as proliferation, apoptosis,
metabolism and invasion. In mammals, Foxk2 is one of the two FOXK
isoforms, however, little is known about the biological role and
potential mechanisms of Foxk2 in tumorigenic driver pathways.
Materials and methods
Cell culture and transfection
Human non-small cell lung cancer cell lines A549,
NCI-H520, H1299, H358, H460 cells and human lung fibroblast cell
line WI-38 cells were purchased from the Type Culture Collection of
the Chinese Academy of Sciences (Shanghai, China). Cells were
maintained in RPMI-1640 medium (Life Sciences, Corning, NY, USA)
containing 10% fetal bovine serum (FBS) (Gibco, Rockville, MD,
USA), in humidified atmosphere of 5% CO2 at 37°C, with
100 units of penicillin/streptomycin (Invitrogen, Carlsbad, CA,
USA).
Transfection was performed using Lipofectamine 2000
(Invitrogen) according to the manufacturer's instructions. The
relative recombinant lentivirus expressing Foxk2 or shRNA were
purchased from Genechem Company (Shanghai, China). At 48–72 h after
transfection, cells were analyzed as required.
Patients and specimens
The 50 samples of NSCLC and the adjacent non-tumor
tissues were obtained from surgical specimens in Tongji Hospital of
Tongji Medical College from the year of 2010-2012. The patients
were collected with complete clinicopathologic information, and the
patients who received chemotherapy and radiotherapy were excluded.
The tissues were obtained immediately frozen and stored at −80°C
until use. This study was approved by the Ethics Committee of
Tongji Medical College, and consent information was obtained from
each patient, followed up for survival.
Cell proliferation assay
Cells were cultured in 24-well plates at a
concentration of 1×104 cells/well and incubated for
various periods, 24 h after transfection, 0.5 ml methyl thiazolyl
tetrazolium (MTT) solution (Sigma-Aldrich, St. Louis, MO, USA) was
added to each well and cells were further incubated for 4 h at
37°C. Dimethyl sulfoxide (0.2 ml) was added to the supernatant
medium to dissolve the purple crystals for 10 min. The optical
density was determined at 490 nm with a SpectraMax
spectrophotometer (Molecular Devices, Sunnyvale, CA, USA).
Edu assay
Edu (5-ethynyl-2¢-deoxyuridine) assay was used to
measure cell proliferation in vitro, according to the
manufacturer's instructions. Cells (1×104) were seeded
into a 96-well plate, after transfected with recombinant lentivirus
expressing Foxk2 or shRNAs, cells were labeled with 50 µM Edu for 2
h before they were formalin fixed and processed. Then the stained
cells were observed under a fluorescent microscope.
Cell cycle analysis
The relative cells were seeded in 10-cm diameter
plates in 10% FBS. At 48 h after transfection, a total of
5×106 cells were harvested, washed once with PBS and
fixed with 70% ice-cold ethanol (diluted with PBS) for 24 h at 4°C.
Then the cells were rinsed with PBS again and incubated with 0.5 ml
propidium iodide, 0.5 mg/ml ribonuclease A and 0.2% Triton X-100
for 30 min. Cell cycle distribution was measured and analyzed by BD
C6 flow cytometry (BD Biosciences, San Jose, CA, USA). Each
experiment was performed in triplicate.
Wound healing assay
For the wound healing assay, cells were seeded into
12-well plates and cultured in complete medium with 10% FBS to form
a monolayer, 24 h after transfection, a uniform straight scratch
was made by a pipette tip. The medium was replaced without FBS and
the wound area was observed under the microscope (Olympus
Corporation, Tokyo, Japan). Cells were further incubated at 37°C
with 5% CO2 for 24 h. The speed of wound closure was
measured. Each experiment was performed in triplicate.
Invasion assay
Cell invasion was measured using Transwell assay,
Transwell chambers were from Corning Inc., Corning, NY, USA.
Matrigel (30%) (BD Biosciences) was placed on the upper surface of
the chamber. A total of 2×105 cells/well without bovine
serum were seeded into the upper chambers, and 1 ml RPMI-1640 with
10% FBS was added to the lower chamber. At 48 h after incubation at
37°C, the penetrated cells were fixed with methanol, stained with
crystal violet and counted, while the remaining cells in the upper
chamber were removed. Three independent experiments were
performed.
RNA extraction and quantitative
real-time PCR analysis
Total RNA was extracted from tumor tissues or from
the relative cells using TRIzol reagent (Thermo Fisher Scientific)
following the manufacturer's protocol. Total RNA (1 µg) was used to
transcribe to cDNA using with M-MLV reverse transcriptase.
Real-time PCR was carried out using the ABI 7500
Real-time PCR system (Applied Biosystems, Foster City, CA, USA).
The expression of TWIST1 was determined using SYBR green qPCR assay
(Takara, Dalian, China) 2-∆∆Ct analysis method was used and
normalized with GAPDH. Specific primers were obtained from
Invitrogen. All reactions were performed in triplicate.
Western blotting
Cells were harvested using sodium dodecyl sulfate
lysis buffer for 30 min at 4°C. The lysates were centrifuged at
13000 rpm for 15 min at 4°C, and the supernatants were harvested.
An equal amount of protein was separated on 10% SDS-PAGE gels and
then were transferred onto polyvinylidene difluoride membranes
(Thermo Fisher Scientific). After blocked with 5% non-fat milk, the
proteins were probed with primary antibodies against Foxk2 (Santa
Cruz Biotechnology, Santa Cruz, CA, USA), or antibodies from EMT
antibody kit (Cell Signaling Technology, Danvers, MA, USA) or
β-actin (cat. no. SC47778; Santa Cruz Biotechnology) overnight at
4°C (1:1000 dilution). Followed by washing five times and incubated
with respective secondary antibodies peroxidase-conjugated anti-IgG
(Abcam), and were visualized using a chemiluminescent detection
system (Western blot detection system; Thermo Fisher Scientific),
according to the manufacturer's instructions. Representative data
are shown from three individual experiments.
ChIP-seq and qChIP analysis
Approximately 5×107 A549 cells were used
for each ChIP-seq or qChIP assay. The chromatin DNA was
precipitated by normal rabbit IgG (negative control) or polyclonal
antibodies against Foxk2 at 4°C overnight. After purified with the
Qiagen PCR purification kit, qChIP was analyzed by quantitative PCR
using specific primers. ChIP-seq was performed by the CapitalBio
Corporation, Beijing, China.
Luciferase reporter analysis
For target gene reporter construction, the sequence
of the target gene promoter and partial first exon was obtained by
PCR, these PCR products were digested and ligated into the pGL3
basic vector (Promega). A549 cells or NCI-H520 cells in 96-well
plates were transfected with target gene luciferase reporter,
Renilla plasmid, and Foxk2 constructs or shRNAs, using
Lipofectamine LTX-Plus (Invitrogen).
The 3′UTR reporter plasmids (RL-Foxk2) were
constructed by Jikai Gene Co. (Shanghai, China). Mutation of the
Foxk2 3′UTR were generated by QuikChang Multi site-directed
mutagenesis kit (Stratagene). The RL reporter plasmids (a firefly
luciferase reporter was used for normalization), together with
miR-1271 and pGL3-control were transfected into A549 cells or
NCI-H520 cells, 48 h after transfection, luciferase reporter assay
was performed.
Statistical analysis
Statistical analysis was carried out using SPSS
software (version 17.0). All assays were performed at least three
times, and the data were expressed as the means ± SD, unless
otherwise mentioned, P<0.05 was considered to be significant.
Survival time was analyzed using the Kaplan-Meier method with
log-rank tests. Two-tailed Student's t-test was used to analyze the
difference between two groups, one-way ANOVA was applied to
evaluate the difference among different lung cancer tissues and
adjacent non-tumor tissues. TargetScan Human (http://www.targetscan.org/vert_61/) was used to
perform prediction as to which of the miRNA target Foxk2.
Results
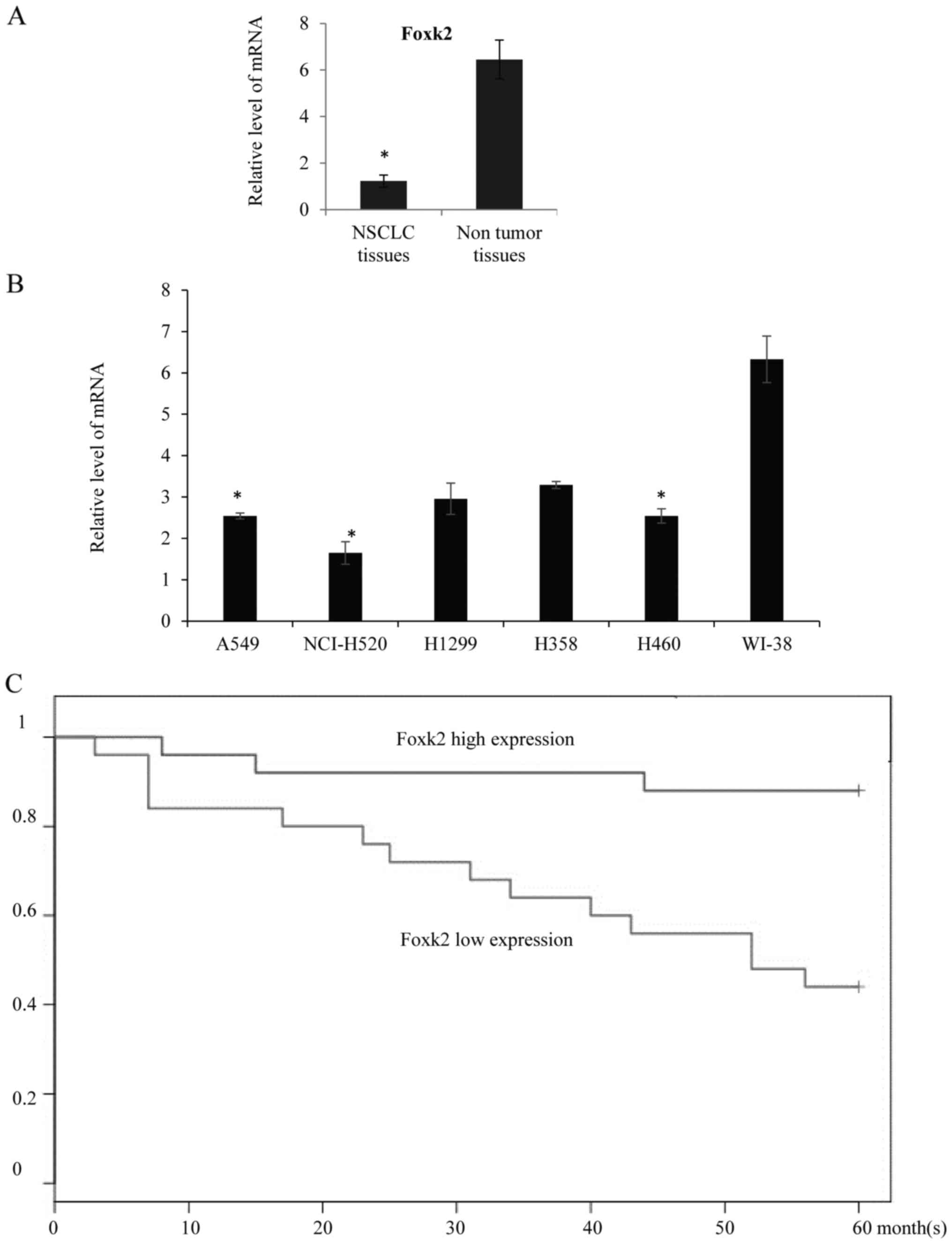
Foxk2 expression was downregulated in
lung adenocarcinoma tissues and NSCLC cell lines
In order to understand the role of Foxk2 during the
progress of NSCLC, we first assayed the expression level of Foxk2
in NSCLC tissues, compared with the adjacent non-tumor tissues,
qRT-PCR showed that the mean expression of Foxk2 in NSCLC tissues
was lower than control (Fig. 1A).
In the NSCLC cell lines, qRT-PCR also revealed that the Foxk2
expression was lower in the NSCLC A549 cells, NCI-H520 cells, H1299
cells, H358 cells and H460 cells compared with the human lung
fibroblast WI-38 cells (Fig. 1B).
The relationship between Foxk2 expression and patient survival were
investigated. Patients collected in the Tongji Hospital Hospital
were divided in to two groups, depending on the median Foxk2
expression level, and found that high Foxk2 expression was
correlated with better overall survival rate, and Kaplan-Meier
curves were drawn (Fig. 1C,
P<0.05).
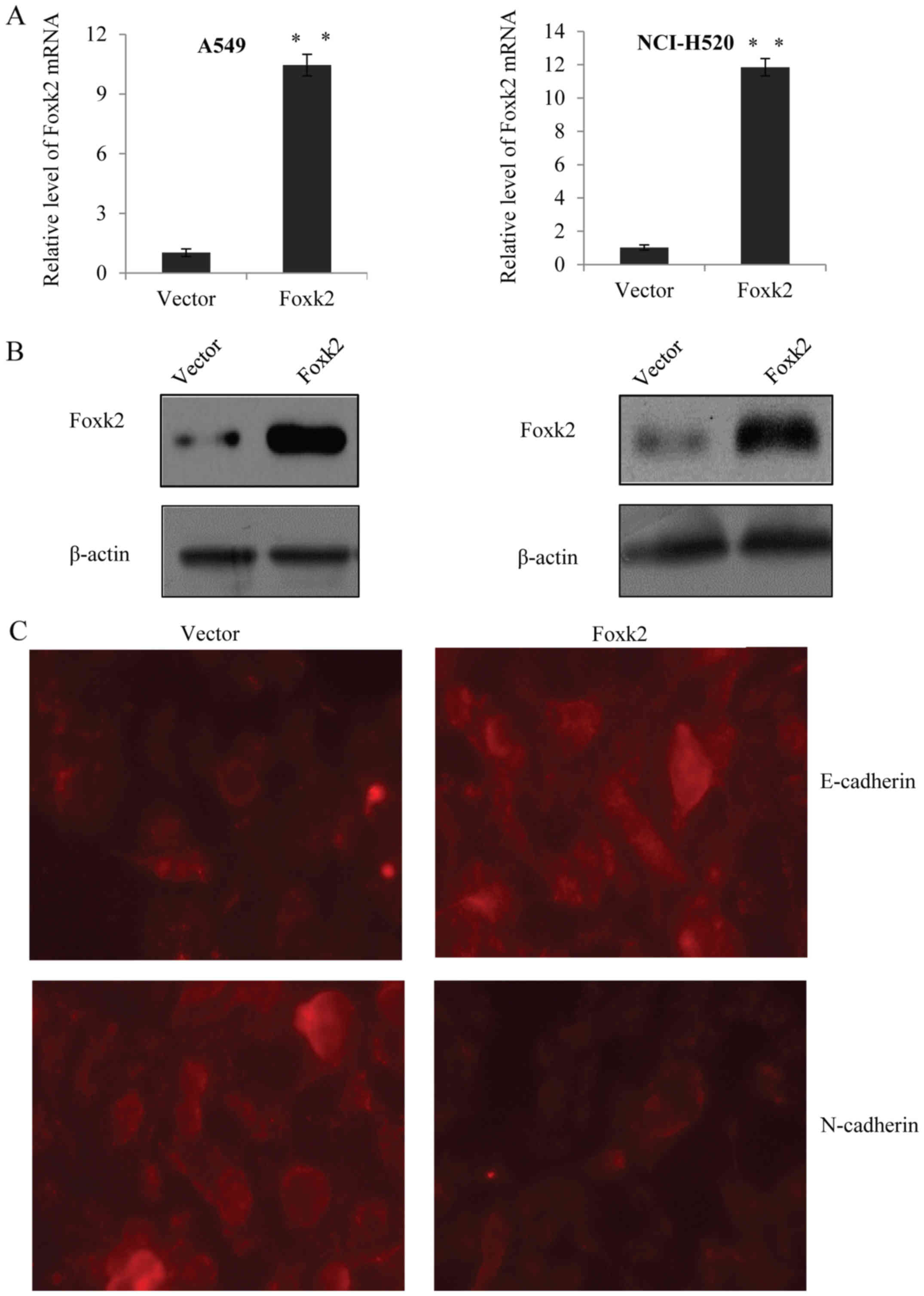
Overexpression of Foxk2 suppresses EMT
in NSCLC cells
In order to understand the biological significance
of Foxk2 involved in non-small cell lung cancer, recombinant
lentivirus expressing Foxk2 constructs were developed and
transfected into A549 cells and NCI-H520 cells, respectively.
qRT-PCR and western blotting was used to detect the transfection
efficiency, β-actin was used as control. As expected, the Foxk2
lentivirus was successfully expressed in the two cell lines
(Fig. 2A and B). To explore whether
Foxk2 inhibits EMT in NSCLC cells, immunofluorescence assay was
performed in A549 cells, and the results revealed that the
overexpression of Foxk2 markedly promoted the expression of
epithelial marker E-cadherin and markedly suppressed the expression
of mesenchymal marker N-cadherin (Fig.
2C). Furthermore, qRT-PCR was used in A549 cells and NCI-H520
cells to detect the mRNA level of E-cadherin and α-catenin, as well
as the level of N-cadherin and vimentin (Fig. 2D). Western blot assays were carried
out in the above two cell lines, and the results showed that
restoration of Foxk2 significantly increased the expression of
epithelial markers (E-cadherin and α-catenin) and significantly
inhibited the expression of mesenchymal markers (N-cadherin and
vimentin) (Fig. 2E). These results
suggested that Foxk2 was responsible for the inhibition of EMT in
NSCLC cells.
Foxk2 inhibits NSCLC cell migration
and invasion in vitro
As known, EMT affects tumors in an uncontrolled
fashion and they acquire invasive ability, we supposed Foxk2 may
have function in NSCLC cell migration and invasion. In support of
our hypothesis, two different lentivirus stably silencing Foxk2 was
developed and infected into A549 cells and NCI-H520 cells. Both
mRNA level and protein level of Foxk2 in different groups were
detected. As shown in Fig. 3A, mRNA
level of Foxk2 was significantly reduced when cells were infected
with ShFoxk2#1, or ShFoxk2#2 compared with the negative control
lentivirus group (SCR). Concomitantly, in western blot analysis
(Fig. 3B), the expression of Foxk2
was also remarkably downregulated in ShFoxk2-infected cells as
compared with the control group. Both lentiviruses were
successfully constructed, but the knockdown efficiency of ShFoxk2#2
was better than that of ShFoxk#1 and was chosen for further
study.
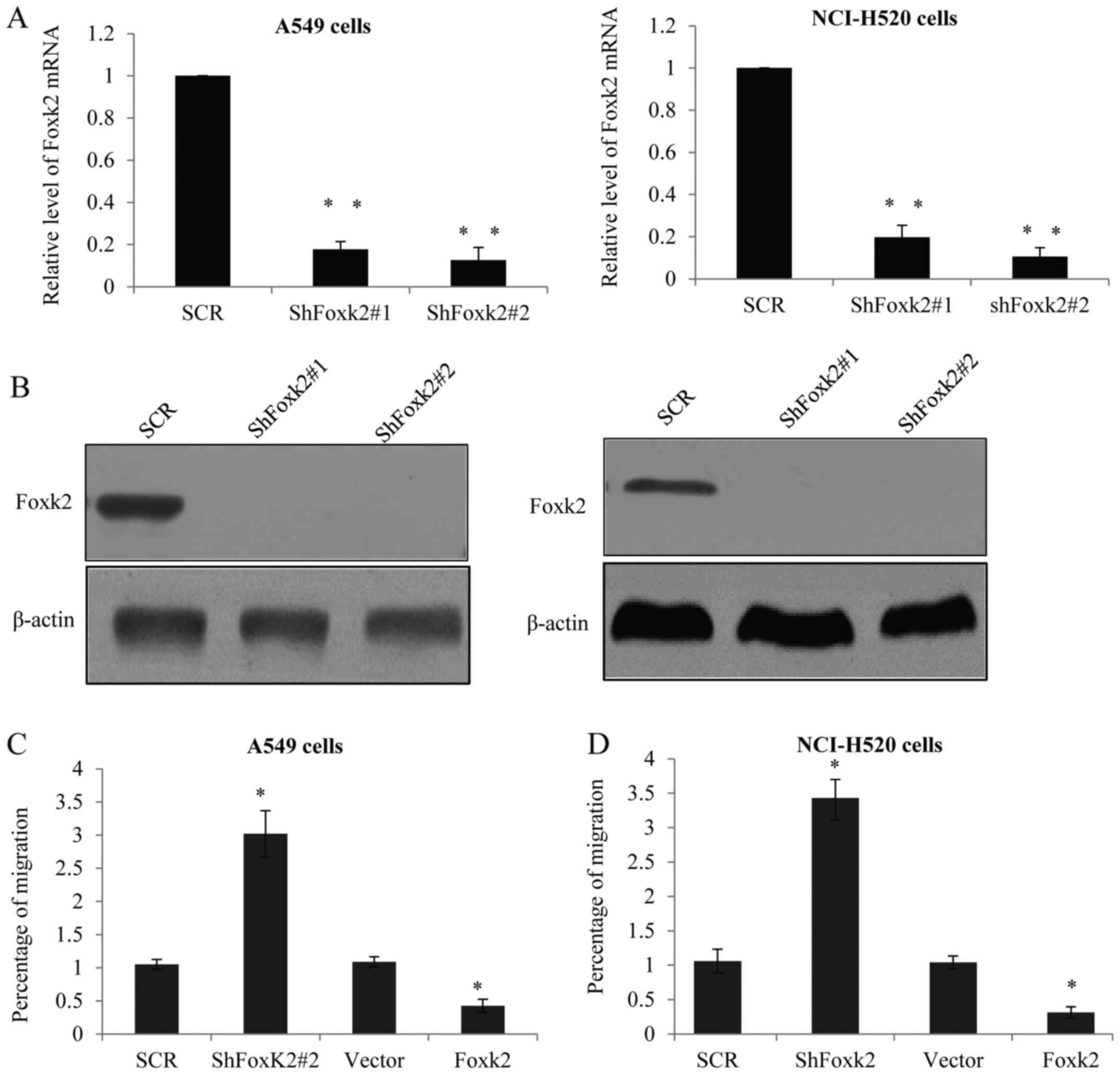 | Figure 3.Foxk2 inhibits NSCLC cell migration
and invasion in vitro. (A) The knock-down efficiencies of
ShFoxk2#1, ShFoxk2#2 were confirmed by qRT-PCR in A549 cells or
NCI-H520 cells, β-actin was used as a normalization control (left
panel), experiments were repeated three times, **P<0.01. (B) The
protein level of Foxk2 was measured by western blotting in A549
cells or NCI-H520 cells. (C) A549 cells transfected with SCR,
ShFoxk2#2, vector, Foxk2 were subjected to wound-healing analysis.
Data are presented as fold change. (D) Wound-healing analysis was
performed in NCI-H520 cells. *P<0.05. Foxk2 inhibited NSCLC
cells migration and invasion in vitro. (E) A549 cells were
transfected with SCR, ShFoxk2#2, vector, Foxk2, Transwell assay was
performed, and invaded cells were stained and counted.
Representative images were shown and statistically analyzed were
represented. (F) Transwell assay was performed in NCI-H520 cells.
*P<0.05. |
We further proved the gain-of-function
effect or loss-of-function effect of Foxk2 on the invasive
potential by wound-healing assays in A549 cells and NCI-H520
cells
As shown in Fig. 3C and
D, Foxk2 knockdown group was more efficient in wound healing,
whereas cells treated with Foxk2 overexpression constructs, were
resistant to wound healing to certain degrees, compared with the
control groups, the overall tendency was shown. Further, when Foxk2
(ShFoxk2#2) was knocked down in A549 cells, there was an increase
in the invasive potential, as measured by the Transwell analysis.
To the contrary, when cells were transferred with Foxk2 lentivirus,
there was a decrease in the invasive potential of A549 cells
(Fig. 3E). Similar tendency was
shown in the NCI-H520 cells transfected with the relative
lentivirus (Fig. 3F).
Foxk2 modulates proliferation and
tumorigenicity of NSCLC cells in vitro
In order to further understand the role of Foxk2
tumorigenicity in NSCLC cells, MTT assays was performed in A549
cells transfected with vector, Foxk2 gain-of-function, Foxk2
loss-of-function lentivirus. As shown in Fig. 4A, Foxk2 overexpression showed an
evident growth inhibition, while Foxk2 was knocked down, there was
an obvious growth promotion, compared with the vector group.
Similar result was also observed in the NCI-H520 cells (Fig. 4B). Edu test further proved the
function of Foxk2 in suppressing proliferation of A549 cells
(Fig. 4C) and NCI-H520 cells
(Fig. 4D) in vitro. To
explore the mechanism of Foxk2 contribution to cell proliferation,
we examined cell cycle distribution using flow cytometry in the
above two cell lines, compared with the control group cells,
ShFoxk2 transfected cells showed a substantial increase in S phase
and a decrease in G1 phase populations, while overexpression of
Foxk2 blocks the G1 phase to S phase (Fig. 4E and F). The above data suggested
that Foxk2 might take part in the inhibition of osteosarcoma cell
grow through induction of G1 arrest.
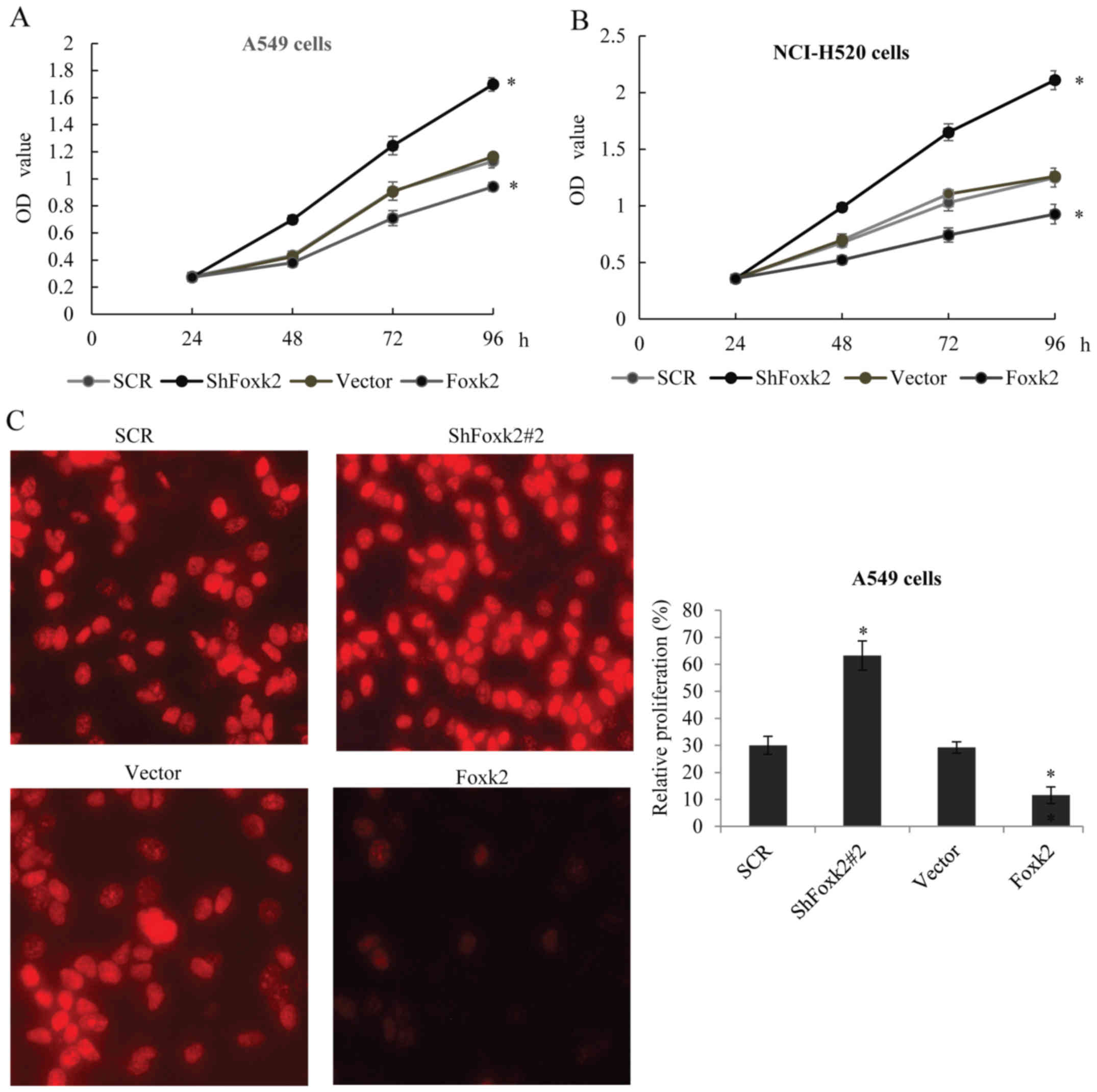 | Figure 4.Foxk2 modulates proliferation and
tumorigenicity of NSCLC cells in vitro. (A) MTT assay was
used to measure the A549 proliferation. Equal numbers of cells
transfected with SCR, ShFoxk2#2, vector, Foxk2 were cultured, MTT
assay was performed every 24 h. (B) The above experiment was
performed in NCI-H520 cells. (C) Edu assay was performed in A549
cells transfected with SCR, ShFoxk2#2, vector, Foxk2.
Representative images are shown, statistical analysis are presented
as fold change. (D) The above experiment was performed in NCI-H520
cells. (E) Cell cycle distribution was performed using flow
cytometry (FCM) in A549 cells transfected with SCR, ShFoxk2#2,
vector, Foxk2. The percentage of cells in the G0/G1, S, and G2/M
phase are shown, experiments were repeated three times, *P<0.05.
(F) Cell cycle distribution was performed in NCI-H520 cells. |
Identification of genome-wide
transcriptional targets for Foxk2
As known, Foxk2 is a transcription factor which acts
to repress gene transcription. In order to understand the molecular
mechanism of Foxk2 in regulating NSCLC, ChIP-seq was performed
using the Foxk2 antibody in A549 cells to identify the potential
target genes, normal IgG was used as negative control. As shown in
Fig. 5A, the Foxk2 ChIP-seq peak
distribution and 806 different gene promoters were targeted by
Foxk2. Those genes were further classified into various signaling
pathways using KEGG, which included focal adhesion, PI3K-Akt
signaling pathway, pathway in cancer, amoebiasis, cell cycle and
TGF-β pathways that were critically involved in EMT, migration,
invasion and proliferation (Fig.
5B). To validate the ChIP-seq result, different target genes
involved in classified pathways were selected to perform qChIP
assay, consistent with the ChIP-seq, on the promoter of CDH2
(N-cadherin), Snail, CCND1 and CDK4, there was obvious bindings of
Foxk2 compared to the normal IgG (Fig.
5C). Real-time PCR was further carried out in A549 cells
(Fig. 5D) with Foxk2 knocked down
or Foxk2 overexpression, to detect the change of the target genes,
as expected, in the Foxk2 knocked down group, the expression of
CDH2 (N-cadherin), Snail, CCND1 and CDK4 increased, while the
overexpression of Foxk2 showed the opposite tendency, similar
result was also observed in the NCI-H520 cells (Fig. 5E).
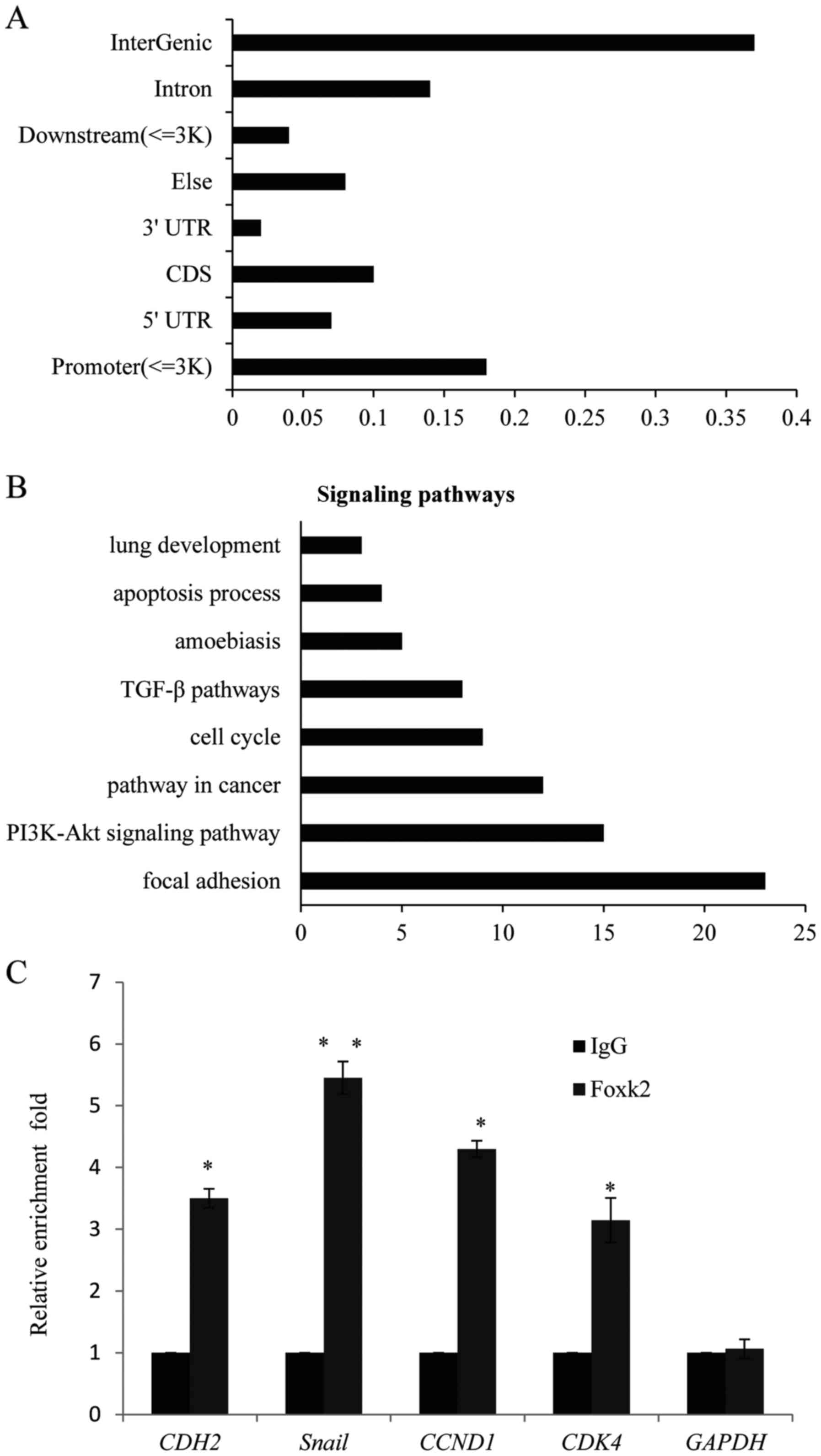 | Figure 5.Identification of genome-wide
transcriptional targets for Foxk2. (A) ChIP-seq assays were
performed in A549 cells with Foxk2 or normal IgG as negative
control, the peak distribution is shown. (B) KEGG was used to
analyze the target genes pathways, the relative pathways are shown.
(C) qChIP experiments were performed in A549 cells with Foxk2 or
normal IgG, the enrichments on the promoter of CDH2 (N-cadherin),
Snail, CCND1 and CDK4 were detected. Each bar indicates mean ± SD,
of three independent experiments. **P<0.01. (D) Foxk2
overexpression led to decrease in N-cadherin, Snail, cyclin D1 or
CDK4, mRNA level was measured by qPCR in A549 cells, while the
knockdown of Foxk2 showed the opposite tendency. (E) The
overexpression or knockdown of Foxk2 was performed in NCI-H520
cells. |
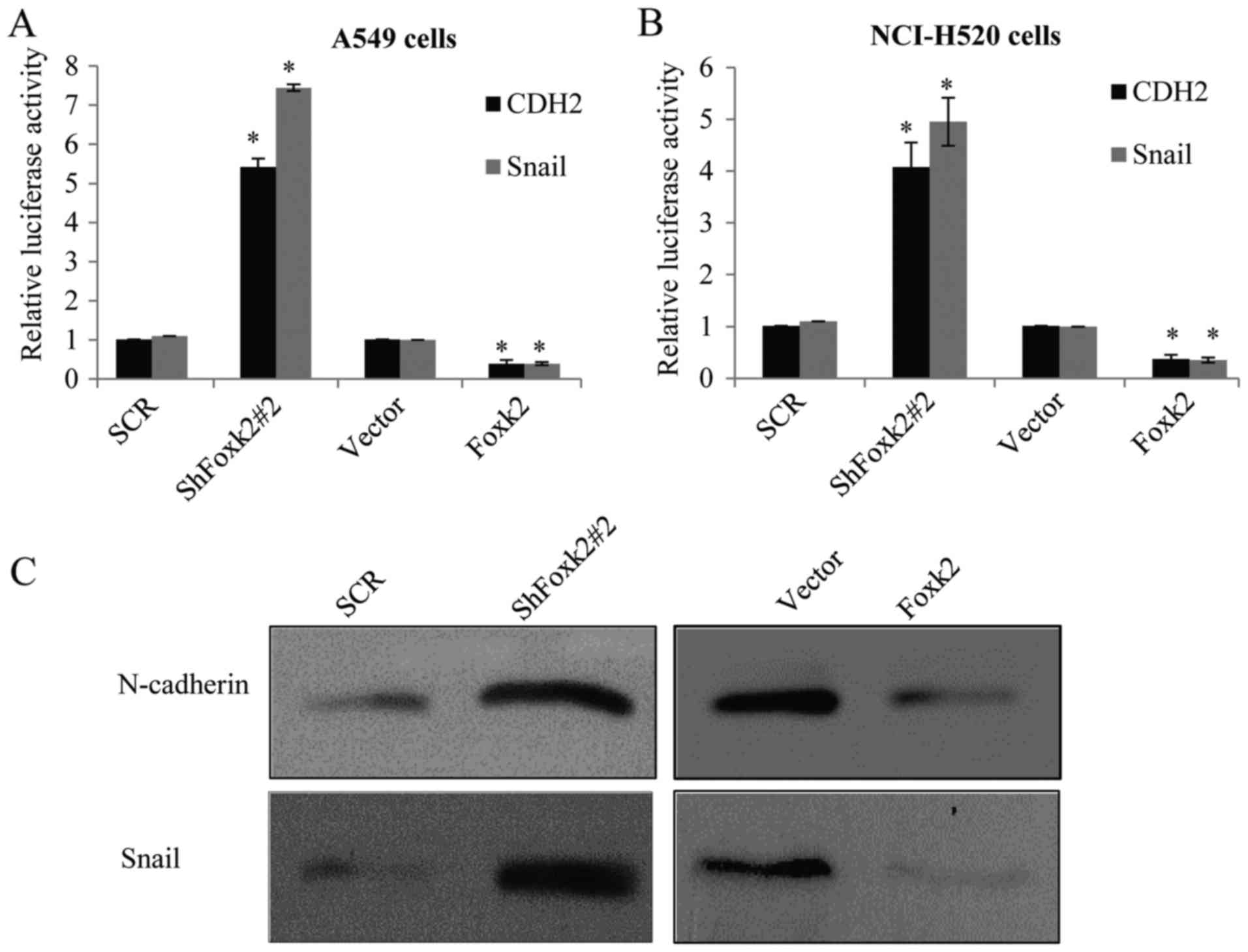
Molecular mechanism of Foxk2
inhibiting NSCLC cell EMT and invasion
As reported, CDH2 (N-cadherin) and Snail are usually
considered to have crucial roles in the progress of EMT, to
understand whether Foxk2 has a direct transcription repression on
the two target genes, luciferase reporter activity assays were
carried out. In Foxk2 depletion or overexpression of A549 cells,
transfected with CDH2 or Snail promoter-driven luciferase reporter,
a significant repression effect of Foxk2 on the indicated reporter
activity was observed (Fig. 6A),
similar result was also observed in the NCI-H520 cells (Fig. 6B). When Foxk2 was knocked down in
A549 cells (Fig. 6C), in protein
level, N-cadherin and Snail increased, while the overexpression of
Foxk2 resulted in the decrease of N-cadherin and Snail, further
supporting the notion that Foxk2 could repress target gene
expression from transcription level, similarly to the NCI-H520
cells (Fig. 6D). In Transwell
assay, A549 transfected with lentiviruses carrying ShFoxk2 plus
ShSnail or ShFoxk2 plus ShN-cadherin could partially rescue the
effect of Foxk2 knockdown on the invasive potential of A549 cells
(Fig. 6E), also in the NCI-H520
cells (Fig. 6F).
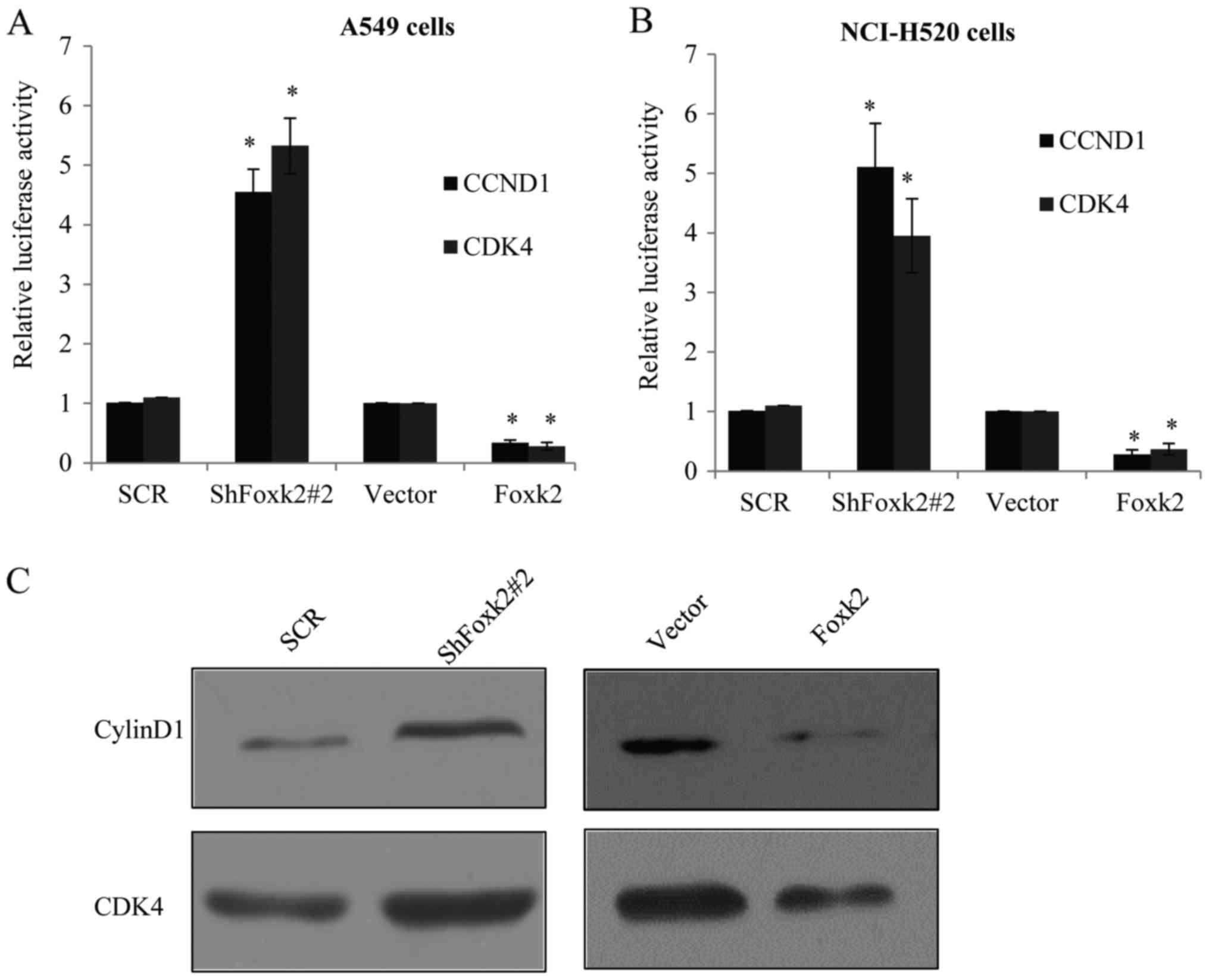
Foxk2 suppresses the activity of
PI3K/AKT/mTOR signaling pathway to inhibit NSCLC cell
tumorigenicity
Attributed to the fact that Foxk2 suppressed NSCLC
cell cycle progression, and as indicated from the ChIP-seq result,
cell cycle G1 to S checkpoint proteins CCND1 and CDK4 were
considered as the two target genes of Foxk2, and luciferase
reporter activity assays were carried out. In Foxk2 depletion or
overexpression of A549 cells, transfected with CCND1 or CDK4
promoter-driven luciferase reporter, Foxk2 has a significant
repression effect on the reporter activity (Fig. 7A), similar result was also observed
in the NCI-H520 cells (Fig.
7B).
Next, we examined the protein expression of cyclin
D1 (CCND1) and CDK4, in both Foxk2-overexpressed or knocked down
A549 cells. We found that introduction of Foxk2 blocked the
expression of cyclin D1 and CDK4, while knocking down endogenous
Foxk2 induced the target gene expression of cyclin D1 and CDK4
(Fig. 7C). Similar tendency was
also shown in the NCI-H520 cells (Fig.
7D). Many studies have indicated the PI3K/Akt signaling pathway
was responsible for the cell cycle, we also examined the effect of
Foxk2 on PI3K/Akt signaling pathway, and found that introduction of
Foxk2 reduced the expression of pPI3K (Tyr458) and pAkt (Ser473),
while knockdown of Foxk2 increased the expression respectively
(Fig. 7C and D), whereas their
total levels remained unchanged (data not shown). MTT assay further
proved that A549 transfected with lentiviruses carrying ShFoxk2
plus shCyclinD1 or ShFoxk2 plus shCDK4 could partially rescue the
effect of Foxk2 knockdown on the tumorigenesis potential of A549
cells (Fig. 7E), also in the
NCI-H520 cells (Fig. 7F).
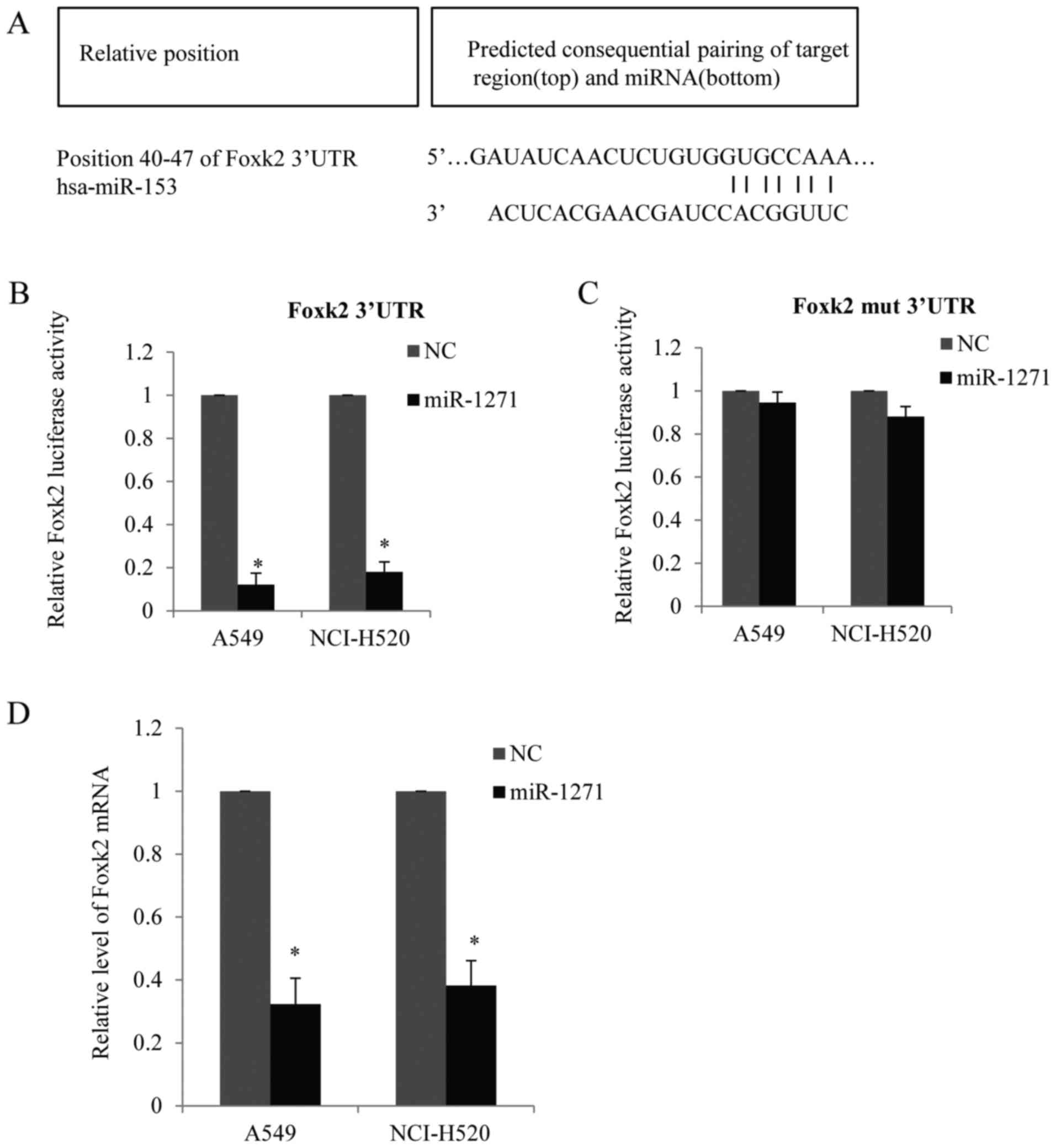
Foxk2 is negatively targeted by
miR-1271
Since Foxk2 was downregulated in the NSCLC cells, we
tried to find which miRNAs target Foxk2. Bioinformatics was used to
predicate the potential miRNAs. Based on their potential relevance
to NSCLC and conservation, we found the Foxk2 3-UTR harbors
potential miR-1271 target sites (position 40–47) (GUGCCAA), the
site was complementary to the human miR-1271 heptamer motif
(CACGGUU) and highly conserved regions in different species
(Fig. 8A). Next, the 3′UTR of Foxk2
was cloned into luciferase reporter plasmids. miR-1271 and the
reporter plasmids were co-transfected into A549 cells or NCI-H520
cells. We found that miR-1271 reduced mainly the Foxk2 luciferase
activity (Fig. 8B). To further
confirm the repression effect of miR-1271 on Foxk2, we mutated
Foxk2 3′UTR as indicated (GUGCAAA) and the mutated UTR was cloned
into luciferase reporter plasmids and co-transfected with miR-1271
into A549 cells or NCI-H520 cells (Fig.
8C). As shown, miR-1271 did not reduce the luciferase activity
in cells transfected with mutated Foxk2 3′UTR. The mRNA level of
Foxk2 was reduced in the A549 cells or NCI-H520 cells transfected
with miR-1271 compared with the control groups (Fig. 8D). Thus, our data indicated that
Foxk2 was negatively targeted by miR-1271.
Discussion
Non-small cell lung cancer is not only a disease
with malignant proliferation, but also with high invasiveness and
metastasis. Foxk2, a member of the forkhead transcription factor
family has been reported to regulate a number of essential
biological progresses (13,14). However, in NSCLC, the role and
molecular mechanism of Foxk2 remain largely unknown.
In this study, the expression of Foxk2 was evaluated
in NSCLC tissues and cell lines. We found that Foxk2 was
significantly downregulated in NSCLC tissues compared with the
corresponding adjacent paracarcinoma tissues. Foxk2 has also low
expression in the NSCLC cell lines. Our data hinted that lost
expression of Foxk2 was involved in the pathogenesis of NSCLC.
Subsequently, we presented evidence that Foxk2 inhibited NSCLC cell
EMT ability. As lentiviral-delivered Foxk2 markedly induced the
expression of epithelial markers (E-cadherin and α-catenin) and
significantly reduced the expression of mesenchymal markers
(N-cadherin and vimentin) in A549 and NCI-H520 cells. Further, the
overexpression of Foxk2 reduced cell invasion, growth,
proliferation and blocked cell cycle progression. On the contrary,
stably knocking down Foxk2 significantly elevated NSCLC cell
malignancy phenotypes. ChIP-seq was used to identify genome-wide
transcriptional targets for Foxk2.
Several classic oncogenic proteins were proved to be
Foxk2 target genes and repressed by Foxk2. Such as N-cadherin,
which was considered not only as a mesenchymal marker but also
promoted cancer cell invasion. Snail was a crucial EMT inducer and
contributed to the NSCLC metastasis (15). CCND1 is involved in promoting cell
proliferation in many cancers (16,17).
CDK4, a member of the cyclin-dependent kinase family, which was
responsible for cell cycle G1-S phase progression (18). By repressing the above target genes,
we underlined the mechanism in which Foxk2 suppressed NSCLC tumor
progression. MicroRNAs are short, non-coding RNAs (18–25
nucleotides) which negatively regulate numerous target genes by
complementary recognized sequences of 3-UTRs at
post-transcriptional level. MicroRNAs play important roles in the
pathogenesis of various cancers, including lung cancer,
particularly non-small cell lung cancer (NSCLC) (19,20).
miR-1271 was reported to promote NSCLC proliferation and invasion
via targeting HOXA5 (21). In our
study, we found that Foxk2 was another target gene of miR-1271, and
it is reasonable to suggest that the low expression of Foxk2 was
possibly due to the repression function of miR-1271.
In conclusion, our study on the function and
mechanism of Foxk2 in NSCLC indicated that miR-1271 may target
Foxk2 in NSCLC. Our study indicates that Foxk2 could be a tumor
suppressor and miR-1271 a potential target in NSCLC.
Acknowledgements
This study was financially supported by the National
Natural Science Foundation of China (no. 81470252). We thank all
our coworkers who contributed to this study.
References
|
1
|
van Zandwijk N and Fong KM: Update in lung
cancer: Prologue to a modern review series. Respirology.
20:183–184. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
O'Dowd EL and Baldwin DR: Early diagnosis
pivotal to survival in lung cancer. Practitioner. 258:21–24, 22–23.
2014.
|
|
3
|
Sharma SP: New therapeutic target for
non-small-cell lung cancer. Lancet Oncol. 15:e5332014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yuan X, Wu H, Han N, Xu H, Chu Q, Yu S,
Chen Y and Wu K: Notch signaling and EMT in non-small cell lung
cancer: Biological significance and therapeutic application. J
Hematol Oncol. 7:872014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Baum B, Settleman J and Quinlan MP:
Transitions between epithelial and mesenchymal states in
development and disease. Semin Cell Dev Biol. 19:294–308. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Baldin V, Lukas J, Marcote MJ, Pagano M
and Draetta G: Cyclin D1 is a nuclear protein required for cell
cycle progression in G1. Genes Dev. 7:812–821. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Thiery JP: Epithelial-mesenchymal
transitions in tumour progression. Nat Rev Cancer. 2:442–454. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ferrarotto R, Goonatilake R, Yoo Young S,
Tong P, Giri U, Peng S, Minna J, Girard L, Wang Y, Wang L, et al:
Epithelial-mesenchymal transition predicts polo-like kinase 1
inhibitor-mediated apoptosis in non-small cell lung cancer. Clin
Cancer Res. 22:1674–1686. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Matsubara D, Kishaba Y, Yoshimoto T,
Sakuma Y, Sakatani T, Tamura T, Endo S, Sugiyama Y, Murakami Y and
Niki T: Immunohistochemical analysis of the expression of
E-cadherin and ZEB1 in non-small cell lung cancer. Pathol Int.
64:560–568. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Merikallio H, Turpeenniemi-Hujanen T,
Pääkkö P, Mäkitaro R, Riitta K, Salo S, Salo T, Harju T and Soini
Y: Snail promotes an invasive phenotype in lung carcinoma. Respir
Res. 13:1042012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu CW, Li CH, Peng YJ, Cheng YW, Chen HW,
Liao PL, Kang JJ and Yeng MH: Snail regulates Nanog status during
the epithelial-mesenchymal transition via the Smad1/Akt/GSK3β
signaling pathway in non-small-cell lung cancer. Oncotarget.
5:3880–3894. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
van der Heide LP, Wijchers PJ, von Oerthel
L, Burbach JP, Hoekman MF and Smidt MP: FoxK2 is required for
cellular proliferation and survival. J Cell Physiol. 230:1013–1023.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ji Z, Donaldson IJ, Liu J, Hayes A, Zeef
LA and Sharrocks AD: The forkhead transcription factor FOXK2
promotes AP-1-mediated transcriptional regulation. Mol Cell Biol.
32:385–398. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Argast GM, Krueger JS, Thomson S,
Sujka-Kwok I, Carey K, Silva S, O'Connor M, Mercado P, Mulford IJ,
Young GD, et al: Inducible expression of TGFβ, snail and Zeb1
recapitulates EMT in vitro and in vivo in a NSCLC model. Clin Exp
Metastasis. 28:593–614. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Su CL, Deng TR, Shang Z and Xiao Y: JARID2
inhibits leukemia cell proliferation by regulating CCND1
expression. Int J Hematol. 102:76–85. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gu H, Yang T, Fu S, Chen X, Guo L and Ni
Y: MicroRNA-490-3p inhibits proliferation of A549 lung cancer cells
by targeting CCND1. Biochem Biophys Res Commun. 444:104–108. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liang YW, Chang CC, Hung CM, Chen TY,
Huang TY and Hsu YC: Preclinical activity of simvastatin induces
cell cycle arrest in G1 via blockade of cyclin D-Cdk4 expression in
non-small cell lung cancer (NSCLC). Int J Mol Sci. 14:5806–5816.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xia Y, Wu Y, Liu B, Wang P and Chen Y:
Downregulation of miR-638 promotes invasion and proliferation by
regulating SOX2 and induces EMT in NSCLC. FEBS Lett. 588:2238–2245.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang JG, Wang JJ, Zhao F, Liu Q, Jiang K
and Yang GH: MicroRNA-21 (miR-21) represses tumor suppressor PTEN
and promotes growth and invasion in non-small cell lung cancer
(NSCLC). Clin Chim Acta. 411:846–852. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang Y, Xu L and Jiang L: miR-1271
promotes non-small-cell lung cancer cell proliferation and invasion
via targeting HOXA5. Biochem Biophys Res Commun. 458:714–719. 2015.
View Article : Google Scholar : PubMed/NCBI
|