|
1
|
Cid-Arregui A and Juarez V: Perspectives
in the treatment of pancreatic adenocarcinoma. World J
Gastroenterol. 21:9297–9316. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Majumder S, Chari ST and Ahlquist DA:
Molecular detection of pancreatic neoplasia: Current status and
future promise. World J Gastroenterol. 21:11387–11395. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gresham GK, Wells GA, Gill S, Cameron C
and Jonker DJ: Chemotherapy regimens for advanced pancreatic
cancer: A systematic review and network meta-analysis. BMC Cancer.
14:4712014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
de Sousa Cavalcante L and Monteiro G:
Gemcitabine: Metabolism and molecular mechanisms of action,
sensitivity and chemoresistance in pancreatic cancer. Eur J
Pharmacol. 741:8–16. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Burris HA III, Moore MJ, Andersen J, Green
MR, Rothenberg ML, Modiano MR, Cripps MC, Portenoy RK, Storniolo
AM, Tarassoff P, et al: Improvements in survival and clinical
benefit with gemcitabine as first-line therapy for patients with
advanced pancreas cancer: A randomized trial. J Clin Oncol.
15:2403–2413. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Karnitz LM, Flatten KS, Wagner JM,
Loegering D, Hackbarth JS, Arlander SJ, Vroman BT, Thomas MB, Baek
YU, Hopkins KM, et al: Gemcitabine-induced activation of checkpoint
signaling pathways that affect tumor cell survival. Mol Pharmacol.
68:1636–1644. 2005.PubMed/NCBI
|
|
7
|
Voutsadakis IA: Molecular predictors of
gemcitabine response in pancreatic cancer. World J Gastrointest
Oncol. 3:153–164. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Qin L, Dong Z and Zhang JT: 14-3-3σ
regulation of and interaction with YAP1 in acquired gemcitabine
resistance via promoting ribonucleotide reductase expression.
Oncotarget. 7:17726–17736. 2016.PubMed/NCBI
|
|
9
|
Maréchal A and Zou L: DNA damage sensing
by the ATM and ATR kinases. Cold Spring Harb Perspect Biol.
5:52013. View Article : Google Scholar
|
|
10
|
Couch FB, Bansbach CE, Driscoll R, Luzwick
JW, Glick GG, Bétous R, Carroll CM, Jung SY, Qin J, Cimprich KA, et
al: ATR phosphorylates SMARCAL1 to prevent replication fork
collapse. Genes Dev. 27:1610–1623. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sørensen CS and Syljuåsen RG: Safeguarding
genome integrity: The checkpoint kinases ATR CHK1 and WEE1 restrain
CDK activity during normal DNA replication. Nucleic Acids Res.
40:477–486. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Buisson R, Boisvert JL, Benes CH and Zou
L: Distinct but concerted roles of ATR DNA-PK, and Chk1 in
countering replication stress during S phase. Mol Cell.
59:1011–1024. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jackson SP and Bartek J: The DNA-damage
response in human biology and disease. Nature. 461:1071–1078. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Farmer H, McCabe N, Lord CJ, Tutt AN,
Johnson DA, Richardson TB, Santarosa M, Dillon KJ, Hickson I,
Knights C, et al: Targeting the DNA repair defect in BRCA mutant
cells as a therapeutic strategy. Nature. 434:917–921. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kwok M, Davies N, Agathanggelou A, Smith
E, Petermann E, Yates E, Brown J, Lau A and Stankovic T: Synthetic
lethality in chronic lymphocytic leukaemia with DNA damage response
defects by targeting the ATR pathway. Lancet. 385 Suppl 1:S582015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fokas E, Prevo R, Hammond EM, Brunner TB,
McKenna WG and Muschel RJ: Targeting ATR in DNA damage response and
cancer therapeutics. Cancer Treat Rev. 40:109–117. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Weber AM and Ryan AJ: ATM and ATR as
therapeutic targets in cancer. Pharmacol Ther. 149:124–138. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Prevo R, Fokas E, Reaper PM, Charlton PA,
Pollard JR, McKenna WG, Muschel RJ and Brunner TB: The novel ATR
inhibitor VE-821 increases sensitivity of pancreatic cancer cells
to radiation and chemotherapy. Cancer Biol Ther. 13:1072–1081.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fokas E, Prevo R, Pollard JR, Reaper PM,
Charlton PA, Cornelissen B, Vallis KA, Hammond EM, Olcina MM,
McKenna W Gillies, et al: Targeting ATR in vivo using the novel
inhibitor VE-822 results in selective sensitization of pancreatic
tumors to radiation. Cell Death Dis. 3:e4412012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Foote KM, Blades K, Cronin A, Fillery S,
Guichard SS, Hassall L, Hickson I, Jacq X, Jewsbury PJ, McGuire TM,
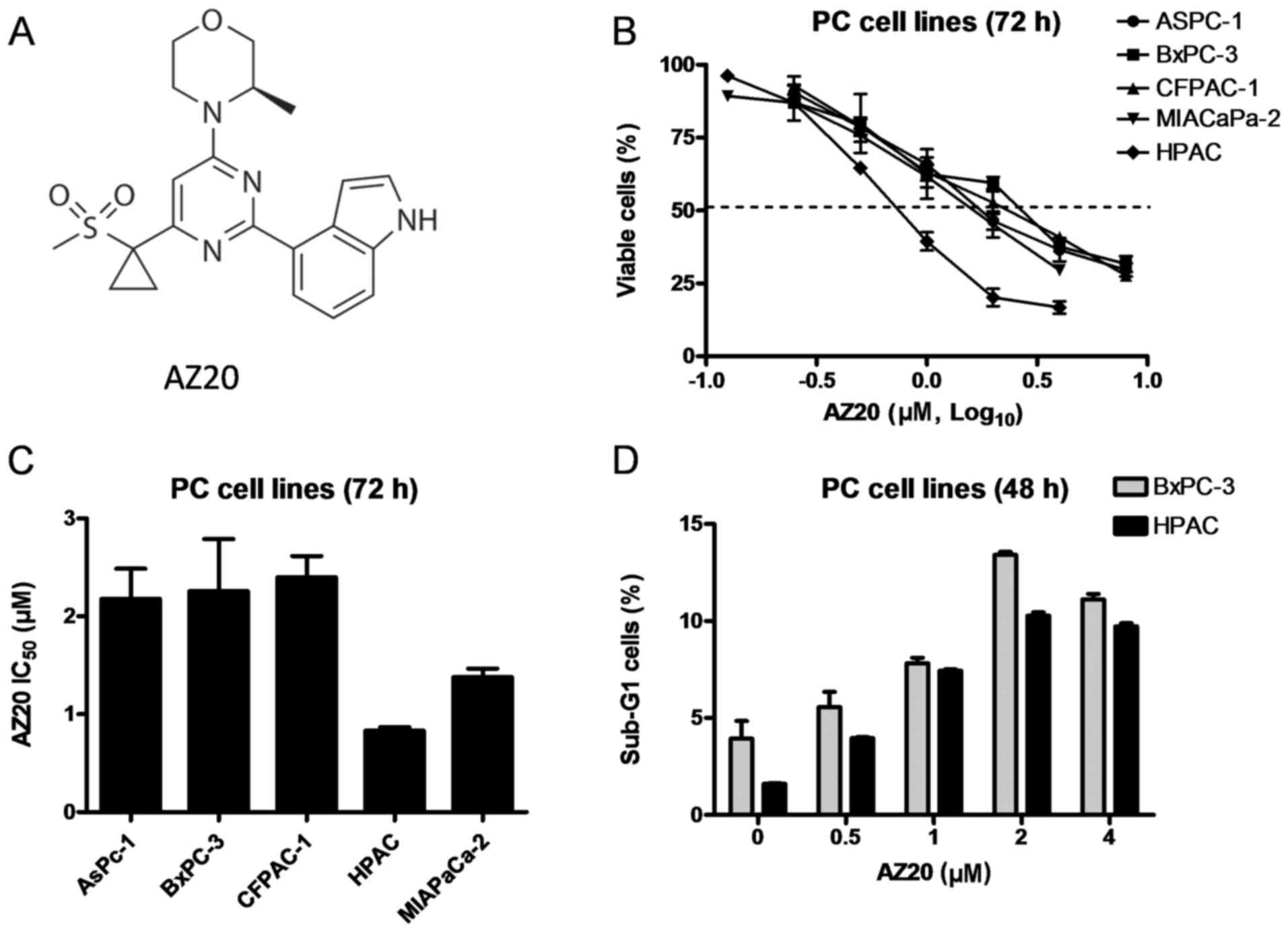
et al: Discovery of
4-{4-[(3R)-3-Methylmorpholin-4-yl]-6-[1-(methylsulfonyl)cyclopropyl]pyrimidin-2-yl}-1H-indole
(AZ20): A potent and selective inhibitor of ATR protein kinase with
monotherapy in vivo antitumor activity. J Med Chem. 56:2125–2138.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nghiem P, Park PK, Kim Y, Vaziri C and
Schreiber SL: ATR inhibition selectively sensitizes G1
checkpoint-deficient cells to lethal premature chromatin
condensation. Proc Natl Acad Sci USA. 98:pp. 9092–9097. 2001;
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mukhopadhyay UK, Senderowicz AM and
Ferbeyre G: RNA silencing of checkpoint regulators sensitizes
p53-defective prostate cancer cells to chemotherapy while sparing
normal cells. Cancer Res. 65:2872–2881. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Toledo LI, Murga M, Zur R, Soria R,
Rodriguez A, Martinez S, Oyarzabal J, Pastor J, Bischoff JR and
Fernandez-Capetillo O: A cell-based screen identifies ATR
inhibitors with synthetic lethal properties for cancer-associated
mutations. Nat Struct Mol Biol. 18:721–727. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ruzankina Y, Schoppy DW, Asare A, Clark
CE, Vonderheide RH and Brown EJ: Tissue regenerative delays and
synthetic lethality in adult mice after combined deletion of Atr
and Trp53. Nat Genet. 41:1144–1149. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang G, Niu X, Zhang W, Caldwell JT,
Edwards H, Chen W, Taub JW, Zhao L and Ge Y: Synergistic antitumor
interactions between MK-1775 and panobinostat in preclinical models
of pancreatic cancer. Cancer Lett. 356:656–668. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xie C, Drenberg C, Edwards H, Caldwell JT,
Chen W, Inaba H, Xu X, Buck SA, Taub JW, Baker SD, et al:
Panobinostat enhances cytarabine and daunorubicin sensitivities in
AML cells through suppressing the expression of BRCA1, CHK1, and
Rad51. PLoS One. 8:e791062013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Redon CE, Nakamura AJ, Zhang YW, Ji JJ,
Bonner WM, Kinders RJ, Parchment RE, Doroshow JH and Pommier Y:
Histone gammaH2AX and poly(ADP-ribose) as clinical pharmacodynamic
biomarkers. Clin Cancer Res. 16:4532–4542. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jossé R, Martin SE, Guha R, Ormanoglu P,
Pfister TD, Reaper PM, Barnes CS, Jones J, Charlton P, Pollard JR,
et al: ATR inhibitors VE-821 and VX-970 sensitize cancer cells to
topoisomerase i inhibitors by disabling DNA replication initiation
and fork elongation responses. Cancer Res. 74:6968–6979. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kastan MB, Zhan Q, el-Deiry WS, Carrier F,
Jacks T, Walsh WV, Plunkett BS, Vogelstein B and Fornace AJ Jr: A
mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is
defective in ataxia-telangiectasia. Cell. 71:587–597. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
D'Angiolella V, Donato V, Forrester FM,
Jeong YT, Pellacani C, Kudo Y, Saraf A, Florens L, Washburn MP and
Pagano M: Cyclin F-mediated degradation of ribonucleotide reductase
M2 controls genome integrity and DNA repair. Cell. 149:1023–1034.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jordheim LP, Sève P, Trédan O and Dumontet
C: The ribonucleotide reductase large subunit (RRM1) as a
predictive factor in patients with cancer. Lancet Oncol.
12:693–702. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhou J, Chen Z, Malysa A, Li X, Oliveira
P, Zhang Y and Bepler G: A kinome screen identifies checkpoint
kinase 1 (CHK1) as a sensitizer for RRM1-dependent gemcitabine
efficacy. PLoS One. 8:e580912013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rahman MA, Amin ARMR, Wang D, Koenig L,
Nannapaneni S, Chen Z, Wang Z, Sica G, Deng X, Chen ZG, et al: RRM2
regulates Bcl-2 in head and neck and lung cancers: A potential
target for cancer therapy. Clin Cancer Res. 19:3416–3428. 2013.
View Article : Google Scholar : PubMed/NCBI
|