Introduction
Polycystic ovary syndrome (PCOS) is a common
endocrine disease affecting 5–20% of women of reproductive age
worldwide (1). PCOS is
characterized by high luteinizing hormone levels, polycystic
ovarian morphology, hyperandrogenism, hyperinsulinemia, and insulin
resistance. Excessive androgen production by the ovaries is the
main feature of PCOS (1,2). The prevalence of PCOS has increased
during the past decade, and its adverse effects on the health of
women are becoming more serious (3,4).
Moreover, women with PCOS are at increased risks for cardiovascular
disease and diabetes (5–7). Therefore, when considering its
increasing prevalence, more effective methods for preventing or
treating PCOS are needed. Presently, the pathogenesis of PCOS is
not fully understood, and requires further investigation. More than
50% of PCOS patients are insulin resistant (2,8), which
can partly explain their increased risk for diabetes and
cardiovascular disease. Furthermore, insulin resistance can also be
the cause for hyperlipidemia and hyperinsulinemia in PCOS patients.
Therefore, identifying key factors involved in the development of
insulin resistance should provide us with a better understanding of
PCOS pathogenesis. For example, glucose transporter 4 (GLUT4) is a
protein that plays critical roles in controlling the translocation
of glucose into adipocytes and regulating glucose homeostasis
(9,10). GLUT4 is strongly associated with
insulin sensitivity, and its dysfunction has been reported to cause
insulin resistance in PCOS patients (11–13).
High mobility group A2 (HMGA2), a non-histone chromosomal protein,
is associated with lipomas, suggesting its roles in adipogenesis
and insulin resistance (14,15).
SREBF1, also known as sterol regulatory element-binding protein 1
(SREBP1), is a major regulator of genes involved in metabolic
pathways and sterol biosynthesis (16–18),
and its expression is significantly increased in the endometrium of
PCOS patients (17). More
importantly, the transcripts for the two hsa-miR-33 isoforms (a and
b) are located in a non-coding region of the SREBF gene,
suggesting a potential role for miR-33 in regulating SREBF1
expression (19). However, the
underlying molecular mechanisms that regulate these key factors and
lead to insulin resistance in PCOS patients remain poorly
understood.
MicroRNAs (miRNAs) consist of a group of small
non-coding RNAs ~20-24 nt in length (20). Previous studies have revealed that
miRNAs play key roles in modulating the functions of human bodies
(20–22). miRNAs mainly exert their effects by
binding to the 3′-untranslated regions (3′-UTRs) of target genes
and inhibiting transcription of the mRNAs of those genes (22). Recent studies have proposed that
miRNAs are important gene regulators involved in human diseases,
including endocrine and metabolic diseases (23,24).
Some miRNAs have been found to play critical roles in modulating
glucose homeostasis, and the aberrant expression of those miRNAs
can result in insulin resistance that is often involved in the
development of metabolic diseases (25,26).
Several miRNAs (i.e. miR-93 and miR-320) have been implicated in
the pathogenesis of PCOS (27–32).
However, our understanding of their involvement in the development
of PCOS is limited, and more studies are needed to clarify their
roles in PCOS pathogenesis. In the present study, we examined the
levels of miR-33b-5p expression in the ovarian tissues of PCOS rats
with insulin resistance, and the correlations between miR-33b-5p
levels and the expression of three different proteins: GLUT4, HMGA2
and SREBF1. We also investigated the possible role of miR-33b-5p in
regulating GLUT4, HMGA2 and SREBF1 in cultured adipocytes.
Materials and methods
Animal model of PCOS
Twenty-four female Sprague-Dawley (SD) rats (180–250
g) were obtained from the Center of Laboratory Animal Science,
Hubei, China. The rats were group-housed in a room with a regular
12-h light/dark cycle, and had free access to food and water. All
animals were treated in accordance with the guidelines of the
National Institutes of Health Guide for the Care and Use of
Laboratory Animals, and the study protocol was approved by the
Animal Ethics Committee of Hubei University of Arts and
Science.
The PCOS model was developed in female rats
according to the method described by Poretsky et al
(33). Female rats (70 to 85 days
old) were subcutaneously injected with increasing doses of insulin
(0.5 IU, 5.0 IU and 5.0 IU/day) for the first 10 days, and then
with 5 IU insulin + 1.5 IU HCG from day 11 through day 22. The rats
were fed a high fat diet for 6 weeks, and their weights were
measured weekly. Rats in the control groups were fed a normal diet
every day. Finally, the PCOS model rats displayed typical
characteristics of PCOS, such as hyperinsulinemia, insulin
resistance, weight gain, and polycystic degeneration in the ovaries
(33). Fasting blood glucose and
fasting insulin levels were assessed and the homeostatic model
assessment-insulin resistance (HOMA-IR) of each rat was calculated.
Next, the rats were assigned to four different groups: a
non-PCOS/non-IR group, a non-PCOS/IR group, a PCOS/non-IR group,
and a PCOS/IR group. The rats were then anesthetized with 10%
chloral hydrate, and laparotomies were performed to collect ovarian
tissue specimens for further processing. Insulin resistance was
assessed by the HOMA-IR index, and rats with a HOMA-IR ≥2.5 were
considered insulin resistant (IR).
Hematoxylin and eosin (H&E) and
immunohistochemical staining
The ovaries of each rat were removed, processed, and
sectioned at 4-mm thickness; after which, the sections were stained
with H&E and examined for evidence of morphological changes.
Five sections were obtained from each ovary. Immunohistochemistry
(IHC) was then used to examine the ovarian tissues for SREBF1,
HMGA2 and GLUT4 expression. Tissue sections used for
immunohistochemistry were first deparaffinized and hydrated, and
endogenous peroxidase activity was quenched with 0.3% hydrogen
peroxide in PBS. Next, the tissue sections were incubated in
blocking buffer for 60 min, and then incubated overnight at 4°C
with primary antibodies that included GLUT4 antibody (1:500; rabbit
polyclonal; cat. no. PB0143; Boster Biological Technology, Wuhan,
China), SREBF1 antibody (1:500; rabbit polyclonal; cat. no.
14088–1-AP; Proteintech; Wuhan Sanying Biotechnology, Wuhan,
China), and HMGA2 antibody (1:500; rabbit polyclonal; cat. no.
bs-0556R; Bioss Antibodies, Beijing, China). They were then
incubated with horseradish peroxidase-conjugated secondary
antibodies [1:5,000; goat anti-rabbit IgG H&L (HRP); cat. no.
ab205718; Abcam, Cambridge, MA, USA] as per the manufacturer's
instructions. Positively-labeled cells were visualized using a
streptavidin-HRP conjugate and DAB chromogen. The tissue sections
were then counterstained with hematoxylin, dehydrated, and mounted
in DPX.
Culturing of human adipocytes in high
glucose medium
Human adipocyte cells (ScienCell Research
Laboratories, Inc., San Diego, CA, USA) were incubated with
adipocyte maintenance medium at 37°C in a humidified incubator
containing 5–10% CO2. The adipocyte cells were then
cultured in normal adipocyte maintenance medium (control group) or
treated with different concentrations of high-glucose adipocyte
maintenance medium (Gibco; Thermo Fisher Scientific, Inc., Waltham,
MA, USA) (0, 3.15, 9.45 or 28.35 g/l) for 24 h. After culture, the
adipocytes were collected and their levels of miR-33b-5p, SREBF1,
HMGA2 and GLUT4 were examined using real-time polymerase chain
reaction (PCR) or and western blotting methods.
Culturing of human adipocytes in
different concentrations of insulin
Human adipocytes were incubated with adipocyte
maintenance medium at 37°C in a humidified incubator containing
5–10% CO2. The adipocyte cells were then treated with
different doses of insulin (Sigma-Aldrich, Merck KGaA, Darmstadt,
Germany) (0, 5, 50 or 500 nM) for 24 h; after which, they were
collected and tested for their levels of miR-33b-5p, SREBF1, HMGA2
and GLUT4 expression using real-time PCR or western blotting
methods.
Treatment of human adipocytes with
different concentrations of miR-33b-5p inhibitor
Human adipocytes were cultured as aforementioned;
after which, they were seeded into 6-well plates and grown to
60–80% confluence for use in transfection studies. A 9-µl volume of
Lipofectamine 2000 was mixed with 150 µl of Opti-MEM medium (both
from Thermo Fisher Scientific, Inc.). A miR-33b-5p inhibitor or NC
inhibitor (Shanghai JiKai Gene Chemical Technology Co., Ltd.,
Shanghai, China) was diluted with Opti-MEM medium and then mixed
with Lipofectamine-Opti-MEM medium at a ratio of 1:1 to achieve
miR-33b-5p inhibitor concentrations of 0, 25, 50 and 100 nM,
respectively. The sequence of miR-33b-5p inhibitor:
5′-GCAAUGCAACAGCAAUGCAC-3′. After allowing the cells to incubate at
room temperature for 5 min, the miR-33b-5p inhibitor was added, and
the cells were incubated with the different concentrations of
miR-inhibitor complex for 48 h. Next, the adipocytes were
collected, and their levels of miR-33b-5p, SREBF1, HMGA2 and GLUT4
were determined using real-time PCR or western blotting
methods.
RNA extraction and real-time PCR
A miRACLE isolation kit (Jinfiniti Biotech, LLC,
Augusta, GA, USA) was used to extract total RNA from the ovarian
tissues of rats and cultured adipocytes. The various cDNAs of mRNAs
or miRNAs were synthesized using a cDNA reverse transcription kit.
Real-time PCR was carried out using SYBR-Green Supermix and an
Applied Biosystems 7300 Real-Time PCR System (Applied Biosystems;
Thermo Fisher Scientific, Inc.). The primers used for PCR are
listed in Table I. U6 and
GAPDH were used as internal control genes. Real-time PCR was
performed on target genes under the following conditions: 35 cycles
each containing 94°C for 30 sec, 58°C for 45 sec and 72°C for 35
sec. The relative levels of mRNA and miR-33b-5p were calculated
using the 2−ΔΔCT method.
 | Table I.PCR primer information. |
Table I.
PCR primer information.
| ID | Sequence (5–3) | Product length
(bp) |
|---|
| U6 F |
CTCGCTTCGGCAGCACA |
|
| U6 R |
AACGCTTCACGAATTTGCGT |
|
| r-GAPDH F |
CCTCGTCTCATAGACAAGATGGT | 169 |
| r-GAPDH R |
GGGTAGAGTCATACTGGAACATG |
|
| h-GAPDH F |
TGTTCGTCATGGGTGTGAAC | 154 |
| h-GAPDH R |
ATGGCATGGACTGTGGTCAT |
|
| r-miR-33-5p RT |
CTCAACTGGTGTCGTGGAGTC
GGCAATTCAGTTGAGTGCAATGC |
|
| r-miR-33-5p F |
ACACTCCAGCTGGGGTGCATTGTAGTTGCAT |
|
| h-miR-33b-5p
RT | CTCAACTGGTGTCGT
GGAGTCGGCAATTCAGTTGAGGCAATGCA |
|
| h-miR-33b-5p F |
ACACTCCAGCTGGGGTGCATTGCTGTTGCA |
|
| ALL R |
CTCAACTGGTGTCGTGGA |
|
| r-SREBF1 F |
TTACAGCACAGCAACCAGAA | 132 |
| r-SREBF1 R |
CATGCCCTCCATAGACACAT |
|
| h-SREBF1 F |
GCAACACAGCAACCAGAAACT | 191 |
| h-SREBF1 R |
GGAAAGGTGAGCCAGCATC |
|
| r-HMGA2 F |
CCTGTGAGCCCTCTCCTAA | 107 |
| r-HMGA2 R |
CCGTTTTTCTCCAATGGTC |
|
| h-HMGA2 F |
AGCAGCAGCAAGAACCAAC | 203 |
| h-HMGA2 R |
CCCAGGCAAGGCAACAT |
|
| r-GLUT4 F |
TGGTTGGGAAGGAAAAGG | 191 |
| r-GLUT4 R |
AGATGAGTGGGGGCGAT |
|
| h-GLUT4 F |
GCCCCCGCTACCTCTACAT | 363 |
| h-GLUT4 R |
CCGCTCCACCAACAAC |
|
Western blotting studies
Proteins were extracted from cultured adipocytes,
and then separated using 10% SDS-PAGE. The separated protein bands
were transferred onto a PVDF membrane, which was then probed with
rabbit monoclonal antibodies against SREBF1 (1:500; rabbit
polyclonal; cat. no. 14088-1-AP; Proteintech); HMGA2 (1:1,000;
rabbit polyclonal; cat. no. bs-0556R; Bioss Antibodies); GLUT4
(1:1,000; rabbit polyclonal; cat. no. PB0143; Boster Biological
Technology) or GAPDH (1:2,000; mouse monoclonal; cat. no. ab8245;
Abcam) as an internal control overnight at 4°C. After incubation at
37°C for 1 h with horseradish peroxidase-conjugated secondary
antibodies (1:5,000; goat polyclonal to HRP; cat. no. ab181658;
Abcam), an Immobilon™ Western Chemiluminescent ECL kit was used to
detect bound antibodies. Protein staining intensity was assessed
using Image-Pro Plus 6.0 software.
Luciferase reporter assay
Wild-type and mutant type HMGA2 3-UTRs were
amplified and inserted into psiCHECK2 luciferase vectors (Promega
Corporation, Madison, WI, USA). Next, the vectors with either the
miR-33b-5p mimics or negative mimics (GenePharma Co., Ltd.,
Shanghai, China) (1.5 mg) were used to transfect 293T cells.
Following 48 h of transfection, cellular luciferase activity was
detected with a Microplate reader (Tecan GENios Pro; Tecan Group,
Ltd., Mannedorf, Switzerland) according to the manufacturer's
protocol.
HMG2 transcription factor binding sites for GLUT4
and SREBF1 5′-promoter luciferase vectors were amplified and
inserted into pGL3 luciferase vectors (Promega Corporation). Next,
the vectors with either the pcDNA3.0-HMGA2 overexpression plasmid
or empty plasmid (0, 0.5, 1.5, 2.5 and 5.0 µg) were transfected
into 293T cells. Luciferase activity was examined after 48 h of
transfection. SREBF1 transcription factor binding sites were used
to establish GLUT4 5′-promoter wild-type and mutant type luciferase
vectors. Vectors with either the pcDNA3.0-SREBF1 overexpression
plasmid or empty plasmid were transfected into 293T cells, and
cellular luciferase activity was assessed.
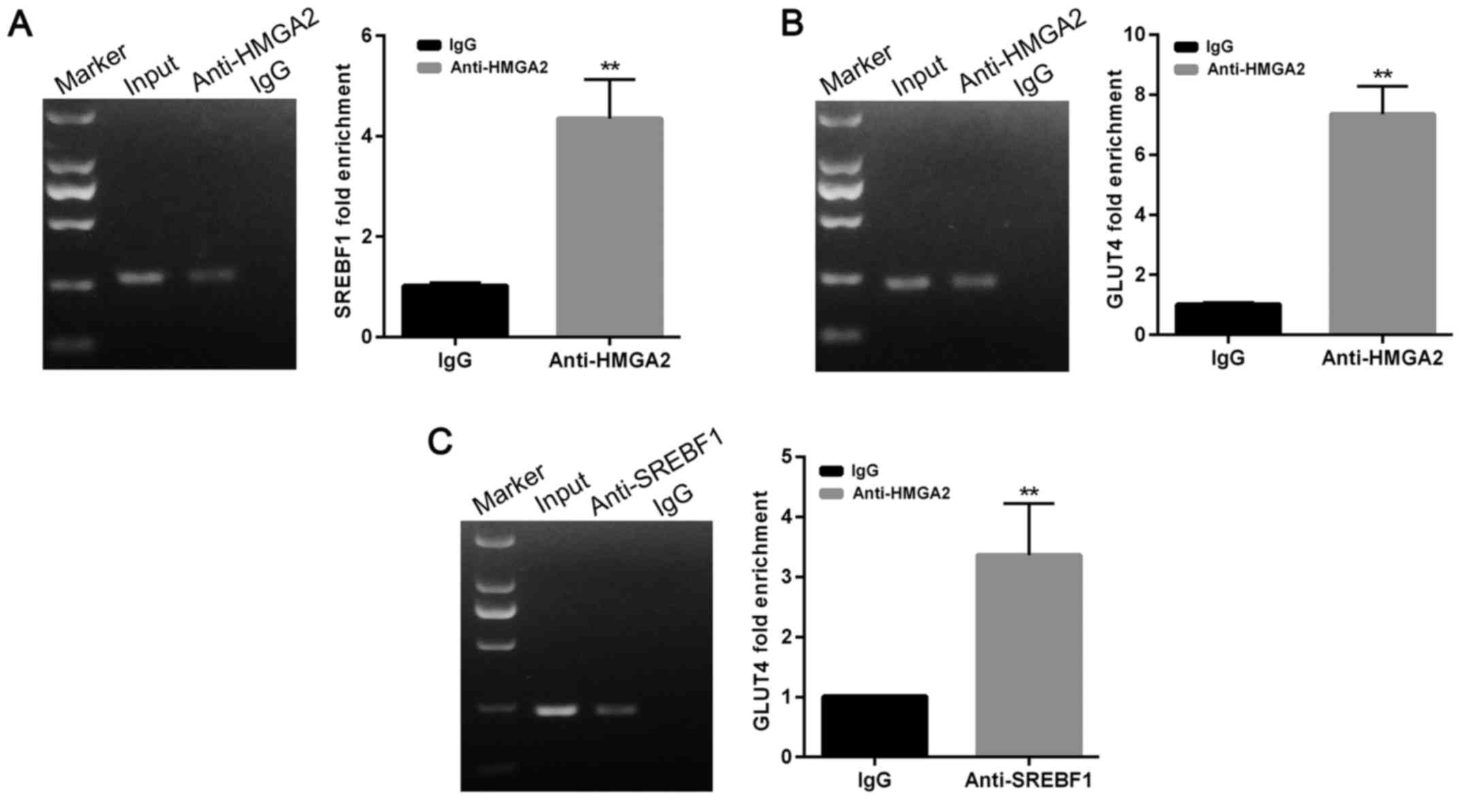
Chromatin immunoprecipitation
(ChIP)
To analyze the binding of SREBF1/HMGA2 protein to
the GLUT4 gene promoter, adipocyte cells transfected with
either SREBF1 or HMGA2 antibodies (Abcam) were used for ChIP
analyses that were performed using a Pierce Agarose Chip Kit
(EpiGentek Group, Inc., Farmingdale, NY, USA). Normal rabbit IgG
was used as a negative control. Non-precipitated genomic DNA input
was amplified as an input control. After purification, the DNAs
were used for PCR analyses that were performed using primers
(Sangon Biotech Co., Ltd., Shanghai, China) that encompassed the
GLUT4 promoter region. The conditions used for PCR were as
follows: denaturation at 95°C for 2 min, followed by 40 cycles at
95°C for 20 sec, 58°C for 20 sec, and 72°C for 20 sec.
Statistical analysis
All statistical analyses were performed using IBM
SPSS Statistics for Windows, version 19.0 (IBM Corporation, Armonk,
NY, USA). The levels of miR-33b-5p and gene expression are shown as
the mean ± SD, and differences between groups were analyzed using
Student's t-test. One-way ANOVA and Tukey's test were used to
evaluate the levels of miR-33b-5p and gene expression in human
adipocytes treated with different concentrations of glucose or
insulin. The association between miR-33b-5p and SREBF1/HMGA2
expression was assessed using Pearson's correlation test. P-values
<0.05 were regarded as statistically significant.
Results
Expression of miR-33b-5p, GLUT4,
SREBF1 and HMGA2 in the ovarian tissues of PCOS rats
The PCOS rat model was successfully developed using
the method described by Poretsky et al (33). Insulin resistance in PCOS rats was
indicated by their significantly increased insulin resistance index
(HOMA-IR) values when compared with those of control rats (Table II). Based on their HOMA-IR score,
the rats were then further classified into four groups: a
non-PCOS/non-IR group, a non-PCOS/IR group, a PCOS/non-IR group,
and a PCOS/IR group. The levels of miR-33b-5p expression in the
ovarian tissues of non-PCOS/IR rats were significantly higher than
those in the ovarian tissues of non-PCOS/non-IR rats (P<0.05).
The PCOS/IR group also exhibited upregulated levels of miR-33b-5p
expression when compared with the PCOS/non-IR group (Fig. 1A). Conversely, the levels of HMGA2,
SREBF1, and GLUT4 expression in the ovarian tissues of non-PCOS/IR
rats were significantly lower than those in the non-PCOS/non-IR
rats (both P<0.05). PCOS/IR rats had lower HMG2 and GLUT4 levels
when compared with PCOS-non-IR rats (Fig. 1B-D). Furthermore, the levels of HMG2
and GLU4 expression were lower in the PCOS/non-IR group than in the
non-PCOS/non-IR group. miR-33b-5p levels were negatively associated
with SREBF1 and HMGA2 expression in the ovarian tissues of rats in
all four groups (P<0.01), and miR-33b-5p levels were negatively
correlated with GLUT4 levels (P<0.01) (Fig. 1E-G).
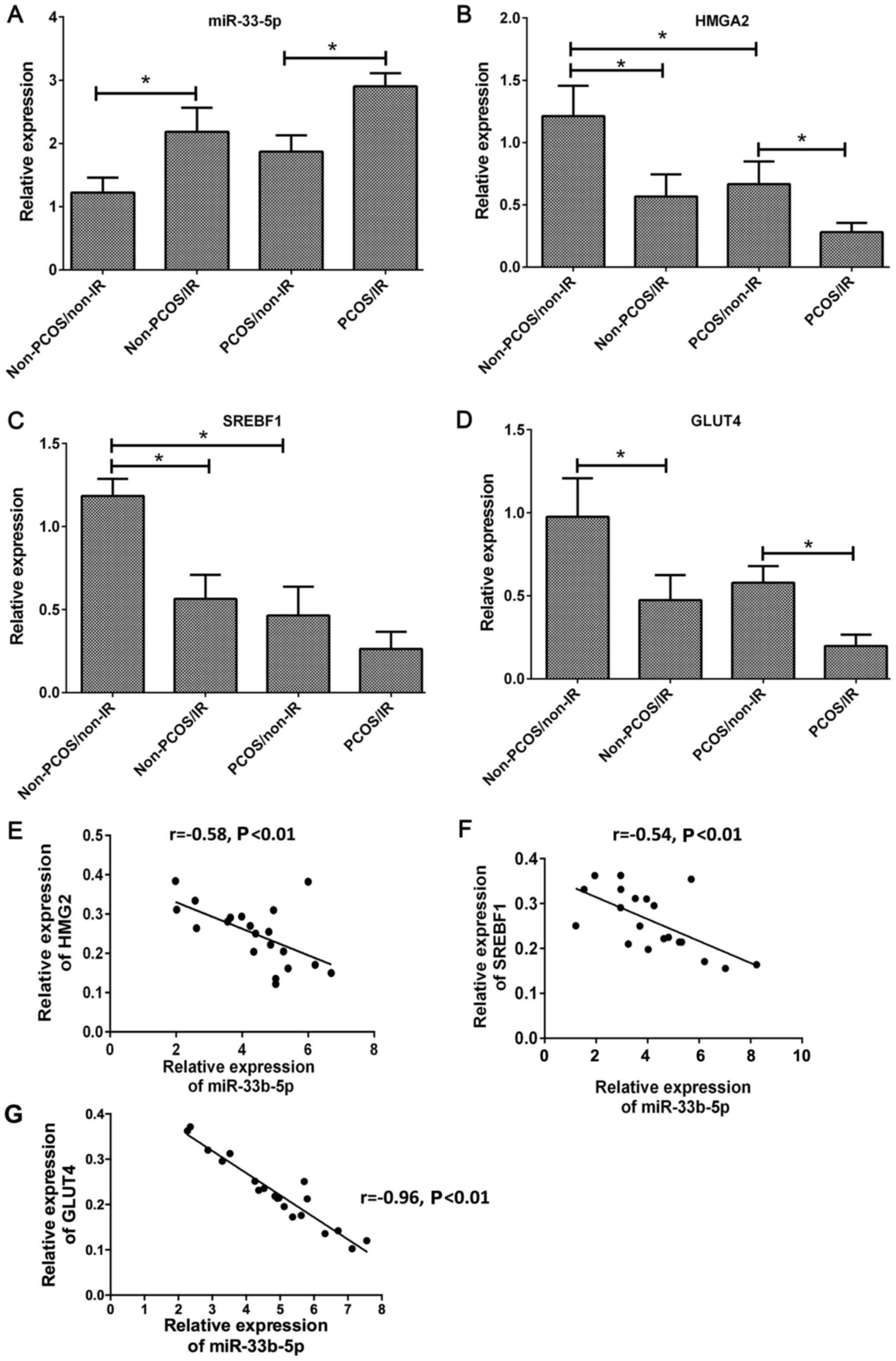 | Figure 1.(A-D) The RNA levels and correlations
of miR-33b-5p, GLUT4, SREBF1, and HMGA2 in rat ovarian tissues from
the non-PCOS/non-IR group, non-PCOS/IR group, PCOS/non-IR group,
and PCOS/IR group (U6 and GAPDH were used as internal controls for
miR-33b-5p and the genes aforementioned, respectively. Relative
expression levels of mRNAs and miRNAs were calculated using the
2−ΔΔCT method). (E-G) The respective correlations
between miR-33b-5p and HMGA2, SREBF1 and GLUT4 in rat ovarian
tissues, *P<0.05. PCOS, polycystic ovary syndrome; IR, insulin
resistance. |
 | Table II.Serum fasting glucose and fasting
insulin levels and HOMA-IR index in the PCOS rats. |
Table II.
Serum fasting glucose and fasting
insulin levels and HOMA-IR index in the PCOS rats.
| Groups | N | Fasting glucose
(mmol/l) | Fasting insulin
(mIU/l) | HOMA-IR |
|---|
| PCOS | 10 |
7.05±0.93a |
27.6±2.3a |
7.5±2.7a |
| Control | 10 | 5.51±0.58 | 11.6±1.9 |
2.7±0.3a |
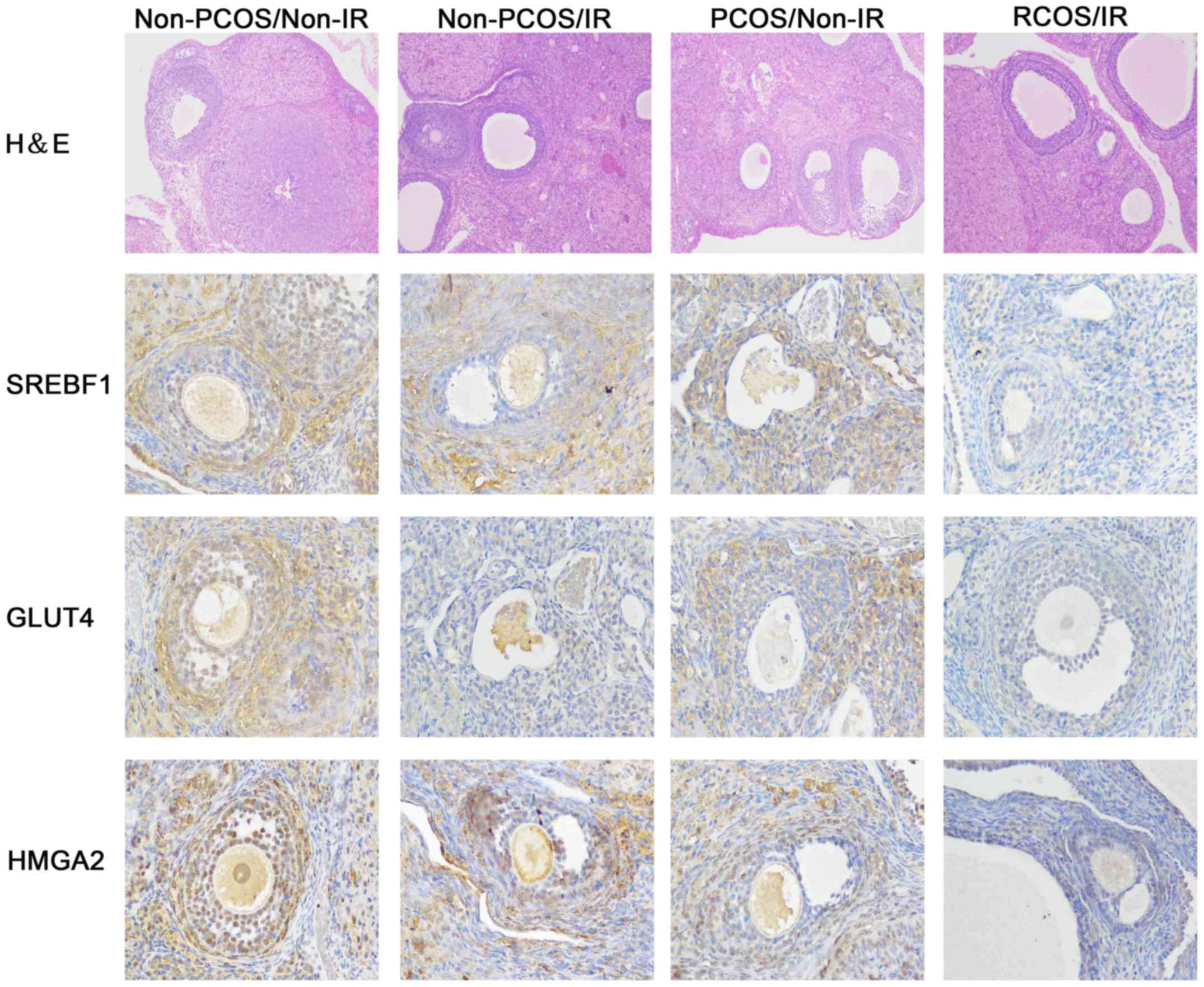
H&E staining of rat ovarian tissues revealed
that ovary oocytes in the non-PCOS/non-IR group were clear, and
large numbers of granulosa cells were present. Ovarian tissues in
the other three groups contained significantly fewer granulosa
cells. The granulosa cells were loosely arranged, and the cortical
stromal cells displayed obvious signs of hyperplasia (Fig. 2). Furthermore, immunohistochemistry
was conducted to detect and compare the expression of GLUT4, SREBF1
and HMGA2, which validated the data by real-time PCR in Fig. 1. Consistently, obvious positive
expression of GLUT4, SREBF1 and HMGA2 (yellow color) were found in
non-PCOS/non-IR group compared to that in the other three groups.
Immunohistochemistry data revealed that GLUT4, SREBF1 and HMGA2
were all highly expressed in the ovarian tissues of rats in the
non-PCOS/non-IR group, however, their expression levels were
significantly decreased in the PCOS/IR group (Fig. 2).
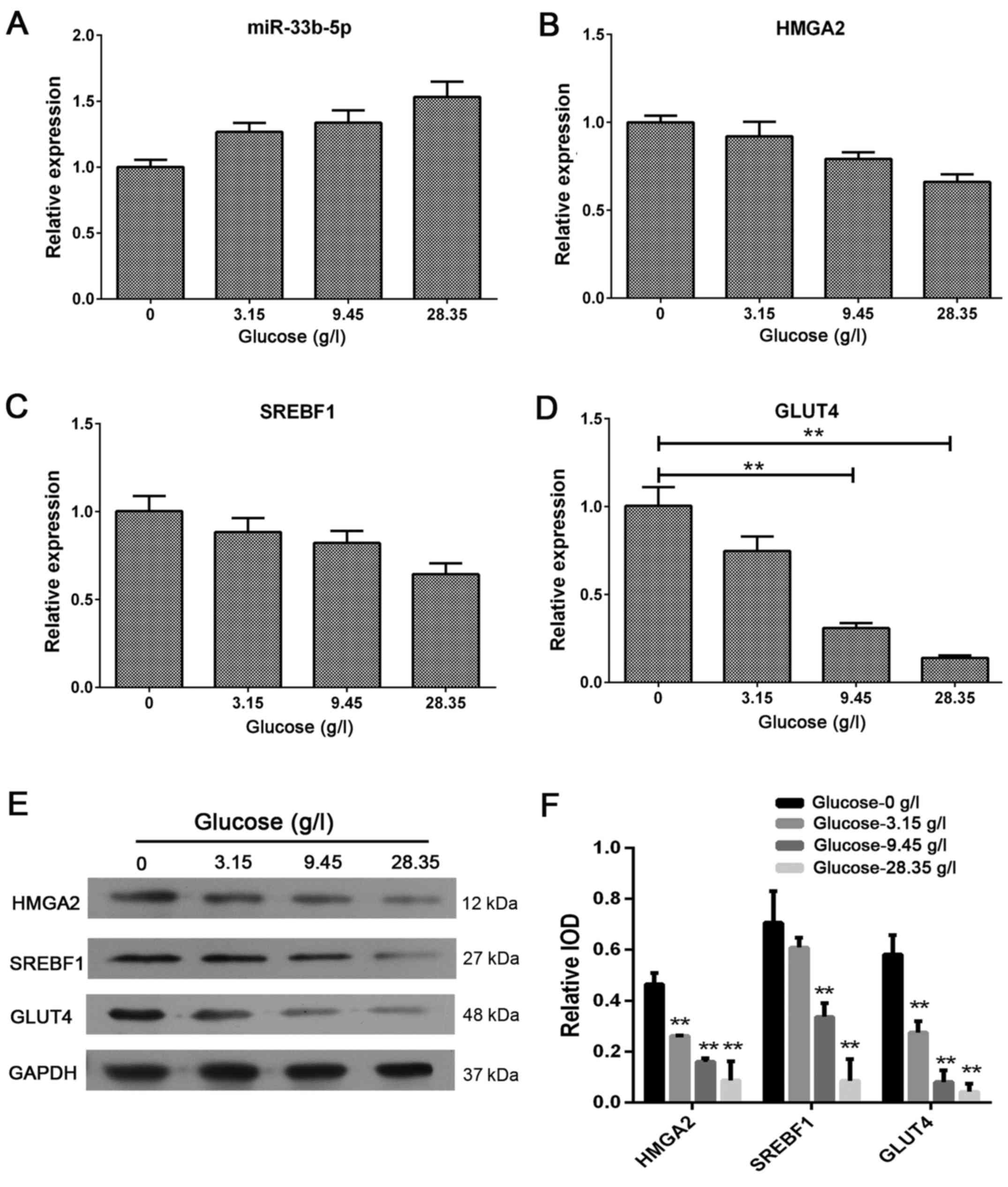
Excess extracellular glucose increases
miR-33b-5p expression and reduces GLUT4, SREBF1 and HMGA2
expression
As shown in Fig. 3,
excess extracellular glucose increased miR-33b-5p mRNA expression
and reduced GLUT4, SREBF1, and HMGA2 expression in adipocytes
(Fig. 3A-D). These findings were
further supported by western blotting data (Fig. 3E-F). These results revealed that
excess glucose can alter the level of miR-33b-5p expression, which
in turn, may further alter the expression of mRNAs for GLUT4,
SREBF1 and HMGA2 in adipocytes.
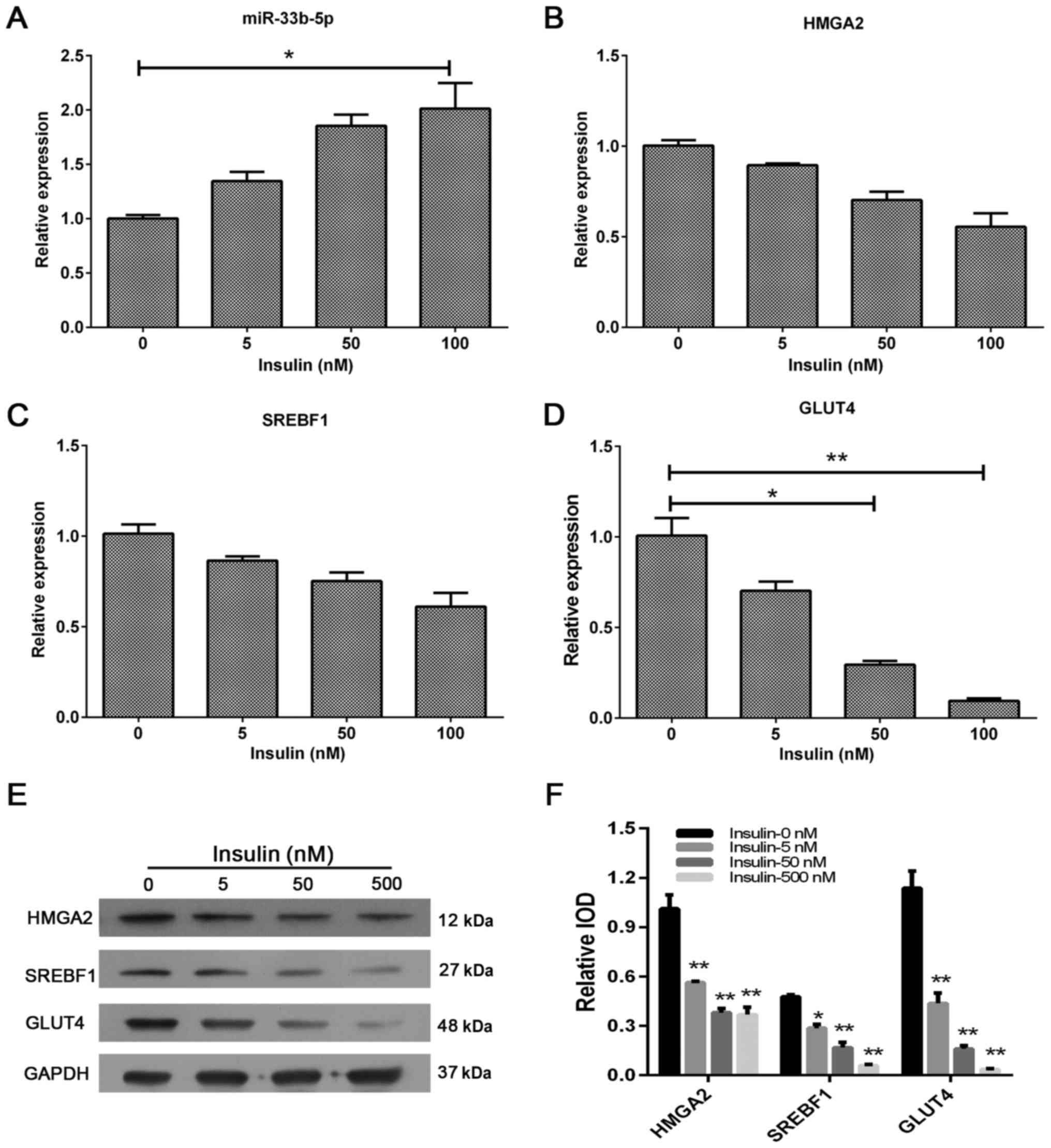
Excess extracellular insulin increases
miR-33b-5p expression and reduces the levels of GLUT4, SREBF1 and
HMGA2 expression
We also assessed the effect of extracellular insulin
on miR-33b-5p, GLUT4, SREBF1 and HMGA2 expression in adipocytes. We
found that insulin produced a similar effect as glucose; it
upregulated miR-33b-5p levels and downregulated GLUT4, SREBF1 and
HMGA2 mRNA and protein levels (Fig.
4). These results revealed that GLUT4, SREBF1 and HMGA2
expression in human adipocytes may also be regulated by changes in
miR-33b-5p levels induced by excess insulin.
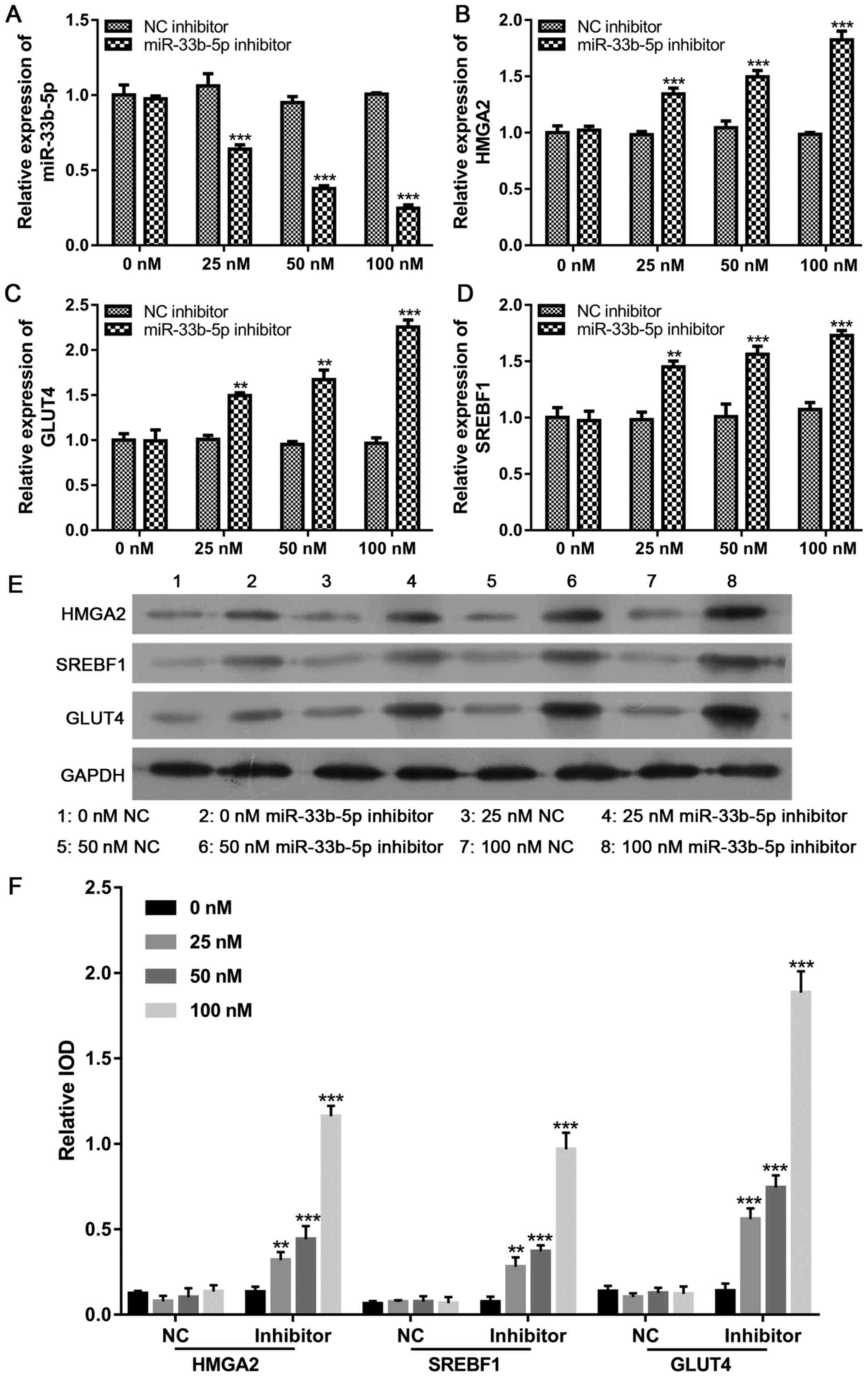
Downregulation of miR-33b-5p induces
GLUT4, SREBF1 and HMGA2 expression
After determining that increased miR-33b-5p levels
produced by treatment with either excess extracellular glucose or
insulin suppressed GLUT4, SREBF1 and HMGA2 expression, we examined
the effect of miR-33b-5p on GLUT4, SREBF1 and HMGA2 levels in
adipocytes by reducing miR-33b-5p expression. Our data revealed
that the expression levels miR-33b-5p gradually decreased as the
concentration of miR-33b-5p inhibitor increased. In contrast, the
GLUT4, SREBF1 and HMGA2 mRNA and protein levels increased in a
concentration-dependent manner, suggesting that miR-33b-5p
negatively regulates the levels of GLUT4, SREBF1 and HMGA2
(Fig. 5).
The role of miR-33b-5p in regulating
HMGA2, SREBF-1 and GLUT4 expression
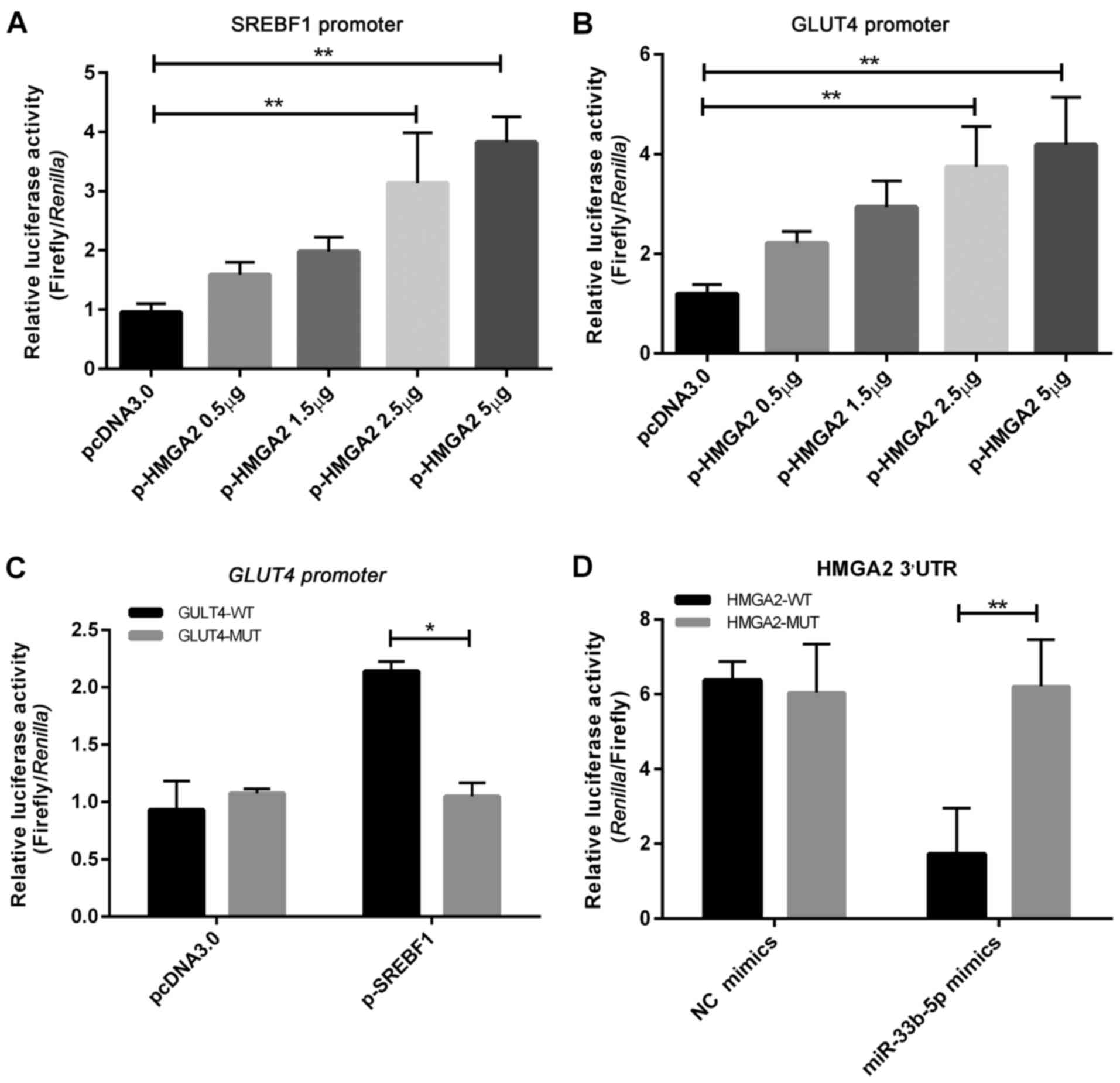
Transient co-transfection of the SREBF1 5′-promoter
luciferase vector with either the pcDNA3.0-HMGA2 overexpression or
empty plasmid revealed that pcDNA3.0-HMGA2 significantly increased
luciferase activity in a dose-dependent manner, suggesting that
HMGA2 could target SREBF1 and promote its expression (Fig. 6A). Chip data (Fig. 7A) revealed that HMGA2 directly binds
to the 5′-promoter region of SREBF1 and promotes its
expression.
Transient co-transfection of the GLUT4 5′-promoter
luciferase vector with either the pcDNA3.0-HMGA2 overexpression or
empty plasmid revealed that pcDNA3.0-HMGA2 also increased
luciferase activity in a dose-dependent manner (Fig. 6B). Chip experiments also
demonstrated that HMGA2 could bind to the 5′-promoter region of
GLUT4 (Fig. 7B). These findings
revealed that HMGA2 can directly bind to the 5′-promoter region of
GLUT4 and promote its expression.
Transient co-transfection of the wild-type GLUT4
5′-promoter or mutant type vectors with either the pcDNA3.0-SREBF1
overexpression or empty plasmid revealed that SREBF1 could
significantly increase luciferase activity in the wild-type GLUT4
5′-promoter group (Fig. 6C). Chip
experiments also demonstrated that SREBF1 could bind to the
5′-promoter region of GLUT4 (Fig. 7C). These findings indicated that
SREBF1 can directly bind to the 5′-promoter region of GLUT4
and promote its expression.
Transient co-transfection of the wild-type
luciferase HMGA2 3′-UTR and mutant vectors with either miR-33b-5p
mimics or NC mimics into 293T cells demonstrated direct binding of
miR-33b-5p to the 3′-UTR of HMGA2, and resulted in significant
reductions in luciferase activity (Fig.
6D). These results revealed that miR-33b-5p could target the
3′-UTR of HMGA2 and inhibit its expression.
Discussion
The present study investigated the role of
miR-33b-5p in PCOS, and particularly its role in regulating GLUT4.
We determined that miR-33b-5p expression was higher in PCOS/IR
rats, and correlated with SREBF1 and HMGA2 expression (P<0.05).
Results of luciferase reporter assays and ChIP studies performed
with adipocytes revealed that miR-33b-5p could target the 3′-UTR of
HMGA2 and inhibit its expression. Moreover, HMGA2 could directly
bind to the 5′-promoter region of GLUT4 and promote its
expression, and could also promote SREBF1 expression. Finally, we
determined that SREBF1 could directly bind to the 5′-promoter
region of GLUT4 and promote its expression. Therefore, our
study results demonstrated that miR-33b-5p was overexpressed in the
ovarian tissues of PCOS/IR rats, and revealed that miR-33b-5p can
inhibit GLUT4 by targeting HMGA2 during the development of
insulin resistance in PCOS patients. Our results also indicated
that HMGA2 and SREBF1 are important molecules involved in
modulating GLUT4 expression.
miRNAs consist of a group of small non-coding RNAs,
some of which play key roles in modulating the functions of human
bodies (25,34,35).
Numerous miRNAs have been demonstrated to be involved in endocrine
and metabolic diseases (36,37),
and several miRNAs (e.g., miR-93 and miR-145) have been reported to
be involved in the pathogenesis of PCOS (27–31,38–42).
miRNA-93 was initially revealed to play an important role in
modulating GLUT4 expression, and thus be involved in the
development of insulin resistance in PCOS patients (27). miR-145 was identified as a molecular
target associated with granulosa cell dysfunction in PCOS, because
it could inhibit IRS1 expression and the MAPK/ERK signaling
pathways (43). The findings
aforementioned revealed that some miRNAs indeed play key roles in
the development of PCOS. In the present study, we found that
miR-33b-5p was highly involved in the development of PCOS, and its
overexpression was observed in our rat model of PCOS. The findings
in our present study provide new additional evidence that miRNAs
play key roles in the development of PCOS.
We further examined how miR-33b-5p affects GLUT4,
SREBF1 and HMGA2 expression. Both excess extracellular glucose and
insulin suppressed the levels of GLUT4, SREBF1 and HMGA2 by
upregulating miR-33b-5p, whereas GLUT4, SREBF1 and HMGA2 levels
became elevated when miR-33b-5p was inhibited, indicating the
powerful effects that glucose and insulin exert in the regulation
of GLUT4, SREBF1 and HMGA2 by miR-33b-5p. Insulin resistance is a
main feature of PCOS, and can adversely affect a woman's health
(8,44,45).
GLUT4 is strongly linked to the development of insulin resistance
in type 2 diabetes or PCOS (46,47).
GLUT4 is a transporter molecule responsible for the uptake of
glucose, and its dysfunction is observed in individuals with
insulin resistance or PCOS (48).
When blood glucose levels are low, GLUT4 is stored intracellularly
in muscle and fat cells, preventing GLUT4 from reaching the cell
surface and transporting glucose into cells (49). When blood glucose levels are high,
GLUT4 is translocated to the cell surface after receiving an
intracellular signal produced by insulin, resulting in glucose
uptake (49). Therefore, adequate
intracellular retention and a quick response to insulin stimulation
are needed to maintain the function of GLUT4 in modulating glucose
homeostasis. GLUT4 is currently regarded as an important molecule
for regulating glucose homeostasis, and also a key factor involved
in the development of insulin resistance (49). A previous study found decreased
levels of GLUT4 expression in the endometrium of PCOS patients
(13); however, the mechanism for
that decrease is largely unclear. In the present study we found
that GLUT4 expression could be regulated by HMGA2 and SREBF1, which
offers a novel explanation for the aberrant expression of GLUT4 in
PCOS patients. Our findings also provided some new promising
therapeutic targets for PCOS, and suggest that increasing GLUT4
expression by inhibiting miR-33b-5p or increasing HMGA2 and SREBF1
levels may be a promising strategy for treating PCOS.
HMGA2 is a non-histone chromosomal protein with
three DNA binding domains, and can act as a transcription
regulating factor (50). HMGA2 can
alter the structure of chromatin by forming a stereospecific
complex with other proteins in the promoter or enhancer region of
certain genes (51). It is also an
essential component of the enhanceosome, and can promote the
assembly of protein complexes and regulate the transcription of
target genes (51). Previous
studies have revealed that HMGA2 is associated with lipomas,
indicating its roles in adipogenesis and insulin resistance
(14,15). In the present study, we demonstrated
that miR-33b-5p was responsible for regulating HMGA2 and HMGA2
could regulate GLUT4 transcription. These findings explain
the roles played by miR-33b-5p and GLUT4 in the pathogenesis of
PCOS at a molecular level.
SREBF1 is a major regulator of genes involved in
metabolic pathways and sterol biosynthesis, as it binds to the
steroid regulatory element of the promoter or enhancer of those
genes (16–18). Previous studies have revealed that
SREBF1 is an important transcription factor that regulates certain
genes related to lipid metabolism, which is also associated with
insulin resistance (52,53). In the present study, we determined
that SREBF1 could directly regulate the target gene of GLUT4, which
is a key gene involved in insulin resistance. We also determined
that miR-33b-5p could target SREBP1 and inhibit its
expression. Moreover, we revealed that SREBP1 could regulate the
expression of GLUT4 as a transcription factor, which further
illustrates the key roles played by miR-33b-5p and GLUT4 in the
development of PCOS.
In summary, the findings from this study revealed
that miR-33b-5p was overexpressed in the ovarian tissues of PCOS
rats with insulin resistance, and thus may play an important role
in the development of insulin resistance in PCOS patients.
miR-33b-5p could inhibit GLUT4 by targeting HMGA2 during the
development of PCOS. Furthermore, HMGA2 and SREBF1 are important
molecules involved in modulating GLUT4 expression. These findings
enhanced our understanding of the molecular mechanisms that
contribute to insulin resistance during the development of PCOS,
and may assist in finding new therapeutic targets for PCOS. Future
studies may determine whether it is feasible to treat PCOS by
targeting miR-33b-5p.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Natural
Science Foundation of Hubei Province (no. 2014CFB251).
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
YY, HJ and XZY conceived and designed the study. YY,
HJ and LX performed the experiments. YY and XZY wrote the paper.
All authors read and approved the manuscript and agree to be
accountable for all aspects of the research in ensuring that the
accuracy or integrity of any part of the work are appropriately
investigated and resolved.
Ethics approval and consent to
participate
All tissue specimens were obtained with permission
from the Affiliated Hospital of Hubei University. All participants
have read and signed the written informed consent.
Consent for publication
All participants have read and signed the written
informed consent for the publication.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Azziz R, Carmina E, Chen Z, Dunaif A,
Laven JS, Legro RS, Lizneva D, Natterson-Horowtiz B, Teede HJ and
Yildiz BO: Polycystic ovary syndrome. Nat Rev Dis Primers.
2:160572016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Barber TM, Dimitriadis GK, Andreou A and
Franks S: Polycystic ovary syndrome: Insight into pathogenesis and
a common association with insulin resistance. Clin Med. 16:262–266.
2016. View Article : Google Scholar
|
|
3
|
Lauritsen MP, Bentzen JG, Pinborg A, Loft
A, Forman JL, Thuesen LL, Cohen A, Hougaard DM and Andersen Nyboe
A: The prevalence of polycystic ovary syndrome in a normal
population according to the Rotterdam criteria versus revised
criteria including anti-Mullerian hormone. Hum Reprod. 29:791–801.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li R, Zhang Q, Yang D, Li S, Lu S, Wu X,
Wei Z, Song X, Wang X, Fu S, et al: Prevalence of polycystic ovary
syndrome in women in China: A large community-based study. Hum
Reprod. 28:2562–2569. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Marciniak A, Rutkowska Nawrocka J,
Brodowska A, Wiśniewska B and Starczewski A: Cardiovascular system
diseases in patients with polycystic ovary syndrome - the role of
inflammation process in this pathology and possibility of early
diagnosis and prevention. Ann Agric Environ Med. 23:537–541. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Merz CNB, Shaw LJ, Azziz R, Stanczyk FZ,
Sopko G, Braunstein GD, Kelsey SF, Kip KE, Cooper-DeHoff RM,
Johnson BD, et al: Cardiovascular disease and 10-year mortality in
postmenopausal women with clinical features of polycystic ovary
syndrome. J Womens Health. 25:875–881. 2016. View Article : Google Scholar
|
|
7
|
Velija-Asimi Z, Burekovic A, Dujic T,
Dizdarevic-Bostandzic A and Semiz S: Incidence of prediabetes and
risk of developing cardiovascular disease in women with polycystic
ovary syndrome. Bosn J Basic Med Sci. 16:298–306. 2016.PubMed/NCBI
|
|
8
|
Jeanes YM and Reeves S: Metabolic
consequences of obesity and insulin resistance in polycystic ovary
syndrome: Diagnostic and methodological challenges. Nutr Res Rev.
30:97–105. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Morgan BJ, Chai SY and Albiston AL: GLUT4
associated proteins as therapeutic targets for diabetes. Recent Pat
Endocr Metab Immune Drug Discov. 5:25–32. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Govers R: Cellular regulation of glucose
uptake by glucose transporter GLUT4. Adv Clin Chem. 66:173–240.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Johansson J, Feng Y, Shao R, Lönn M,
Billig H and Stener-Victorin E: Intense electroacupuncture
normalizes insulin sensitivity, increases muscle GLUT4 content, and
improves lipid profile in a rat model of polycystic ovary syndrome.
Am J Physiol Endocrinol Metab. 299:E551–E559. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mozzanega B, Mioni R, Granzotto M,
Chiarelli S, Xamin N, Zuliani L, Sicolo N, Marchesoni D and Vettor
R: Obesity reduces the expression of GLUT4 in the endometrium of
normoinsulinemic women affected by the polycystic ovary syndrome.
Ann NY Acad Sci. 1034:364–374. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhai J, Liu CX, Tian ZR, Jiang QH and Sun
YP: Effects of metformin on the expression of GLUT4 in endometrium
of obese women with polycystic ovary syndrome. Biol Reprod.
87:292012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Xi Y, Shen W, Ma L, Zhao M, Zheng J, Bu S,
Hino S and Nakao M: HMGA2 promotes adipogenesis by activating
C/EBPβ-mediated expression of PPARγ. Biochem Biophys Res Commun.
472:617–623. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yuan Y, Xi Y, Chen J, Zhu P, Kang J, Zou
Z, Wang F and Bu S: STAT3 stimulates adipogenic stem cell
proliferation and cooperates with HMGA2 during the early stage of
differentiation to promote adipogenesis. Biochem Biophys Res
Commun. 482:1360–1366. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Oishi Y, Spann NJ, Link VM, Muse ED, Strid
T, Edillor C, Kolar MJ, Matsuzaka T, Hayakawa S, Tao J, et al:
SREBP1 contributes to resolution of pro-inflammatory TLR4 signaling
by reprogramming fatty acid metabolism. Cell Metab. 25:412–427.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shafiee MN, Mongan N, Seedhouse C, Chapman
C, Deen S, Abu J and Atiomo W: Sterol regulatory element binding
protein-1 (SREBP1) gene expression is similarly increased in
polycystic ovary syndrome and endometrial cancer. Acta Obstet
Gynecol Scand. 96:556–562. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang L, Chen J, Li Y, Wang Y, Liang S, Shi
Y, Shi S and Xu Y: Association between SCAP and
SREBF1 gene polymorphisms and metabolic syndrome in
schizophrenia patients treated with atypical antipsychotics. World
J Biol Psychiatry. 17:467–474. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Flowers E, Froelicher ES and Aouizerat BE:
MicroRNA regulation of lipid metabolism. Metabolism. 62:12–20.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ebert MS and Sharp PA: Roles for microRNAs
in conferring robustness to biological processes. Cell.
149:515–524. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bartel DP: MicroRNAs: Target recognition
and regulatory functions. Cell. 136:215–233. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yates LA, Norbury CJ and Gilbert RJC: The
long and short of microRNA. Cell. 153:516–519. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Osmai M, Osmai Y, Bang-Berthelsen CH,
Pallesen EM, Vestergaard AL, Novotny GW, Pociot F and
Mandrup-Poulsen T: MicroRNAs as regulators of beta-cell function
and dysfunction. Diabetes Metab Res Rev. 32:334–349. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vienberg S, Geiger J, Madsen S and
Dalgaard LT: MicroRNAs in metabolism. Acta Physiol. 219:346–361.
2017. View Article : Google Scholar
|
|
25
|
Cruz KJ, de Oliveira AR, Morais JB, Severo
JS and Marreiro DD: Role of microRNAs on adipogenesis, chronic
low-grade inflammation, and insulin resistance in obesity.
Nutrition. 35:28–35. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Williams MD and Mitchell GM: MicroRNAs in
insulin resistance and obesity. Exp Diabetes Res. 2012:4846962012.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen YH, Heneidi S, Lee JM, Layman LC,
Stepp DW, Gamboa GM, Chen BS, Chazenbalk G and Azziz R: miRNA-93
inhibits GLUT4 and is overexpressed in adipose tissue of polycystic
ovary syndrome patients and women with insulin resistance.
Diabetes. 62:2278–2286. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hossain MM, Cao M, Wang Q, Kim JY,
Schellander K, Tesfaye D and Tsang BK: Altered expression of miRNAs
in a dihydrotestosterone-induced rat PCOS model. J Ovarian Res.
6:362013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ilie IR and Georgescu CE: Polycystic ovary
syndrome-epigenetic mechanisms and aberrant microRNA. Adv Clin
Chem. 71:25–45. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jiang L, Huang J, Li L, Chen Y, Chen X,
Zhao X and Yang D: MicroRNA-93 promotes ovarian granulosa cells
proliferation through targeting CDKN1A in polycystic ovarian
syndrome. J Clin Endocrinol Metab. 100:E729–E738. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sørensen AE, Udesen PB, Wissing ML,
Englund AL and Dalgaard LT: MicroRNAs related to androgen
metabolism and polycystic ovary syndrome. Chem Biol Interact.
259:8–16. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yin M, Wang X, Yao G, Lü M, Liang M, Sun Y
and Sun F: Transactivation of microRNA-320 by microRNA-383
regulates granulosa cell functions by targeting E2F1 and SF-1
proteins. J Biol Chem. 289:18239–18257. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Poretsky L, Clemons J and Bogovich K:
Hyperinsulinemia and human chorionic gonadotropin synergistically
promote the growth of ovarian follicular cysts in rats. Metabolism.
41:903–910. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Adams BD, Parsons C, Walker L, Zhang WC
and Slack FJ: Targeting noncoding RNAs in disease. J Clin Invest.
127:761–771. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yee D, Coles MC and Lagos D: microRnAs in
the lymphatic endothelium: Master regulators of lineage plasticity
and inflammation. Front Immunol. 8:1042017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Butz H, Kinga N, Racz K and Patocs A:
Circulating miRNAs as biomarkers for endocrine disorders. J
Endocrinol Invest. 39:1–10. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Derghal A, Djelloul M, Trouslard J and
Mounien L: An emerging role of micro-RNA in the effect of the
endocrine disruptors. Front Neurosci. 10:3182016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Jiang L, Huang J, Chen Y, Yang Y, Li R, Li
Y, Chen X and Yang D: Identification of several circulating
microRNAs from a genome-wide circulating microRNA expression
profile as potential biomarkers for impaired glucose metabolism in
polycystic ovarian syndrome. Endocrine. 53:280–290. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li C, Chen L, Zhao Y, Chen S, Fu L, Jiang
Y, Gao S, Liu Z, Wang F, Zhu X, et al: Altered expression of miRNAs
in the uterus from a letrozole-induced rat PCOS model. Gene.
598:20–26. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Salimi-Asl M, Mozdarani H and Kadivar M:
Up-regulation of miR-21 and 146a expression and increased DNA
damage frequency in a mouse model of polycystic ovary syndrome
(PCOS). Bioimpacts. 6:85–91. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Sørensen AE, Wissing ML, Englund ALM and
Dalgaard LT: MicroRNA species in follicular fluid associating with
polycystic ovary syndrome and related intermediary phenotypes. J
Clin Endocrinol Metab. 101:1579–1589. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang CL, Wang H, Yan CY, Gao XF and Ling
XJ: Deregulation of RUNX2 by miR-320a deficiency impairs
steroidogenesis in cumulus granulosa cells from polycystic ovary
syndrome (PCOS) patients. Biochem Biophys Res Commun.
482:1469–1476. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cai G, Ma X, Chen B, Huang Y, Liu S, Yang
H and Zou W: MicroRNA-145 negatively regulates cell proliferation
through targeting IRS1 in isolated ovarian granulosa cells from
patients with polycystic ovary syndrome. Reprod Sci. 24:902–910.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Abruzzese GA, Cerrrone GE, Gamez JM,
Graffigna MN, Belli S, Lioy G, Mormandi E, Otero P, Levalle OA and
Motta AB: Lipid accumulation product (LAP) and visceral adiposity
index (VAI) as markers of insulin resistance and metabolic
associated disturbances in young argentine women with polycystic
ovary syndrome. Horm Metab Res. 49:23–29. 2017.PubMed/NCBI
|
|
45
|
Temur M, Yılmaz Ö, Aksun S, Calan M, Özbay
Özün P, Kumbasar S and Sever E: The relationship of urocortin-2
with insulin resistance patients having PCOS. Gynecol Endocrinol.
33:124–127. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Boden G, Homko C, Barrero CA, Stein TP,
Chen X, Cheung P, Fecchio C, Koller S and Merali S: Excessive
caloric intake acutely causes oxidative stress, GLUT4
carbonylation, and insulin resistance in healthy men. Sci Transl
Med. 7:304re72015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Fazakerley DJ, Naghiloo S, Chaudhuri R,
Koumanov F, Burchfield JG, Thomas KC, Krycer JR, Prior MJ, Parker
BL, Murrow BA, et al: Proteomic analysis of GLUT4 storage vesicles
reveals tumor suppressor candidate 5 (TUSC5) as a novel regulator
of insulin action in adipocytes. J Biol Chem. 290:23528–23542.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Belman JP, Bian RR, Habtemichael EN, Li
DT, Jurczak MJ, Alcázar-Román A, McNally LJ, Shulman GI and Bogan
JS: Acetylation of TUG protein promotes the accumulation of GLUT4
glucose transporters in an insulin-responsive intracellular
compartment. J Biol Chem. 290:4447–4463. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Leto D and Saltiel AR: Regulation of
glucose transport by insulin: Traffic control of GLUT4. Nat Rev Mol
Cell Biol. 13:383–396. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Rowe RG, Wang LD, Coma S, Han A, Mathieu
R, Pearson DS, Ross S, Sousa P, Nguyen PT, Rodriguez A, et al:
Developmental regulation of myeloerythroid progenitor function by
the Lin28b-let-7-Hmga2 axis. J Exp Med. 213:1497–1512. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Fedele M, Palmieri D and Fusco A: HMGA2: A
pituitary tumour subtype-specific oncogene? Mol Cell Endocrinol.
326:19–24. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Han J, Li E, Chen L, Zhang Y, Wei F, Liu
J, Deng H and Wang Y: The CREB coactivator CRTC2 controls hepatic
lipid metabolism by regulating SREBP1. Nature. 524:243–246. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Gorgani-Firuzjaee S and Meshkani R: SH2
domain-containing inositol 5-phosphatase (SHIP2) inhibition
ameliorates high glucose-induced de-novo lipogenesis and VLDL
production through regulating AMPK/mTOR/SREBP1 pathway and ROS
production in HepG2 cells. Free Radic Biol Med. 89:679–689. 2015.
View Article : Google Scholar : PubMed/NCBI
|