Introduction
Gastric cancer (GC), the fourth most commonly
diagnosed malignant disease following lung, breast and colorectal
cancer, is the second leading cause of cancer-related mortality
worldwide. The National Comprehensive Cancer Network (NCCN)
guidelines recommend surgery, chemotherapy and radiation therapy
for GC patients (1). However,
despite the fact that the incidence of gastric cancer has decreased
worldwide over the last 3 decades, its burden remains substantial
due to common relapses originating from a residual nidus (2). Therefore, there is an urgent need to
identify novel anticancer agents with milder toxicity profiles to
enhance the efficacy of GC treatments.
Accumulating evidence suggests that natural,
bioactive substances from plants (phytochemicals) may be promising
chemopreventive and chemotherapeutic agents in the treatment of
several human cancers. A number of phytochemicals have been found
to exert anticancer effects in in vitro studies, some of
which have been confirmed in vivo, such as certain
polyphenols (e.g., resveratrol and gallocatechins) and flavonoids
(e.g., methoxy licoflavanone and alpinumisoflavone), among others
(3). More importantly, clinical
trial evaluations of several phytochemicals have been conducted in
cancer patients. For example, the anticancer activity of
resveratrol was evaluated in patients with colorectal cancer and
hepatic metastases in a phase I randomized double-blind pilot
study, which reported that resveratrol significantly increased the
apoptosis of hepatic cancer cells (4). Xanthohumol [Xn;
3-(3,3-dimethylallyl)-2,4,4-trihydroxy-6′-methoxychalcone], the
most abundant prenylated flavonoid (0.1–1% of dry weight) in Hops
(Humulus lupulus L.), may be isolated from the female
inflorescences, as shown in Fig. 1.
Xn is also a constituent of beer, a major dietary source of
prenylated flavonoids, where it has been found at concentrations of
up to 0.96 mg/l (1.95 µM) (5). In recent years, an increasing number
of studies demonstrated the broad-spectrum anticancer activity of
Xn in NSCLC (6), hepatocellular
carcinoma (7), breast cancer
(8), leukemia (9), prostate cancer (10) and glioblastoma (11). Exposure of cancer cells to Xn may
inhibit their proliferation, migration and invasion, as well as
induce apoptosis and cell cycle arrest. However, to the best of our
knowledge, the effects of Xn on GC have not been investigated to
date.
The present study aimed to investigate the
anticancer activity of Xn against GC cells in vitro, and
preliminarily explore the underlying mechanism. The effects of Xn
on the proliferation, apoptosis, migration and invasiveness of GC
cells were evaluated; in addition, whether these effects involved
reactive oxygen species (ROS) production and nuclear factor (NF)-κB
signaling was further investigated.
Materials and methods
Cell culture
GC cells (AGS, SGC-7901 and MGC-803) and normal
gastric epithelial cells GES-1 were obtained from the American Type
Culture Collection (Rockville, MD, USA). These cell lines were
cultured in RPMI-1640 (HyClone, Logan, UT, USA), supplemented with
10% heat-inactivated fetal bovine serum (FBS; Gibco-Invitrogen;
Thermo Fisher Scientific, Inc., Carlsbad, CA, USA) in a humidified
atmosphere with 5% CO2 at 37°C.
Reagents
Xn was obtained from Sigma-Aldrich; Merck KGaA (St.
Louis, MO, USA). CellTiter 96® AQueous One Solution Cell
Proliferation Assay kit was purchased from Promega Corporation
(Madison, WI, USA). The 5-ethynyl-20-deoxyuridine (EdU)
incorporation assay kit was purchased from RiboBio (Guangzhou
China), the FITC Annexin V Apoptosis Detection Kit I was obtained
from BD Pharmingen (BD Biosciences, Franklin Lakes, NJ, USA).
Antibodies against Bcl-2 (rabbit polyclonal antibody, dilution
1:1,000; cat. no. ab194583), Bax (rabbit monoclonal antibody,
dilution 1:1,000; cat. no. ab32503), p-IκBα (rabbit monoclonal
antibody, dilution 1:1,000; cat. no. ab133462), IκBα (rabbit
monoclonal antibody, dilution 1:1,000; cat. no. ab32518), p65
(rabbit polyclonal antibody, dilution 1:1,000; cat. no. ab16502),
histone H3 (rabbit polyclonal antibody, dilution 1:1,000; cat. no.
ab1791) and GAPDH (rabbit polyclonal antibody, dilution 1:1,000;
cat. no. ab9485) were obtained from Abcam (Cambridge, UK). ROS and
superoxide dismutase (SOD) assay kits were purchased from Beyotime
Institute of Biotechnology, Ltd. (Shanghai, China).
Cell viability assay
Cell proliferation was measured by the CellTiter
96® AQueous One Solution Cell Proliferation Assay kit,
as reported previously (12). After
treatment with Xn for 24 h, the cells were incubated with 20
µl/well MTS solution for 1 h, and then measured using an
optical density reader at 570 nm (BioTek, Winooski, VT, USA).
EdU incorporation assay
A total of 100 µl of 50 µM EdU diluent
medium was added into each well for 3 h. The cells were fixed using
4% paraformaldehyde and incubated with 2 mg/ml aminoacetic acid for
5 min with oscillation. The cells were then incubated with 100
µl 0.5% Triton X-100 added into each well with 10 min of
oscillation followed by 100 µl of 1X Apollo® 488
fluorescent staining reaction liquid for 30 min at 37°C. DAPI was
used to stain the cell nuclei. The EdU incorporation rate was
expressed as the ratio of EdU-positive cells (green cells) to total
DAPI-positive cells (blue cells).
Flow cytometric analysis
Flow cytometric analysis was performed according to
the manufacturers' instructions (BD Biosciences). Cells were washed
twice with cold phosphate-buffered saline (PBS) and then
resuspended in 1X binding buffer at a concentration of
1×106 cells/ml. A total of 100 µl of the solution
(1×105 cells) was transferred to a 5-ml culture tube.
After adding 5 µl FITC Annexin V and 5 µl propidium
iodide (PI), the cells were gently vortexed and incubated for 15
min at room temperature (25°C) in the dark, followed by the
addition of 400 µl of 1X binding buffer to each tube.
Analysis by flow cytometry was performed within 1 h.
Western blotting
Proteins were extracted with RIPA buffer (containing
0.1% PMSF), separated by 10% SDS/PAGE and transferred to
polyvinylidene fluoride membranes. The membranes were then
incubated with primary antibodies overnight at 4°C, and horseradish
peroxidase-conjugated goat anti-mouse or goat anti-rabbit secondary
antibody (sc-2005, 1:2,000 and sc-2030, 1:5,000; Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA) for 1 h. The
chemiluminescence signals were detected with the EasySee Western
Blot Kit (Beijing TransGen Biotech, Beijing, China). ImageJ 1.43
software (National Institutes of Health, Bethesda, MD, USA) was
used for densitometric analysis.
Wound healing assay
A p200 pipette tip was used to create a scratch
after the cells had grown to 80–90% confluence in 6-well culture
plates. Images were captured at 0, 24 and 48 h after wounding. To
ensure that all wounds were of the same width at the beginning of
each experiment, an ocular ruler was used to measure wound
width.
Transwell assay
Transwell inserts (24-well) with an 8-µm pore
size (Corning Incorporated, Corning, NY, USA) were used in the
migration and invasion assays. The invasion assay was performed by
precoating the Transwell inserts with Matrigel Basement Membrane
Matrix (BD Biosciences). Briefly, the protocol was as follows:
Cells suspended in serum-free medium with different concentrations
of Xn were seeded in the upper chamber and were allowed to
transmigrate towards the bottom chamber, which contained medium
with 15% FBS for 24 h. The membrane inserts were then fixed with 4%
paraformaldehyde and stained with 1% gentian violet solution.
Images were captured from each membrane, and the number of
migrating and invading cells was counted under a microscope.
Intracellular ROS and SOD activity
measurement
The fluorescent probe dihydroethidium (DHE) was used
to monitor intracellular ROS levels. Intracellular DHE is oxidized
to ethidium, which binds to DNA and stains the nuclei bright
fluorescent red. Cells were incubated with different concentrations
of Xn and 5 mM DHE for 0, 1 and 3 h. The cultures were washed twice
with ice-cold PBS, then visualized using a Zeiss inverted
fluorescence microscope (Carl Zeiss AG, Oberkochen, Germany). The
total red fluorescence intensities of 5 views per well were
quantitated using ImageJ analysis software (National Institutes of
Health). Total SOD activity was determined by detecting superoxide
radicals generated from hypoxanthine and xanthine oxidase,
according to the manufacturer's instructions (Beyotime Institute of
Biotechnology, Ltd.).
Statistical analysis
The results are presented as means ± standard error
of the mean. Statistical analysis was performed by analysis of
variance followed by the Newman-Student-Keuls test for multiple
comparisons. The results were considered statistically significant
when P<0.05.
Results
Cytotoxicity of Xn on GC cells and
normal gastric epithelial cells
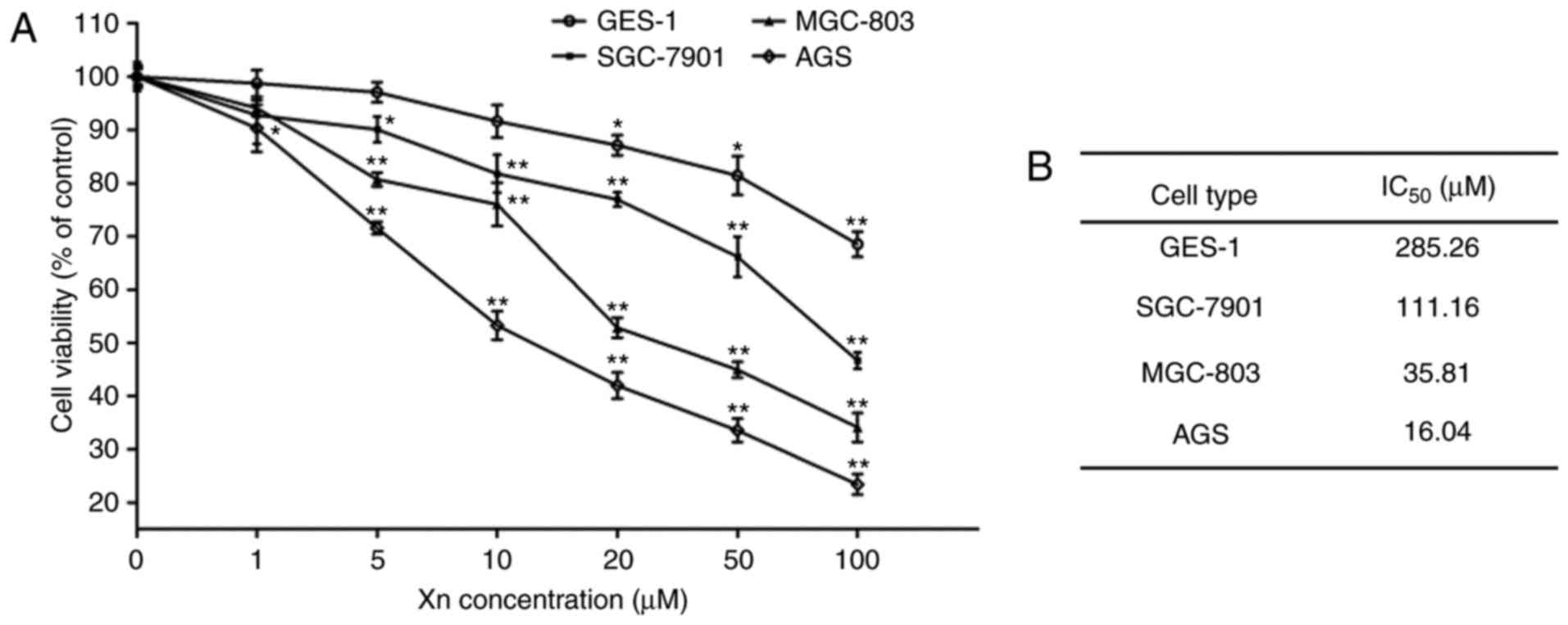
To investigate the effect of Xn on GC, the
cytotoxicity of Xn on GC cell lines (AGS, SGC-7901 and MGC-803) was
determined by the MTS assay after 24 h. As shown in Fig. 2, Xn significantly inhibited the
viability of AGS, SGC-7901 and MGC-803 cells in a dose-dependent
manner, with an IC50 of 16.04, 35.81 and 111.16
µM, respectively. The highest inhibitory activity of Xn was
observed in AGS cells. Thus, AGS cells and three concentrations
distributed around its IC50 value (5, 10 and 20
µM) were selected for further experiments.
To investigate whether the inhibition of Xn was
selective for cancer cells, the effect of Xn on normal gastric
epithelial cells (GES-1) was also tested. The results demonstrated
that Xn exerted very mild or no toxic effects on GES-1 cells
compared with GC cells, with an IC50 of up to 285.26
µM (Fig. 2A and B). There
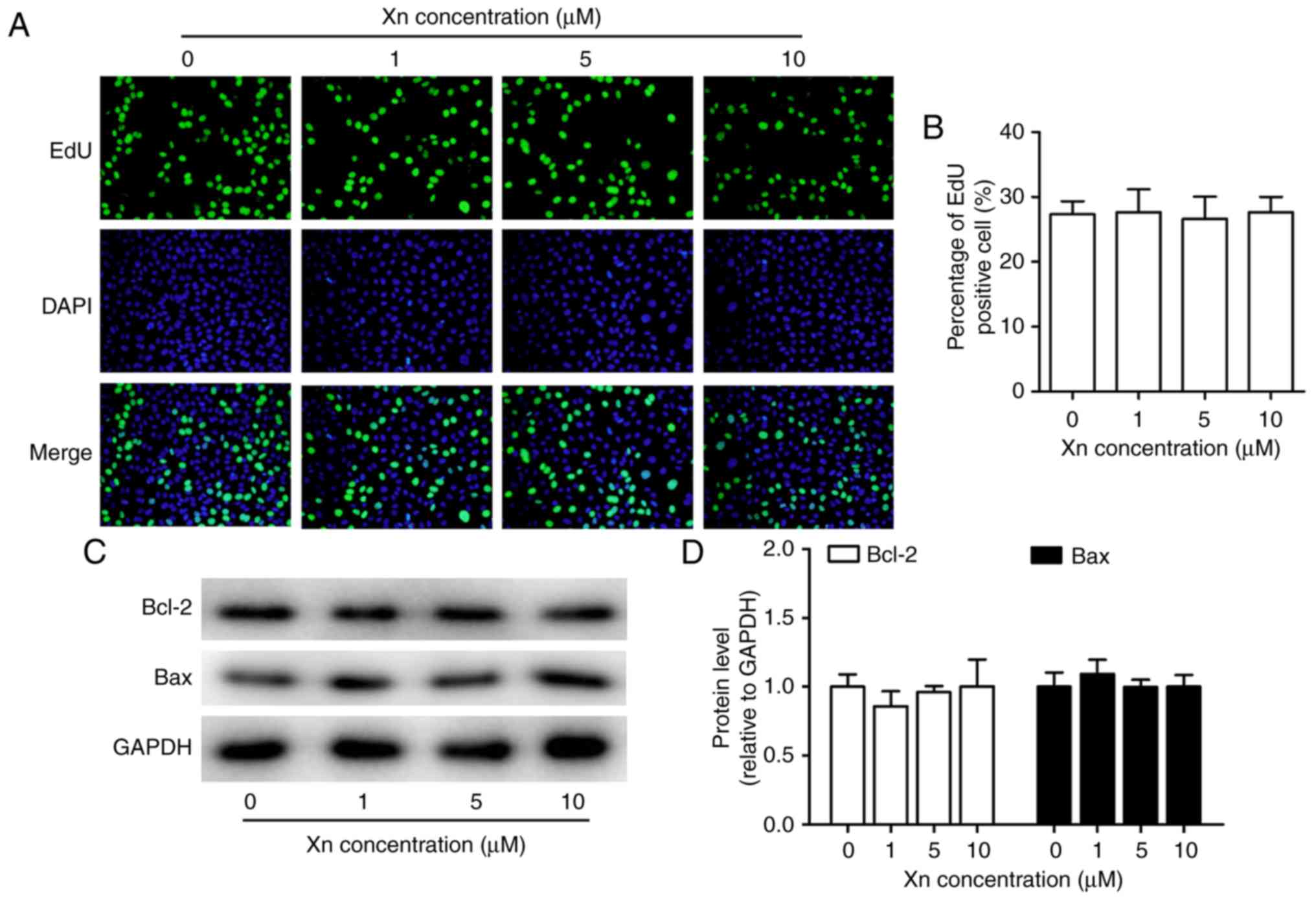
was no cytotoxicity at concentrations of 0–10 µM. Moreover,
Xn exerted no effect on the proliferation or apoptosis of GES-1
cells (Fig. 3A-D), as indicated by
the unchanged percentage of EdU-positive cells and the expression
of apoptosis-related proteins (Bcl-2 and Bax) following treatment
with Xn. These results indicated that Xn selectively targets GC
cells, but not normal cells.
Effect of Xn on the proliferation and
apoptosis of AGS cells
In view of the abnormal proliferation of cancer
cells playing a key role in the development and progression of GC
(13), the effect of Xn on the
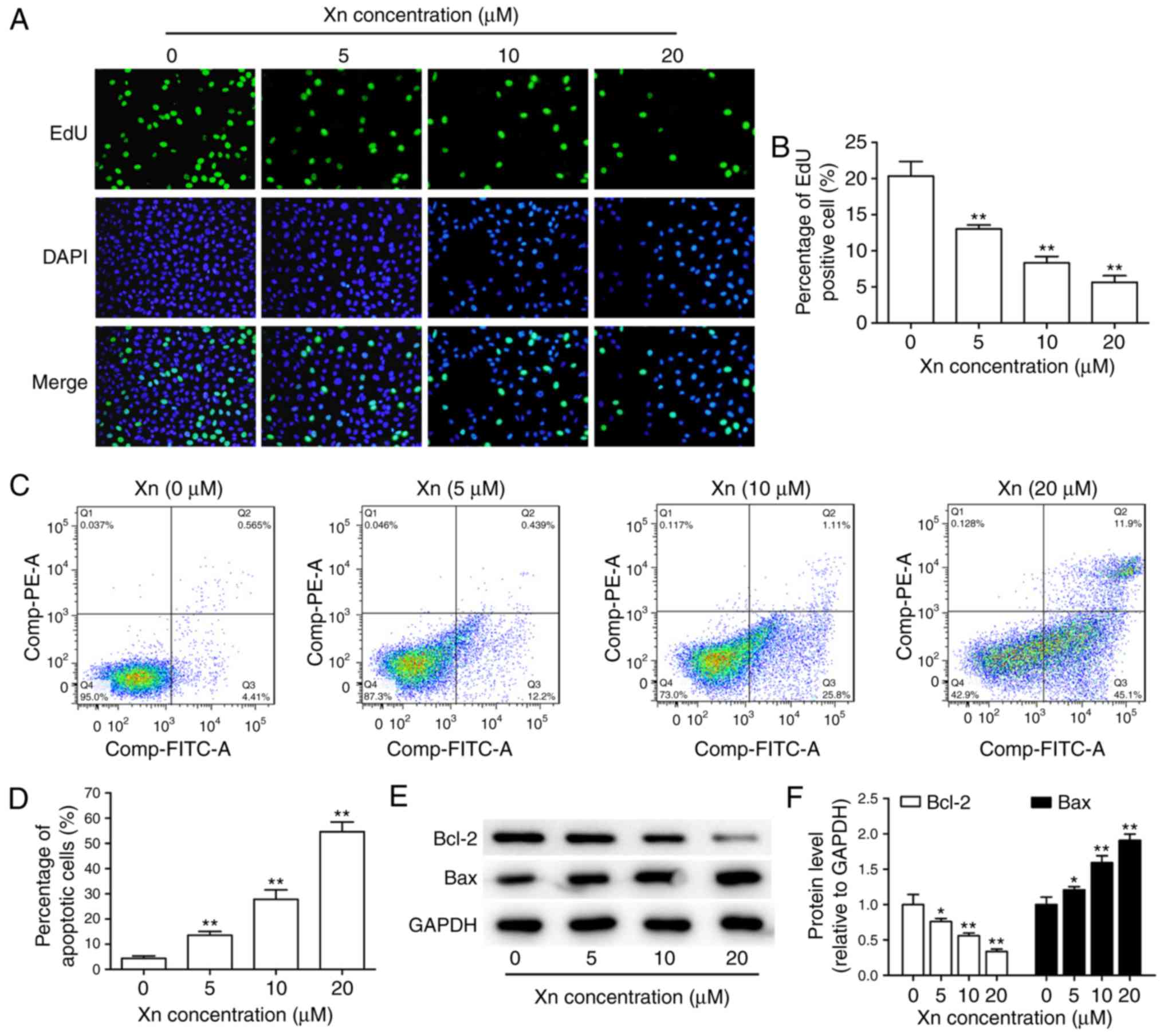
proliferation of AGS cells was investigated through EdU assay.
Following treatment with different concentrations of Xn for 24 h,
both the total number of cells and the percentage of EdU-positive
cells were dose-dependently decreased (Fig. 4A and B), suggesting that Xn
effectively suppresses the proliferation of AGS cells.
The decreased total number of cells under Xn
treatment may be due to apoptosis induction in addition to
proliferation inhibition. Therefore, the effect of Xn on the
apoptosis of AGS cells was assessed by flow cytometry and the
expression of apoptosis-related proteins. The results of flow
cytometry demonstrated that Xn increased the percentage of
apoptotic cells in a dose-dependent manner. Early apoptosis was
observed at concentrations of 5 and 10 µM, while incubation
with 20 µM Xn induced late apoptosis of AGS cells (Fig. 4C and D). Moreover, the expression of
apoptosis-related proteins, including the pro-apoptotic protein Bax
and the anti-apoptotic protein Bcl-2, may reflect the apoptosis
level. As shown in Fig. 4E and F,
Bcl-2 expression was decreased, while Bax expression was increased
following treatment with Xn for 24 h. Taken together, these results
indicate that Xn induces the apoptosis of AGS cells.
Effect of Xn on the migration and
invasion of AGS cells
Unrestricted metastasis is an important trait of GC,
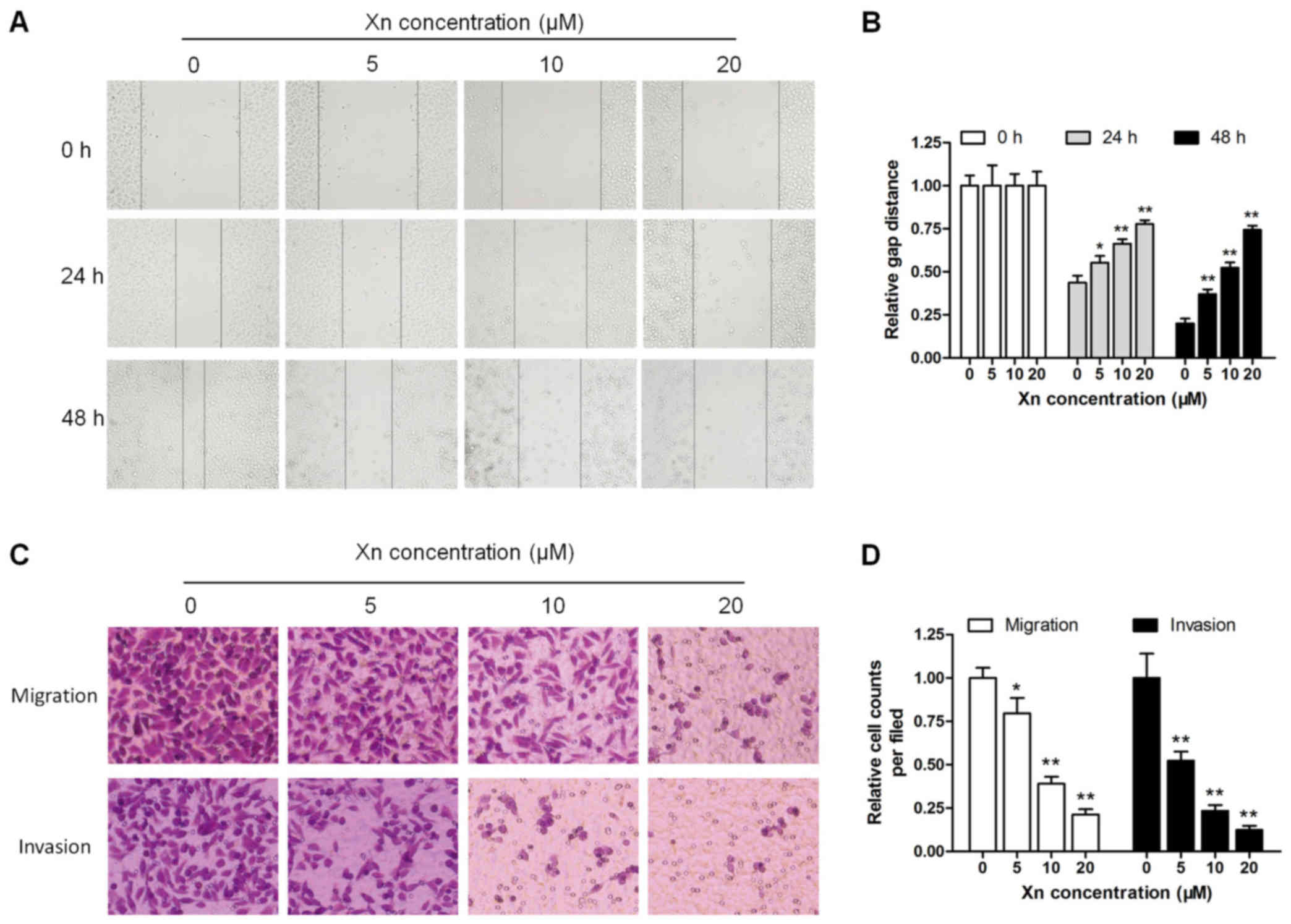
apart from disruptions in proliferation and apoptosis (14). The wound healing and Transwell
assays were performed to determine the effect of Xn on cell
motility. As shown in Fig. 5A and
B, wound recovery was significantly delayed by Xn in a time-
and dose-dependent manner; the inhibitory effect increased
gradually with increasing incubation time and Xn concentration. In
the Transwell assay, the migrating and invading cells were counted
and normalized to mock. Xn treatment for 24 h significantly
decreased the number of migrating and invading cells in a
dose-dependent manner (Fig. 5C and
D).
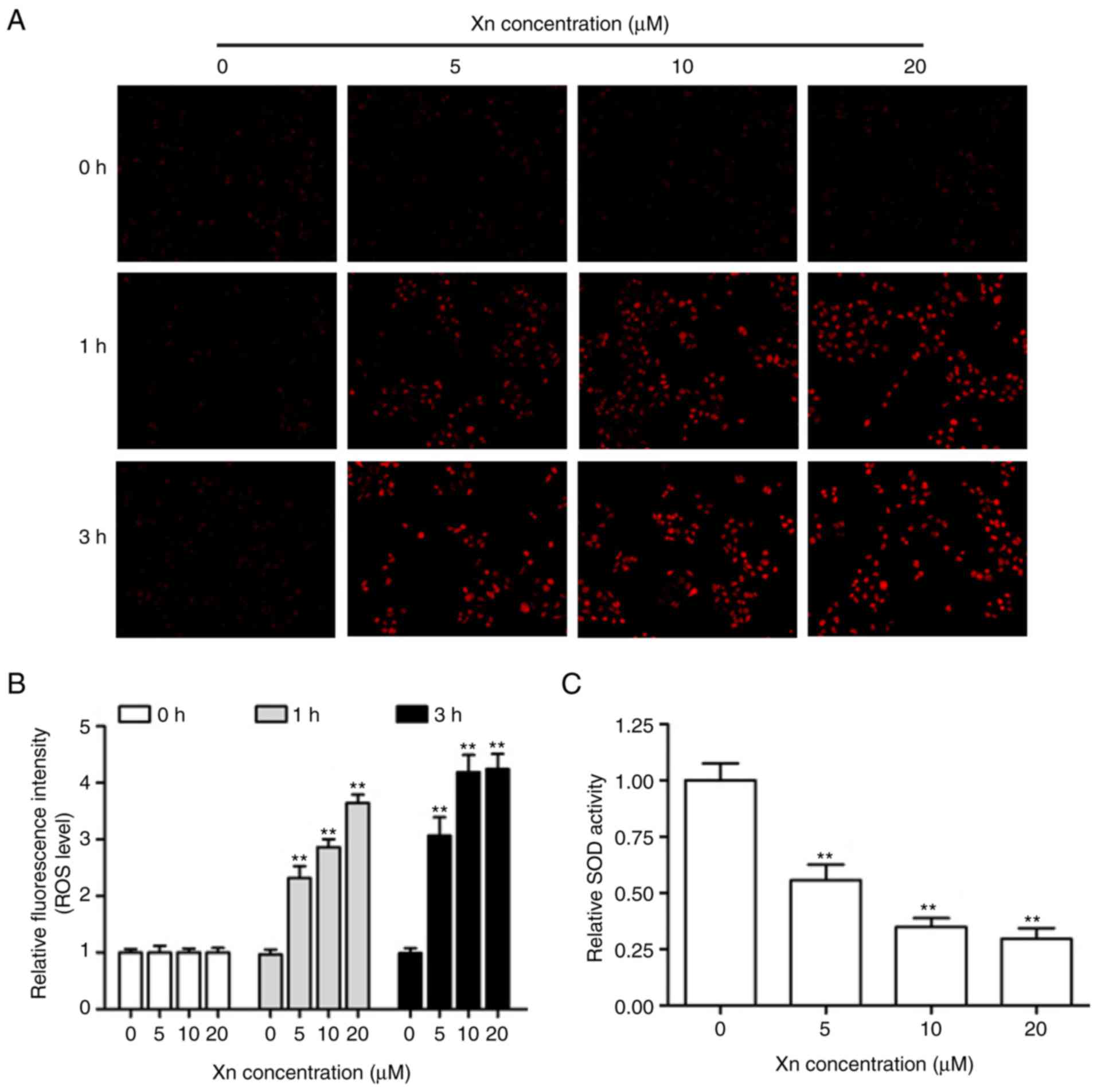
Effect of Xn on ROS production and SOD
activity in AGS cells
Alterations of the intracellular redox balance are
known to play a regulatory role in cell proliferation and apoptosis
(15). To explore whether the
proliferation inhibition and apoptosis induction by Xn involves ROS
production, DHE was used to monitor the intracellular ROS levels
derived from superoxide anion and superoxide. The representative
fluorescent images and corresponding statistics (Fig. 6A and B) revealed that treatment with
different concentrations of Xn for various times (0, 1 and 3 h)
increased DHE fluorescence intensity in a time- and dose-dependent
manner, suggesting that Xn induces ROS overproduction in AGS cells.
It is well-known that SOD activity reflects the endogenous
antioxidant ability against superoxide radicals. As shown in
Fig. 6C, the relative SOD activity
in AGS cells was decreased following exposure to Xn for 3 h. These
results indicated that Xn promotes ROS production and suppresses
SOD activity, resulting in intracellular redox imbalance.
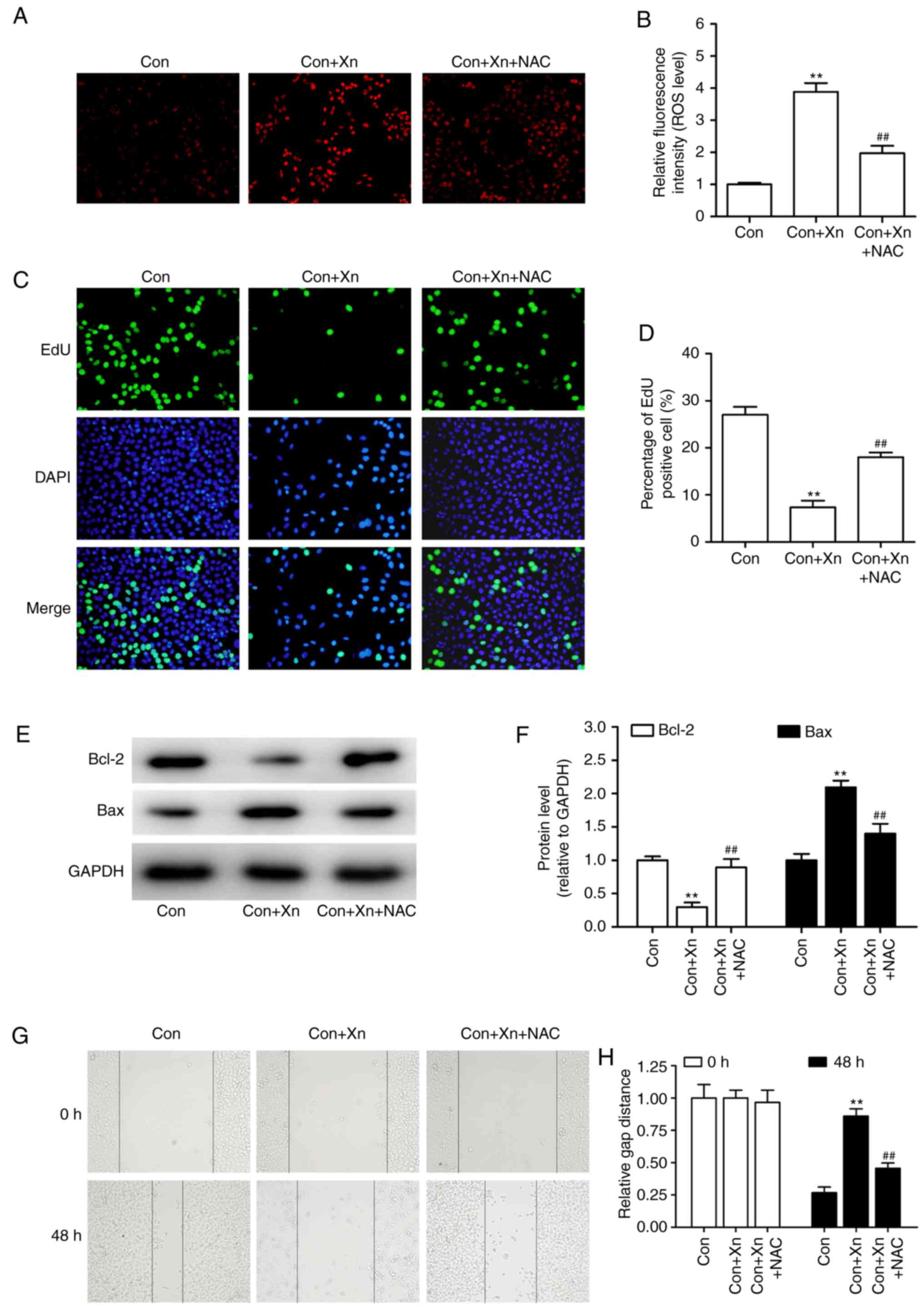
Effect of ROS inhibitor
N-acetylcysteine (NAC) on the anticancer activity of Xn against
GC
On the basis of abovementioned findings, to further
determine whether ROS mediated the anticancer activity of Xn
against GC, AGS cells were pre-treated with the ROS inhibitor NAC
(5 mM) prior to treatment with Xn (20 µM). As shown in
Fig. 7A and B, NAC inhibited the
ROS overproduction induced by Xn (20 µM). Furthermore, Xn
decreased the percentage of EdU-positive cells, delayed wound
recovery and induced cell apoptosis, as evidence by decreased Bcl-2
and increased Bax expression. However, these effects of Xn were
reversed by pretreatment with NAC (Fig.
7C-H), indicating the inhibitory effect of the ROS inhibitor
NAC on the anticancer activity of Xn.
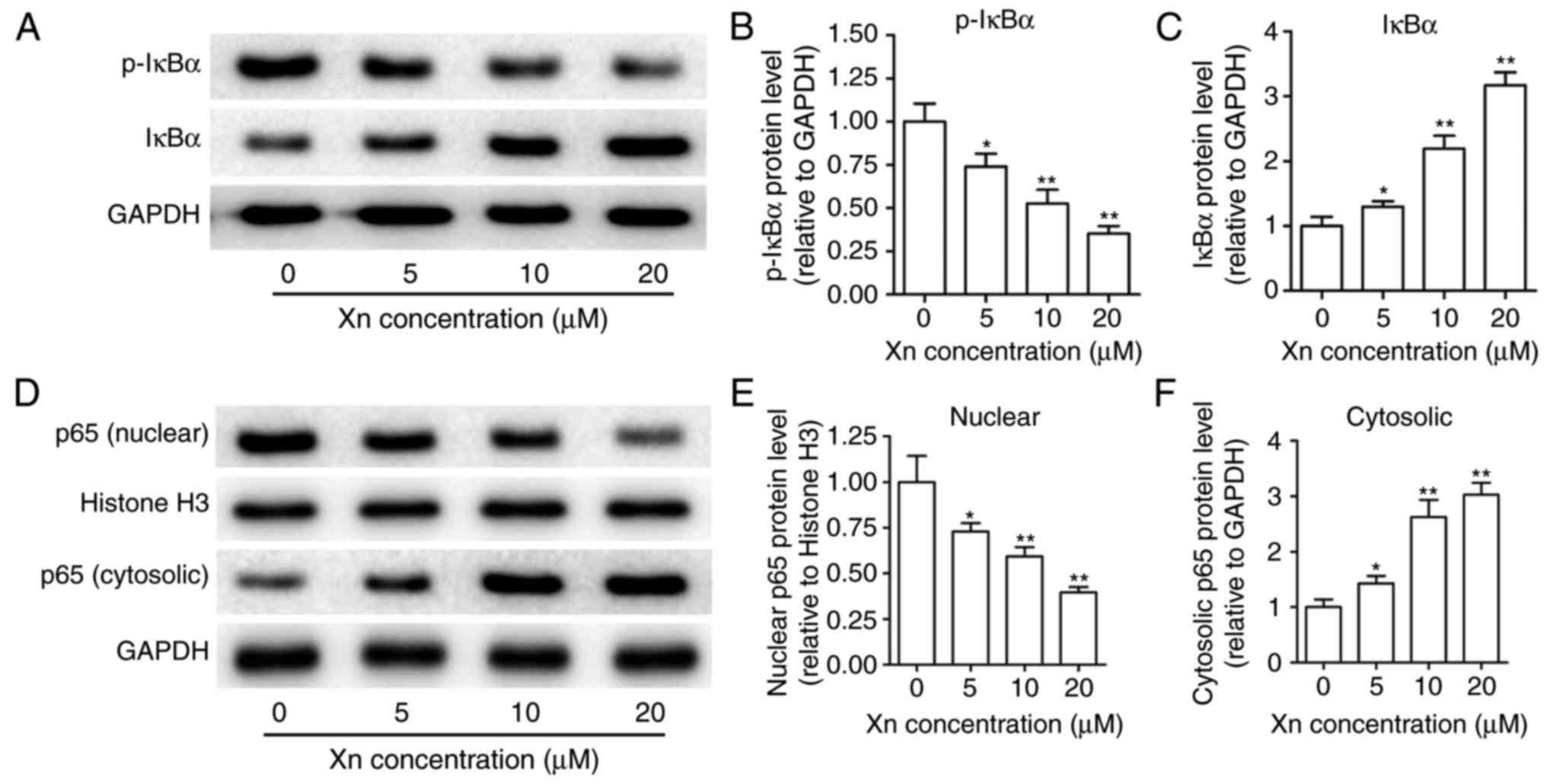
Effect of Xn on the NF-κB signaling
pathway in AGS cells
NF-κB is known to modulate apoptosis, acting as a
‘pro-survival’ factor (16). NF-κB
signaling is responsible for regulating transcription through NF-κB
p65 translocation into the nucleus, which is controlled by the
targeted phosphorylation and subsequent degradation of IκBα. To
examine the effect of Xn on NF-κB signaling, the expression of
p-IκBα, IκBα, p65 (nuclear) and p65 (cytosolic) were measured upon
Xn treatment for 24 h. As shown in Fig.
8A-C, Xn decreased the expression of p-IκBα and increased the
expression of IκBα, suggesting that the phosphorylation and
subsequent degradation of IκBα was inhibited by Xn. Moreover, the
expression of nuclear p65 was decreased, while the expression of
cytosolic p65 was increased with Xn treatment (Fig. 8D-F), suggesting a suppressive effect
of Xn on the NF-κB p65 nuclear translocation. These results
indicate that Xn inhibits NF-κB signaling.
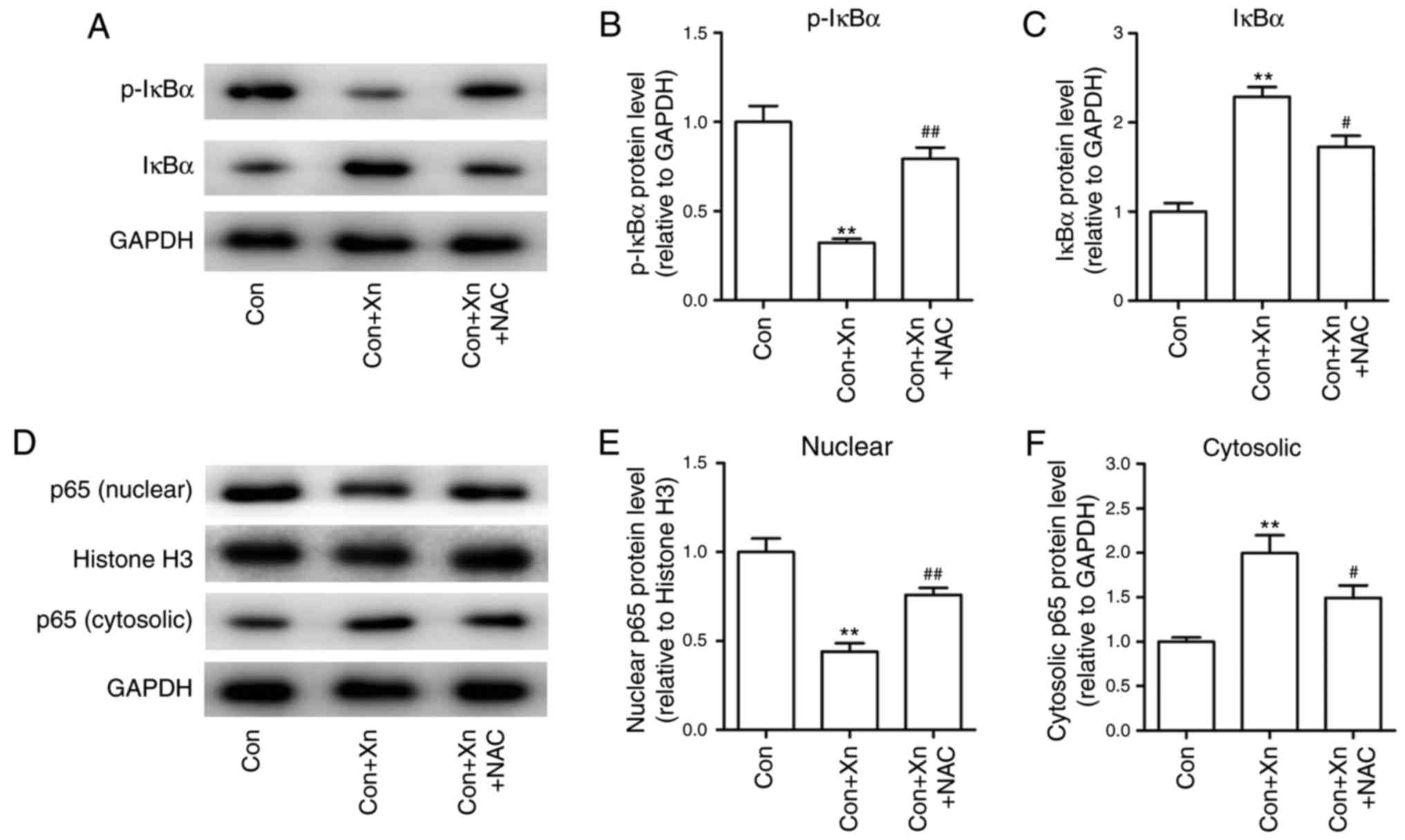
Effect of the ROS inhibitor NAC on the
NF-κB signaling pathway in Xn-treated AGS cells
High ROS levels have been shown to inhibit NF-κB
activation, which in turn regulates cancer cell survival (16). To determine whether ROS mediated the
anticancer activity of Xn through regulating NF-κB activation, the
expression of NF-κB activation-related proteins was measured
following pre-treatment with the ROS inhibitor NAC in Xn-treated
AGS cells. The results demonstrated that Xn decreased the
phosphorylation and subsequent degradation of IκBα, and suppressed
the nuclear translocation of NF-κB p65, which were inhibited by NAC
(Fig. 9A-F). Collectively, these
results suggest that ROS mediates the anticancer activity of Xn
against GC via the NF-κB signaling pathway.
Discussion
Hops, a principle raw material of beer, is widely
used in the brewing industry worldwide. Hops acts as a preservative
and gives beer its unique aroma and flavor (17,18).
In addition, hops has long been used as a medicinal plant, as it is
rich in a variety of phenolic compounds (19). Dried hops contains 4–14%
polyphenols, mainly phenolic acids, prenylated chalcones,
flavonoids, catechins and proanthocyanidins (20). As the most abundant prenylated
flavonoid, Xn exhibits extensive biological activities.
The structure of Xn was first identified by Verzele
et al in 1957 (21).
However, the beneficial pharmacological properties of Xn, including
antioxidant, anti-inflammatory, antibacterial, antiviral,
antifungal and antiplasmodial activities, were not fully elucidated
until the 1990s (22). Recently,
the anticancer activity of Xn was validated in a variety of cancer
cells. However, to the best of our knowledge, the present study is
the first to investigate the anticancer activity of Xn in GC. We
found that Xn dose-dependently decreased the viability of GC cells,
particularly AGS cells, with an IC50 as low as 16.04
µM. It has been reported that Xn exerts very mild or no
toxic effects on normal cells, including human lung fibroblast
cells (MRC-5), primary human hepatocytes, oligodendroglia-derived
cells (OLN-93) and human skin fibroblasts (23–25).
Encouraged by the results of these studies, we further investigated
the cytotoxicity of Xn on the normal gastric epithelial cell line
GES-1. No cytotoxicity was observed up to a concentration of 10
µM, with an IC50 of up to 285.26 µM.
Moreover, Xn exerted no effects on the proliferation and apoptosis
of GES-1 cells at concentrations of 0–10 µM. By contrast, Xn
significantly decreased the viability of GC cells from a
concentration of 5 µM, and all the IC50 values in
GC cells were higher compared with that in GES-1 cells. These
findings indicate that Xn specifically targets GC cells; therefore,
Xn may be a safe and effective treatment for GC.
The decreased cell viability may be attributed to
inhibition of proliferation or induction of apoptosis. Xn
suppressed the proliferation of AGS cells, as indicated by the
decreased number of EdU-positive cells. Moreover, flow cytometric
analysis revealed an increased number of apoptotic cells upon Xn
treatment. Numerous apoptotic-related proteins are involved in the
apoptosis process, particularly the Bcl-2 family members. Xn has
been reported to induce apoptosis through regulating the expression
of Bcl-2 family proteins in several types of cancer (8,10).
Bcl-2 family members may be classified into anti- and pro-apoptotic
proteins. Most Bcl-2 family members, including Bcl-2, Bcl-XL,
Bcl-w, Mcl-l, Bfl1/A-1 and Bcl-B, have anti-apoptotic properties;
however, a subset display pro-apoptotic properties, including Bax,
Bak and Bid. Among these Bcl-2 family members, the pro-apoptotic
protein Bax has been identified as an inhibitory binding partner of
Bcl-2, and their expression is commonly used to predict apoptosis
(26). In the present study,
downregulated Bcl-2 expression and upregulated Bax expression were
observed following Xn treatment; this finding, in combination with
the results of flow cytometric analysis, suggest that Xn induces
apoptosis of AGS cells. As one of the major causes of
cancer-related mortality worldwide, GC has a poor prognosis.
Metastasis accounts for the majority of deaths and the poor
prognosis, indicating that the prevention and control of metastasis
would contribute to improved GC treatment outcome (27). The metastatic ability of AGS cells
under Xn treatment was further determined and it was observed that
Xn dose-dependently delayed wound healing, cell migration and
invasion, suggesting that metastasis of AGS cells is suppressed by
Xn. Taken together, these findings indicated that Xn may be a
potential anticancer agent via affecting the proliferation,
apoptosis and metastasis of GC cells.
It is well-known that oxidative stress plays a key
role in several aspects of cancer development and progression,
including cellular proliferation, evasion of apoptosis or anoikis,
tissue invasion, metastasis and angiogenesis. Thus, cancer
treatment is highly associated with regulation of oxidative stress.
Although oxidative stress caused by ROS accumulation promotes tumor
growth, it can also increase the sensitivity to treatment (28). Numerous commonly used
chemotherapeutic agents and phytochemicals with anticancer activity
induce ROS production. For example, the cytotoxicity induced by the
chemotherapeutic drugs 5-fluorouracil and oxaliplatin is attributed
to increased ROS levels (29).
Piperlongumine, a bioactive agent derived from the long pepper
plant, potently inhibits the growth of breast tumors and metastases
in mice by increasing ROS levels (30). Moreover, Xn induced ROS
overproduction in A549 NSCLC cells and T98G glioblastoma cells,
resulting in cancer cell apoptosis (6,11). The
present study further demonstrated that Xn promoted intracellular
ROS production in AGS cells. The damage that ROS causes to cells
depends not only on their intracellular concentration, but also on
the equilibrium between the ROS and the endogenous antioxidant
species (e.g., SOD) (28). In the
present study, the relative SOD activity in AGS cells was decreased
following exposure to Xn, resulting in ROS overproduction.
Furthermore, reduction of ROS by NAC suppressed the effect of Xn on
the proliferation, apoptosis and metastasis of AGS cells. These
results indicate that ROS mediate the anticancer activity of Xn
against GC.
At sublethal levels, ROS have been shown to activate
the pro-inflammatory transcription factor, NF-κB, which in turn
controls the expression of signaling molecules associated with
cancer cell survival. However, high ROS levels reduce NF-κB
activity, resulting in apoptosis of cancer cells (16). Melatonin suppresses thyroid cancer
growth and overcomes radioresistance via inhibition of p65
phosphorylation and induction of ROS (31). The induction of ROS overproduction
mentioned above prompted us to further explore the effect of Xn on
NF-κB signaling in AGS cells. The results demonstrated that Xn
inhibited NF-κB activity via suppressing IκBα degradation and p65
nuclear translocation. However, the inhibitory effect of Xn on
NF-κB signaling may be reversed by ROS reduction, suggesting that
ROS mediates the anticancer activity of Xn against GC via the NF-κB
signaling pathway.
In conclusion, the present study demonstrated that
Xn significantly decreases the viability of GC cells, but not that
of normal gastric epithelial cells. Xn was shown to inhibit the
proliferation, induce apoptosis and suppress metastasis of AGS
cells. The underlying mechanism appears to involve ROS
overproduction and subsequent inhibition of NF-κB activity. These
results may provide a scientific basis supporting further use of Xn
in the treatment of GC.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the
National Natural Scientific Foundation of China (nos. 81703518,
81703592 and 81573718), the Hunan Provincial Natural Scientific
Foundation (no. 2018JJ3571), the Scientific Research Project of
Hunan Provincial Health and Family Planning Commission (no.
B20180253) and the Open-End Fund for the Valuable and Precision
Instruments of Central South University (no. CSUZC201837).
Availability of data and materials
The datasets generated during the present study are
available from the corresponding author on reasonable request.
Authors' contributions
WL, DX and BZ conceived and designed the
experiments. SW and TS performed the experiments. JD analyzed the
data. WL and SW wrote the manuscript. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests to disclose.
Glossary
Abbreviations
Abbreviations:
|
GC
|
gastric cancer
|
|
Xn
|
xanthohumol
|
|
NSCLC
|
non-small-cell lung cancer
|
|
DHE
|
dihydroethidium
|
|
ROS
|
reactive oxygen species
|
|
SOD
|
superoxide dismutase
|
|
NF-κB
|
nuclear factor κB
|
References
|
1
|
Ajani JA, Bentrem DJ, Besh S, D'Amico TA,
Das P, Denlinger C, Fakih MG, Fuchs CS, Gerdes H, Glasgow RE, et
al: Gastric cancer, version 2.2013: Featured updates to the NCCN
Guidelines. J Natl Compr Cancer Netw. 11:531–546. 2013. View Article : Google Scholar
|
|
2
|
Gong J, Cao J, Liu G and Huo JR: Function
and mechanism of F-box proteins in gastric cancer (Review). Int J
Oncol. 47:43–50. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Singh S, Sharma B, Kanwar SS and Kumar A:
Lead phytochemicals for anticancer drug development. Front Plant
Sci. 7:16672016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hosseini A and Ghorbani A: Cancer therapy
with phytochemicals: Evidence from clinical studies. Avicenna J
Phytomed. 5:84–97. 2015.PubMed/NCBI
|
|
5
|
Chen QH, Fu ML, Chen MM, Liu J, Liu XJ, He
GQ and Pu SC: Preparative isolation and purification of xanthohumol
from hops (Humulus lupulus L.) by high-speed counter-current
chromatography. Food Chem. 132:619–623. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhang B, Chu W, Wei P, Liu Y and Wei T:
Xanthohumol induces generation of reactive oxygen species and
triggers apoptosis through inhibition of mitochondrial electron
transfer chain complex I. Free Radic Biol Med. 89:486–497. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kunnimalaiyaan S, Sokolowski KM,
Balamurugan M, Gamblin TC and Kunnimalaiyaan M: Xanthohumol
inhibits Notch signaling and induces apoptosis in hepatocellular
carcinoma. PLoS One. 10:e01274642015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yoo YB, Park KS, Kim JB, Kang HJ, Yang JH,
Lee EK and Kim HY: Xanthohumol inhibits cellular proliferation in a
breast cancer cell line (MDA-MB231) through an intrinsic
mitochondrial-dependent pathway. Indian J Cancer. 51:518–523. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Benelli R, Venè R, Ciarlo M, Carlone S,
Barbieri O and Ferrari N: The AKT/NF-κB inhibitor xanthohumol is a
potent anti-lymphocytic leukemia drug overcoming chemoresistance
and cell infiltration. Biochem Pharmacol. 83:1634–1642. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Deeb D, Gao X, Jiang H, Arbab AS,
Dulchavsky SA and Gautam SC: Growth inhibitory and
apoptosis-inducing effects of xanthohumol, a prenylated chalone
present in hops, in human prostate cancer cells. Anticancer Res.
30:3333–3339. 2010.PubMed/NCBI
|
|
11
|
Festa M, Capasso A, DAcunto CW, Masullo M,
Rossi AG, Pizza C and Piacente S: Xanthohumol induces apoptosis in
human malignant glioblastoma cells by increasing reactive oxygen
species and activating MAPK pathways. J Nat Prod. 74:2505–2513.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li Y, Fan S, Koo J, Yue P, Chen ZG,
Owonikoko TK, Ramalingam SS, Khuri FR and Sun SY: Elevated
expression of eukaryotic translation initiation factor 4E is
associated with proliferation, invasion and acquired resistance to
erlotinib in lung cancer. Cancer Biol Ther. 13:272–280. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen FZ, Mo XM, Wang QP, Li J and Zhang L:
Effects of rosiglitazone on the growth and lymphangiogenesis of
human gastric cancer transplanted in nude mice. Oncol Rep.
30:2705–2712. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zuo ZK, Gong Y, Chen XH, Ye F, Yin ZM,
Gong QN and Huang JS: TGFβ1-induced lncRNA UCA1 upregulation
promotes gastric cancer invasion and migration. DNA Cell Biol.
36:159–167. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tong XP, Ma YX, Quan DN, Zhang L, Yan M
and Fan XR: Rosemary extracts upregulate Nrf2, Sestrin2, and MRP2
protein level in human hepatoma HepG2 cells. Evid Based Complement
Alternat Med. 2017:73598062017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fouani L, Kovacevic Z and Richardson DR:
Targeting oncogenic nuclear factor kappa B signaling with
redox-active agents for cancer treatment. Antioxid Redox Signal.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
McAdam EL, Freeman JS, Whittock SP, Buck
EJ, Jakse J, Cerenak A, Javornik B, Kilian A, Wang CH, Andersen D,
et al: Quantitative trait loci in hop (Humulus lupulus L.) reveal
complex genetic architecture underlying variation in sex, yield and
cone chemistry. BMC Genom. 14:3602013. View Article : Google Scholar
|
|
18
|
Dostálek P, Karabín M and Jelínek L: Hop
phytochemicals and their potential role in metabolic syndrome
prevention and therapy. Molecules. 22:E17612017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zanoli P and Zavatti M: Pharmacognostic
and pharmacological profile of Humulus lupulus L. J Ethnopharmacol.
116:383–396. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nikolic D and van Breemen RB: Analytical
methods for quantitation of prenylated flavonoids from hops. Curr
Anal Chem. 9:71–85. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Verzele M, Stockx J, Fontijn F and
Anteunis M: Xanthohumol, a new natural chalkone. J Agric Food Chem.
66:452–475. 1957.
|
|
22
|
Liu M, Hansen PE, Wang G, Qiu L, Dong J,
Yin H, Qian Z, Yang M and Miao J: Pharmacological profile of
xanthohumol, a prenylated flavonoid from hops (Humulus lupulus).
Molecules. 20:754–779. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dorn C, Weiss TS, Heilmann J and
Hellerbrand C: Xanthohumol, a prenylated chalcone derived from
hops, inhibits proliferation, migration and interleukin-8
expression of hepatocellular carcinoma cells. Int J Oncol.
36:435–441. 2010.PubMed/NCBI
|
|
24
|
Sławińska-Brych A, Król SK,
Dmoszyńska-Graniczka M, Zdzisińska B, Stepulak A and Gagoś M:
Xanthohumol inhibits cell cycle progression and proliferation of
larynx cancer cells in vitro. Chem Biol Interact. 240:110–118.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yong WK, Ho YF and Malek SN: Xanthohumol
induces apoptosis and S phase cell cycle arrest in A549 non-small
cell lung cancer cells. Pharmacogn Mag. 11 Suppl 2:S275–S283. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hardwick JM and Soane L: Multiple
functions of BCL-2 family proteins. Cold Spring Harb Perspect Biol.
5:a0087222013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Obermannová R and Lordick F: Management of
metastatic gastric cancer. Hematol Oncol Clin North Am. 31:469–483.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sosa V, Moliné T, Somoza R, Paciucci R,
Kondoh H and ME LL: Oxidative stress and cancer: An overview.
Ageing Res Rev. 12:376–390. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Afzal S, Jensen SA, Sørensen JB, Henriksen
T, Weimann A and Poulsen HE: Oxidative damage to guanine
nucleosides following combination chemotherapy with 5-fluorouracil
and oxaliplatin. Cancer Chemother Pharmacol. 69:301–307. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Raj L, Ide T, Gurkar AU, Foley M, Schenone
M, Li X, Tolliday NJ, Golub TR, Carr SA, Shamji AF, et al:
Selective killing of cancer cells by a small molecule targeting the
stress response to ROS. Nature. 475:231–234. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zou ZW, Liu T, Li Y, Chen P, Peng X, Ma C,
Zhang WJ and Li PD: Melatonin suppresses thyroid cancer growth and
overcomes radioresistance via inhibition of p65 phosphorylation and
induction of ROS. Redox Biol. 16:226–236. 2018. View Article : Google Scholar : PubMed/NCBI
|