Introduction
Abdominal adhesion refers to an abnormal adhesion
between intestinal tubes, between the intestines and viscera, or
between the intestines and peritoneum in the abdominal cavity
(1). Abdominal adhesions can be
caused by congenital or acquired factors, with acquired factors
accounting for 80% of abdominal adhesion cases, including
mechanical damage, peritoneal dryness, and introduction of foreign
bodies (e.g., microorganisms), suture lines and the talcum powder
present in surgical gloves (2),
which will cause long-term abdominal pain in patients (3).
The mechanisms underlying the formation of
postoperative abdominal adhesions remain unclear. Most experts
support the theory of injury and inflammation, and claim that
disorders of inflammatory cells, the secretion of inflammatory
factors, as well as fibrin formation and dissolution at the site of
injury are responsible for the formation of postoperative abdominal
adhesions (this process is usually completed within 7 days of
injury) (3). To prevent adhesions,
surgical or adjuvant treatments are usually applied in the clinical
setting (4). However, surgery
cannot completely eliminate the risk of adhesions, and almost a
quarter of abdominal re-exploration procedures accidentally cause
intestinal injury, which may lead to the formation of a new
adhesion after surgery (5). The
adjuvants include drugs (anticoagulant, antibiotic and fibrinolytic
drugs, as well as Chinese medicine preparations) as well as other
materials (membrane materials, adhesive glue and intraperitoneal
crystal solutions (6,7). With regard to drugs, their effects on
preventing abdominal adhesions are still unclear, and some of them
may even increase the occurrence of adverse reactions, specifically
bleeding of the wound after surgery and delayed wound healing
(8). Regarding materials, membrane
materials are difficult to apply during laparoscopic surgery or to
tissues with complex geometric structures, and they become fragile
and difficult to position when attached to moist surgical equipment
or tissues (9). The crystal
solution can easily become dislodged by photographic processing
during surgery, and the strong absorption capacity of the abdominal
cavity makes it a challenge to achieve a good curative effect
(10).
Intestinal microbiota are a microecology system that
is dominated by bacteria. There are approximately 1,000 different
types of bacteria in the human intestine, and maintaining a balance
is important for health (11,12).
Under physiological conditions, symbiotic physiological anaerobic
bacteria coexist with symbiotic conditional pathogenic bacteria and
other harmful bacteria in the intestinal tract, and their
cooperation with the nonspecific immune system can protect
intestinal epithelial cells from inflammation and fibrosis
(13). A study revealed that
intestinal inflammation was closely related to the Toll-like
receptor 4/nuclear factor-κ B (TLR4/NF-κB) signalling pathway, in
which TLR4 mainly recognizes lipopolysaccharide (LPS) of
gram-negative bacteria. When TLR4 is combined with LPS, it
activates the NF-κB pathway, causing the release of numerous
pro-inflammatory factors [interleukin-1β, (IL-1β); tumour necrosis
factor-α (TNF-α)] to mediate the intestinal inflammation process
(14). In addition, researchers
contend that transforming growth factor-β1 (TGF-β1) is the
initiator of fibrosis, which can transmit signals to Smad through
specific receptors on the cell membrane (15,16),
and is closely related to multiple organ fibrosis (17).
The intestinal microbiota are known to be associated
with obesity, cardiovascular disease, malnutrition, osteoporosis,
Clostridium difficile infection, type 2 diabetes, colorectal
cancer and inflammatory bowel disease (18,19).
Fedorak et al (20) reported
that probiotics could effectively reduce the levels of inflammatory
cytokines in the mucosa and helped to prevent the recurrence of
Crohn's disease after surgery. In addition, a study by Liu et
al (21) revealed that
probiotics had sound anti-inflammatory effects in animal models of
LPS-induced colitis by inhibiting the activation of inflammatory
signalling pathways [NF-κB and mitogen-activated protein kinase
(MAPK)] and reducing colon fibrosis (TGF-β1). To date, little
research has been conducted to explore the relationship between the
microbiota and abdominal adhesions, especially the potential of
probiotic treatment to prevent abdominal adhesions. In China, no
probiotic drug has been approved in recent decades, and the
probiotic drugs currently used are older drugs with certain
problems, e.g. they contain Enterococcus faecalis (found in
>75% of Chinese probiotic drugs; this strain is readily drug
resistant and can spread resistant genes) (22,23)
and Bacillus cereus (some strains cause infection) (24,25).
The Lactobacillus and Bifidobacteriium species have
been revealed to have anti-inflammatory/immunomodulatory effects in
experimental animal as well as human studies (26,27).
The beneficial effects of Lactobacillus plantarum, L.
acidophilus, L. rhamnosus or Bifidobacterium animalis
have been widely reported in these studies. Moreover, these
probiotic strains were regarded as safe and edible by the Ministry
of Health, China.
Therefore, to obtain a robust probiotic effect, a
combination of these four aforementioned probiotic strains were
used to investigate whether administration of a probiotic
combination could help to prevent abdominal adhesions in a rat
model, and to elucidate the underlying mechanism.
Materials and methods
Cultivation of probiotic bacteria
Four strains of L. plantarum MH-301 (isolated
from human faeces, and stored as a patented bacterium at the
Institute of Microbiology, Chinese Academy of Sciences), L.
acidophilus (isolated from fermented milk), L. rhamnosus
L12 and B. animalis subsp. lactis LPL-RH (both
provided by Harbin Meihua Biotechnology Co., Ltd.) were used in the
present study. These strains were cultured in De Man, Rogosa and
Sharpe (MRS; HB0384-1; Qingdao Hope Bio-Technology, Co., Ltd.)
broth for 24 h in an incubator at 37°C under anaerobic or aerobic
conditions. All strains were twice activated in MRS broth, and the
bacterial density of each strain was approximately 109
CFU/ml via spectrophotometry.
Animal model and treatments
Forty-five male Sprague Dawley rats (8 weeks,
200–220 g) were provided by Hunan Si Lake King of Experimental
Animal Co., Ltd.. Rats were housed in specific pathogen-free cages
in a room with a controlled temperature (22±2°C) and humidity
(55–60%), under a 12 h light/dark cycle, and were allowed free
access to food and water. Rats were acclimated for 7 days before
the study.
The animals were randomised into four groups: The
control group (C, n=9), treated with gelatine physiological saline
(day 7 to end); the model group (M, n=12), treated with gelatine
physiological saline (day 7 to end) and surgery (day 10); the
treatment group (T, n=12), treated with gelatine physiological
saline containing probiotic combinations with L. plantarum
(109 CFU/ml), L. acidophilus (109
CFU/ml), L. rhamnosus (109 CFU/ml) and B.
animalis (109 CFU/ml) (day 17 to end) and surgery
(day 10) (the dose of probiotics were determined based on our
preliminary test) (28), with the
rest of the treatment time (day 7 to 16) maintained the same as
group M; and the prevention treatment group (PT, n=12), pretreated
with the probiotic combinations (day 7 to end) and surgery (day
10). The gelatine physiological saline contained 0.01% gelatine to
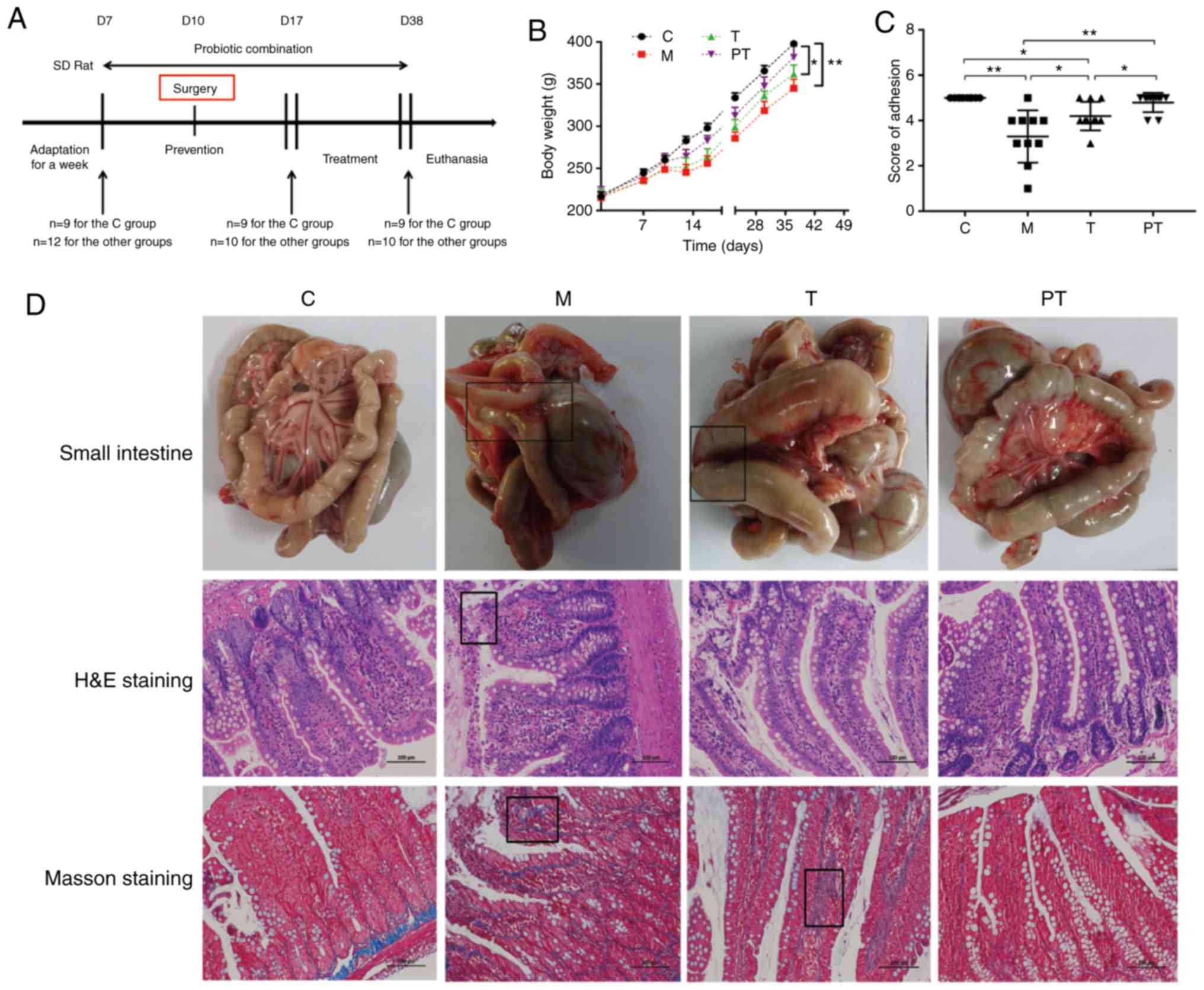
coat the probiotics and prevent digestion in the stomach (Fig. 1A). The weight of the rats was
measured every week before the surgery and once every 3 days during
the first week after the surgery. Detailed records regarding the
activities of the rats, the healing of the surgical incision and
their survival condition, among others, were performed at the same
intervals, and once per week thereafter.
Animal experiments were performed in a sterile
environment using the clamp trauma method. The rats were fasted for
12 h preoperatively. Animals were anaesthetised by intraperitoneal
injection with sodium pentobarbital (40 mg/kg; cat. no. B1202-005;
Fluka). Once anaesthesia was achieved, the abdomen was shaved and
disinfected with povidone iodine (cat. no. MDS093904; Medline
Industries) and a sterile surgical towel was laid. Abdominal
laparotomy was performed by an incision of ~2 cm in diameter into
the right side of the abdominal cavity. The ileocecal part was
gently removed from the incision, starting 5 cm away from the
ileocecal part. The intestinal tract on the opposite side of the
mesentery was clamped with toothed tweezers and toothless tweezers
at 1 cm intervals until subserosal haemorrhage and punctate
bleeding appeared, totalling 10 places. The abdominal incision was
sutured with a 3-0 nylon thread. After surgery, all rats were
transferred to separate cages according to groups for monitoring,
and their body weight, activity and survival condition were
observed (10).
The present study was approved by the Ethics
Committee of the Second Affiliated Hospital of Nanchang University,
and all experiments were conducted in accordance with the approved
guidelines.
Sample collection and evaluation of
adhesion
On day 28 after surgery, all rats were euthanised
with an overdose of sodium pentobarbital (200 mg/kg; cat. no.
B1202-005; Fluka), and a U-shaped incision was made to open the
abdominal cavity. The degree of abdominal adhesion was scored as
follows: Grade 0, no adhesions (5 points); grade 1, thin filmy
adhesion (4 points); grade 2, thick adhesions in a limited area (3
points); grade 3, widespread adhesions (2 points); and grade 4,
widespread adhesions plus adherence of visceral organs to the
abdominal wall (1 point). Tissue was collected from the typical
abdominal adhesion site or intestinal tissue from the modelling
site, and specimens were stored in 4% paraformaldehyde in
preparation for hematoxylin and eosin (H&E) staining and Masson
staining. Venous blood was obtained from the inferior vena cava of
rats, centrifuged at 1,000 x g for 20 min at 4°C, and then the
supernatant was carefully removed and stored at −80°C. Rat faeces
were stored in glycerol and maintained in a refrigerator at −80°C
for later use.
H&E and Masson staining
Adhered tissues or intestinal tissue were fixed in
4% paraformaldehyde at 4°C for 24 h and then embedded in paraffin.
The samples were cut into sections of 5–6 µm thickness and then
rehydrated with xylene and declining grades of ethanol for 5–6 min.
Specimens were washed three times with PBS for another 5 min, then
H&E and Masson staining were performed (29).
RNA preparation and quantitative
PCR
For the evaluation of cytokine mRNA expression
levels of IL-1β, IL-6 and TNF-α, total RNA from adhered tissues or
intestinal tissue were prepared by adding TRIzol reagent (Gibco
BRL; Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol (30). In
addition, the purity and integrity of RNA were evaluated using a
NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Inc.).
RNA (1 mg) was reversed-transcribed into cDNA using the PrimeScript
RT Master Mix reverse transcription kit (Takara Biotechnology Co.,
Ltd.). Quantitative real-time PCR was performed using a 7900HT fast
real-time PCR system (ABI; Thermo Fisher Scientific, Inc.) using 2X
SYBR-Green Master Mix (Bio-Rad Laboratories, Inc.). Forty cycles at
95°C for 30 sec and 60°C for 30 sec were conducted, preceded by 1
min at 95°C. The following primers were used: IL-1β sense,
5′-GTGTCTTTCCCGTGGACCTTC-3′ and antisense,
5′-TCATCTCGGAGCCTGTAGTGC-3′; TNF-α sense,
5′-GTGGAACTGGCAGAAGAGGCA-3′ and antisense,
5′-AGAGGGAGGCCATTTGGGAAC-3′; IL-6 sense, 5′-GAAATCGTGGAAATGAG-3′
and antisense, 5′-GCTTAGGCATAACGCACT-3′; and GAPDH sense,
5′-CTCGTGGAGTCTACTGGTGT-3′ and antisense
5′-GTCATCATACTTGGCAGGTT-3′.
Measurement of cytokines
Venous blood was obtained from the inferior vena
cava of rats, and then centrifuged at 1,000 x g for 20 min at 4°C.
The concentrations of cytokines IL-1β (cat. no. RK00009; rat;
ABclonal, Inc.; detection range, 62.5–4000 pg/ml; concentrations
used for generating calibration curves: 4000, 2000, 1,000, 500,
250, 125, 62.5, 31.2 and 0 pg/ml), IL-6 (cat. no. RK00020; rat;
ABclonal, Inc.; detection range, 125–8000 pg/ml; concentrations
used for generating calibration curves: 4000, 2000, 1,000, 500,
250, 125, 62.5 and 0 pg/ml) and TNF-α (cat. no. RK00029; rat;
ABclonal, Inc.; detection range, 62.5–4000 pg/ml; concentrations
used for generating calibration curves: 2000, 1,000, 500, 250, 125,
62.5, 31.2 and 0 pg/ml) in rat venous blood serum were measured
using enzyme-linked immunosorbent assay (ELISA) kits according to
the manufacturer's protocol.
Western blot analysis
Intestinal samples were extracted using Cell Lysis
buffer (cat. no. R0020; Solarbio, Inc.) supplemented with protease
inhibitor cocktail (cat. no. 78429) and 1 mM phenylmethanesulfonyl
fluoride (PMSF) (cat. no. 36978; both from Thermo Fisher
Scientific, Inc.). The protein concentration was determined using a
BCA protein assay kit. After degeneration, proteins were separated
using 10% polyacrylamide resolving gels and then transferred onto
polyvinylidene fluoride (PVDF) membranes. Protein concentrations
were mixed and then resolved by polyacrylamide gel electrophoresis.
Nonspecific binding sites were blocked using 5% skim milk with
Tris-buffered saline with Tween-20 (TBST) for 90 min at room
temperature (RT). Then, membranes were co-incubated overnight at
4°C with the following primary antibodies: Mouse anti-β-actin
(1:5,000; cat. no. 60008-1-Ig; ProteinTech Group, Inc.; RRID:
AB_2289225; having reactivity with rat), mouse anti-TLR4 (1:750;
cat. no. sc-293072; Santa Cruz Biotechnology, Inc.; RRID:
AB_10611320; having reactivity with rat), and rabbit anti-myeloid
differentiation primary response 88 (MyD88; 1:1,000; cat. no.
A0980; ABclonal, Inc.; RRID: AB_2722690; having reactivity with
rat), rabbit anti-NF-κB p65 (1:2,000; cat. no. 10745-1-AP;
ProteinTech Group, Inc.; RRID: AB_2178878; having reactivity with
rat), rabbit anti-phosphorylated (p)-NF-κB p65 (1:1,000; cat. no.
3033; Cell Signaling Technology, Inc.; RRID: AB_331284; having
reactivity with rat), rabbit anti-TGF-β1 (1:1,000; cat. no.
21898-1-AP; ProteinTech Group, Inc.; RRID: Not registered; having
reactivity with rat), rabbit anti-p-Smad2 (1:1,000; cat. no. 18338;
Cell Signaling Technology, Inc.; RRID: AB_2798798; having
reactivity with rat), rabbit anti-Smad2 (1:1,000; cat. no. A11498;
ABclonal, Inc.; RRID: AB_2758585; having reactivity with rat),
rabbit anti-p-Smad3 (1:1,000; cat. no. 9520; Cell Signaling
Technology, Inc.; RRID: AB_2193207; having reactivity with rat),
rabbit anti-Smad3 (1:1,000; cat. no. A7536; ABclonal, Inc.; RRID:
AB_2768063; having reactivity with rat), and rabbit anti-alpha
smooth muscle actin (α-SMA; 1:1,000; cat. no. A1011; ABclonal,
Inc.; RRID: AB_2757633; having reactivity with rat). After primary
incubation, the membranes were washed with TBST buffer three times
for 10 min each, then incubated with goat anti-rabbit secondary
antibody (1:5,000; cat. no. SA00001-2; ProteinTech Group, Inc.;
RRID: AB_2722564) or goat anti-mouse secondary antibody (1:5,000;
cat. no. SA00001-1; ProteinTech Group, Inc.; RRID: AB_2722565) to
bind with horseradish peroxidase (HRP) for 60 min at RT. Finally,
the proteins were visualised using enhanced chemiluminescence (ECL
Western blot kit; cat. no. CW0049S; CwbioTech, Inc.) (31,32).
Densitometry was performed using Image Lab Software (version 4.0;
Bio-Rad Laboratories, Inc.).
Total bacterial genomic DNA extraction
and high-throughput sequencing
For microbial DNA extraction, rat faecal samples for
the C (n=8), M (n=8), T (n=8) and PT (n=8) groups were collected.
The bead-beating method was combined with genomic DNA kits (Tiangen
Biotech Co., Ltd.), and a spectrophotometer (NanoDrop; Thermo
Fisher Scientific, Inc.) was used to determine the concentration
and quality of purified DNA. Then, 515F/806R primers (515F,
5′-GCACCTAAYTGGGYDTAAAGNG-3′; 806R, 5′-TACNVGGGTATCTAATCC-3′) were
used to amplify the V4 region of the 16S rDNA genes in each sample.
These PCR products were sequenced with an IlluminaHiSeq 2000
platform (GenBank accession no. PRJNA542549) (33).
Data analysis
To analyse the high-throughput sequencing data,
Cutadapt (version 1.9.1, http://cutadapt.readthedocs.io/en/stable/), UCHIME
algorithm http://www.drive5.com/usearch/manual/uchime_algo.html,
UPARSE software package (version 7.0.100), QIIME software (version
1.9.1), QIIME software package (version 1.8.0) and SIMCA-P software
(version 11.5; Umetrics; Sartorius Stedim) were used to determine
the α diversity (within a sample) and β diversity (among samples)
(34,35).
Statistical analyses were performed using Prism
software (version 7.0; GraphPad Software, Inc.). Data are presented
as the means ± standard deviation (SD). Statistical significance
was determined using one-way analysis of variance (ANOVA) followed
by Tukey's multiple comparison test. Error probabilities of
P<0.05 were considered to indicate a statistically significant
difference.
Results
Probiotic combinations prevent and
treat abdominal adhesions
During the model development process, two rats in
the M, T and PT groups respectively died of surgery-related causes
within 24 h, and no abdominal wall dehiscence or bowel perforation
were found in these animals (data not shown). As revealed in
Fig. 1B, there was a significant
reduction in the body weight of rats in the M group after surgery,
while rats treated with probiotics, especially those in the PT
group, had increased body weight compared with rats in the M group
(P<0.05). The degree of abdominal adhesion was compared using
the abdominal adhesion score, and it was revealed that surgery
significantly reduced the degree of abdominal adhesion in group M
compared to group C (3.24 vs. 5.0, P<0.01). The groups treated
with probiotics exhibited a significantly improved abdominal
adhesion score compared with the M group (T group, 4.2 vs. 3.24,
P<0.05; PT group, 4.8 vs. 3.24, P<0.05; Fig. 1C). H&E and Masson staining
further demonstrated that probiotics reduced inflammatory cell
infiltration, the production of collagenous fibres caused by
surgery and intestinal villus destruction compared with the M group
(Fig. 1D).
Probiotic combinations reduce
fibrosis
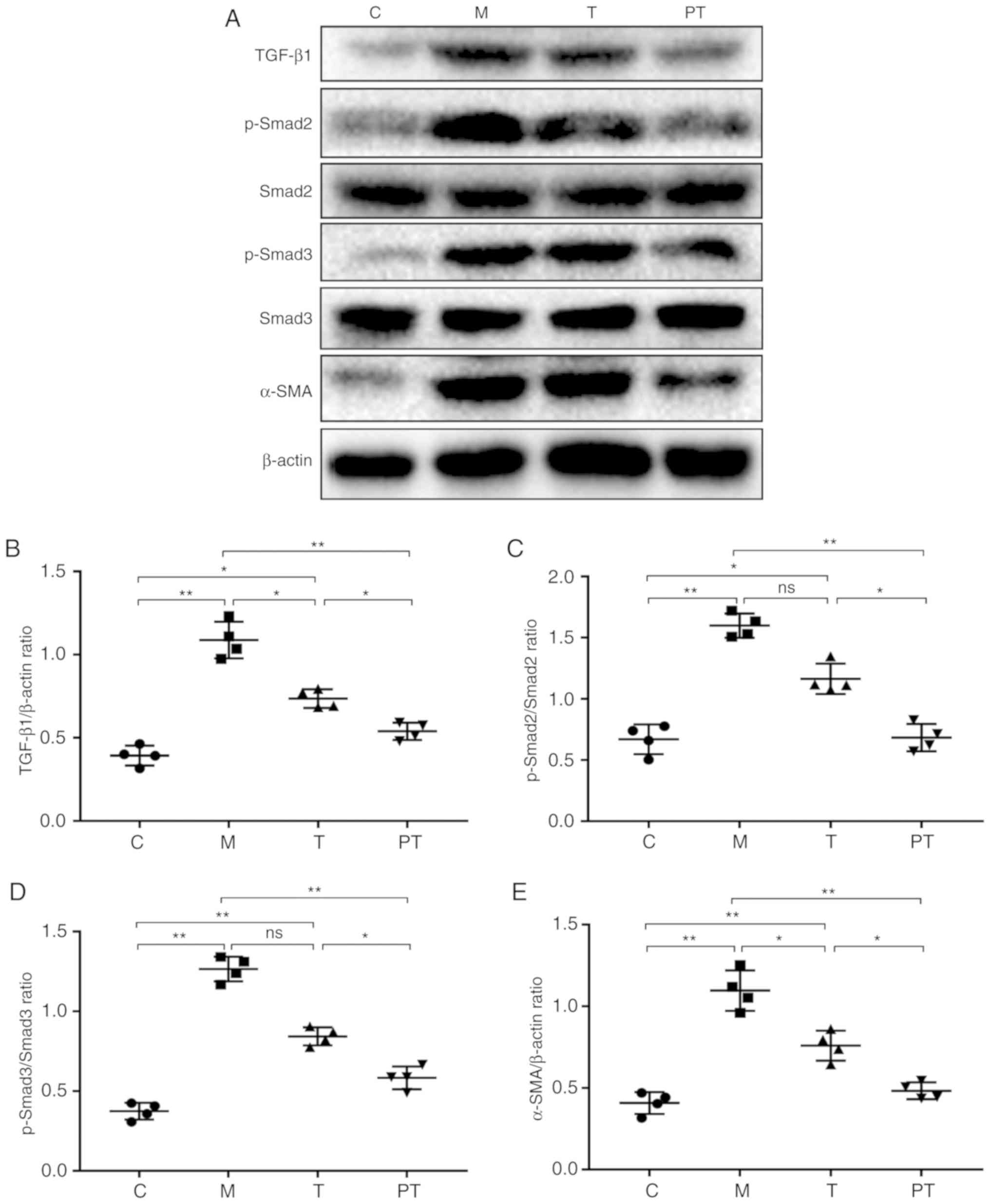
The formation of adhesions is closely related to the
TGF-β1/Smad signalling pathway; therefore, the level of their
expression at the adhesion site was evaluated. As revealed in
Fig. 2, surgery led to a
significant increase in the expression of TGF-β1 (0.39 vs. 1.09,
P<0.01), p-Smad2 (0.71 vs. 1.54, P<0.01) and p-Smad3 (0.37
vs. 1.27, P<0.01) compared with the control group, but treatment
with probiotics reversed this trend and recovered the TGF-β1,
p-Smad2 and p-Smad3 expression levels in the T group to 0.74, 1.22
and 0.84 and in the PT group to 0.54, 0.66 and 0.58, respectively.
Τhe expression of the myofibroblast marker α-SMA was then studied,
and it was revealed that probiotics inhibited the formation of
fibres caused by surgery. In the PT group, probiotic treatment
reduced the α-SMA expression compared with the M group (0.48 vs.
1.09; P<0.01).
Probiotic combinations decrease the
concentration of inflammatory mediators
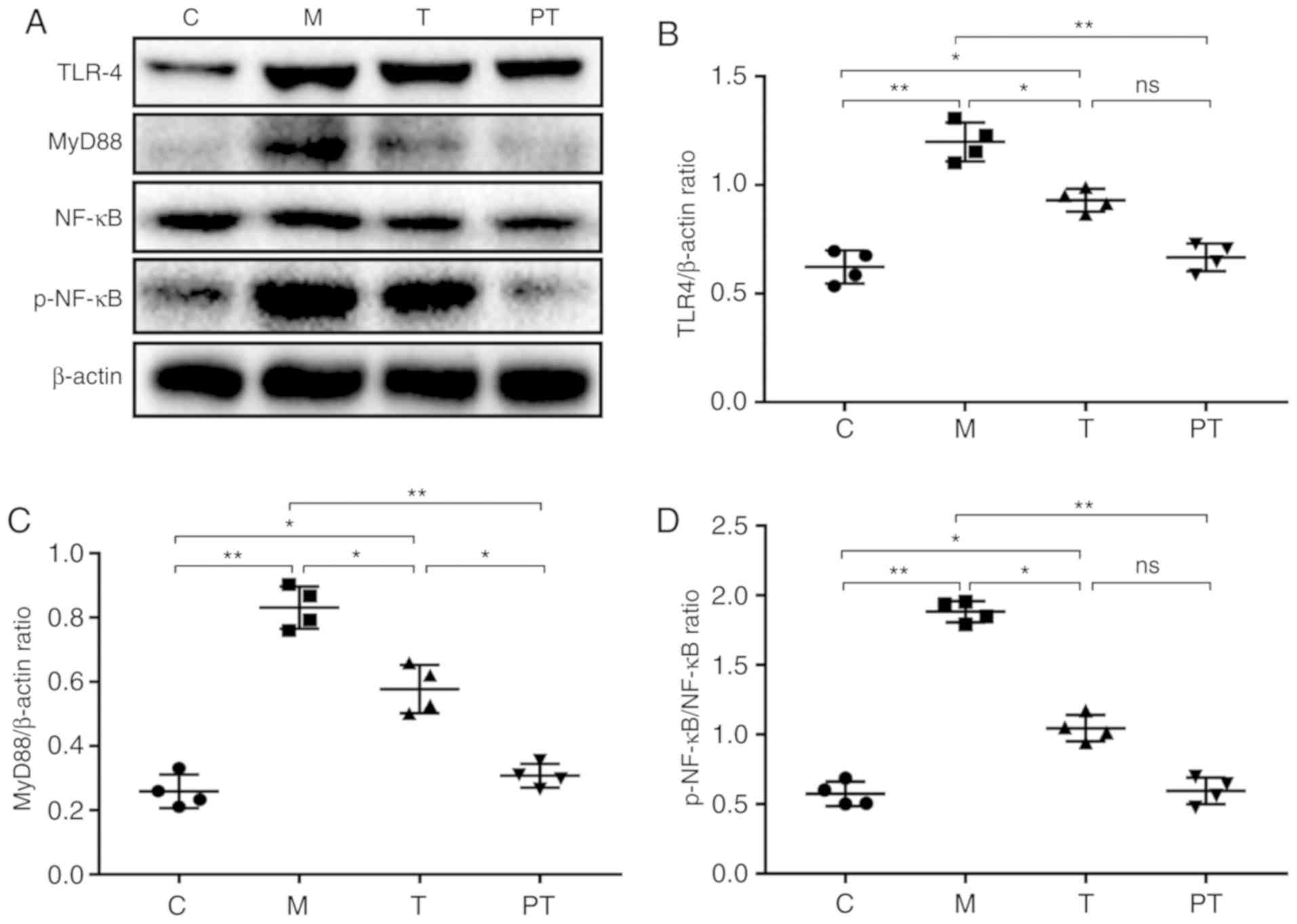
To verify whether the occurrence of abdominal
adhesion was related to inflammation, the classical inflammatory
TLR4/NF-κB signalling pathway was investigated using western
blotting, and it was revealed that surgery significantly
upregulated the expression of TLR4 (0.62 vs. 1.20, P<0.01),
MyD88 (0.26 vs. 0.83, P<0.01) and p-NF-κB/NF-κB (0.57 vs. 1.88,
P<0.01) compared with the C group, and the probiotic
combinations administered to the T and PT groups significantly
reduced the TLR4, MyD88 and p-NF-κB expression levels to 0.93, 0.58
and 1.05 for the T group and 0.67, 0.31 and 0.59 for the PT group,
respectively (Fig. 3).
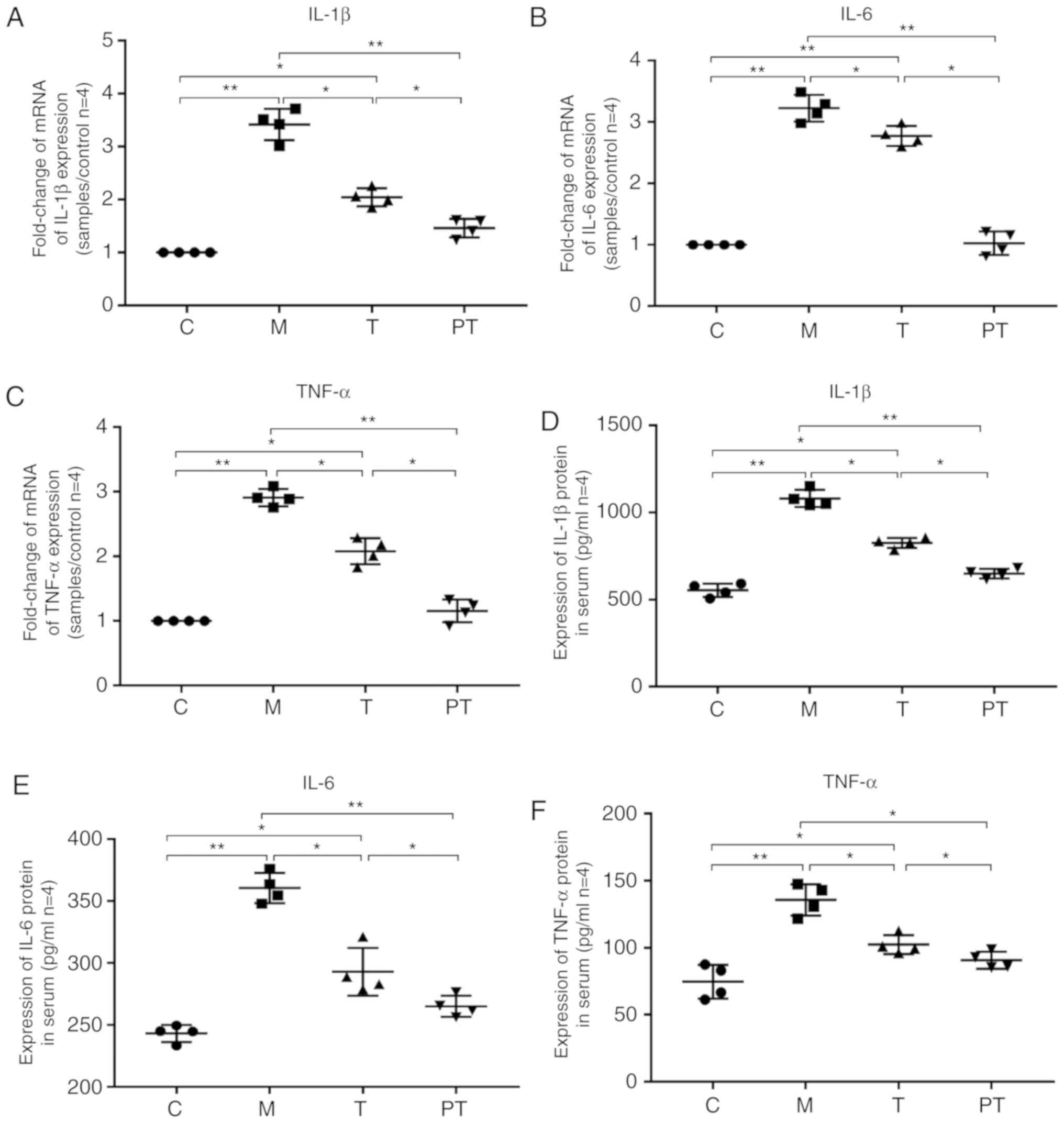
It is known that activation of the TLR4/NF-κB
signalling pathway causes the release of proinflammatory factors;
therefore, the effect of the probiotic combinations on the release
of inflammatory factors at the gene (q-PCR) and protein (ELISA)
levels, were investigated. Surgery significantly enhanced the
transcriptional levels of IL-1β (1.00 vs. 3.42, P<0.05), IL-6
(1.00 vs. 3.23, P<0.05) and TNF-α (1.00 vs. 2.91, P<0.05) in
the M group compared with the C group, and administration of
probiotics significantly reduced the expression of the
proinflammatory factors IL-1β, IL-6 and TNF-α to 2.04, 2.77 and
2.08 in the T group and 1.46, 1.03 and 1.16 in the PT group,
respectively. Likewise, the ELISA results supported the finding
that probiotics significantly inhibited the production of
proinflammatory factors in rat blood, with decreasing levels of
IL-1β, IL-6 and TNF-α from 1081, 360 and 136 in the M group,
respectively, to 826, 293 and 102 in the T group (P<0.05) and
649, 265 and 91 in the PT group (P<0.01), respectively (Fig. 4).
Probiotic combinations restore the
intestinal microbiota to a normal state
The intestinal microbiota is associated with many
intestinal diseases, including intestinal fibrosis. High-throughput
sequencing indicated that surgery significantly disturbed the
microbial balance, and reduced the Shannon diversity, the richness
of the microbiome (P<0.05) and the abundance-based coverage
estimators (ACE) index (P<0.01), although small statistical
differences were observed among these 3 indexes due to their
different statistical approaches (Fig.
5A-C). However, administration of probiotics in the T and PT
groups recovered the relative abundance of microbiota to normal
levels (P<0.05). In addition, the principal coordinate analysis
(PCoA) confirmed that the intestinal microbiota in the M group were
located far away from the C group, and the C, T and PT groups were
close to each other in distance (Fig.
5D). At the order level (Fig.
5E), communities of gut microbiota in the M group exhibited a
decrease in Bacteroidales (0.45 vs. 0.25) and
Lactobacillales (0.20 vs. 0.16), but exhibited an increase
in Clostridiales (0.27 vs. 0.37) and Coriobacteriales
(0.045 vs. 0.036) when compared with rats in the C group. However,
it was determined that supplementation with the probiotic
combinations altered the relative abundance of Bacteroidales
(0.29 vs. 0.27), Lactobacillales (0.27 vs. 0.35),
Clostridiales (0.30 vs. 0.28) and Coriobacteriales
(0.025 vs. 0.013) in the T group and PT group. At the genus level,
similar with the aforementioned results, supplementation of
probiotics significantly changed the composition of intestinal
microorganisms, greatly enhanced the richness of
Lactobacillus (levels in the C, M, T and PT groups were
0.19, 0.15, 0.25 and 0.34, respectively), Ruminococcus
(0.033, 0.012, 0.015 and 0.030, respectively), while the relative
levels of Oscillospira (0.041, 0.061, 0.035 and 0.045,
respectively) were markedly decreased (Fig. 5F).
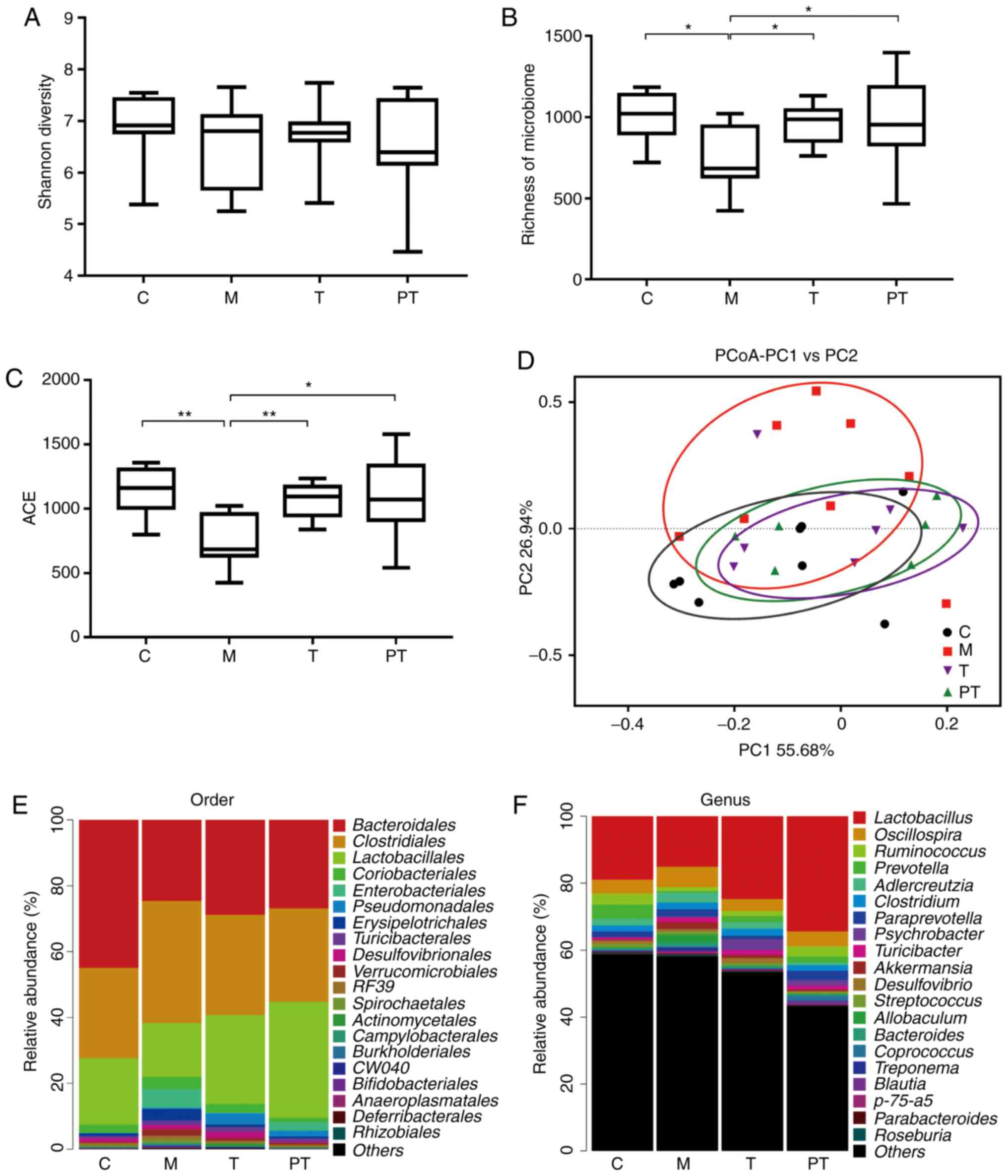 | Figure 5.Evaluation of the overall effect of
the probiotic combinations on microorganisms in rat faeces,
determined using high-throughput sequencing technology. Evaluation
of the effect of the probiotic combinations on intestinal
microbiota regarding (A) the Shannon Diversity, (B) the richness of
the microbiome, (C) the ACE index, (D) the PCoA of the β diversity
index. Effect of the probiotic combinations on intestinal
microorganisms regarding the relative abundance (E) at the order
level and (F) at the genus level. (n=8). C, control group; M, model
group; T, treatment group (fed 109 CFU probiotic
combinations 7 days after remodelling); PT, prevention and
treatment group (fed 109 CFU probiotic combinations 3
days before remodelling). Data are presented as the means ± SD.
*P<0.05; **P<0.01. ACE, abundance-based coverage estimators;
PCoA, principal coordinate analysis. |
In order to further analyse intestinal
microorganisms in rats, the relative abundance of some probiotic
bacteria as well as pathogens that were closely associated with
intestinal diseases were compared. The results revealed that
surgery tended to decrease the relative abundance of
Bacteroidetes, but increase the abundance of
Firmicutes and Proteobacteria (P<0.05) at the
phylum level (Fig. 6A-C). At the
order level, communities of gut microbiota in the M group exhibited
a decrease in Bacteroidales (0.45 vs. 0.25) and
Lactobacillales (0.20 vs. 0.16) but an increase in
Erysipelotrichales (0.0075 vs. 0.034) and
Verrucomicrobiales (0.0022 vs. 0.0198) levels compared with
C group (P<0.05; Fig. 6D-G),
while the probiotic combinations administered to the T and PT
groups significantly increased Bacteroidales (0.29 vs. 0.27)
and Lactobacillales (0.27 vs. 0.35) and decreased
Erysipelotrichales (0.0074 vs. 0.0095) and
Verrucomicrobiales (0.0095 vs. 0.0053). At the genus level
(Fig. 6H-K), it was observed that
surgery markedly reduced the richness of beneficial bacteria
Lactobacillus (0.19 vs. 0.15%) and Anaerostipes
(0.028 vs. 0.001%), but enhanced the abundance of pathogenic
bacteria Klebsiella (0 vs. 0.44%) and Serratia (0 vs.
0.075%) compared with the C group (P<0.05). Supplementation of
the probiotic combinations significantly reversed this trend, with
T and PT groups exhibiting a similar abundance of
Lactobacillus (0.25 vs. 0.34%), Anaerostipes (0.009
vs. 0.013%), Klebsiella (0.003 vs. 0.002%) and
Serratia (0.003 vs. 0%) to controls.
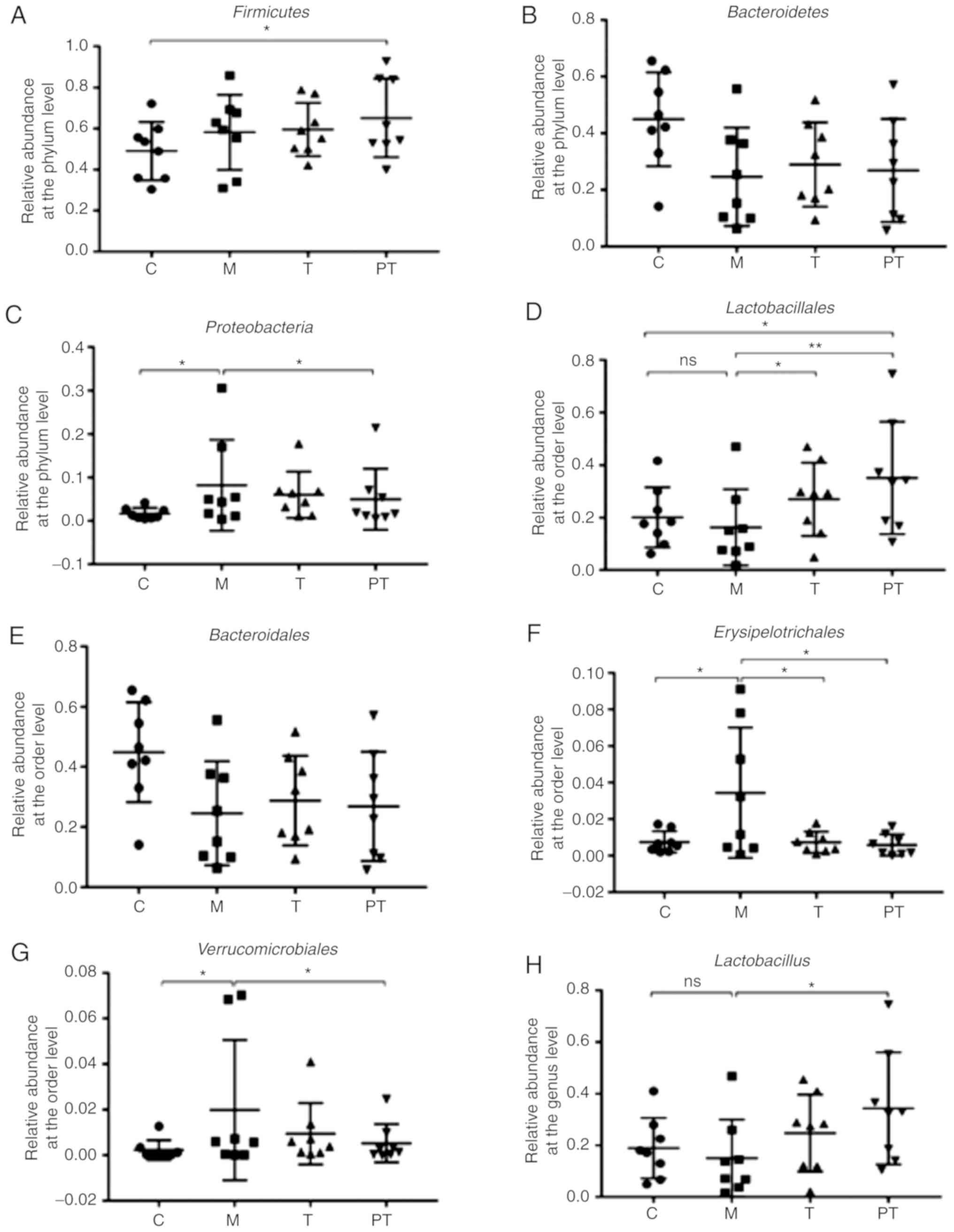 | Figure 6.Composition and relative abundance of
intestinal flora in rat faeces by phylum, order and genus,
determined using high-throughput sequencing technology. Evaluation
of the effect of the probiotic combinations on the relative
abundance of phyla (A) Firmicutes, (B) Bacteroidetes
(C) and Proteobacteria (n=8). Evaluation of the effect of
the probiotic combinations on the relative abundance of orders (D)
Lactobacillales, (E) Bacteroidales, (F)
Erysipelotrichales and (G) Verrucomicrobiales (n=8).
Evaluation of the effect of the probiotic combinations on the
relative abundance of genera (H) Lactobacillus, (I)
Anaerostipes, (J) Serratia and (K) Klebsiella
(n=8). C, control group; M, model group; T, treatment group (fed
109 CFU probiotic combinations 7 days after
remodelling); PT, prevention and treatment group (fed
109 CFU probiotic combinations 3 days before
remodelling). Data are presented as the means ± SD. ns, P>0.05;
*P<0.05; **P<0.01. |
Discussion
Although the mechanism underlying abdominal adhesion
formation has not yet been completely elucidated, the general
assumption is that they develop through processes related to
healing after injury (36).
Briefly, abdominal adhesions can be attributed to inflammation
caused by peritoneal stimulation, traction, drying and the
introduction of foreign bodies during abdominal surgery. With
inflammation which will initiate tissue repairing, during this
repairing process, fibrin formation and dissolution are out of
balance, collagen is deposited and, consequently, inappropriate
adhesion occurs (37). Although
researchers have been trying to solve this problem for a long time,
the current interventions still have little effect.
The human intestinal tract is occupied by a large
number of microorganisms which are commonly referred to as the
intestinal microflora. This complex and dynamic bacterial community
plays an important role in human health (38). Common probiotics include
Lactobacillus, Bifidobacterium and some Escherichia coli.
Lactobacillus and Bifidobacterium are vital members of
the normal microflora of the gastrointestinal tract, and will
accompany the host for a whole life. They contribute a great deal
to maintaining the microbial balance of the body. In addition, they
can stimulate the production of immunoglobulins, induce the
expression of interferons in macrophages, and enhance the
anti-inflammatory immunity of the host (39). A series of studies have reported
that intestinal tract stimulation, intestinal tract preparation,
use of preventive antibiotics and an imbalance in water and
electrolytes after abdominal surgery may lead to disruptions in the
intestinal microbial balance, resulting in endogenous infection and
intestinal inflammation (40,41).
Accounts of probiotics inhibiting inflammatory processes are no
longer unique, as there are numerous studies of their application
in the treatment of ulcerative colitis, Crohn's disease, diarrhoea,
constipation and even colorectal cancer (18,19,26).
However, the role of probiotics in the prevention of abdominal
adhesions has not yet been studied. The occurrence and development
of abdominal adhesions depend to a great extent on the inflammatory
reaction of the damaged tissues (3). In the present study, attempts to
prevent abdominal adhesions were made by administering a probiotic
combination of Lactobacillus and Bifidobacterium
(including L. plantarum, L. acidophilus, L. rhamnosus and
B. animalis), since probiotic mixtures appear to exhibit
greater efficacy than any single strain, although it is not clear
whether this is attributable to synergism or is the consequence of
the higher probiotic dose used in the studies investigating
them.
Firstly, the effect of probiotics on the body weight
and abdominal adhesion of rats following surgery were evaluated,
and it was revealed that probiotics significantly increased their
body weight when compared to the model group, and led to a
significant reduction in the inflammatory cellular infiltration and
fibrosis caused by surgery (Fig.
1). Notably, rats represented different scores in each groups
due to individual differences (some people will not form abdominal
adhesions after surgery in practice), and 1 rat in the M group had
an abdominal adhesion score of 5, 4 rats had 4 points for the
self-recovery capability of rats with not serious abdominal
adhesion after the treatment stage. Numerous studies have revealed
that TGF-β1/Smad signal transduction promotes fibroblast
proliferation, the occurrence and development of fibrosis and
adhesions, as well as the fibrosis in various tissues and organs,
including renal, myocardial, liver and pulmonary fibrosis (42,43).
Most researchers maintain that TGF-β is the initiating factor of
fibrosis, and that it has a significant effect on the fibrosis of
various tissues and organs. TGF-β can activate fibroblasts to
produce collagen, block plasminogen activator and disrupt the
balance between fibrinolysis and synthesis, leading to
extracellular matrix deposition and the formation of adhesions
(17). TGF-β1 has been revealed to
be the most active cytokine in the TGF-β family, and the level of
TGF-β1 increased gradually during the early postoperative period
(15). Furthermore, Bi et al
(44) revealed that TGF-β1 was
significantly upregulated in a mouse model of abdominal adhesions.
In addition, the Smad signalling pathway is one of the major
pathways by which members of the TGF-β superfamily transmit signals
through specific receptors on the cell membrane. Studies have
revealed that TGF-β1 could activate Smad3 to promote myocardial
interstitial fibrosis, causing myocardial fibrosis and other
irreversible changes, and eventually leading to chronic heart
failure (45). Furthermore,
research by Guo et al (46)
revealed that downregulating the expression of Smad2/Smad3 markedly
reduced the development of abdominal adhesions. The present study,
confirmed that surgery increased fibrosis (α-SMA is a marker
protein of myofibroblasts) in model rats, while the administration
of probiotics observably reduced the levels of key proteins
(TGF-β1, p-Smad2, p-Smad3 and α-SMA) associated with the
TGF-β1/Smad pathway.
Inflammation plays an important role in various
diseases, including abdominal adhesions (3,47).
Therefore, the effect of probiotics on the inflammatory state of
rats was evaluated, and it was revealed that administration of
probiotics significantly reduced the proinflammatory factors IL-1β,
IL-6 and TNF-α and downregulated the TLR4/NF-κB inflammatory
signalling pathway. The Toll-like receptor (TLR) family is one of
the most characteristic pattern recognition receptor families,
whose downstream molecules include NF-κB and MyD88. When a
molecular pattern is recognised by TLRs, downstream signals are
activated, leading to the release of a large amount of the
proinflammatory factors TNF-α and IL-1β (48). Therefore, downregulating the
expression of key proteins in the TLR4/NF-κB signalling pathway can
reduce the inflammatory response. Studies have revealed that the
expression levels of TLR4 and NF-κB are significantly upregulated
when an inflammatory reaction occurs in the intestinal tract of
mice, along with a marked increase in the release of related
proinflammatory cytokines TNF-α and IL-6 (49). Research by Wei et al
(47) demonstrated that
downregulation of the NF-κB signalling pathway could observably
reduce the inflammatory response and ultimately reduce abdominal
adhesions. Therefore, the decreased expression of key proteins in
the TLR4/NF-κB signalling pathway was observed in the groups
receiving probiotics, which indicated that probiotics could prevent
the occurrence of abdominal adhesions by suppressing
inflammation.
Finally, the V4 hypervariable region of microbiota
was sequenced using 16S rRNA amplicon sequencing analysis, and the
presence of intestinal microorganisms in rats was detected to
further explore the mechanism underlying the protective effect of
probiotics. The present results revealed that the combination of
probiotics greatly enhanced the abundance of Lactobacillus
and Anaerostipes, whereas it decreased the abundance of
pathogenic Klebsiella and Serratia bacteria. As is
known, Lactobacillus is often closely related to health.
Studies have revealed that the abundance of Lactobacillus is
significantly reduced in the intestinal tract of patients with
irritable bowel syndrome (26). In
addition, it has been reported that Anaerostipes can produce
butyric acid, which plays a positive role in maintaining
gastrointestinal health (50). A
recent study by Wopereis et al (51) revealed that the abundance of the
butyric acid-producing bacteria Anaerostipes was markedly
decreased in infantile eczema. Klebsiella exists in the
intestinal and respiratory tracts of humans, and can cause
bronchitis, pneumonia, diarrhoea, urinary system infections and
wound infection, and has even been linked to septicaemia,
meningitis and peritonitis. A previous study revealed that the
detection rate of Klebsiella pneumoniae was significantly
increased in children with acute intestinal diseases, while the
detection rate in healthy children without any clinical
manifestations was relatively low (52). This indicates that Klebsiella
has an aetiological relationship with acute bowel disease.
Serratia is an important genus of Enterobacteriaceae
that can cause hospital-related infections. It is invasive and
resistant to numerous commonly used antibacterial drugs. In
particular, Serratia marcescens has become an important
conditioned pathogen, and has been identified as the main pathogen
causing extraintestinal infection (53). In addition, a study revealed that
the abundance of Serratia marcesens in patients with Crohn's
disease was often higher than that in healthy family members, and
these bacteria could cooperate with other pathogenic bacteria to
aggravate intestinal inflammation (54).
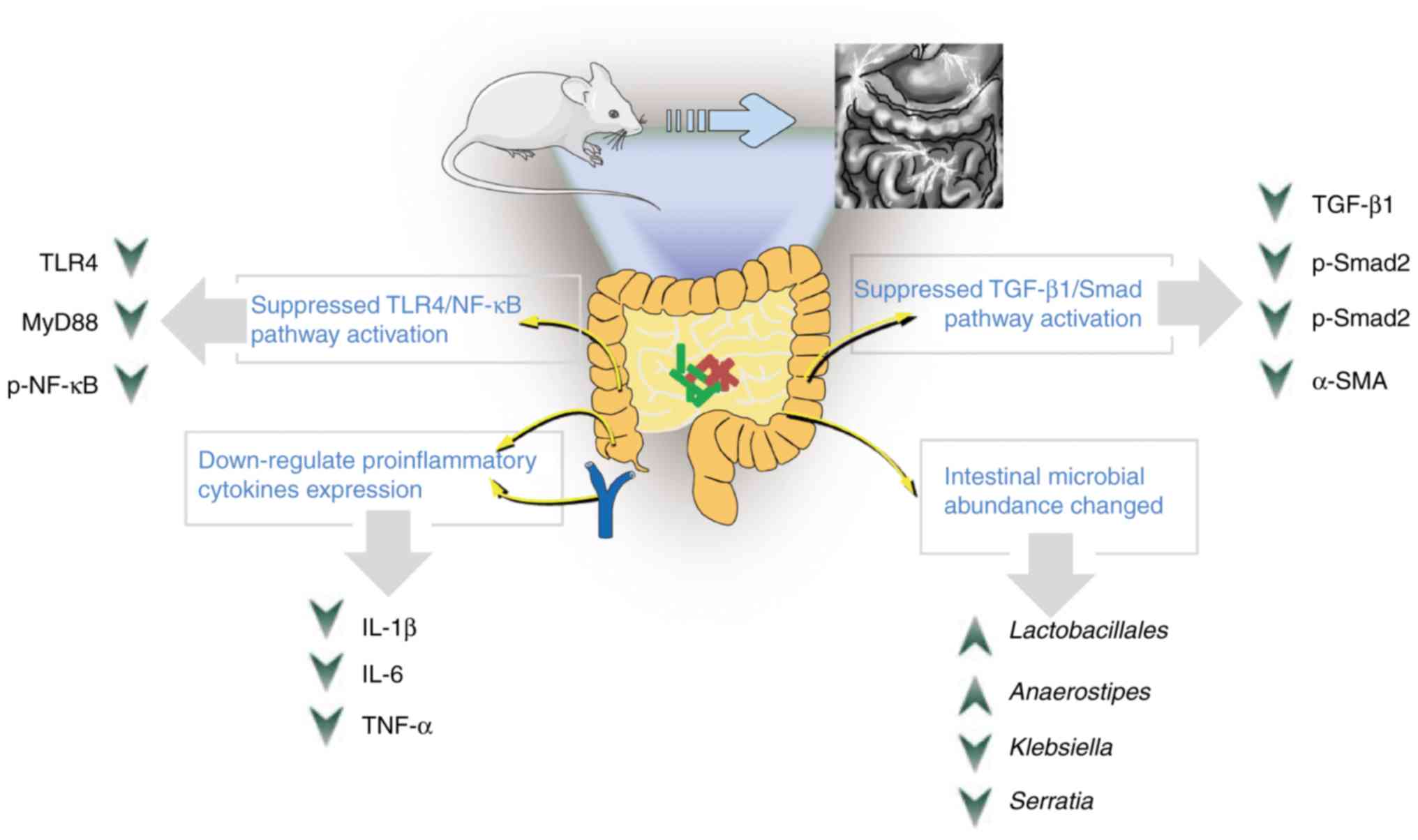
The present study revealed that the probiotic
combination administration was conducive to preventing abdominal
adhesion by restoring microbial diversity and reducing inflammation
and collagen deposition, and the effect was even more apparent when
administered early (Fig. 7).
Therefore, it is surmised, that in cases of extensive abdominal
surgery, the administration of oral probiotic combinations during
the perioperative period could have a positive effect on the
prevention of abdominal adhesions. However, the present study was
limited by the absence of an objective assessment tool for
abdominal adhesions at the anatomical level in this animal model,
and the discrepancies in the physiology and microbial composition
between humans and rats. A clinical trial to assess these
conclusions is required in the near future.
Acknowledgements
Not applicable.
Funding
This present study was supported by grants from the
National Natural Science Foundation of China (81960103 to XD,
31560264 to TC), the Natural Science Foundation of Jiangxi province
(20192ACBL20034 to ZL), the Excellent Youth Foundation of the
Jiangxi Scientific Committee (20171BCB23028 to TC), the Science and
Technology Plan of the Jiangxi Health Planning Committee (20175526
to TC), the Science and Technology Project of Jiangxi
(20181BBG70028 and 20181BCB24003 both to TC).
Availability of data and materials
All data generated or analysed during this study are
included in this published article.
Authors' contributions
TC, XD and ZL designed the experiments, analysed the
data and wrote the manuscript. CZ, SW and RY performed the
experiments. All authors discussed the results and commented on the
final manuscript. All authors read and approved the manuscript and
agree to be accountable for all aspects of the research in ensuring
that the accuracy or integrity of any part of the work are
appropriately investigated and resolved.
Ethics approval and consent to
participate
The protocol was approved by the Committee on the
Ethics of Animal Experiments of the Second Affiliated Hospital of
Nanchang University (Jiangxi, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that there are no competing
interests regarding the publication of this study and regarding the
funding that they have received.
References
|
1
|
Taylan E, Akdemir A, Ergenoglu AM, Yeniel
AO and Tekindal MA: Can we predict the presence and severity of
intra-abdominal adhesions before cesarean delivery. Gynecol Obstet
Invest. 82:521–526. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Vrijland WW, Tseng LN, Eijkman HJ, Hop WC,
Jakimowicz JJ, Leguit P, Stassen LP, Swank DJ, Haverlag R, Bonjer
HJ and Jeekel H: Fewer intraperitoneal adhesions with use of
hyaluronic acid-carboxymethylcellulose membrane: A randomized
clinical trial. Ann Surg. 235:193–199. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tsaousi G, Stavrou G, Fotiadis K,
Kotzampassi K and Kolios G: Implementation of phospholipids as
pharmacological modalities for postoperative adhesions prevention.
Eur J Pharmacol. 842:189–196. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tanaka K, Hashimoto H, Misawa T and Akiba
T: The prevention of carboxymethylcellulose on bowel adhesions
induced by talc peritonitis in mice. J Surg Res. 234:311–316. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bento SV, Nunes TA, Araujo ID, Silva RCOE,
Vidigal PVT and Carvalhais RM: Hyperbaric oxygenation on adhesions
prevention after laparotomy in rats. Acta Cir Bras. 33:824–833.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Robb WB and Mariette C: Strategies in the
prevention of the formation of postoperative adhesions in digestive
surgery: A systematic review of the literature. Dis Colon Rectum.
57:1228–1240. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Risberg B: Adhesions: Preventive
strategies. Eur J Surg Suppl. 32–39. 1997.PubMed/NCBI
|
|
8
|
Song Z, Zhang Y, Shao H, Ying Y, Chen X,
Mei L, Ma X, Chen L, Ling P and Liu F: Effect of xanthan gum on the
prevention of intra-abdominal adhesion in rats. Int J Biol
Macromol. 126:531–538. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lin LX, Luo JW, Yuan F, Zhang HH, Ye CQ
and Sun YL: In situ cross-linking carbodiimide-modified chitosan
hydrogel for postoperative adhesion prevention in a rat model. Mat
Sci Eng C Mater Biol Appl. 81:380–385. 2017. View Article : Google Scholar
|
|
10
|
Kuckelman J, Barron M, Kniery K, Kay J,
Kononchik J, Hoffer Z and Sohn V: Crystalloid fluid suspension
results in decreased adhesion burden when compared to bioresorbable
membranes in a rat model. Am J Surg. 217:954–958. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dinan TG and Cryan JF: The impact of gut
microbiota on brain and behaviour: Implications for psychiatry.
Curr Opin Clin Nutr Metab Care. 18:552–558. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Meng F, Chen T, Wang X, Wang X, Wei H,
Tian P, Wang H, Zhao X, Shen L and Xin H: Evaluation of the
accuracy and sensitivity of highthroughput sequencing technology
using known microbiota. Mol Med Rep. 17:408–413. 2018.PubMed/NCBI
|
|
13
|
Lee YK and Mazmanian SK: Has the
microbiota played a critical role in the evolution of the adaptive
immune system? Science. 330:1768–1773. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bing X, Xuelei L, Wanwei D, Linlang L and
Keyan C: EGCG maintains Th1/Th2 balance and mitigates ulcerative
colitis induced by dextran sulfate sodium through TLR4/MyD88/NF-κB
signaling pathway in rats. Can J Gastroenterol Hepatol.
2017:30572682017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Munireddy S, Kavalukas SL and Barbul A:
Intra-abdominal healing: Gastrointestinal tract and adhesions. Surg
Clin North Am. 90:1227–1236. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Duron JJ: Postoperative intraperitoneal
adhesion pathophysiology. Colorectal Dis. 9:14–24. 2010. View Article : Google Scholar
|
|
17
|
Yao QY, Xu BL, Wang JY, Liu HC, Zhang SC
and Tu CT: Inhibition by curcumin of multiple sites of the
transforming growth factor-beta1 signalling pathway ameliorates the
progression of liver fibrosis induced by carbon tetrachloride in
rats. BMC Complement Altern Med. 12:1562012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tojo R, Suarez A, Clemente MG, de los
Reyes-Gavilan CG, Margolles A, Gueimonde M and Ruas-Madiedo P:
Intestinal microbiota in health and disease: Role of bifidobacteria
in gut homeostasis. World J Gastroenterol. 20:15163–15176. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nie P, Li Z, Wang Y, Zhang Y, Zhao M, Luo
J, Du S, Deng Z, Chen J, Wang Y, et al: Gut microbiome
interventions in human health and diseases. Med Res Rev.
39:2286–2313. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fedorak RN, Feagan BG, Hotte N, Leddin D,
Dieleman LA, Petrunia DM, Enns R, Bitton A, Chiba N, Paré P, et al:
The probiotic VSL#3 has anti-inflammatory effects and could reduce
endoscopic recurrence after surgery for crohn's disease. Clin
Gastroenterol Hepatol. 13:928–935. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Liu M, Zhang X, Hao Y, Ding J, Shen J, Xue
Z, Qi W, Li Z, Song Y, Zhang T and Wang N: Protective effects of a
novel probiotic strain, Lactococcus lactis ML2018, in
colitis: In vivo and in vitro evidence. Food Funct. 10:1132–1145.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Munozprice LS, Lolans K and Quinn JP:
Emergence of resistance to daptomycin during treatment of
vancomycin-resistant enterococcus faecalis infection. Clin
Infect Dis. 41:565–566. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Abdelkareem MZ, Sayed M, Hassuna NA,
Mahmoud MS and Abdelwahab SF: Multi-Drug-Resistant enterococcus
faecalis among Egyptian patients with urinary tract infection.
J Chemother. 29:74–82. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tuladhar R, Patole SK, Kon TH, Norton R
and Whitehall JS: Refractory bacillus cereus infection in a
neonate. Int J Clin Pract. 54:345–347. 2000.PubMed/NCBI
|
|
25
|
Gröschel D, Burgress MA and Bodey GP Sr:
Gas gangrene-like infection with bacillus cereus in a
lymphoma patient. Cancer. 37:988–991. 2015. View Article : Google Scholar
|
|
26
|
Heeney DD, Gareau MG and Marco ML:
Intestinal Lactobacillus in health and disease, a driver or
just along for the ride? Curr Opin Biotechnol. 49:140–147. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hidalgo-Cantabrana C, Delgado S, Ruiz L,
Ruas-Madiedo P, Sanchez B and Margolles A: Bifidobacteria and their
health-promoting effects. Microbiol Spectr. 5:32017.
|
|
28
|
Chen T, Wu Q, Li S, Xiong S, Wei H, Jiang
S, Tan Q, Zhang Z and Zhu D: Microbiological quality and
characteristics of probiotic products in China. J Sci Food Agric.
94:131–138. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang L, Yuan D, Zheng J, Wu X, Wang J, Liu
X, He Y, Zhang C, Liu C, Wang T and Zhou Z: Chikusetsu saponin IVa
attenuates isoprenaline-induced myocardial fibrosis in mice through
activation autophagy mediated by AMPK/mTOR/ULK1 signaling.
Phytomedicine. 58:1527642018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chen T, Tian P, Huang Z, Zhao X, Wang H,
Xia C, Wang L and Wei H: Engineered commensal bacteria prevent
systemic inflammation-induced memory impairment and amyloidogenesis
via producing GLP-1. Appl Microbiol Biotechnol. 102:7565–7575.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tian P, Xu D, Huang Z, Meng F, Fu J, Wei H
and Chen T: Evaluation of truncated g protein delivered by live
attenuated salmonella as a vaccine against respiratory syncytial
virus. Microb Pathog. 115:299–303. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Fang X, Tian P, Zhao X, Jiang C and Chen
T: Neuroprotective effects of an engineered commensal bacterium in
the 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine Parkinson
disease mouse model via producing glucagon-like peptide-1. J
Neurochem. 150:441–452. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zheng C, Chen T, Wang Y, Gao Y, Kong Y,
Liu Z and Deng X: A randomised trial of probiotics to reduce
severity of physiological and microbial disorders induced by
partial gastrectomy for patients with gastric cancer. J Cancer.
10:568–576. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Bolger AM, Lohse M and Usadel B:
Trimmomatic: A flexible trimmer for Illumina sequence data.
Bioinformatics. 30:2114–2120. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Edgar RC: UPARSE: Highly accurate OTU
sequences from microbial amplicon reads. Nat Methods. 10:996–998.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Arung W, Meurisse M and Detry O:
Pathophysiology and prevention of postoperative peritoneal
adhesions. World J Gastroenterol. 17:4545–4553. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gemmati D, Occhionorelli S, Tisato V,
Vigliano M, Longo G, Gonelli A, Sibilla MG, Serino ML and Zamboni
P: Inherited genetic predispositions in F13A1 and F13B genes
predict abdominal adhesion formation: Identification of gender
prognostic indicators. Sci Rep. 8:169162018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li S, Chen T, Xu F, Dong S, Xu H, Xiong Y
and Wei H: The beneficial effect of exopolysaccharides from
bifidobacterium bifidum WBIN03 on microbial diversity in
mouse intestine. J Sci Food Agric. 94:256–264. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mary Ellen S, Francisco G, Richard G, Holt
PR, Quigley EM, Sartor RB, Sherman PM and Mayer EA: An update on
the use and investigation of probiotics in health and disease. Gut.
62:787–796. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Floch MH: The role of prebiotics and
probiotics in gastrointestinal disease. Gastroenterol Clin North
Am. 47:179–191. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhu D, Chen X, Wu J, Ju Y, Feng J, Lu G,
Ouyang M, Ren B and Li Y: Effect of perioperative intestinal
probiotics on intestinal flora and immune function in patients with
colorectal cancer. Nan Fang Yi Ke Da Xue Xue Bao. 32:1190–1193.
2012.(In Chinese). PubMed/NCBI
|
|
42
|
Li JH, Zhu HJ, Huang XR, Lai KN, Johnson
RJ and Lan HY: Smad7 inhibits fibrotic effect of TGF-Beta on renal
tubular epithelial cells by blocking Smad2 activation. J Am Soc
Nephrol. 13:1464–1472. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Pan R, Zhang Y, Zang B, Tan L and Jin M:
Hydroxysafflor yellow A inhibits TGF-β1-induced activation of human
fetal lung fibroblasts in vitro. J Pharm Pharmacol. 68:1320–1330.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bi J, Zhang S, Du Z and Zhang J, Deng Y,
Liu C and Zhang J: Peripheral serotonin regulates postoperative
intra-abdominal adhesion formation in mice. Sci Rep. 7:100012017.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Elmadhun NY, Sabe AA, Lassaletta AD, Dalal
RS and Sellke FW: Effects of alcohol on postoperative adhesion
formation in ischemic myocardium and pericardium. Ann Thorac Surg.
104:545–552. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Guo H, Leung JC, Cheung JS, Chan LY, Wu EX
and Lai KN: Non-Viral Smad7 gene delivery and attenuation of
postoperative peritoneal adhesion in an experimental model. Br J
Surg. 96:1323–1335. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wei G, Wu Y, Gao Q, Shen C, Chen Z, Wang
K, Yu J, Li X and Sun X: Gallic acid attenuates postoperative
intra-abdominal adhesion by inhibiting inflammatory reaction in a
rat model. Med Sci Monit. 24:827–838. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Xu J, Lu C, Liu Z, Zhang P, Guo H and Wang
T: Schizandrin B protects LPS-induced sepsis via TLR4/NF-κB/MyD88
signaling pathway. Am J Transl Res. 1155–1163. 2018.PubMed/NCBI
|
|
49
|
Wu W, Wang F, Gao X, Niu T, Zhu X, Yan X
and Chen H: Synergistic effect of kappa-carrageenan on
oxazolone-induced inflammation in BALB/c mice. BMC Gastroenterol.
16:412016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kant R, Rasinkangas P, Satokari R, Pietila
TE and Palva A: Genome sequence of the butyrate-producing anaerobic
bacterium anaerostipes hadrus PEL 85. Genome Announc.
3:e00224–e00215. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wopereis H, Sim K, Shaw A, Warner JO, Knol
J and Kroll JS: Intestinal microbiota in infants at high risk for
allergy: Effects of prebiotics and role in eczema development. J
Allergy Clin Immunol. 141:1334–1342. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Kiseleva MN, Nikulenkova TS and Kuznetsova
LS: Etiologic role of klebsiella in children with acute
intestinal diseases. Zh Mikrobiol Epidemiol Immunobiol. 50–52.
1976.(In Russian). PubMed/NCBI
|
|
53
|
Ochieng JB, Boisen N, Lindsay B, Santiago
A, Ouma C, Ombok M, Fields B, Stine OC and Nataro JP:
Serratia marcescens is injurious to intestinal epithelial
cells. Gut Microbes. 5:729–736. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Hager CL and Ghannoum MA: The mycobiome:
Role in health and disease, and as a potential probiotic target in
gastrointestinal disease. Dig Liver Dis. 49:1171–1176. 2017.
View Article : Google Scholar : PubMed/NCBI
|