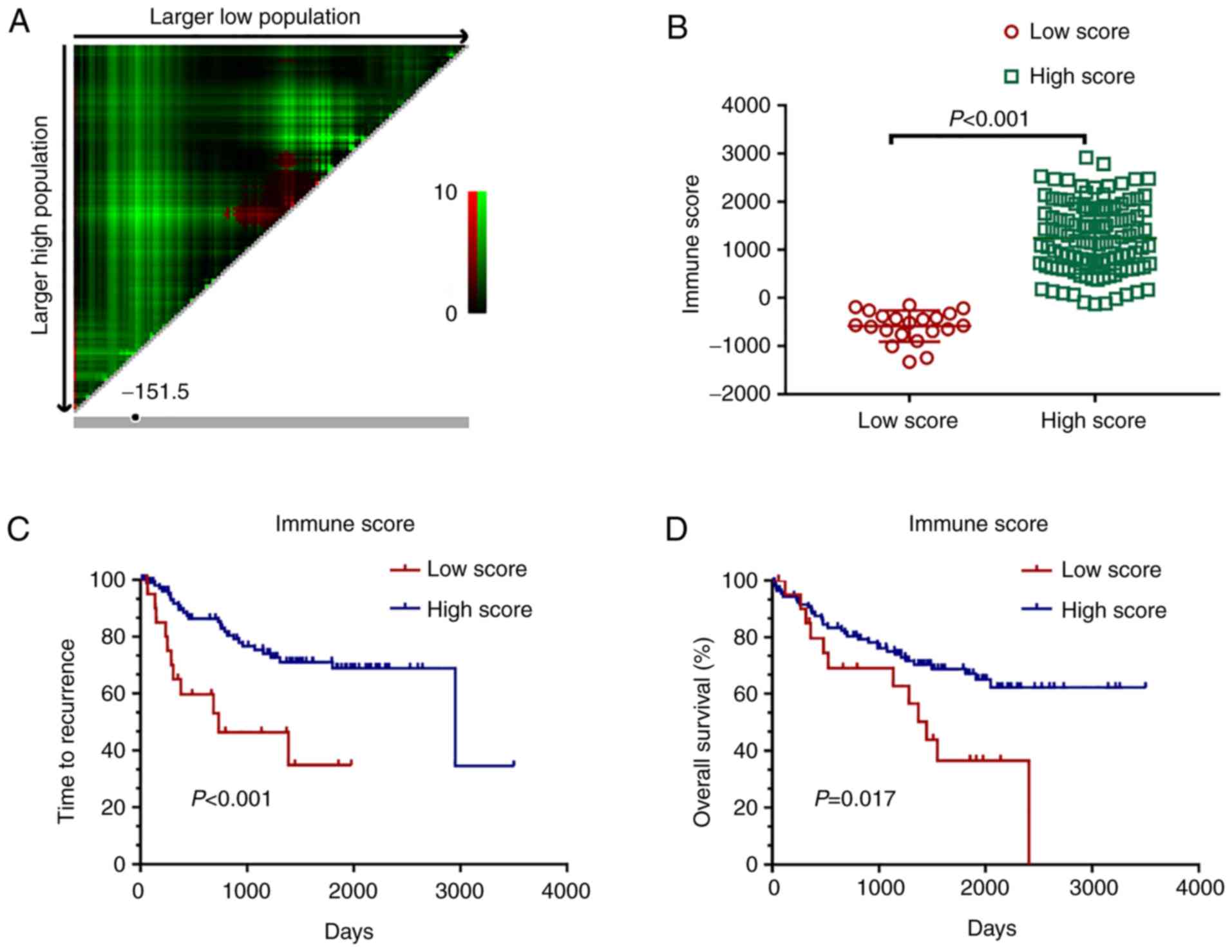
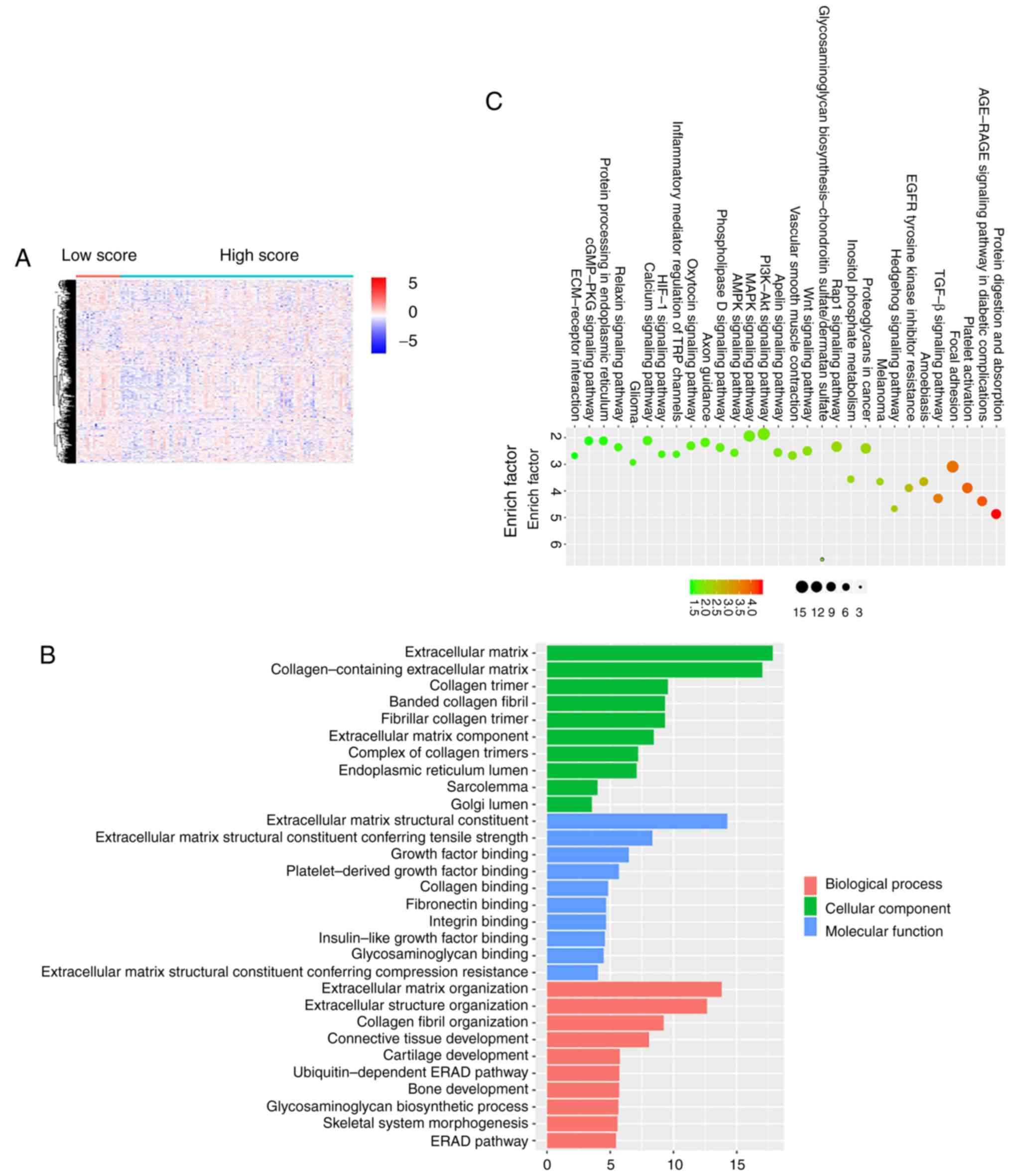
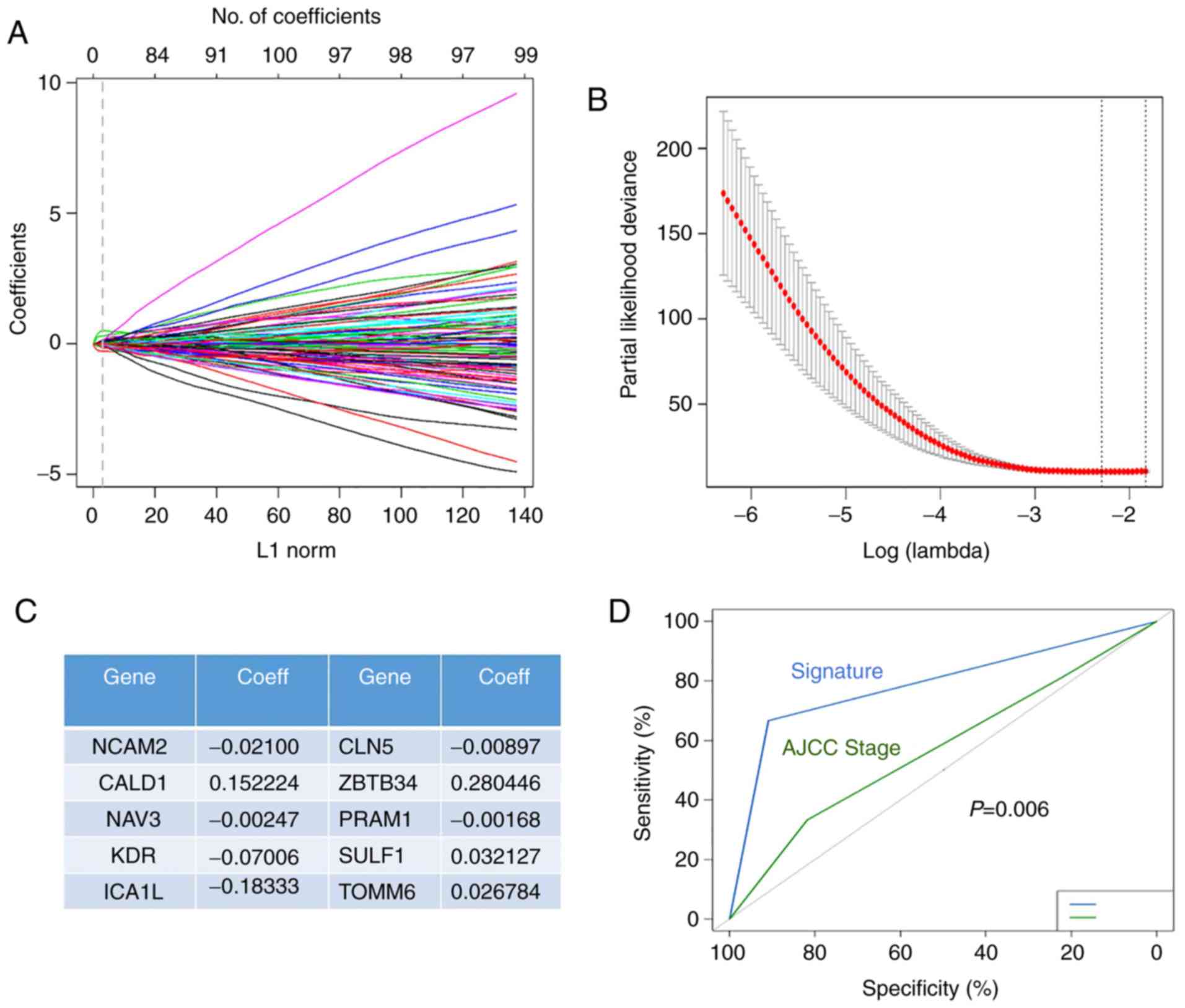
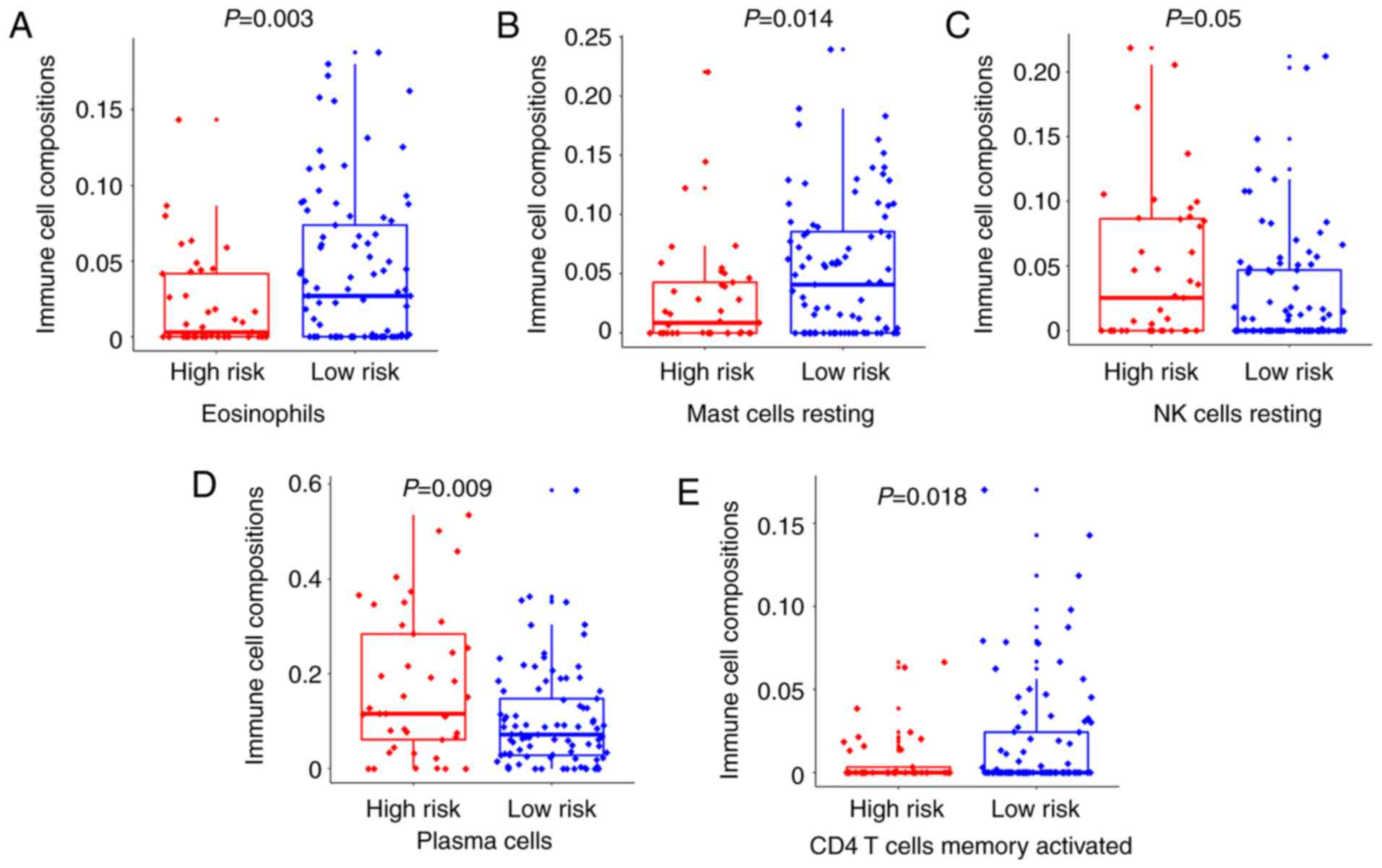
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Song P, Cui X, Bai L, Zhou X, Zhu X, Zhang
J, Jin F, Zhao J, Zhou C, Zhou Y, et al: Molecular characterization
of clinical responses to PD-1/PD-L1 inhibitors in non-small cell
lung cancer: Predictive value of multidimensional immunomarker
detection for the efficacy of PD-1 inhibitors in Chinese patients.
Thorac Cancer. 10:1303–1309. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang C, Leighl NB, Wu YL and Zhong WZ:
Emerging therapies for non-small cell lung cancer. J Hematol Oncol.
12:452019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chansky K, Sculier JP, Crowley JJ, Giroux
D, Van Meerbeeck J and Goldstraw P; International Staging Committee
and Participating Institutions, : The international association for
the study of lung cancer staging project: Prognostic factors and
pathologic TNM stage in surgically managed non-small cell lung
cancer. J Thorac Oncol. 4:792–801. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goldstraw P, Ball D, Jett JR, Le Chevalier
T, Lim E, Nicholson AG and Shepherd FA: Non-small-cell lung cancer.
Lancet. 378:1727–1740. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hanahan D and Weinberg RA: The hallmarks
of cancer. Cell. 100:57–70. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hanahan D and Coussens LM: Accessories to
the crime: Functions of cells recruited to the tumor
microenvironment. Cancer Cell. 21:309–322. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Stankovic B, Bjørhovde HA, Skarshaug R,
Aamodt H, Frafjord A, Müller E, Hammarström C, Beraki K, Bækkevold
ES, Woldbæk PR, et al: Immune cell composition in human non-small
cell lung cancer. Front Immunol. 9:31012019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Galon J, Pagès F, Marincola FM, Angell HK,
Thurin M, Lugli A, Zlobec I, Berger A, Bifulco C, Botti G, et al:
Cancer classification using the immunoscore: A worldwide task
force. J Transl Med. 10:2052012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xiong Y, Liu L, Xia Y, Qi Y, Chen Y, Chen
L, Zhang P, Kong Y, Qu Y, Wang Z, et al: Tumor infiltrating mast
cells determine oncogenic HIF-2α-conferred immune evasion in clear
cell renal cell carcinoma. Cancer Immunol Immunother. 68:731–741.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rohr-Udilova N, Klinglmüller F,
Schulte-Hermann R, Stift J, Herac M, Salzmann M, Finotello F,
Timelthaler G, Oberhuber G, Pinter M, et al: Deviations of the
immune cell landscape between healthy liver and hepatocellular
carcinoma. Sci Rep. 8:62202018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen B, Khodadoust MS, Liu CL, Newman AM
and Alizadeh AA: Profiling tumor infiltrating immune cells with
CIBERSORT. Methods Mol Biol. 1711:243–259. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yoshihara K, Shahmoradgoli M, Martínez E,
Vegesna R, Kim H, Torres-Garcia W, Treviño V, Shen H, Laird PW,
Levine DA, et al: Inferring tumour purity and stromal and immune
cell admixture from expression data. Nat Commun. 4:26122013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bakr S, Gevaert O, Echegaray S, Ayers K,
Zhou M, Shafiq M, Zheng H, Benson JA, Zhang W, Leung ANC, et al: A
radiogenomic dataset of non-small cell lung cancer. Sci Data.
5:1802022018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Okayama H, Kohno T, Ishii Y, Shimada Y,
Shiraishi K, Iwakawa R, Furuta K, Tsuta K, Shibata T, Yamamoto S,
et al: Identification of genes upregulated in ALK-positive and
EGFR/KRAS/ALK-negative lung adenocarcinomas. Cancer Res.
72:100–111. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Camp RL, Dolled-Filhart M and Rimm DL:
X-tile: A new bio-informatics tool for biomarker assessment and
outcome- based cut-point optimization. Clin Cancer Res.
10:7252–7259. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW,
Shi W and Smyth GK: Limma powers differential expression analyses
for RNA-sequencing and microarray studies. Nucleic Acids Res.
43:e472015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Huang da W, Sherman BT and Lempicki RA:
Systematic and integrative analysis of large gene lists using DAVID
bioinformatics resources. Nat Protoc. 4:44–57. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tibshirani R: The lasso method for
variable selection in the Cox model. Stat Med. 16:385–395. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Friedman J, Hastie T and Tibshirani R:
Regularization paths for generalized linear models via coordinate
descent. J Stat Softw. 33:1–22. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Newman AM, Liu CL, Green MR, Gentles AJ,
Feng W, Xu Y, Hoang CD, Diehn M and Alizadeh AA: Robust enumeration
of cell subsets from tissue expression profiles. Nat Methods.
12:453–457. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
O'Quigley J and Moreau T: Cox's regression
model: Computing a goodness of fit statistic. Comput Methods
Programs Biomed. 22:253–256. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Heagerty PJ, Lumley T and Pepe MS:
Time-dependent ROC curves for censored survival data and a
diagnostic marker. Biometrics. 56:337–344. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fridman WH, Pagès F, Sautès-Fridman C and
Galon J: The immune contexture in human tumours: Impact on clinical
outcome. Nat Rev Cancer. 12:298–306. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wojas-Krawczyk K, Kalinka E, Grenda A,
Krawczyk P and Milanowski J: Beyond PD-L1 markers for lung cancer
immunotherapy. Int J Mol Sci. 20:E19152019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Binnewies M, Roberts EW, Kersten K, Chan
V, Fearon DF, Merad M, Coussens LM, Gabrilovich DI,
Ostrand-Rosenberg S, Hedrick CC, et al: Understanding the tumor
immune microenvironment (TIME) for effective therapy. Nat Med.
24:541–550. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kilic A, Landreneau RJ, Luketich JD,
Pennathur A and Schuchert MJ: Density of tumor-infiltrating
lymphocytes correlates with disease recurrence and survival in
patients with large non-small-cell lung cancer tumors. J Surg Res.
167:207–210. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Horne ZD, Jack R, Gray ZT, Siegfried JM,
Wilson DO, Yousem SA, Nason KS, Landreneau RJ, Luketich JD and
Schuchert MJ: Increased levels of tumor-infiltrating lymphocytes
are associated with improved recurrence-free survival in stage 1A
non-small-cell lung cancer. J Surg Res. 171:1–5. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Domagala-Kulawik J: The role of the immune
system in non-small cell lung carcinoma and potential for
therapeutic intervention. Transl Lung Cancer Res. 4:177–190.
2015.PubMed/NCBI
|
|
30
|
Pabla S, Conroy JM, Nesline MK, Glenn ST,
Papanicolau-Sengos A, Burgher B, Hagen J, Giamo V, Andreas J, Lenzo
FL, et al: Proliferative potential and resistance to immune
checkpoint blockade in lung cancer patients. J Immunother Cancer.
7:272019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shin DS, Zaretsky JM, Escuin-Ordinas H,
Garcia-Diaz A, Hu-Lieskovan S, Kalbasi A, Grasso CS, Hugo W,
Sandoval S, Torrejon DY, et al: Primary resistance to PD-1 blockade
mediated by JAK1/2 mutations. Cancer Discov. 7:188–201. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kato S, Goodman A, Walavalkar V,
Barkauskas DA, Sharabi A and Kurzrock R: Hyperprogressors after
immunotherapy: Analysis of genomic alterations associated with
accelerated growth rate. Clin Cancer Res. 23:4242–4250. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Champiat S, Dercle L, Ammari S, Massard C,
Hollebecque A, Postel-Vinay S, Chaput N, Eggermont A, Marabelle A,
Soria JC and Ferté C: Hyperprogressive disease is a new pattern of
progression in cancer patients treated by anti-PD-1/PD-L1. Clin
Cancer Res. 23:1920–1928. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Paoloni-Giacobino A, Chen H and
Antonarakis SE: Cloning of a novel human neural cell adhesion
molecule gene (NCAM2) that maps to chromosome region 21q21 and is
potentially involved in Down syndrome. Genomics. 43:43–51. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Rasmussen KK, Falkesgaard MH, Winther M,
Roed NK, Quistgaard CL, Teisen MN, Edslev SM, Petersen DL,
Aljubouri A, Christensen C, et al: NCAM2 Fibronectin type-III
domains form a rigid structure that binds and activates the
Fibroblast Growth Factor Receptor. Sci Rep. 8:89572018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Edwards S, Campbell C, Flohr P, Shipley J,
Giddings I, Te-Poele R, Dodson A, Foster C, Clark J, Jhavar S, et
al: Expression analysis onto microarrays of randomly selected cDNA
clones highlights HOXB13 as a marker of human prostate cancer. Br J
Cancer. 92:376–381. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Romanuik TL, Ueda T, Le N, Haile S, Yong
TM, Thomson T, Vessella RL and Sadar MD: Novel biomarkers for
prostate cancer including noncoding transcripts. Am J Pathol.
175:2264–2276. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
von der Heyde S, Wagner S, Czerny A,
Nietert M, Ludewig F, Salinas-Riester G, Arlt D and Beißbarth T:
mRNA profiling reveals determinants of trastuzumab efficiency in
HER2-positive breast cancer. PLoS One. 10:e01178182015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lim B, Park JL, Kim HJ, Park YK, Kim JH,
Sohn HA, Noh SM, Song KS, Kim WH, Kim YS and Kim SY: Integrative
genomics analysis reveals the multilevel dysregulation and
oncogenic characteristics of TEAD4 in gastric cancer.
Carcinogenesis. 35:1020–1027. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Mayanagi T, Morita T, Hayashi K, Fukumoto
K and Sobue K: Glucocorticoid receptor-mediated expression of
caldesmon regulates cell migration via the reorganization of the
actin cytoskeleton. J Biol Chem. 283:31183–31196. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Thorsen K, Sørensen KD, Brems-Eskildsen
AS, Modin C, Gaustadnes M, Hein AM, Kruhøffer M, Laurberg S, Borre
M, Wang K, et al: Alternative splicing in colon, bladder, and
prostate cancer identified by exon array analysis. Mol Cell
Proteomics. 7:1214–1224. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Liu J, Li H, Shen S, Sun L, Yuan Y and
Xing C: Alternative splicing events implicated in carcinogenesis
and prognosis of colorectal cancer. J Cancer. 9:1754–1764. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Liu Y, Wu X, Wang G, Hu S, Zhang Y and
Zhao S: CALD1, CNN1, and TAGLN identified as potential prognostic
molecular markers of bladder cancer by bioinformatics analysis.
Medicine (Baltimore). 98:e138472019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Chauvin A, Wang CS, Geha S, Garde-Granger
P, Mathieu AA, Lacasse V and Boisvert FM: The response to
neoadjuvant chemoradiotherapy with 5-fluorouracil in locally
advanced rectal cancer patients: A predictive proteomic signature.
Clin Proteomics. 15:162018. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yokota M, Kojima M, Higuchi Y, Nishizawa
Y, Kobayashi A, Ito M, Saito N and Ochiai A: Gene expression
profile in the activation of subperitoneal fibroblasts reflects
prognosis of patients with colon cancer. Int J Cancer.
138:1422–1431. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wood LD, Parsons DW, Jones S, Lin J,
Sjöblom T, Leary RJ, Shen D, Boca SM, Barber T, Ptak J, et al: The
genomic landscapes of human breast and colorectal cancers. Science.
318:1108–1113. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Coy JF, Wiemann S, Bechmann I, Bächner D,
Nitsch R, Kretz O, Christiansen H and Poustka A: Pore membrane
and/or filament interacting like protein 1 (POMFIL1) is
predominantly expressed in the nervous system and encodes different
protein isoforms. Gene. 290:73–94. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Soon PS, Gill AJ, Benn DE, Clarkson A,
Robinson BG, McDonald KL and Sidhu SB: Microarray gene expression
and immunohistochemistry analyses of adrenocortical tumors identify
IGF2 and Ki-67 as useful in differentiating carcinomas from
adenomas. Endocr Relat Cancer. 16:573–583. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Bleeker FE, Lamba S, Rodolfo M, Scarpa A,
Leenstra S, Vandertop WP and Bardelli A: Mutational profiling of
cancer candidate genes in glioblastoma, melanoma and pancreatic
carcinoma reveals a snapshot of their genomic landscapes. Hum
Mutat. 30:E451–E459. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Hahtola S, Burghart E, Puputti M, Karenko
L, Abdel-Rahman WM, Väkevä L, Jeskanen L, Virolainen S, Karvonen J,
Salmenkivi K, et al: Cutaneous T-cell lymphoma-associated lung
cancers show chromosomal aberrations differing from primary lung
cancer. Genes Chromosomes Cancer. 47:107–117. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Karenko L, Hahtola S, Päivinen S, Karhu R,
Syrjä S, Kähkönen M, Nedoszytko B, Kytölä S, Zhou Y, Blazevic V, et
al: Primary cutaneous T-cell lymphomas show a deletion or
translocation affecting NAV3, the human UNC-53 homologue. Cancer
Res. 65:8101–8110. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hahtola S, Burghart E, Jeskanen L, Karenko
L, Abdel-Rahman WM, Polzer B, Kajanti M, Peltomäki P, Pettersson T,
Klein CA and Ranki A: Clinicopathological characterization and
genomic aberrations in subcutaneous panniculitis-like T-cell
lymphoma. J Invest Dermatol. 128:2304–2309. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Zhang SD, McCrudden CM, Meng C, Lin Y and
Kwok HF: The significance of combining VEGFA, FLT1, and KDR
expressions in colon cancer patient prognosis and predicting
response to bevacizumab. Onco Targets Ther. 8:835–843.
2015.PubMed/NCBI
|
|
54
|
Kershaw MH, Westwood JA, Zhu Z, Witte L,
Libutti SK and Hwu P: Generation of gene-modified T cells reactive
against the angiogenic kinase insert domain-containing receptor
(KDR) found on tumor vasculature. Hum Gene Ther. 11:2445–2452.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Edelbauer M, Datta D, Vos IH, Basu A,
Stack MP, Reinders ME, Sho M, Calzadilla K, Ganz P and Briscoe DM:
Effect of vascular endothelial growth factor and its receptor KDR
on the transendothelial migration and local trafficking of human T
cells in vitro and in vivo. Blood. 116:1980–1989. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
He J, Xia M, Tsang WH, Chow KL and Xia J:
ICA1L forms BAR-domain complexes with PICK1 and is crucial for
acrosome formation in spermiogenesis. J Cell Sci. 128:3822–3836.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Parvin S, Rezazadeh M, Hosseinzadeh H,
Moradi M, Shiva S and Gharesouran J: The neuronal ceroid
lipofuscinoses-linked loss of function CLN5 and CLN8 variants
disrupt normal lysosomal function. Neuromolecular Med. 21:160–169.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Qi J, Zhang X, Zhang HK, Yang HM, Zhou YB
and Han ZG: ZBTB34, a novel human BTB/POZ zinc finger protein, is a
potential transcriptional repressor. Mol Cell Biochem. 290:159–167.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Clemens RA, Newbrough SA, Chung EY, Gheith
S, Singer AL, Koretzky GA and Peterson EJ: PRAM-1 is required for
optimal integrin-dependent neutrophil function. Mol Cell Biol.
24:10923–10932. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Moog-Lutz C, Peterson EJ, Lutz PG, Eliason
S, Cavé-Riant F, Singer A, Di Gioia Y, Dmowski S, Kamens J, Cayre
YE and Koretzky G: PRAM-1 is a novel adaptor protein regulated by
retinoic acid (RA) and promyelocytic leukemia (PML)-RA receptor
alpha in acute promyelocytic leukemia cells. J Biol Chem.
276:22375–22381. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Gill RM, Michael A, Westley L, Kocher HM,
Murphy JI and Dhoot GK: SULF1/SULF2 splice variants differentially
regulate pancreatic tumour growth progression. Exp Cell Res.
324:157–171. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Morimoto-Tomita M, Uchimura K, Werb Z,
Hemmerich S and Rosen SD: Cloning and characterization of two
extracellular heparin-degrading endosulfatases in mice and humans.
J Biol Chem. 277:49175–49185. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Hur K, Han TS, Jung EJ, Yu J, Lee HJ, Kim
WH, Goel A and Yang HK: Up-regulated expression of sulfatases
(SULF1 and SULF2) as prognostic and metastasis predictive markers
in human gastric cancer. J Pathol. 228:88–98. 2012.PubMed/NCBI
|